Submitted:
12 February 2026
Posted:
14 February 2026
You are already at the latest version
Abstract
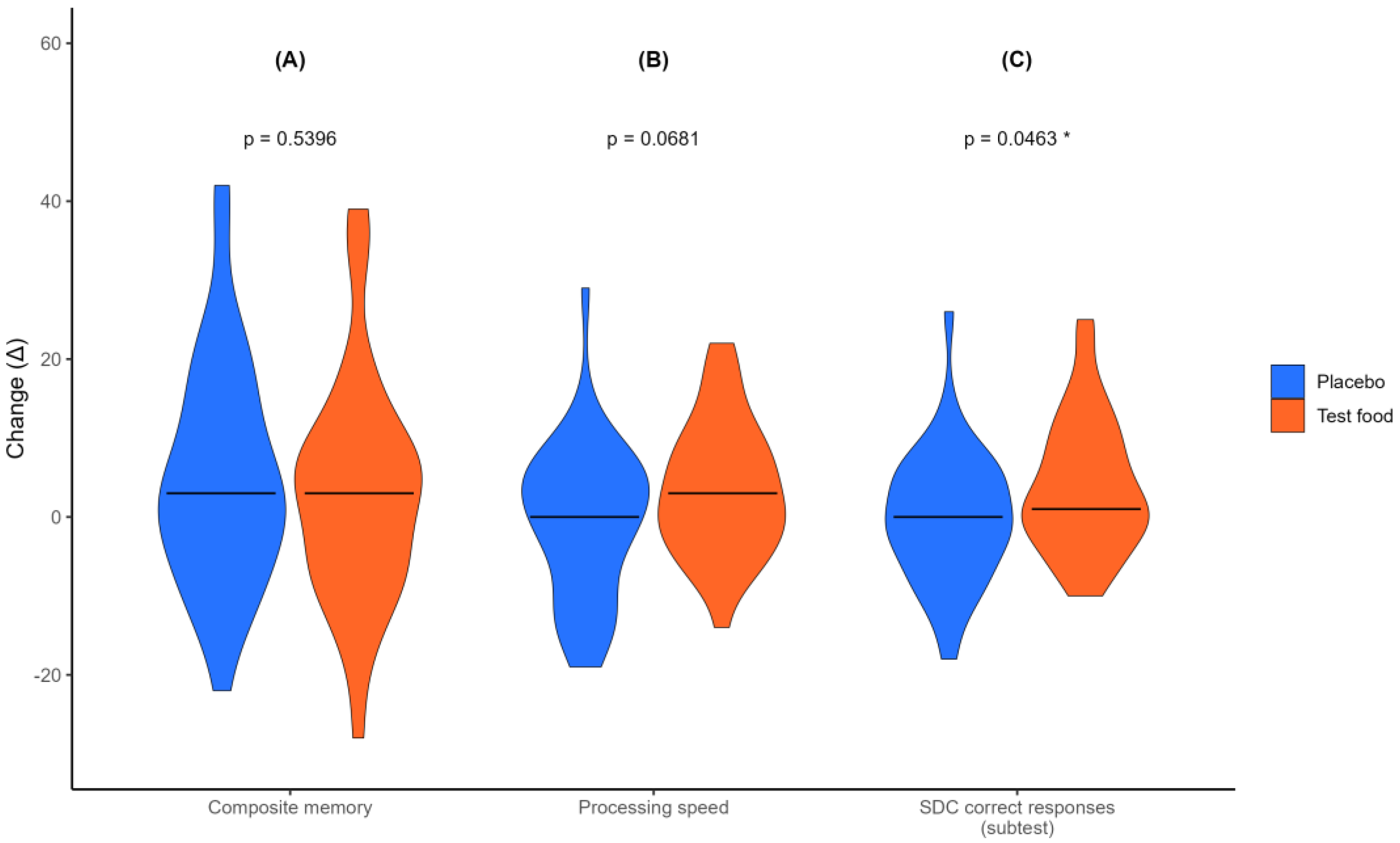
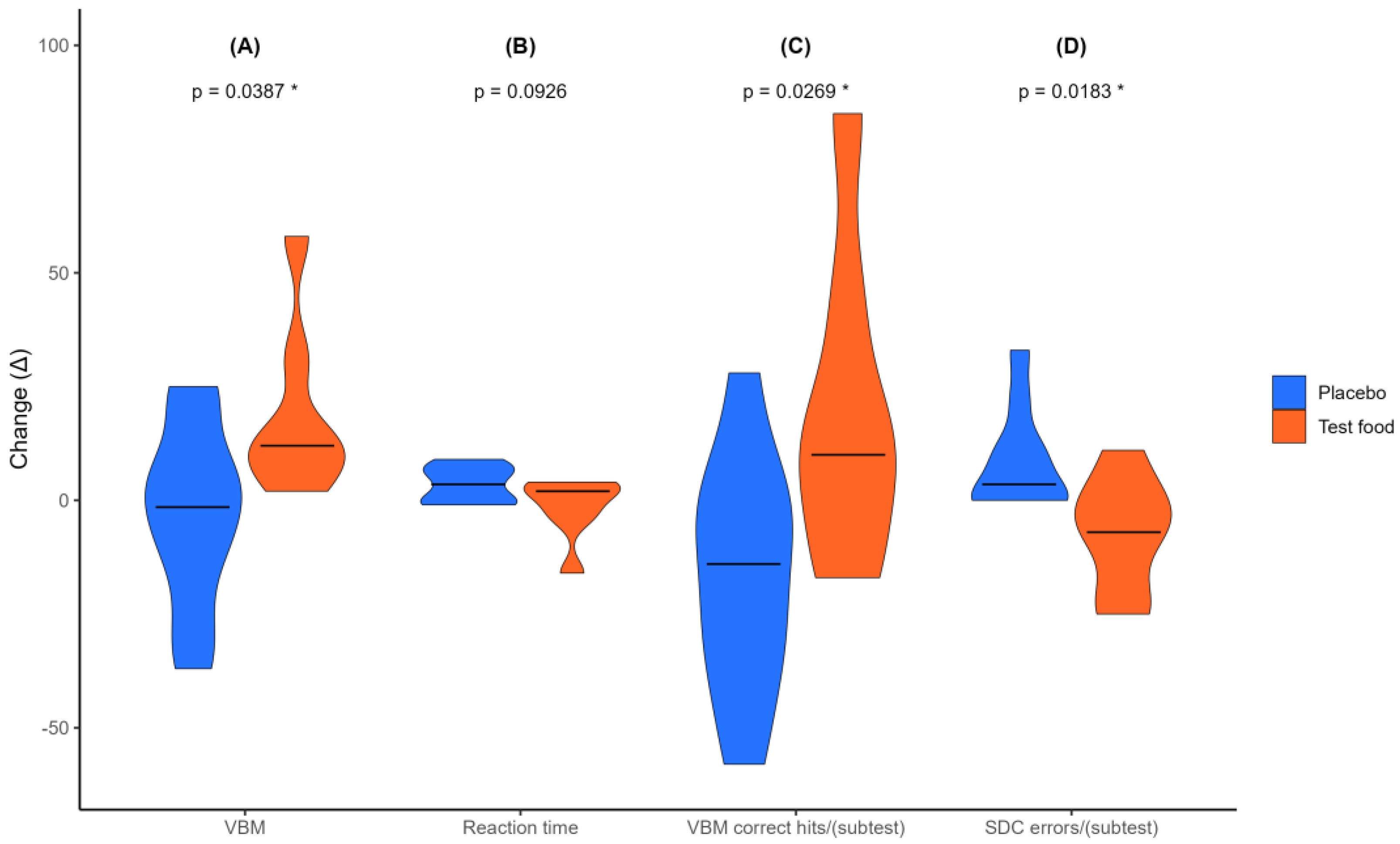
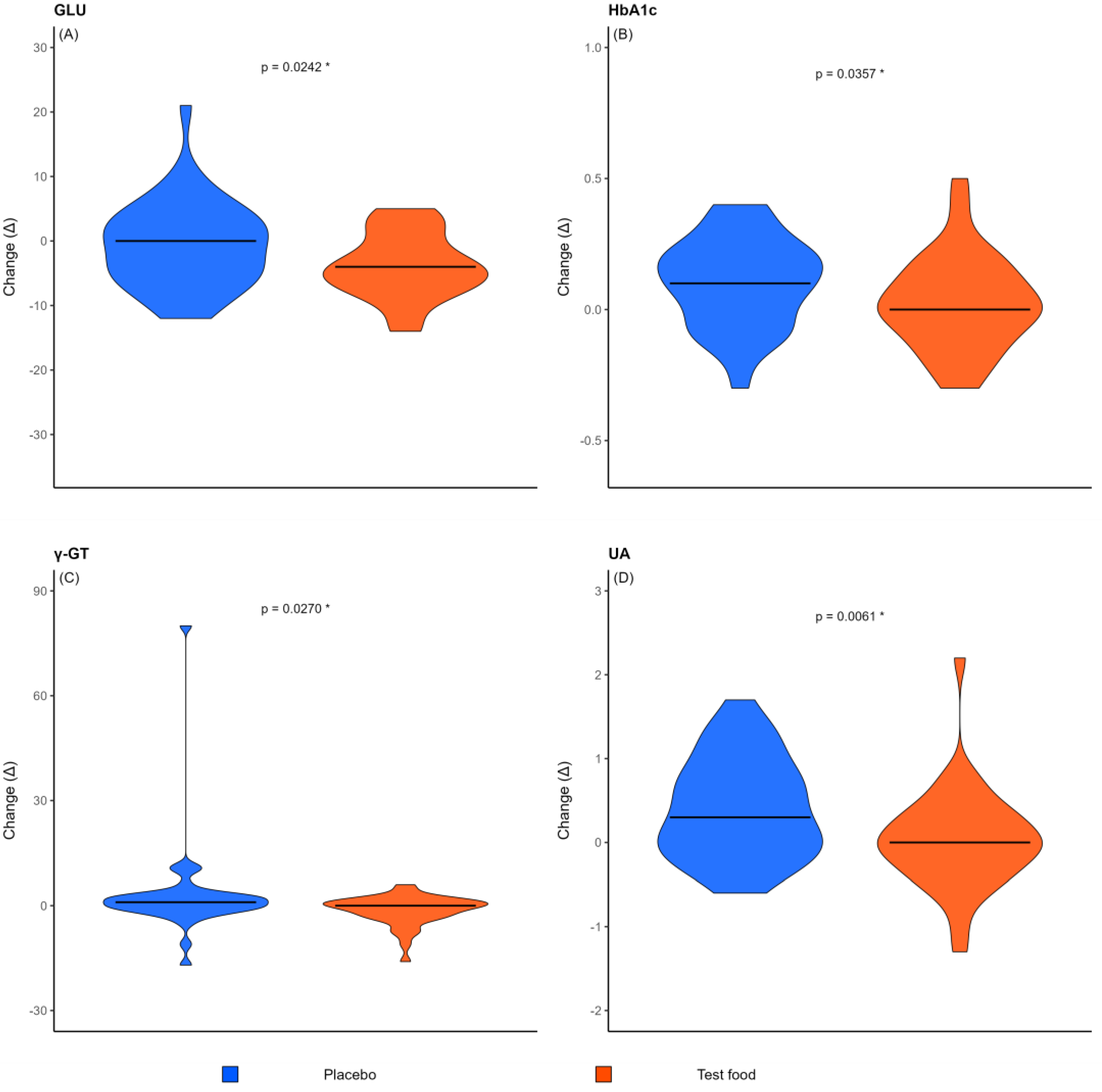
Background: As the prevention of dementia onset and the slowing of its progression become a global challenge, nutritional interventions focusing on the gut-brain axis are garnering attention. This study examined the effects of a complex functional food containing Clostridium butyricum, 3-(4-hydroxy-3-methoxyphenyl)propionic acid (HMPA) derived from fermented rice bran, and salmon milt-derived DNA on cognitive function and bowel habits in middle-aged and older adults. Methods: A randomized, double-blind, placebo-controlled, parallel-group study was conducted on 80 men and women aged 55–79 years with subjective memory complaints and an MMSE-J score of 24 or higher (UMIN ID: UMIN000057405). Participants ingested either the Test food (containing 1.4 × 10⁷ CFU of C. butyricum, 11.5 mg of HMPA, and 45 mg of salmon milt-derived DNA) or a Placebo once daily for 12 weeks. The primary outcome was cognitive function assessed by Cognitrax. Secondary outcomes included subjective cognition, mood state, bowel habits, blood biochemical parameters, and safety. A subgroup analysis was performed for participants aged ≥65 years with MMSE-J scores of 24–27. Results: In the overall analysis, no significant difference was observed in Composite Memory between groups; however, the number of correct responses in the Symbol Digit Coding (SDC) test, which is related to attention and processing speed, significantly improved in the Test food group. In the subgroup analysis, significant improvements in verbal memory-related indices were observed in the Test food group. Bowel habit indices showed no consistent between-group differences. Exploratory evaluations showed significant improvements in metabolic markers such as fasting plasma glucose, HbA1c, γ-GT, and uric acid in the Test food group. No serious adverse events occurred, and adherence to intake was high. Conclusions: Twelve-week intake of this complex functional food improved indices of attention and processing speed in the overall population, improved verbal memory indices in the high-risk subgroup, and positively affected metabolic markers. However, no clear effects on bowel habits were confirmed. Larger-scale and longer-term verification is needed in the future.
Keywords:
1. Introduction
