Submitted:
06 February 2026
Posted:
06 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
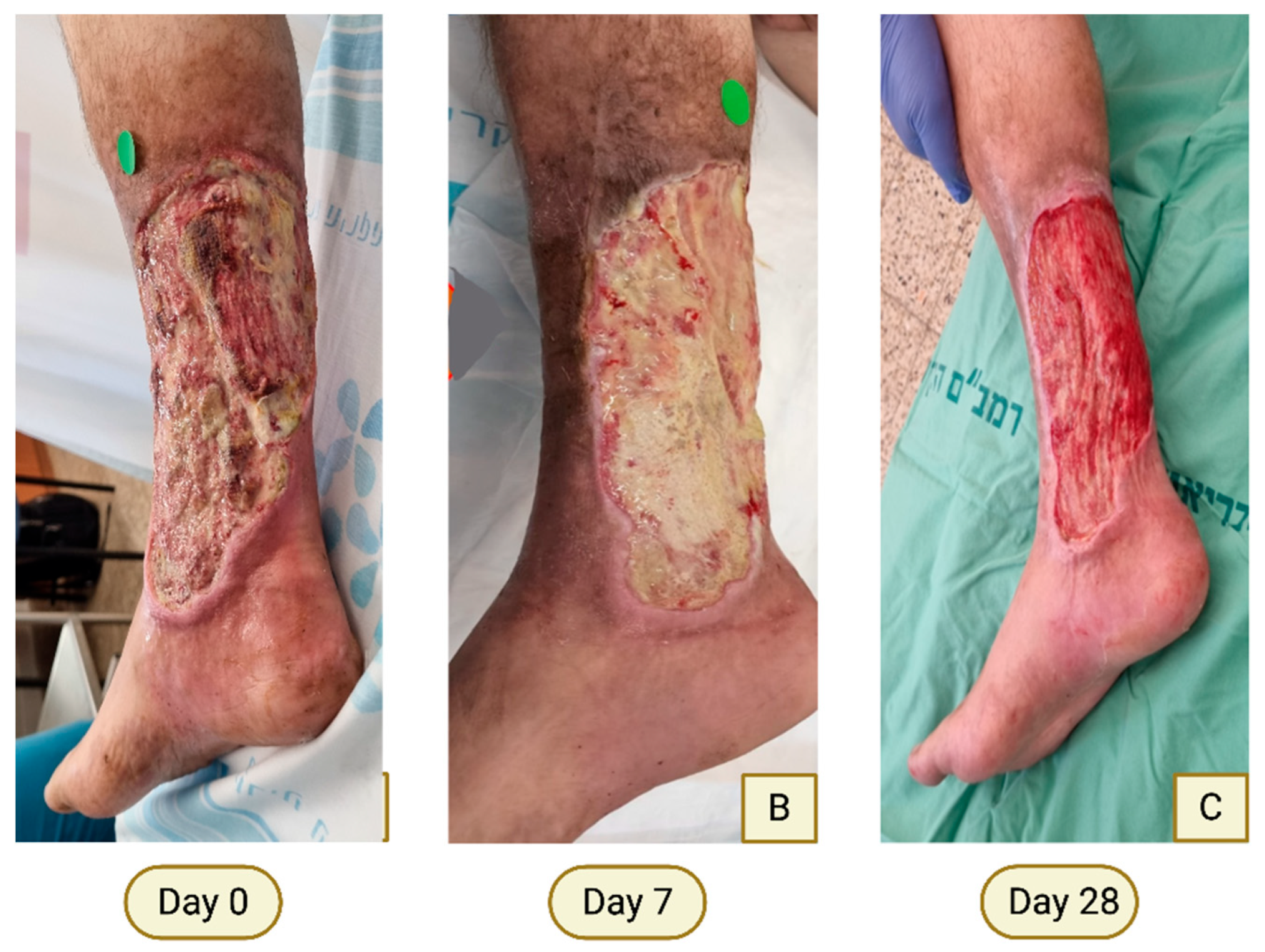
Case 1—Chronic Venous Leg Ulcer in a Patient with APLA Syndrome
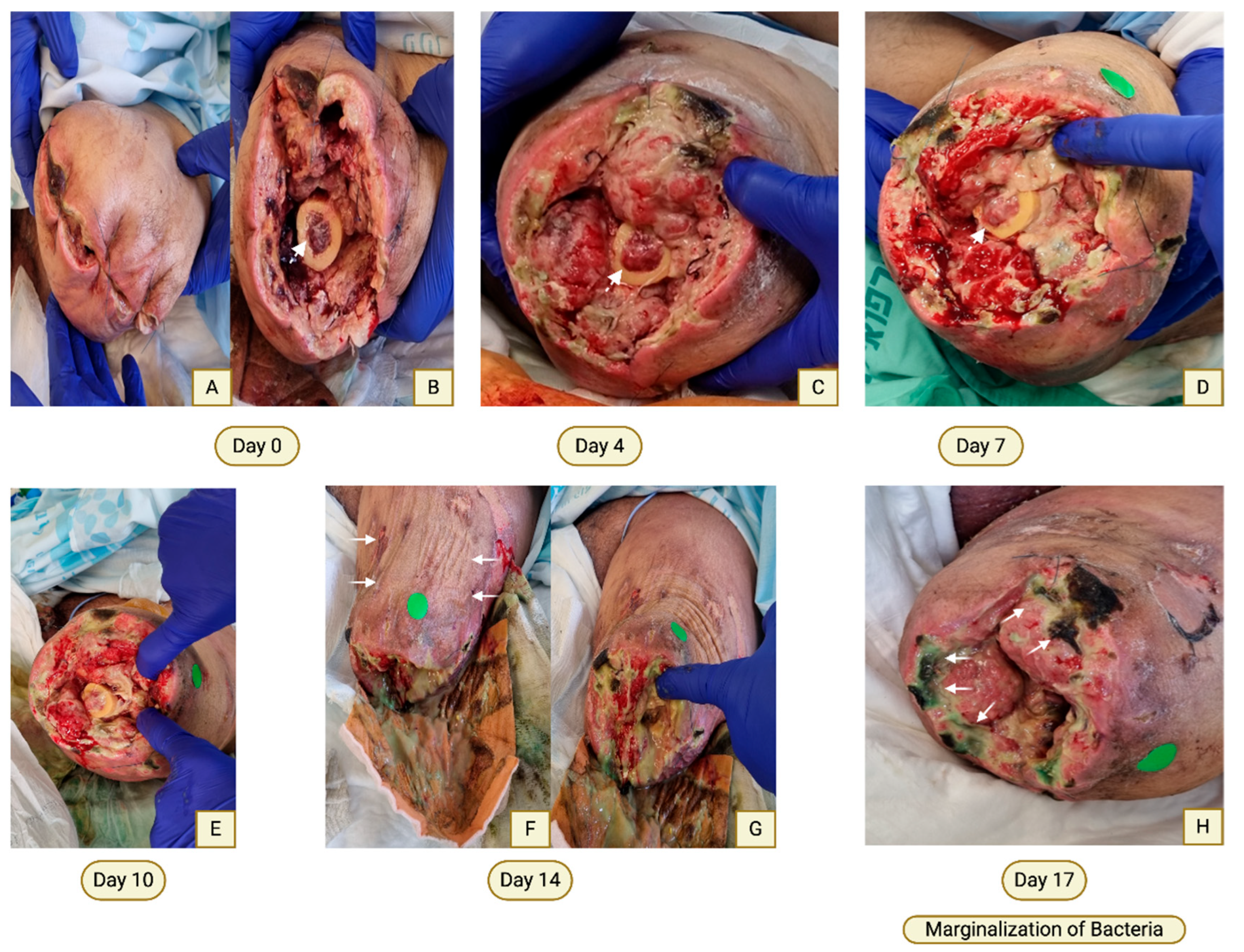
Case 2—Debridement of Marginal Wound (Stump) Necrosis
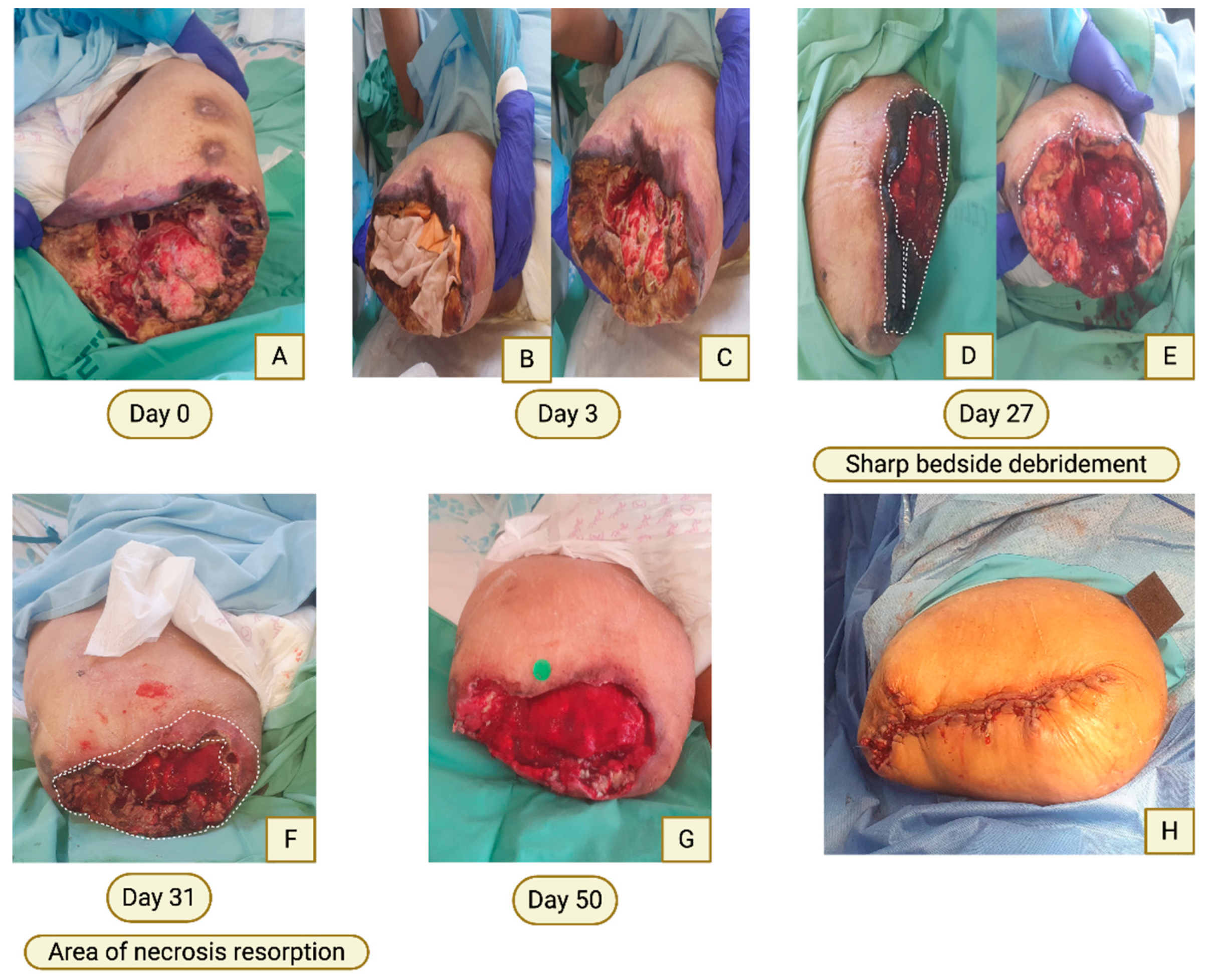
Case 3—Biological Reactivation of a Critically Ischemic Amputation Stump
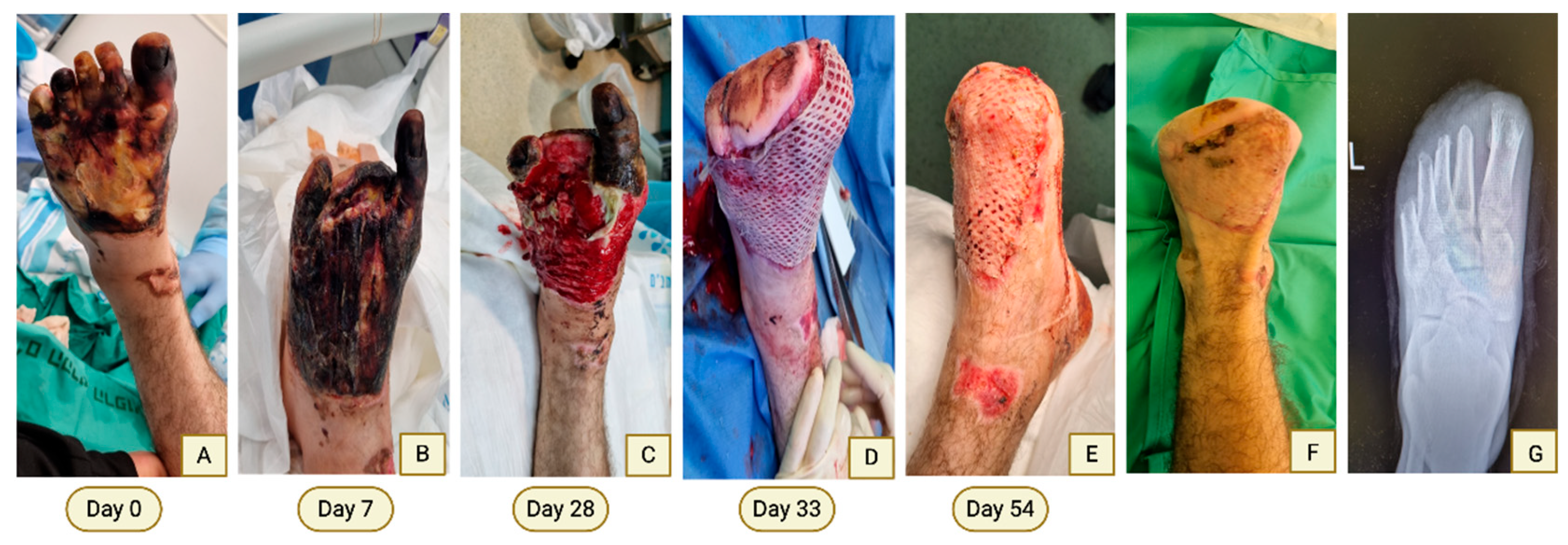
Case 4—Debridement of Extensive Eschar Following Electrical Burn
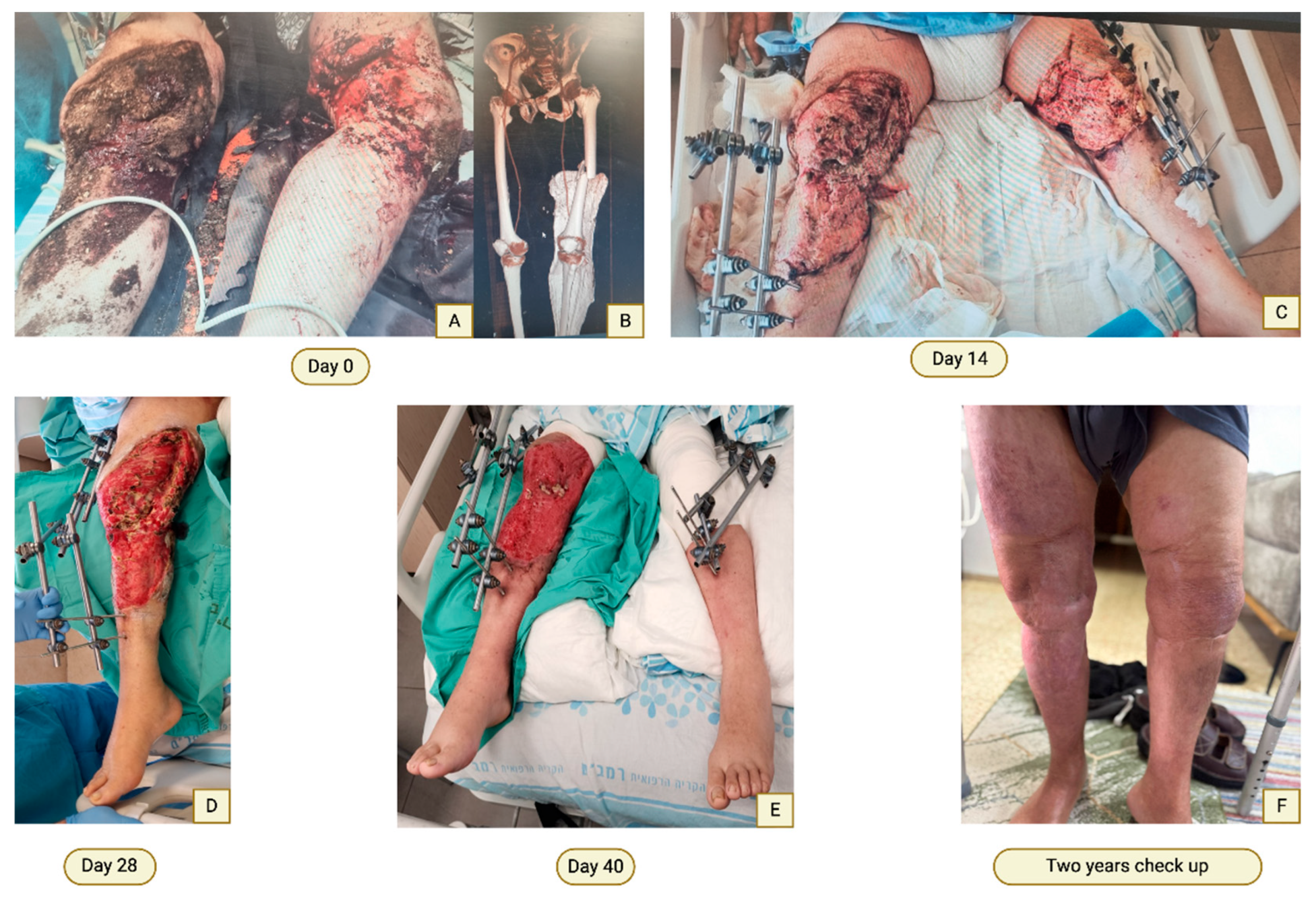
Case 5—Debridement and Revascularization of Extensive Devitalized Tissue Following an Open Fracture
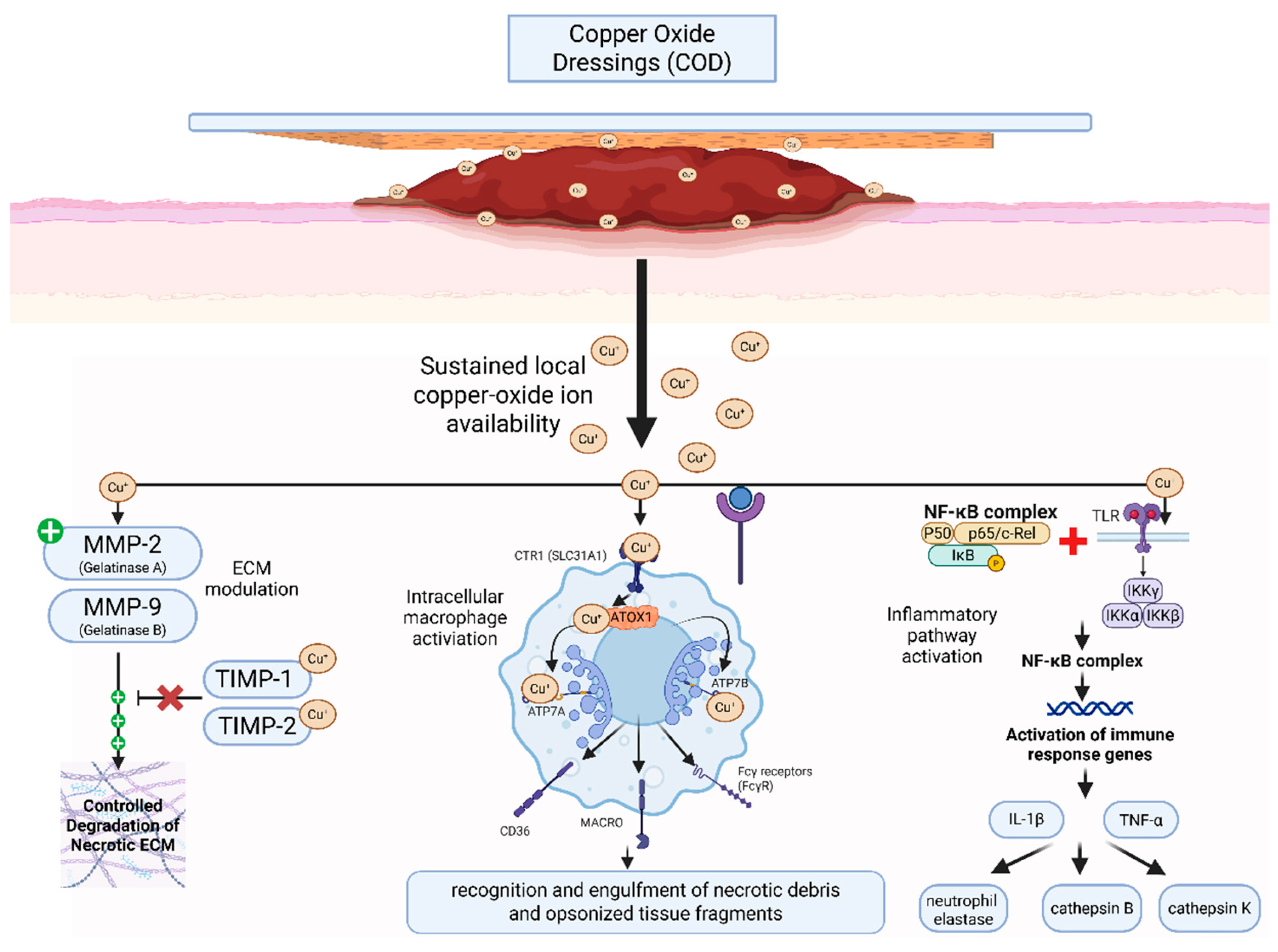
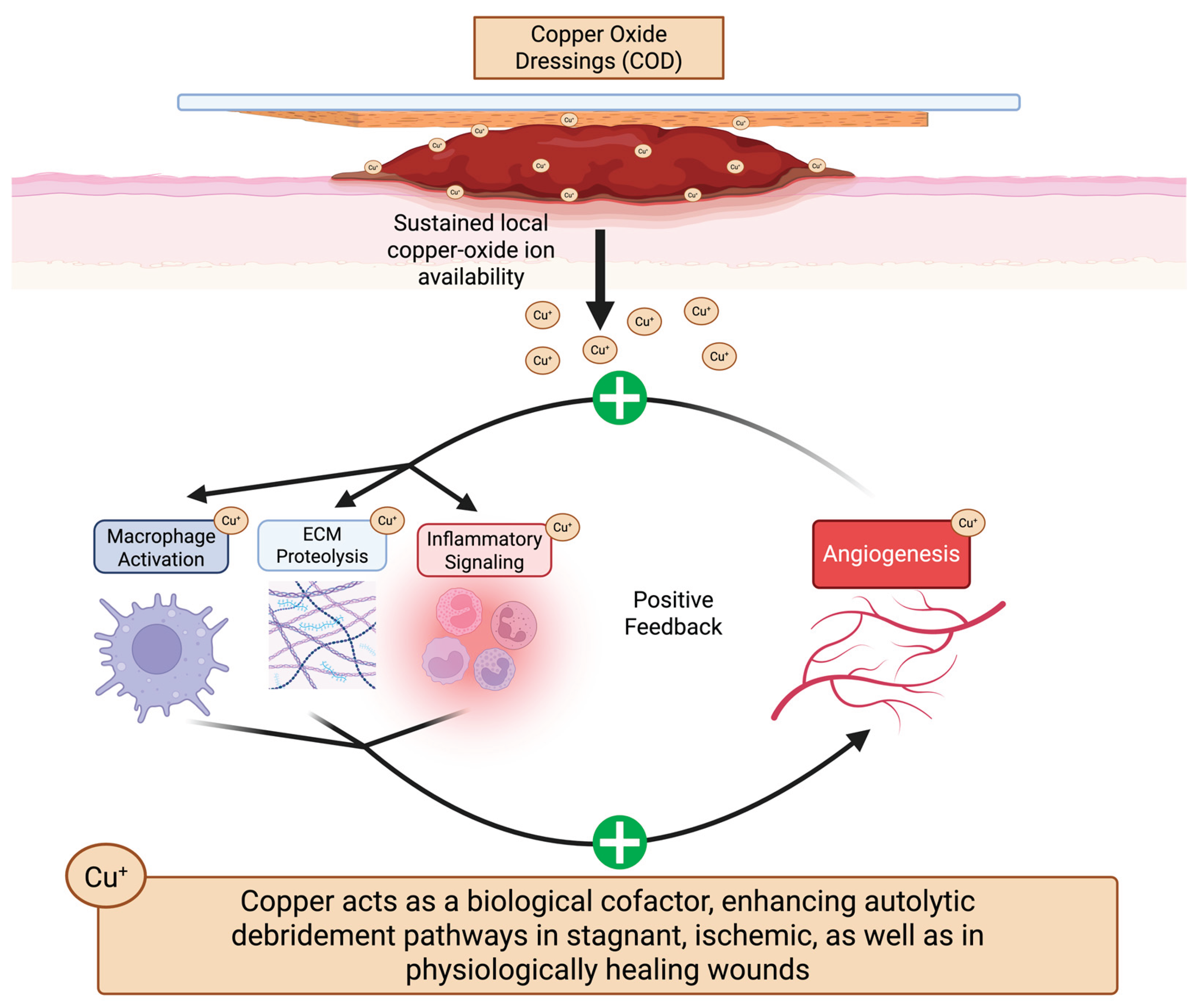
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| APLA | Antiphospholipid Antibody Syndrome |
| ATP7A | ATPase Copper Transporting Alpha |
| ATP7B | ATPase Copper Transporting Beta |
| ATOX1 | Antioxidant Protein 1 (Copper Chaperone) |
| CD36 | Cluster of Differentiation 36 |
| CDP | Copper-Dependent Pathways |
| COD | Copper Oxide Dressings |
| CTR1 (SLC31A1) | Copper Transporter 1 |
| ECM | Extracellular Matrix |
| FcγR | Fc Gamma Receptor |
