1. Introduction
Craft beer has evolved from traditional small-scale brewing practices (often household- or microbrewery-based), in which greater emphasis is typically placed on ingredient quality and product attributes [
1]. With increasing consumer demand for novel olfactory and gustatory experiences, the diversity of beer styles has expanded substantially, and fruit beers have garnered increasing interest owing to their distinctive aroma, pleasant mouthfeel, and overall sensory attractiveness [
2]. Most fruit beers are crafted by directly integrating fruits or fruit-derived ingredients during the brewing process [
2,
3], with a wide variety of fruits applicable—including stone fruits (e.g., peach, plum, and cherry), pome fruits (e.g., pear and apple), tropical fruits (e.g., mango, pineapple, and banana), and berries (e.g., strawberry and blueberry). Prior studies have demonstrated that incorporating Cornelian cherry into beer can significantly improve its antioxidant capacity [
4]. Furthermore, the feasibility of mango as a brewing ingredient has been explored, with studies reporting that mango incorporation can increase the total polyphenol content by up to 44% relative to the fruit-free control and enhance antioxidant capacity, and sensory evaluation results from trained panelists have further demonstrated that mango beer exhibits more prominent aromatic characteristics and superior overall palatability compared with the control beer without fruit addition [
5].
Different brewing systems and beer styles, especially fruit-flavored craft beers like mango craft beer, require distinct yeast strains to tailor fermentation characteristics and flavor profiles [
6]. Fermentation serves as a pivotal stage in beer production, during which yeast metabolism not only dictates the conversion efficiency of fermentable sugars (e.g., maltose and maltotriose) into ethanol but also exerts a decisive influence on the formation, composition, and intensity of flavor- and aroma-active compounds—key factors that distinguish mango craft beer from conventional beers [
7]. Therefore, maintaining stable yeast growth, proliferation, and metabolic activity throughout the fermentation process is indispensable for ensuring consistent flavor quality, retaining fruit-specific nutrients (such as β-carotene in mango), and optimizing fermentation efficiency. ICT refers to the formation of a solid-phase biocatalytic system in which physiologically active cells are immobilized onto carrier materials via physical and/or chemical approaches.
Under continuous operation or repeated use, ICT enables improved cell retention and reusability, while sustaining relatively stable yeast growth and metabolic activity—critical merits for industrial-scale brewing that demands cost-effectiveness and process consistency [
8]. ICT is commonly categorized into immobilized microbial, animal, and plant cells based on the type of cells immobilized [
9]. In beer fermentation, relevant studies have further validated the feasibility of this technology; for instance, Ogawa et al. [
10] reported that yeast biocapsules enhanced cell reutilization during the fermentation, which supports the applicability of immobilized yeast for intensifying brewing process. Benucci et al. [
11] applied a commercial Saccharomyces cerevisiae Nottingham Ale yeast entrapped in chitosan–calcium alginate double-layer microcapsules for primary wort fermentation, and the study found that the kinetics of specific gravity decrease and oxygen consumption were slower for the encapsulated yeast than for free cells. Milinčić et al. [
12] further developed and evaluated an immobilized-yeast craft beer fermentation process supplemented with Prokupac grape pomace seed powder (2.5% and 5%), aiming to enrich phenolic compounds and improve sensory characteristics. In terms of carrier composition, immobilization carriers are generally classified as organic, inorganic, or natural materials; among these, natural materials are often preferred in food fermentations due to their structural broad availability, cost-effectiveness, porous architecture, and good biocompatibility [
13,
14]. Nevertheless, reports on the application of ICT to the production of fruit-based craft beers remain limited.
In this study, it was hypothesized that immobilizing yeast cells on plant-derived carrier materials could improve fermentation performance and the quality of products in fruit-based craft beer production. Immobilized yeast was first conducted using corncob, loofah sponge, and corncob-filled loofah sponge as carriers, after which the carrier system exhibiting the optimal immobilization efficiency was selected. Subsequently, mango craft beer was employed as a model to carry out batch fermentation experiments, and quality characteristics were assessed between beers inoculated with 1% immobilized yeast and those inoculated with 1% free yeast under identical inoculation conditions. The results were intended to provide a theoretical basis and experimental support for the application of immobilized-yeast technology in the fermentation and production of fruit-based craft beers.
2. Materials and Methods
2.1. Experimental Design
Two experiments were designed in this work: in Experiment 1, immobilized yeast was prepared using corncob, loofah sponge, and corncob-filled loofah sponge as carriers, and the fermentation performance and quality attributes of wort inoculated with 1% immobilized yeast were compared with those wort inoculated with 1% free yeast under identical inoculation conditions; in Experiment 2, batch fermentation was performed using mango craft beer as the model system, and quality differences between beers fermented with 1% immobilized yeast and 1% free yeast were evaluated.
2.2. Strain Source and Culture Conditions
American ale yeast (US-05) was purchased from Fermentis followed by activation and propagation. Briefly, the dry yeast was rehydrated in sterile water for 30 min, and 1 mL of the suspension was inoculated into yeast extract peptone dextrose medium (YPD) broth and incubated at 30 ℃ with shaking at 220 r/min for 48 h. Subsequently, a portion of the cultured broth was streaked onto YPD agar plates and incubated at 30 ℃ for 48 h to obtain isolated colonies. This streaking-incubation cycle was repeated three times to ensure culture purity. A single isolated colony was then picked and inoculated into fresh YPD broth, with incubation at 30 ℃ and 220 r/min for another 48 h. The resulting yeast culture served as the working inoculum.
2.3. Preparation of Natural Immobilization Carriers
A growth-associated adsorption method was employed to immobilize American ale yeast onto natural carriers. Specifically, loofah sponge was cut into small segments, and diced corncob was loaded into the center of the loofah matrix to prepare a corncob-filled loofah composite carrier. Into sterile culture vessels, the composite carrier and YPD broth were added at a volume ratio of 1:60 (carrier: medium), followed by inoculation with the yeast seed culture at a 1% (v/v) ratio. The mixture was incubated at 30 ℃ with shaking at 220 r/min for 48 h, allowing yeast cells to be adsorbed and immobilized on the carrier surface and within its porous structure during cultivation.
2.4. Immobilized Yeast Fermentation
Fermentation was carried out with wort adjusted to a concentration of 12 °P. Briefly, 1000 g of concentrated wort (75 °P) was reconstituted in a suitable volume of warm purified water, followed by boiling for 60 min at a boil-off rate of 10%. Horehound was added at a dosage of 0.4 g/L at the onset of boiling, with a subsequent supplementation of 0.6 g/L at the 55th minute of the boiling process. Upon completion of boiling, the wort was sampled for extract determination, adjusted to 12 °P as needed, and cooled in an ice-water bath prior to fermentation. The cooled wort was separately inoculated with immobilized yeast or free yeast, with the initial fermentation temperature set at 19 ℃. During the first 12 h post-inoculation, the cultures were incubated at 19 ℃ with agitation at 220 r/min in a temperature-controlled shaker, after which fermentation was continued under static conditions. Samples were collected at 8-h intervals for extract measurement and 12-h intervals for ethanol determination throughout the entire 96-h fermentation period.
