Submitted:
09 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
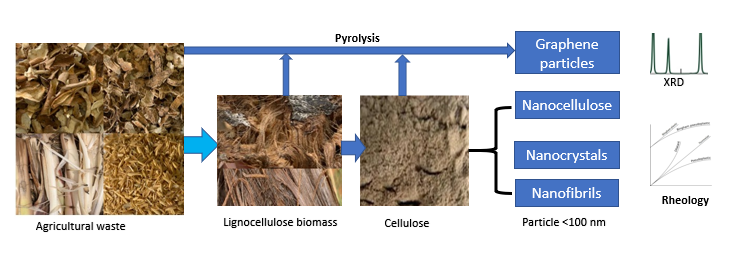
Keywords:
1. Introduction
2. Composition and Extraction of Lignocelluloses
2.1. Lignin Content
2.2. Hemicellulose Content
2.3. Cellulose Content
3. Green Methods for Extraction of Cellulose
3.1. Ionic Liquid Solvents
3.2. Enzymatic Process
4. Production of Nanoparticles
4.1. Carbonization
4.2. Biochar Activation
4.3. Production of Nanocellulose
4.4. Mechanical Milling
5. Characterization of Nanoparticles
5.1. Surface Charge
5.2. Particle Size and Shape
5.3. Surface Area
5.4. Morphologies of Nanocellulose
5.5. Crystallinity
5.6. Thermal Properties
| Nanoparticles | Source of Biomass | Technique | Thermal Properties | Reference |
|---|---|---|---|---|
| Cellulose | Eucalyptus saw dust | TGA, heat 20 - 600°C. instrument: STA 6000 (PerkinElmer, USA |
Tmax 367oC | Ferreira, R.R. et al. [65] |
| Nano cellulose | Eucalyptus saw dust | TGA, heat 20 - 600 °C. instrument: STA 6000 (PerkinElmer, USA) |
Tmaz 321-342o | Ferreira, R.R. et al. [65] |
| Nanocellulose | Cumin husk | TGA, 20 – 600oC, heat rate 20oC/min, STA 6000 (PerkinElmer, USA) |
Initial weight loss (50-150oC), Tmax (275–365oC), thermal decomposition (>400oC) | Hoseinpour, Z. et al. [66] |
| Extracted fibre | Bamboo | Thermal Analyzer, temp 40 - 700oC, Hitachi STA 7300, heating rate of 10oC/min. Sample weight (3-5 mg). | minor weight 5-7% (100-300o C), major weight loss 40-41% at peak decomposition (300-400oC), final weight loss 10-12% at final decomposition (>350oC) | Verma, Y.K. et al. [93] |
| Nanocellulose | Bamboo | Thermal Analyzer, temp 40 - 700oC, Hitachi STA 7300, heating rate of 10oC/min. Sample weight (3-5 mg). | minor weight 5-6% (100-300o C), major weight loss 40-41% at peak decomposition (300-400oC), final weight loss 10-15% at final decomposition (>350oC) | Verma, Y.K. et al. [93] |
| Cellulose | Cellulose paper | TGA, thermal Analyzer (DTG-60H, Shimadzu), temp 20 - 600oC, Heating rate 10oC/min | major degradation temp (370oC), weight loss (87%) and residue weight loss (6%) at 600oC. | Phanthong, P. et al. [94] |
| Nanocellulose | Cellulose paper | TGA, thermal Analyzer (DTG-60H, Shimadzu), temp 20 - 600oC, Heating rate 10oC/min | major degradation temp (200-420oC), weight loss (75%), max decomposition temp (340-350oC). residue weight loss (15%) at 600oC. | Phanthong, P. et al. [94] |
| Fibrous | Pineapple pee (hot, bleach and alkali treated) | TGA, thermal analyser (TGA550, TA Instruments, New Castle, DE, USA), temp 30 – 600o C, heating rate 10oC/min | Onset temp (220-240oC) and max temp (340-345oC), char residue (5-19%). | Wang, Y. et al. [60] |
| Cellulose nanofibrils | Pineapple peel | TGA, thermal analyser (TGA550, TA Instruments, New Castle, DE, USA), temp 30 – 600o C, heating rate 10oC/min | Onset temp (230-245oC) and max temp (316-330oC), char residue (14-17%). | Wang, Y. et al. [60] |
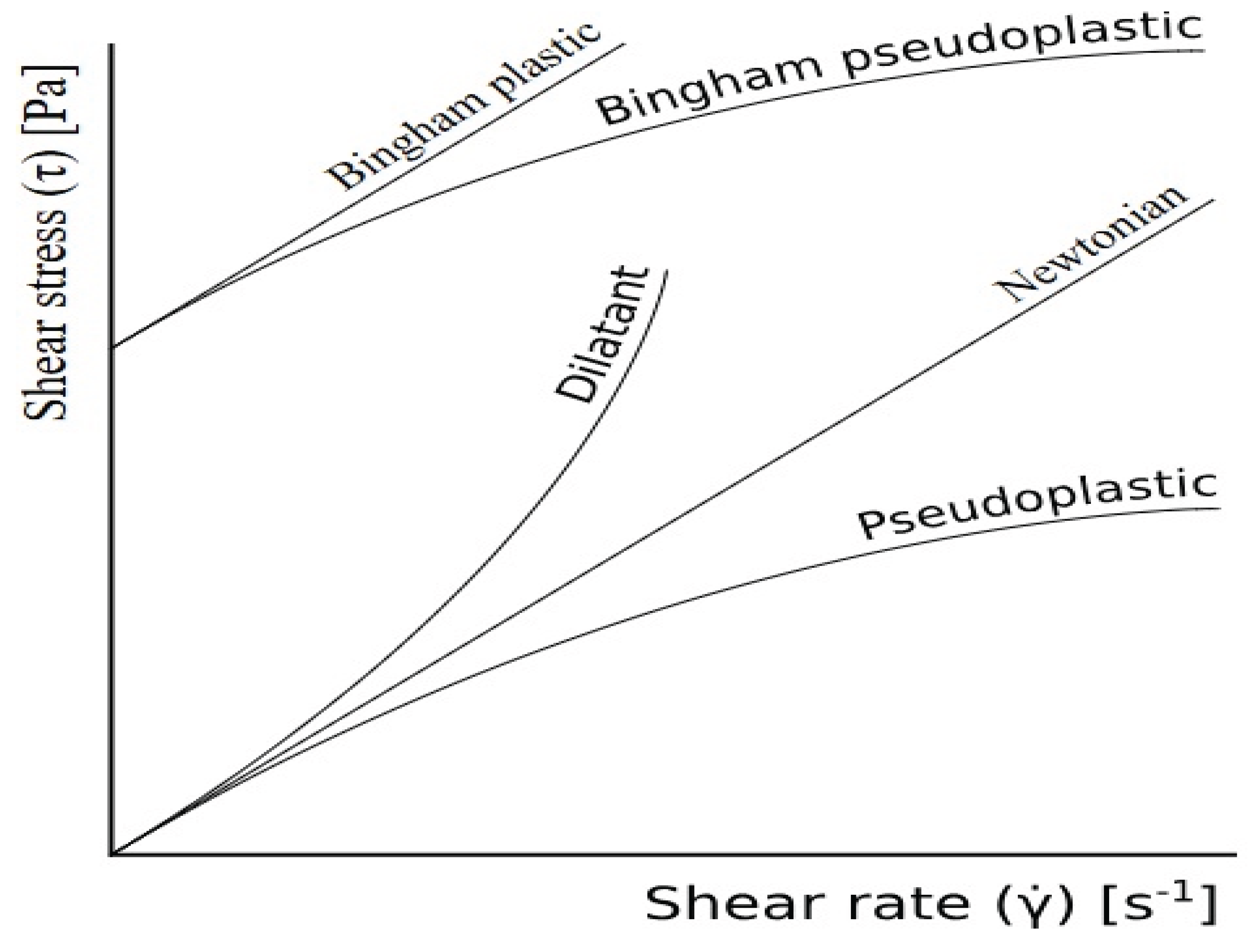
6. Rheology Behaviour of Nanocellulose
7. Application of Cellulose-Derived Nanoparticles
7.1. Packaging
7.2. Rheology Modifiers and Thickeners
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Imma, P. Bioactives from agricultural food by-products: application in food and health; 2017. [Google Scholar]
- Nargotra, P.; Yadav, A.; Sharma, D.; Tsai, M.-L.; Bajaj, B.K.; Sharma, V. Integrated technologies for agro-waste valorization, in Agriculture-Bioenergy Nexus; Elsevier, 2026; pp. 351–385. [Google Scholar]
- Mikus, M.; Galus, S. Biopolymers from agriculture waste and by-products, in Biopolymers: Recent Updates, Challenges and Opportunities; Springer, 2022; pp. 111–128. [Google Scholar]
- Phiri, R.; Rangappa, S.M.; Siengchin, S.; Oladijo, O.P.; Dhakal, H.N. Development of sustainable biopolymer-based composites for lightweight applications from agricultural waste biomass: A review . Adv. Ind. Eng. Polym. Res. 2023, 6, 436–450. [Google Scholar] [CrossRef]
- Choudhary, R.; Kumar, A.; Kumar, V.; Elmnifi, M.H.; Dhar, S.; Alqahtani, F.M.; Sharma, D.; Mahata, C.; Chaudhari, S.K.; Kumari, S. Case study on biopolymers derived from food and agro-industrial wastes, in Food and Agro-Industrial Wastes; Elsevier, 2026; pp. 439–454. [Google Scholar]
- Teli, M.; Jadhav, A.C. Extraction and characterization of novel lignocellulosic fibre . J. Bionanosci. 2016, 10, 418–423. [Google Scholar] [CrossRef]
- Rencoret, J.; Marques, G.; Rosado, M.J.; Benito, J.; Barro, F.; Gutiérrez, A.; Del Rio, J.C. Variations in the composition and structure of the lignins of oat (Avena sativa L.) straws according to variety and planting season. Int. J. Biol. Macromol. 2023, 242, 124811. [Google Scholar] [CrossRef] [PubMed]
- Lama-Muñoz, A.; del Mar Contreras, M.; Espínola, F.; Moya, M.; Romero, I.; Castro, E. Characterization of the lignocellulosic and sugars composition of different olive leaves cultivars. Food Chem. 2020, 329, 127153. [Google Scholar] [CrossRef]
- De, S.; Mishra, S.; Poonguzhali, E.; Rajesh, M.; Tamilarasan, K. Fractionation and characterization of lignin from waste rice straw: Biomass surface chemical composition analysis. Int. J. Biol. Macromol. 2020, 145, 795–803. [Google Scholar] [CrossRef]
- Bhat, R.; Ahmad, A.; Jõudu, I. Applications of lignin in the agri-food industry, in Lignin: Biosynthesis and transformation for industrial applications; Springer, 2020; pp. 275–298. [Google Scholar]
- Vasile, C.; Baican, M. Lignins as promising renewable biopolymers and bioactive compounds for high-performance materials. Polymers. 2023, 15, 3177. [Google Scholar] [CrossRef]
