Submitted:
11 February 2026
Posted:
13 February 2026
You are already at the latest version
Abstract
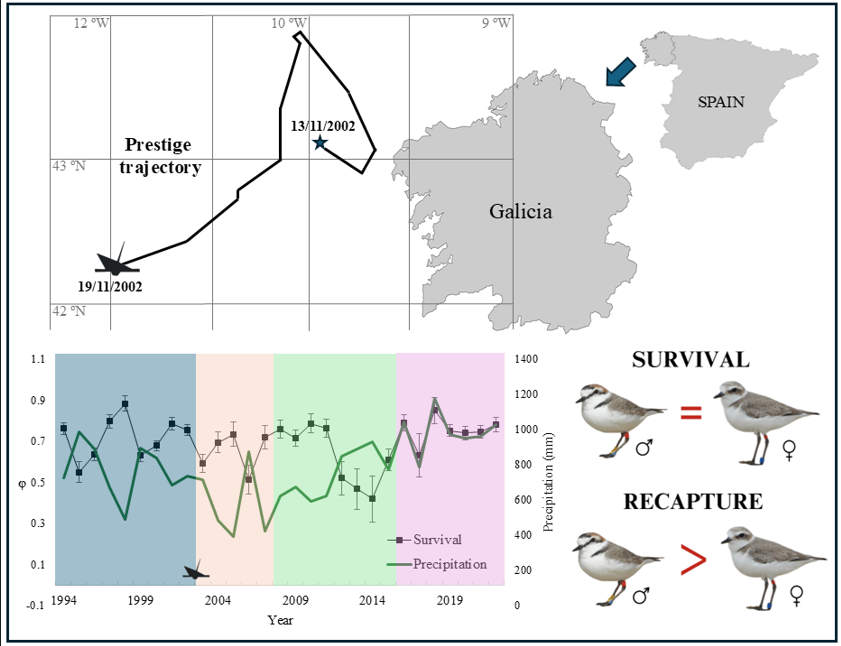
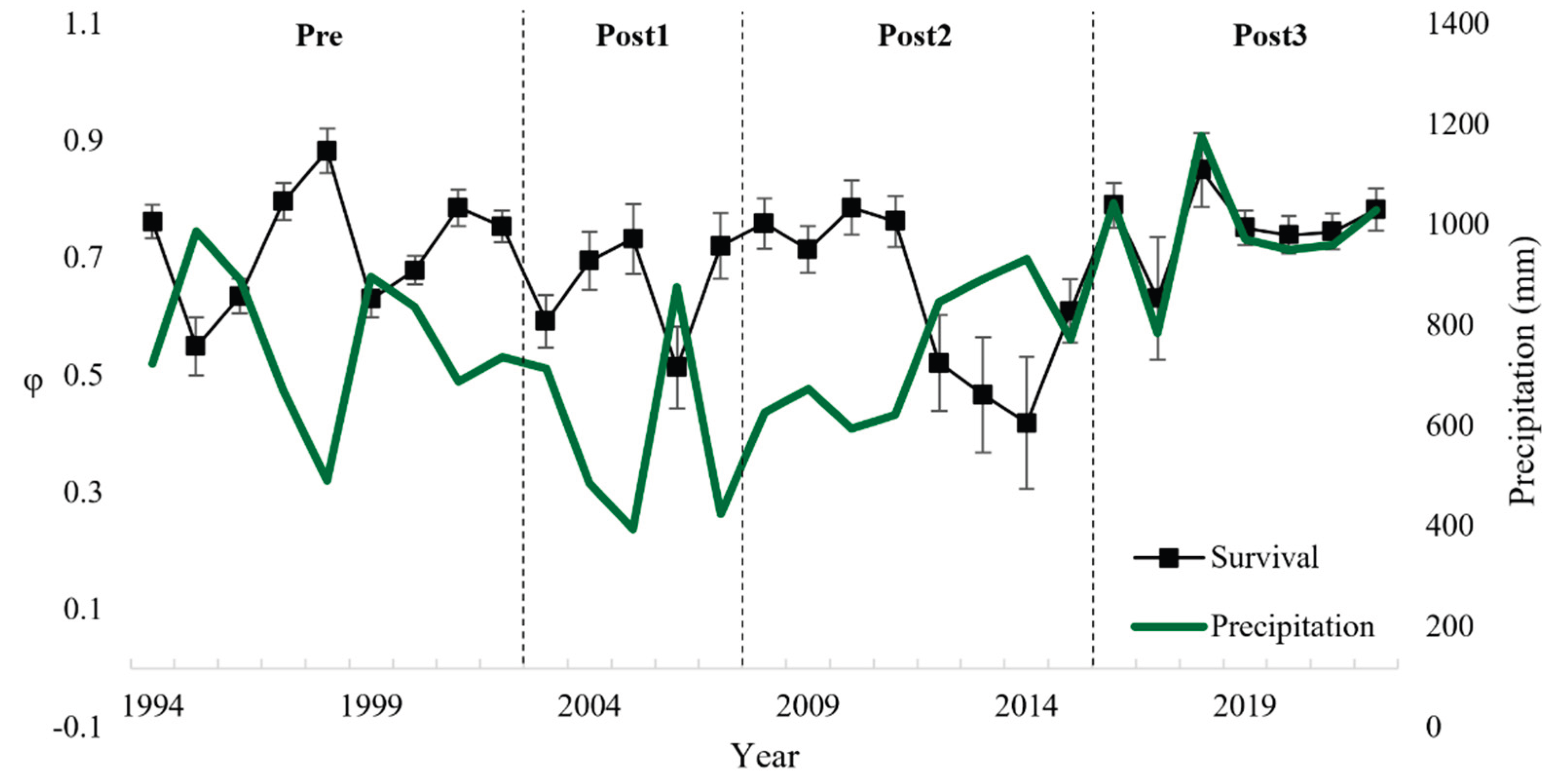
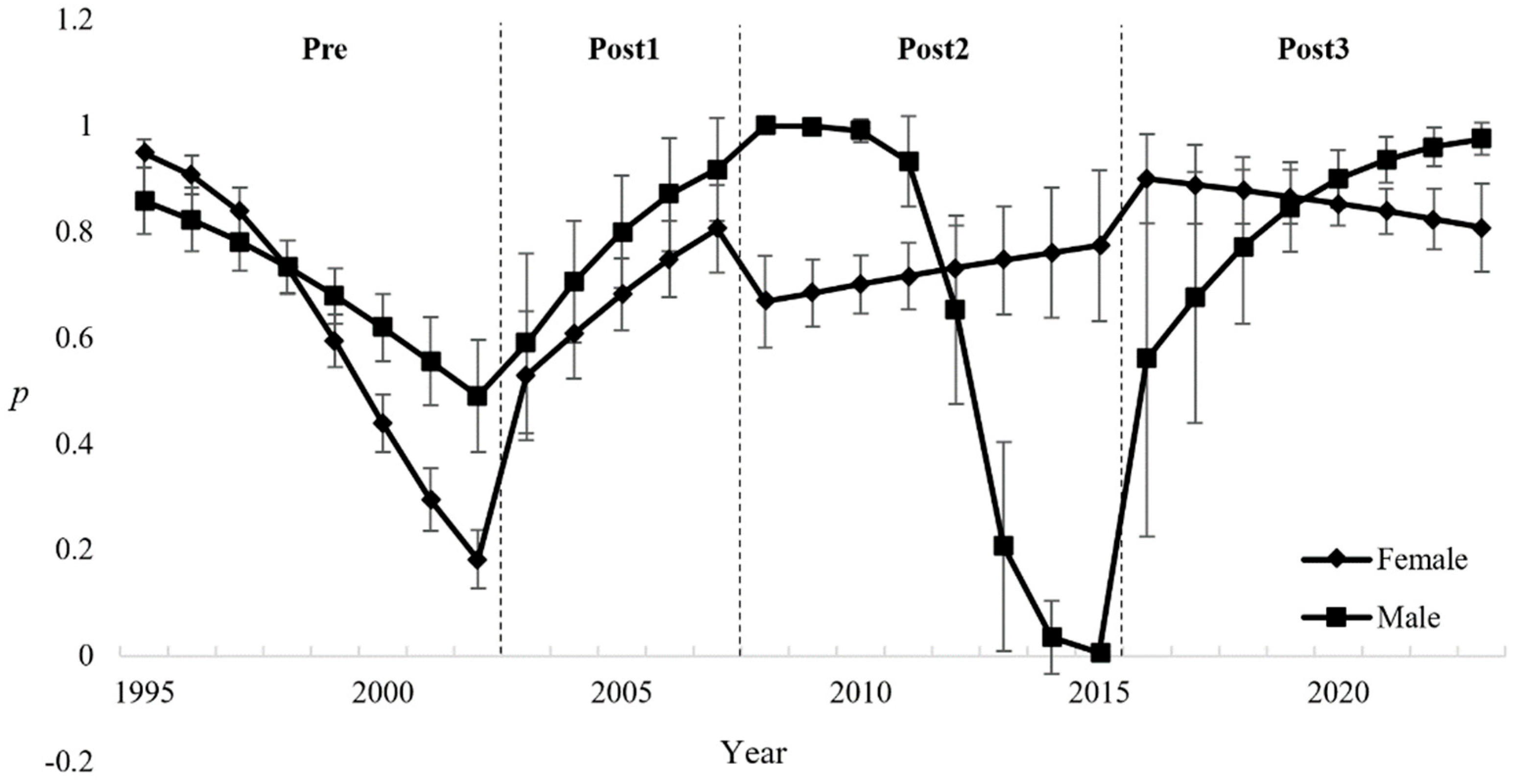
Bird survival is influenced by both natural and anthropogenic factors, including weather conditions and oil spills. In this study, we examined the impact of a major oil spill (Prestige oil tanker) and climatic conditions (precipitation and wind) on survival and recapture probability in the Kentish plover (Anarhynchus alexandrinus) population in Galicia (NW Spain). To this end, we applied the Cormack-Jolly-Seber (CJS) live recapture model to a sample of 372 adult birds captured between 1994 and 2023. The best-fit model indicated that survival was best explained by the interaction between precipitation and the Prestige oil spill, indicating a decrease in survival post-spill, especially in the periods Post1 (years 2003–2007) (βP1 = -0.70 ± 0.26) and Post2 (2008–2015) (βP2 = -0.56 ± 0.27). Precipitation showed a negative influence on adult survival (βₚₚ = -0.71 ± 0.20), but wind had no significant influence. Recapture probability was influenced by the interaction between time, sex and Prestige, with males showing higher values (βsexMale = 3.67 ± 1.47), probably due to behavioural and detectability differences. Environmental monitoring and preparedness for pollution events are therefore essential to improve the long-term viability of the species.