2.5. Fermentation Protocol for Mango Craft Beer
To investigate the effects of key process variables on mango craft beer, this study was conducted by varying the original wort extract (8, 10, 12, 14, and 16 °P) and incorporating mango purée (supplied by Guangxi Guotianxia Food Co., Ltd.) at different fermentation stages (days 0, 4, 12, and 20) with varying dosages of 2.5%, 5%, 10%, 15%, and 20% (v/v) relative to the fermentation volume. Single-factor experiments were first carried out to delineate the effective range of original wort extract affecting sensory scores and β-carotene retention, based on which response surface methodology was applied to determine the optimal fermentation conditions, followed by the implementation of scale-up fermentation under these optimized parameters. For the sensory evaluation of the mango craft beer produced via scale-up fermentation, the assessment was performed in strict accordance with the relevant requirements specified in GB/T 4928-2008, GB/T 13868-2009 and ISO 4120:2021. This study was approved by the Medical Ethics Committee of Guangxi University (Approval No.: GXU-2024-085), and written informed consent was obtained from all participants. The sensory evaluation panel consisted of 10 panelists aged 18–55 years, all selected from teachers and students majoring in food science and engineering. Candidates received unified training provided by professional beer tasting experts with more than ten years of working experience in the beer industry; after training, the triangle test was adopted for screening, and only those who could correctly identify the distinct sample in three consecutive repeated tests were selected as formal panelists. The sensory evaluation employed four core indicators, namely appearance, aroma, taste, and coordination, along with a 100-point scoring system, with the detailed scoring criteria presented in
Table 1.
2.6. Determination of Alcohol Content and Extract
The alcohol content of the beer was determined by gas chromatography (GC, GC-2030)under the following conditions: the separation was performed on an Rtx-Wax capillary column (30 m × 0.25 mm × 0.25 μm) maintained at 200 ℃; the temperatures of both the injector and detector were set at 240 ℃; high-purity nitrogen was used as the carrier gas at 40 mL/min, with hydrogen and air flow rates of 40 mL/min and 500 mL/min, respectively; and the injection volume was fixed at 1.0 μL. The extract (°P) was measured using an Anton Paar EasyDens smart density meter.
2.7. Electronic Nose–Based Volatile Profile Analysis
The volatile aroma profiles of the beer samples were analyzed using a PEN3 electronic nose (E-nose), followed by principal component analysis (PCA). At room temperature, A 20-mL aliquot of each sample was added to a 50-mL beaker., which was immediately sealed with plastic film and equilibrated for 30 min to achieve headspace saturation. The E-nose probe was then inserted into the headspace for volatile compound detection. All measurements were conducted in triplicate. Data collected during the stable sensor-response phase were selected for analysis, with five data points extracted per sample, and data processing and PCA were conducted using the Winmuster software provided with the PEN3 system. Instrumental parameters were set according to the manufacturer’s protocol: pre-sampling time, 5 s; signal acquisition time, 120 s; purge time, 100 s; zero-point adjustment time, 5 s; measurement time, 60 s; and carrier-gas flow rate, 400 mL/min, allowing the carrier gas to deliver headspace volatiles across the sensor array to generate response signals.
2.8. Electronic Tongue–Based Taste Profile Analysis
The taste attributes of mango craft beer were evaluated using an electronic tongue (SA402B). Sensor activation (24 h) and calibration were performed according to the manufacturer’s instructions to ensure measurement stability and reliability. All measurements were conducted at 25 ℃ using 100 mL of filtered sample placed in the instrument’s dedicated sample cup. The “Sample Measurement (2 steps washing)” program was applied to quantify five basic taste modalities (sourness, bitterness, astringency, umami, and saltiness), with sample acquisition alternating with rinsing steps. Each sample was measured four times; the first measurement was discarded to minimize systematic error, and the mean of the subsequent three measurements was used for data processing and statistical analysis.
The measurement sequence was programmed as follows: the sensors were initially rinsed in an anion or cation cleaning solution for 90 s, followed by washing in Reference solutions 1 and 2 for 120 s each. Zero-point calibration was performed twice in Reference solution 3 (30 s per cycle) to reach equilibrium, after which the measurement of basic taste was conducted in the sample for 3 s. The sensors were then briefly washed in Reference solutions 4 and 5 (3 s each), and the evaluation of aftertaste was performed in Reference solution 6 for 30 s.
2.9. Quantification of β-Carotene, Total Phenolics, and Alcohol Compounds
The determination of β-carotene was performed with reference to the method described by Guadalupe Ortega et al. [
15], with appropriate modifications. Briefly, 10.0 g of thoroughly mango purée craft beer was mixed with 30 mL of an acetone/petroleum ether solvent (v/v = 4:1), followed by the addition of 1.0 g MgCO₃. The mixture was shaken for 1 min, ultrasonically extracted for 30 min, and centrifuged at 4 ℃ and 9000 r/min for 20 min. The supernatant was transferred to a separatory funnel; and the extraction was repeated once, and the two supernatants were combined. Subsequently, 75 mL of 20% (w/v) NaCl solution was added for liquid–liquid extraction; after standing for 2 h, the aqueous phase was discarded and the organic phase was diluted to 50 mL with petroleum ether. Finally, the absorbance of the organic phase was measured at a wavelength of 450 nm using a UV-visible spectrophotometer.
Total phenolic content was determined using the Folin–Ciocalteu method with gallic acid (GAE) as the standard. For both GAE standards and sample solutions, 2.5 mL of 10% Folin–Ciocalteu reagent was added, and the mixture was kept in the dark for 3–8 min; 2.0 mL of 7.5% Na₂CO₃ solution was then added, and the volume was adjusted to 10 mL with water. After incubation in the dark for 2 h, absorbance was recorded at 760 nm.
Higher alcohols, including n-propanol, n-butanol, isobutanol, n-pentanol, isoamyl alcohol, and β-phenylethanol, were quantified by gas chromatography. Separation was achieved on a DB-FFAP capillary column (60 m × 0.25 mm × 0.25 μm), with high-purity nitrogen used as the carrier gas at a flow rate of 0.5–1.0 mL/min. The split ratio was configured at 37:1, and the make-up (tail) gas flow rate was set at 25.0 mL/min; the flow rates of hydrogen and air were maintained at 30 mL/min and 400 mL/min, respectively. Both the injector and detector temperatures were held constant at 250 ℃. The column oven temperature program was optimized as follows: the initial temperature was set at 40 ℃ and held for 3 min, then ramped up to 70 ℃ at a rate of 1 ℃/min, further elevated to 180 ℃ at 5 ℃/min, and finally increased to 220 ℃ at 3 °C/min with a final 5-min isothermal hold.
2.10. Analysis of Volatile Flavor Compounds
A 10 mL aliquot of degassed beer sample was transferred into a 50 mL headspace vial designed for solid-phase microextraction (SPME). Subsequently, 10 μL of 3-decanone (1‰, v/v) was added as the internal standard, and 1.5 g NaCl was introduced to increase ionic strength and promote the partitioning of volatile compounds into the headspace. After sealing, the vial was equilibrated at 50 ℃ for 5 min, followed by extraction with an SPME fiber for 50 min. Upon completion of extraction, thermal desorption was performed at the GC injector for 5 min. Volatile flavor compounds were determined by gas Chromatography-Mass Spectrometry (GC-MS), with 3-decanone employed as the internal standard for quantitative correction.
GC separation was carried out on a DB-5ms capillary column (30 m × 250 μm × 0.25 μm) using high-purity helium as the carrier gas at a constant flow rate of 1.0 mL/min. The oven temperature program was set as follows: 40 ℃ (held for 5 min), increased to 120 ℃ at 4 ℃/min (held for 3 min), increased to 220 ℃ at 10 ℃/min (held for 10 min), and finally increased to 280 ℃ at 30 ℃/min (held for 10 min). Samples were injected in splitless mode. Mass spectrometric (MS) detection was performed using electron ionization (EI) at 70 eV with an emission current of 200 μA. The ion source temperature, quadrupole temperature, and transfer line temperature were held constant at 250 ℃, 150 ℃, and 250 ℃, respectively. Data were acquired in full-scan mode over an m/z range of 35–450, with no solvent delay applied.