- Gautam, A.; Kumar, A.; Bharti, A.K.; Dutt, D. Rice straw fermentation by Schizophyllum commune ARC-11 to produce high level of xylanase for its application in pre-bleaching. J. Genet. Eng. & Biotechnol. 2018, 16, 693–701. [Google Scholar]
- Wu, W.; Li, P.; Huang, L.; Wei, Y.; Li, J.; Zhang, L.; Jin, Y. The role of lignin structure on cellulase adsorption and enzymatic hydrolysis. Biomass 2023, 3, 96–107. [Google Scholar] [CrossRef]
- Xu, R.; Doskaliuk, N.; Pang, B.; Xu, J.; Xu, W.; Xu, C.; Antonietti, M.; Filonenko, S. Hemicellulose from mild extraction of biomass: Revealing structural insights and advancing potential value. Carbohydr. Polym. Technol. Appl. 2025, 100843. [Google Scholar] [CrossRef]
- Zhao, Y.; Sun, H.; Yang, B.; Weng, Y. Hemicellulose-based film: potential green films for food packaging. Polymers. 2020, 12, 1775. [Google Scholar] [CrossRef]
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Biochemistry (loose-leaf); Macmillan, 2007. [Google Scholar]
- Heinze, T. Cellulose: structure and properties, in Cellulose chemistry and properties: fibers, nanocelluloses and advanced materials; Springer, 2015; pp. 1–52. [Google Scholar]
- Tomisawa, R.; Nagata, M.; Otsuka, Y.; Ikaga, T.; Kim, K.; Ohkoshi, Y.; Okada, K.; Kanaya, T.; Katsuta, H. Tensile strength of polyester fiber estimated by molecular-chain extension prior to structure formation. Sci. Rep. 2023, 13, 11759. [Google Scholar] [CrossRef] [PubMed]
- Glińska, K.; Gitalt, J.; Torrens, E.; Plechkova, N.; Bengoa, C. Extraction of cellulose from corn stover using designed ionic liquids with improved reusing capabilities. Process Saf. Environ. Prot. 2021, 147, 181–191. [Google Scholar] [CrossRef]
- Freixo, R.; Casanova, F.; Ribeiro, A.B.; Pereira, C.F.; Costa, E.M.; Pintado, M.E.; Ramos, Ó.L. Extraction methods and characterization of cellulose fractions from a sugarcane by-product for potential industry applications. Ind. Crop. Prod. 2023, 197, 116615. [Google Scholar] [CrossRef]
- Foroushani, M.Y.; Foroushani, A.Y.; Yarahmadi, H. Analysis of mechanical techniques in extracting cellulose fibers from sugarcane bagasse. Biomass Convers. Biorefin. 2025, 1–14. [Google Scholar] [CrossRef]
- Rizwan, M.; Gilani, S.R.; Durrani, A.I.; Naseem, S. Cellulose extraction of Alstonia scholaris: A comparative study on efficiency of different bleaching reagents for its isolation and characterization. Int. J. Biol. Macromol. 2021, 191, 964–972. [Google Scholar] [CrossRef]
- Benali, M.; Oulmekki, A.; Toyir, J. The impact of the alkali-bleaching treatment on the isolation of natural cellulosic fibers from Juncus Effesus L plant. Fiber Polym. 2024, 25, 525–533. [Google Scholar] [CrossRef]
- Zhang, M.; Song, X.; Sun, X.; Wang, Z.; Li, Z.; Ji, H.; Xu, X.; Li, J. The relationship between cellulose content and the contents of sugars and minerals during fiber development in colored cotton cultivars. Cellul. 2012, 19, 2003–2014. [Google Scholar] [CrossRef]
- Djahedi, C.; Berglund, L.A.; Wohlert, J. Molecular deformation mechanisms in cellulose allomorphs and the role of hydrogen bonds. Carbohydr. Polym. 2015, 130, 175–182. [Google Scholar] [CrossRef]
- Baghaei, B.; Skrifvars, M. All-cellulose composites: a review of recent studies on structure, properties and applications. MOL. 2020, 25, 2836. [Google Scholar] [CrossRef]
- Sousa, L.d.C.; Humpula, J.; Balan, V.; Dale, B.E.; Chundawat, S.P. Impact of ammonia pretreatment conditions on the cellulose III allomorph ultrastructure and its enzymatic digestibility. ACS Sustain. Chem. Eng. 2019, 7, 14411–14424. [Google Scholar] [CrossRef]
- Chen, J. Synthetic textile fibers: regenerated cellulose fibers, in Textiles and fashion; Elsevier, 2015; pp. 79–95. [Google Scholar]
- Swatloski, R.P.; Spear, S.K.; Holbrey, J.D.; Rogers, R.D. Dissolution of cellose with ionic liquids. J. Am. Chem. Soc. 2002, 124, 4974–4975. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.; Yu, X.; Ji, Q.; Chen, L.; Yagoub, A.E.-G.; Olugbenga, F.; Zhou, C. Preparation and characterization of lignin-containing cellulose nanocrystals from peanut shells using a deep eutectic solvent containing lignin-derived phenol. Ind. Crop. Prod. 2023, 195, 116415. [Google Scholar] [CrossRef]
- Zhang, X.; Bao, D.; Huang, Y.; Dong, H.; Zhang, X.; Zhang, S. Gas–liquid mass-transfer properties in CO2 absorption system with ionic liquids. Am. Inst. Chem. Eng. 2014, 60, 2929–2939. [Google Scholar] [CrossRef]
- Andlar, M.; Rezić, T.; Marđetko, N.; Kracher, D.; Ludwig, R.; Šantek, B. Lignocellulose degradation: An overview of fungi and fungal enzymes involved in lignocellulose degradation. Eng. Life Sci. 2018, 18, 768–778. [Google Scholar] [CrossRef]
- Kumar, A.; Chandra, R. Ligninolytic enzymes and its mechanisms for degradation of lignocellulosic waste in environment. Heliyon. 2020, 6. [Google Scholar] [CrossRef]
- Radotić, K.; Mićić, M. Methods for extraction and purification of lignin and cellulose from plant tissues, in Sample preparation techniques for soil, plant, and animal samples; Springer, 2016; pp. 365–376. [Google Scholar]
- Gonzalez-Gonzalez, M.d.R.; Miranda-Lopez, R. Cellulases, hemicellulases and ligninolytic enzymes: mechanism of action, optimal processing conditions and obtaining value-added compounds in plant matrices. MOJ food process. technol. 2022, 10, 30–37. [Google Scholar]
- Sigma-Aldrich®. Ilya Koltover, Ph. D. Aldrich® Materials Science Sigma-Aldrich® Corporation.