2. Materials and Methods
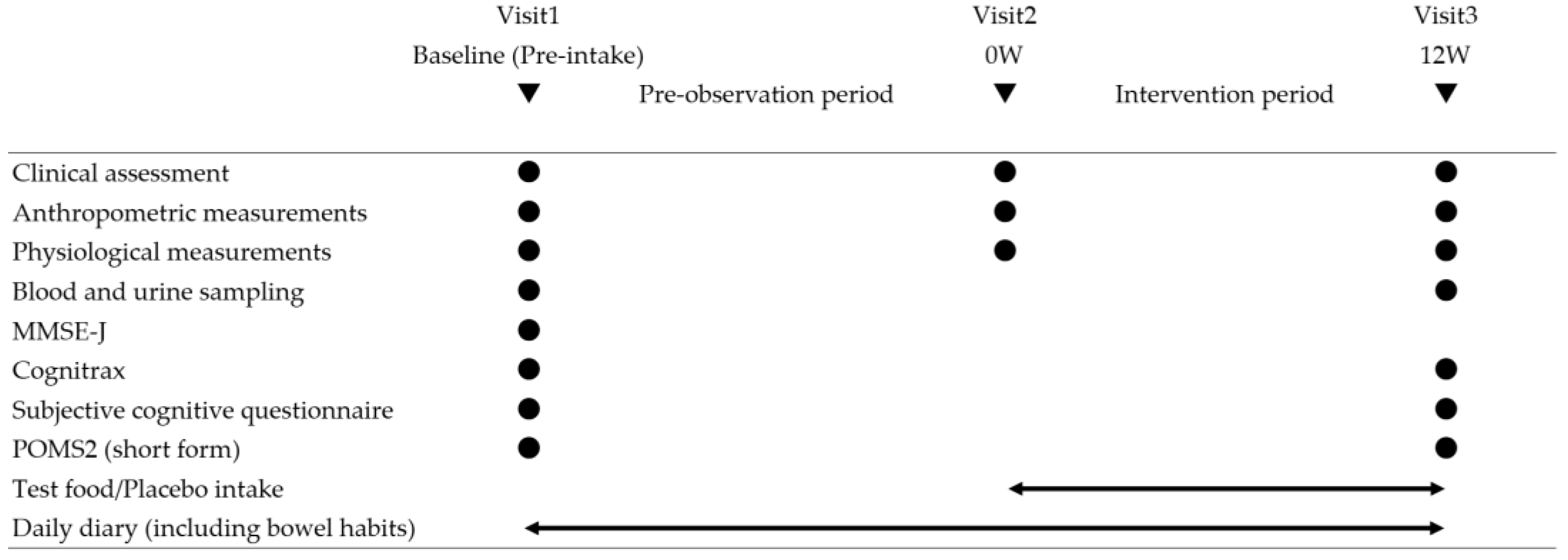
2.1. Study Design and Ethical Considerations
2.2. Participants
2.3. Randomization and Blinding
2.4. Intervention
2.5. Outcome Measures
2.5.1. Cognitive Function (Primary Outcome)
2.5.2. Subjective Cognitive Function
2.5.3. Psychological State
2.5.4. Biochemical Parameters
2.5.5. Bowel Habits
2.6. Compliance and Safety
2.7. Statistical Analysis
2.8. Use of Generative AI
3. Results
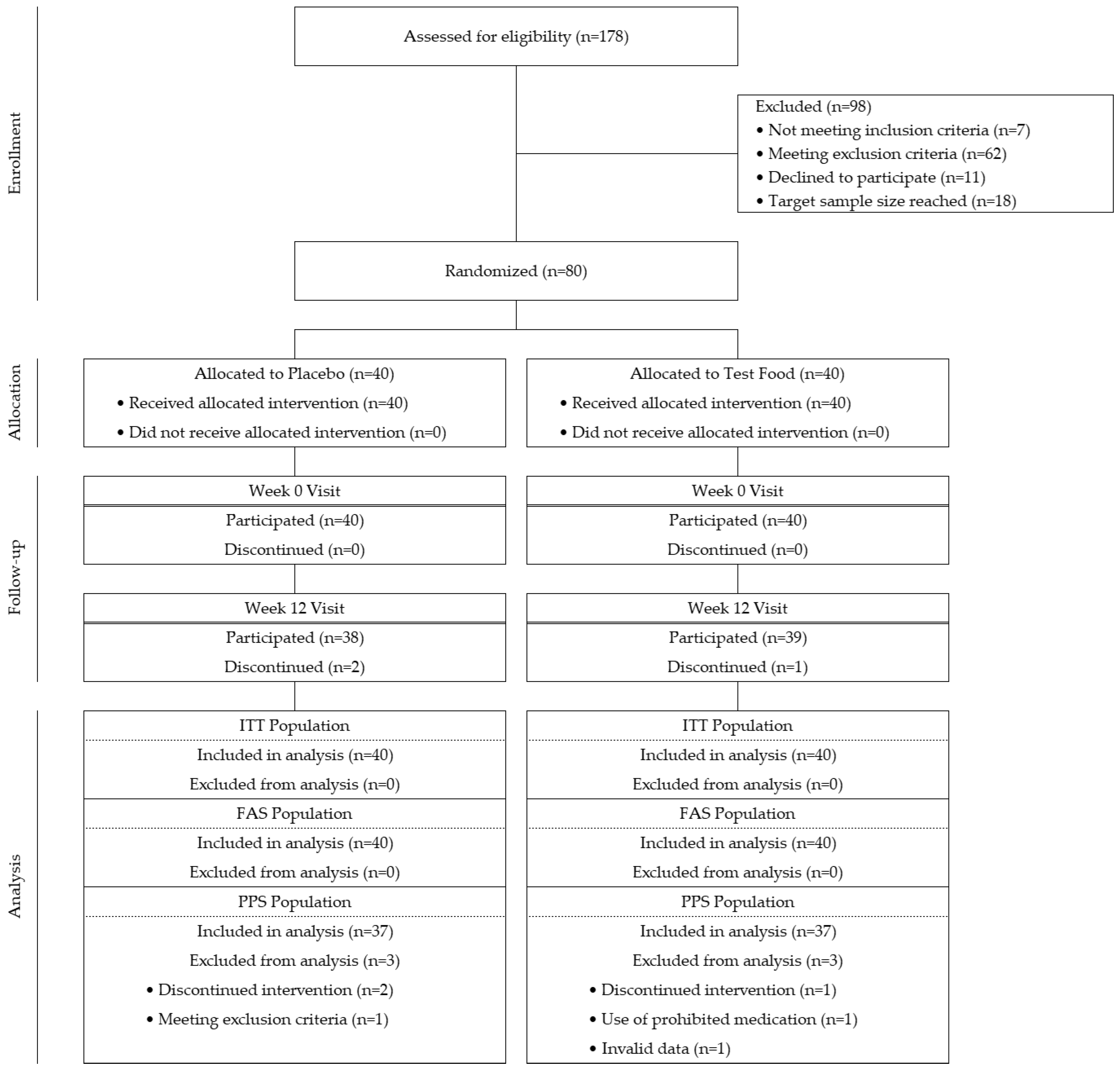
3.1. Participant Flow and Analysis Sets
3.2. Baseline Characteristics
| Items (Unit) | Placebo (n = 37) | Test food (n = 37) | p-value |
|---|---|---|---|
| Mean (SD) | Mean (SD) | ||
| Age (years) | 65.6 (4.3) | 65.6 (4.1) | 0.9265 a |
| Male | 24 | 23 | 1.0000 b |
| Female | 13 | 14 | |
| Height (cm) | 164.74 (8.97) | 164.66 (7.94) | 0.9684 c |
| Body weight (kg) | 63.23 (11.61) | 62.10 (9.18) | 0.6431 c |
| BMI (kg/m²) | 23.15 (2.89) | 22.85 (2.59) | 0.8161 a |
| Variable | Placebo (n = 37) | Test food (n = 37) | p-value |
|---|---|---|---|
| Mean (SD) | Mean (SD) | ||
| MMSE-J score | 27.4 (1.9) | 27.8 (1.5) | 0.3688 a |
| NCI (Cognitrax) | 102.4 (8.2) | 103.1 (9.1) | 0.7480 c |
| Composite Memory (Cognitrax) | 92.9 (16.5) | 96.1 (19.9) | 0.4481 c |
| BDNF (pg/mL) | 67.05 (50.37) | 330.85 (968.43) | 0.1520 a |
| Defecation frequency (times/week) | 7.9 (3.9) | 7.9 (2.8) | 0.7905 a |
| Glucose (mg/dL) | 96.0 (8.2) | 96.1 (7.4) | 0.9645 c |
| HbA1c (NGSP, %) | 5.50 (0.28) | 5.57 (0.32) | 0.2633 a |
| Aspartate aminotransferase (U/L) | 23.1 (4.6) | 22.5 (5.9) | 0.6456 c |
| Alanine aminotransferase (U/L) | 18.5 (6.9) | 19.6 (8.3) | 0.5190 c |
| Gamma-glutamyl transferase (U/L) | 26.2 (12.4) | 24.2 (13.6) | 0.3520 a |
| Total cholesterol (mg/dL) | 210.7 (26.2) | 206.7 (25.9) | 0.5110 c |
| Triglycerides (mg/dL) | 105.9 (57.1) | 93.5 (37.5) | 0.5377 a |
| HDL cholesterol (mg/dL) | 66.2 (18.6) | 66.7 (15.3) | 0.7661 a |
| LDL cholesterol (mg/dL) | 123.6 (24.4) | 122.9 (27.4) | 0.9146 c |
3.3. Effects on Cognitive Function (Primary Outcome)
3.4. Subgroup Analysis of Cognitive Function
3.5. Effects on Metabolic and Biochemical Parameters
3.6. Other Secondary Outcomes
3.6.1. Subjective Cognitive Function
3.6.2. Psychological State
3.6.3. Bowel Habits
3.7. Compliance and Safety
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GBD 2021 Nervous System Disorders Collaborators. Global, regional, and national burden of disorders affecting the nervous system, 1990–2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Neurol. 2024, 23, 344–381. [CrossRef]