3. Results
3.1. Naturally Immobilized Yeast Improves Fermentation Extract, Alcohol Content, and Fermentation Aroma
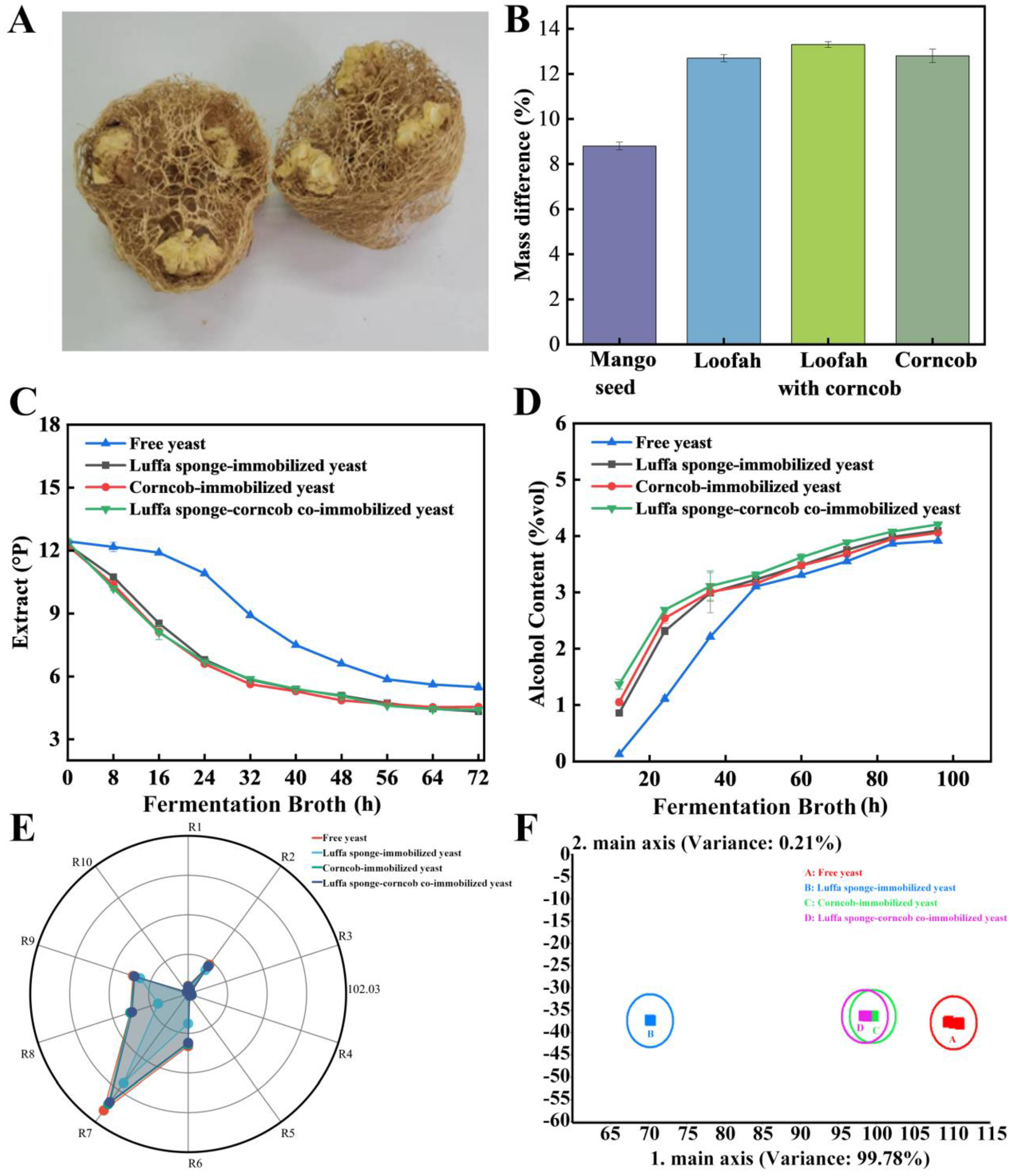
To enhance the effective biocatalyst concentration, shorten fermentation time, and improve overall flavor attributes, an immobilized yeast system was established using a corncob-filled loofah sponge as a composite natural carrier (
Figure 1A). When the adsorption performance of several natural materials (including mango seed, loofah sponge, and corncob) was compared, the corncob-filled loofah carrier exhibited the highest immobilization efficiency, with a unit-mass difference of 13.3% before versus after immobilization (
Figure 1B). Under an identical inoculum level (1%, v/v), pronounced differences in primary-fermentation kinetics were observed between immobilized and free yeast. At 48 h, wort fermented with immobilized yeast reached an extract of 5.0 °P and subsequently approached a plateau, whereas the free-yeast fermentation required 72 h to decrease to approximately 5.0 °P, indicating that the primary fermentation period was reduced from ~72 h to ~48 h (33.3% shorter) by immobilization (
Figure 1C). During primary fermentation, the mean extract depletion rate of the immobilized-yeast treatments was 0.15 °P/h, which was 1.58-fold higher than that of free yeast (0.096 °P/h), and the mean alcohol production rate (
Figure 1D) was 0.068 %vol/h, exceeding that of free yeast (0.049 %vol/h; 1.39-fold). The corncob-filled loofah immobilized-yeast system achieved the highest alcohol production rate and final alcohol content among the immobilized carriers evaluated. With respect to aroma characteristics, radar-plot responses of beers fermented with corncob- and corncob-filled-loofah-immobilized yeast were closer to those of the free-yeast control (
Figure 1E), suggesting a broadly similar aroma composition. The consistent separation pattern was also verified by PCA (
Figure 1F). Collectively, corncob and the corncob-filled loofah sponge were identified as more suitable carriers for yeast immobilization when both fermentation performance and aroma similarity to free-yeast fermentation were considered.
