1. Introduction
Nanotechnology has emerged as a transformative field in modern science due to its ability to manipulate matter at the nanoscale, where materials often exhibit properties distinct from their bulk counterparts. The term “nanotechnology” was first introduced by Norio Taniguchi in 1974, building upon earlier conceptual foundations proposed in the late 1950s [
1]. Since then, advances in nanoscale science have enabled the development of materials with unique physicochemical characteristics, including enhanced surface area–to–volume ratios, altered optical behavior, and size-dependent thermodynamic properties [
2]. These attributes have positioned nanoparticles as critical components in diverse disciplines such as electronics, optics, biomedicine, materials science, and food technology, where they contribute to innovation in processing, preservation, and quality control [
3].
Among metal oxide nanomaterials, zinc oxide nanoparticles (ZnO NPs) have attracted substantial attention due to their multifunctional nature and broad application potential. Nanoparticles are generally defined as materials with at least one dimension in the range of 1–100 nm, within which quantum confinement and surface effects become prominent [
4]. ZnO NPs exhibit a unique combination of semiconducting, piezoelectric, and pyroelectric properties, alongside high chemical stability and photostability, making them valuable for applications spanning electronics, environmental remediation, and biomedical engineering [
5]. Their wide direct bandgap (~3.37 eV) and high exciton binding energy (~60 meV) at room temperature further support their use in optoelectronic devices such as light-emitting diodes, sensors, and solar cells [
6].
A wide range of synthesis strategies—including chemical precipitation, sol–gel processing, hydrothermal methods, and green synthesis using biological resources—have been reported for ZnO nanoparticles, each offering varying degrees of control over particle size, morphology, and surface characteristics [
7]. This tunability plays a critical role in determining functional performance, particularly in biological and environmental contexts. ZnO NPs have been incorporated into numerous commercial products, including ceramics, pigments, cosmetics, sunscreens, food supplements, and antimicrobial coatings, underscoring their industrial relevance [
8].
In biomedical research, ZnO nanoparticles have demonstrated antimicrobial, anticancer, and drug-delivery-related functionalities. Proposed mechanisms underlying their biological activity include reactive oxygen species generation, Zn
2+ ion release, membrane disruption, and cellular internalization, though the relative contribution of each mechanism remains debated [
9,
10]. While many studies report favorable biocompatibility, others highlight dose-dependent cytotoxicity and environmental concerns, reflecting ongoing controversy regarding their safe and sustainable use [
11].
The primary aim of this review is to critically examine the synthesis methods, physicochemical characteristics, biological interactions, and application-driven performance of zinc oxide nanoparticles. By consolidating current knowledge, addressing divergent findings, and identifying unresolved challenges, this article seeks to provide a balanced perspective on the future development and responsible deployment of ZnO-based nanomaterials across biomedical and technological domains.
2. Structure
Zinc oxide (ZnO) is a well-established II–VI semiconductor composed of zinc and oxygen, elements belonging to Groups II and VI of the periodic table, respectively. Owing to its unique combination of physicochemical properties—including high optical transparency, wide bandgap energy, strong room-temperature luminescence, high electron mobility, and excellent mechanical and thermal stability—ZnO has attracted sustained interest in both fundamental research and applied materials science [
1]. The wide direct bandgap of ZnO, approximately 3.37 eV at room temperature, positions it at the interface between covalent and ionic semiconductors, enabling diverse electronic and optoelectronic applications [
2].
