Submitted:
10 February 2026
Posted:
12 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Starting Materials
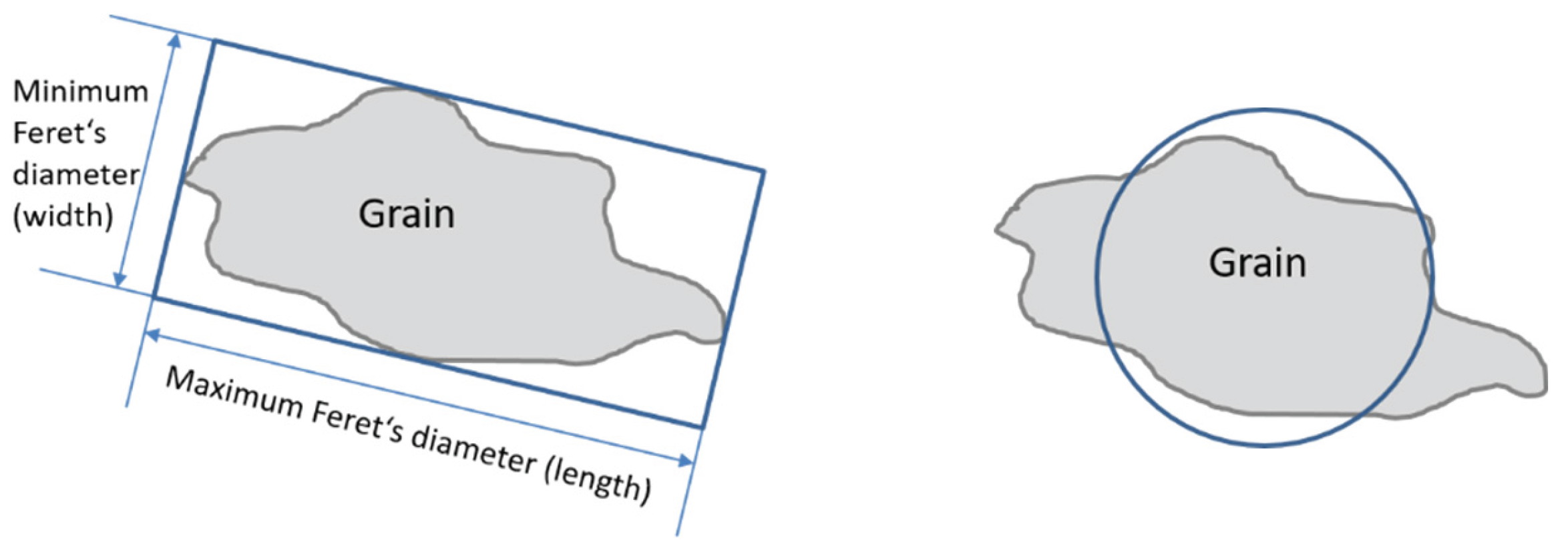
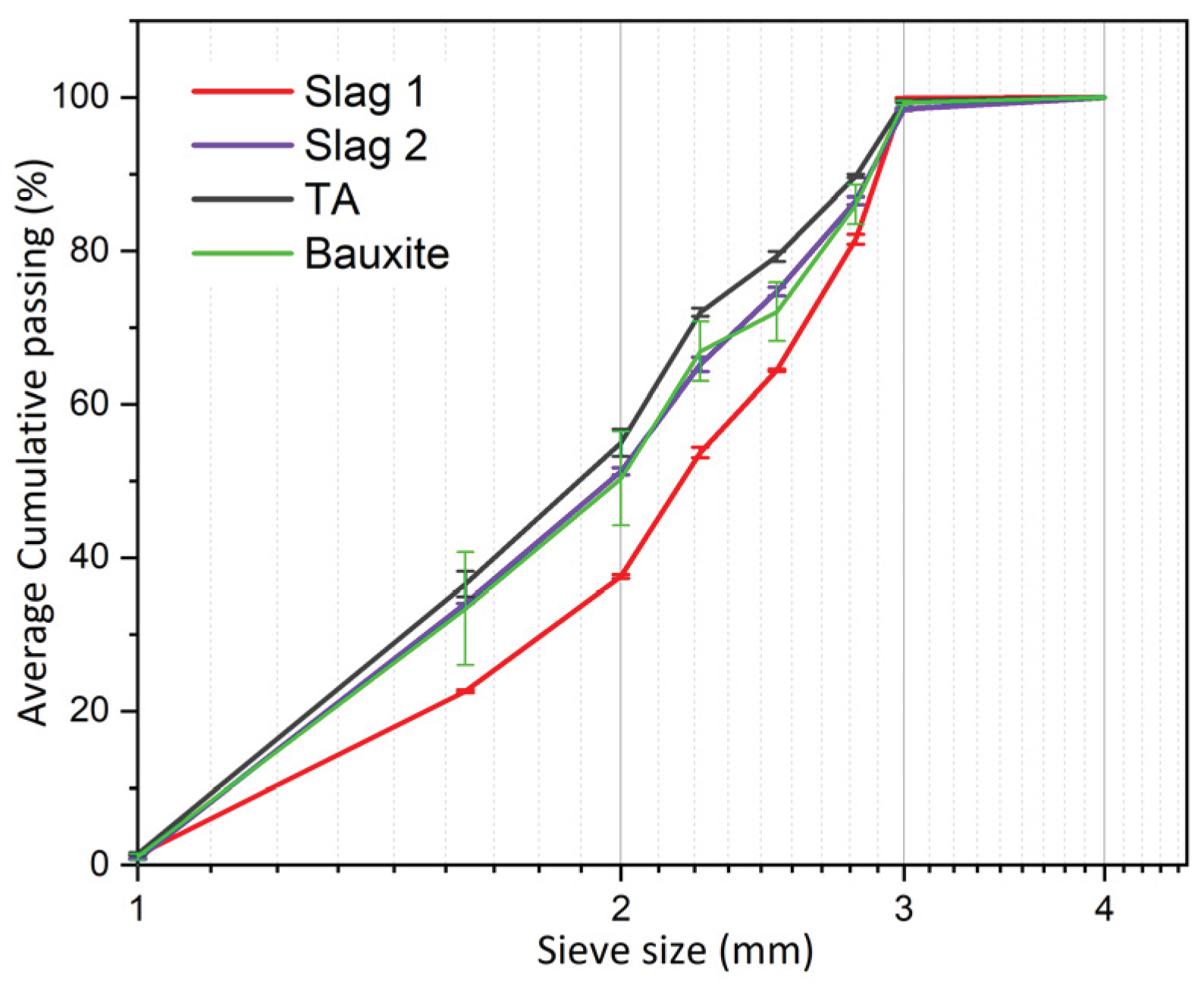
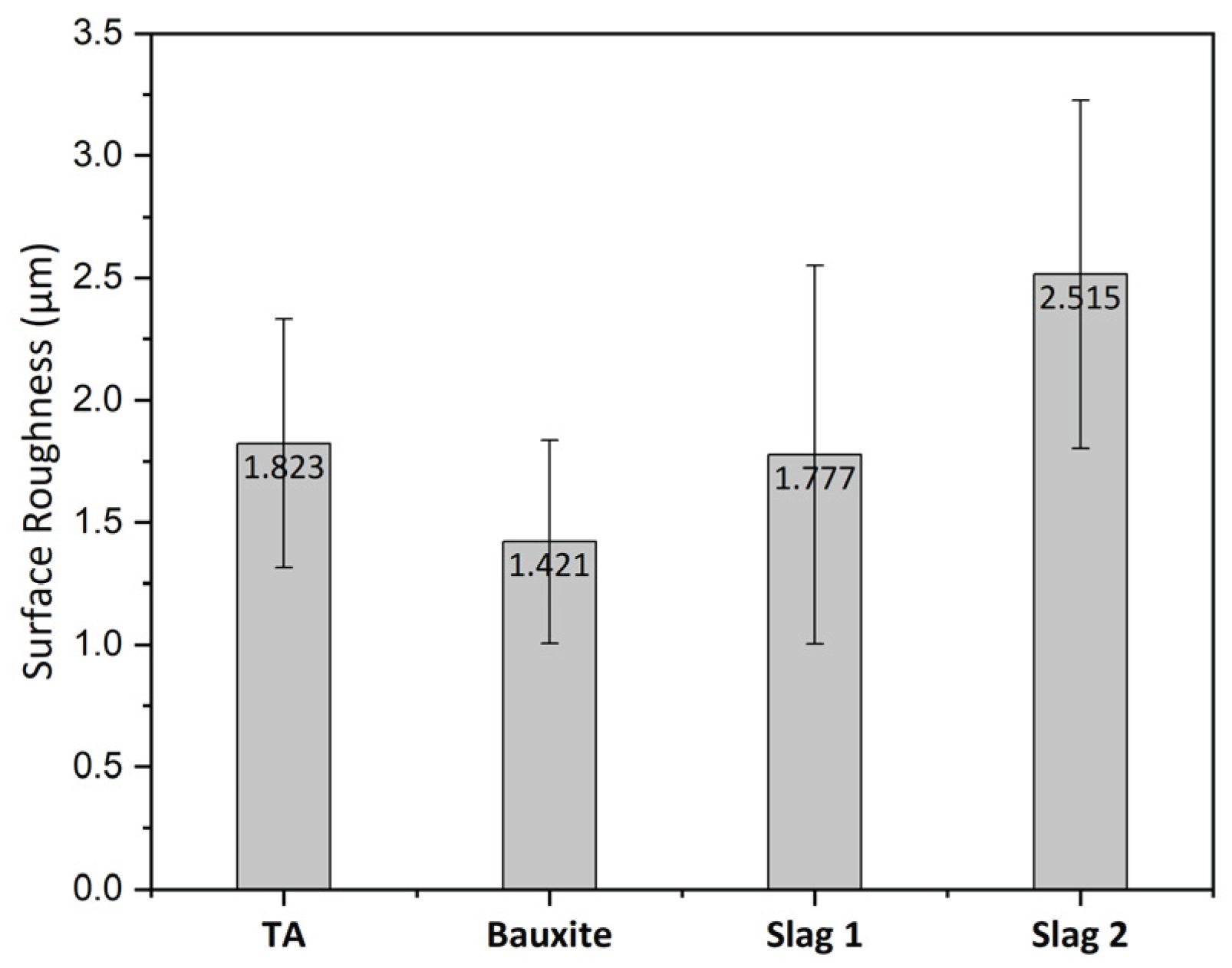
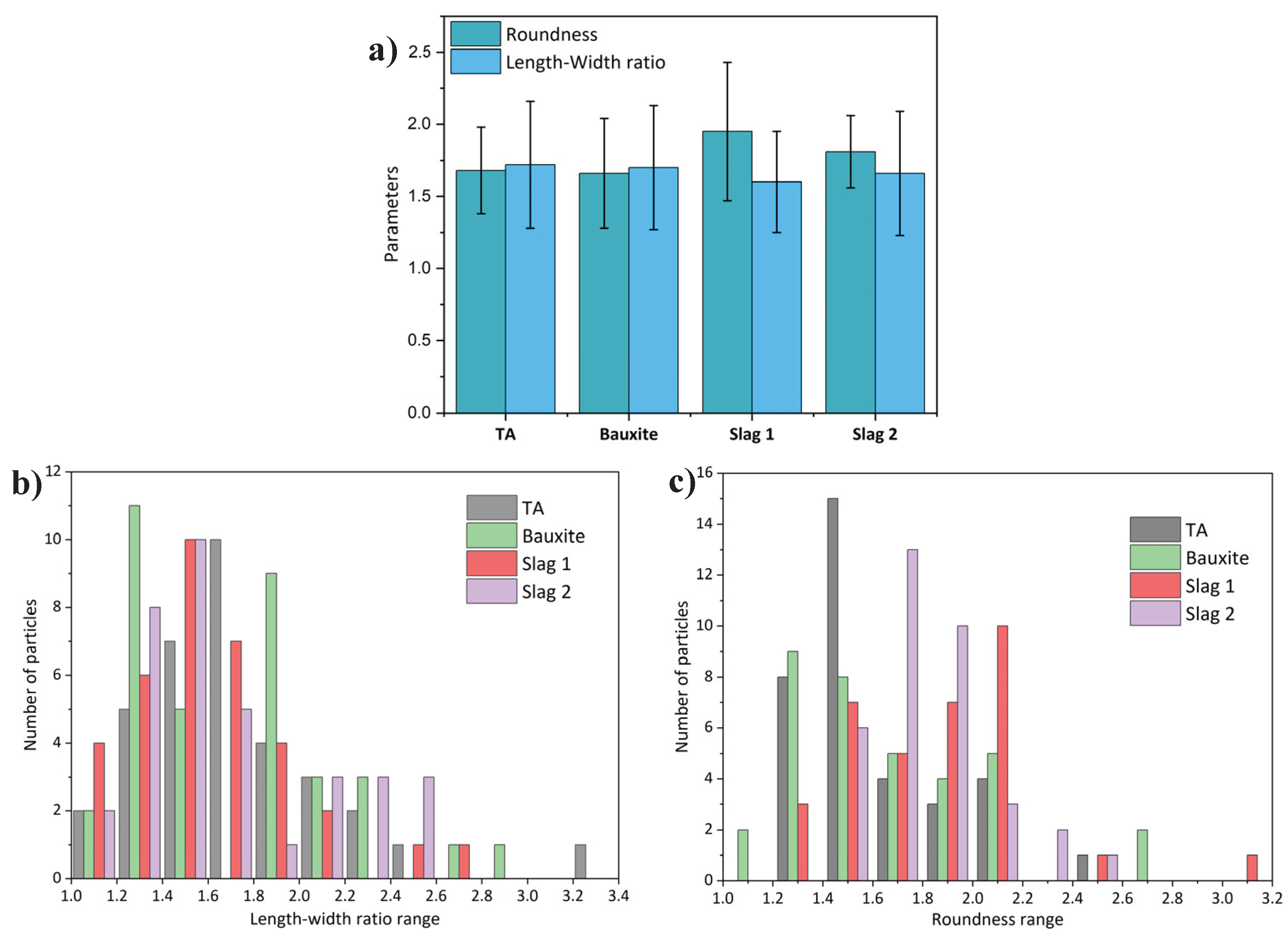
2.2. Aggregates Characterization Methods
2.3. Castables Preparation