3.2. Optimization of the Fermentation Process for Mango Craft Beer
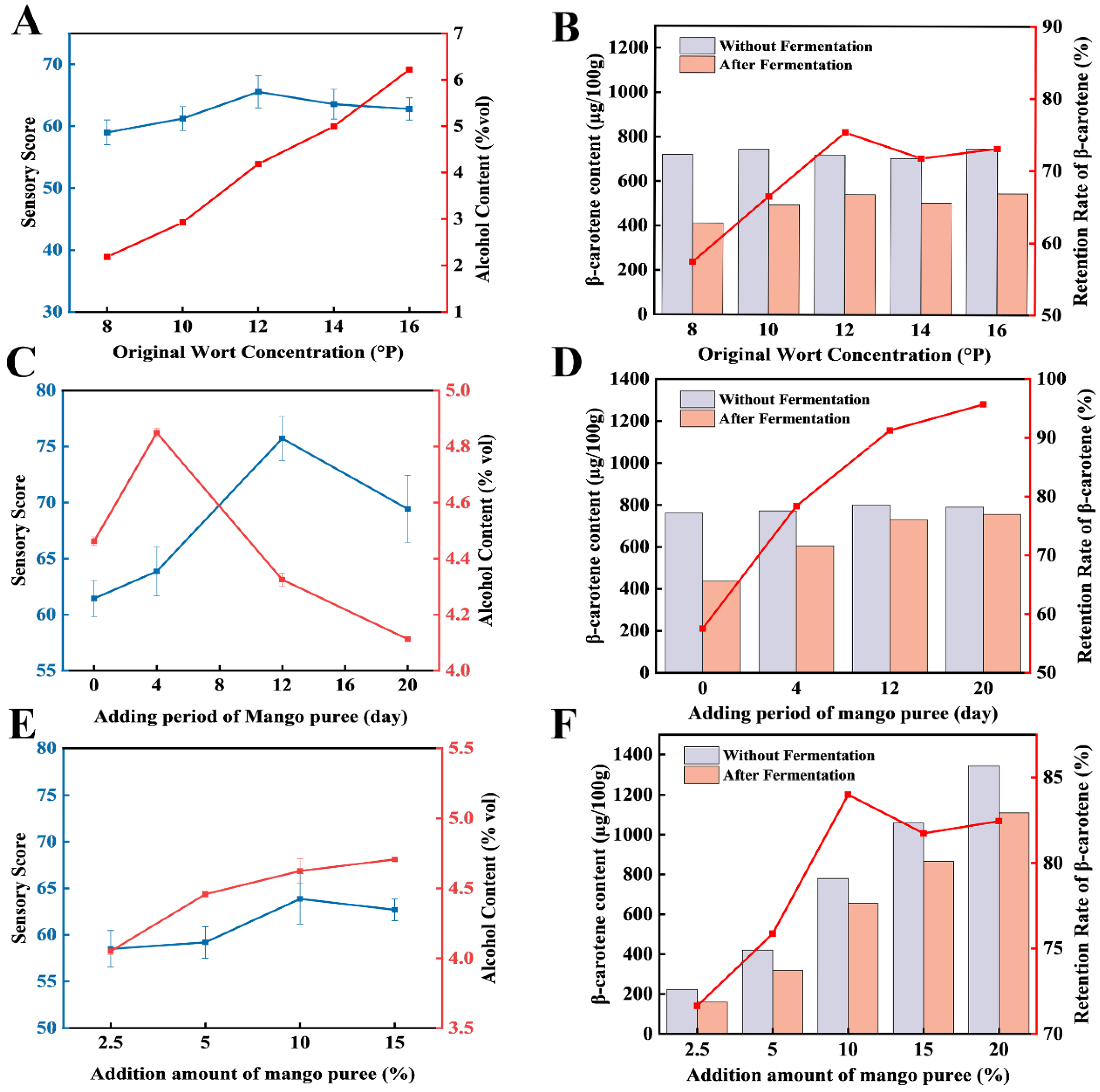
To determine the optimal fermentation process for mango craft beer, single-factor experiments were first conducted to evaluate the effects of original wort extract (
Figure 2A-B), the timing of mango purée addition (
Figure 2C-D), and the purée dosage (
Figure 2E-F), and to define appropriate ranges for subsequent optimization. As shown in the
Figure 2A and B, alcohol content increased progressively with increasing original wort extract, whereas both the sensory score and β-carotene retention exhibited a rise–fall pattern, reaching their maxima at 12 °P (65 points and 75.28%, respectively). The effects of the timing of mango purée addition are presented in
Figure 2C and 2D. With the addition time shifted to later stages, the sensory score and alcohol content initially increased and then decreased; the highest sensory score (75.7 points) was obtained when mango purée was added on day 12, which differed very significantly from the other time points. In contrast, β-carotene retention increased steadily as the addition time was delayed. The effects of purée dosage are shown in
Figure 2E and 2F: with increasing purée dosage, the sensory score and β-carotene retention first increased and then decreased, while alcohol content showed a continuous upward trend. Based on these single-factor results, the factor ranges selected for response surface methodology (RSM) were 10–14 °P for original wort extract, 4–20 d for the timing of mango purée addition, and 5%–15% for the purée dosage.
Subsequently, RSM was performed using original wort extract (A), mango purée dosage (B), and mango purée addition timing (C) as independent variables, with sensory score and β-carotene retention as response variables (
Table 2). The relative contributions of the factors to both responses followed the same order (
Table 3): mango purée addition timing (C) > mango purée dosage (B) > original wort extract (A). On the basis of the RSM optimization and practical feasibility of the fermentation process, the optimal conditions were determined to be an original wort extract of 12 °P, a mango purée dosage of 10%, and mango purée addition on day 18.
3.3. Naturally Immobilized Yeast Increases Alcohol Concentration and Modulates Fermentable Extract and pH of Mango Craft Beer
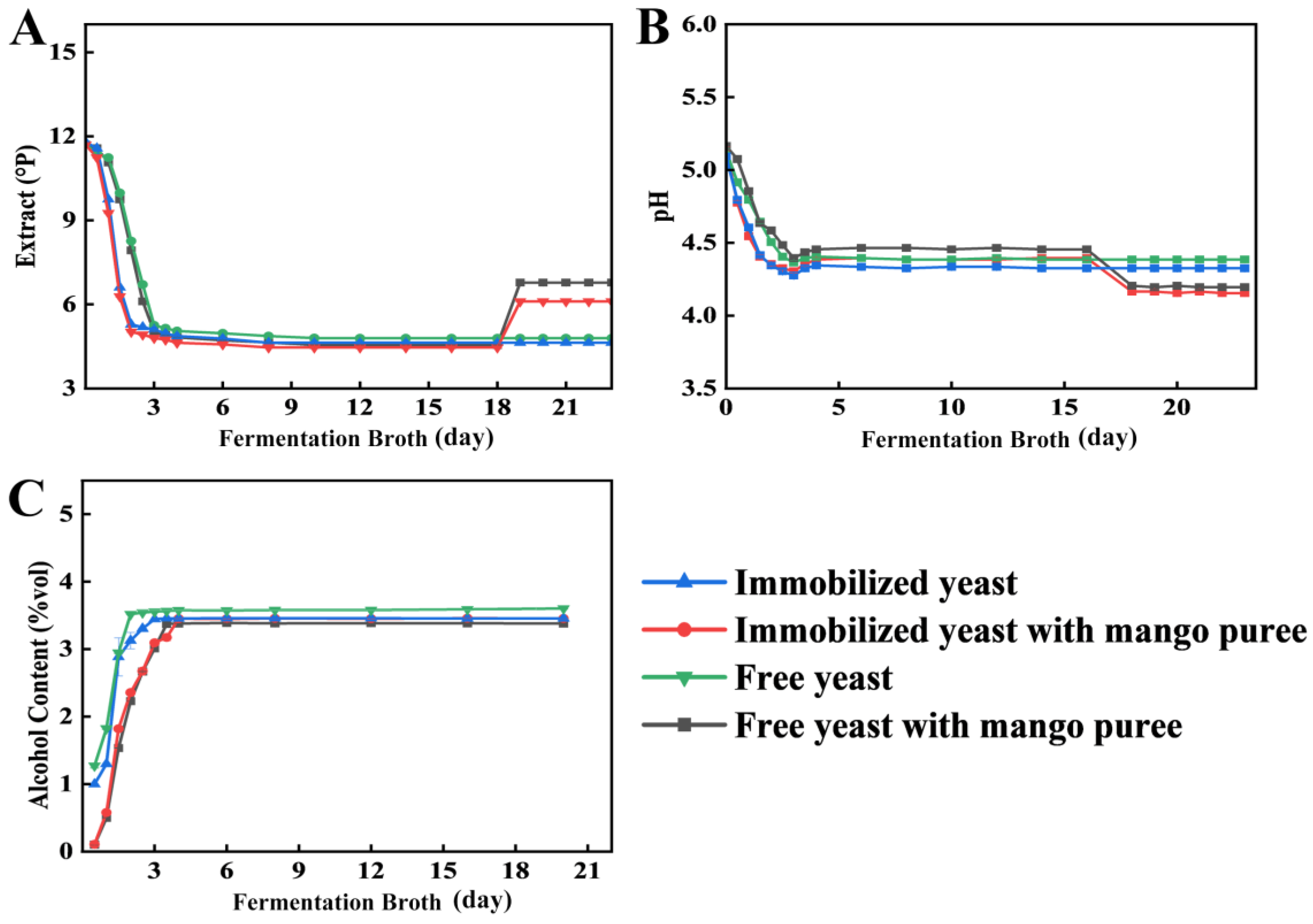
Fermentation was performed using the corncob-filled loofah sponge immobilized yeast prepared in this study under the previously optimized conditions for mango craft beer, and changes in extract (
Figure 3A), pH (
Figure 3B), and alcohol content (
Figure 3C) were comparatively evaluated. Relative to free yeast, faster decreases in both extract and pH were observed throughout fermentation when immobilized yeast was applied. The primary fermentation phase lasted approximately 48 h in the immobilized-yeast treatment, whereas 72 h was required in the free-yeast treatment, corresponding to a 33.3% reduction in primary fermentation time. The mean extract attenuation rate achieved with immobilized yeast was 0.14 °P/h, which was 1.50-fold higher than that obtained with free yeast (0.093 °P/h). In addition, the rate of pH decline in immobilized-yeast fermentations was 1.45-fold that of the free-yeast control.
