Submitted:
09 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Interest in microalga-based technologies has emerged in recent years as a response to environmental challenges and the global food crisis for providing alternative and sustainable food products. This study used temperature variations between 18 and 32 °C, and nitrogen-to-phosphorus (N:P) ratios between 1.9 and 42.6, to model and optimize growth and composition of Chlorella vulgaris, a nutritionally interesting species. Lower temperatures appear ideal for this strain. An increase in average biomass productivity was observed with decreasing temperature, leading to a maximum of 122.27 mgdw L-1 d-1 at 18 °C on the 4th day of cultivation. The maximum productivities for total proteins, fatty acids, carbohydrates, and pigments were, respectively, 26.9 mg L-1 d-1, 26.4 mg L-1 d-1, 16.0 mg L-1 d-1, and 2.41 mg L-1 d-1, all referring to 18 °C. The fatty acid, carotenoid, and amino acid profiles were also ascertained; several indicators suggested that cultivation of this microalga under the aforementioned optimal conditions holds potential for the food industry. The high proportion of polyunsaturated fatty acids, including two essential fatty acids; the high production of lutein; and the presence of several essential amino acids are among the favorable indicators. Overall, the information generated by this study is helpful to support future pilot studies aimed at the commercial production of microalga-derived products.
Keywords:
1. Introduction
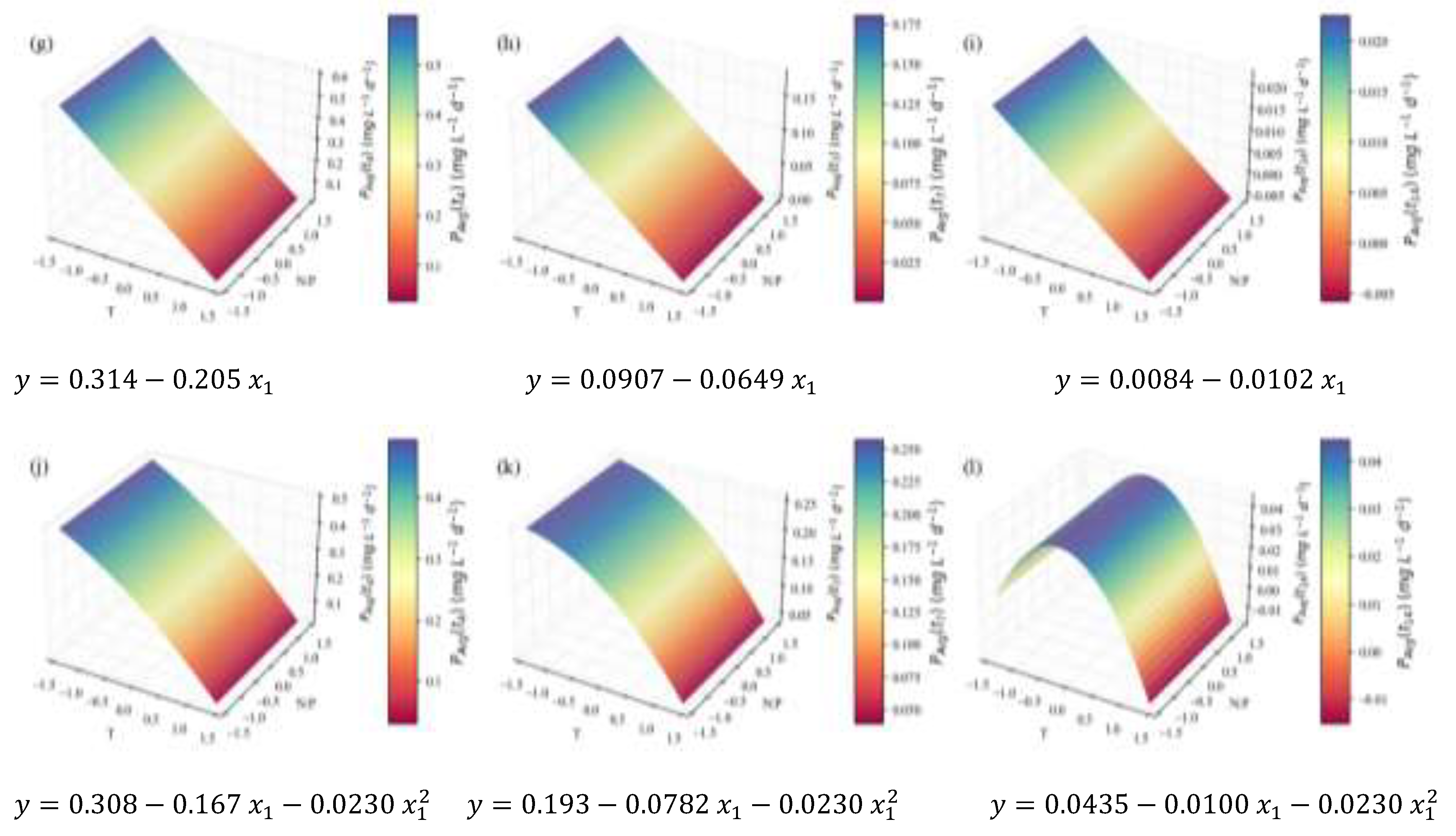
2. Results and Discussion
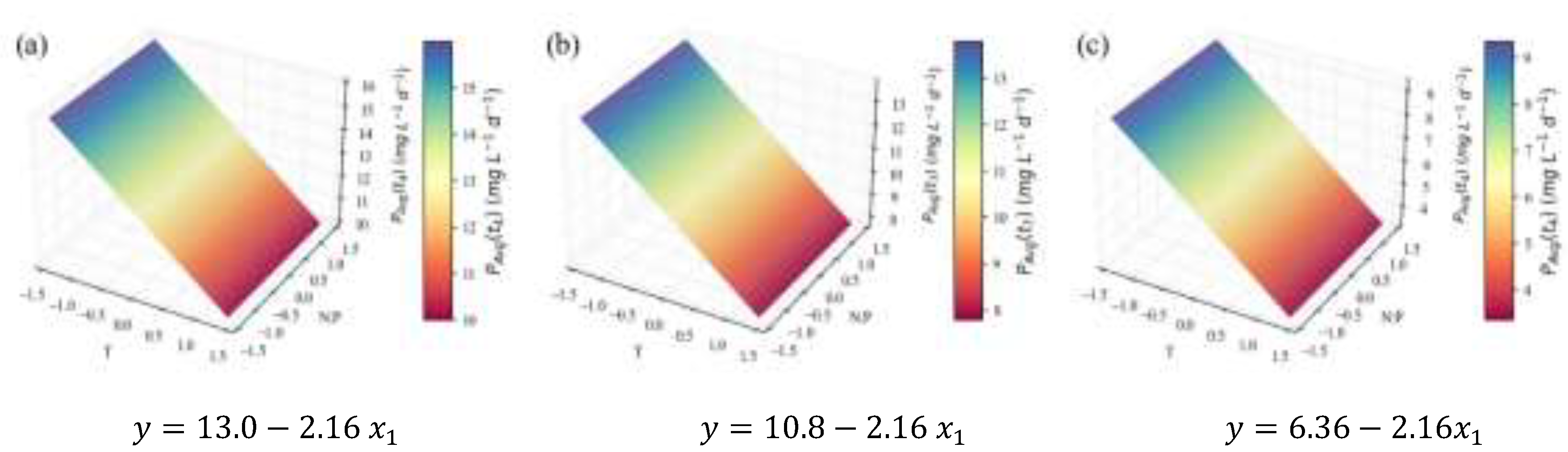
2.1. Biomass Growth and Nutrient Consumption
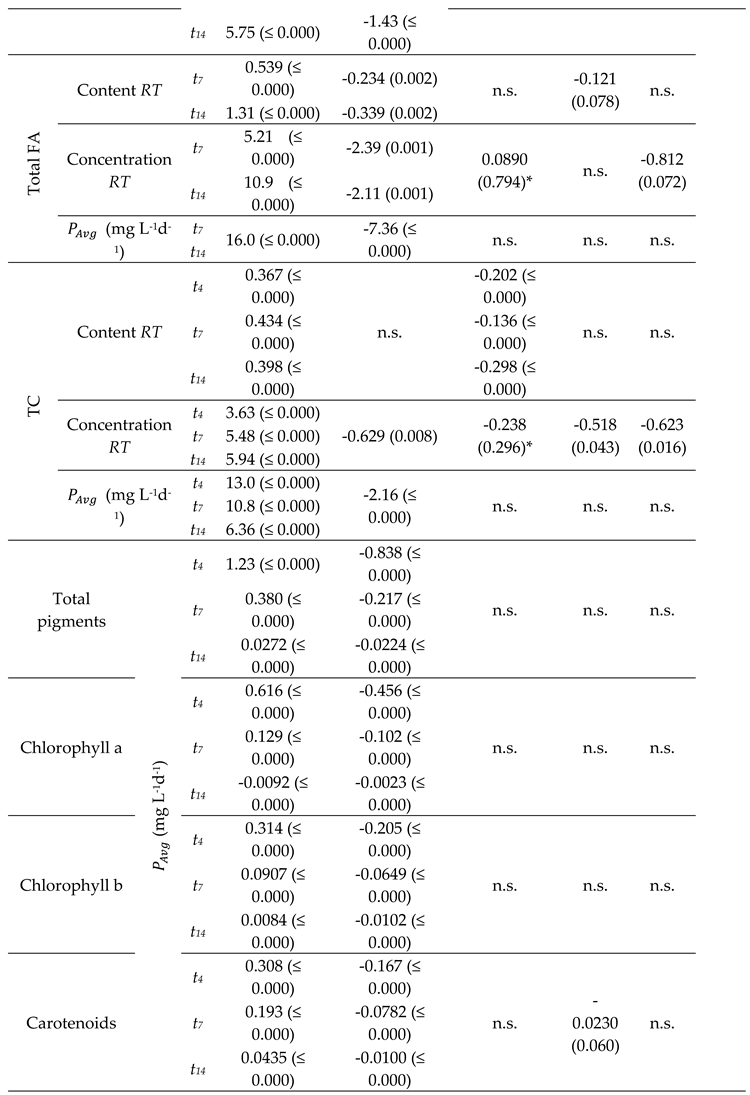
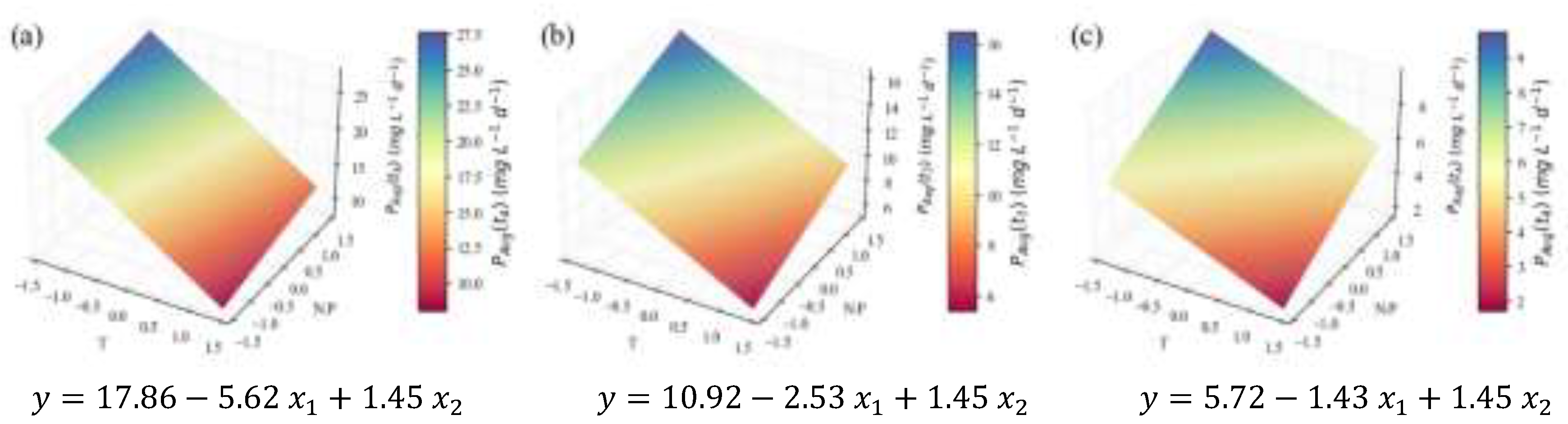
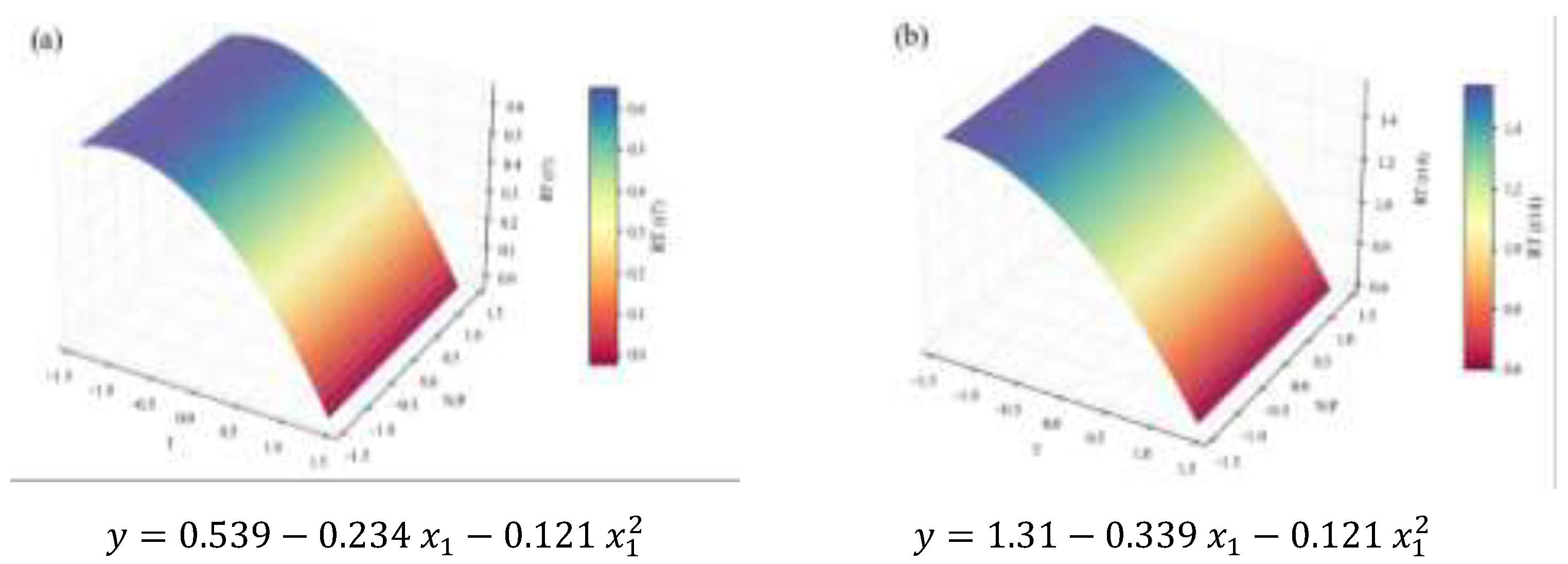
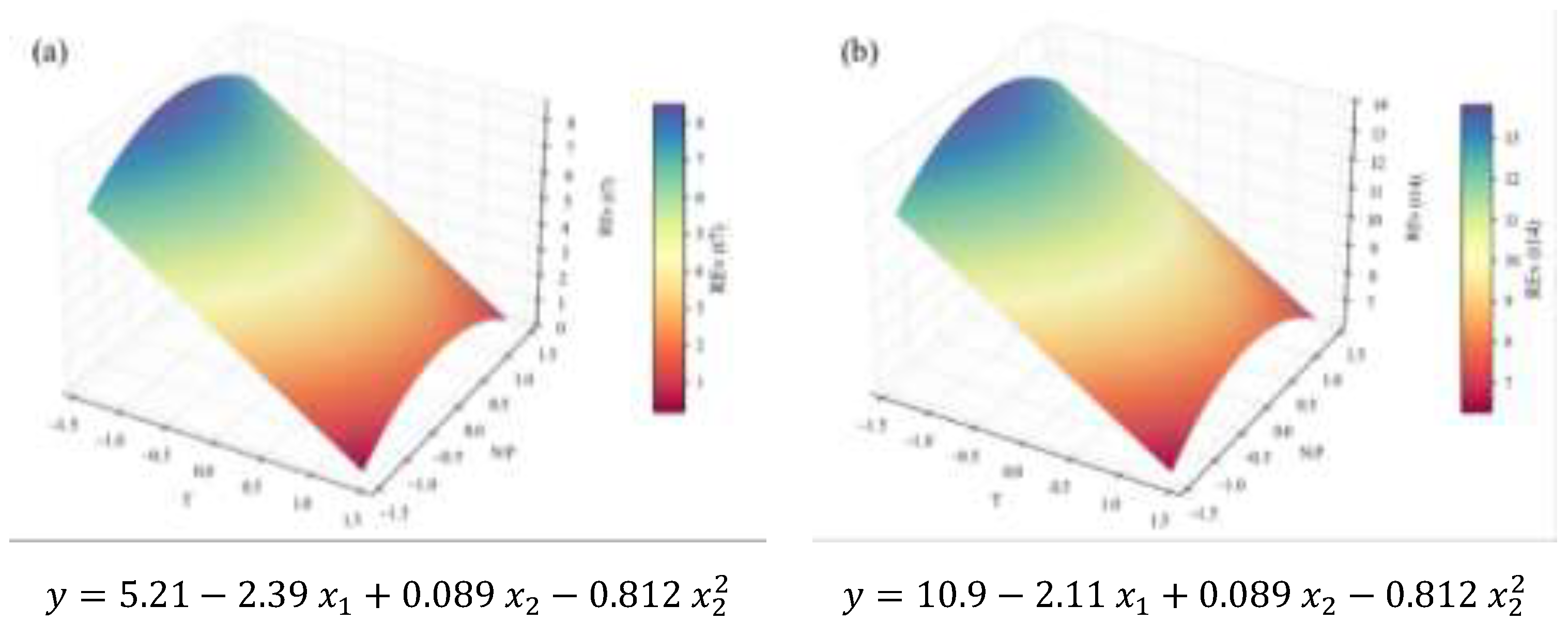
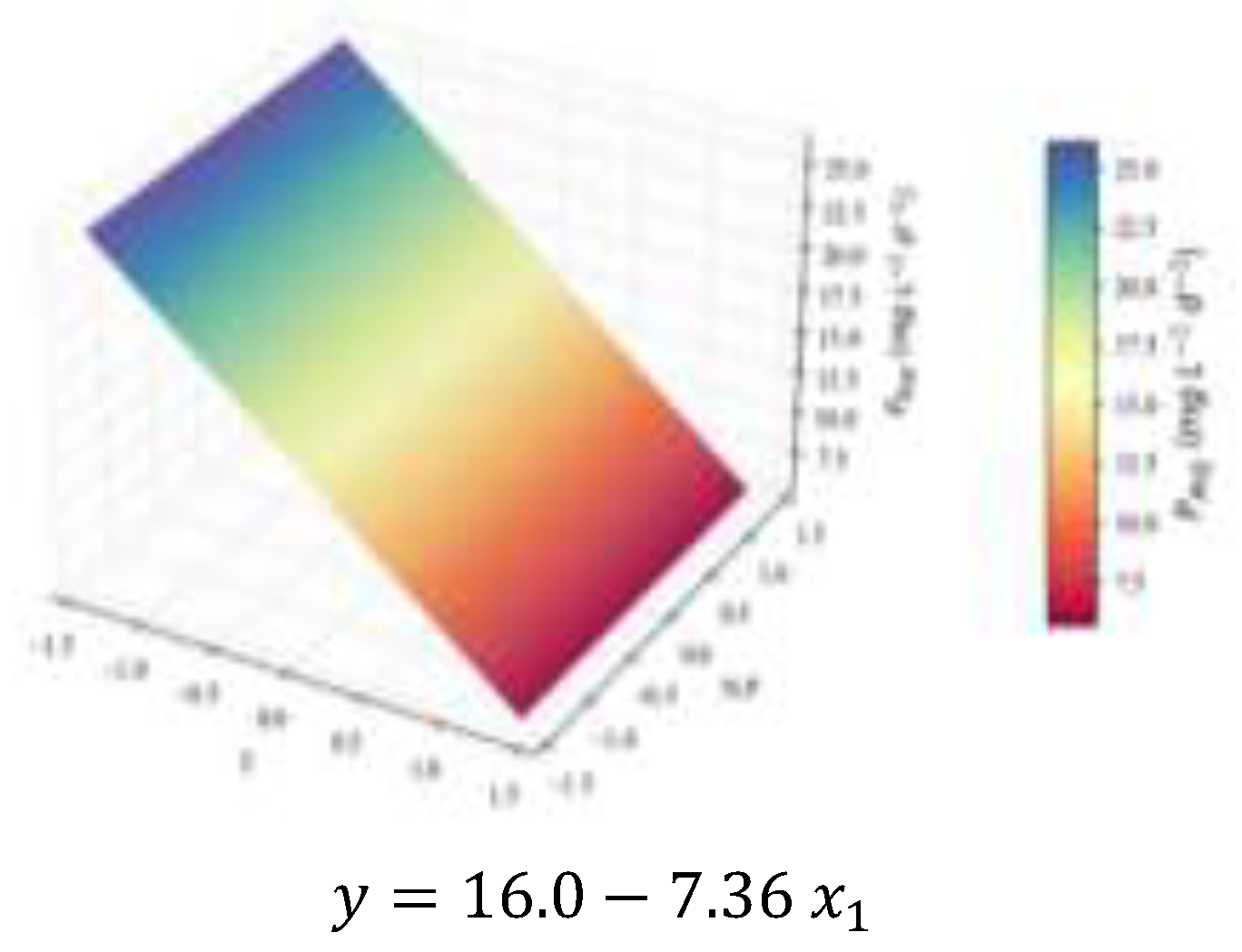
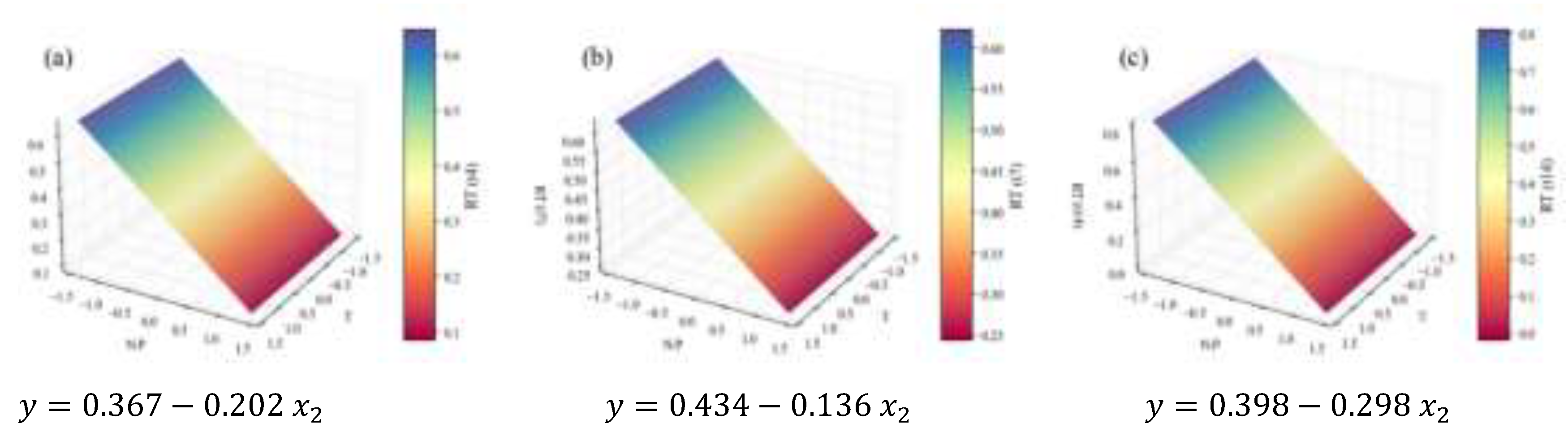
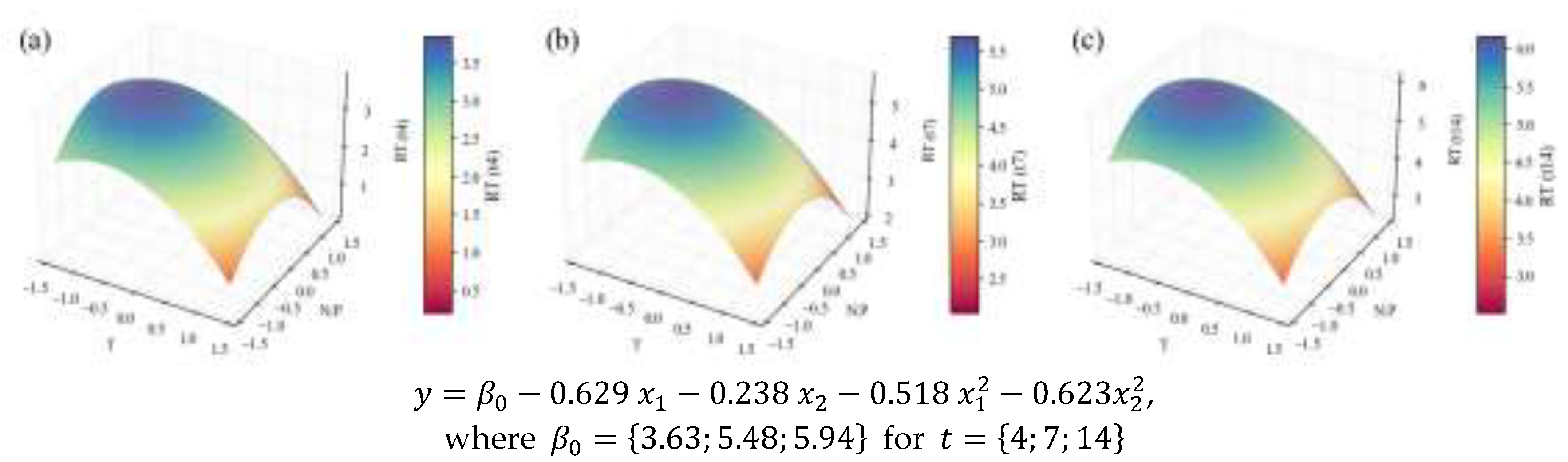
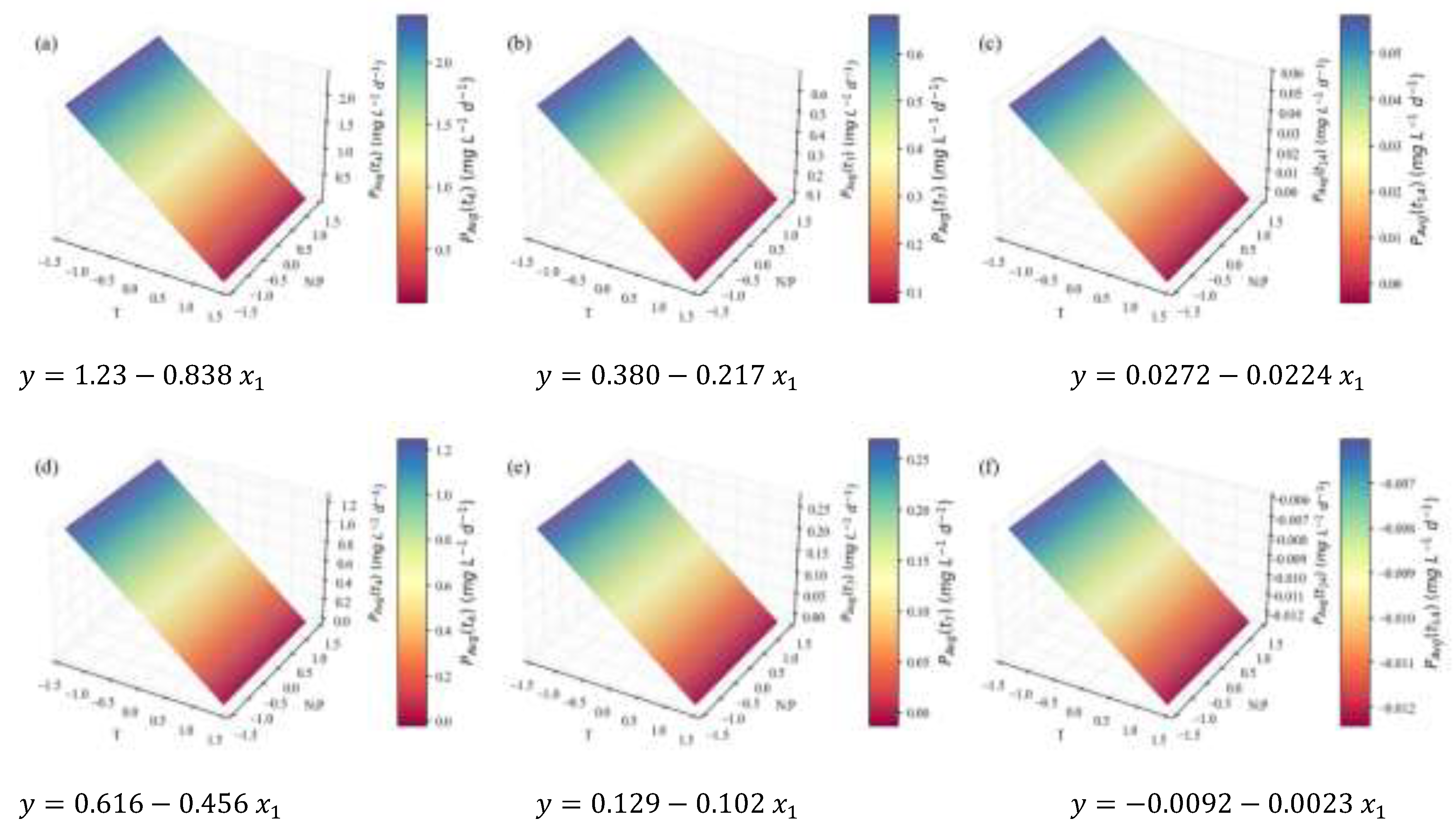
2.2. Biochemical Composition – Total Analyses
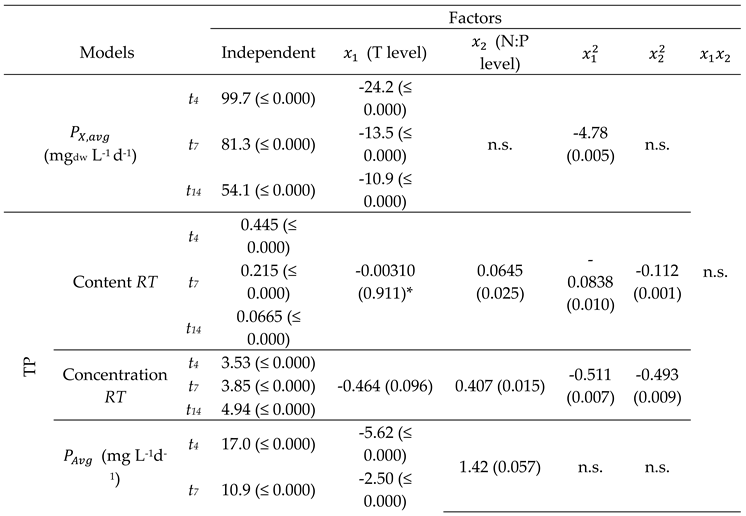