Keywords:
1. Introduction
2. Study Area and Methods
2.1. Study Area
2.2. Methods
Apparent Adult Survival
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brlík, V.; Pakanen, V.-M.; Jaakkonen, T.; Arppe, H.; Jokinen, J.; Lakka, J.; Blomqvist, D.; Hahn, S.; Valkama, J.; Koivula, K. Survival fluctuation is linked to precipitation variation during staging in a migratory shorebird. Sci. Rep. 2022, 12, 19830. [Google Scholar] [CrossRef]
- Loiseau, V.; Gendreau, Y.; Calosi, P.; Cusson, M. Freshwater input significantly reduces specific and functional diversity of small subarctic estuaries. Estuar. Coast. Shelf Sci. 2024, 305, 108856. [Google Scholar] [CrossRef]
- Convertino, M.; Elsner, J.B.; Muñoz-Carpena, R.; Kiker, G.A.; Martinez, C.J.; Fischer, R.A.; Linkov, I. Do Tropical Cyclones Shape Shorebird Habitat Patterns? Biogeoclimatology of Snowy Plovers in Florida. PLoS One 2011, 6, e15683. [Google Scholar] [CrossRef] [PubMed]
- Engel, N.; Sandercock, B.K.; Kosztolányi, A.; Adrião, A.; Tavares, A.; Rice, R.; Székely, T. Climatic variation influences annual survival of an island-breeding tropical shorebird. J. Avian Biol. 2024, e03191. [Google Scholar] [CrossRef]
- Martin, K.; Wilson, S.; MacDonald, E.C.; Camfield, A.F.; Martin, M.; Trefry, S.A. Effects of severe weather on reproduction for sympatric songbirds in an alpine environment: Interactions of climate extremes influence nesting success. Auk 2017, 134, 696–709. [Google Scholar] [CrossRef]
- Sexson, M.G.; Farley, G.H. Snowy plover nest survival in Kansas and effective management to counter negative effects of precipitation. J. Wildl. Manag. 2012, 76, 1587–1596. [Google Scholar] [CrossRef]
- Gardner, J.L.; Clayton, M.; Allen, R.; Stein, J.; Bonnet, T. The effects of temperature extremes on survival in two semi-arid Australian bird communities over three decades, with predictions to 2104. Glob. Ecol. Biogeogr. 2022, 31, 2498–2509. [Google Scholar] [CrossRef]
- Rockwell, S.M.; Wunderle, J.M.; Sillett, T.S.; Bocetti, C.I.; Ewert, D.N.; Currie, D.; White, J.D.; Marra, P.P. Seasonal survival estimation for a long-distance migratory bird and the influence of winter precipitation. Oecologia 2017, 183, 715–726. [Google Scholar] [CrossRef]
- Stenzel, L.E.; Hudgens, B.R.; Page, G.W.; Neuman, K.K.; Palkovic, A.L.; Erbes, J.L.; Eyster, C.R.; Ramer, B.A.; George, D.E. Climate change consequences for differential adult survival and the mating system of a temperate breeding shorebird. Ecosphere 2023, 14, e4608. [Google Scholar] [CrossRef]
- Votier, S.C.; Hatchwell, B.J.; Beckerman, A.; McCleery, R.H.; Hunter, F.M.; Pellatt, J.; Trinder, M.; Birkhead, T.R. Oil pollution and climate have wide-scale impacts on seabird demographics. Ecol. Lett. 2005, 8, 1157–1164. [Google Scholar] [CrossRef]
- Allen, M.C.; Clinchy, M.; Zanette, L.Y. Fear of predators in free-living wildlife reduces population growth over generations. Proc. Natl. Acad. Sci. 2022, 119, e2112404119. [Google Scholar] [CrossRef]
- Dinsmore, S.J.; Gaines, E.P.; Pearson, S.F.; Lauten, D.J.; Castelein, K.A. Factors affecting Snowy Plover chick survival in a managed population. Condor 2017, 119, 34–43. [Google Scholar] [CrossRef]
- Elas, M.; Witkowska, M.; Meissner, W. Factors Affecting Survival of Common Sandpiper (Actitis hypoleucos) Nests along the Semi-Natural Vistula River in Poland. Animals 2024, 14, 2055. [Google Scholar] [CrossRef]
- Kaasiku, T.; Rannap, R.; Männil, P. Predation-mediated edge effects reduce survival of wader nests at a wet grassland-forest edge. Anim. Conserv. 2022, 25, 692–703. [Google Scholar] [CrossRef]
- Nichols, C.R.; Zinnert, J.; Young, D.R. Degradation of coastal ecosystems: causes, impacts and mitigation efforts. In Tomorrow's Coasts: Complex and Impermanent; Wright, L.D., Nichols, C.R., Eds.; Coastal Research Library: Springer, Cham., 2019; Volume 27, pp. 119–136. [Google Scholar]
- Amelia, T.S.M.; Khalik, W.M.A.W.M.; Ong, M.C.; Shao, Y.T.; Pan, H.-J.; Bhubalan, K. Marine microplastics as vectors of major ocean pollutants and its hazards to the marine ecosystem and humans. Prog. Earth Planet. Sci. 2021, 8, 1–26. [Google Scholar] [CrossRef]
- Wang, L.; Nabi, G.; Yin, L.; Wang, Y.; Li, S.; Hao, Z.; Li, D. Birds and plastic pollution: recent advances. Avian Res. 2021, 12, 1–9. [Google Scholar] [CrossRef] [PubMed]
- ITOPF. Oil tanker spill statistics 2024. Available online: https://www.itopf.org/knowledge-resources/data-statistics/oil-tanker-spill-statistics-2024/?utm_source=chatgpt.com.