Changes in alcohol content during fermentation are shown in
Figure 3C. The alcohol content increased with fermentation time and subsequently reached a plateau. However, alcohol production proceeded more rapidly in the immobilized-yeast fermentations. During primary fermentation, the mean alcohol production rate in batches supplemented with mango purée was 0.073 %vol/h for immobilized yeast, compared with 0.046 %vol/h for free yeast. Similarly, in batches without mango purée, the mean alcohol production rate was 0.070 %vol/h for immobilized yeast and 0.045 %vol/h for free yeast. Overall, the mean alcohol production rate obtained with immobilized yeast was approximately 1.59-fold that of the free-yeast treatment.
3.4. Flavor Evaluation of Mango Craft Beer Using an Electronic Nose and Electronic Tongue
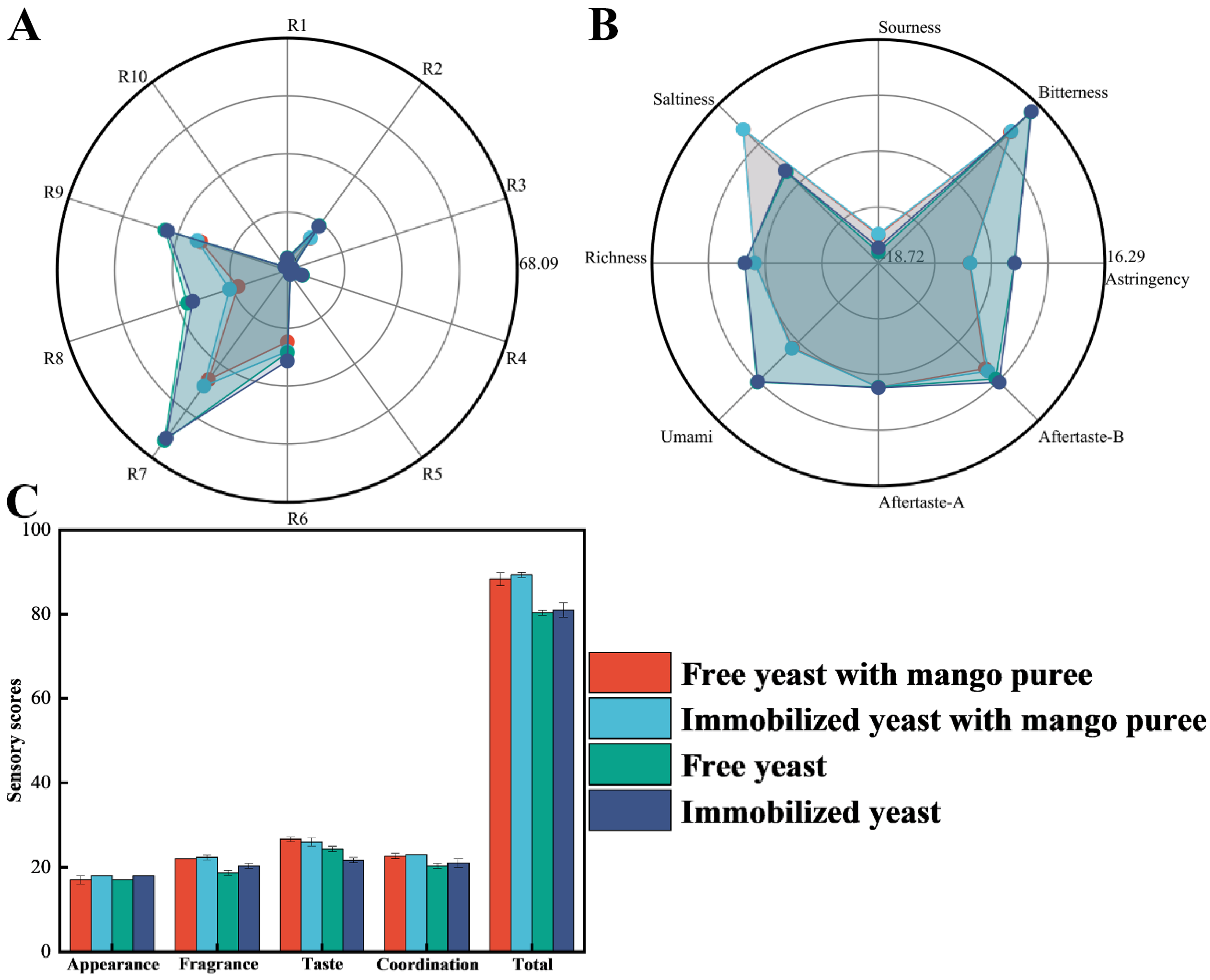
To characterize flavor attributes of mango craft beer, four pilot-scale beer batches were produced using an immobilized-yeast system, including two beers fermented with mango purée addition and two beers fermented without mango purée. Aroma profiles were evaluated using an electronic nose. As shown in
Figure 4A, responses were detected for all four beers across the ten sensors, and overall sensor-response magnitudes were broadly comparable between the mango-added beers and their non-mango counterparts. The significant differences were observed among the four beers in sensor responses associated with sulfur compounds (R7), aldehydes/ketones, and aromatic constituents. Relative to beers fermented without mango purée, lower signals related to sulfur compounds, aldehydes/ketones, and aromatic constituents were recorded in mango craft beers, with the most pronounced decrease observed for sulfur-related responses. Because excessive sulfur compounds and certain aldehydes/ketones may contribute to “green” notes and sulfur off-odors, leading to immature or unbalanced aroma and taste, these results suggest that mango purée addition can improve aroma composition and may alleviate harshness/bitterness, thereby enhancing overall sensory quality.
The five taste modalities (sourness, saltiness, umami, astringency, and bitterness) were further quantified for the four craft beers, as shown in
Figure 4B. All five taste attributes were detectable in each sample. Overall, partial overlap in sensor responses was observed between beers fermented with immobilized yeast and those fermented with free yeast, indicating broadly comparable taste profiles between the two fermentation modes. Sensory evaluation results indicated that all beer samples achieved a generally high level of overall acceptability (
Figure 4C). In contrast, significant differences were detected between mango-purée–fermented beers and beers fermented without mango purée in bitterness, astringency, umami, and saltiness. Notably, bitterness and astringency were significantly reduced in mango craft beers relative to the non-mango controls, suggesting that mango purée addition effectively mitigated the bitter/astringent mouthfeel.