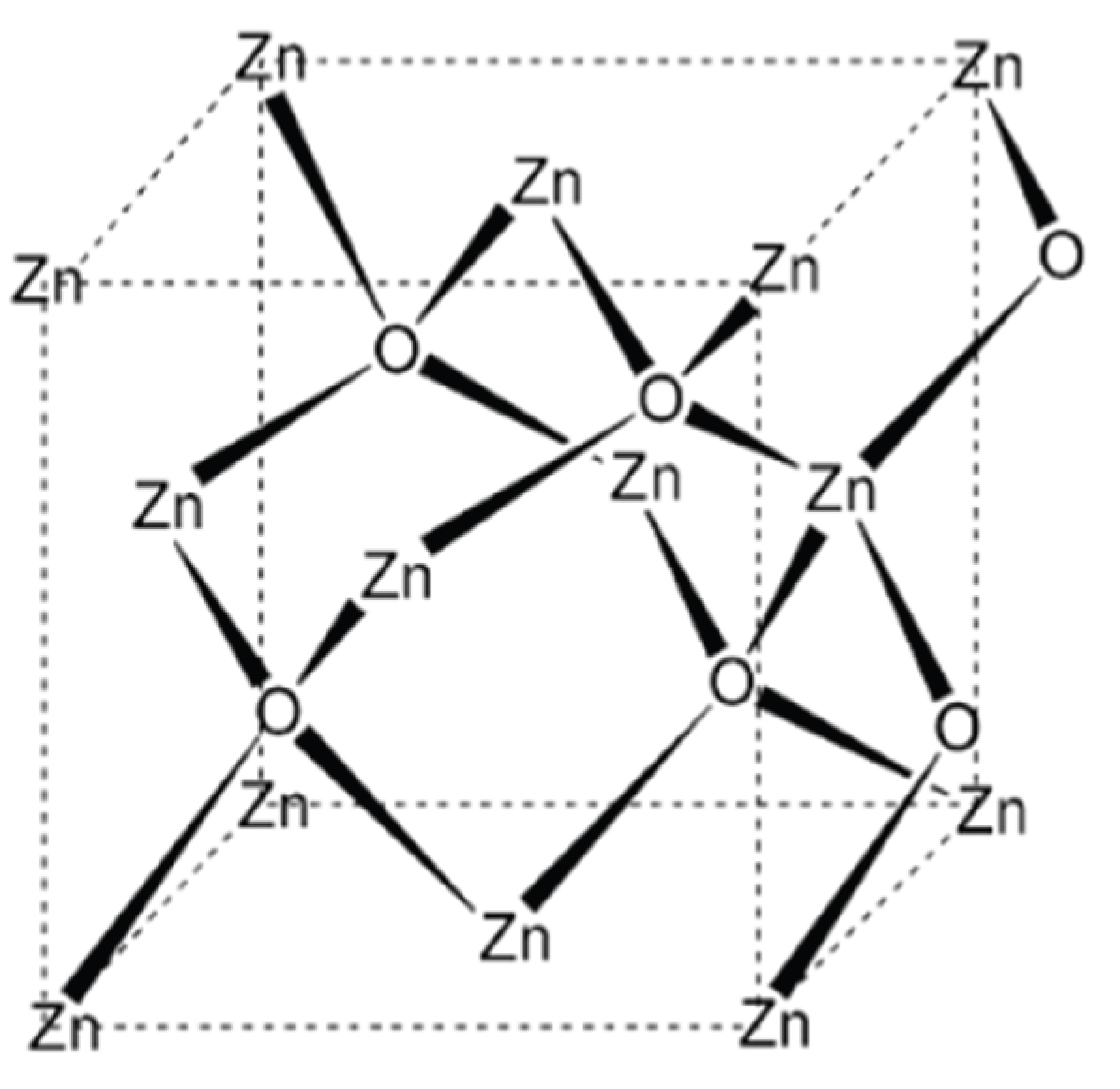
Crystalline ZnO predominantly adopts a hexagonal wurtzite (B4) structure under ambient conditions, which is thermodynamically the most stable phase. The hexagonal unit cell is defined by lattice parameters a ≈ 0.325 nm and c ≈ 0.521 nm [
3]. In this structure, each zinc ion is tetrahedrally coordinated by four oxygen ions, and vice versa, resulting in a non-centrosymmetric lattice characterized by sp
3-type covalent bonding [
4]. This lack of inversion symmetry is responsible for ZnO’s notable piezoelectric and pyroelectric properties, which are critical for sensing and energy-harvesting applications.
In addition to the wurtzite phase, ZnO can also crystallize in the cubic zincblende structure under specific synthesis conditions, although this phase is metastable and less commonly observed in bulk materials [
5]. Naturally occurring ZnO is found as the mineral zincite; however, most commercial ZnO is produced synthetically, typically through the high-temperature oxidation of metallic zinc or zinc-containing ores [
6].
From a chemical standpoint, ZnO is an inorganic compound that is insoluble in water but readily dissolves in dilute acids and bases, reflecting its amphoteric nature. It exhibits a high melting point of approximately 1975 °C, at which decomposition also occurs [
7]. These structural and chemical characteristics underpin the versatility of ZnO and strongly influence the behavior of ZnO nanoparticles, particularly with respect to their stability, surface reactivity, and functional performance in technological and biomedical applications.
Figure 1.