| Model summary | Response optimization | |||||||
|---|---|---|---|---|---|---|---|---|
| RMSE | y fit | Setting | ||||||
| TP | Content RT | 0.16 | 0.61 | 0.54 | 0.45 | 0.45 | ||
| Concentration RT | 0.907 | 0.78 | 0.74 | 0.69 | 4.23 | |||
| (mg L-1d-1) | 3.5 | 0.76 | 0.71 | 0.64 | 26.9 | †† | ||
| Total FA | Content RT | 0.25 | 0.86 | 0.83 | 0.73 | 1.54 | ||
| Concentration RT | 1.64 | 0.92 | 0.89 | 0.82 | 13.9 | † | ||
| (mg L-1d-1) | 4.5 | 0.68 | 0.66 | 0.58 | 26.4 | † | ||
| TC | Content RT | 0.20 | 0.61 | 0.54 | 0.40 | 0.82 | † | |
| Concentration RT | 1.25 | 0.79 | 0.75 | 0.68 | 6.15 | |||
| (mg L-1d-1) | 2.3 | 0.70 | 0.67 | 0.62 | 16.0 | † | ||
| Total pigments |
(mg L-1d-1) |
0.15 | 0.95 | 0.94 | 0.93 | 2.41 | † | |
| Chlorophyll a | 0.083 | 0.94 | 0.93 | 0.92 | 1.26 | † | ||
| Chlorophyll b | 0.044 | 0.93 | 0.92 | 0.91 | 0.60 | † | ||
| Carotenoids | 0.043 | 0.92 | 0.90 | 0.86 | 0.50 | † | ||
 |
 |
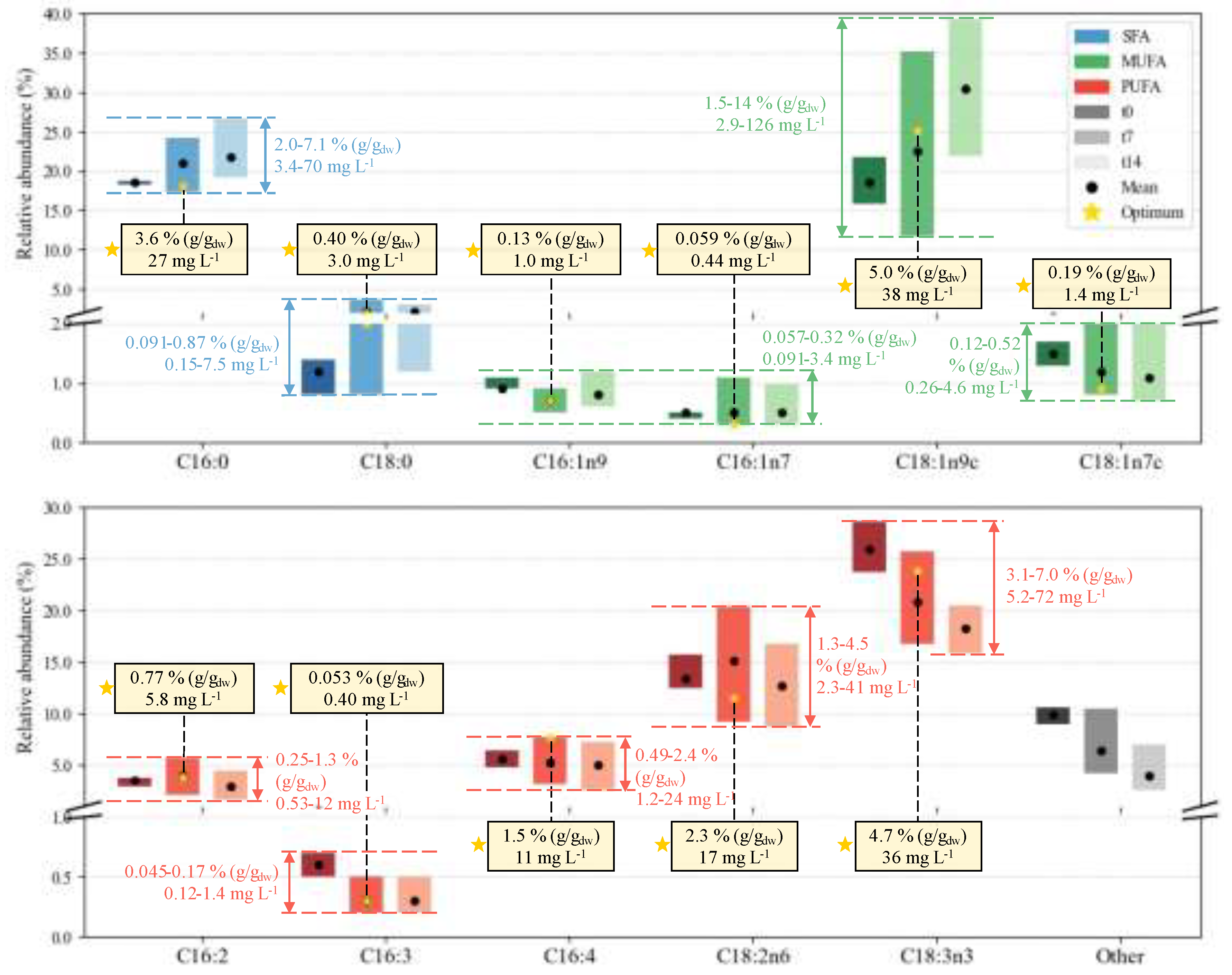
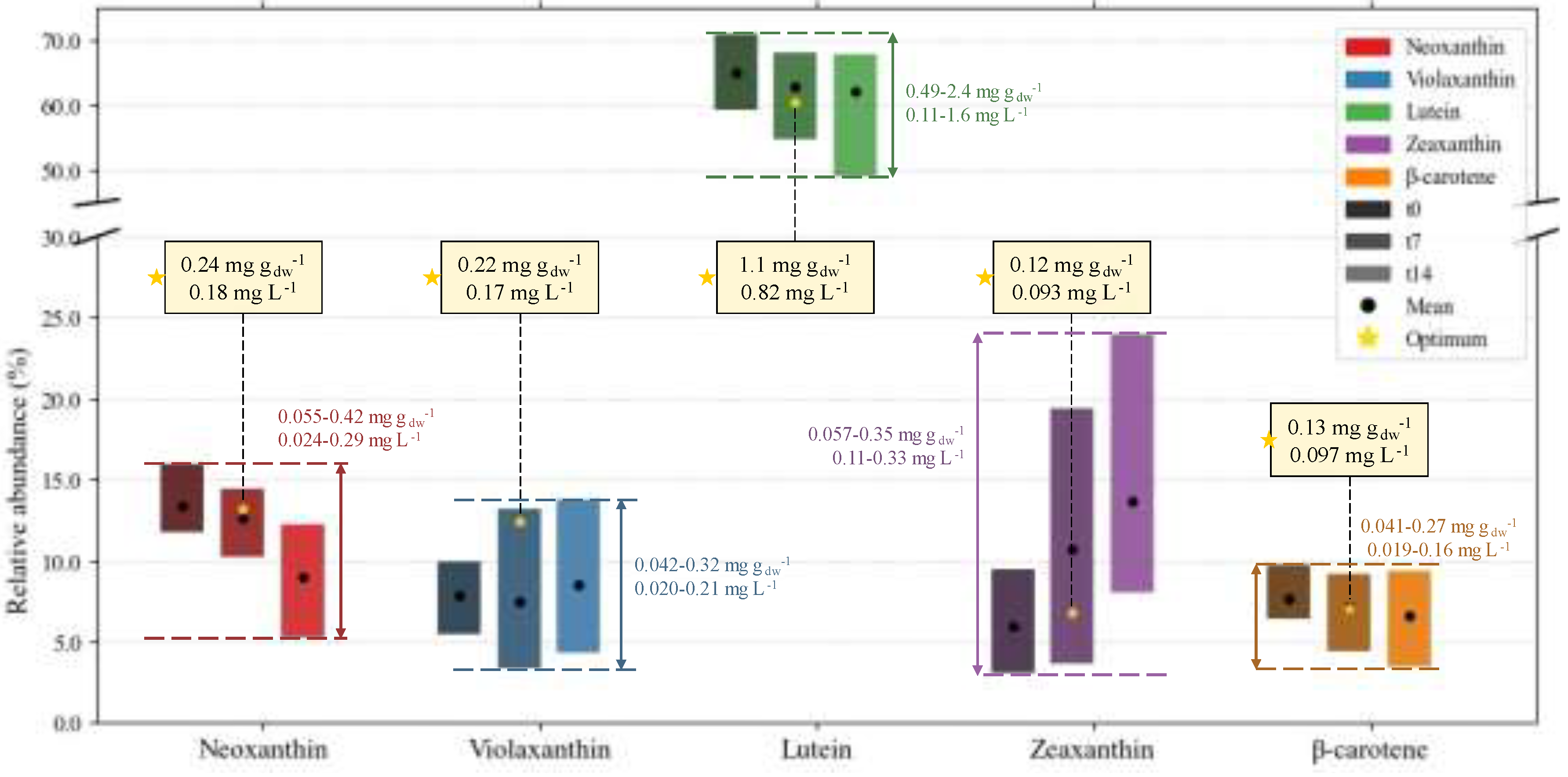
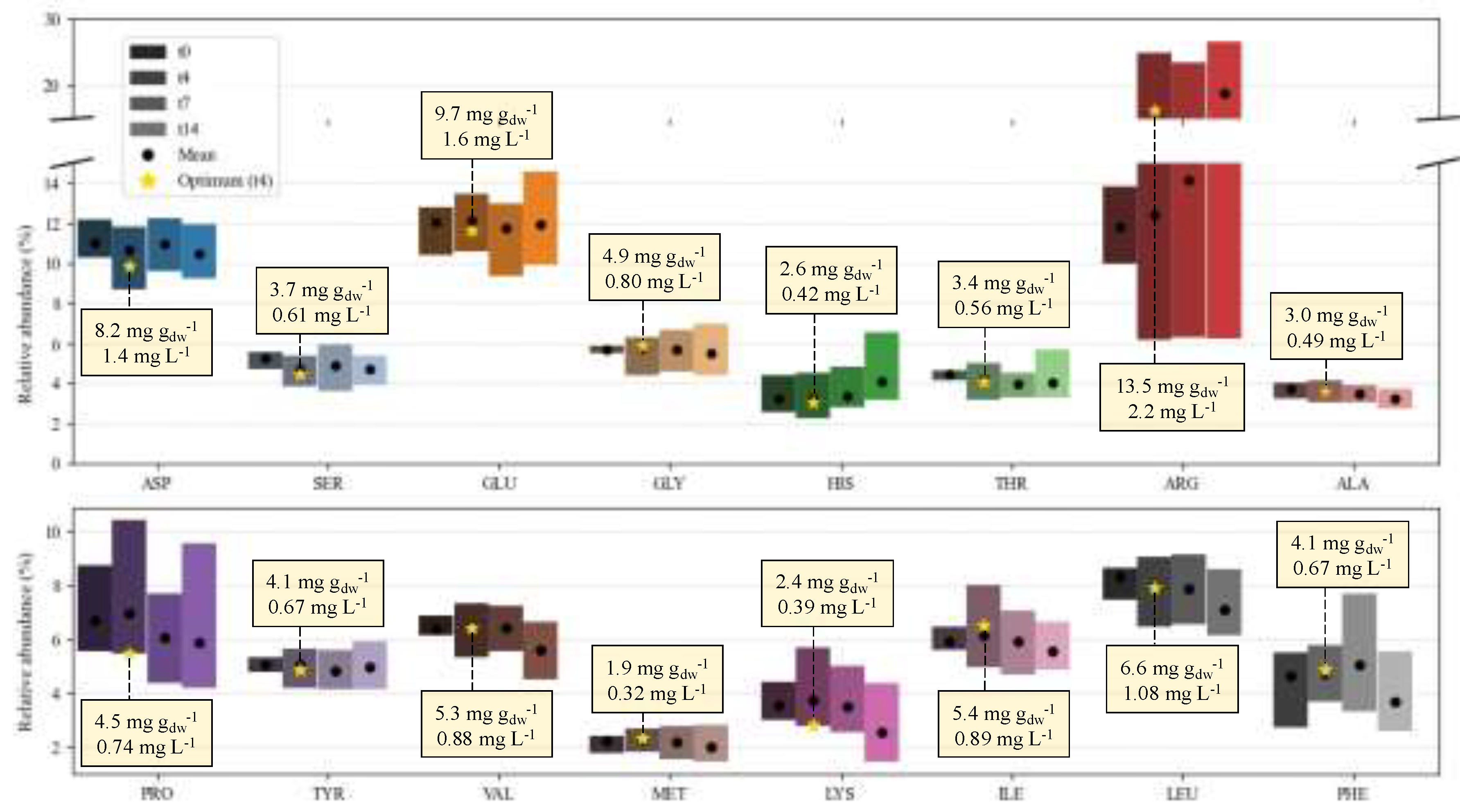
2.3. Biochemical Composition – Profiles
2.3.1. FA Profile
2.3.2. Carotenoid Profile
2.3.3. Amino Acid Profile
3. Materials and Methods
3.1. Microalgae
3.2. Inoculation and Culture Conditions
3.3. Design of Experiments and Response Surface Methodology
3.4. Biomass Growth Monitoring
3.5. Nutrient Consumption Monitoring
3.6. Harvesting and Lyophilization
3.7. Extraction and Analyses: General Biochemical Composition
3.8. Extraction and Analyses: Profiles
3.9. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ALA | Alpha-linoleic acid |
| ANN | Artificial Neural Network |
| ANOVA | Analysis of Variance |
| ATP | Adenosine Triphosphate |
| CAGR | Compound Annual Growth Rate |
| CCAP | Culture Collection of Algae and Protozoa |
| CCD | Central Composite Design |
| DNA | Deoxyribonucleic acid |
| DoE | Design of Experiments |
| EAA | Essential Amino Acid |
| EPA | Eicosapentaenoic acid |
| FA | Fatty Acid |
| FAME | Fatty Acid Methyl Ester |
| GC | Gas Chromatography |
| GM | Growth Medium |
| HPLC | High-Performance Liquid Chromatography |