| HIF-1α | Hypoxia-Inducible Factor 1 Alpha |
| ICU | Intensive Care Unit |
| IKK | IκB Kinase |
| IL-1β | Interleukin 1 Beta |
| MARCO | Macrophage Receptor with Collagenous Structure |
| MMP | Matrix Metalloproteinase |
| MMP-2 | Matrix Metalloproteinase-2 (Gelatinase A) |
| MMP-9 | Matrix Metalloproteinase-9 (Gelatinase B) |
| NF-κB | Nuclear Factor Kappa B |
| NPWT | Negative Pressure Wound Therapy |
| PDGF | Platelet-Derived Growth Factor |
| TIMP | Tissue Inhibitor of Metalloproteinases |
| TIMP-1 | Tissue Inhibitor of Metalloproteinases-1 |
| TIMP-2 | Tissue Inhibitor of Metalloproteinases-2 |
| TNF-α | Tumor Necrosis Factor Alpha |
| VEGF | Vascular Endothelial Growth Factor |
References
- Halim, A.S.; Khoo, T.L.; Saad, A.Z. Wound Bed Preparation from a Clinical Perspective. Indian Journal of Plastic Surgery 2012, 45, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.C.; Tsu, C.L.; Nain, R.A. The Role of Debridement in Wound Bed Preparation in Chronic Wound: A Narrative Review. Annals of Medicine and Surgery 2021, 71, 102876. [Google Scholar] [CrossRef]