Schematic representation of the crystal structure of zinc oxide (ZnO), illustrating the tetrahedral coordination between Zn2+ and O2− ions within the lattice.
Figure 1.
Schematic representation of the crystal structure of zinc oxide (ZnO), illustrating the tetrahedral coordination between Zn2+ and O2− ions within the lattice.
3. Synthesis of ZnO Nanoparticles
3.1. Precipitation Method
The wet chemical precipitation method is one of the most widely employed approaches for the synthesis of zinc oxide (ZnO) nanoparticles due to its simplicity, cost-effectiveness, and scalability. In this method, a soluble zinc precursor such as zinc nitrate or zinc acetate is dissolved in an aqueous medium, followed by the gradual addition of a base—commonly sodium hydroxide—under constant stirring. The increase in pH induces the formation of a zinc hydroxide precipitate, which subsequently undergoes dehydration upon thermal treatment to yield ZnO nanoparticles [
1].
Reaction parameters such as precursor concentration, pH, temperature, stirring rate, and aging time play a crucial role in controlling particle size, morphology, and crystallinity. The precipitated product is typically separated by centrifugation, thoroughly washed to remove residual ions, and dried. Calcination at elevated temperatures (commonly around 400–500 °C) promotes phase purity and crystallization of ZnO [
2]. Compared with other synthesis routes, the precipitation method offers good control over nanoparticle formation while avoiding complex equipment or extreme reaction conditions.
Characterization of precipitated ZnO nanoparticles is commonly performed using ultraviolet–visible (UV–Vis) spectroscopy to confirm nanoparticle formation through characteristic absorption features. Morphological features and particle size distribution are evaluated using transmission electron microscopy (TEM) and dynamic light scattering (DLS), which together provide complementary insights into nanoscale structure and dispersion behavior [
3]. Numerous studies have reported successful synthesis of ZnO nanoparticles via this approach, demonstrating its reliability for producing nanostructures suitable for optical, environmental, and biomedical applications [
4].
3.2. Chemical Methods
Chemical solution-based methods are widely employed for the synthesis of zinc oxide (ZnO) nanoparticles due to their ability to produce relatively uniform particles under controlled reaction conditions. A commonly reported approach involves the use of zinc acetate dihydrate as the zinc precursor and potassium hydroxide (KOH) as the alkaline source, typically dissolved in an alcoholic medium such as methanol [
1]. In this method, partial dissolution of zinc acetate occurs initially, followed by the gradual addition of the alkaline solution, which promotes nucleation and growth of zinc-containing intermediates.
Upon mixing, the formation of a white precipitate is generally observed, indicating the generation of zinc hydroxide species. Continued stirring and controlled heating facilitate the conversion of these intermediates into ZnO nanoparticles through dehydration and crystallization processes. Prolonged aging of the reaction mixture often leads to sediment formation, which is subsequently separated by centrifugation and washed with deionized water to remove residual reactants and by-products [
2].
The resulting ZnO nanoparticles are frequently described as gel-like or colloidal in nature prior to drying, reflecting their nanoscale dimensions and high surface reactivity. Structural and morphological characterization is typically carried out using X-ray diffraction (XRD) to confirm phase purity and crystallinity, while transmission electron microscopy (TEM) is employed to assess particle size, shape, and dispersion behavior [
3]. Chemical synthesis routes of this type offer good reproducibility and tunability, although careful control of solvent composition, temperature, and reactant ratios is required to avoid agglomeration and achieve consistent nanoparticle properties [
4].
3.3. Sol–Gel Method
The sol–gel method is a versatile and widely reported chemical route for the synthesis of zinc oxide (ZnO) nanostructures, offering good control over composition, homogeneity, and particle size. In this approach, zinc acetate dihydrate is commonly employed as the precursor, while alkaline agents such as sodium hydroxide facilitate hydrolysis and condensation reactions leading to gel formation. Distilled water typically serves as the solvent medium, whereas alcohols such as ethanol are used to promote uniform mixing and regulate the rate of nucleation and growth [
1].
During sol–gel processing, the gradual transformation from a colloidal sol to a three-dimensional gel network enables controlled formation of zinc-containing intermediates, which subsequently convert to ZnO upon aging and thermal treatment. The appearance of a white precipitate is generally indicative of successful nanoparticle formation. Reaction parameters such as precursor concentration, solvent composition, pH, and stirring conditions significantly influence the morphology, crystallinity, and dispersion of the resulting ZnO nanostructures [
2].