- King, M.D.; Elliott, J.E.; Williams, T.D. Effects of petroleum exposure on birds: A review. Sci. Total Environ. 2021, 755, 142834. [Google Scholar] [CrossRef] [PubMed]
- Munilla, I.; Arcos, J.M.; Oro, D.; Álvarez, D.; Leyenda, P.M.; Velando, A. Mass mortality of seabirds in the aftermath of the Prestige oil spill. Ecosphere 2011, 2, 1–14. [Google Scholar] [CrossRef]
- Votier, S.C.; Birkhead, T.R.; Oro, D.; Trinder, M.; Grantham, M.; Clark, J.A.; McCleery, R.H.; Hatchwell, B.J. Recruitment and survival of immature seabirds in relation to oil spills and climate variability. J. Anim. Ecol. 2008, 77, 974–983. [Google Scholar] [CrossRef]
- Bianchini, K.; Morrissey, C.A. Polycyclic aromatic hydrocarbon exposure impairs pre-migratory fuelling in captively-dosed Sanderling (Calidris alba). Ecotoxicol. Environ. Saf. 2018, 161, 383–391. [Google Scholar] [CrossRef]
- Henkel, J.R.; Sigel, B.J.; Taylor, C.M. Large-scale impacts of the Deepwater Horizon oil spill: can local disturbance affect distant ecosystems through migratory shorebirds? J. Biosci. 2012, 62, 676–685. [Google Scholar] [CrossRef]
- Maggini, I.; Kennedy, L.V.; Elliott, K.H.; Dean, K.M.; MacCurdy, R.; Macmillan, A.; Pritsos, C.A.; Guglielmo, C.G. Trouble on takeoff: crude oil on feathers reduces escape performance of shorebirds. Ecotoxicol. Environ. Saf. 2017, 141, 171–177. [Google Scholar] [CrossRef]
- Ellis, K.S.; Anteau, M.J.; Cuthbert, F.J.; Gratto-Trevor, C.L.; Jorgensen, J.G.; Newstead, D.J.; Powell, L.A.; Ring, M.M.; Sherfy, M.H.; Swift, R.J. Impacts of extreme environmental disturbances on piping plover survival are partially moderated by migratory connectivity. Biol. Conserv. 2021, 264, 109371. [Google Scholar] [CrossRef]
- Gibson, D.; Catlin, D.H.; Hunt, K.L.; Fraser, J.D.; Karpanty, S.M.; Friedrich, M.J.; Bimbi, M.K.; Cohen, J.B.; Maddock, S.B. Evaluating the impact of man-made disasters on imperiled species: Piping plovers and the Deepwater Horizon oil spill. Biol. Conserv. 2017, 212, 48–62. [Google Scholar] [CrossRef]
- Viada, C.; Moreno-Opo, R.; Carboneras, C. Impacto de la marea negra del “Prestige” sobre las aves marinas; SEO/Birdlife: Madrid, 2003. [Google Scholar]
- Albaigés, J.; Morales-Nin, B.; Vilas, F. The Prestige oil spill: a scientific response. Mar. Pollut. Bull. 2006, 53, 205–207. [Google Scholar] [CrossRef] [PubMed]
- Junoy, J.; Castellanos, C.; Viéitez, J.; De La Huz, M.; Lastra, M. The macroinfauna of the Galician sandy beaches (NW Spain) affected by the Prestige oil-spill. Mar. Pollut. Bull. 2005, 50, 526–536. [Google Scholar] [CrossRef]
- Freire, J.; Labarta, U. El" Prestige": impactos sobre los recursos y ecosistemas marinos. In La huella del fuel. Ensayos sobre el "Prestige".; Fundación Santiago Rey Fernández-Latorre, Ed.: A Coruña, 2003; pp. 104–135. [Google Scholar]
- Moreno, R.; Jover, L.; Díez, C.; Sardà, F.; Sanpera, C. Ten Years after the Prestige Oil Spill: Seabird Trophic Ecology as Indicator of Long-Term Effects on the Coastal Marine Ecosystem. PLoS One 2013, 8, e77360. [Google Scholar] [CrossRef]
- Velando, A.; Álvarez, D.; Mouriño, J.; Arcos, F.; Barros, A. Population trends and reproductive success of the European shag Phalacrocorax aristotelis on the Iberian Peninsula following the Prestige oil spill. J. Ornithol. 2005, 146, 116–120. [Google Scholar] [CrossRef]
- Bronskov, O.; Keller, V. Charadrius alexandrinus, Kentish Plover. In European Breeding Bird Atlas 2: Distribution, Abundance and Change.; Keller, V., Herrando, S., Voříšek, P., Franch, M., Kipson, M., Milanesi, P., Martí, D., Anton, M., Klvaňová, A., Kalyakin, M.V., et al., Eds.; European Bird Census Council & Lynx Edicions: Barcelona, 2020; pp. 300–301. [Google Scholar]
- Meininger, P.; Székely, T.; Scott, D.A. Kentish Plover (Charadrius alexandrinus). In An Atlas of Wader Populations in Africa and Western Eurasia; Delany, S., Scott, D., Dodman, T., Stroud, S., Eds.; Wetlands International: Wageninger, 2009; pp. 230–235. [Google Scholar]
- Delany, S.; Scott, D.A.; Dodman, T.; Stroud, D.A. An Atlas of Wader Populations in Africa and Western Eurasia.; Wetlands International: Wageningen., 2009. [Google Scholar]
- Gómez-Serrano, M.Á.; Castro, E.M.; Domínguez, J.; Pérez-Hurtado, A.; Tejera, G.; Vidal, M. Chorlitejo patinegro Charadrius alexandrinus. In Libro Rojo de las Aves de España; López-Jiménez, A.N., Ed.; SEO/Birdlife: Madrid, 2021; pp. 375–385. [Google Scholar]
- Lorenzo, J.A.; Barone, R. Atlas de las aves nidificantes en el archipiélago canario:(1997-2003); Dirección General de Conservación de la Naturaleza-SEO/BirdLife: Madrid., 2007. [Google Scholar]
- Gómez-Serrano, M.Á.; Hortas, F. Chorlitejo patinegro Charadrius alexandrinus. In III Atlas de las aves en época de reproducción en España; Molina, B., Nebreda, A., Muñoz, R.A., Seoane, J., Real, R., Bustamante, J., Del Moral, J.C., Eds.; SEO/BirdLife: Madrid, 2022. [Google Scholar]
- De Souza, J.A.; Caeiro, M.L.; Rosende, F.; Monteagudo, A.; Fafián, J.M. Estacionamientos, estructura y patrones de residencia de la población invernante del Chorlitejo patinegro (Charadrius alexandrinus) en Galicia: un análisis preliminar. Chioglossa 1999, 1, 23–45. [Google Scholar]
- Domínguez, J.; Vidal, M. Plan de Conservación del Chorlitejo patinegro (Charadrius alexandrinus) en Galicia; Consellería de Medio Ambiente e Desenvolvemento Sostible: Santiago de Compostela, 2008. [Google Scholar]
