Submitted:
05 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
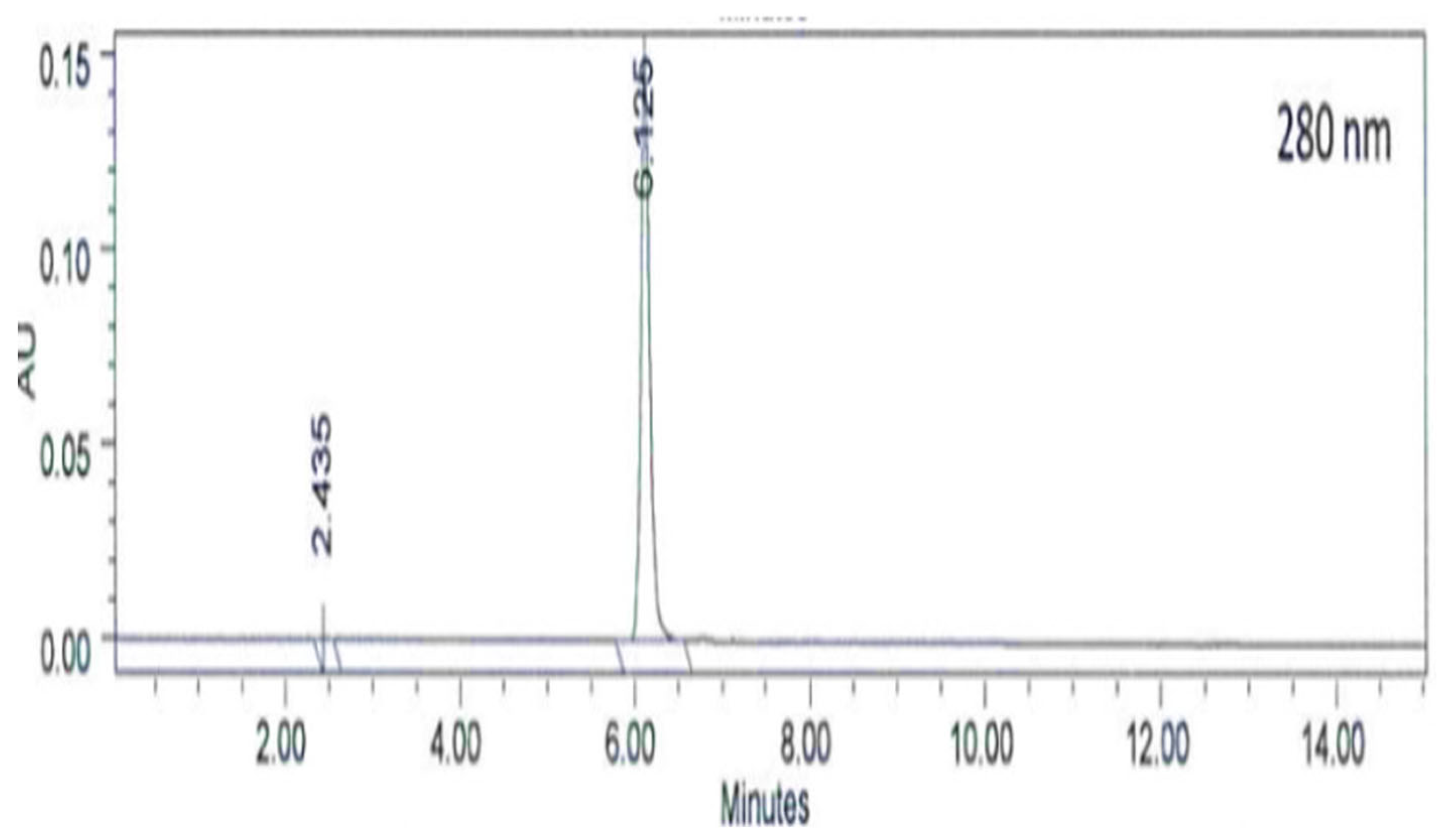
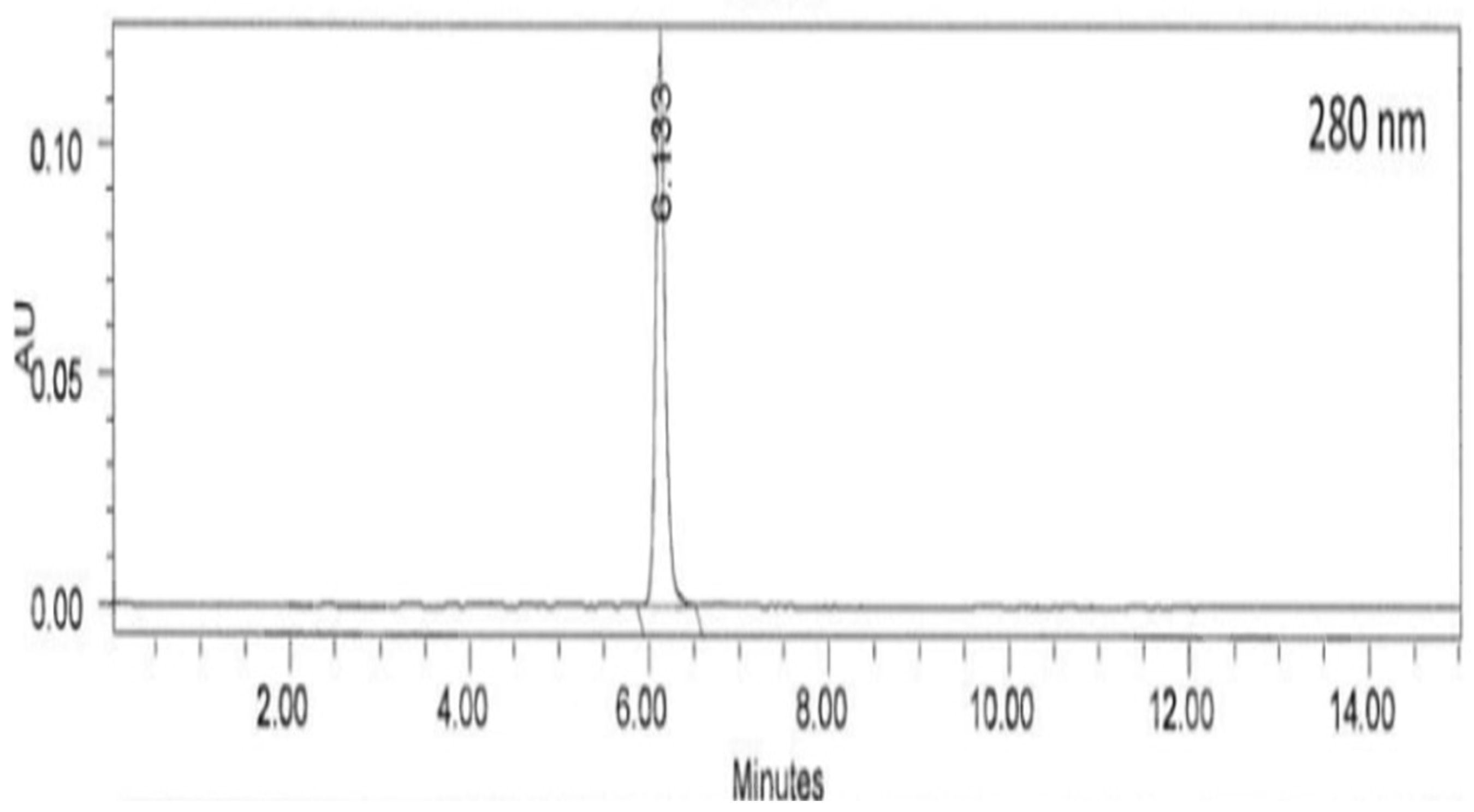
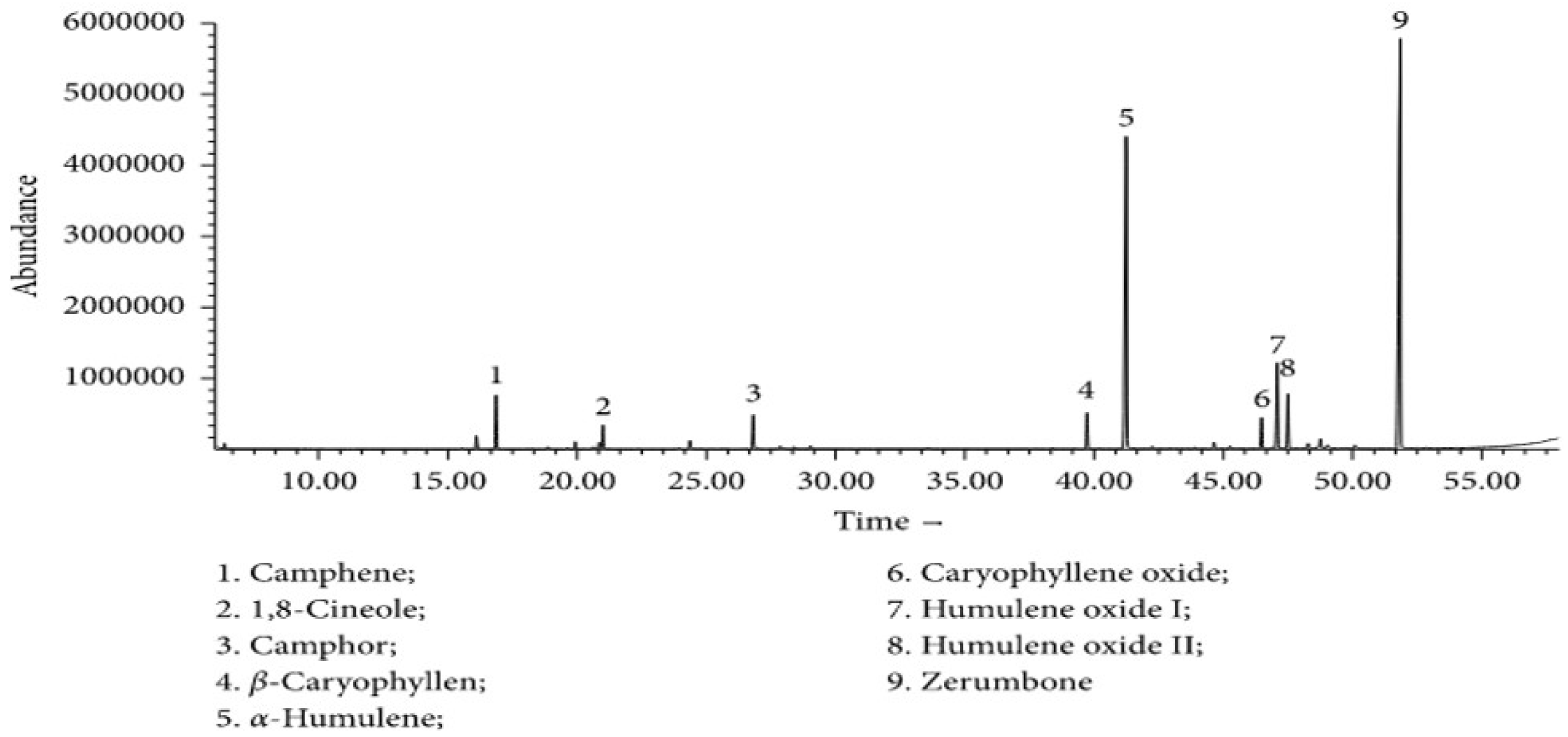
The oil of the Zingiber zerumbet has been gaining traction amongst the researchers due to its phytochemical benefits mainly zerumbone. Due to the presence of a complex mixture of terpenoids in the oil, recrystallization is an effective method to obtain the active compound, zerumbone. Objective: This study was to optimized the recrystallization via decanting with hexane and evaporation method to produce the most yield that contain purified zerumbone crystals. Materials and methods: Ground dried and intact dried Zingiber zerumbet were compared to determine the highest yield of zerumbone crystals. A yield comparison between ground and intact dried Zingiber zerumbet was carried out through 6 hours of hydrodistillation followed by decanting with hexane. HPLC qualitative analysis was done on the purity of zerumbone crystals from ground and intact material respectively at wavelength 280nm. Results: After 6 hours of hydrodistillation, intact dried crude plant material produced 0.29%w/w more zerumbone crystals than the ground dried Zingiber zerumbet. HPLC qualitative analysis done on the purity of zerumbone crystals from ground dried and intact plant material were 98.51% and 99.68% respectively at wavelength 280nm. Discussion: A yield comparison between ground and intact dried Zingiber zerumbet that was carried out through hydrodistillation, exhibited significant difference (p<0.05). The low yield of oil from the ground crude plant material, which in turn produced lesser zerumbone crystals can be contributed to the heat emanated by the blades of the grinder resulting in melted oil from the