The sol–gel method is particularly valued for its low processing temperature, simplicity, and suitability for producing uniform and high-purity ZnO nanoparticles. However, challenges such as particle agglomeration and solvent removal must be carefully managed to ensure reproducibility and scalability. Owing to its flexibility and cost-effectiveness, the sol–gel technique remains a prominent synthesis route for ZnO nanoparticles intended for optical, electronic, and biomedical applications [
3].
3.4. Green Synthesis Approaches for ZnO Nanoparticles
Biological or green synthesis has emerged as an attractive alternative to conventional physical and chemical routes for the production of zinc oxide nanoparticles (ZnO NPs), primarily due to its environmental compatibility, cost-effectiveness, and reduced reliance on hazardous reagents. In this approach, plant-derived extracts are commonly employed as reducing, stabilizing, and capping agents, while zinc salts such as zinc nitrate serve as the metal precursor [
1].
Leaf extracts are frequently used owing to their rich content of phytochemicals, including polyphenols, flavonoids, proteins, and organic acids, which facilitate nanoparticle formation and stabilization. Typically, aqueous plant extracts are prepared through controlled heating and filtration, followed by their reaction with zinc nitrate solutions under mild temperature and stirring conditions. The interaction between zinc ions and bioactive compounds leads to the formation of ZnO nanoparticle precursors, which are subsequently recovered by filtration or centrifugation and subjected to thermal treatment to improve crystallinity and phase purity [
2].
Green-synthesized ZnO nanoparticles are commonly characterized using X-ray diffraction (XRD) to confirm crystal structure, transmission electron microscopy (TEM) to evaluate morphology and particle size, ultraviolet–visible (UV–Vis) spectroscopy to assess optical properties, and Fourier transform infrared (FTIR) spectroscopy to identify functional groups involved in stabilization [
3]. While biological synthesis offers advantages such as biocompatibility and reduced environmental impact, challenges remain in achieving consistent particle size, reproducibility, and precise control over morphology. Nevertheless, continued optimization of biological routes holds promise for the sustainable production of ZnO nanoparticles, particularly for biomedical and environmental applications [
4].
Table 1.
Comparison of Synthesis Methods for Zinc Oxide Nanoparticles.
Table 1.
Comparison of Synthesis Methods for Zinc Oxide Nanoparticles.
| Synthesis Method |
Typical Precursors |
Key Conditions |
Particle Control |
Major Advantages |
Limitations |
Representative Applications |
| Precipitation method |
Zinc nitrate, zinc acetate |
Aqueous medium, alkaline pH, moderate temperature, calcination |
Moderate control over size and morphology |
Simple, cost-effective, scalable |
Agglomeration, limited morphology control |
Antibacterial agents, photocatalysis |
| Chemical (solution-based) method |
Zinc acetate, KOH |
Alcoholic solvents, controlled stirring and heating |
Good control over size and crystallinity |
Reproducible, uniform particles |
Use of organic solvents, post-treatment required |
Drug delivery, optoelectronics |
| Sol–gel method |
Zinc acetate, NaOH |
Hydrolysis–condensation, aging, low-temperature processing |
Excellent compositional homogeneity |
Low processing temperature, high purity |
Gel shrinkage, solvent removal issues |
Thin films, sensors, biomedical coatings |
| Green (biological) method |
Zinc nitrate with plant or microbial extracts |
Mild temperature, aqueous medium, calcination |
Limited but tunable via extract composition |
Eco-friendly, biocompatible, low toxicity |
Reproducibility and size uniformity challenges |
Biomedical, antimicrobial, wound healing |
4. Characterization of ZnO Nanoparticles
Characterization of zinc oxide (ZnO) nanoparticles is essential for determining their size, morphology, crystallinity, surface properties, and stability, all of which directly influence their performance in biomedical, catalytic, electronic, and environmental applications. Reliable characterization enables optimization of synthesis conditions, improves reproducibility, and supports safe and application-specific nanoparticle design.
Microscopic techniques such as transmission electron microscopy (TEM) and scanning electron microscopy (SEM) are widely used to evaluate particle size, shape, and agglomeration. X-ray diffraction (XRD) provides information on crystal structure, phase purity, and crystallite size. In colloidal systems, dynamic light scattering (DLS) is employed to assess hydrodynamic particle size, while zeta potential analysis offers insight into surface charge and colloidal stability.