| HSD | Honestly Significant Difference |
| LA | Linoleic acid |
| LOD | Limit of Detection |
| LOQ | Limit of Quantification |
| MUFA | Monounsaturated Fatty Acid |
| OD | Optical Density |
| OECD | Organization for Economic Cooperation and Development |
| PUFA | Polyunsaturated Fatty Acid |
| RMSE | Root Mean Square Error |
| RNA | Ribonucleic acid |
| ROS | Reactive Oxygen Species |
| RSM | Response Surface Methodology |
| SD | Standard Deviation |
| SDG | Sustainable Development Goals |
| SE | Standard Error |
| SFA | Saturated Fatty Acid |
| T | Temperature |
| TAG | Triacylglycerol |
| TC | Total Carboydrates |
| TP | Total Proteins |
| UFA | Unsaturated Fatty Acid |
| UN | United Nations |
Nomenclature
| x1 | Independent variable 1 (level of temperature) | |
| x2 | Independent variable 2 (level of N:P molar ratio) | |
| Z1 | Variable representing temperature | °C |
| Z2 | Variable representing N:P molar ratio | |
| Regression coefficient | ||
| y | Response variable | |
| α | CCD parameter | |
| E(X) | Experiment number X | |
| R2 | Coefficient of determination | % |
| X | Biomass concentration | mgdw L-1 |
| μ | Growth rate | d-1 |
| t | Time | d |
| P | Productivity | mg L-1 d-1 |
| RE | Removal Efficiency | % |
| RR | Average Removal Rate | mg L-1 d-1 |
| S | Substrate/Nutrient | mg L-1 |
| λ | Wavelength | nm |
| RT | Relative Tendency |
| i; j | Variable counter |
| 0 | Initial |
| f | Final |
| exp | Exponential |
| dw | Dry weight |
| X | Biomass |
| Avg | Average |
| Adj | Adjusted |
| Pred | Predicted |
References
- Su, Y. Revisiting carbon, nitrogen, and phosphorus metabolisms in microalgae for wastewater treatment. Science of The Total Environment 2021, 762, 144590. [Google Scholar] [CrossRef]
- Esteves, A.F.; Gonçalves, A.L.; Vilar, V.J.P.; Pires, J.C.M. Is it possible to shape the microalgal biomass composition with operational parameters for target compound accumulation? Biotechnol. Adv. 2025, 79, 108493. [Google Scholar] [CrossRef]