- Vidal, M.; Domínguez, J.; Dxpn. La población reproductora de Chorlitejo patinegro en Galicia (2014-2023). 2024. [Google Scholar]
- Domínguez, J.; Vidal, M. Factors affecting plumage oiling levels in a Kentish Plover Charadrius alexandrinus population after a major oil spill. Acta Ornithol. 2009, 44, 119–126. [Google Scholar] [CrossRef]
- Vidal, M.; Domínguez, J.; Luís, A. Spatial and temporal patterns of polycyclic aromatic hydrocarbons (PAHs) in eggs of a coastal bird from northwestern Iberia after a major oil spill. Sci. Total Environ. 2011, 409, 2668–2673. [Google Scholar] [CrossRef]
- Vidal, M.; Domínguez, J. Did the Prestige oil spill compromise bird reproductive performance? Evidences from long-term data on the Kentish Plover (Charadrius alexandrinus) in NW Iberian Peninsula. Biol. Conserv. 2015, 191, 178–184. [Google Scholar] [CrossRef]
- Foppen, R.P.; Majoor, F.A.; Willems, F.J.; Meininger, P.L.; Van Houwelingen, G.C.; Wolf, P.A. Survival and emigration rates in Kentish Charadrius alexandrinus and Ringed Plovers Ch. hiaticula in the Delta area, SW-Netherlands. Ardea 2006, 94, 159–173. [Google Scholar]
- Garcias, P.J.; Tavecchia, G. Apparent survival and long-term population growth rate of The Kentish Plover Charadrius alexandrinus in Mallorca, Archipelago, Spain. Ardeola 2018, 65, 59–68. [Google Scholar] [CrossRef]
- Stenzel, L.E.; Page, G.W.; Warriner, J.C.; Warriner, J.S.; George, D.E.; Eyster, C.R.; Ramer, B.A.; Neuman, K.K. Survival and natal dispersal of juvenile Snowy Plovers (Charadrius alexandrinus) in central coastal California. Auk 2007, 124, 1023–1036. [Google Scholar] [CrossRef]
- Stenzel, L.E.; Page, G.W.; Warriner, J.C.; Warriner, J.S.; Neuman, K.K.; George, D.E.; Eyster, C.R.; Bidstrup, F.C. Male-skewed adult sex ratio, survival, mating opportunity and annual productivity in the Snowy Plover Charadrius alexandrinus. Ibis 2011, 153, 312–322. [Google Scholar] [CrossRef]
- Tejera, G.; Amat, J.A.; Rodríguez, B.; Arizaga, J. Apparent survival, reproduction, and population growth estimation of a Kentish plover population in the Canary Islands. Eur. J. Wildl. Res. 2022, 68, 52. [Google Scholar] [CrossRef]
- Vidal, M.; Domínguez, J. Long-term population trends of breeding Kentish Plovers (Charadrius alexandrinus) in Northwestern Spain under the effects of a major oil spill. Bird Conserv. Int. 2013, 23, 386–397. [Google Scholar] [CrossRef]
- Székely, T.; Argüelles-Ticó, A.; Kosztolányi, A.; Küpper, C. Practical guide for investigating breeding ecology of Kentish plover Charadrius alexandrinus; Department of Biology & Biochemistry. University of Bath. UK., 2011. [Google Scholar]
- Cramp, S.; Simmons, K.E.L. (Eds.) BWP: Birds of the Western Palearctic App.; Nature Guides Ltd., 2020. [Google Scholar]
- Cormack, R.M. Estimates of survival from the sighting of marked animals. Biometrika 1964, 51, 429–438. [Google Scholar] [CrossRef]
- Jolly, G.M. Explicit estimates from capture-recapture data with both death and immigration-stochastic model. Biometrika 1965, 52, 225–247. [Google Scholar] [CrossRef]
- Seber, G.A. A note on the multiple-recapture census. Biometrika 1965, 52, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Laake, J.L. RMark: an R interface for analysis of capture-recapture data with MARK; AFSC Processed Rep. 2013-01; Alaska Fish. Sci. Cent., NOAA, Natl. Mar. Fish. Serv.: 7600 Sand Point Way NE, Seattle, 2013. [Google Scholar]
- White, G.C.; Burnham, K.P. Program MARK: survival estimation from populations of marked animals. Bird Stud. 1999, 46, 120–139. [Google Scholar] [CrossRef]
- Sandercock, B.K.; Székely, T.; Kosztolanyi, A. The effects of age and sex on the apparent survival of Kentish Plovers breeding in southern Turkey. Condor 2005, 107, 583–596. [Google Scholar] [CrossRef]
- Mullin, S.M.; Colwell, M.A.; MCallister, S.E.; Dinsmore, S.J. Apparent Survival and Population Growth of Snowy Plovers in Coastal Northern California. J. Wildl. Manag. 2010, 74, 1792–1798. [Google Scholar] [CrossRef]
- Sandercock, B.K. Estimation of survival rates for waders populations: a review of mark-recapture methods. Wader Study Group Bull. 2003, 100, 163–174. [Google Scholar]
- Domínguez, J.; Vidal, M. Proxecto Píllara. Potenciación e Estudo da Poboación reproductora da Píllara das dunas (Charadrius alexandrinus) nas praias de Galicia tras a catástrofe do Prestige.; Fundación Arao: Santiago de Compostela, 2007. [Google Scholar]
- Alonso-Álvarez, C.; Munilla, I.; López-Alonso, M.; Velando, A. Sublethal toxicity of the Prestige oil spill on yellow-legged gulls. Environ. Int. 2007, 33, 773–781. [Google Scholar] [CrossRef]
- De Souza, J.; Barros, A.; Sandoval, A.; Bao, R. A review of the status of Great Northern Diver Gavia immer in Galicia, northwest Spain. Seabird 2010, 23, 76–90. [Google Scholar] [CrossRef]
- Laffon, B.; Rábade, T.; Pásaro, E.; Méndez, J. Monitoring of the impact of Prestige oil spill on Mytilus galloprovincialis from Galician coast. Environ. Int. 2006, 32, 342–348. [Google Scholar] [CrossRef]
- Velando, A.; Munilla, I.; Leyenda, P.M. Short-term indirect effects of the ‘Prestige’oil spill on European shags: changes in availability of prey. Mar. Ecol. Prog. Ser. 2005, 302, 263–274. [Google Scholar] [CrossRef]
- Pérez-del-Olmo, A.; Raga, J.A.; Kostadinova, A. Parasite communities in a marine fish indicate ecological recovery from the impacts of the Prestige oil-spill 12–13 years after the disaster. Sci. Total Environ. 2022, 847, 157354. [Google Scholar] [CrossRef] [PubMed]