- Zhang, J.; Li, P.; Yu, Y.; Xu, Y.; Jia, W.; Zhao, S. A review of natural polysaccharides-based flocculants derived from waste: application efficiency, function mechanism, and development prospects. Ind. Eng. Chem. Res. 2023, 62, 15774–15789. [Google Scholar] [CrossRef]
- Zhao, J.; Wu, X.; Yuan, X.; Yang, X.; Guo, H.; Yao, W.; Ji, D.; Li, X.; Zhang, L. Nanocellulose and cellulose making with bio-enzymes from different particle sizes of neosinocalamus affinis. Coatings. 2022, 12, 1734. [Google Scholar] [CrossRef]
- Himmel, M.E.; Baker, J.O.; Adney, W.S.; Decker, S.R. Cellulases, hemicellulases, and pectinases. MOJ food process. technol. 2007, 596–610. [Google Scholar]
- Fenila, F.; Shastri, Y. Optimal control of enzymatic hydrolysis of lignocellulosic biomass. Resour.-Effic. Technol. 2016, 2, S96–S104. [Google Scholar]
- Jiang, H.; Nie, J.; Zeng, L.; Zhu, F.; Gao, Z.; Zhang, A.; Xie, J.; Chen, Y. Selective Removal of Hemicellulose by Diluted Sulfuric Acid Assisted by Aluminum Sulfate. MOL. 2024, 29, 2027. [Google Scholar] [CrossRef]
- Yuan, X.; Zhao, J.; Wu, X.; Yao, W.; Guo, H.; Ji, D.; Yu, Q.; Luo, L.; Li, X.; Zhang, L. Extraction of corn bract cellulose by the ammonia-coordinated bio-enzymatic method. Polymers. 2022, 15, 206. [Google Scholar] [CrossRef] [PubMed]
- Ren, Q.; Zhao, C.; Chen, X.; Duan, L.; Li, Y.; Ma, C. NOx and N2O precursors (NH3 and HCN) from biomass pyrolysis: Co-pyrolysis of amino acids and cellulose, hemicellulose and lignin. Proc. Combust. Inst. 2011, 33, 1715–1722. [Google Scholar] [CrossRef]
- Mahasweta, L. Thermochemical Conversion of Biomass to Energy: The Pyrolysis Pathway, in The Principles of Green Energy and Technology. CRC Press, 2025; Volume 2, pp. 89–106. [Google Scholar]
- Pusceddu, E.; Montanaro, A.; Fioravanti, G.; Santilli, S.; Foscolo, P.; Criscuoli, I.; Raschi, A.; Miglietta, F. Comparison between ancient and fresh biochar samples, a study on the recalcitrance of carbonaceous structures during soil incubation. Int. J. New Technol. Res 2017, 3, 39–46. [Google Scholar]
- Moradi-Choghamarani, F.; Moosavi, A.A.; Baghernejad, M. Determining organo-chemical composition of sugarcane bagasse-derived biochar as a function of pyrolysis temperature using proximate and Fourier transform infrared analyses. J. Therm. Anal. Calorim. 2019, 138, 331–342. [Google Scholar] [CrossRef]
- Aboelela, D.; Saleh, H.; Attia, A.M.; Elhenawy, Y.; Majozi, T.; Bassyouni, M. Recent advances in biomass pyrolysis processes for bioenergy production: optimization of operating conditions. Sustainability. 2023, 15, 1–30. [Google Scholar] [CrossRef]
- Duman, G.; Yanik, J. Two-step steam pyrolysis of biomass for hydrogen production. Int. J. Hydrog. Energy. 2017, 42, 17000–17008. [Google Scholar] [CrossRef]
- Pütün, A.; Özbay, N.; Pütün, E. Effect of steam on the pyrolysis of biomass. Energ. Sources Part A. 2006, 28, 253–262. [Google Scholar] [CrossRef]
- Aro-Modiu, O.; Osobamiro, M.; Osundeko, A. Synthesis of graphene oxide from agricultural waste. Sci. Afr. 2019, 18, 143–151. [Google Scholar]
- Sukoyo, A.; Djoyowasito, G.; Wibisono, Y. Unravelling the potency of activated carbon powder derived from cultivated marine microalgae as a promising filler in mixed matrix membranes. J. Agric. Eng. 2019, 1, 188–204. [Google Scholar] [CrossRef]
- Anthonysamy, S.; Lahijani, P.; Mohammadi, M.; Mohamed, A. Alkali-modified biochar as a sustainable adsorbent for the low-temperature uptake of nitric oxide. Int. J. Environ. Sci. Technol. 2022, 19, 7127–7140. [Google Scholar] [CrossRef]
- Baral, K.R.; McIlroy, J.; Lyons, G.; Johnston, C. The effect of biochar and acid activated biochar on ammonia emissions during manure storage. Environ. Pollut. 2023, 317, 120815. [Google Scholar] [CrossRef] [PubMed]
- Iriarte-Velasco, U.; Sierra, I.; Zudaire, L.; Ayastuy, J.L. Preparation of a porous biochar from the acid activation of pork bones. Food Bioprod. Process. 2016, 98, 341–353. [Google Scholar] [CrossRef]
- Kreuder, A.D.; House-Knight, T.; Whitford, J.; Ponnusamy, E.; Miller, P.; Jesse, N.; Rodenborn, R.; Sayag, S.; Gebel, M.; Aped, I. A method for assessing greener alternatives between chemical products following the 12 principles of green chemistry. ACS Sustain. Chem. Eng. 2017, 5, 2927–2935. [Google Scholar] [CrossRef]
- Lonappan, L.; Liu, Y.; Rouissi, T.; Brar, S.K.; Surampalli, R.Y. Development of biochar-based green functional materials using organic acids for environmental applications. J. Clean. Prod. 2020, 244, 118841. [Google Scholar] [CrossRef]
- Eslami, E.; Carpentieri, S.; Pataro, G.; Ferrari, G. A comprehensive overview of tomato processing by-product valorization by conventional methods versus emerging technologies. Foods. 2022, 12, 166. [Google Scholar] [CrossRef]
- Moskovskikh, D. Overview of Planetary Ball Milling Strategies for Tailoring Composite Powder Morphology and Reactivity. Int. J. Self-Propagating High-Temperature Synth. 2025, 34, 330–341. [Google Scholar] [CrossRef]
- Naghdi, M.; Taheran, M.; Brar, S.K.; Rouissi, T.; Verma, M.; Surampalli, R.Y.; Valero, J.R. A green method for production of nanobiochar by ball milling-optimization and characterization. J. Clean. Prod. 2017, 164, 1394–1405. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, Y.; Hu, S.; Yu, J.; Huang, Y.; Dai, H. Preparation of Agrowaste-Based Nanocellulose by NaOH-Assisted Ball Milling Technique: Influence of Component Intervention. Gels. 2025, 11, 631. [Google Scholar] [CrossRef]
- Phanthong, P.; Guan, G.; Ma, Y.; Hao, X.; Abudula, A. Effect of ball milling on the production of nanocellulose using mild acid hydrolysis method. J. Taiwan Inst. Chem. Eng. 2016, 60, 617–622. [Google Scholar] [CrossRef]
- dos Santos, D.F.; Ferreira, R.R.; Souza, A.G.; Lenhani, G.C.; Deon, V.G.; Rosa, D.S.; Pinto, V.Z. UPCYCLING CORN CROPPING WASTE BY NANOCELLULOSE PRODUCTION. Biocatal Agric Biotechnol. 2025, 103624. [Google Scholar] [CrossRef]
- Júnior, J.A.A.; Baldo, J.B. The behavior of zeta potential of silica suspensions. New J. Glass Ceram. 2014, 4, 29. [Google Scholar] [CrossRef]
- Asha, A.B.; Narain, R. Nanomaterials properties, in Polymer science and nanotechnology; Elsevier, 2020; pp. 343–359. [Google Scholar]
- Ferreira, R.R.; Souza, A.G.; Nunes, L.L.; Shahi, N.; Rangari, V.K.; dos Santos Rosa, D. Use of ball mill to prepare nanocellulose from eucalyptus biomass: challenges and process optimization by combined method. Mater. Today Commun. 2020, 22, 100755. [Google Scholar] [CrossRef]