- Cabinet Office; Government of Japan. Annual Report on the Aging Society: 2023. Available online: https://www8.cao.go.jp/kourei/whitepaper/w-2023/zenbun/05pdf_index.html (accessed on 16 December 2025).
- Livingston, G.; Sommerlad, A.; Orgeta, V.; Costafreda, S. G.; Huntley, J.; Ames, D.; Ballard, C.; Banerjee, S.; Burns, A.; Cohen-Mansfield, J.; et al. Dementia prevention, intervention, and care. Lancet 2017, 390, 2673–2734. [Google Scholar] [CrossRef]
- Tao, W.; Zhang, Y.; Wang, B.; Nie, S.; Fang, L.; Xiao, J.; Wu, Y. Advances in molecular mechanisms and therapeutic strategies for central nervous system diseases based on gut microbiota imbalance. J. Adv. Res. 2025, 69, 261–278. [Google Scholar] [CrossRef]
- Loh, J. S.; Mak, W. Q.; Tan, L. K. S.; Ng, C. X.; Chan, H. H.; Yeow, S. H.; Foo, J. B.; Ong, Y. S.; How, C. W.; Khaw, K. Y. Microbiota–gut–brain axis and its therapeutic applications in neurodegenerative diseases. Signal Transduct. Target. Ther. 2024, 9, 37. [Google Scholar] [CrossRef] [PubMed]
- Nakhal, M. M.; Yassin, L. K.; Alyaqoubi, R.; Saeed, S.; Alderei, A.; Alhammadi, A.; Alshehhi, M.; Almehairbi, A.; Al Houqani, S.; BaniYas, S.; et al. The microbiota–gut–brain axis and neurological disorders: A comprehensive review. Life 2024, 14, 1234. [Google Scholar] [CrossRef]
- Verhaar, B. J. H.; Hendriksen, H. M. A.; de Leeuw, F. A.; Doorduijn, A. S.; van Leeuwenstijn, M.; Teunissen, C. E.; Barkhof, F.; Scheltens, P.; Kraaij, R.; van Duijn, C. M.; et al. Gut microbiota composition is related to AD pathology. Front. Immunol. 2022, 12, 794519. [Google Scholar] [CrossRef] [PubMed]
- Nishiwaki, H.; Ito, M.; Ishida, T.; Hamaguchi, T.; Maeda, T.; Kashihara, K.; Tsuboi, Y.; Ueyama, J.; Shimamura, T.; Mori, H.; et al. Meta-analysis of gut dysbiosis in Parkinson’s disease. Mov. Disord. 2020, 35, 1626–1635. [Google Scholar] [CrossRef] [PubMed]
- Fekete, M.; Lehoczki, A.; Major, D.; Fazekas-Pongor, V.; Csípő, T.; Tarantini, S.; Csizmadia, Z.; Varga, J. T. Exploring the influence of gut–brain axis modulation on cognitive health: A comprehensive review of prebiotics, probiotics, and symbiotics. Nutrients 2024, 16, 789. [Google Scholar] [CrossRef]
- Yamashiro, K.; Takabayashi, K.; Kamagata, K.; Nishimoto, Y.; Togashi, Y.; Yamauchi, Y.; Ogaki, K.; Li, Y.; Hatano, T.; Motoi, Y.; et al. Free water in gray matter linked to gut microbiota changes with decreased butyrate producers in Alzheimer’s disease and mild cognitive impairment. Neurobiol. Dis. 2024, 193, 106464. [Google Scholar] [CrossRef]