- Sousa, S.A.; Esteves, A.F.; Salgado, E.M.; Pires, J.C.M. Enhancing urban wastewater treatment: Chlorella vulgaris performance in tertiary treatment and the impact of anaerobic digestate addition. Environ. Technol. Innov. 2024, 34, 103601. [Google Scholar] [CrossRef]
- Oliveira, J.; Pardilhó, S.; Costa, E.; Pires, J.C.; Dias, J.M. Exploring microalgae species for integrated bioenergy Production: A Multi-Fuel cascade valorisation approach. Energy Convers. Manag. 2025, 332, 119736. [Google Scholar] [CrossRef]
- Esteves, A.F.; Soares, O.S.G.P.; Vilar, V.J.P.; Pires, J.C.M.; Gonçalves, A.L. The Effect of Light Wavelength on CO2 Capture, Biomass Production and Nutrient Uptake by Green Microalgae: A Step Forward on Process Integration and Optimisation. Energies (Basel) 2020, 13, 333. [Google Scholar] [CrossRef]
- Olabi, A.G.; et al. Role of microalgae in achieving sustainable development goals and circular economy. Science of The Total Environment 2023, 854, 158689. [Google Scholar] [CrossRef]
- Garrido-Cardenas, J.A.; Manzano-Agugliaro, F.; Acien-Fernandez, F.G.; Molina-Grima, E. Microalgae research worldwide. Algal Res. 2018, 35, 50–60. [Google Scholar] [CrossRef]
- Beijerinck, M.W. Culturversuche mit Zoochlorellen, Lichenengonidien und anderen niederen Algen. Botanische Zeitung 1890, 47, 725–772. [Google Scholar]
- Mendes, A.R.; Spínola, M.P.; Lordelo, M.; Prates, J.A.M. Chemical Compounds, Bioactivities, and Applications of Chlorella vulgaris in Food, Feed and Medicine. Applied Sciences 2024, 14, 10810. [Google Scholar] [CrossRef]
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef]
- Barbarino, E.; Lourenço, S.O. An evaluation of methods for extraction and quantification of protein from marine macro- and microalgae. J. Appl. Phycol. 2005, 17, 447–460. [Google Scholar] [CrossRef]
- Barkia, I.; Saari, N.; Manning, S.R. Microalgae for High-Value Products Towards Human Health and Nutrition. Mar. Drugs 2019, 17, 304. [Google Scholar] [CrossRef]
- Esteves, A.F.C. Shaping microalgal biomass composition for targeted product accumulation: impact of operational parameters. Oct 2024. Available online: https://repositorio-aberto.up.pt/handle/10216/162001.