- Hoseinpour, Z.; Niazmand, R.; Heydari-Majd, M. Extraction and characterization of nanocellulose from Cuminum cyminum L. husk by ball-milling-assisted ultrasound. Carbohydr. Polym. Technol. Appl. 2025, 100934. [Google Scholar] [CrossRef]
- Nakatuka, Y.; Yoshida, H.; Fukui, K.; Matuzawa, M. The effect of particle size distribution on effective zeta-potential by use of the sedimentation method. Adv. Powder Technol. 2015, 26, 650–656. [Google Scholar] [CrossRef]
- Saleh, T.A. Nanomaterials: Classification, properties, and environmental toxicities. Environ. Technol. Innov. 2020, 20, 101067. [Google Scholar] [CrossRef]
- Serrano-Lotina, A.; Portela, R.; Baeza, P.; Alcolea-Rodríguez, V.; Villarroel, M.; Ávila, P. Zeta potential as a tool for functional materials development. Catal. Today. 2023, 423, 113862. [Google Scholar] [CrossRef]
- Ali, W.A.; Richards, S.E.; Alzard, R.H. Unlocking the potential of ball milling for nanomaterial Synthesis: An overview. J. Ind. Eng. Chem. 2025. [Google Scholar] [CrossRef]
- Yadav, T.P.; Yadav, R.M.; Singh, D.P. Mechanical milling: a top down approach for the synthesis of nanomaterials and nanocomposites. J. Nanosci. Nanotechnol. 2012, 2, 22–48. [Google Scholar] [CrossRef]
- Du Plessis, M. Relationship between specific surface area and pore dimension of high porosity nanoporous silicon–model and experiment. Phys. Status Solidi A. 2007, 204, 2319–2328. [Google Scholar] [CrossRef]
- Tessema, B.; Gonfa, G.; Hailegiorgis, S. Mekuria; Prabhu, S. Venkatesa. An overview of current and prognostic trends on synthesis, characterization, and applications of biobased silica. Adv. mater. sci. eng. 2023, 4865273. [Google Scholar] [CrossRef]
- Liu, Z.; Li, J.; Wang, Q.; Lu, X.; Zhang, Y.; Zhu, M.; Zhang, Z.; Zhang, D. An experimental investigation into mineral transformation, particle agglomeration and ash deposition during combustion of Zhundong lignite in a laboratory-scale circulating fluidized bed. Fuel. 2019, 243, 458–468. [Google Scholar] [CrossRef]
- Antony Jose, S.; Cowan, N.; Davidson, M.; Godina, G.; Smith, I.; Xin, J.; Menezes, P.L. A comprehensive review on cellulose Nanofibers, nanomaterials, and composites: Manufacturing, properties, and applications. Nanomaterials. 2025, 15, 356. [Google Scholar] [CrossRef] [PubMed]
- Kondor, A.; Santmarti, A.; Mautner, A.; Williams, D.; Bismarck, A.; Lee, K.-Y. On the BET surface area of nanocellulose determined using volumetric, gravimetric and chromatographic adsorption methods. Front. Chem. Eng. 2021, 3, 738995. [Google Scholar] [CrossRef]
- Osorio, M.; Fernández-Morales, P.; Gañán, P.; Zuluaga, R.; Kerguelen, H.; Ortiz, I.; Castro, C. Development of novel three-dimensional scaffolds based on bacterial nanocellulose for tissue engineering and regenerative medicine: Effect of processing methods, pore size, and surface area. J. Biomed. Mater. Res. A. 2019, 107, 348–359. [Google Scholar] [CrossRef]
- Krishnaiah, P.; Ratnam, C.T.; Manickam, S. Enhancements in crystallinity, thermal stability, tensile modulus and strength of sisal fibres and their PP composites induced by the synergistic effects of alkali and high intensity ultrasound (HIU) treatments. Ultrason. Sonochem. 2017, 34, 729–742. [Google Scholar] [CrossRef]
- Rana, M.S.; Rahim, M.A.; Mosharraf, M.P.; Tipu, M.F.K.; Chowdhury, J.A.; Haque, M.R.; Kabir, S.; Amran, M.S.; Chowdhury, A.A. Morphological, spectroscopic and thermal analysis of cellulose nanocrystals extracted from waste jute fiber by acid hydrolysis. Polymers. 2023, 15, 1530. [Google Scholar] [CrossRef]
- Moon, R.; Johnston, L.; Land-Hensdal, C.; Batchelor, W. Perspectives on cellulose nanofibril size measurement using scanning electron microscopy. Cellul. 2025, 1–18. [Google Scholar] [CrossRef]
- Mehanny, S.; Abu-El Magd, E.E.; Ibrahim, M.; Farag, M.; Gil-San-Millan, R.; Navarro, J.; El Habbak, A.E.H.; El-Kashif, E. Extraction and characterization of nanocellulose from three types of palm residues. J. Mater. Res. Technol. 2021, 10, 526–537. [Google Scholar] [CrossRef]
- Schroeder, A.B.; Dobson, E.T.; Rueden, C.T.; Tomancak, P.; Jug, F.; Eliceiri, K.W. The ImageJ ecosystem: Open-source software for image visualization, processing, and analysis. Protein Sci. 2021, 30, 234–249. [Google Scholar] [CrossRef]
- Hotaling, N.A.; Bharti, K.; Kriel, H.; Simon, C.G., Jr. DiameterJ: A validated open source nanofiber diameter measurement tool. Biomaterials. 2015, 61, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Ho, H.V.; Makkar, P.; Padalhin, A.R.; Le, T.T.T.; Lee, S.Y.; Jaegyoung, G.; Lee, B.-T. Preliminary studies on the in vivo performance of various kinds of nanocellulose for biomedical applications. J. Biomater. Appl. 2020, 34, 942–951. [Google Scholar] [CrossRef] [PubMed]
- Tarrés, Q.; Aguado, R.; Zoppe, J.O.; Mutjé, P.; Fiol, N.; Delgado-Aguilar, M. Dynamic light scattering plus scanning electron microscopy: usefulness and limitations of a simplified estimation of nanocellulose dimensions. Nanomaterials. 2022, 12, 4288. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, B.; Li, H.; Yang, W.; Ding, Y.; Sinogeikin, S.V.; Meng, Y.; Liu, Z.; Zeng, X.C.; Mao, W.L. Long-range ordered carbon clusters: a crystalline material with amorphous building blocks. J. Sci. 2012, 337, 825–828. [Google Scholar] [CrossRef]
- Du, H.; Liu, K.; Xu, T.; Xu, C.; Lin, M.; Fang, Z.; Kim, S.-W.; Seo, J.-Y.; Chen, J.; Ma, H. Lignocellulosic Films: Preparation, Properties, and Applications. Chem. Rev. 2025. [Google Scholar] [CrossRef]
- Santmartí, A.; Lee, K.-Y. Crystallinity and thermal stability of nanocellulose, in Nanocellulose and sustainability; CRC Press, 2018; pp. 67–86. [Google Scholar]
- Nam, S.; Liu, Y.; He, Z.; Hinchliffe, D.J.; Fang, D. Assessment of Segal method for identifying crystallinity evolution in developing cotton fibers. Agric. environ. lett. 2024, 9, e20138. [Google Scholar] [CrossRef]
- Rashid, S.; Dutta, H. Characterization of nanocellulose extracted from short, medium and long grain rice husks. Ind. Crop. Prod. 2020, 154, 112627. [Google Scholar] [CrossRef]
- Raza, M.; Abu-Jdayil, B.; Banat, F.; Al-Marzouqi, A.H. Isolation and characterization of cellulose nanocrystals from date palm waste. ACS Omega. 2022, 7, 25366–25379. [Google Scholar] [CrossRef]
- Seifert, E. OriginPro 9.1: Scientific data analysis and graphing software software review; ACS Publications, 2014. [Google Scholar]