- Borkow, G.; Gabbay, J.; Dardik, R.; Eidelman, A.I.; Lavie, Y.; Grunfeld, Y.; Ikher, S.; Huszar, M.; Zatcoff, R.C.; Marikovsky, M. Molecular Mechanisms of Enhanced Wound Healing by Copper Oxide-Impregnated Dressings. Wound Repair and Regeneration 2010, 18, 266–275. [Google Scholar] [CrossRef]
- White, C.; Lee, J.; Kambe, T.; Fritsche, K.; Petris, M.J. A Role for the ATP7A Copper-Transporting ATPase in Macrophage Bactericidal Activity. Journal of Biological Chemistry 2009, 284, 33949–33956. [Google Scholar] [CrossRef] [PubMed]
- Uriu-Adams, J.Y.; Keen, C.L. Copper, Oxidative Stress, and Human Health. Mol. Aspects Med. 2005, 26, 268–298. [Google Scholar] [CrossRef]
- Percival, S. Copper and Immunity. Am. J. Clin. Nutr. 1998, 67, 1064S–1068S. [Google Scholar] [CrossRef] [PubMed]
- Borkow, G.; Cheyne, I.; Melamed, E. Copper Dressings as Part of the Armamentarium in the Fight against Wounds – Much More than an Antimicrobial. Wound Healing Southern Africa 2024, 17, 38–40. [Google Scholar] [CrossRef]
- Cheyne, I.; Rybka, M.; Mazurek, Ł.; Jurak, J.; Konop, M. Shifting the Clinical Paradigm: Copper versus Silver Wound Dressings - Where We Are and What We Are Looking For. J. Biomater. Appl. 2025. [Google Scholar] [CrossRef]