Spectroscopic methods, including ultraviolet–visible (UV–Vis), Fourier transform infrared (FTIR), and Raman spectroscopy, are used to study optical properties, chemical bonding, and surface functionalization. Elemental composition and surface chemistry are commonly examined using energy-dispersive X-ray spectroscopy (EDS) and X-ray photoelectron spectroscopy (XPS). Additionally, atomic force microscopy (AFM) provides nanoscale surface topography and roughness information.
Collectively, these techniques play a critical role in correlating nanoparticle properties with functional performance and addressing challenges related to stability and toxicity, thereby supporting the effective utilization of ZnO nanoparticles across diverse applications.
4.1. Transmission Electron Microscopy (TEM)
Transmission electron microscopy (TEM) provides high-resolution images of ZnO nanoparticles by transmitting an electron beam through ultrathin samples. It enables direct determination of particle size, shape, morphology, and internal structure, making it one of the most powerful tools for nanoscale characterization.
4.2. Scanning Electron Microscopy (SEM)
Scanning electron microscopy (SEM) is used to examine surface morphology and topography by scanning a focused electron beam across the sample surface. The detection of emitted secondary electrons generates detailed, three-dimensional–like images, allowing assessment of particle shape, surface texture, and agglomeration.
4.3. X-ray Diffraction (XRD)
X-ray diffraction (XRD) is employed to analyze the crystallographic structure, phase purity, and crystallite size of ZnO nanoparticles. Diffraction patterns generated by interaction of X-rays with the crystal lattice provide essential information on structural organization and crystalline phases.
4.4. Energy-Dispersive X-ray Spectroscopy (EDS)
Energy-dispersive X-ray spectroscopy (EDS), typically coupled with SEM or TEM, is used to determine elemental composition. Characteristic X-rays emitted during electron beam interaction are analyzed to identify and confirm the presence of constituent elements.
4.5. Atomic Force Microscopy (AFM)
Atomic force microscopy (AFM) provides high-resolution surface topography and roughness information by scanning the sample with a nanoscale mechanical probe. It enables three-dimensional surface mapping and is particularly useful for analyzing surface features at the atomic or nanometer scale.
In addition to these techniques, methods such as dynamic light scattering (DLS), zeta potential analysis, ultraviolet–visible (UV–Vis) spectroscopy, Fourier transform infrared (FTIR) spectroscopy, Raman spectroscopy, and X-ray photoelectron spectroscopy (XPS) are widely employed to assess particle size distribution, surface charge, optical properties, functional groups, and surface chemistry. Among these, TEM, DLS, and XRD are the most commonly used due to their versatility and reliability. The selection of appropriate characterization techniques depends on the specific properties of interest, and their combined use provides a comprehensive understanding of ZnO nanoparticle behavior across biomedical, catalytic, and electronic applications.
5. Applications of ZnO Nanoparticles
Zinc oxide nanoparticles (ZnO NPs) have attracted significant research interest due to their unique optical, electrical, chemical, and biological properties, enabling their use across a wide range of technological and industrial applications. Their wide bandgap, high exciton binding energy, and strong photocatalytic activity have led to extensive utilization in transparent electronics, ultraviolet (UV) light emitters, piezoelectric devices, chemical sensors, optoelectronic components, solar cells, and spintronic systems.
ZnO nanoparticles are also widely recognized for their photocatalytic efficiency in degrading organic pollutants, making them valuable for environmental remediation and wastewater treatment. Owing to their relative biocompatibility and UV-absorbing capability, ZnO NPs have found increasing application in the pharmaceutical and cosmetic industries, particularly as UV filters in sunscreens and as antimicrobial agents in topical formulations. In addition, they are employed as pigments and functional additives in paints, concrete, rubber, plastics, and textiles, where they enhance durability, UV resistance, and antimicrobial performance.
The versatility of ZnO nanoparticles has further expanded their use in biomedical and biological fields, including drug delivery, wound healing, and antimicrobial coatings. Collectively, these diverse applications highlight the multifunctional nature of ZnO nanoparticles and underscore their growing importance in advancing materials science, environmental technologies, and healthcare-related innovations.
5.1. Antibacterial Activity
Zinc oxide nanoparticles (ZnO NPs) have attracted considerable attention due to their broad-spectrum antibacterial activity against both Gram-positive and Gram-negative bacteria. Model organisms such as Staphylococcus aureus and Escherichia coli are frequently employed to evaluate antibacterial efficacy, alongside other clinically relevant species including Bacillus subtilis, Enterococcus faecalis, Pseudomonas aeruginosa, Vibrio cholerae, and Proteus vulgaris. The antibacterial performance of ZnO NPs is strongly influenced by particle size, concentration, morphology, and surface defects, which collectively modulate their interaction with bacterial cells.