3.5. Analysis of β-Carotene, Total Phenolics, and Alcohol Compounds in Mango Craft Beer
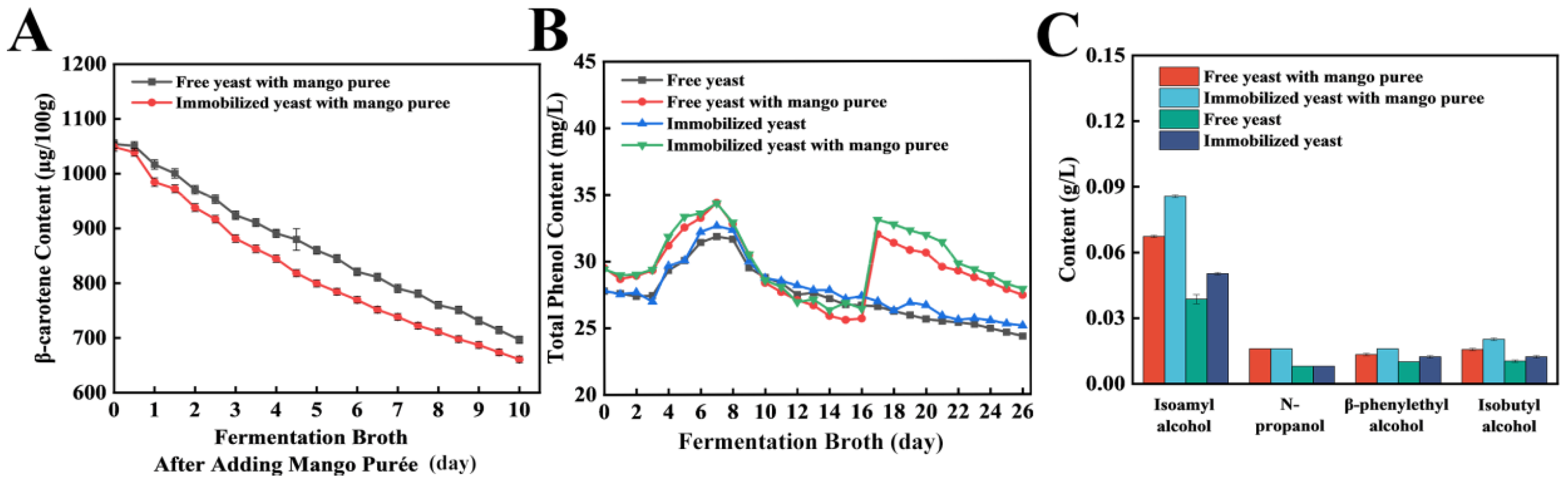
To evaluate the effect of mango purée addition on functional constituents in craft beer, mango purée was introduced during the late conditioning stage, after which β-carotene content was monitored (
Figure 5A). Following mango purée addition, a gradual decrease in β-carotene content was observed with increasing fermentation/conditioning time, which was consistent with the declining trend of total anthocyanins reported during mulberry wine fermentation by Zhou et al. [
16]. During the fermentation period following mango purée addition, a slightly faster decline in β-carotene was observed in the immobilized-yeast system than in the free-yeast system. However, the total losses were comparable in the immobilized-yeast system and the free-yeast system, reaching 325 μg/100 g and 310 μg/100 g, respectively. Ultimately, β-carotene retention in beers fermented with immobilized or free yeast was approximately 70%, representing a decrease of 22% relative to the shake-flask stage. Subsequently, dynamic changes in total phenolic content during fermentation were compared (
Figure 5B). Overall, total phenolics exhibited an increase followed by a decrease and then approached a plateau, which was in agreement with the trend reported [
17]. Given that mango purée inherently contains phenolic compounds [
18], a marked elevation in total phenolic content was observed immediately after mango purée addition, followed by a gradual decline as fermentation progressed. Compared with free-yeast fermentation, slightly higher total phenolic levels were generally maintained in beers fermented with immobilized yeast. In addition, mango purée addition increased the total phenolic content by 24.5%.
Higher alcohols (e.g., n-propanol, isobutanol, n-butanol, n-pentanol, isoamyl alcohol, and β-phenylethanol) constitute key components of the beer flavor matrix, and their concentrations can exert bidirectional effects on sensory quality [
19]. To facilitate process-oriented modulation of higher alcohols for improved flavor and drinking experience, the influence of pilot-scale fermentation with immobilized yeast on higher-alcohol composition was further evaluated (
Figure 5C). Across the four craft beer samples, n-propanol, isobutanol, isoamyl alcohol, and β-phenylethanol were detected, whereas n-butanol and n-pentanol were not detected. All detected compounds were within the usual concentration ranges reported for beer [
20,
21], and no obvious adverse sensations were observed during sensory evaluation. Total higher-alcohol levels were higher in beers fermented with mango purée than in their non-mango counterparts. In addition, within the mango beers, higher alcohol concentrations were observed when fermentation was carried out with immobilized yeast compared to free yeast. Overall, these findings demonstrate that both mango purée addition and yeast immobilization significantly affected higher-alcohol accumulation, providing a basis for process optimization to modulate higher-alcohol profiles and improve beer flavor quality.
3.6. Identification of Volatile Flavor Compounds in Mango Craft Beer
Volatile compounds in beer are highly diverse, among which alcohols and esters represent the two most characteristic classes and play a decisive role in the overall aroma profile. Volatile flavor compounds in the four craft beers were identified and semi-quantified by solid-phase microextraction coupled with gas chromatography–mass spectrometry (SPME–GC–MS), and the results are summarized in
Table 4. Across all four beers, alcohols exhibited the highest relative abundance, followed by esters. Given that terpenes have been reported as major volatile constituents of mango [
22], terpene compounds—besides esters—were also present at markedly higher relative levels in mango-purée beers, ranking second only to alcohols. Overall, 13 alcohols, 22 esters, 4 acids, 3 aldehydes/ketones, 4 alkanes, 1 phenol, 18 terpenes, and 1 estragole were detected. Compared with beers produced without mango purée, several characteristic compounds were uniquely detected in mango-purée beers, including linalool and p-methylphenyl isopropanol (alcohols); ethyl (Z)-9-tetradecenoate, ethyl myristate, diisobutyl phthalate, methyl palmitate, and ethyl (E)-11-hexadecenoate (esters); octanoic acid and lauric acid (acids); β-ionone (aldehydes/ketones); and 4-carene, α-ocimene, γ-terpinene, p-mentha-1,3,8-triene, (2E)-2,6-dimethyl-1,3,5,7-octatetraene, and β-junipene (terpenes). In addition, alkanes, phenolic compounds, and estragole were detected in all four beers, whereas isobutyl octanoate, valencene, and cubebene were detected only in beers fermented with free yeast and at low relative abundances. Additionally, compared with beers fermented without mango purée, ethyl decanoate was increased by 28.7% in mango-purée–fermented beers, a compound associated with pineapple-like and fruity aroma notes. Meanwhile, the total relative abundance of terpenes was 6.5-fold higher than that in beers fermented without mango purée. Among the terpenes, terpinolene exhibited the highest relative abundance and was approximately 100-fold higher than in the non-mango beers. The volatile-compound results were consistent with electronic-nose, electronic-tongue, and sensory-evaluation outcomes, indicating that mango purée addition alleviated bitter/astringent perception and markedly improved aroma expression, flavor complexity, body harmony, and overall acceptance. Collectively, the enriched and elevated volatile profile conferred more distinctive flavor attributes and a more pleasant mouthfeel to the mango craft beer.