- Safi, C.; Zebib, B.; Merah, O.; Pontalier, P.Y.; Vaca-Garcia, C. Morphology, composition, production, processing and applications of Chlorella vulgaris: A review. Renewable and Sustainable Energy Reviews 2014, 35, 265–278. [Google Scholar] [CrossRef]
- Arguelles, E.D.; Martinez-Goss, M.R. Lipid accumulation and profiling in microalgae Chlorolobion sp. (BIOTECH 4031) and Chlorella sp. (BIOTECH 4026) during nitrogen starvation for biodiesel production. J. Appl. Phycol. 2021, 33, 1–11. [Google Scholar] [CrossRef]
- Dong, L.; Li, D.; Li, C. Characteristics of lipid biosynthesis of Chlorella pyrenoidosa under stress conditions. Bioprocess Biosyst. Eng. 2020, 43, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Yuan, Y.; Cheng, D.; Zhao, Q. Effect of light quality on growth rate, carbohydrate accumulation, fatty acid profile and lutein biosynthesis of Chlorella sp. AE10. Bioresour. Technol. 2019, 291, 121783. [Google Scholar] [CrossRef]
- Magyar, T.; Németh, B.; Tamás, J.; Nagy, P.T. Improvement of N and P ratio for enhanced biomass productivity and sustainable cultivation of Chlorella vulgaris microalgae. Heliyon 2024, 10. [Google Scholar] [CrossRef]
- Salgado, E.M.; Esteves, A.F.; Gonçalves, A.L.; Pires, J.C.M. Microalgal cultures for the remediation of wastewaters with different nitrogen to phosphorus ratios: Process modelling using artificial neural networks. Environ. Res. 2023, 231, 116076. [Google Scholar] [CrossRef] [PubMed]
- Montoya-Vallejo, C.; Duque, F.L.G.; Díaz, J.C.Q. Biomass and lipid production by the native green microalgae Chlorella sorokiniana in response to nutrients, light intensity, and carbon dioxide: experimental and modeling approach. Front. Bioeng. Biotechnol. 2023, 11, 1149762. [Google Scholar] [CrossRef]
- Santhakumaran, P.; Kookal, S.K.; Mathew, L.; Ray, J.G. Experimental evaluation of the culture parameters for optimum yield of lipids and other nutraceutically valuable compounds in Chloroidium saccharophillum. Renew. Energy 2020, 147, 1082–1097. [Google Scholar] [CrossRef]
- Qari, H.A.; Oves, M. Fatty acid synthesis by Chlamydomonas reinhardtii in phosphorus limitation. J. Bioenerg. Biomembr. 2020, 52, 27–38. [Google Scholar] [CrossRef]
- Bajwa, K.; Bishnoi, N.R.; Kirrolia, A.; Gupta, S.; Selvan, S.T. Response surface methodology as a statistical tool for optimization of physio-biochemical cellular components of microalgae Chlorella pyrenoidosa for biodiesel production. Appl. Water Sci. 2019, 9, 1–16. [Google Scholar] [CrossRef]
- Xu, K.; Zou, X.; Wen, H.; Xue, Y.; Qu, Y.; Li, Y. Effects of multi-temperature regimes on cultivation of microalgae in municipal wastewater to simultaneously remove nutrients and produce biomass. Appl. Microbiol. Biotechnol. 2019, 103, 8255–8265. [Google Scholar] [CrossRef]
- Converti, A.; Casazza, A.A.; Ortiz, E.Y.; Perego, P.; Del Borghi, M. Effect of temperature and nitrogen concentration on the growth and lipid content of Nannochloropsis oculata and Chlorella vulgaris for biodiesel production. Chemical Engineering and Processing: Process Intensification 2009, 48, 1146–1151. [Google Scholar] [CrossRef]
- Zhao, T.; Han, X.; Cao, H. Effect of Temperature on Biological Macromolecules of Three Microalgae and Application of FT-IR for Evaluating Microalgal Lipid Characterization. ACS Omega 2020, 5, 33262–33268. [Google Scholar] [CrossRef]
- Chauhan, D.S.; Sahoo, L.; Mohanty, K. Acclimation-driven microalgal cultivation improved temperature and light stress tolerance, CO2 sequestration and metabolite regulation for bioenergy production. Bioresour. Technol. 2023, 385, 129386. [Google Scholar] [CrossRef]
- Maneechote, W.; Cheirsilp, B. Stepwise-incremental physicochemical factors induced acclimation and tolerance in oleaginous microalgae to crucial outdoor stresses and improved properties as biodiesel feedstocks. Bioresour. Technol. 2021, 328, 124850. [Google Scholar] [CrossRef] [PubMed]
- Pereira, S.; Otero, A. Haematococcus pluvialis bioprocess optimization: Effect of light quality, temperature and irradiance on growth, pigment content and photosynthetic response. Algal Res. 2020, 51, 102027. [Google Scholar] [CrossRef]
- Ördög, V.; et al. Effect of temperature and nitrogen concentration on lipid productivity and fatty acid composition in three Chlorella strains. Algal Res. 2016, 16, 141–149. [Google Scholar] [CrossRef]
- Kazeem, M.A.; Hossain, S.M.Z.; Hossain, M.M.; Razzak, S.A. Application of central composite design to optimize culture conditions of Chlorella vulgaris in a batch photobioreactor: An efficient modeling approach. Chemical Product and Process Modeling 2018, 13. [Google Scholar] [CrossRef]
- Geada, P.; et al. Food and Bioproducts Processing Multivariable optimization process of heterotrophic growth of Chlorella vulgaris. In Allmicroalgae Natural Products S.A; 2022. [Google Scholar] [CrossRef]
- Dunn, J.; Grider, M.H. Physiology, Adenosine Triphosphate. StatPearls. Feb 2023. Available online: http://europepmc.org/books/NBK553175.
- Umdu, E.S.; Univ, Y. Building integrated photobioreactor. in Bio-based Materials and Biotechnologies for Eco-efficient Construction; Pacheco-Torgal, F., Ivanov, V., Tsang, D., Eds.; Woodhead Publishing, 2020; pp. 243–258. [Google Scholar] [CrossRef]
- Song, X.; Liu, B.F.; Kong, F.; Ren, N.Q.; Ren, H.Y. Overview on stress-induced strategies for enhanced microalgae lipid production: Application, mechanisms and challenges. Resour. Conserv. Recycl. 2022, 183, 106355. [Google Scholar] [CrossRef]
- Mustapha, S.I.; Rawat, I.; Bux, F.; Isa, Y.M. Enhancing the efficiency of thermal conversion of microalgae: a review. Biomass Convers. Biorefin. 2023, 13, 8813–8827. [Google Scholar] [CrossRef]
- Sui, Y.; Vlaeminck, S.E. Dunaliella Microalgae for Nutritional Protein: An Undervalued Asset. Trends Biotechnol. 2020, 38, 10–12. [Google Scholar] [CrossRef]
- Gauthier, M.R.; Senhorinho, G.N.A.; Scott, J.A. Microalgae under environmental stress as a source of antioxidants. Algal Res. 2020, 52, 102104. [Google Scholar] [CrossRef]
- Maltseva, Y.M.K.; Maltsev, Y.; Maltseva, K.; Khmelnytskyi, B. Fatty acids of microalgae: diversity and applications. Reviews in Environmental Science and Bio/Technology 2021, 20, 515–547. [Google Scholar] [CrossRef]
- Fu, L.; Li, Q.; Yan, G.; Zhou, D.; Crittenden, J.C. Hormesis effects of phosphorus on the viability of Chlorella regularis cells under nitrogen limitation. Biotechnol. Biofuels 2019, 12, 1–9. [Google Scholar] [CrossRef]
- Chu, F.; Cheng, J.; Zhang, X.; Ye, Q.; Zhou, J. Enhancing lipid production in microalgae Chlorella PY-ZU1 with phosphorus excess and nitrogen starvation under 15% CO2 in a continuous two-step cultivation process. Chemical Engineering Journal 2019, 375, 121912. [Google Scholar] [CrossRef]
- Oslan, S.N.H.; et al. A Review on Haematococcus pluvialis Bioprocess Optimization of Green and Red Stage Culture Conditions for the Production of Natural Astaxanthin. Biomolecules 2021, 11, 256. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Chen, A.; Huang, L.; Zhang, C.; Gao, B. Transcriptomic analysis unravels the modulating mechanisms of the biomass and value-added bioproducts accumulation by light spectrum in Eustigmatos cf. polyphem (Eustigmatophyceae). Bioresour. Technol. 2021, 338, 125523. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; et al. Effects of different abiotic stresses on carotenoid and fatty acid metabolism in the green microalga Dunaliella salina Y6. Ann. Microbiol. 2020, 70, 1–9. [Google Scholar] [CrossRef]
- Schüler, L.M.; et al. Improved production of lutein and β-carotene by thermal and light intensity upshifts in the marine microalga Tetraselmis sp. CTP4. Algal Res. 2020, 45, 101732. [Google Scholar] [CrossRef]
- Costa, J.A.V.; de Morais, M.G. An Open Pond System for Microalgal Cultivation. in Biofuels from Algae; Pandey, A., Lee, D.-J., Chisti, Y., Soccol, C., Eds.; Elsevier, 2014; pp. 1–22. [Google Scholar] [CrossRef]
- Gao, B.; Hong, J.; Chen, J.; Zhang, H.; Hu, R.; Zhang, C. The growth, lipid accumulation and adaptation mechanism in response to variation of temperature and nitrogen supply in psychrotrophic filamentous microalga Xanthonema hormidioides (Xanthophyceae). Biotechnology for Biofuels and Bioproducts 2023, 16, 1–16. [Google Scholar] [CrossRef]
- Ma, R.; et al. Co-production of lutein and fatty acid in microalga Chlamydomonas sp. JSC4 in response to different temperatures with gene expression profiles. Algal Res. 2020, 47, 101821. [Google Scholar] [CrossRef]
- Li, X.; Slavens, S.; Crunkleton, D.W.; Johannes, T.W. Interactive effect of light quality and temperature on Chlamydomonas reinhardtii growth kinetics and lipid synthesis. Algal Res. 2021, 53, 102127. [Google Scholar] [CrossRef]
- Yang, Y.F.; et al. Utilization of lipidic food waste as low-cost nutrients for enhancing the potentiality of biofuel production from engineered diatom under temperature variations. Bioresour. Technol. 2023, 387, 129611. [Google Scholar] [CrossRef] [PubMed]
- Ugya, A.Y.; Imam, T.S.; Li, A.; Ma, J.; Hua, X. Antioxidant response mechanism of freshwater microalgae species to reactive oxygen species production: a mini review. Chemistry and Ecology 2020, 36, 174–193. [Google Scholar] [CrossRef]
- Pinto, A.S.; Maia, C.; Sousa, S.A.; Tavares, T.; Pires, J.C.M. Amino Acid and Carotenoid Profiles of Chlorella vulgaris During Two-Stage Cultivation at Different Salinities. Bioengineering 2025, 12, 284. [Google Scholar] [CrossRef]
- Barten, R.J.P.; Wijffels, R.H.; Barbosa, M.J. Bioprospecting and characterization of temperature tolerant microalgae from Bonaire. Algal Res. 2020, 50, 102008. [Google Scholar] [CrossRef]
- Xing, C.; Li, J.; Yuan, H.; Yang, J. Physiological and transcription level responses of microalgae Auxenochlorella protothecoides to cold and heat induced oxidative stress. Environ. Res. 2022, 211, 113023. [Google Scholar] [CrossRef] [PubMed]
- Peter, A.P.; et al. Continuous cultivation of microalgae in photobioreactors as a source of renewable energy: Current status and future challenges. Renewable and Sustainable Energy Reviews 2022, 154, 111852. [Google Scholar] [CrossRef]
- Gifuni, I.; Pollio, A.; Safi, C.; Marzocchella, A.; Olivieri, G. Current Bottlenecks and Challenges of the Microalgal Biorefinery. Trends Biotechnol. 2019, 37, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Serra-Maia, R.; Bernard, O.; Gonçalves, A.; Bensalem, S.; Lopes, F. Influence of temperature on Chlorella vulgaris growth and mortality rates in a photobioreactor. Algal Res. 2016, 18, 352–359. [Google Scholar] [CrossRef]
- Deniz, İ. Determination of growth conditions for Chlorella vulgaris. Mar. Sci. Tech. Bull 2020, 9, 114–117. [Google Scholar] [CrossRef]
- Mathimani, T.; et al. Semicontinuous outdoor cultivation and efficient harvesting of marine Chlorella vulgaris BDUG 91771 with minimum solid co-precipitation and high floc recovery for biodiesel. Energy Convers. Manag. 2017, 149, 13–25. [Google Scholar] [CrossRef]
- Theses, U.U.K.; Engineering, A.; Cassidy, K.O. Evaluating Algal Growth at Different Temperatures. Theses and Dissertations--Biosystems and Agricultural Engineering. Jan 2011. Available online: https://uknowledge.uky.edu/bae_etds/3.