- Verma, Y.K.; Singh, A.K.; Paswan, M.; Gurmaita, P.K. Preparation and characterization of bamboo based nanocellulose by ball milling and used as a filler for preparation of nanocomposite. Polymer. 2024, 308, 127396. [Google Scholar] [CrossRef]
- Phanthong, P.; Karnjanakom, S.; Reubroycharoen, P.; Hao, X.; Abudula, A.; Guan, G. A facile one-step way for extraction of nanocellulose with high yield by ball milling with ionic liquid. Cellul. 2017, 24, 2083–2093. [Google Scholar] [CrossRef]
- Lichtenstein, K.; Lavoine, N. Toward a deeper understanding of the thermal degradation mechanism of nanocellulose. Polym. Degrad. Stabil. 2017, 146, 53–60. [Google Scholar] [CrossRef]
- Ballesteros-Mártinez, L.; Pérez-Cervera, C.; Andrade-Pizarro, R. Effect of glycerol and sorbitol concentrations on mechanical, optical, and barrier properties of sweet potato starch film. NFS J. 2020, 20, 1–9. [Google Scholar] [CrossRef]
- Bonifacio, A.; Bonetti, L.; Piantanida, E.; De Nardo, L. Plasticizer design strategies enabling advanced applications of cellulose acetate. Eur. Polym. J. 2023, 197, 112360. [Google Scholar] [CrossRef]
- Sanyang, M.L.; Sapuan, S.M.; Jawaid, M.; Ishak, M.R.; Sahari, J. Effect of plasticizer type and concentration on tensile, thermal and barrier properties of biodegradable films based on sugar palm (Arenga pinnata) starch. Polymers. 2015, 7, 1106–1124. [Google Scholar] [CrossRef]
- Shin, S. Rheological Design and Applications of Cellulose Nanofiber in Additive Manufacturing. J. of Korea TAPPI. 2025, 57, 19–31. [Google Scholar] [CrossRef]
- Ospennikov, A.S.; Kwiatkowski, A.L.; Philippova, O.E. Cellulose nanofibrils vs nanocrystals: Rheology of suspensions and hydrogels. Gels. 2025, 11, 926. [Google Scholar] [CrossRef]
- Ait Balla, M.; Maazouz, A.; Lamnawar, K.; Arrakhiz, F.E. Biowastes as Reinforcements for Sustainable PLA-Biobased Composites Designed for 3D Printing Applications: Structure–Rheology–Process–Properties Relationships. Polymers. 2025, 18, 128. [Google Scholar] [CrossRef]
- Aigaje, E.; Riofrio, A.; Baykara, H. Processing, properties, modifications, and environmental impact of nanocellulose/biopolymer composites: a review. Polymers. 2023, 15, 1–19. [Google Scholar] [CrossRef]
- Shojaeiarani, J.; Bajwa, D.S.; Stark, N.M.; Bajwa, S.G. Rheological properties of cellulose nanocrystals engineered polylactic acid nanocomposites. Compos. B Eng. 2019, 161, 483–489. [Google Scholar] [CrossRef]
- Meng, W.; Zhang, X.; Hu, X.; Liu, Y.; Zhang, J.; Qu, X.; Abdel-Magid, B. Mechanical Properties and Non-Isothermal Crystallization Kinetics of Polylactic Acid Modified by Polyacrylic Elastomers and Cellulose Nanocrystals. Polymers. 2023, 15, 3767. [Google Scholar] [CrossRef]
- Tang, K.; Wang, S.; Liu, H.; Zhang, C.; Yao, Y.; Lu, S.; Liao, X. Manipulation of crystallization nucleation and mechanical properties of PBAT films by octadecylamine-cellulose. ACS Sustain. Chem. Eng. 2024, 12, 4276–4285. [Google Scholar] [CrossRef]
- Alonso-Cañon, S.; Alonso-Estébanez, A.; Yoris-Nobile, A.I.; Brunčič, A.; Blanco-Fernandez, E.; Castanon-Jano, L. Rheological parameter ranges for 3D printing sustainable mortars using a new low-cost rotational rheometer. Int. J. Adv. Manuf. Tech. 2025, 140, 1945–1961. [Google Scholar] [CrossRef]
- Lasseuguette, E.; Roux, D.; Nishiyama, Y. Rheological properties of microfibrillar suspension of TEMPO-oxidized pulp. Cellul. 2008, 15, 425–433. [Google Scholar] [CrossRef]
- Gharib, N.; Bharathan, B.; Amiri, L.; McGuinness, M.; Hassani, F.P.; Sasmito, A.P. Flow characteristics and wear prediction of Herschel-Bulkley non-Newtonian paste backfill in pipe elbows. Can. J. Chem. Eng. 2017, 95, 1181–1191. [Google Scholar]
- Zou, L.; Tang, M.; Li, B. Bingham and herschel-bulkley fluids flow regimes in rough-walled rock fractures. Int. J. Rock Mech. Min. Sci. 2024, 180, 105832. [Google Scholar] [CrossRef]
- Mahmood, F.T.; Chowdhury, T.S.; Hasan, M.N. Fluid-structure interaction induced mixed convection characteristics in a lid-driven square cavity with non-Newtonian power law fluids. Int. J. Thermofluids. 2024, 22, 100687. [Google Scholar] [CrossRef]
- Nabels-Sneiders, M.; Barkane, A.; Platnieks, O.; Orlova, L.; Gaidukovs, S. Biodegradable poly (butylene succinate) laminate with nanocellulose interphase layer for high-barrier packaging film application. Foods. 2023, 12, 4136. [Google Scholar] [CrossRef]
- Ren, Y.; Fan, X.; Cao, L.; Chen, Y. Water-resistant and barrier properties of poly (vinyl alcohol)/nanocellulose films enhanced by metal ion crosslinking. Int. J. Biol. Macromol. 2024, 277, 134245. [Google Scholar]
- Riaz, T.; Zeng, X.; Ye, X.; Yu, T.; Xia, Z.; Khan, M.M.; Ahmad, T.; Kalsoom, A.; Rauf, M.A.; Ashraf, M.A. The Biophysics of Nanocellulose Packaging: Linking Nanoscale Structure to Food Preservation and Safety. Food Biophys. 2025, 20, 196. [Google Scholar] [CrossRef]
- Yu, K.; Yang, L.; Zhang, S.; Zhang, N.; Zhu, D.; He, Y.; Cao, X.; Liu, H. Tough, antibacterial, antioxidant, antifogging and washable chitosan/nanocellulose-based edible coatings for grape preservation. Food Chem. 2025, 468, 142513. [Google Scholar] [CrossRef]
- Zhang, L.; Cui, M.; Tong, H.; Zhang, J.; Li, Q.; Gao, X.; Qi, W.; Lam, H.L.; Huang, R.; Su, R. Multi-functional edible coatings tailored with nanocellulose for perishable fruits. Carbohydr. Polym. 2025, 358, 123520. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Cai, J.; Huang, Y.; Luo, J. Nanocellulose-based carbon sphere immobilized AgNPs for antibacterial cellulosic paper. Ind. Crop. Prod. 2025, 230, 121120. [Google Scholar] [CrossRef]
- Zhang, K.; Li, C.; Liu, L.; Wang, X.; Li, N.; Yang, C.; Huang, Y.; Lu, H.; Liu, H.; Li, J. Developing lightweight silanized cellulose ultramaterials as the strongest engineered aerogel. Adv. Funct. Mater. 2025, 35, 2415963. [Google Scholar] [CrossRef]
- Alves, Z.; Brites, P.; Ferreira, N.M.; Figueiredo, G.; Otero-Irurueta, G.; Gonçalves, I.; Mendo, S.; Ferreira, P.; Nunes, C. Thermoplastic starch-based films loaded with biochar-ZnO particles for active food packaging. J. Food Eng. 2024, 361, 111741. [Google Scholar] [CrossRef]
- Feng, Q.; Fan, B.; He, Y.-C.; Ma, C. Antibacterial, antioxidant and fruit packaging ability of biochar-based silver nanoparticles-polyvinyl alcohol-chitosan composite film. Int. J. Biol. Macromol. 2024, 256, 128297. [Google Scholar] [CrossRef]