compound sticking to the surface of the grinder. It is also lamented that only 1% of the energy transmitted to the grinder is used for size reduction, the balance of the energy is converted into heat. Also, the colour of the compound from traditional grinding also intensified due to the heated compound. Besides that, the method of recrystallization that produced a higher yield of zerumbone crystals can be performed for future research. Conclusion: Intact dried Zingiber zerumbet produced higher yield of zerumbone crystals through an improved method of recrystallization.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Extraction
2.3. Extraction of Zingiber zerumbet
2.4. Optimization of Recrystallization and Isolation of Zerumbone
2.5. Identification of Zerumbone
3. Statistical Analysis
4. Results
4.1. Extraction of Zingiber zerumbet
5. Discussion
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Akhtar, N. M. Y., Jantan, I., Arshad, L., and Haque, M. A. (2019). Standardized ethanol extract, essential oil and zerumbone of zingiber zerumbet rhizome suppress phagocytic activity of human neutrophils. BMC Complementary and Alternative Medicine, 19(1), 1–12. [CrossRef]
- Al-Amin, M., Salhimi, S., Eltayeb, N. M., Siddiqui, M. A., Ruma, S. A., Nurun Nahar Sultana, G., Salhimi, S. M., and Faiz Hossain, C. (2019). Antimicrobial activity of the crude extract, fractions and isolation of zerumbone from rhizomes of zingiber roseum. Journal of Research in Pharmacy, 23(3), 559–566. [CrossRef]
- Albaayit, S. F. A., Rasedee, A., and Abdullah, N. (2020). Zerumbone-loaded nanostructured lipid carrier gel facilitates wound healing in rats. Revista Brasileira de Farmacognosia, 30(2), 272–278. [CrossRef]