- Blinová, L.; Bartošová, A.; Gerulová, K.; Blinová, I.L.; Bartošová, I.A.; Gerulová, I.K. Cultivation of microalgae (Chlorella vulgaris) for biodiesel production. 2015, 23, 36. [Google Scholar] [CrossRef]
- James, C.M.; Al-Hinty, S.; Salman, A.E. Growth and ω3 fatty acid and amino acid composition of microalgae under different temperature regimes. Aquaculture 1989, 77, 337–351. [Google Scholar] [CrossRef]
- Chu, R.; Hu, D.; Zhu, L.; Li, S.; Yin, Z.; Yu, Y. Recycling spent water from microalgae harvesting by fungal pellets to re-cultivate Chlorella vulgaris under different nutrient loads for biodiesel production. Bioresour. Technol. 2022, 344, 126227. [Google Scholar] [CrossRef]
- Silva, N.F.P.; et al. Towards sustainable microalgal biomass production by phycoremediation of a synthetic wastewater: A kinetic study. Algal Res. 2015, 11, 350–358. [Google Scholar] [CrossRef]
- Rodrigues-Sousa, A.E.; Nunes, I.V.O.; Muniz-Junior, A.B.; Carvalho, J.C.M.; Mejia-da-Silva, L.C.; Matsudo, M.C. Nitrogen supplementation for the production of Chlorella vulgaris biomass in secondary effluent from dairy industry. Biochem. Eng. J. 2021, 165, 107818. [Google Scholar] [CrossRef]
- Chen, C.Y.; et al. Improving protein production of indigenous microalga Chlorella vulgaris FSP-E by photobioreactor design and cultivation strategies. Biotechnol. J. 2015, 10, 905–914. [Google Scholar] [CrossRef] [PubMed]
- Mahboob, S.; et al. High-density growth and crude protein productivity of a thermotolerant Chlorella vulgaris: Production kinetics and thermodynamics. Aquaculture International 2012, 20, 455–466. [Google Scholar] [CrossRef]
- Metsoviti, M.N.; Katsoulas, N.; Karapanagiotidis, I.T.; Papapolymerou, G. Effect of nitrogen concentration, two-stage and prolonged cultivation on growth rate, lipid and protein content of Chlorella vulgaris. In Journal of Chemical Technology and Biotechnology; STRING:PUBLICATION: WGROUP, 2019; Volume 94, pp. 1466–1473. [Google Scholar] [CrossRef]
- Sui, Y.; Muys, M.; Vermeir, P.; D’Adamo, S.; Vlaeminck, S.E. Light regime and growth phase affect the microalgal production of protein quantity and quality with Dunaliella salina. Bioresour. Technol. 2019, 275, 145–152. [Google Scholar] [CrossRef]
- Fernández-Reiriz, M.J.; et al. Biomass production and variation in the biochemical profile (total protein, carbohydrates, RNA, lipids and fatty acids) of seven species of marine microalgae. Aquaculture 1989, 83, 17–37. [Google Scholar] [CrossRef]
- Sajjadi, B.; Chen, W.Y.; Raman, A.A.A.; Ibrahim, S. Microalgae lipid and biomass for biofuel production: A comprehensive review on lipid enhancement strategies and their effects on fatty acid composition. Renewable and Sustainable Energy Reviews 2018, 97, 200–232. [Google Scholar] [CrossRef]
- Klin, M.; Pniewski, F.; Latała, A. Growth phase-dependent biochemical composition of green microalgae: Theoretical considerations for biogas production. Bioresour. Technol. 2020, 303, 122875. [Google Scholar] [CrossRef] [PubMed]
- Ju, J.H.; et al. Regulation of lipid accumulation using nitrogen for microalgae lipid production in Schizochytrium sp. ABC101. Renew. Energy 2020, 153, 580–587. [Google Scholar] [CrossRef]
- Ho, S.H.; Chen, C.Y.; Chang, J.S. Effect of light intensity and nitrogen starvation on CO2 fixation and lipid/carbohydrate production of an indigenous microalga Scenedesmus obliquus CNW-N. Bioresour. Technol. 2012, 113, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Ikaran, Z.; Suárez-Alvarez, S.; Urreta, I.; Castañón, S. The effect of nitrogen limitation on the physiology and metabolism of Chlorella vulgaris var L3. Algal Res. 2015, 10, 134–144. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. [34] Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar] [CrossRef]
- 11.1: Fatty Acids - Chemistry LibreTexts. Available online: https://chem.libretexts.org/Courses/American_River_College/CHEM_309%3A_Applied_Chemistry_for_the_Health_Sciences/11%3A_Lipids_-_An_Introduction/11.01%3A_Fatty_Acids (accessed on Sep. 06 2025).
- Patel, A.; Rova, U.; Christakopoulos, P.; Matsakas, L. Introduction to essential fatty acids. In Nutraceutical Fatty Acids from Oleaginous Microalgae: A Human Health Perspective; JOURNAL:JOURNAL:BOOKS: WGROUP; STRING:PUBLICATION, Jun 2020; pp. 1–22. [Google Scholar] [CrossRef]
- Moradi-Kheibari, N.; Ahmadzadeh, H.; Lyon, S.R. Correlation of Total Lipid Content of Chlorella vulgaris With the Dynamics of Individual Fatty Acid Growth Rates. Front. Mar. Sci. 2022, 9, 837067. [Google Scholar] [CrossRef]
- Yun, H.S.; Kim, Y.S.; Yoon, H.S. Characterization of Chlorella sorokiniana and Chlorella vulgaris fatty acid components under a wide range of light intensity and growth temperature for their use as biological resources. Heliyon 2020, vol. 6. [Google Scholar] [CrossRef]
- Kapoor, B.; Kapoor, D.; Gautam, S.; Singh, R.; Bhardwaj, S. Dietary Polyunsaturated Fatty Acids (PUFAs): Uses and Potential Health Benefits. Curr. Nutr. Rep. 2021, 10, 232–242. [Google Scholar] [CrossRef]
- Wang, C.A.; Onyeaka, H.; Miri, T.; Soltani, F. Chlorella vulgaris as a food substitute: Applications and benefits in the food industry. J. Food Sci. 2024, 89, 8231–8247. [Google Scholar] [CrossRef]
- Anthony, J.; Sivashankarasubbiah, K.T.; Thonthula, S.; Rangamaran, V.R.; Gopal, D.; Ramalingam, K. An efficient method for the sequential production of lipid and carotenoids from the Chlorella Growth Factor-extracted biomass of Chlorella vulgaris. J. Appl. Phycol. 2018, 30, 2325–2335. [Google Scholar] [CrossRef]
- Wu, K.; et al. Optimizing Chlorella vulgaris Cultivation to Enhance Biomass and Lutein Production. Foods 2024, 13, 2514. [Google Scholar] [CrossRef] [PubMed]
- McClure, D.D.; Nightingale, J.K.; Luiz, A.; Black, S.; Zhu, J.; Kavanagh, J.M. Pilot-scale production of lutein using Chlorella vulgaris. Algal Res. 2019, 44, 101707. [Google Scholar] [CrossRef]
- Hammond, B.R.; Renzi, L.M. Carotenoids. Advances in Nutrition 2013, vol. 4, 474. [Google Scholar] [CrossRef] [PubMed]
- Semba, R.D.; Dagnelie, G. Are lutein and zeaxanthin conditionally essential nutrients for eye health? Med. Hypotheses 2003, 61, 465–472. [Google Scholar] [CrossRef]
- Yang, J.; et al. Lutein protected the retina from light induced retinal damage by inhibiting increasing oxidative stress and inflammation. J. Funct. Foods 2020, 73, 104107. [Google Scholar] [CrossRef]
- Bone, R.A.; Landrum, J.T.; Guerra, L.H.; Ruiz, C.A. Lutein and Zeaxanthin Dietary Supplements Raise Macular Pigment Density and Serum Concentrations of these Carotenoids in Humans. J. Nutr. 2003, 133, 992–998. [Google Scholar] [CrossRef] [PubMed]
- Caetano, P.A.; Nass, P.P.; Deprá, M.C.; Nascimento, T.C.D.; Jacob-Lopes, E.; Zepka, L.Q. Trade-Off Between Growth Regimes in Chlorella vulgaris: Impact on Carotenoid Production. Colorants 2024. [Google Scholar] [CrossRef]
- Gouveia, L.; Veloso, V.; Reis, A.; Fernandes, H.; Novais, J.; Empis, J. Evolution of pigment composition in Chlorella vulgaris. Bioresour. Technol. 1996, 57, 157–163. [Google Scholar] [CrossRef]
- Hynstova, V.; Sterbova, D.; Klejdus, B.; Hedbavny, J.; Huska, D.; Adam, V. Separation, identification and quantification of carotenoids and chlorophylls in dietary supplements containing Chlorella vulgaris and Spirulina platensis using High Performance Thin Layer Chromatography. J. Pharm. Biomed. Anal. 2018, 148, 108–118. [Google Scholar] [CrossRef]
- Hou, Y.; Wu, G. Nutritionally Essential Amino Acids. Advances in Nutrition 2018, 9, 849–851. [Google Scholar] [CrossRef]
- Wu, G. Amino Acids: Biochemistry and Nutrition, Second Edition. Amino Acids: Biochemistry and Nutrition, Second Edition 2021, 1–788. [Google Scholar] [CrossRef]
- Lorenzo, K.; et al. Bioactivity of Macronutrients from Chlorella in Physical Exercise. Nutrients 2023, 15, 2168. [Google Scholar] [CrossRef] [PubMed]
- Wells, M.L.; et al. Algae as nutritional and functional food sources: revisiting our understanding. J. Appl. Phycol. 2016, 29, 949. [Google Scholar] [CrossRef]
- Wu, G. Principles of Animal Nutrition. In Principles of Animal Nutrition; Jan 2017; pp. 1–772. [Google Scholar] [CrossRef]
- Sousa, S.A.; Esteves, A.F.; Salgado, E.M.; Pires, J.C.M. Enhancing urban wastewater treatment: Chlorella vulgaris performance in tertiary treatment and the impact of anaerobic digestate addition. Environ. Technol. Innov. 2024, 34, 103601. [Google Scholar] [CrossRef]
- FMT DSA SERIES | Bench Tio Fermenter, Pilot Scale Fermenter, Air Lift Bioreactor, Beer fermenter | FERMENTEC. Available online: http://fermentec.co.kr/eng/product/bench-top-fermenter/fmt-dsa-series/?ckattempt=2 (accessed on 12 May 2025).