Several studies report that green-synthesized ZnO nanoparticles exhibit enhanced antibacterial activity compared with chemically synthesized counterparts, likely due to improved surface reactivity and the presence of bio-derived capping agents. Microscopic observations indicate that ZnO NP exposure can lead to bacterial membrane disruption, structural deformation, and eventual cell death, with higher nanoparticle concentrations causing more pronounced damage. Although multiple mechanisms—such as reactive oxygen species generation, Zn2+ ion release, and membrane interaction—have been proposed, the precise antibacterial pathways remain incompletely understood. Owing to their strong antibacterial potential, ZnO nanoparticles are being explored for use in antimicrobial coatings, wound dressings, medical devices, and personal-care formulations such as ointments and lotions.
5.2. Drug Delivery
Advances in nanotechnology have enabled the development of more efficient and targeted drug delivery systems, with ZnO nanoparticles emerging as promising carriers due to their tunable surface properties, pH sensitivity, and relative biocompatibility. Numerous studies have demonstrated the effectiveness of ZnO NPs in delivering therapeutic agents for cancer treatment and gene therapy. Surface modification with biopolymers such as chitosan has been shown to improve nanoparticle stability, cellular uptake, and controlled drug release.
ZnO-based nanocarriers have been successfully employed for the delivery of chemotherapeutic agents, including doxorubicin, often exhibiting pH-responsive drug release behavior that enhances cytotoxicity in tumor cells while reducing off-target effects. Targeted delivery strategies, such as folic acid–functionalized ZnO nanostructures, have further improved selective uptake by cancer cells. In addition, ZnO nanoparticles have demonstrated potential in gene delivery and gene silencing applications, where their surface chemistry facilitates efficient binding and transport of genetic material. Recent studies also highlight the use of ZnO nanostructures in photodynamic therapy, where reactive oxygen species generation contributes to anticancer activity. Collectively, these findings underscore the growing potential of ZnO nanoparticles as multifunctional platforms for drug and gene delivery applications.
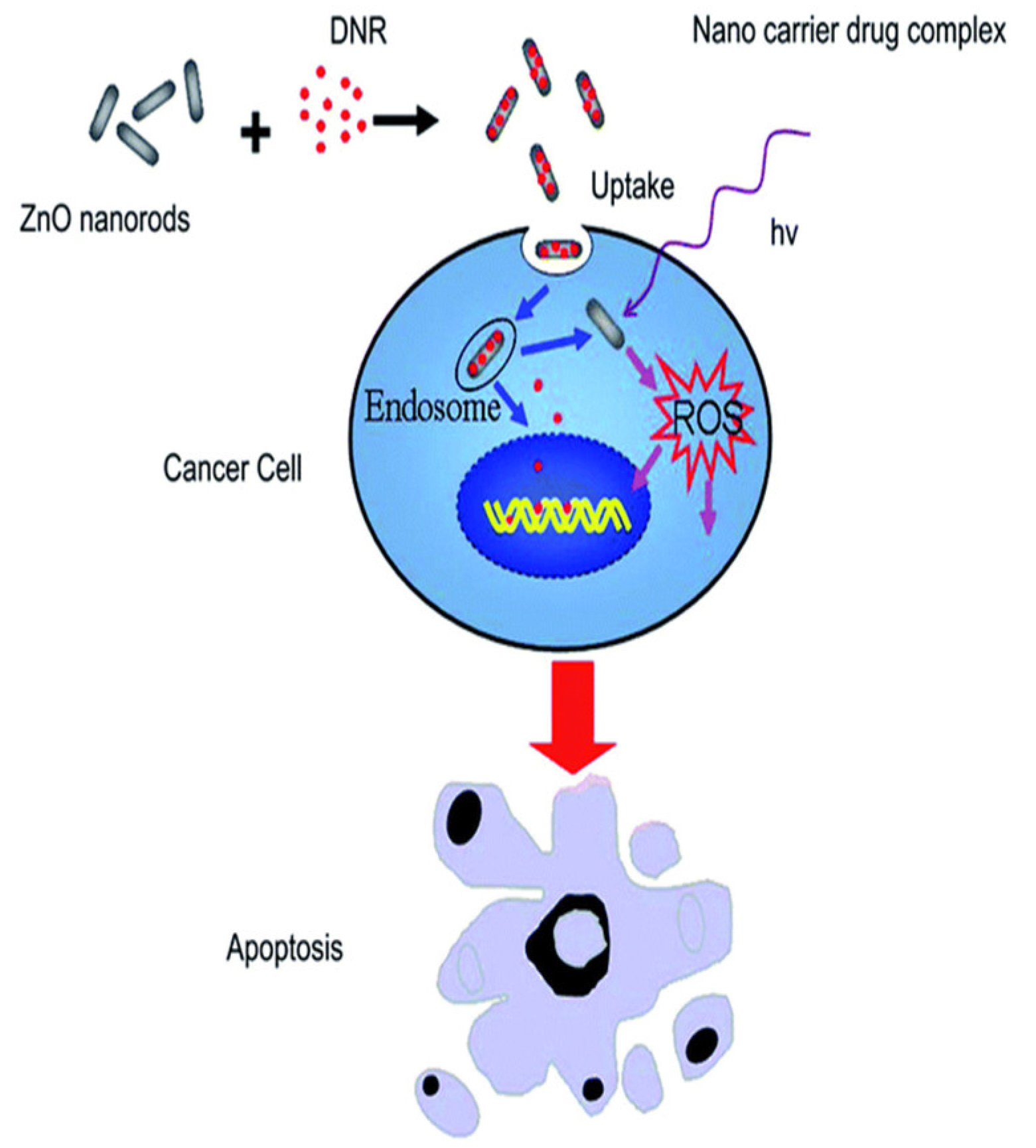
Figure 2.
Schematic representation of ZnO nanorod-assisted drug delivery and ROS-induced apoptosis in cancer cells.
Figure 2.
Schematic representation of ZnO nanorod-assisted drug delivery and ROS-induced apoptosis in cancer cells.
5.3. Anticancer Activity