- Azelan, N. A., Aziz, R., and Hasham, R. (2018). Optimisation of essential oil yield and zerumbone content in zingiber zerumbet extract using hydrodistillation process. Chemical Engineering Transactions, 63, 595–600. [CrossRef]
- Chia, J. S. M., Izham, N. A. M., Farouk, A. A. O., Sulaiman, M. R., Mustafa, S., Hutchinson, M. R., and Perimal, E. K. (2020). Zerumbone modulates α2A-adrenergic, TRPV1, and NMDA NR2B receptors plasticity in CCI-induced neuropathic pain in vivo and LPS-induced SH-SY5Y neuroblastoma in vitro models. Frontiers in Pharmacology, 11, 1–24. [CrossRef]
- Chien, T. Y., Huang, S. K. H., Lee, C. J., Tsai, P. W., and Wang, C. C. (2016). Antinociceptive and anti-inflammatory effects of zerumbone against mono-iodoacetate-induced arthritis. International Journal of Molecular Sciences, 17(2), 249. [CrossRef]
- Cirri, M., Bragagni, M., Mennini, N., and Mura, P. (2012). Development of a new delivery system consisting in “drug - In cyclodextrin - In nanostructured lipid carriers” for ketoprofen topical delivery. European Journal of Pharmaceutics and Biopharmaceutics, 80(1), 46–53. [CrossRef]
- Foong, J. N., Selvarajah, G. T., Rasedee, A., Rahman, H. S., How, C. W., Beh, C. Y., Teo, G. Y., and Ku, C. L. (2018). Zerumbone-loaded nanostructured lipid carrier induces apoptosis of canine mammary adenocarcinoma cells. BioMed Research International, 2018(8691569), 1–18. [CrossRef]
- Ghasemzadeh, A., Jaafar, H. Z. E., Ashkani, S., Rahmat, A., Juraimi, A. S., Puteh, A., and Muda Mohamed, M. T. (2016). Variation in secondary metabolite production as well as antioxidant and antibacterial activities of zingiber zerumbet (L.) at different stages of growth. BMC Complementary and Alternative Medicine, 16(1), 104. [CrossRef]
- Ghasemzadeh, A., Jaafar, H. Z. E., Rahmat, A., and Swamy, M. K. (2017). Optimization of microwave-assisted extraction of zerumbone from zingiber zerumbet L. rhizome and evaluation of antiproliferative activity of optimized extracts. Chemistry Central Journal, 11(1), 5. [CrossRef]