4. Discussion
With the continuing rise in consumer demand for higher-quality alcoholic beverages with diversified flavor profiles, flavored beers characterized by distinctive aroma and mouthfeel have become a focal point of both research and market interest. To shorten the brewing cycle and promote the formation of aroma-active compounds during fermentation, yeast immobilization has attracted considerable attention because yeast stability, cell reusability, and fermentation efficiency can be enhanced, while natural immobilization supports are particularly attractive due to their wide availability, good biocompatibility, low cost, and environmental friendliness. In this study, a composite natural carrier was constructed by packing corncob into a loofah sponge matrix, and brewing yeast was immobilized via adsorption to intensify fermentation, enabling the successful production of mango craft beer. An adsorption-based approach was employed to construct an immobilized-yeast system on natural carriers, and a unit-mass difference of 13.3% was obtained before versus after immobilization. Compared with the reinforced alginate carrier reported by Bangrak et al. [
23], which yielded a unit-mass difference of 9.7% for Saccharomyces cerevisiae immobilization in fuel-alcohol production, a higher yeast-retention capacity was prepared in this study, supporting its favorable immobilization performance. This superiority may be attributable to the more porous and loosely structured nature of corncob and loofah sponge relative to sweet sorghum stalk, with increased specific surface area and porosity providing additional sites and space for cell attachment [
24]. Natural supports have also been investigated for yeast immobilization in beer fermentation; for example, peanut shells and coconut shells were reported to increase ester and alcohol contents while reducing diacetyl and dimethyl sulfide, and peanut-shell immobilization was shown to shorten fermentation time while enhancing reducing-sugar consumption and alcohol yield [
25]. However, peanut and coconut shells typically require NaOH pretreatment to remove lignin and generate tubular cellulose structures to achieve satisfactory immobilization efficiency [
26]. In contrast, the corncob-loofah support employed here required no elaborate pretreatment and enabled effective immobilization without chemical reagents or additives, thereby improving fermentation efficiency and shortening primary fermentation.
Fermentation process parameters were further shown to exert pronounced effects on the sensory quality and nutritional/functional constituents of mango craft beer [
27]. Variations in mango purée dosage were expected to alter the nutrients released into the fermentation matrix, thereby modulating sweetness perception and alcohol formation, while the initial original wort extract was also associated with changes in final alcohol content and β-carotene levels. Because yeast physiology differs across fermentation stages-rapid proliferation and high metabolic activity during primary fermentation versus by-product conversion during maturation-the stage at which fruit is added was also expected to influence sensory scores and nutrient retention [
4,
28]. Accordingly, the optimal conditions were determined as an original wort extract of 12 °P, a mango purée dosage of 10%, and mango purée addition on day 18. Under these conditions, β-carotene degradation was still observed during fermentation, which may arise from thermal degradation, photooxidation, chemical oxidation, and potentially biological degradation [
29]. Varakumar et al. [
30] reported β-carotene degradation rates of 17.9–60.7% in mango wines produced from different cultivars fermented with Saccharomyces bayanus. In contrast, a β-carotene retention of 91.25% was achieved under the response-surface-optimized process established here, exceeding that reported for the S. bayanus mango-wine system; concurrently, high sensory scores were obtained, indicating that intense mango-derived flavor characteristics were preserved while high β-carotene retention was achieved.
Mango purée addition exerted a systemic influence on the flavor attributes of craft beer as well as on the levels of key constituents [
31]. During fermentation, β-carotene was progressively degraded, with total losses of 325 μg/100 g and 310 μg/100 g observed in the immobilized-yeast and free-yeast systems, respectively; the final retention in both systems remained at approximately 70%, representing a decrease of 22% relative to the shake-flask stage. This degradation was more likely attributable to oxygen ingress and the resulting oxidative reactions during fermentation, rather than to differences in alcohol content. An oxidation-related effect was also reflected in the temporal behavior of total phenolics, which increased after mango purée addition and subsequently decreased. With respect to volatile constituents, the relative abundance of terpenes was markedly increased by mango purée addition, reaching 6.5-fold that of beers produced without purée. Among these, terpinolene exhibited the most pronounced increase, with an approximately 100-fold elevation relative to the non-mango beers; this compound has been recognized as a key contributor to the characteristic aroma of mango. In addition, β-carotene degradation may have promoted the formation of aroma-active products such as β-ionone, thereby enriching aroma complexity. Notably, several volatile compounds that have been rarely reported in previous studies, including 3-methylbutyl octanoate and ethyl myristate, were also identified; their occurrence may be associated with varietal and geographical differences in the mango raw material. Taken together, instrumental analyses in combination with sensory evaluation demonstrated that mango purée addition effectively attenuated bitterness and astringency while significantly enhancing aroma complexity, body harmony, and overall acceptability, indicating its potential to improve both sensory quality and the nutritional/flavor profile of craft beer.
5. Conclusions
During mango craft beer fermentation, synergistic improvements in both production efficiency and product quality were achieved when immobilized yeast was integrated with a mango purée addition strategy. Response surface optimization identified the optimal fermentation conditions as an original wort extract of 12 °P, a mango purée dosage of 10%, and purée addition on day 18. Under the optimized fermentation conditions, the mango craft beer achieved a sensory score of 81, with a β-carotene retention rate of 91.25%. In addition, the mango craft beer exhibited a more diverse profile of volatile flavor compounds and enhanced nutritional composition compared with the control. Collectively, the development of mango craft beer provides a basis for value-added processing of mango, enhances the economic value of mango-derived products, diversifies the beer market, and offers a practical reference for the development and utilization of fruit-flavored beers.