- Hatayama, K.; Ebara, A.; Okuma, K.; Tokuno, H.; Hasuko, K.; Masuyama, H.; Ashikari, I.; Shirasawa, T. Characteristics of intestinal microbiota in Japanese patients with mild cognitive impairment and a risk-estimating method for the disorder. Biomedicines 2023, 11, 1789. [Google Scholar] [CrossRef]
- Zheng, M.; Ye, H.; Yang, X.; Shen, L.; Dang, X.; Liu, X.; Gong, Y.; Wu, Q.; Wang, L.; Ge, X.; et al. Probiotic Clostridium butyricum ameliorates cognitive impairment in obesity via the microbiota–gut–brain axis. Brain Behav. Immun. 2024, 115, 565–587. [Google Scholar] [CrossRef]
- Su, Y.; Wang, D.; Liu, N.; Yang, J.; Sun, R.; Zhang, Z. Clostridium butyricum improves cognitive dysfunction in ICV-STZ-induced Alzheimer’s disease mice via suppressing TLR4 signaling pathway through the gut–brain axis. PLoS One 2023, 18, e0286086. [Google Scholar] [CrossRef]
- Cavaliere, G.; Catapano, A.; Trinchese, G.; Cimmino, F.; Penna, E.; Pizzella, A.; Cristiano, C.; Lama, A.; Crispino, M.; Mollica, M. P. Butyrate improves neuroinflammation and mitochondrial impairment in cerebral cortex and synaptic fraction in an animal model of diet-induced obesity. Antioxidants 2022, 12, 4. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.-L.; Liu, L.-Z.; Li, L.; Hu, Y.-Y.; Xian, X.-H.; Li, W.-B. Sodium butyrate improves cognitive dysfunction in high-fat diet/streptozotocin-induced type 2 diabetic mice by ameliorating hippocampal mitochondrial damage through regulating AMPK/PGC-1α pathway. Neuropharmacology 2024, 261, 110139. [Google Scholar] [CrossRef]
- Shiwaku, R.; Yoshino, S.; Tagawa, T.; Kuwahara, H.; Tanaka, A.; Kagami-Katsuyama, H.; Homma, N.; Nishihira, J. Improvement of cognitive function by combining a food containing fermented rice bran with light exercise: A randomized, placebo-controlled, double-blind, parallel-group study. Pharmacometrics 2024, 107, 103–117. [Google Scholar]
- Mori, M.; Nakano, H.; Hikishima, S.; Minamikawa, J.; Muramatsu, D.; Sakashita, Y.; Ikeda, T.; Noguchi-Shinohara, M.; Ono, K. Inhibitory effects of 3-(4-hydroxy-3-methoxyphenyl) propionic acid on amyloid β-peptide aggregation in vitro. Biomedicines 2025, 13, 1649. [Google Scholar] [CrossRef]