- Ghosh, S., Majumder, P. B., and Sen Mandi, S. (2011). Species-specific AFLP markers for identification of zingiber officinale, Z. montanum and Z. zerumbet (Zingiberaceae). Genetics and Molecular Research : GMR, 10(1), 218–229. [CrossRef]
- Girisa, S., Shabnam, B., Monisha, J., Fan, L., Halim, C. E., Arfuso, F., Ahn, K. S., Sethi, G., and Kunnumakkara, A. B. (2019). Potential of zerumbone as an anti-cancer agent. Molecules, 24(4), 734. [CrossRef]
- Huong, L. T., Chinh, H. V., An, N. T. G., Viet, N. T., Hung, N. H., Thuong, N. T. H., Giwa-Ajeniya, A. O., and Ogunwande, I. A. (2019). Zingiber zerumbet rhizome essential oil: Chemical composition, antimicrobial and mosquito larvicidal activities. European Journal of Medicinal Plants, 30(4), 1–12. [CrossRef]
- Jalil, M., Mohamad Annuar, M. S., Tan, B. C., and Khalid, N. (2015). Effects of selected physicochemical parameters on zerumbone production of zingiber zerumbet smith cell suspension culture. Evidence-Based Complementary and Alternative Medicine, 2015(757514), 1–7. [CrossRef]
- Jalili-Nik, M., Sadeghi, M. M., Mohtashami, E., Mollazadeh, H., Afshari, A. R., and Sahebkar, A. (2020). Zerumbone promotes cytotoxicity in human malignant glioblastoma cells through reactive oxygen species (ROS) generation. Oxidative Medicine and Cellular Longevity, 2020(3237983), 1–9. [CrossRef]
- Jung, H., Lee, Y. J., and Yoon, W. B. (2018). Effect of moisture content on the grinding process and powder properties in food: A review. Processes, 6(69), 1–16. [CrossRef]
- Kalantari, K., Moniri, M., Moghaddam, A. B., Rahim, R. A., Ariff, A. B., Izadiyan, Z., and Mohamad, R. (2017). A Review of the biomedical applications of zerumbone and the techniques for its extraction from ginger rhizomes. In Molecules, 22(10), 1–26. [CrossRef]