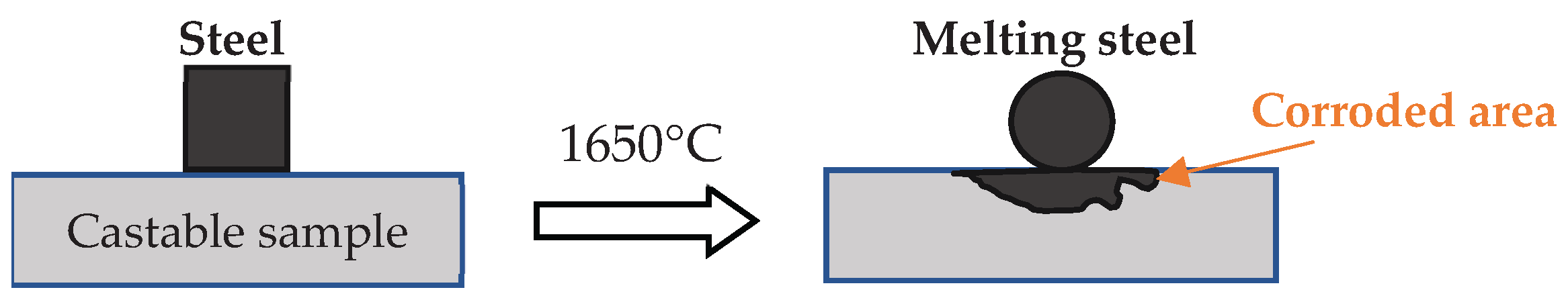
2.4. Castables Characterization
3. Results and Discussion
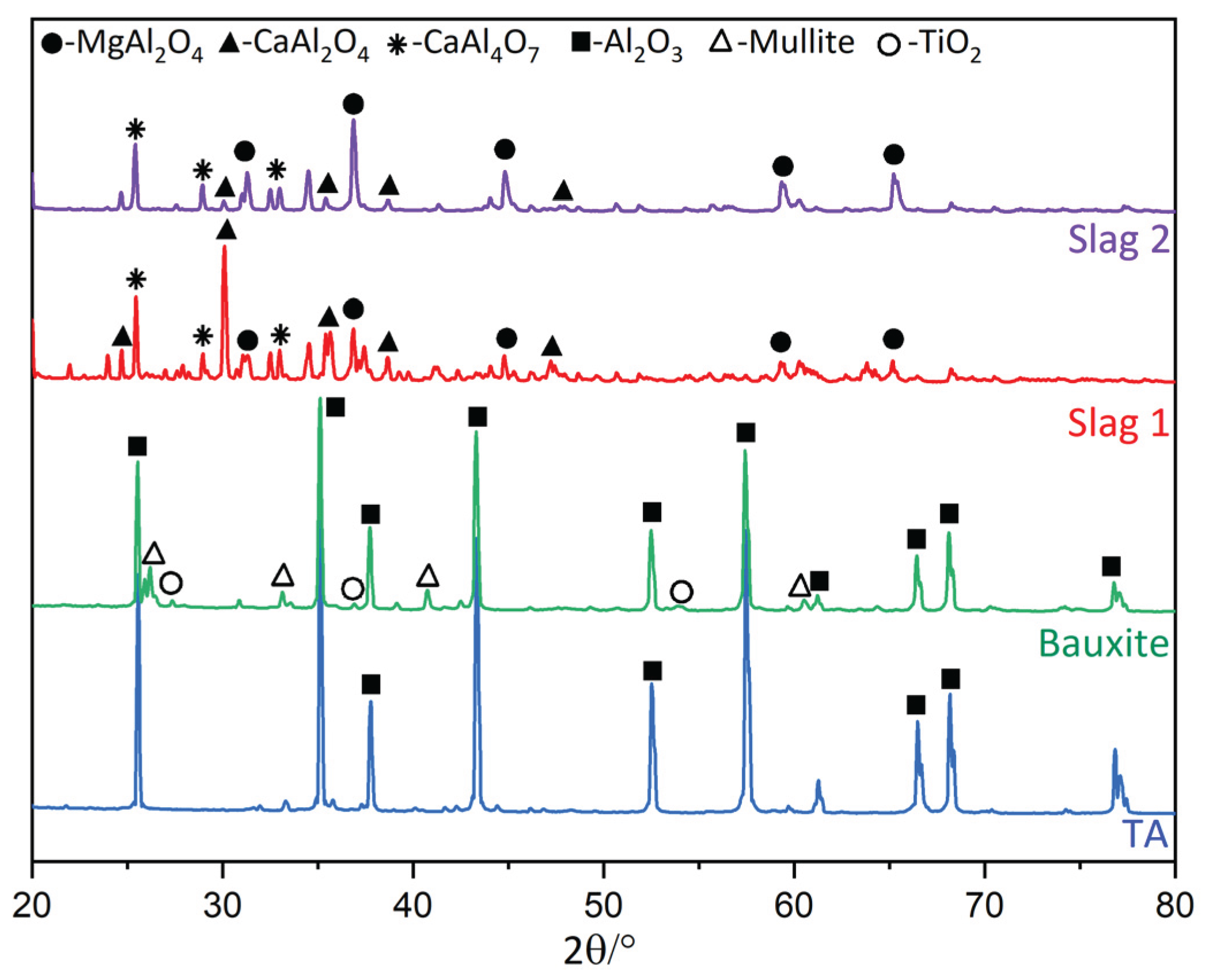
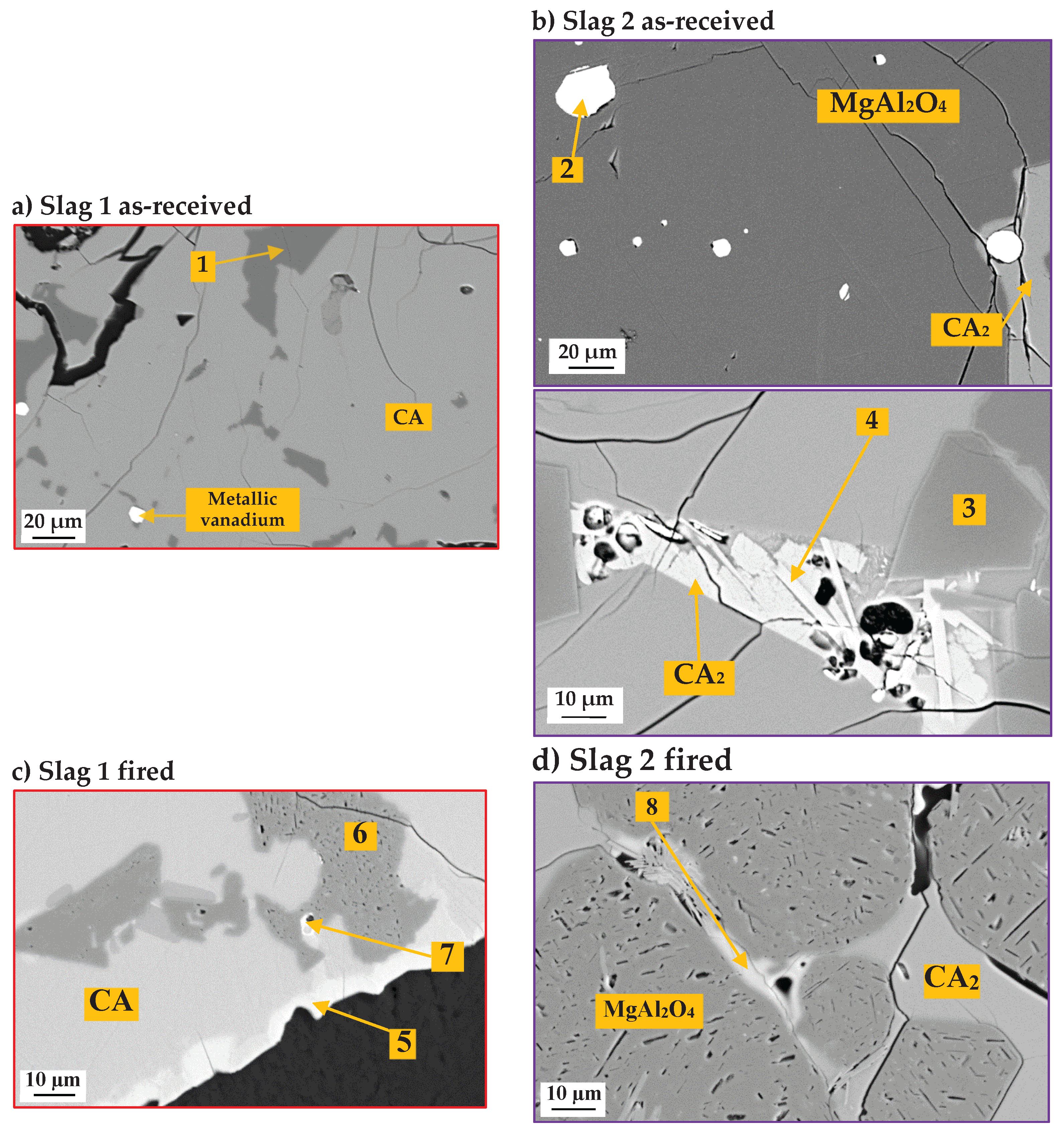
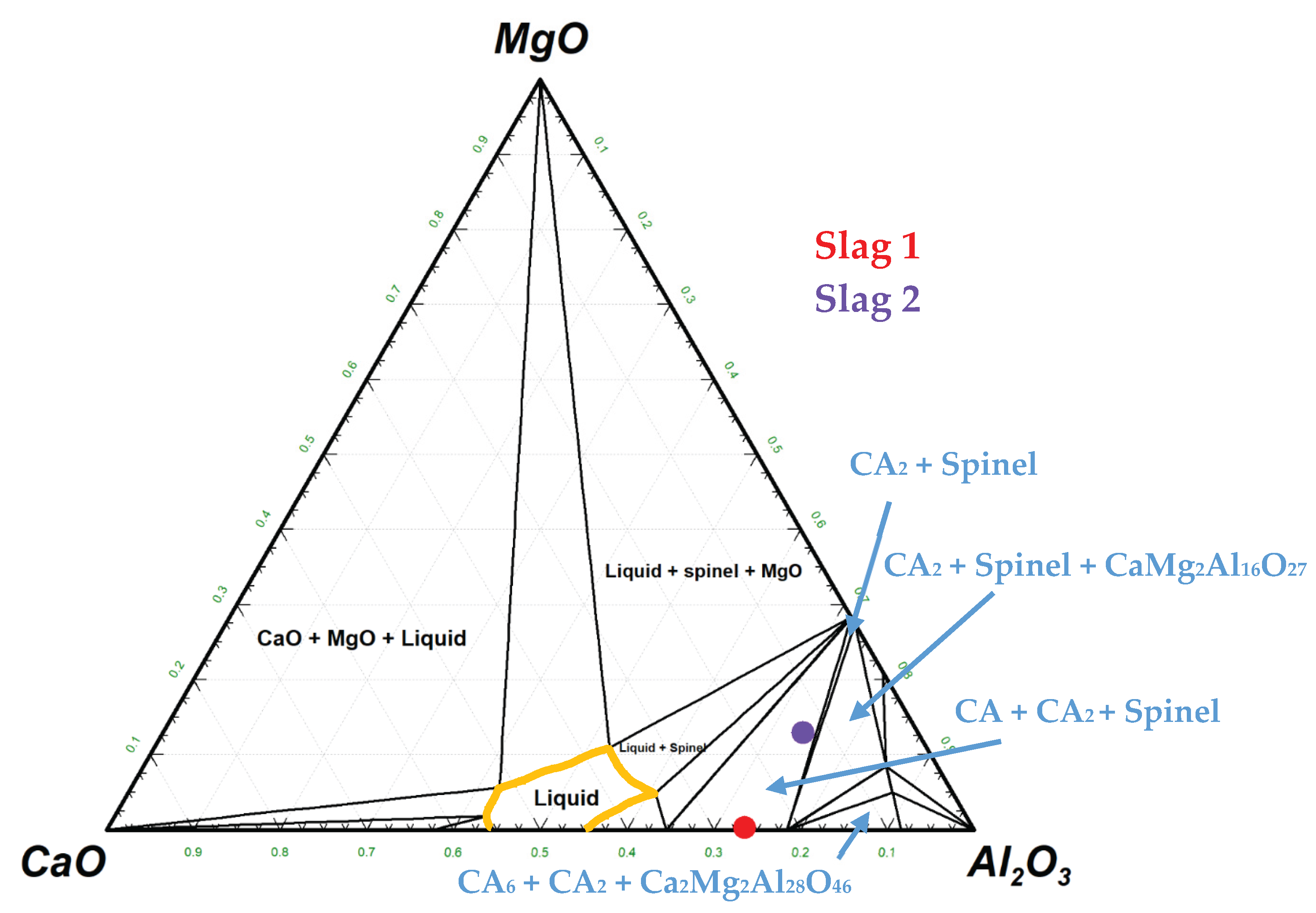
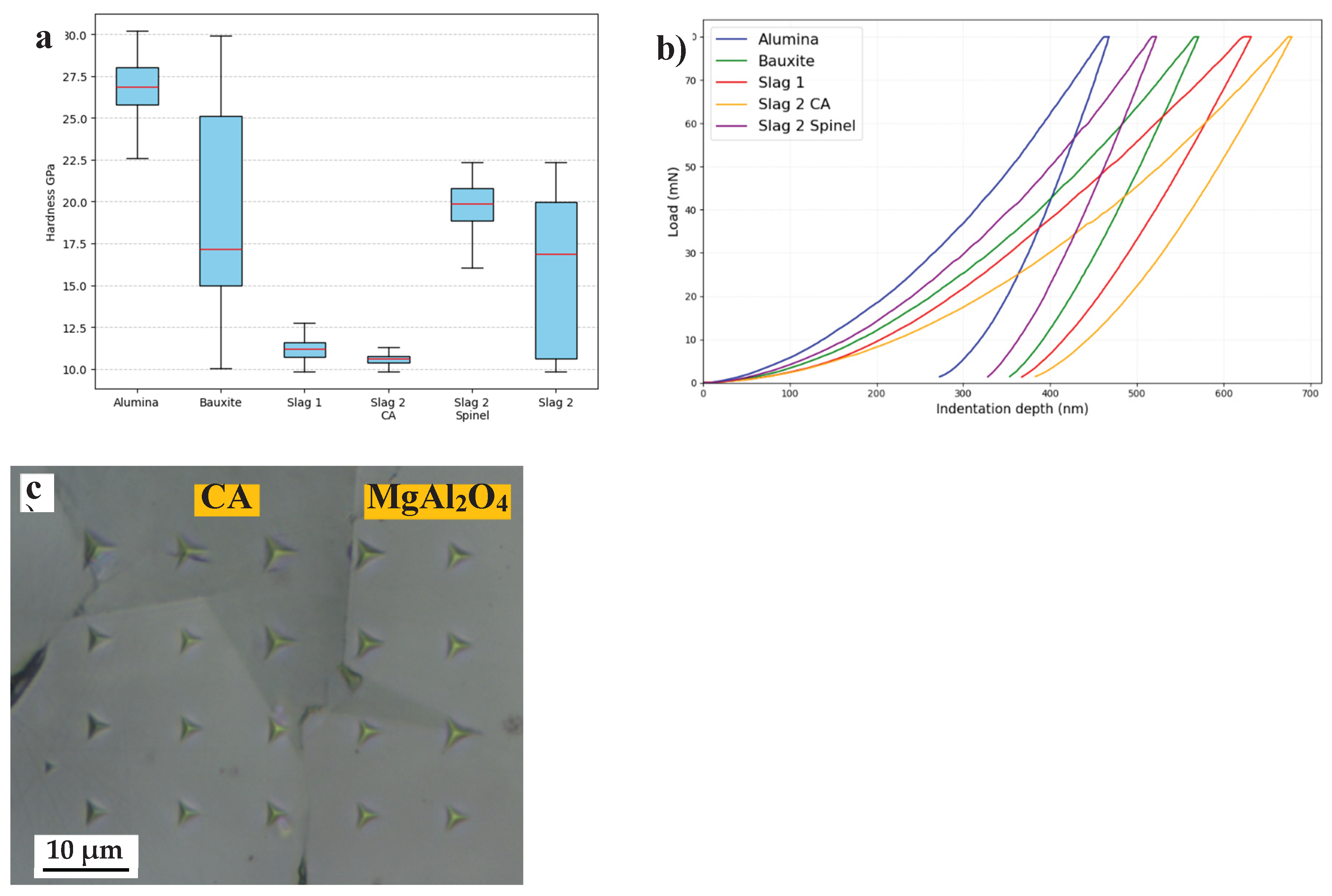
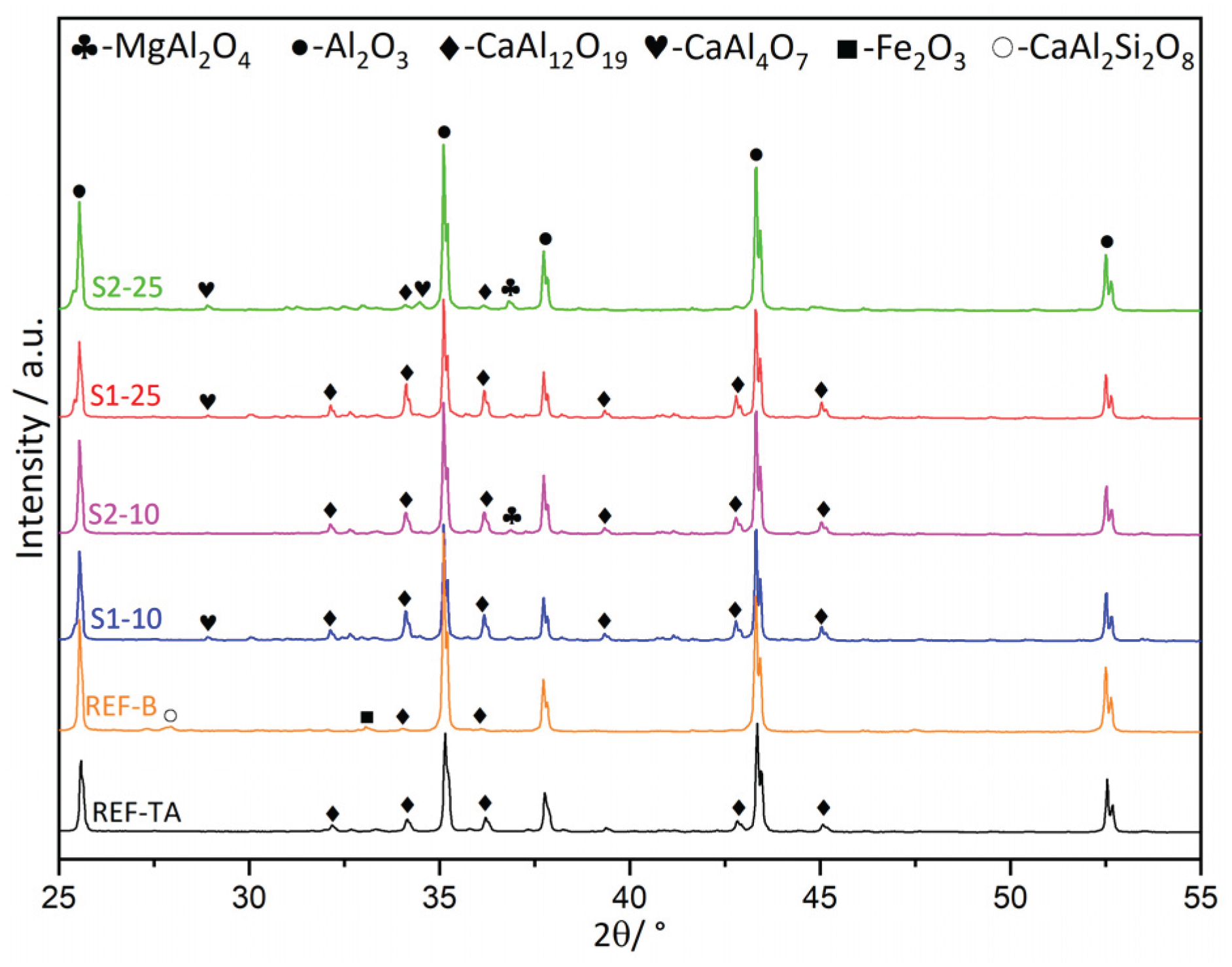
3.1. Slags Characterization
| Composition (wt.%) | ||||