- Nakamichi, N.; Nakao, S.; Masuo, Y.; Koike, A.; Matsumura, N.; Nishiyama, M.; Al-Shammari, A. H.; Sekiguchi, H.; Sutoh, K.; Usumi, K.; et al. Hydrolyzed salmon milt extract enhances object recognition and location memory through an increase in hippocampal cytidine nucleoside levels in normal mice. J. Med. Food 2019, 22, 408–415. [Google Scholar] [CrossRef]
- Zhu, N.; Liu, R.; Xu, M.-H.; Li, Y. Neuroprotective actions of different exogenous nucleotides in H2O2-induced cell death in PC-12 cells. Molecules 2023, 28, 1226. [Google Scholar] [CrossRef]
- Shimbo, I.; Yamaguchi, T.; Odaka, T.; Nakajima, K.; Koide, A.; Koyama, H.; Saisho, H. Effect of Clostridium butyricum on fecal flora in Helicobacter pylori eradication therapy. World J. Gastroenterol. 2005, 11, 7520–7524. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, K.; Kudo, H.; Oka, K.; Hayashi, A.; Onizuka, M.; Kusakabe, S.; Hino, A.; Takahashi, M.; Takeda, K.; Mori, M.; et al. Clostridium butyricum MIYAIRI 588 contributes to the maintenance of intestinal microbiota diversity early after haematopoietic cell transplantation. Bone Marrow Transplant. 2024, 59, 795–802. [Google Scholar] [CrossRef]
- Oginome, N.; Yokokawa, T.; Najima, M.; Miyata, A.; Shirado, N. Study on improvement of intestinal environment by intake of foods containing Clostridium butyricum and 3-(4-hydroxy-3-methoxyphenyl)propionic acid: A randomized, double-blind, placebo-controlled parallel-group trial. Med Cons New-Remed. 2022, 59, 17–32. [Google Scholar]
- Yokokawa, T.; Najima, M.; Miyata, A.; Shirado, N. Evaluation of the effect on intestinal environment by intake of foods containing Clostridium butyricum and 3-(4-hydroxy-3-methoxyphenyl)propionic acid in subjects with mild constipation: Reanalysis results. Med Cons New-Remed. 2023, 60, 95–103. [Google Scholar]
- Perraudeau, F.; McMurdie, P.; Bullard, J.; Cheng, A.; Cutcliffe, C.; Deo, A.; Eid, J.; Gines, J.; Iyer, M.; Justice, N.; et al. Improvements to postprandial glucose control in subjects with type 2 diabetes: A multicenter, double blind, randomized placebo-controlled trial of a novel probiotic formulation. BMJ Open Diabetes Res. Care 2020, 8, e001319. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, Z.; Cao, J.; Sun, H.; Niu, W. Clostridium butyricum 337279 shapes the gut microbiota to attenuate metabolic disorder in diet-induced obese mice. Front. Microbiol. 2025, 16, 1580847. [Google Scholar] [CrossRef] [PubMed]
- Shang, H.; Sun, J.; Chen, Y. Q. Clostridium butyricum CGMCC0313.1 modulates lipid profile, insulin resistance and colon homeostasis in obese mice. PLoS One 2016, 11, e0154373. [Google Scholar] [CrossRef]