- Hubbe, M.A.; Tayeb, P.; Joyce, M.; Tyagi, P.; Kehoe, M.; Dimic-Misic, K.; Pal, L. Rheology of nanocellulose-rich aqueous suspensions: a review. BioResources. 2017, 12, 9556–9661. [Google Scholar]
- Wu, J.; Gao, Y.; Shen, H.; Yan, S.; Zhao, R.; Wang, F.; Shen, X.; Li, Z.; Yao, X.; Wang, Y. Application potential of wheat bran cellulose nanofibers as Pickering emulsion stabilizers and stabilization mechanisms. Food Chem. X. 2024, 24, 101922. [Google Scholar] [CrossRef]
- Wu, J.; Tang, X.; Gao, W.; Li, J.; Li, J.; Cao, D.; Zeng, J.; Wang, B.; Xu, J.; Chen, K. Excellently stable Camellia oleifera oil Pickering emulsion fabricated based on high carboxyl content cellulose nanofibrils. Carbohydr. Polym. 2025, 123694. [Google Scholar] [CrossRef]
- Zhang, Z.; Lin, X.; Lin, J.; Liu, N.; Wan, B.; Fang, X.; Shui, L.; Li, Y.; Tam, K.C.; Huang, J. Cellulose supported and strengthened shear stiffening gel with enhanced impact-resistant performance. Chem. Eng. J. 2023, 473, 145435. [Google Scholar] [CrossRef]
- Hamouda, R.A.; Qarabai, F.A.; Shahabuddin, F.S.; Al-Shaikh, T.M.; Makharita, R.R. Antibacterial activity of ulva/nanocellulose and ulva/Ag/cellulose nanocomposites and both blended with fluoride against bacteria causing dental decay. Polymers. 2023, 15, 1047. [Google Scholar]
- Hafid, H.S.; Omar, F.N.; Zhu, J.; Wakisaka, M. Enhanced crystallinity and thermal properties of cellulose from rice husk using acid hydrolysis treatment. Carbohydr. Polym. 2021, 260, 117789. [Google Scholar] [CrossRef] [PubMed]
- Ponce, J.; da Silva Andrade, J.G.; dos Santos, L.N.; Bulla, M.K.; Barros, B.C.B.; Favaro, S.L.; Hioka, N.; Caetano, W.; Batistela, V.R. Alkali pretreated sugarcane bagasse, rice husk and corn husk wastes as lignocellulosic biosorbents for dyes. Carbohydr. Polym. Technol. Appl. 2021, 2, 100061. [Google Scholar] [CrossRef]
- Vu, A.N.; Nguyen, L.H.; Tran, H.-C.V.; Yoshimura, K.; Tran, T.D.; Van Le, H.; Nguyen, N.-U.T. Cellulose nanocrystals extracted from rice husk using the formic/peroxyformic acid process: isolation and structural characterization. RSC Adv. 2024, 14, 2048–2060. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; E, Y.; Sun, Q.; Chen, Y.; Dai, W.; Bao, Z.; Niu, W.; Meng, J. Analysis of the pyrolysis kinetics, reaction mechanisms, and by-products of Rice husk and rice straw via TG-FTIR and Py-GC/MS. MOL. 2024, 30, 10. [Google Scholar] [CrossRef]
- Rodrı́guez-Chong, A.; Ramı́rez, J.A.; Garrote, G.; Vázquez, M. Hydrolysis of sugar cane bagasse using nitric acid: a kinetic assessment. J. Food Eng. 2004, 61, 143–152. [Google Scholar] [CrossRef]
- Mahmud, M.A.; Anannya, F.R. Sugarcane bagasse-A source of cellulosic fiber for diverse applications. Heliyon. 2021, 7. [Google Scholar] [CrossRef]
- Song, G.; Madadi, M.; Meng, X.; Sun, C.; Aghbashlo, M.; Sun, F.; Ragauskas, A.J.; Tabatabaei, M.; Ashori, A. Double in-situ lignin modification in surfactant-assisted glycerol organosolv pretreatment of sugarcane bagasse towards efficient enzymatic hydrolysis. Chem. Eng. J. 2024, 481, 148713. [Google Scholar]
- Husna, M.; Vasantharuba, S. Analysis and comparison of peanut shell’s cellulose content; 2022. [Google Scholar]
- Anike, F.; Yusuf, M.; Isikhuemhen, O. Co-substrating of peanut shells with cornstalks enhances biodegradation by Pleurotus ostreatus. J Bioremed Biodeg. 2016, 7, 1–7. [Google Scholar]
- Pączkowski, P.; Puszka, A.; Gawdzik, B. Effect of eco-friendly peanut shell powder on the chemical resistance, physical, thermal, and thermomechanical properties of unsaturated polyester resin composites. Polymers. 2021, 13, 3690. [Google Scholar]
- Garcia-Maraver, A.; Salvachúa, D.; Martínez, M.; Diaz, L.; Zamorano, M. Analysis of the relation between the cellulose, hemicellulose and lignin content and the thermal behavior of residual biomass from olive trees. Waste Manag. 2013, 33, 2245–2249. [Google Scholar] [CrossRef]
- Mansora, A.M.; Lima, J.S.; Anib, F.N.; Hashima, H.; Hoa, W.S. Characteristics of cellulose, hemicellulose and lignin of MD2 pineapple biomass. Chem. Eng. 2019, 72, 79–84. [Google Scholar]
- Daud, Z.; Awang, H.; Kassim, A.S.M.; Hatta, M.Z.M.; Aripin, A.M. Comparison of pineapple leaf and cassava peel by chemical properties and morphology characterization. Adv. Mater. Res. 2014, 974, 384–388. [Google Scholar] [CrossRef]
- Aripin, A.M.; Kassim, A.S.M.; Daud, Z.; Hatta, M.Z.M. Cassava peels for alternative fibre in pulp and paper industry: chemical properties and morphology characterization. Int. J. Integr. Eng. 2013, 5. [Google Scholar]
- Mubarik, S.; Qureshi, N.; Sattar, Z.; Shaheen, A.; Kalsoom, A.; Imran, M.; Hanif, F. Synthetic approach to rice waste-derived carbon-based nanomaterials and their applications. J. Nanomanuf. 2021, 1, 109–159. [Google Scholar] [CrossRef]
- Liu, J.; Xue, Y.; Zhang, M.; Dai, L. Graphene-based materials for energy applications. MRS Bull. 2012, 37, 1265–1272. [Google Scholar] [CrossRef]
- Terea, H.; Selloum, D.; Rebiai, A.; Bouafia, A.; Ben Mya, O. Preparation and characterization of cellulose/ZnO nanoparticles extracted from peanut shells: effects on antibacterial and antifungal activities. Biomass Convers. Biorefin. 2024, 14, 19489–19500. [Google Scholar] [CrossRef]
- Abdul Rahman, N.H.; Chieng, B.W.; Ibrahim, N.A.; Abdul Rahman, N. Extraction and characterization of cellulose nanocrystals from tea leaf waste fibers. Polymers 2017, 9, 588. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, Y.; Zheng, D.; Li, M.; Yue, J. Isolation and characterization of nanocellulose crystals via acid hydrolysis from agricultural waste-tea stalk. Int. J. Biol. Macromol. 2020, 163, 927–933. [Google Scholar] [CrossRef]
- Abdul Rahman, N.H.; Chieng, B.W.; Ibrahim, N.A.; Abdul Rahman, N. Extraction and characterization of cellulose nanocrystals from tea leaf waste fibers. Polymers. 2017, 9, 1–11. [Google Scholar] [CrossRef]
- Mohd, N.K.; Wee, N.N.A.N.; Azmi, A.A. Green synthesis of silica nanoparticles using sugarcane bagasse. in AIP conference proceedings, 2017; AIP Publishing LLC. [Google Scholar]
- Pelissari, F.M.; Andrade-Mahecha, M.M.; do Amaral Sobral, P.J.; Menegalli, F.C. Nanocomposites based on banana starch reinforced with cellulose nanofibers isolated from banana peels. J. Colloid Interface Sci. 2017, 505, 154–167. [Google Scholar] [CrossRef]
- Paras, K. Yadav, P. Kumar, D.R. Teja, S. Chakraborty, M. Chakraborty, S.S. Mohapatra, A. Sahoo, M.M. Chou, C.-T.J.N. Liang. A review on low-dimensional nanomaterials: nanofabrication, characterization and applications 2022, 13, 160.