- Karthikeyan, R. H., Vian, M. A., Tao, Y., Degrou, A., Costil, J., Trespeuch, C., & Chemat, F. (2019). Alternative solvents for lipid extraction and their effect on protein quality in black soldier fly (Hermetia illucens) larvae. Journal of Cleaner Production, 238(117861), 1–40. [CrossRef]
- Khalid, M. H., Akhtar, M. N., Mohamad, A. S., Perimal, E. K., Akira, A., Israf, D. A., and Sulaiman, M. R. (2011). Antinociceptive effect of the essential oil of zingiber zerumbet in mice: Possible mechanisms. Journal of Ethnopharmacology, 137(1), 345–351. https://scihub.wikicn.top/10.1016/j.jep.2011.05.043.
- Kitayama, T., Okamoto, T., Hill, R. K., Kawai, Y., Takahashi, S., Yonemori, S., Yamamoto, Y., Ohe, K., Uemura, S., and Sawada, S. (1999). Chemistry of zerumbone. Simplified isolation, conjugate addition reactions, and a unique ring contracting transannular reaction of its dibromide. Journal of Organic Chemistry, 64(8), 2667–2672. [CrossRef]
- Kitayama, T., Yamamoto, K., Utsumi, R., Takatani, M., Hill, R. K., Kawai, Y., Sawada, S., and Okamoto, T. (2001). Chemistry of zerumbone. 2. Regulation of ring bond cleavage and unique antibacterial activities of zerumbone derivatives. Bioscience, Biotechnology and Biochemistry, 65(10), 2193–2199. [CrossRef]
- Kiyama, R. (2021). Nutritional implications of ginger: chemistry, biological activities and signaling pathways. Journal of Nutritional Biochemistry, 86(108486), 1–15. Accessed on 29 September 2021 from https://reader.elsevier.com/reader/sd/pii/S0955286320305180?token=4D3CED86C6DDD8B32B6F67767A6A18B26FC6BE19E0DF747C3D427C2B0E637BF9BE4408D3E06E33085B7141B03D2B1D93&originRegion=eu-west-1&originCreation=20210929073719.