| Slag 1 | Slag 2 | Bauxite | Tabular Alumina | |
| Al2O3 | 68.5 ± 0.05 | 71.2 ± 0.05 | 81.5 | 99.0 |
| CaO | 25.4 ± 0.03 | 12.8 ± 0.05 | 0.55 | - |
| MgO | 2.86 ± 0.07 | 12.4 ± 0.09 | 0.21 | - |
| SiO2 | 1.34 ± 0.08 | 1.08 ± 0.05 | 11.2 | 0.40 |
| V2O5 | 0.73 ± 0.02 | 1.13 ± 0.01 | - | - |
| Na2O | 0.24 ± 0 02 | 0.18 ± 0.01 | 0.15 | - |
| Fe2O3 | 0.23 ± 0.01 | 0.32 ± 0.02 | 2.18 | 0.60 |
| TiO2 | 0.13 ± 0.05 | 0.21 ± 0.00 | 3.78 | - |
| SO3 | 0.04 ± 0 | 0.39 ± 0 | 0.02 | - |
| K2O | 0.50 ± 0.01 | 0.22 ± 0 | 0.22 | - |
| MnO | 0.03 ± 0 | 0.07 ± 0 | 0.02 | - |
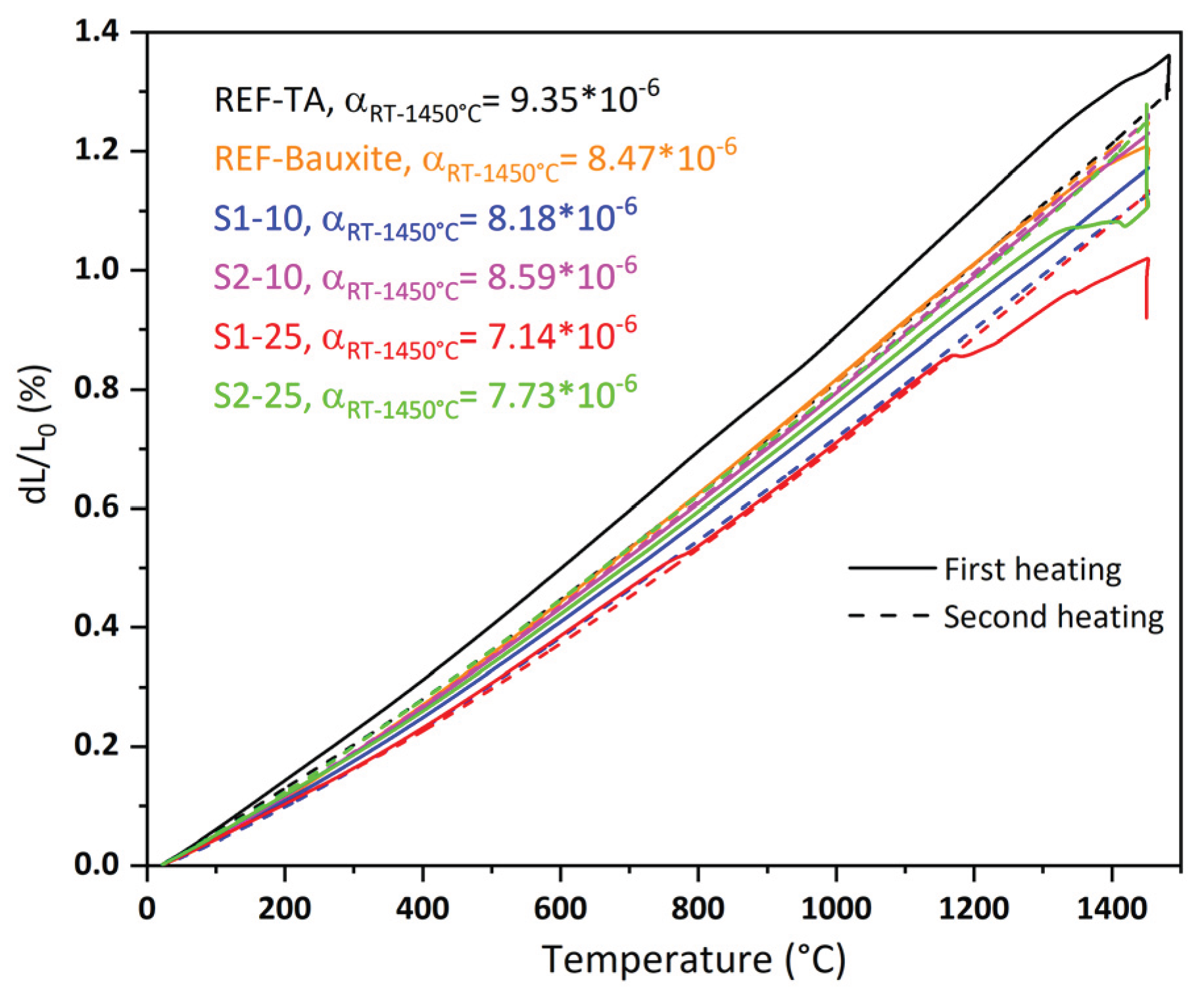
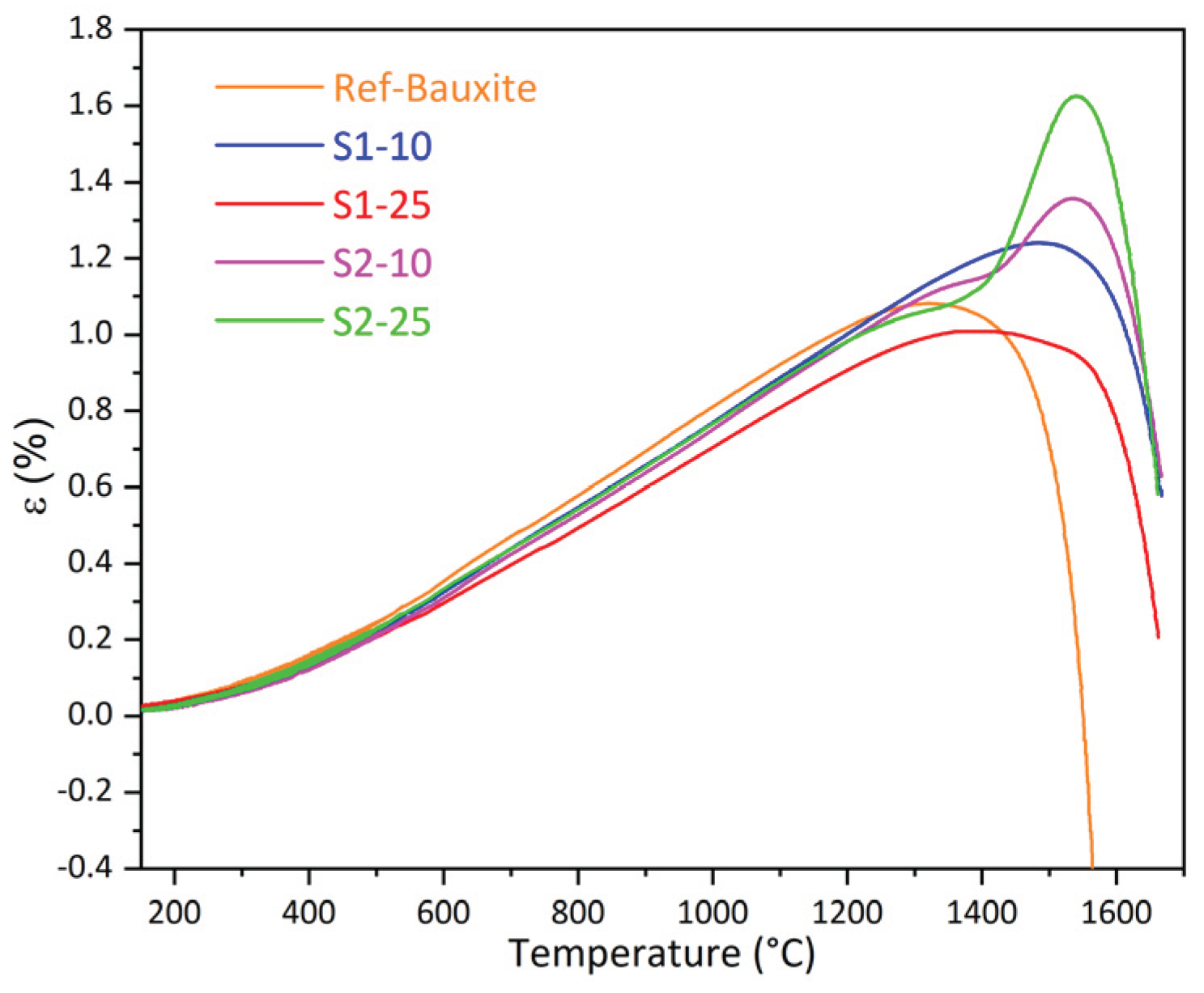
3.2. Castables Characterization
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Herrington, R. Mining Our Green Future. Nat Rev Mater 2021, 6, 456–458. [Google Scholar] [CrossRef]
- Afrouzi, H.; Bhattarai, S.; Wu, E. Relative-Price Changes as Aggregate Supply Shocks Revisited: Theory and Evidence. Journal of Monetary Economics 2024, 148, 103650. [Google Scholar] [CrossRef]
- Santos, D.A.; Dixit, M.K.; Pradeep Kumar, P.; Banerjee, S. Assessing the Role of Vanadium Technologies in Decarbonizing Hard-to-Abate Sectors and Enabling the Energy Transition. iScience 2021, 24, 103277. [Google Scholar] [CrossRef]
- OECD Global Material Resources Outlook to 2060: Economic Drivers and Environmental Consequences; OECD, 2019; ISBN 978-92-64-30744-5.