- Su, B.; Wu, Y.; Jiang, L. The art of aligning one-dimensional (1D) nanostructures. Chem. Soc. Rev. 2012, 41, 7832–7856. [Google Scholar] [CrossRef]
- Li, X.; Wang, J. One-dimensional and two-dimensional synergized nanostructures for high-performing energy storage and conversion. InfoMat. 2020, 2, 3–32. [Google Scholar] [CrossRef]
- Dutta, S.; Kim, J.; Ide, Y.; Kim, J.H.; Hossain, M.S.A.; Bando, Y.; Yamauchi, Y.; Wu, K.C.-W. 3D network of cellulose-based energy storage devices and related emerging applications. Mater. Horiz. 2017, 4, 522–545. [Google Scholar] [CrossRef]
- Sumarago, E.C.; dela Cerna, M.F.M.; Leyson, A.K.B.; Tan, N.P.B.; Magsico, K.F. Production and characterization of nanocellulose from maguey (Agave cantala) fiber. Polymers. 2024, 16, 1312. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.I.; Rahman, M.M.; Ghos, B.C.; Gafur, M.A.; Alam, M.A.; Rabbi, M.A. Preparation and characterization of crystalline nanocellulose from keya (Pandanus tectorius) L. fiber as potential reinforcement in sustainable bionanocomposite: A waste to wealth scheme. Carbohydr. Polym. Technol. Appl. 2024, 8, 100600. [Google Scholar] [CrossRef]
- Punnadiyil, R.K.; Sreejith, M.; Purushothaman, E. Isolation of microcrystalline and nano cellulose from peanut shells. J. Chem. Pharm. Sci. 2016, 974, 2115. [Google Scholar]
- Mishra, S.; Prabhakar, B.; Kharkar, P.S.; Pethe, A.M. Banana peel waste: An emerging cellulosic material to extract nanocrystalline cellulose. ACS Omega. 2022, 8, 1140–1145. [Google Scholar] [CrossRef]
| Fibrous Biomass | Cellulose | Lignin | Hemicellulose | Treatment/Extraction Process | Reference |
|---|---|---|---|---|---|
| Rice husk | 30-70 | 3-25 | 10-20 | Alkaline, bleaching agent, acid hydrolysis | Hafid, H.S. et al. [125] |
| Rice husk | 50 | 27 | 10 | NaOH treatment, water washing | Ponce, J. et al. [126] |
| Rice husk | 22-57 | - | - | Acetic acid, bleaching (NaOH/H2O2) | Vu, A.N. et al. [127] |
| Rice husk | 16 | 21 | 36 | Removal of dust, without treatments | Lin, L. et al. [128] |
| Rice straw | 18 | 14 | 47 | Removal of dust, without treatments | Lin, L. et al. [128] |
| Sugarcane bagasse | 58 | 12 | 17 | NaOH treatment, water washing | Ponce, J. et al. [126] |
| Sugarcane bagasse | 39 | 26 | 24 | Acid hydrolysis | Rodrı́guez-Chong, A. et al. [129] |
| Sugarcane bagasse | 26-47 | 19-23 | 14-23 | Alkaline treatment, hot water wash | Mahmud, M.A. and Anannya, F.R. [130] |
| Sugarcane bagasse | 41 | 23 | 21 | High temperature alkaline treatment | Song, G. et al. [131] |
| Peanut shell | 36-59 | 27 | 7 | Deep eutectic solvents | Lu, A. et al. [30] |
| Peanut shell | 32-34 | 32 | 18 | Standard and extraction methods | Husna, M. and Vasantharuba, S. [132] |
| Peanut shell | 32-37 | 27-30 | 7-9 | Inoculum of culture incubation process | Anike, F. et al. [133] |
| Peanut shell | 45 | 36 | 6 | - | Pączkowski, P. et al. [134] |
| Corn husk | 55 | 9 | 27 | NaOH treatment, water washing | Ponce, J. et al. [126] |
| Corn stalk | 38-44 | 5-10 | 17-27 | Inoculum of culture incubation process | Anike, F. et al. [133] |
| Corn bract | 36 | 39 | 15 | Enzymatic hydrolysis without pre-treatment | Anike, F. et al. [133] |
| Corn bract | 52 | 35 | 4 | Acetic acid, enzymatic hydrolysis | Anike, F. et al. [133] |
| Corn bract | 76 | 13 | 2 | Ammonium, enzymatic hydrolysis | Anike, F. et al. [133] |
| Olive leaves | 6-9 | 40 | 4-9 | Acid hydrolysis | Garcia-Maraver, A. et al. [135] |
| Olive pruning | 20 | 27 | 10-11 | Acid hydrolysis | Garcia-Maraver, A. et al. [135] |
| Olive wood | 31-32 | 24 | 11-15 | Acid hydrolysis | Garcia-Maraver, A. et al. [135] |
| Pineapple leaf | 30 | 22 | 37 | NaOH and sodium chlorite treatment | Mansora, A.M. et al. [136] |
| Pineapple leaf | 66 | 4 | 20 | Bleaching agent | Daud, Z. et al. [137] |
| Pineapple stem | 37 | 20 | 34 | NaOH and sodium chlorite treatment | Mansora, A.M. et al. [136] |
| Pineapple root | 42 | 19 | 32 | NaOH and sodium chlorite treatment | Mansora, A.M. et al. [136] |
| Banana stem | 35 | 12 | 25 | NaOH and sodium chlorite treatment | Mansora, A.M. et al. [136] |
| Banana stem (outer) | 40 | 13 | 25 | NaOH and sodium chlorite treatment | Mansora, A.M. et al. [136] |
| Cassava peel | 38 | 8 | 37 | Dust removal, without treatment | Aripin, A.M. et al. [138] |
| Cassava peel | 40 | 12 | 21 | Dust removal, without treatment | Aripin, A.M. et al. [138] |
| Cassava peel | 38 | 8 | 37 | Bleaching agent | Daud, Z. et al. [137] |
| Nanomaterial | Material | Method | Activation Process | Surface Effect | Reference |
|---|---|---|---|---|---|
| Graphene | Rice husk | Carbonization Pyrosis 400 oC | Mixture of biochar with KOH in 1:2 ratio. annealing at 800oC, washing and drying | few-layered graphene with agglomeration of silica particles (porous) | Mubarik, S. et al. [139] |
| Graphene | Peanut shells | Carbonization Pyrosis 200 oC | Mixture of biochar and FeCl3.6H2O in water. Adjust pH HCl (98% Betaine Hydrochloride). Washing and drying at 100oC, milling | Two-dimensional (2D) material, network of SP2-bonded carbon atoms | Aro-Modiu, O. et al. [50] |
| Graphene oxide | Graphene | - | Mixture of graphene powder, sodium nitrate and H2SO4 in ice bath. Adding KMnO4. Reaction termination with H2O2.washing, filtration. Drying 30oC. | Polar groups (surface HO- groups), surface area≈2630 m2 g-1. | Aro-Modiu, O. et al. [50] [140] |
| Graphene | Rubber seed shell | Pyrolysis 700 °C for 90 min | Mixture of biochar to KOH in 1:3 ratio under N2 at different temperatures (600, 700, 800 and 900°C), 3h | Surface area 712 m2g-1 | Anthonysamy, S. et al. [52] |
| cellulose nanocrystals | Peanut shells | lignocellulose | Peanut shell powder (500-μm sieve), hot rinse and dry. Treatment: HCl, NaOH. precipitate washed, centrifuged. Residual fibres sonicated and dried. Fibres hydrolysed with H2SO4, cold water wash, centrifuge, ultrasonication, drying | High antifungal and antibacterial activity. positive reaction and sensitivity against Gram (−) and Gram (+) strains of pathogens | Terea, H. et al. [141] |
| Cellulose nanocrystal | Tea leaf waste fiber | Cellulose extraction | Acid hydrolysis: cellulose treated with H2SO4 (pre-heated). Processes of dialysis of suspension and ultrasonic. | - | Abdul Rahman, N.H. et al. [142] |