- He, Z.; Xiong, H.; Cai, Y.; Chen, W.; Shi, M.; Liu, L.; Wu, K.; Deng, X.; Deng, X.; Chen, T. Clostridium butyricum ameliorates post-gastrectomy insulin resistance by regulating the mTORC1 signaling pathway through the gut-liver axis. Microbiol. Res. 2025, 297, 128154. [Google Scholar] [CrossRef]
- Yan, M.; Pan, D.; Chen, L.; Pang, J.; Shao, Y.; Cheng, Q.; Liu, Y.; Yin, Z.; Jiang, Z.; Sha, P.; et al. Role of intestinal SCFAs homeostasis in the hepatoprotective effect of Clostridium butyricum in T2DM. NPJ Biofilms Microbiomes 2025, 11, 206. [Google Scholar] [CrossRef]
- Yoshino, S.; Kawakami, H.; Shimizu, T.; Ono, T. Food containing rice bran fermented product improve glucose metabolism—A randomized, double-blind, placebo-controlled study. Jpn. Pharmacol. Ther. 2022, 50, 791–799. [Google Scholar]
- Yoshino, S.; Kayaki, H.; Awa, R.; Nisitani, Y.; Shimizu, T.; Ono, T.; Kuwabara, H. Reduction of LDL-cholesterol by the food containing rice bran fermented product—A randomized, double-blind, placebo-controlled study. Jpn. Pharmacol. Ther. 2023, 51, 831–840. [Google Scholar]
- Yoshino, S.; Kayaki, H.; Awa, R.; Nisitani, Y.; Shimizu, T.; Ono, T.; Kuwabara, H. Effect of food containing rice bran fermented product on visceral abdominal fat area—A randomized, double-blind, placebo-controlled parallel-group study. Jpn. Pharmacol. Ther. 2022, 50, 1031–1040. [Google Scholar]
- Ohue-Kitano, R.; Masujima, Y.; Nishikawa, S.; Iwasa, M.; Nishitani, Y.; Kawakami, H.; Kuwahara, H.; Kimura, I. 3-(4-hydroxy-3-methoxyphenyl) propionic acid contributes to improved hepatic lipid metabolism via GPR41. Sci. Rep. 2023, 13, 21246. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Konishi, T.; Nishimura, M.; Nishihira, J. Evaluation of the efficacy and safety of chum salmon milt deoxyribonucleic acid for improvement of hepatic functions: A placebo-controlled, randomised, double-blind, and parallel-group, pilot clinical trial. Food Funct. 2022, 13, 9372–9382. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y. Effects of a Food Containing Clostridium butyricum, 3-(4-Hydroxy-3-methoxyphenyl)propionic Acid, and Salmon Milt-Derived DNA on Cognitive Function: A 12-Week Two-Dose Pilot Study in Older Adults. In Research and Development Team, NICORIO Co., Ltd.; manuscript in preparation; Tokyo 154-0004, Japan, 2026. [Google Scholar]