- Lebreton, J.-D.; Burnham, K.P.; Clobert, J.; Anderson, D.R. Modeling survival and testing biological hypotheses using marked animals: a unified approach with case studies. Ecol. Monogr. 1992, 62, 67–118. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model selection and multimodel inference: a practical information-theoretic approach.; Springer-Verlag.: New York., 2002. [Google Scholar]
- Burnham, K.P.; Anderson, D.R.; Huyvaert, K.P. AIC model selection and multimodel inference in behavioral ecology: some background, observations, and comparisons. Behav. Ecol. Sociobiol. 2011, 65, 23–35. [Google Scholar] [CrossRef]
- Skalski, J.; Hoffman, A.; Smith, S.G. Testing the significance of individual-and cohort-level covariates in animal survival studies. In Marked individuals in the study of bird population; Lebreton, J.D., North, P.M., Eds.; Birkhäuser Verlag: Basel, Switzerland., 1993; pp. 9–28. [Google Scholar]
- Briggs, K.T.; Gershwin, M.E.; Anderson, D.W. Consequences of petrochemical ingestion and stress on the immune system of seabirds. ICES J. Mar. Sci. 1997, 54, 718–725. [Google Scholar] [CrossRef]
- Troisi, G.; Barton, S.; Bexton, S. Impacts of oil spills on seabirds: Unsustainable impacts of non-renewable energy. Int. J. Hydrogen Energy. 2016, 41, 16549–16555. [Google Scholar] [CrossRef]
- Irons, D.B.; Kendall, S.J.; Erickson, W.P.; McDonald, L.L.; Lance, B.K. Nine years after the Exxon Valdez oil spill: effects on marine bird populations in Prince William Sound, Alaska. Condor 2000, 102, 723–737. [Google Scholar] [CrossRef]
- De la Huz, R.; Lastra, M.; Junoy, J.; Castellanos, C.; Vieitez, J. Biological impacts of oil pollution and cleaning in the intertidal zone of exposed sandy beaches: preliminary study of the “Prestige” oil spill. Estuar. Coast. Shelf Sci. 2005, 65, 19–29. [Google Scholar] [CrossRef]
- Bowman, T.D.; Schempf, P.F.; Hodges, J.I. Bald eagle population in Prince William Sound after the Exxon Valdez oil spill. J. Wildl. Manage. 1997, 61, 962–967. [Google Scholar] [CrossRef]
- Day, R.H.; Murphy, S.M.; Wiens, J.A.; Hayward, G.D.; Harner, E.J.; Lawhead, B.E. Effects of the Exxon Valdez oil spill on habitat use by birds along the Kenai Peninsula, Alaska. Condor 1997, 99, 728–742. [Google Scholar] [CrossRef]
- Day, R.H.; Murphy, S.M.; Wiens, J.A.; Hayward, G.D.; Harner, E.J.; Smith, L.N. Effects of the Exxon Valdez oil spill on habitat use by birds in Prince William Sound, Alaska. Ecol. Appl. 1997, 7, 593–613. [Google Scholar] [CrossRef]
- Murphy, S.M.; Day, R.H.; Wiens, J.A.; Parker, K.R. Effects of the Exxon Valdez oil spill on birds: comparisons of pre-and post-spill surveys in Prince William Sound, Alaska. Condor 1997, 99, 299–313. [Google Scholar] [CrossRef]
- Clark, J.A. Ringing recoveries confirm higher wader mortality in severe winters. Ring. Migr. 2004, 22, 43–50. [Google Scholar] [CrossRef]
- Mitchell, P.; Scott, I.; Evans, P. Vulnerability to severe weather and regulation of body mass of Icelandic and British Redshank Tringa totanus. J. Avian Biol. 2000, 31, 511–521. [Google Scholar] [CrossRef]
- Clark, J.A. Selective mortality of waders during severe weather. Bird Stud. 2009, 56, 96–102. [Google Scholar] [CrossRef]
- Stantial, M.L.; Cohen, J.B.; Darrah, A.J.; Farrell, S.; Maslo, B. Habitat-specific behavior, growth rate, and survival of Piping Plover chicks in New Jersey, USA. Ecosphere 2021, 12, e03782. [Google Scholar] [CrossRef]
- MeteoGalicia. Informe Climatolóxico Inverno 2013-2014; Consellería de Medioambiente, Territorio e Infraestructuras; Xunta de Galicia, 2014. [Google Scholar]
- MeteoGalicia. Informe Climatolóxico Inverno 2012-2013; Consellería de Medioambiente, Territorio e Infraestructuras; Xunta de Galicia, 2013. [Google Scholar]
- Marigómez, I.; Garmendia, L.; Soto, M.; Orbea, A.; Izagirre, U.; Cajaraville, M.P. Marine ecosystem health status assessment through integrative biomarker indices: a comparative study after the Prestige oil spill “Mussel Watch”. Ecotoxicology 2013, 22, 486–505. [Google Scholar] [CrossRef]
- Zhao, Q.; Heath-Acre, K.; Collins, D.; Conway, W.; Weegman, M.D. Integrated population modelling reveals potential drivers of demography from partially aligned data: a case study of snowy plover declines under human stressors. PeerJ 2021, 9, e12475. [Google Scholar] [CrossRef]
- Saunders, S.P.; Arnold, T.W.; Roche, E.A.; Cuthbert, F.J. Age-specific survival and recruitment of piping plovers Charadrius melodus in the Great Lakes region. J. Avian Biol. 2014, 45, 437–449. [Google Scholar] [CrossRef]
- Allen, M.R.; Ingram, W.J. Constraints on future changes in climate and the hydrologic cycle. Nature 2002, 419, 224–232. [Google Scholar] [CrossRef]
- Giorgi, F.; Raffaele, F.; Coppola, E. The response of precipitation characteristics to global warming from climate projections. Earth Syst. Dyn. 2019, 10, 73–89. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2023: Synthesis Report. Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2023; pp. 1–34. [Google Scholar]
- Pokhrel, Y.; Felfelani, F.; Satoh, Y.; Boulange, J.; Burek, P.; Gädeke, A.; Gerten, D.; Gosling, S.N.; Grillakis, M.; Gudmundsson, L. Global terrestrial water storage and drought severity under climate change. Nat. Clim. Chang. 2021, 11, 226–233. [Google Scholar] [CrossRef]