- Amadeh, A.; Mohebbi, N.; Amadeh, Z. others Comparative Efficacy of Autolytic and Collagenase-Based Enzymatic Debridement in Chronic Wound Healing: A Comprehensive Systematic Review. Int. Wound J. 2025, 22, e70177. [Google Scholar] [CrossRef]
- Atkin, L. Understanding Methods of Wound Debridement. British Journal of Nursing 2014, 23, S10–S15. [Google Scholar] [CrossRef]
- Borkow, G.; Melamed, E. The Journey of Copper-Impregnated Dressings in Wound Healing: From a Medical Hypothesis to Clinical Practice. Biomedicines 2025, 13, 562. [Google Scholar] [CrossRef]
- Borkow, G.; Melamed, E. Copper, an Abandoned Player Returning to the Wound Healing Battle. In Recent Advances in Wound Healing; IntechOpen, 2022. [Google Scholar]
- Borkow, G.; Melamed, E. Multi-Function Modality of Copper Dressings in Managing Hard-to-Heal Wounds. J. Wound Care 2026. [Google Scholar] [CrossRef]
- Bogadi, S.; Uddin, M.E.; Karri, V. others Therapeutic Potential of Copper II Oxide in Treating Diabetic Wounds: An Emerging Approach for Enhanced Healing. Acta Diabetol. 2025. [Google Scholar] [CrossRef] [PubMed]
- Melamed, E.; Borkow, G. Continuum of Care in Hard-to-Heal Wounds by Copper Dressings: A Case Series. J. Wound Care 2023, 32, 788–796. [Google Scholar] [CrossRef]