- Koga, A. Y., Beltrame, F. L., and Pereira, A. V. (2016). Several aspects of zingiber zerumbet: A review. Revista Brasileira de Farmacognosia, 26, 385–391. [CrossRef]
- Li, L., Wu, X. H., Zhao, X. J., Xu, L., Pan, C. L., and Zhang, Z. Y. (2020). Zerumbone ameliorates behavioral impairments and neuropathology in transgenic APP/PS1 mice by suppressing MAPK signaling. Journal of Neuroinflammation, 17(1), 61. [CrossRef]
- Liu, H., Zheng, J., Liu, P., and Zeng, F. (2018). Pulverizing processes affect the chemical quality and thermal property of black, white, and green pepper (Piper nigrum L.). Journal of Food Science and Technology, 55(6), 2130. [CrossRef]
- Md, S., Kit, B. C. M., Jagdish, S., David, D. J. P., Pandey, M., and Chatterjee, L. A. (2018). Development and in vitro evaluation of a zerumbone loaded nanosuspension drug delivery system. Crystals, 8(7), 2–13. [CrossRef]
- Moreira Da Silva, T., Danniel Pinheiro, C., Orlandi, P. P., Pinheiro, C. C., and Soares Pontes, G. (2018). Zerumbone from zingiber zerumbet (L.) smith: a potential prophylactic and therapeutic agent against the cariogenic bacterium streptococcus mutans. BMC Complementary and Alternative Medicine, 18(1), 301. [CrossRef]
- Murthy, C. T., Rani, M., and Rao, P. N. S. (1999). Optimal grinding characteristics of black pepper for essential oil yield. Journal of Food Process Engineering, 22(2), 161–173. [CrossRef]
- Nik Norulaini, N. A., Anuar, O., Omar, A. K. M., AlKarkhi, A. F. M., Setianto, W. B., Fatehah, M. O., Sahena, F., and Zaidul, I. S. M. (2009). Optimization of SC-CO2 extraction of zerumbone from zingiber zerumbet (L) smith. Food Chemistry, 114(2), 702–705. [CrossRef]
- Noor, N. F. M., and Sirat, H. M. (2016). Isolation, characterization and modification of zerumbone from zingiber zerumbet. Journal of Teknologi, 1(1), 1–10. Accessed on 15 January 2021 from http://eprints.utm.my/id/eprint/68333/.
- Padalia, R. C., Verma, R. S., Chauhan, A., Singh, V. R., Goswami, P., Singh, S., Verma, S. K., Luqman, S., Chanotiya, C. S., and Darokar, M. P. (2018). Zingiber zerumbet (L.) Roscoe ex Sm. from northern India: Potential source of zerumbone rich essential oil for antiproliferative and antibacterial applications. Industrial Crops and Products, 112, 749–754. [CrossRef]
- Perez-Hurtado, P., Palmer, E., Owen, T., Aldcroft, C., Allen, M. H., Jones, J., Creaser, C. S., Lindley, M. R., Turner, M. A., and Reynolds, J. C. (2017). Direct analysis of volatile organic compounds in foods by headspace extraction atmospheric pressure chemical ionisation mass spectrometry. Rapid Communications in Mass Spectrometry, 31(22), 1947–1956. [CrossRef]
- Rahman, H. S., Rasedee, A., How, C. W., Abdul, A. B., Zeenathul, N. A., Othman, H. H., and Yeap, S. K. (2013). Zerumbone-loaded nanostructured lipid carriers: Preparation, characterization, and antileukemic effect. International Journal of Nanomedicine, 8, 2769–2781. [CrossRef]
- Rahman, H. S., Rasedee, A., Yeap, S. K., Othman, H. H., Chartrand, M. S., Namvar, F., Abdul, A. B., & How, C. W. (2014). Biomedical properties of a natural dietary plant metabolite, zerumbone, in cancer therapy and chemoprevention trials. BioMed Research International, 2014(920742), 1–10. [CrossRef]
- Rossi, F., Sharon, M., Irudayaraj, J., Vega-Vásquez, P., and Mosier, N. S. (2020). Nanoscale drug delivery systems: From medicine to agriculture. Frontiers in Bioengineering and Biotechnology, 8(79), 1–16. [CrossRef]
- Sachin, S. K., Ashwini, K. V, and Kawade Ashwini, C. V. (2017). Isolation and standardization of gingerol from ginger rhizome by using TLC, HPLC, and identification Tests. The Pharma Innovation Journal, 6(2), 179–182. Accessed on 15 January 2021 from https://www.thepharmajournal.com/archives/2017/vol6issue2/PartC/5-8-19-645.pdf.