- Gualtieri, C. T.; Johnson, L. G. Reliability and validity of a computerized neurocognitive test battery, CNS Vital Signs. Arch. Clin. Neuropsychol. 2006, 21, 623–643. [Google Scholar] [CrossRef]
- Ura, C.; Miyamae, F.; Sakuma, N.; Niikawa, H.; Inagaki, H.; Ijuin, M.; Ito, K.; Okamura, T.; Sugiyama, M.; Awata, S. Development and validation of a self-administered dementia checklist for community-dwelling older adults. Jpn. J. Geriatr. 2015, 52, 243–253. [Google Scholar] [CrossRef]
- Heuchert, J. P.; McNair, D. M. POMS 2®: Profile of Mood States, Second Edition; Multi-Health Systems Inc.: North Tonawanda, NY, USA, 2012. [Google Scholar]
- Heuchert, J. P.; McNair, D. M.; Yokoyama, K.; Watanabe, K. POMS 2 Japanese Version Manual; Kaneko Shobo: Tokyo, Japan, 2015. [Google Scholar]
- Konuma, H.; Hirose, H.; Yokoyama, K. Relationship of the Japanese translation of the Profile of Mood States Second Edition (POMS 2®) to the First Edition (POMS®). Juntendo Med. J. 2015, 61, 517–519. [Google Scholar] [CrossRef]
- Campbell, J.; Lavoie, L.; Farraia, M.; Huelin, R.; Zhang, Q.; Tahami Monfared, A. A. Quality of life in mild cognitive impairment and mild dementia associated with Alzheimer’s disease: A systematic review. Neurol. Ther. 2025, 14, 7–26. [Google Scholar] [CrossRef]
- Ogata, S.; Yamada, T.; Hashimoto, N.; Yamagata, R.; Amano, E.; Shinotojiri, J.; Yoshii, F.; Ishii, T.; Tanaka, S. Reliability, validity, and generalizability of CogHealth: evaluation in a Japanese elderly population. Ninchishinkeikagaku 2008, 10, 119–129. [Google Scholar] [CrossRef]
- Sugishita, M.; Koshizuka, Y.; Suto, S.; Sugishita, K.; Henmi, I.; Karasawa, H.; Inohara, M.; Asada, T.; Mihara, I. MMSE-J (Mini-Mental State Examination-Japan) original method: reliability and validity. Ninchishinkeikagaku 2018, 20, 91–110. [Google Scholar] [CrossRef]
- Prince, M.; Wimo, A.; Guerchet, M.; Ali, G. C.; Wu, Y. T.; Prina, M. World Alzheimer Report 2015: The Global Impact of Dementia; Alzheimer’s Disease International: London, UK, 2015. [Google Scholar]
- Scoville, W. B.; Milner, B. Loss of recent memory after bilateral hippocampal lesions. J. Neurol. Neurosurg. Psychiatry 1957, 20, 11–21. [Google Scholar] [CrossRef]
- Eichenbaum, H. Hippocampus: cognitive processes and neural representations that underlie declarative memory. Neuron 2004, 44, 109–120. [Google Scholar] [CrossRef]
- Ezzati, A.; Katz, M. J.; Zammit, A. R.; Lipton, M. L.; Zimmerman, M. E.; Sliwinski, M. J.; Lipton, R. B. Differential association of left and right hippocampal volumes with verbal episodic and spatial memory in older adults. Neuropsychologia 2016, 93, 380–385. [Google Scholar] [CrossRef]