- Melamed, E.; Dabbah, J.; Israel, T.; Kan, I.; Pinzur, M.S.; Roth, T.; Borkow, G. Noninferiority of Copper Dressings Than Negative Pressure Wound Therapy in Healing Diabetic Wounds: A Randomized Clinical Trial. In Adv. Wound Care (New Rochelle).; 2025. [Google Scholar] [CrossRef]
- Melamed, E.; Kiambi, P.; Okoth, D.; Honigber, I.; Tamir, E.; Borkow, G. Healing of Chronic Wounds by Copper Oxide-Impregnated Wound Dressings—Case Series. Medicina (B Aires). 2021, 57, 296. [Google Scholar] [CrossRef]
- Melamed, E.; Rovitsky, A.; Roth, T.; Assa, L.; Borkow, G. Stimulation of Healing of Non-Infected Stagnated Diabetic Wounds by Copper Oxide-Impregnated Wound Dressings. Medicina (B Aires). 2021, 57, 1129. [Google Scholar] [CrossRef]
- Melamed, E.; Rovitsky, A.; Roth, T.; Borkow, G. Anterior Ankle Full Thickness Skin Necrosis Treated with Copper Oxide Dressings without Debridement and Skin Grafting – A Case Report. Arch. Clin. Med. Case Rep. 2022, 06. [Google Scholar] [CrossRef]
- Gorel, O.; Hamuda, M.; Feldman, I.; Kucyn-Gabovich, I. Enhanced Healing of Wounds That Responded Poorly to Silver Dressing by Copper Wound Dressings: Prospective Single Arm Treatment Study. Health Sci. Rep. 2024, 7. [Google Scholar] [CrossRef] [PubMed]