- Oliveira, J.; Pardilhó, S.; Dias, J.M.; Pires, J.C.M. Microalgae to Bioenergy: Optimization of Aurantiochytrium sp. Saccharification. Biology (Basel) 2023, 12, 935. [Google Scholar] [CrossRef]
- Collos, Y.; Mornet, F.; Sciandra, A.; Waser, N.; Larson, A.; Harrison, P.J. An optical method for the rapid measurement of micromolar concentrations of nitrate in marine phytoplankton cultures. J. Appl. Phycol. 1999, vol. 11, 179–184. [Google Scholar] [CrossRef]
- Lee, B.; Park, S.Y.; Heo, Y.S.; Yea, S.S.; Kim, D.E. Efficient Colorimetric Assay of RNA Polymerase Activity Using Inorganic Pyrophosphatase and Ammonium Molybdate. Bull. Korean Chem. Soc. 2009, vol. 30, 2485–2488. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. PROTEIN MEASUREMENT WITH THE FOLIN PHENOL REAGENT*. Journal of Biological Chemistry vol. 193, 265–275, 1951. [CrossRef]
- Clément-Larosière, B.; et al. Carbon dioxide biofixation by Chlorella vulgaris at different CO2 concentrations and light intensities. Eng. Life Sci. 2014, 14, 509–519. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric Method for Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Van Wychen, S.; Laurens, L.M.L. Determination of Total Carbohydrates in Algal Biomass: Laboratory Analytical Procedure (LAP) (Revised). 2013. Available online: www.nrel.gov/publications.
- Pagels, F.; Pereira, R.N.; Vicente, A.A.; Guedes, A.C. Extraction of Pigments from Microalgae and Cyanobacteria—A Review on Current Methodologies. Applied Sciences 2021. [Google Scholar] [CrossRef]
- Cohen, S.A.; De Antonis, K.M. Applications of amino acid derivatization with 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate: Analysis of feed grains, intravenous solutions and glycoproteins. J. Chromatogr. A 1994, vol. 661(no. 1–2), 25–34. [Google Scholar] [CrossRef]
- Pagels, F. Effects of irradiance of red and blue:red LEDs on Scenedesmus obliquus M2-1 optimization of biomass and high added-value compounds. J. Appl. Phycol. 2021, 33, 1379–1388. [Google Scholar] [CrossRef]
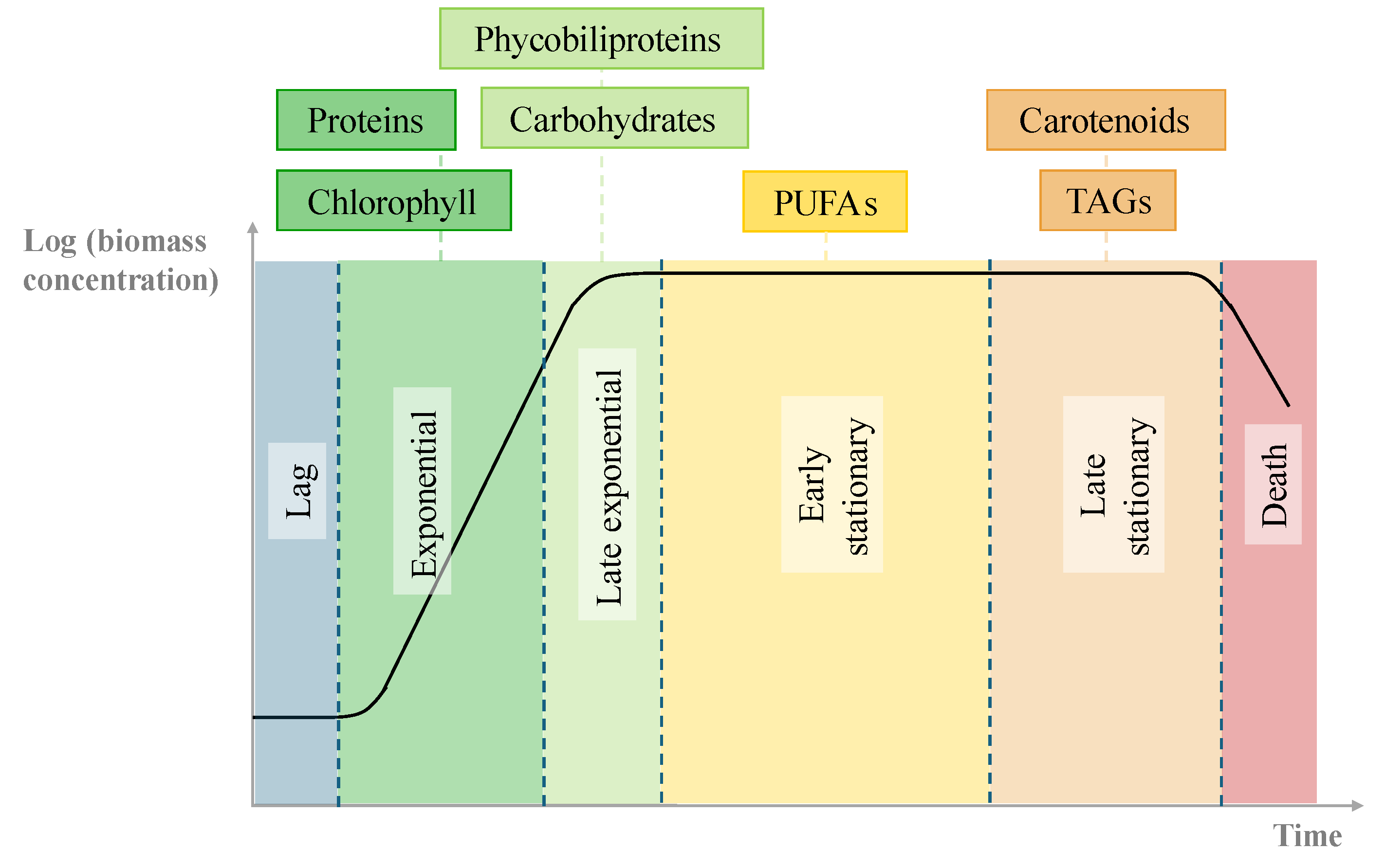
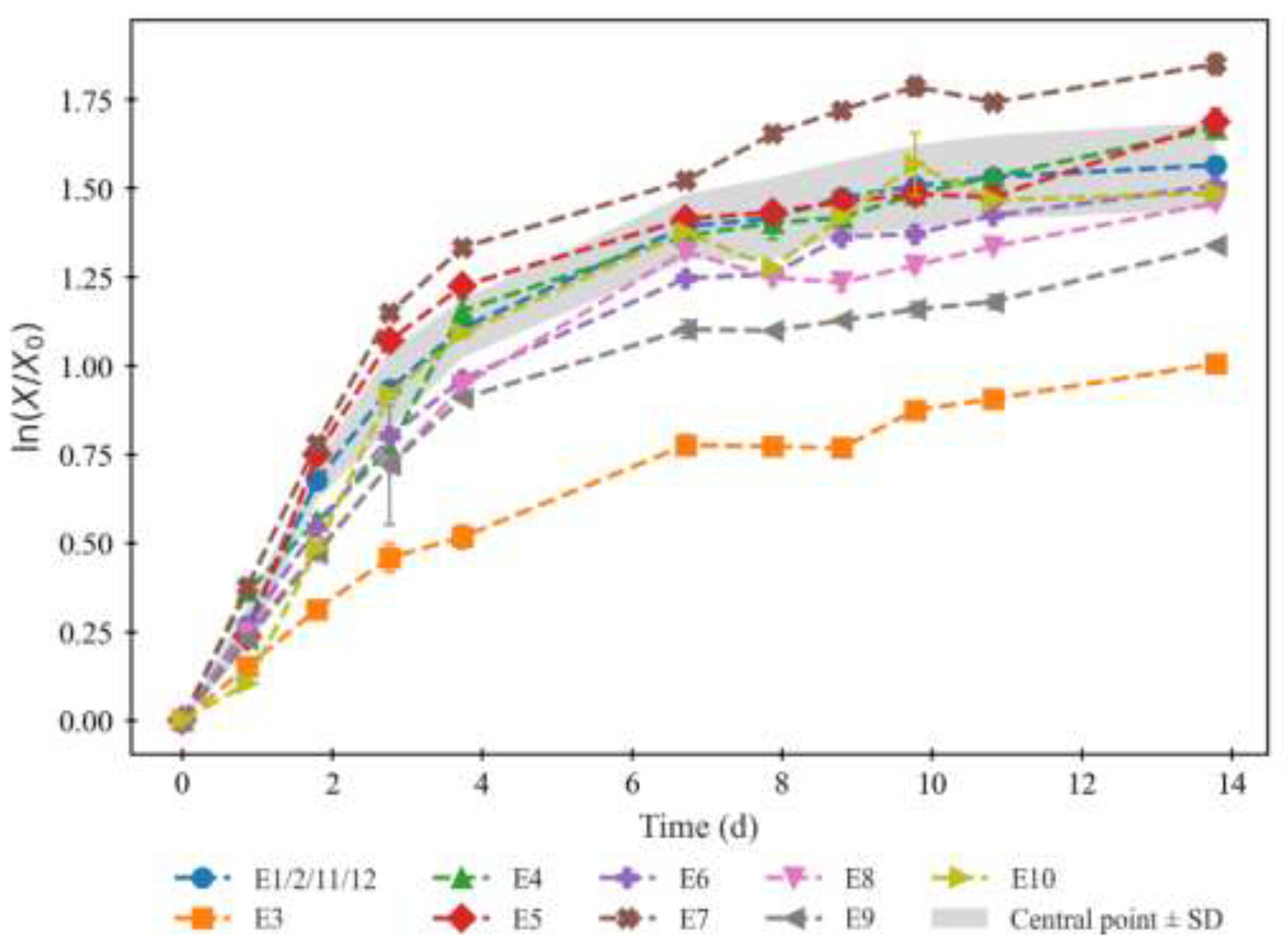
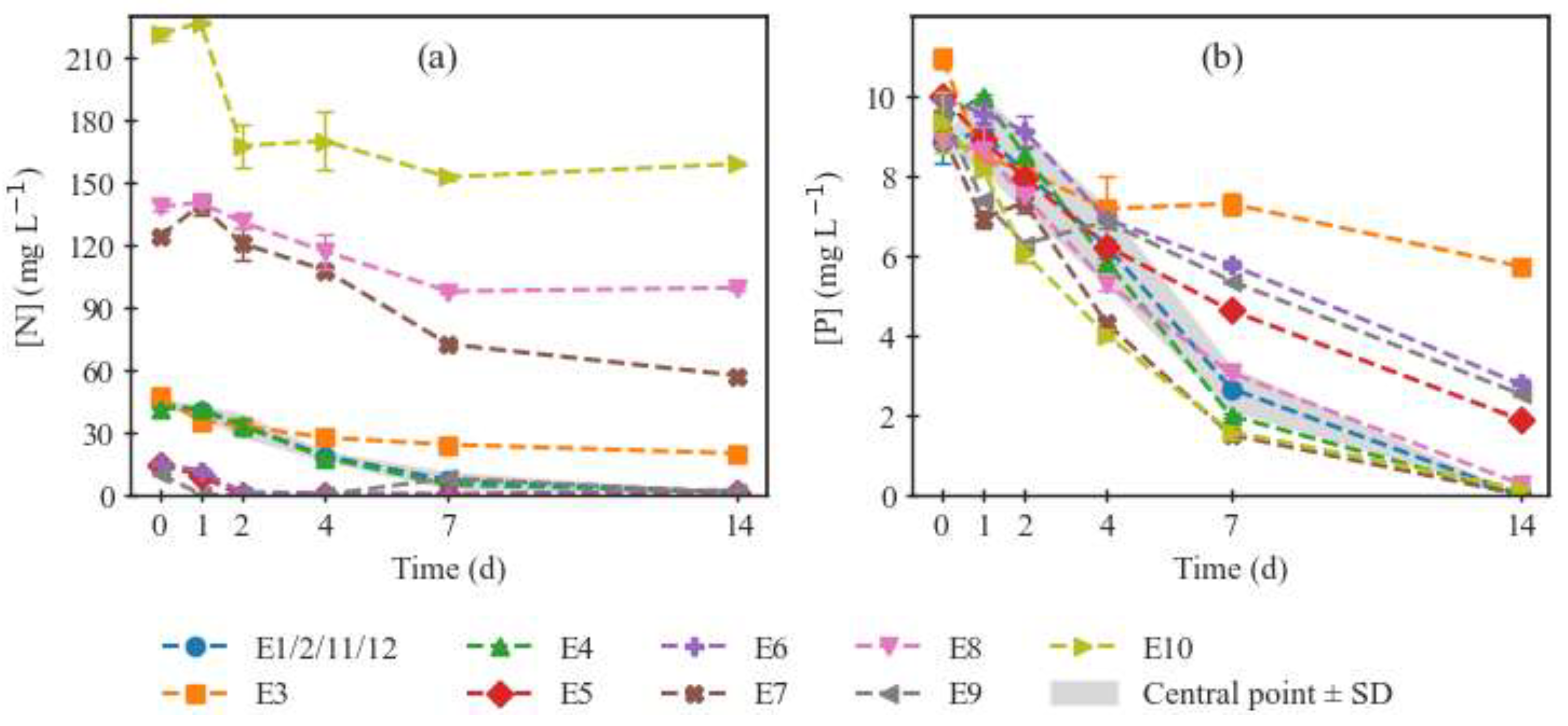
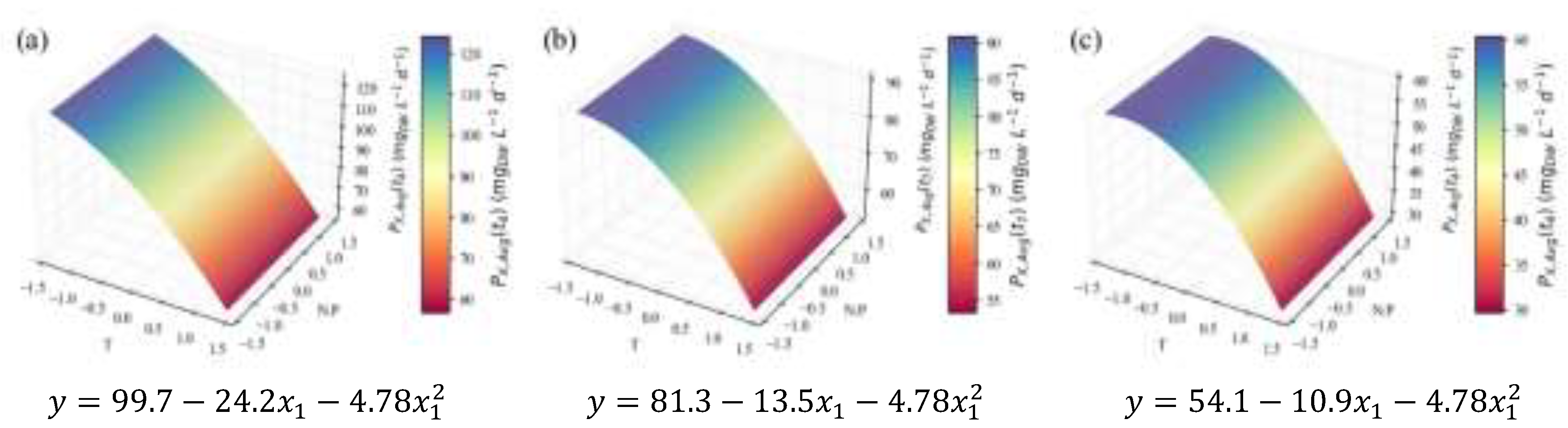
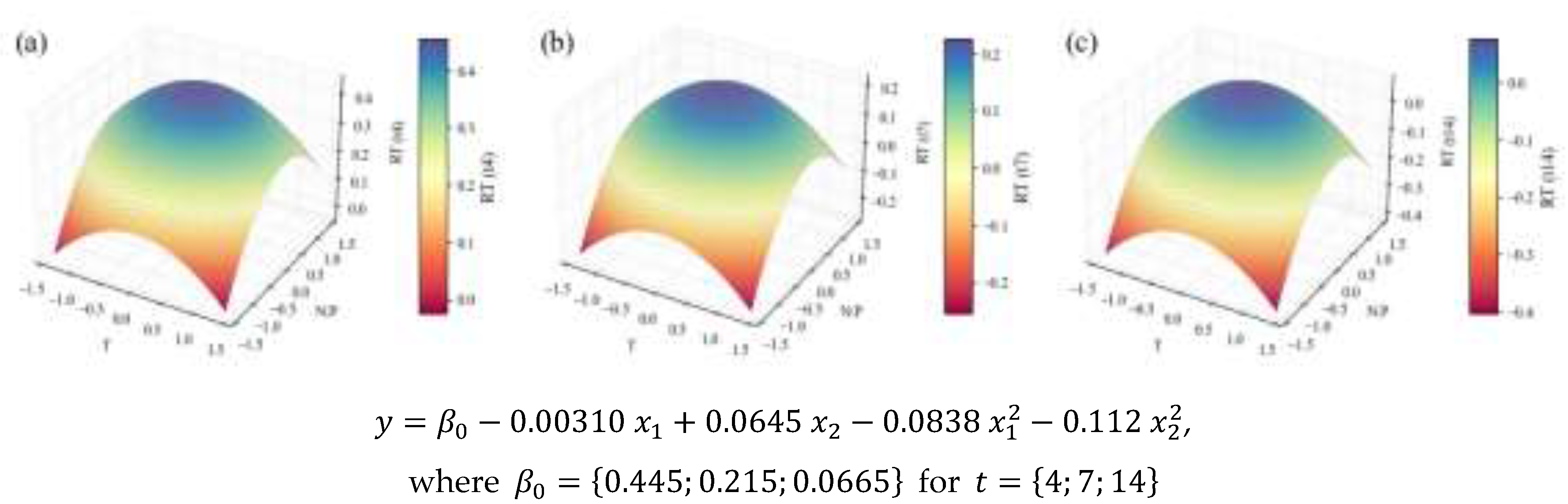
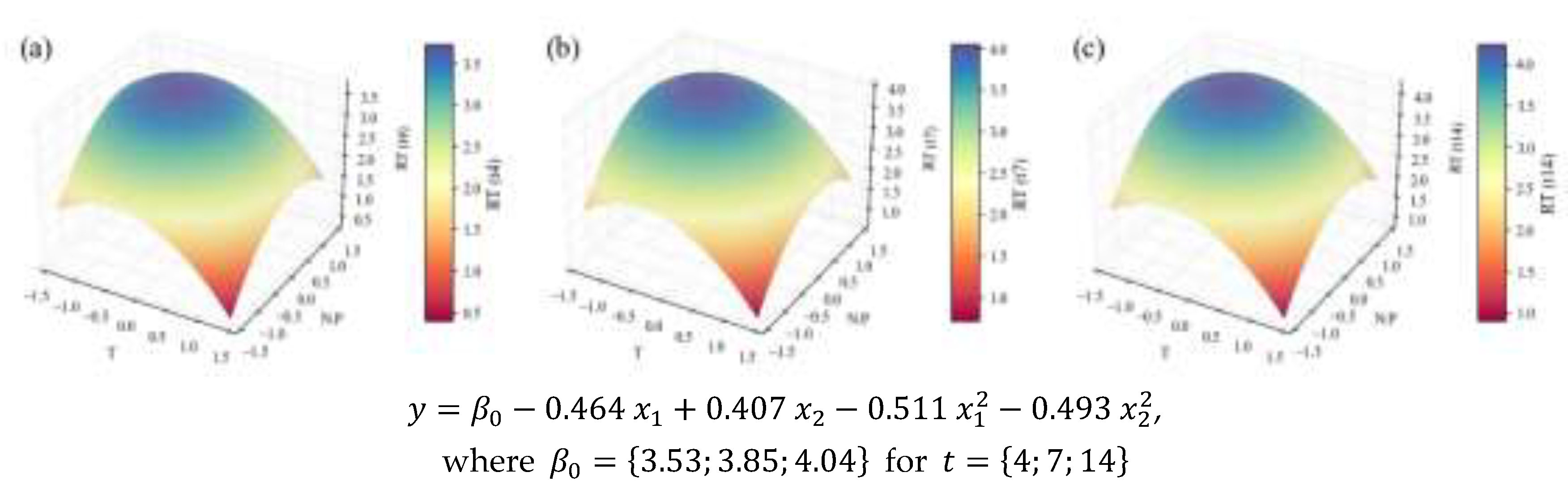
| Temperature | N:P ratio | |||||
|---|---|---|---|---|---|---|
| Experiment | Positioning in CCD | Level (x1) | Value (Z1) | Level (x2) | log3 (N:P) (Z2) | Value |
| E1 | Central (1st replica) | 0 | 25°C | 0 | 2 | 9 |