- Aslaksen, P. M.; Bystad, M. K.; Ørbo, M. C.; Vangberg, T. R. The relation of hippocampal subfield volumes to verbal episodic memory measured by the California Verbal Learning Test II in healthy adults. Behav. Brain Res. 2018, 351, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Kochunov, P.; Coyle, T.; Lancaster, J.; Robin, D. A.; Hardies, J.; Kochunov, V.; Bartzokis, G.; Stanley, J.; Royall, D.; Schlosser, A. E.; et al. Processing speed is correlated with cerebral health markers in the frontal lobes as quantified by neuroimaging. NeuroImage 2010, 49, 1190–1199. [Google Scholar] [CrossRef]
- Moran, K. L.; Smith, C. J.; McManus, E.; Allan, S. M.; Montaldi, D.; Muhlert, N. Cerebrovascular health impacts processing speed through anterior white matter alterations: a UK biobank study. Sci. Rep. 2025, 15, 9860. [Google Scholar] [CrossRef]
- Tsai, Y. S.; Chen, C. C.; Lee, L. Y.; Lin, S. W.; Chen, Y. L.; Chen, C. C. Health-promoting effects of Clostridium butyricum GKB7 on the gastrointestinal tract in murine models. Biochem. Biophys. Rep. 2025, 43, 102145. [Google Scholar] [CrossRef]
- Ekblad, L. L.; Rinne, J. O.; Puukka, P.; Laine, H.; Ahtiluoto, S.; Sulkava, R.; Viitanen, M.; Jula, A. Insulin resistance predicts cognitive decline: An 11-year follow-up of a nationally representative adult population sample. Diabetes Care 2017, 40, 751–758. [Google Scholar] [CrossRef]
- Willette, A. A.; Xu, G.; Johnson, S. C.; Birdsill, A. C.; Jonaitis, E. M.; Sager, M. A.; Hermann, B. P.; La Rue, A.; Asthana, S.; Bendlin, B. B. Insulin resistance, brain atrophy, and cognitive performance in late middle-aged adults. Diabetes Care 2013, 36, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Shima, A.; Noguchi-Shinohara, M.; Shibata, S.; Usui, Y.; Tatewaki, Y.; Thyreau, B.; Hata, J.; Ohara, T.; Honda, T.; Taki, Y.; et al. Glucose metabolism and smaller hippocampal volume in elderly people with normal cognitive function. NPJ Aging 2024, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Samaras, K.; Makkar, S.; Crawford, J. D.; Kochan, N. A.; Wen, W.; Draper, B.; Trollor, J. N.; Brodaty, H.; Sachdev, P. S. Metformin use is associated with slowed cognitive decline and reduced incident dementia in older adults with type 2 diabetes: The Sydney Memory and Ageing Study. Diabetes Care 2020, 43, 2691–2701. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