- Chausha Weitman, C.; Roth, T.; Borkow, G. Copper Dressings to the Wound Rescue after Everything Else Failed: Case Report. Arch. Clin. Med. Case Rep. 2022, 06. [Google Scholar] [CrossRef]
- Harries, R.L.; Bosanquet, D.C.; Harding, K.G. Wound Bed Preparation: TIME for an Update. Int. Wound J. 2016, 13, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Giaquinto-Cilliers, M.G.C.; Nair, A.; Von Pressentin, K.B.; others. A “Game of Dressings”: Strategies for Wound Management in Primary Health Care. South African Family Practice 2022, 64, e1–e8. [Google Scholar] [CrossRef] [PubMed]
- Lauerman, M.H.; Scalea, T.M.; Eglseder, W.A. others Efficacy of Wound Coverage Techniques in Extremity Necrotizing Soft Tissue Infections. American Surgeon 2018, 84, 1790–1795. [Google Scholar] [CrossRef] [PubMed]
- Philips, N.; Hwang, H.; Chauhan, S.; Leonardi, D.; Gonzalez, S. Stimulation of Cell Proliferation and Expression of Matrixmetalloproteinase-1 and Interluekin-8 Genes in Dermal Fibroblasts by Copper. Connect. Tissue Res. 2010, 51, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Ogen-Shtern, N.; Chumin, K.; Silberstein, E.; Borkow, G. Copper Ions Ameliorated Thermal Burn-Induced Damage in Ex Vivo Human Skin Organ Culture. Skin Pharmacol. Physiol. 