| E2 | Central (2nd replica) | 0 | 25°C | 0 | 2 | 9 |
| E3 | Axial (right) | +α | 32°C | 0 | 2 | 9 |
| E4 | Axial (left) | −α | 18°C | 0 | 2 | 9 |
| E5 | Factorial (bottom left) | −1 | 20°C | −1 | 1 | 3 |
| E6 | Factorial (bottom right) | +1 | 30°C | −1 | 1 | 3 |
| E7 | Factorial (upper left) | −1 | 20°C | +1 | 3 | 27 |
| E8 | Factorial (upper right) | +1 | 30°C | +1 | 3 | 27 |
| E9 | Axial (bottom) | 0 | 25°C | −α | 0.59 | 1.90 |
| E10 | Axial (upper) | 0 | 25°C | +α | 3.41 | 42.6 |
| E11 | Central (3rd replica) | 0 | 25°C | 0 | 2 | 9 |
| E12 | Central (4th replica) | 0 | 25°C | 0 | 2 | 9 |
| Growth and biomass productivity | Nutrient consumption | |||||||
|---|---|---|---|---|---|---|---|---|
| µ (d-1) | PX,avg (mgdw L-1 d-1) | NO3-N | Nitrogen | PO4-P | Phosphorus | |||||
| t0 → t4 | t0 → t7 | t0 → t14 | RE (%) |
RR
(mg L -1 d -1 ) |
RE (%) |
RR
(mg L -1 d -1 ) |
||
| E1 | 0.296 ± 0.004 | 99 ± 1 | 87.6 ± 0.5 | 54.7 ± 0.3 | 97.9 ± 0.2 | 3.17 ± 0.04 | 100.01 ± 0.05 | 0.76 ± 0.05 |
| E2 | 0.3343 ± 0.0001 | 107.2 ± 0.3 | 85.4 ± 0.3 | 52.5 ± 0.2 | 96.9 ± 0.3 | 3.14 ± 0.05 | 99.87 ± 0.09 | 0.76 ± 0.05 |
| E3 | 0.145 ± 0.002 | 46 ± 2 | 43 ± 1 | 31.2 ± 0.3 | 58 ± 1 | 1.94 ± 0.09 | 48 ± 2 | 0.37 ± 0.03 |
| E4 | 0.281 ± 0.005 | 119 ± 3 | 94 ± 4 | 68 ± 2 | 96.1 ± 0.1 | 2.87 ± 0.01 | 100.09 ± 0.03 | 0.67 ± 0.02 |
| E5 | 0.35 ± 0.001 | 117.7 ± 0.9 | 84 ± 0.3 | 58 ± 2 | 90 ± 2 | 0.96 ± 0.04 | 81.1 ± 0.4 | 0.579 ± 0.003 |
| E6 | 0.267 ± 0.003 | 77 ± 1 | 65.3 ± 0.5 | 45.5 ± 0.8 | 83.6 ± 0.2 | 0.91 ± 0.02 | 72 ± 1 | 0.51 ± 0.008 |
| E7 | 0.4234 ± 0.0005 | 122.3 ± 0.4 | 88 ± 2 | 64 ± 1 | 54 ± 2 | 4.7 ± 0.2 | 99.73 ± 0.1 | 0.64 ± 0.01 |
| E8 | 0.254 ± 0.001 | 72.4 ± 0.2 | 70.9 ± 0.4 | 41.4 ± 0.4 | 28 ± 2 | 2.8 ± 0.2 | 96.8 ± 0.1 | 0.614 ± 0.01 |
| E9 | 0.25 ± 0.04 | 93.6 ± 0.5 | 69 ± 2 | 47.1 ± 0.2 | 90.9 ± 0.2 | 0.671 ± 0.007 | 73.5 ± 0.4 | 0.503 ± 0.003 |
| E10 | 0.3407 ± 0.001 | 100.3 ± 1 | 83.4 ± 0.2 | 46.2 ± 0.6 | 28 ± 1 | 4.4 ± 0.3 | 98.4 ± 0.2 | 0.66 ± 0.05 |
| E11 | 0.3171 ± 0.0007 | 110.9 ± 0.4 | 92 ± 1 | 59 ± 3 | 97.74 ± 0.05 | 2.9 ± 0.04 | 100.2 ± 0.1 | 0.64 ± 0.02 |
| E12 | 0.266 ± 0.002 | 93 ± 1 | 75 ± 1 | 43 ± 1 | 97.1 ± 0.1 | 3.03 ± 0.08 | 100.1 ± 0.2 | 0.62 ± 0.04 |
| Model summary | Response optimization | |||||
|---|---|---|---|---|---|---|
| RMSE | y-value fit (mgdw L-1 d-1) | Setting | ||||
| 7.03 | 0.93 | 0.92 | 0.89 | 122.27 | † | t=4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