- CEDEX. Evaluación del impacto del Cambio Climático en los recursos hídricos y sequías en España; Madrid, 2017. [Google Scholar]
- Pereira, S.C.; Carvalho, D.; Rocha, A. Temperature and precipitation extremes over the iberian peninsula under climate change scenarios: A review. Climate 2021, 9, 139. [Google Scholar] [CrossRef]
- Pereira, S.C.; Marta-Almeida, M.; Carvalho, A.C.; Rocha, A. Extreme precipitation events under climate change in the Iberian Peninsula. Int. J. Climatol. 2020, 40, 1255–1278. [Google Scholar] [CrossRef]
- Donat, M.; Alexander, L.V.; Yang, H.; Durre, I.; Vose, R.; Dunn, R.J.; Willett, K.M.; Aguilar, E.; Brunet, M.; Caesar, J. Updated analyses of temperature and precipitation extreme indices since the beginning of the twentieth century: The HadEX2 dataset. J. Geophys. Res. 2013, 118, 2098–2118. [Google Scholar] [CrossRef]
- Hosseinzadehtalaei, P.; Tabari, H.; Willems, P. Climate change impact on short-duration extreme precipitation and intensity–duration–frequency curves over Europe. J. Hydrol. 2020, 590, 125249. [Google Scholar] [CrossRef]
- Jin, K.; Wu, Y.; Sun, X.; Sun, Y.; Gao, C. Spatial–temporal assessment of future extreme precipitation and extreme high-temperature exposure across China. Int. J. Climatol. 2024, 44, 2265–2282. [Google Scholar] [CrossRef]
- Zareian, M.J.; Dehban, H.; Gohari, A. Changes in temperature and precipitation extremes over Western Asia: A regional ensemble from CMIP6. Atmos. Res. 2024, 311, 107707. [Google Scholar] [CrossRef]
- Cutillas-Lozano, L.G.; López, M.S.C.; Velasco, A.P.; Andrés-Doménech, I.; Olcina-Cantos, J. Local-scale regionalisation of climate change effects on rainfall pattern: application to Alicante City (Spain). Theor. Appl. Climatol. 2023, 154, 377–402. [Google Scholar] [CrossRef]
- Viceto, C.; Marta-Almeida, M.; Rocha, A. Future climate change of stability indices for the Iberian Peninsula. Int. J. Climatol. 2017, 37, 4390–4408. [Google Scholar] [CrossRef]
- Zittis, G.; Bruggeman, A.; Lelieveld, J. Revisiting future extreme precipitation trends in the Mediterranean. Weather Clim. Extrem. 2021, 34, 100380. [Google Scholar] [CrossRef]
- AEMET. Informe sobre el estado del clima de España 2024; Madrid, 2025. [Google Scholar]
- Cruz Guerrero, R.; Lage, A. Análisis de la evolución de la temperatura y precipitación en el periodo 1973-2004 en Galicia. In Clima, sociedad y medio ambiente; Cuadrat Prats, J.M., Saz Sánchez, M.A., Serrano, Vicente, Lanjeri, S., De Luis Arrillaga, M., González-Hidalgo, J.C., Eds.; Asociación Española de Climatología: Zaragoza, 2006; pp. 1–12. [Google Scholar]
- Gómez, G.; Cabos, W.D.; Liguori, G.; Sein, D.; Lozano-Galeana, S.; Fita, L.; Fernández, J.; Magariño, M.E.; Jiménez-Guerrero, P.; Montávez, J.P. Characterization of the wind speed variability and future change in the Iberian Peninsula and the Balearic Islands. Wind Energy 2016, 19, 1223–1237. [Google Scholar] [CrossRef]
- Martins, J.; Rocha, A.; Viceto, C.; Pereira, S.C.; Santos, J.A. Future projections for wind, wind shear and helicity in the Iberian Peninsula. Atmosphere 2020, 11, 1001. [Google Scholar] [CrossRef]
- Álvarez, I.; Pereira, H.; Picado, A.; Sousa, M.; Lorenzo, M.; Dias, J. Projection of compound wind and precipitation extreme events in the Iberian Peninsula based on CMIP6. Earth Syst. Environ. 2024, 8, 801–814. [Google Scholar] [CrossRef]
- Cimiotti, D.V.; Eberhart-Hertel, L.; Audevard, A.; Garcias Salas, P.J.; Gelinaud, G.; Günther, K.; Rocha, A.; Schulz, R.; van der Winden, J.; Schmaljohann, H. Dispersal in Kentish plovers (Charadrius alexandrinus): Adult females perform furthest movements. J. Ornithol. 2024, 165, 301–314. [Google Scholar] [CrossRef]
- Küpper, C.; Edwards, S.V.; Kosztolányi, A.; Alrashidi, M.; Burke, T.; Herrmann, P.; Argüelles-Tico, A.; Amat, J.A.; Amezian, M.; Rocha, A. High gene flow on a continental scale in the polyandrous Kentish plover Charadrius alexandrinus. Mol. Ecol. 2012, 21, 5864–5879. [Google Scholar] [CrossRef]
- Stenzel, L.E.; Warriner, J.C.; Warriner, J.S.; Wilson, K.S.; Bidstrup, F.C.; Page, G.W. Long-distance breeding dispersal of Snowy Plovers in western North America. J. Anim. Ecol. 1994, 63, 887–902. [Google Scholar] [CrossRef]
- Donald, P.F. Adult sex ratios in wild bird populations. Ibis 2007, 149, 671–692. [Google Scholar] [CrossRef]
- Badzinski, D.S. Population dynamics of semipalmated plovers (Charadrius semipalmatus) breeding at Churchill, Manitoba; Trent University: Peterborough, ON, 2001. [Google Scholar]
- Paton, P.W. Survival estimates for snowy plovers breeding at Great Salt Lake, Utah. Condor 1994, 1106–1109. [Google Scholar] [CrossRef]
- Amat, J.A.; Fraga, R.M.; Arroyo, G.M. Brood desertion and polygamous breeding in the Kentish Plover Charadrius alexandrinus. Ibis 1999, 141, 596–607. [Google Scholar] [CrossRef]
- Székely, T.; Cuthill, I.C. Brood desertion in Kentish plover: the value of parental care. Behav. Ecol. 1999, 10, 191–197. [Google Scholar] [CrossRef]
- Székely, T.; Lessells, C. Mate change by Kentish plovers Charadrius alexandrinus. Ornis Scand. 1993, 317–322. [Google Scholar] [CrossRef]
- Székely, T.; Williams, T.D. Costs and benefits of brood desertion in female Kentish plovers, Charadrius alexandrinus. Behav. Ecol. Sociobiol. 1995, 37, 155–161. [Google Scholar] [CrossRef]
 location of the weather stations where climatic data were recorded.