Author Contributions
Methodology, C.C., T.W. and Y.Q.; validation, S.L. and H.L.; resources, L.W., Q.S. and J.M.; investigation, L.D. and Q.S.; formal analysis, L.D.; supervision, Q.S. and J.M.; writing—original draft preparation, C.C.; writing—review and editing, C.C., T.W., S.L., H.L., L.W., L.L. and J.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Research Projects of Guangxi Vocational University of Agriculture (XKJ2320, XTKJ2515).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Gobbi, L.; Stankovic, M.; Ruggeri, M.; Savastano, M. Craft Beer in Food Science: A Review and Conceptual Framework. Beverages 2024, 10, 91. [Google Scholar] [CrossRef]
- Glowacki, A.; Paszkot, J.; Pietrzak, W.; Kawa-Rygielska, J. Sour Fruit Beers-Ethanol and Lactic Acid Fermentation in Beer Production. Molecules 2025, 30, 3358. [Google Scholar] [CrossRef] [PubMed]
- Castro Marin, A.; Baris, F.; Romanini, E.; Lambri, M.; Montevecchi, G.; Chinnici, F. Physico-Chemical and Sensory Characterization of a Fruit Beer Obtained with the Addition of Cv. Lambrusco Grapes Must. Beverages 2021, 7, 34. [Google Scholar] [CrossRef]
- Kawa-Rygielska, J.; Adamenko, K.; Kucharska, A.Z.; Prorok, P.; Piorecki, N. Physicochemical and Antioxidative Properties of Cornelian Cherry Beer. Food Chem. 2019, 281, 147–153. [Google Scholar] [CrossRef]
- Gasinski, A.; Kawa-Rygielska, J.; Szumny, A.; Czubaszek, A.; Gasior, J.; Pietrzak, W. Volatile Compounds Content, Physicochemical Parameters, and Antioxidant Activity of Beers with Addition of Mango Fruit (Mangifera Indica). Molecules 2020, 25, 3033. [Google Scholar] [CrossRef]
- Bonatto, D. The Diversity of Commercially Available Ale and Lager Yeast Strains and the Impact of Brewer’s Preferential Yeast Choice on the Fermentative Beer Profiles. Food Research International 2021, 141, 110125. [Google Scholar] [CrossRef]
- Iattici, F.; Catallo, M.; Solieri, L. Designing New Yeasts for Craft Brewing: When Natural Biodiversity Meets Biotechnology. Beverages 2020, 6(1), 3. [Google Scholar] [CrossRef]
- Pilkington, P.H.; Margaritis, A.; Mensour, N.A.; Russell, I. Fundamentals of Immobilised Yeast Cells for Continuous Beer Fermentation: A Review. J. Inst. Brew. 1998, 104, 19–31. [Google Scholar] [CrossRef]
- Verbelen, P.J.; De Schutter, D.P.; Delvaux, F.; Verstrepen, K.J.; Delvaux, F.R. Immobilized Yeast Cell Systems for Continuous Fermentation Applications. Biotechnol. Lett. 2006, 28, 1515–1525. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, M.; Carmona-Jiménez, P.; García-Martínez, T.; Jorrín-Novo, J.V.; Moreno, J.; Rey, M.D.; Moreno-García, J. Use of Yeast Biocapsules as a Fungal-Based Immobilized Cell Technology for Indian Pale Ale-Type Beer Brewing. Applied Microbiology and Biotechnology 2022, 106(22), 7615–7625. [Google Scholar] [CrossRef] [PubMed]
- Benucci, I.; Cecchi, T.; Lombardelli, C.; Maresca, D.; Mauriello, G.; Esti, M. Novel Microencapsulated Yeast for the Primary Fermentation of Green Beer: Kinetic Behavior, Volatiles and Sensory Profile. Food Chem. 2021, 340, 127900. [Google Scholar] [CrossRef] [PubMed]
- Milinčić, D.D.; Salević Jelić, A.S.; Lević, S.M.; Stanisavljević, N.S.; Milošević, T.; Pavlović, V.B.; Gašić, U.M.; Obradović, N.S.; Nedović, V.A.; Pešić, M.B. Craft Beer Produced by Immobilized Yeast Cells with the Addition of Grape Pomace Seed Powder: Physico-Chemical Characterization and Antioxidant Properties. Foods 2024, 13(17), 2801. [Google Scholar] [CrossRef] [PubMed]
- Brunet, S.I.; Erceg, T.; Janjusevic, L.; Birgermajer, S.; Odanovic, M.; Puskas, V.; Miljic, U. Materials for Yeast Immobilization in Alcoholic Fermentation: Bridging Conventional Techniques and 3D Bioprinting. Polymers 2025, 17, 3094. [Google Scholar] [CrossRef]
- Lei, Y.; Yang, W.; Yuan, Y.; Wei, G.; Guo, L. Functionalization of Loofah Sponge as a Natural Cellulose-Based Material for Water Environment Remediation and Sustainability Assessment. Carbohydr. Polym. 2025, 358, 123514. [Google Scholar] [CrossRef]
- Guadalupe Ortega, V.; Alberto Ramirez, J.; Velazquez, G.; Tovar, B.; Mata, M.; Montalvo, E. Effect of High Hydrostatic Pressure on Antioxidant Content of “ataulfo” Mango during Postharvest Maturation. Food Sci. Technol. 2013, 33, 561–568. [Google Scholar] [CrossRef]
- Zhou, H.; Chai, Y.; Huang, W.; Zhan, J.; You, Y. Screening for High-Yielding Pyruvate and Acetaldehyde Yeasts and Their Application in Improving the Stability of Anthocyanin in Mulberry Wine. Foods 2025, 14(13), 2278. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, D.O.; Guido, L.F. A Review on the Fate of Phenolic Compounds during Malting and Brewing: Technological Strategies and Beer Styles. Food Chem. 2022, 372, 131093. [Google Scholar] [CrossRef]
- Ankeli Jack, A.; Igbum Ogbene, G.; Okibe Friday, G. Phytochemical Composition and Functional Properties of Fruit Purees Produced from Some Indigenous Varieties of Mango, Orange and Watermelon. Journal of Food and Nutrition Sciences 2024, 12(6), 294–301. [Google Scholar] [CrossRef]
- Pires, E.J.; Teixeira, J.A.; Branyik, T.; Vicente, A.A. Yeast: The Soul of Beer’s Aroma-a Review of Flavour-Active Esters and Higher Alcohols Produced by the Brewing Yeast. Appl. Microbiol. Biotechnol. 2014, 98, 1937–1949. [Google Scholar] [CrossRef]
- Nasuti, C.; Solieri, L. Yeast Bioflavoring in Beer: Complexity Decoded and Built up Again. Fermentation 2024, 10(4), 183. [Google Scholar] [CrossRef]
- Sun, Z.; Xiao, D. Review in Metabolic Modulation of Higher Alcohols in Top-Fermenting Yeast. Advances in Applied Biotechnology 2018, 444, 767–773. [Google Scholar] [CrossRef]
- Bonneau, A.; Boulanger, R.; Lebrun, M.; Maraval, I.; Gunata, Z. Aroma Compounds in Fresh and Dried Mango Fruit (Mangifera Indica L. Cv. Kent): Impact of Drying on Volatile Composition. Int. J. Food Sci. Technol. 2016, 51, 789–800. [Google Scholar] [CrossRef]
- Bangrak, P.; Limtong, S.; Phisalaphong, M. Continuous Ethanol Production Using Immobilized Yeast Cells Entrapped in Loofa-Reinforced Alginate Carriers. Braz. J. Microbiol. 2011, 42, 676–684. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Gan, L.; Huang, J. Design, Manufacturing and Functions of Pore-Structured Materials: From Biomimetics to Artificial. Biomimetics 2023, 8, 140. [Google Scholar] [CrossRef]
- Kandylis, P.; Dimitrellou, D.; Gousi, M.; Kordouli, E.; Kanellaki, M. Effect of Immobilization Support and Fermentation Temperature on Beer and Fermented Milk Aroma Profiles. Beverages 2021, 7(3), 47. [Google Scholar] [CrossRef]
- Estela-Escalante, W.D.; Rosales-Mendoza, S.; Moscosa-Santill; aacute, M. N Yeast Immobilization on Peanut Hulls for Potential Application in Beer Fermentation. Am. J. Biochem. Biotechnol. 2019, 15, 101–109. [Google Scholar] [CrossRef]
- Kitessa, D.A. Review on Effect of Fermentation on Physicochemical Properties, Anti-Nutritional Factors and Sensory Properties of Cereal-Based Fermented Foods and Beverages. Ann. Microbiol. 2024, 74(1), 32. [Google Scholar] [CrossRef]
- Ducruet, J.; Rébénaque, P.; Diserens, S.; Kosińska-Cagnazzo, A.; Héritier, I.; Andlauer, W. Amber Ale Beer Enriched with Goji Berries - the Effect on Bioactive Compound Content and Sensorial Properties. Food Chem. 2017, 226, 109–118. [Google Scholar] [CrossRef]
- Zorn, H.; Langhoff, S.; Scheibner, M.; Nimtz, M.; Berger, R.G. A Peroxidase from Lepista Irina Cleaves β,β-Carotene to Flavor Compounds. Biol. Chem. 2003, 384(7), 1049–1056. [Google Scholar] [CrossRef]
- Varakumar, S.; Kumar, Y.S.; Reddy, O.V.S. Carotenoid Composition of Mango (Mangifera Indica L.) Wine and Its Antioxidant Activity. J. Food Biochem. 2011, 35(5), 1538–1547. [Google Scholar] [CrossRef]
- Ledeker, C.N.; Suwonsichon, S.; Chambers, D.H.; Adhikari, K. Comparison of Sensory Attributes in Fresh Mangoes and Heat-Treated Mango Purees Prepared from Thai Cultivars. LWT 2014, 56(1), 138–144. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).