- Perkins, L.; Royal, A.C.D.; Jefferson, I.; Hills, C.D. The Use of Recycled and Secondary Aggregates to Achieve a Circular Economy within Geotechnical Engineering. Geotechnics 2021, 1, 416–438. [Google Scholar] [CrossRef]
- GRand View Research Recycled Refractories Market Size, Share & Trends Analysis Report By Product (Silica, Alumina, Magnesia), By End-Use (Iron & Steel, Cement & Lime, Glass & Ceramics, Non-Ferrous Metals), By Region, And Segment Forecasts, 2025–2030; 2025; p. 100.
- Idoine, N.E.; Raycraft, E.R.; Price, F.; Hobbs, S.F.; Deady, E.A.; Everett, P.; Shaw, R.A.; Evans, E.J.; Mills, A.J. World Mineral Production 2017-2021; British Geological Survey, 2023; ISBN ISBN 978-0-85272-797-3.
- Mohamad, N.; Muthusamy, K.; Embong, R.; Kusbiantoro, A.; Hashim, M.H. Environmental Impact of Cement Production and Solutions: A Review. Materials Today: Proceedings 2022, 48, 741–746. [Google Scholar] [CrossRef]
- Luz, A.P.; Braulio, M.A.L.; Pandolfelli, V.C. Refractory Castable Engineering; F.I.R.E. compendium series; Göller: Baden-Baden, 2015; ISBN 978-3-87264-004-8.
- Wu, M.; Jin, S. Morphology Characterization for Refractory Aggregates. Open Ceramics 2023, 15, 100408. [Google Scholar] [CrossRef]
- Issa Fares, A.; Md Abu Sohel, K.; Al-Jabri, K.; Al-Saidy, A. Influence of Ferrochrome Slag as Coarse and Fine Aggregates on Thermal and Strength Properties of Concrete at High Temperatures. Construction and Building Materials 2023, 400, 132807. [Google Scholar] [CrossRef]
- Schafföner, S.; Dietze, C.; Möhmel, S.; Fruhstorfer, J.; Aneziris, C.G. Refractories Containing Fused and Sintered Alumina Aggregates: Investigations on Processing, Particle Size Distribution and Particle Morphology. Ceramics International 2017, 43, 4252–4262. [Google Scholar] [CrossRef]
- Büchel, G.; Liu, X.; Buhr, A.; Dutton, J. Review of Tabular Alumina as High Performance Refractory Material. InterCeram: International Ceramic Review 2007, 6–12. [Google Scholar]
- He, X.; Lv, G.; Wang, S.; Li, Q.; Yun, Z.; Zhang, T. Innovative Starch-Based Alkaline Thermal Reduction of Hematite: A Fundamental Study on Mineral Phase Reconstruction and Its Potential in the Bayer Process. Minerals Engineering 2025, 231, 109452. [Google Scholar] [CrossRef]
- Wang, T.; Cui, P.; Tang, Y.; Tan, J.; Qin, M.; Cui, X. A Green Process for Treatment of Bayer Red Mud into Synthetic Soil. Journal of Environmental Chemical Engineering 2025, 13, 118281. [Google Scholar] [CrossRef]
- Matinde, E.; Msibi, S.L. Effect of Reclaimed Bauxite on Andalusite-Based Refractory Castables for Tundish Applications. J. S. Afr. Inst. Min. Metall. 2019, 119. [Google Scholar] [CrossRef]
- de Bortoli, A. Understanding the Environmental Impacts of Virgin Aggregates: Critical Literature Review and Primary Comprehensive Life Cycle Assessments. Journal of Cleaner Production 2023, 415, 137629. [Google Scholar] [CrossRef]
- Zhang, Y.; Luo, W.; Wang, J.; Wang, Y.; Xu, Y.; Xiao, J. A Review of Life Cycle Assessment of Recycled Aggregate Concrete. Construction and Building Materials 2019, 209, 115–125. [Google Scholar] [CrossRef]
- Seifert, S.; Dittrich, S.; Bach, J. Recovery of Raw Materials from Ceramic Waste Materials for the Refractory Industry. Processes 2021, 9, 228. [Google Scholar] [CrossRef]
- Global Growth Insights Aggregates Market Size, Share, Growth, and Industry Analysis, By Types (Crushed Stone, Sand, Gravel, Others), Applications (Residential, Commercial, Industrial) and Regional Insights and Forecast to 2033; Global Growth Insights, 2024; p. 91.
- Chen, Z.; Huang, L.; Yan, L.; Cai, H.; Luo, X.; Li, Y. Autoclaved Steel Slag Coarse Aggregate: A Potential Solution for Sustainable Concrete Production. Construction and Building Materials 2023, 400, 132627. [Google Scholar] [CrossRef]
- Hobson, A.J.; Stewart, D.I.; Bray, A.W.; Mortimer, R.J.G.; Mayes, W.M.; Rogerson, M.; Burke, I.T. Mechanism of Vanadium Leaching during Surface Weathering of Basic Oxygen Furnace Steel Slag Blocks: A Microfocus X-Ray Absorption Spectroscopy and Electron Microscopy Study. Environ. Sci. Technol. 2017, 51, 7823–7830. [Google Scholar] [CrossRef]
- Karhu, M.; Talling, B.; Piotrowska, P.; Matas Adams, A.; Sengottuvelan, A.; Huttunen-Saarivirta, E.; Boccaccini, A.R.; Lintunen, P. Ferrochrome Slag Feasibility as a Raw Material in Refractories: Evaluation of Thermo-Physical and High Temperature Mechanical Properties. Waste Biomass Valor 2020, 11, 7147–7157. [Google Scholar] [CrossRef]
- Astoveza, J.; Trauchessec, R.; Migot-Choux, S.; Soth, R.; Pontikes, Y. Iron-Rich Slag Addition in Ternary Binders of Portland Cement, Aluminate Cement and Calcium Sulfate. Cement and Concrete Research 2022, 153, 106689. [Google Scholar] [CrossRef]
- Branca, T.A.; Colla, V.; Algermissen, D.; Granbom, H.; Martini, U.; Morillon, A.; Pietruck, R.; Rosendahl, S. Reuse and Recycling of By-Products in the Steel Sector: Recent Achievements Paving the Way to Circular Economy and Industrial Symbiosis in Europe. Metals 2020, 10, 345. [Google Scholar] [CrossRef]
- CEMBUREAU Cement, Concrete & the Circular Economy; CEMBUREAU, 2016.
- FEhS—Institute for Building Materials Research; Cement Lime Gypsum+4 Slags Replace over 1.1 Billion t of Natural Rock 2023.
- Dong, Q.; Wang, G.; Chen, X.; Tan, J.; Gu, X. Recycling of Steel Slag Aggregate in Portland Cement Concrete: An Overview. Journal of Cleaner Production 2021, 282, 124447. [Google Scholar] [CrossRef]
- Qasrawi, H.; Shalabi, F.; Asi, I. Use of Low CaO Unprocessed Steel Slag in Concrete as Fine Aggregate. Construction and Building Materials 2009, 23, 1118–1125. [Google Scholar] [CrossRef]
- Ma, S.; Shi, K.; Xia, Y. Effect of Modified Tabular Alumina Aggregates on Mechanical Properties and Microstructure of Al2O3–Al–C Material. Ceramics International 2020, 46, 9773–9779. [Google Scholar] [CrossRef]
- Kumar, P.H.; Srivastava, A.; Kumar, V.; Majhi, M.R.; Singh, V.K. Implementation of Industrial Waste Ferrochrome Slag in Conventional and Low Cement Castables: Effect of Microsilica Addition. Journal of Asian Ceramic Societies 2014, 2, 169–175. [Google Scholar] [CrossRef]
- Liu, S.; Wang, L.; Chen, J.; Ye, L.; Du, J. Research Progress of Vanadium Extraction Processes from Vanadium Slag: A Review. Separation and Purification Technology 2024, 342, 127035. [Google Scholar] [CrossRef]
- Yildirim, I.Z.; Prezzi, M. Chemical, Mineralogical, and Morphological Properties of Steel Slag. Advances in Civil Engineering 2011, 2011, 1–13. [Google Scholar] [CrossRef]
- Wang, G.C. Slag Processing. In The Utilization of Slag in Civil Infrastructure Construction; Elsevier, 2016; pp. 87–113 ISBN 978-0-08-100994-9.