location of the weather stations where climatic data were recorded.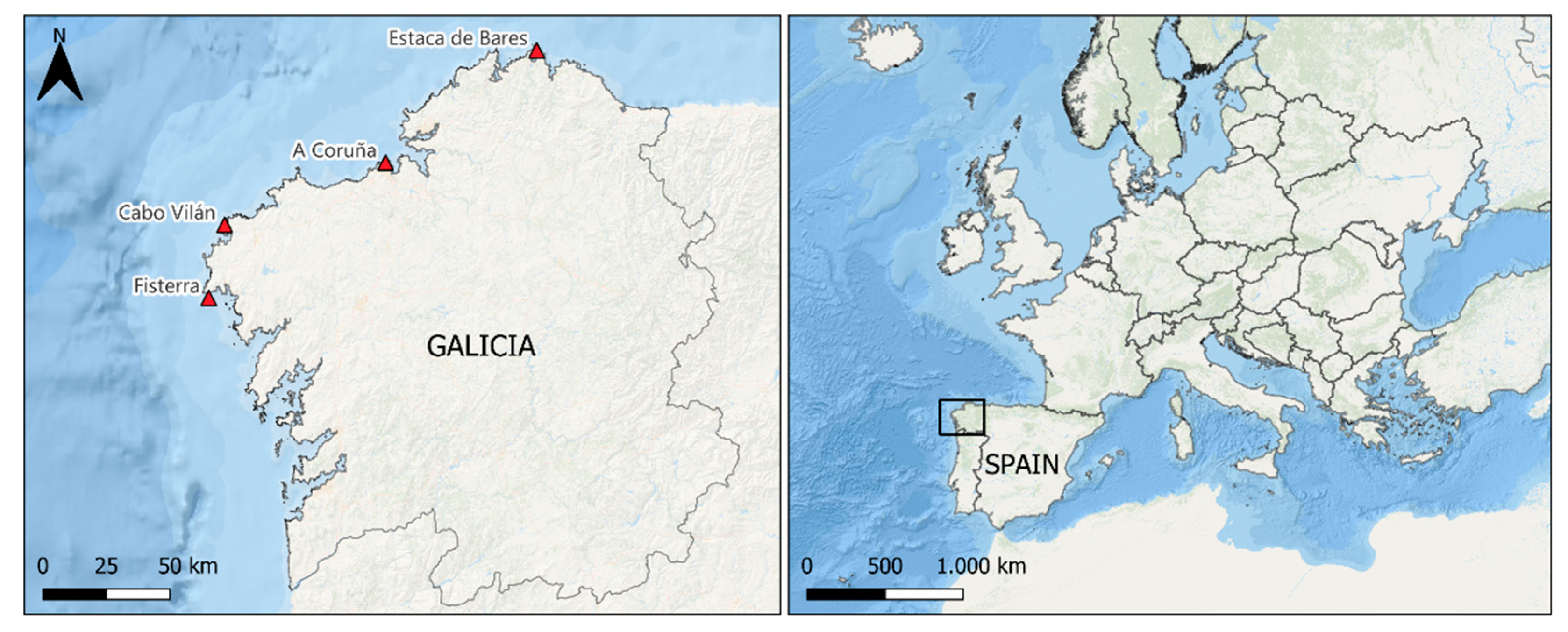
| Model1 | AICc | ΔAICc | wi | NP2 | Deviance | |
|---|---|---|---|---|---|---|
| φ(PP*P) | p(t*P*sex) | 1710.23 | 0 | 0.3424 | 24 | 649.00 |
| φ(PP+P+W) | p(t*P*sex) | 1712.53 | 2.30 | 0.1085 | 22 | 655.51 |
| φ(PP+P) | p(t*P*sex) | 1713.35 | 3.12 | 0.0720 | 21 | 658.44 |
| φ(sex+PP+P+W) | p(t*P*sex) | 1713.40 | 3.18 | 0.0700 | 23 | 654.28 |
| φ(P*PP) | p(t*P) | 1714.05 | 3.82 | 0.0507 | 16 | 669.58 |
| Variable | Estimate | SE | Lower | Upper |
|---|---|---|---|---|
| Intercept | 0.9371 | 0.1203 | 0.7012 | 1.1730 |
| Precipitation | -0.7145 | 0.2025 | -1.1115 | -0.3175 |
| Prestige Post1 | -0.7053 | 0.2589 | -1.2128 | -0.1979 |
| Prestige Post2 | -0.5606 | 0.2713 | -1.0925 | -0.0287 |
| Prestige Post3 | -0.3935 | 0.4628 | -1.3007 | 0.5136 |
| Precipitation*Prestige Post1 | 0.3292 | 0.2745 | -0.2087 | 0.8672 |
| Precipitation* Prestige Post2 | -0.2211 | 0.4121 | -1.0288 | 0.5866 |
| Precipitation* Prestige Post3 | 1.3079 | 0.4855 | 0.3562 | 2.2597 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.