2021, 34, 317–327. [Google Scholar] [CrossRef]
- Caley, M.P.; Martins, V.L.C.; O’Toole, E.A. Metalloproteinases and Wound Healing. Adv. Wound Care (New Rochelle). 2015, 4, 225–234. [Google Scholar] [CrossRef]
- Fukai, T.; Ushio-Fukai, M.; Kaplan, J.H. Copper Transporters and Copper Chaperones: Roles in Cardiovascular Physiology and Disease. American Journal of Physiology-Cell Physiology 2018, 315, C186–C201. [Google Scholar] [CrossRef]
- Stafford, S.L.; Bokil, N.J.; Achard, M.E.S.; Kapetanovic, R.; Schembri, M.A.; McEwan, A.G.; Sweet, M.J. Metal Ions in Macrophage Antimicrobial Pathways: Emerging Roles for Zinc and Copper. Biosci. Rep. 2013, 33. [Google Scholar] [CrossRef]
- Flemming, A. Copper Boosts Pro-Inflammatory State of Macrophages. Nat. Rev. Immunol. 2023, 23, 344–344. [Google Scholar] [CrossRef]
- White, C.; Lee, J.; Kambe, T.; Fritsche, K.; Petris, M.J. A Role for the ATP7A Copper-Transporting ATPase in Macrophage Bactericidal Activity. Journal of Biological Chemistry 2009, 284, 33949–33956. [Google Scholar] [CrossRef]