| Nanocellulose crystals | Tea stalk | Cellulose isolation | Acid hydrolysis | Lower zeta (ζ) potential -33 mV | Guo, Y. et al. [143] |
| Nanoparticles | Cassava peel | Starch extraction, | Sulphuric acid hydrolysis, washing and drying | Abdul Rahman, N.H. et al. [144] | |
| Silica Nanoparticles |
Sugarcane bagasse | Extraction, precipitation | Acid hydrolysis, alkali hydrolysis | surface area 111 m2g-1 | Mohd, N.K. et al. [145] |
| Cellulose nanofibers | Banana peel | Alkaline hydrolysis (KOH), bleaching (NaClO2), acid hydrolysis (H2SO4) Mechanical process: two-stage high-pressure homogenizer |
Between -16 mV and -44 mV | Pelissari, F.M. et al. [146] |
| Type of Ball Mills | Source & Specification | Process-input | Sample | Primary Properties | Reference |
|---|---|---|---|---|---|
| Planetary Ball Mill | PM100; Retsch Corporation, stainless steel jar (500 mL), stainless steel balls 2.4 mm in diameter x 800 balls | settings at 5 min ON and 5 min OFF Milling speed of 510-630 rpm |
Pinewood Biochar, initial size of around 3 mm. | Fine powder 212 - 453 nm Zeta potential (mV) −31.3 ± 2.6 |
Naghdi, M. et al. [59] |
| Planetary ball mill | ITO LP-1. 80 mL of jar with different ball diameters of 10, 5, and 2 mm x 5, 4, and 3 balls, respectively | Speed of 300 rpm, time treatment 0.5, 1, 2, and 3 h, acid hydrolysis-assisted | Grade 3 mm Chr cellulose 38 µm (400 mesh) size of cellulose powder | Yield 77-90%, native cellulose type I, size 3-13 nm, maximum decomposition temperature 300–330 °C with mass loss 75 wt% | Phanthong, P. et al. [61] |
| Tumbler Ball Mill | MA500, Marconi, with alumina balls. Jar 1 L 70 % of balls and 30 % samples | speed 200 rpm, Treatment times: 1 h, 2 h, 3 h, and 4 h. ethanol-ultrasonic-assisted | Cellulose extracted from eucalyptus sawdust using NaClO2, NaOH and KOH. | Yield ˜80 wt.%. Zeta potential (mV) -24 to -60 | Ferreira, R.R. et al. [65] |
| Planetary ball mill | ITO LP-1 Planetary pot mill, | milling speed of 400 rpm for 2 h | cellulose powder | Crystal size 2-4 nm, degradation 220–410 oC with thermal decomposition peak at 373 oC with 80 wt% loss | Phanthong, P. et al. [94] |
| Planetary ball mill | XQM-0.4A, Tencan powder, Changsha, China) |
speed of 400 rpm, 3 h in cycles of 20 min/milling per 10 min/rest. NaOH-assisted. | Cellulose from pineapple peel | Nanofibrils size 19-24 nm, Zeta potential (mV) -22 to -28. | Wang, Y. et al. [60] |
| Planetary ball mill | Naraya-MPM-2 × 250H mill (Amin Asis Fanavar Pars), 250 mL jar, 60% stainless steel balls of 0.5–2 cm, 30% sample | speed 200 rpm for 2 h. dispersed in deionized water prior ultrasonic-assisted, | Cellulose from Cuminum cyminum waste | Zeta potential (mV) -25. Size 25 nm. degradation peak around 330 °C | Hoseinpour, Z. et al. [66] |
| Tumbler Ball Mill | impact ball mill (MA500, Marconi Ltda., Piracicaba, SP, Brazil) | alumina balls (diameter of 21 mm), 70 g of alumina balls for 1 g of cellulose fiber. Time 6, 9, and 12 h. ethanol-assisted | Cellulose from corn stalks, cobs, and husks | Zeta potential (mV) -15 to -41 (stalk), -13 to -42 (husk), -30 to -38 (comb). Size 70-195 nm | dos Santos, D.F. et al. [62] |
| Dimension | Nanoscale | Shape | Nanomaterials | Reference |
|---|---|---|---|---|
| Zero dimension (0D) | Dimensions length, breadth, height (x,y,z) < 100 nm |
Spherical, quasi-sphere, cubic, polygonal | Carbon dots, fullerene, clusters, grains, nanoparticles | Paras et al. [147] |
| One dimensional (1D) | Two dimensions (x,y) < 100 nm, and third dimension (z) >100 nm | needle-like | linear structures, carbon nanotubes, metals or metal oxides nanowires, polymer nanowires, nanofibers, hybrid materials | Su, B. et al. [148] |
| Two dimensional (2D) | One dimension (x) <100 nm, other two dimensions (y,z) undefined | atomically thin sheets, sheet-like honeycomb | Nanofilms, nanolayers, and nanocoatings | Li, X. and Wang, J. [149] |
| Three-dimensional (3D) | Dimensions (x,y,z) > 100 nm, not confined to the nanoscale. |
Nano-cubes, fullerenes, dendrimers, and nanocages |
Cellulose Nanocrystals (CNCs) and Nanofibrils (CNFs) | Dutta, S. et al. [150] |
| Nanoparticles | Source of Waste Biomass | Technique | Crystalline Properties | Reference |
|---|---|---|---|---|
| Cellulose nanofibrils | Pineapples peel | XRD (X’Pert3 Powder, Malvern Panalytical, Almelo, The Netherlands) | Crl 38-44%, Crystal size (D) 2-3 nm |
Wang, Y. et al. [60] |
| Nanocellulose | Cumin husk | XRD, Karlsruhe instrument (Germany), input: CuKα radiation (λ = 0.1542 nm, 40 kV, 40 mA). Position: 2θ range of 5–65o, scan rate of 3o/min | XRD peaks at 2θ =18o and 22.6o Clr 69.3% at 2θ=22.6o Crystal size 3.76 nm |
Hoseinpour, Z. et al. [66] |
| Nanocellulose | Maguey fibre | XRD, LabX-6000, SHIMADZU, Japan. Input: 40.0 kV and 30.0 mA with Cu Kα radiation (1.5148 Å). 2θ range of 2-70°, scan speed 1o/min | XRD peaks ~ 2θ = 15.52° and 22.64°, Crl 74.80% at 2θ=22.64o |
Sumarago, E.C. et al. [151] |
| Raw fibre | Keya leaf (alkali and bleach treatment) | Wider angle XRD, BRUKER D8 ADVANCE, input: Cu Kα radiation (α = 0.154 nm) at 40 mA and 50 kV. | Raw fibre Crl 45.35% at 2θ = 14.79o, 22.59o, 24.25o, and 29.97. Alkali fibre Crl 54.69% at 2θ=22.97o Bleached fibre Crl 43.43% at 2θ=22.8o |
Hossain, M.I. et al. [152] |
| Nanocellulose crystals | Keya leaf | Wider angle XRD, BRUKER D8 ADVANCE, input: Cu Kα radiation (α = 0.154 nm) at 40 mA and 50 kV. | Clr 61.31% at 2θ=19.9o and 22.44o | Hossain, M.I. et al. [152] |
| Nanocellulose crystals | Peanut shells | XRD, RIGAKU MINIFLEX-600 input: Cu Kα (λ=1.54 A0), scanning in a 2θ range of 10-50o | Crl 26-66% at 2θ=22.6o | Punnadiyil, R.K. et al. [153] |
| Nanocrystalline cellulose | Banana fibre | XRD, Shimadzu XRD-6000. Input: voltage of 30 kV and current of 30 mA, 2θ=5-60°, scan rate 2 °C/min. | XRD peaks at 2θ = 16.1°, 22.8° and 34.9° Crl 62% at 2θ = 19 and 22.2° |
Mishra, S. et al. [154] |
| Nanocellulose | Rice husks (variety) |
XRD, MiniFlex 600 (Rigaku, Japan). Input: Cu Kα λ = 1.54, acceleration potential =40 kV, current = 15 mA. scan range of 2θ = 2-60°, scan rate 3°/min | XRD peaks 2θ = 16.3°, 22.4° and 34.5° for raw husk. Crl for husk 40-56% at 2θ=22.4o Crl for cellulose 46-66% at 2θ=22.4o Crl for nanocellulose 59-77% at 2θ=22.4o |
Rashid, S. and Dutta, H. [90] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