- Lee, J.; Kurniawan; Kim, E.; Chung, K.W.; Kim, R.; Jeon, H.-S. A Review on the Metallurgical Recycling of Vanadium from Slags: Towards a Sustainable Vanadium Production. Journal of Materials Research and Technology 2021, 12, 343–364. [Google Scholar] [CrossRef]
- Hu, P.; Hu, P.; Vu, T.D.; Li, M.; Wang, S.; Ke, Y.; Zeng, X.; Mai, L.; Long, Y. Vanadium Oxide: Phase Diagrams, Structures, Synthesis, and Applications. Chem. Rev. 2023, 123, 4353–4415. [Google Scholar] [CrossRef]
- Chan, C.-F.; Ko, Y.-C. Effect of CaO Content on the Hot Strength of Alumina-Spinel Castables in the Temperature Range of 1000° to 1500 °C. Journal of the American Ceramic Society 2005, 81, 2957–2960. [Google Scholar] [CrossRef]
- Cao, Z.; Wang, N.; Xie, W.; Qiao, Z.; Jung, I.-H. Critical Evaluation and Thermodynamic Assessment of the MgO-V2O5 and CaO-V2O5 Systems in Air. Calphad 2017, 56, 72–79. [Google Scholar] [CrossRef]
- Shen, W.; Yang, Z.; Cao, L.; Cao, L.; Liu, Y.; Yang, H.; Lu, Z.; Bai, J. Characterization of Manufactured Sand: Particle Shape, Surface Texture and Behavior in Concrete. Construction and Building Materials 2016, 114, 595–601. [Google Scholar] [CrossRef]
- Li, N.; Zhang, Z.; Pang, G.; Dou, H.; Si, W.; Wang, H. Three-Dimensional Roughness Prediction of Gravels Using Laser Scanning Models and Two-Dimensional Photos. Construction and Building Materials 2025, 484, 141903. [Google Scholar] [CrossRef]
- Vanadium: Extraction, Manufacturing and Applications; Yang, B., He, J., Zhang, G., Guo, J., Eds.; Elsevier: Amsterdam Kidlington Cambridge, 2021; ISBN 978-0-12-818898-9.
- Schnabel, M.; Buhr, A.; Schmidtmeier, D.; Chatterjee, S.; Dutton, J. Perceptions and Characteristics of Fused and Sintered Refractory Aggregates. 2015.









| Raw Material | Grain Size | Sample code | |||||
|---|---|---|---|---|---|---|---|
| REF-TA | REF-B | S1-25 | S2-25 | S1-10 | S2-10 | ||
| Tabular Alumina | 1-3 mm | 34.2 | 34.2 | 25.7 | 25.7 | 30.8 | 30.8 |
| 0.5-1 mm | 9.8 | ||||||
| 0.2-0.6 mm | 9.8 | ||||||
| 0-0.3 mm | 17.1 | ||||||
| 0-0.045 mm | 9.8 | ||||||
| Reactive Alumina | <0.001 mm | 12.9 | |||||
| Secar 71 | 6.4 | ||||||
| Slag 2242 | 1-3 mm | - | - | 8.6 | - | 3.4 | - |
| Slag 2256 | 1-3 mm | - | - | - | 8.6 | - | 3.4 |
| Slag 2242 | <63 µm | - | - | - | - | - | - |
| Slag 2256 | <63µm | - | - | - | - | - | - |
| Water | 5 | ||||||
| Deflocculant | FS65 | 0.1 | |||||
| Raw material | True density g.cm-3 | Porosity % | Specific surface area m2.g-1 |
|---|---|---|---|
| Slag 1 | 3.02 | 7.3 | 0.59 ± 0.03 |
| Slag 2 | 3.06 | 2.1 | 0.24 ± 0.01 |
| Tabular Alumina | 3.92 | 0.9 | 1.00 ± 0.06 |
| Bauxite | 3.81 | 8.9 | 0.74 ± 0.04 |
| Point. | Phase | O | Mg | Al | Ca | Si | S | V | Fe | Ti | Mn |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Spinel Mg(Al,V)2O4 | 55.4 | 14.6 | 29.5 | 0.13 | - | - | 0.31 | - | - | |
| 2 | Metallic V + Si, Mn, Fe | 25.5 | 71.1 | 1.8 | 1.6 | ||||||
| 3 | Spinel Mg(Al,V)2O4 | 52.5 | 13.2 | 31.9 | 2.38 | ||||||
| 4 | Goldmanite Ca3(Al,V,Ti)2(Si,Mg)3O12 | 54.6 | 2.80 | 21.0 | 7.00 | 4.30 | 6.43 | 3.64 | |||
| 5 | Krotite-type (Ca12(Al,V,Si,S)14O33) | 54.8 | - | 22.8 | 16.4 | 2.36 | 0.55 | 3.17 | - | - | |
| 6 | Spinel Mg(Al,V)2O4 | 56.2 | 14.2 | 27.9 | 0.87 | - | - | 0.57 | 0.32 | - | |
| 7 | Mayenite-type (Ca12(Al,V,Si,Ti,Fe)14O33 | 60.9 | 0.67 | 10.8 | 16.6 | 1.05 | - | 1.85 | 0.92 | 7.21 | |
| 8 | Krotite-type | 54.3 | - | 22.0 | 15.6 | 4.23 | - | 3.87 | - | - |
| Sample | Open porosity % | True density g/cm3 |
CCS (MPa) |
CMOR (MPa) |
HMOR 1400°C (MPa) |
E modulus (GPa) | G modulus (GPa) |
| REF-TA | 17.5 | 4.02 | 259.0 ± 16 | 42.5 ± 2.2 | 23.8 ± 1.6 | 133 ± 8.5 | 59.4 ± 1.8 |
| REF-Bauxite | 17.8 | 3.98 | 225.3 ± 9.2 | 34.8 ± 2.2 | 1.30 ± 0.18 | 159 ± 4.6 | 16.2 ± 0.3 |
| S1-10 | 20.9 | 3.86 | 95.38 ± 23 | 21.8 ± 2.1 | 7.49 ± 1.89 | 122 ± 8.2 | 13.0 ± 0.6 |
| S1-25 | 22.0 | 3.86 | 81.57 ± 11 | 9.25 ± 1.2 | 4.69± 0.23 | 54.7 ± 2.5 | 21.2 ± 1.7 |
| S2-10 | 20.9 | 3.93 | 204.5 ± 23 | 24.8 ± 3.5 | 12.8 ± 1.25 | 126 ± 1.6 | 13.6 ± 0.3 |
| S2-25 | 24.0 | 3.89 | 50.10 ± 3.5 | 12.1 ± 2.4 | 3.4± 1.48 | 70.0 ± 3.8 | 33.1 ± 0.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