- Saxena, S. N., Barnwal, P., Balasubramanian, S., Yadav, D. ., Lal, G., and Singh, K. . (2018). Cryogenic grinding for better aroma retention and improved quality of Indian spices and herbs: A review. Journal of Food Process Engineering, 41(6), 1–20. [CrossRef]
- Saxena, S. N., Rathore, S. S., Saxena, R., Barnwal, P., Sharma, L. K., and Singh, B. (2014). Effect of cryogenic grinding on essential oil constituents of coriander (coriandrum sativum L.) genotypes. Journal of Essential Oil-Bearing Plants, 17(3), 385–392. [CrossRef]
- Shahrajabian, M. H., Sun, W., and Cheng, Q. (2019). Pharmacological uses and health benefits of ginger (zingiber officinale) in traditional asian and ancient chinese medicine, and modern practice. Notulae Scientia Biologicae, 11(3), 309–319. [CrossRef]
- Sharma, L. K., Agarwal, D., Rathore, S. S., Malhotra, S. K., and Saxena, S. N. (2016). Effect of cryogenic grinding on volatile and fatty oil constituents of cumin (cuminum cyminum L.) genotypes. Journal of Food Science and Technology, 53(6), 2827–2834. [CrossRef]
- Singh, C. B., Chanu, S. B., Kh, L., Swapana, N., Cantrell, C., and Ross, S. A. (2014). Chemical composition and biological activity of the essential oil of rhizome of zingiber zerumbet (L.) smith. Journal of Pharmacognosy and Phytochemistry, 3(3), 130–133. Accessed on 15 January 2021 from https://naldc.nal.usda.gov/download/62328/PDF.
- Singh, Y. P., Girisa, S., Banik, K., Ghosh, S., Swathi, P., Deka, M., Padmavathi, G., Kotoky, J., Sethi, G., Fan, L., Mao, X., Halim, C. E., Arfuso, F., and Kunnumakkara, A. B. (2019). Potential application of zerumbone in the prevention and therapy of chronic human diseases. Journal of Functional Foods, 53, 248–258. [CrossRef]
- Somchit, M. N., Mak, J. H., Bustamam, A. A., Zuraini, A., Arifah, A. K., Adam, Y., and Zakaria, Z. A. (2012). Zerumbone isolated from zingiber zerumbet inhibits inflammation and pain in rats. Journal of Medicinal Plants Research, 6(2), 177–180. [CrossRef]
- Sulaiman, M. R., Perimal, E. K., Zakaria, Z. A., Mokhtar, F., Akhtar, M. N., Lajis, N. H., and Israf, D. A. (2009). Preliminary analysis of the antinociceptive activity of zerumbone. Fitoterapia, 80(4), 230–232. [CrossRef]
- Tan, J. W., Israf, D. A., and Tham, C. L. (2018). Major bioactive compounds in essential oils extracted from the rhizomes of zingiber zerumbet (L) Smith: A mini-review on the anti-allergic and immunomodulatory properties. Frontiers in Pharmacology, 9(652), 1–8. [CrossRef]
- Tian, M., Wu, X., Hong, Y., Wang, H., Deng, G., and Zhou, Y. (2020). Comparison of chemical composition and bioactivities of essential oils from fresh and dry rhizomes of zingiber zerumbet (L.) smith. BioMed Research International, 2020(9641284), 1–9. [CrossRef]
- Zakaria, Z. A., Mohamad, A. S., Chear, C. T., Wong, Y. Y., Israf, D. A., and Sulaiman, M. R. (2010). Antiinflammatory and antinociceptive activities of zingiber zerumbet methanol extract in experimental model systems. Medical Principles and Practice, 19(4), 287–294. [CrossRef]
- Zakaria, Z. A., Yob, N. J., Jofrry, S. M., Affandi, M. M. R. M. M., Teh, L. K., and Salleh, M. Z. (2011). zingiber zerumbet (L.) smith: s review of its ethnomedicinal, chemical, and pharmacological uses. Evidence-Based Complementary and Alternative Medicine, 2011(543216), 1–12. [CrossRef]
- Zhang, C., Liu, J., Zhao, F., Lu, C., Zhao, G. R., and Lu, W. (2018). Production of sesquiterpenoid zerumbone from metabolic engineered saccharomyces cerevisiae. Metabolic Engineering, 49, 28–35. [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