- Vidak, E.; Javoršek, U.; Vizovišek, M.; Turk, B. Cysteine Cathepsins and Their Extracellular Roles: Shaping the Microenvironment. Cells 2019, 8, 264. [Google Scholar] [CrossRef]
- Chevriaux, A.; Pilot, T.; Derangère, V.; Simonin, H.; Martine, P.; Chalmin, F.; Ghiringhelli, F.; Rébé, C. Cathepsin B Is Required for NLRP3 Inflammasome Activation in Macrophages, Through NLRP3 Interaction. Front. Cell Dev. Biol. 2020, 8. [Google Scholar] [CrossRef] [PubMed]
- Korkmaz, B.; Horwitz, M.S.; Jenne, D.E.; Gauthier, F. Neutrophil Elastase, Proteinase 3, and Cathepsin G as Therapeutic Targets in Human Diseases. Pharmacol. Rev. 2010, 62, 726–759. [Google Scholar] [CrossRef] [PubMed]
- Ganapathy, N.; Venkataraman, S.S.; Daniel, R.; Aravind, R.J.; Kumarakrishnan, V.B. Molecular Biology of Wound Healing. J. Pharm. Bioallied Sci. 2012, 4, S334–7. [Google Scholar] [CrossRef]
- Guo, H.; Jing, L.; Xia, C.; Zhu, Y.; Xie, Y.; Ma, X.; Fang, J.; Wang, Z.; Zuo, Z. Copper Promotes LPS-Induced Inflammation via the NF-КB Pathway in Bovine Macrophages. Biol. Trace Elem. Res. 2024, 202, 5479–5488. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