Conventional cancer treatments such as chemotherapy, radiotherapy, and surgery remain effective but are often associated with significant off-target toxicity. Nanomaterial-based strategies have therefore gained attention for their potential to improve selectivity, biocompatibility, and therapeutic efficacy. Among these, zinc oxide nanoparticles (ZnO NPs) have emerged as promising anticancer agents owing to their tunable surface chemistry, biodegradability, and ability to induce cancer-selective cytotoxicity.
Multiple in vitro studies report dose-dependent reductions in cell viability across diverse cancer cell lines, including MCF-7, A549, HL-60, and HepG2, while exhibiting comparatively lower toxicity toward normal cells. Proposed mechanisms underlying ZnO NP-mediated anticancer effects include intracellular dissolution of Zn2+ ions, enhanced reactive oxygen species (ROS) generation, mitochondrial membrane depolarization, and activation of apoptosis-related signaling pathways. Mechanistic investigations in HepG2 cells have demonstrated loss of mitochondrial membrane potential, altered Bcl-2/Bax ratios, cytochrome-c release, and caspase-9 activation, culminating in apoptosis. Activation of stress-related pathways and p53 phosphorylation has also been reported. Although these findings highlight the therapeutic potential of ZnO NPs, further studies are required to establish in vivo safety, pharmacokinetics, and long-term effects.
5.4. Anti-Inflammatory Activity
Inflammation is a complex biological response to infection, injury, or irritation and plays a critical role in tissue repair. However, chronic or dysregulated inflammation contributes to a wide range of pathological conditions. Zinc oxide nanoparticles have attracted growing interest for their anti-inflammatory properties, particularly in dermatological applications.
Atopic dermatitis (AD), a chronic inflammatory skin disorder, has been extensively studied in this context. Recent investigations demonstrate that textiles functionalized with ZnO NPs can reduce oxidative stress and improve clinical symptoms in AD patients, including pruritus and sleep disturbance. Experimental models indicate that nano-sized ZnO penetrates inflamed skin more effectively than bulk ZnO and significantly reduces pro-inflammatory cytokine expression, resulting in enhanced anti-inflammatory outcomes. Additional studies using macrophage cell lines have shown that biosynthesized ZnO NPs suppress inflammatory mediators such as nitric oxide, iNOS, COX-2, and pro-inflammatory cytokines in a dose-dependent manner. These findings suggest that ZnO nanoparticles hold promise as anti-inflammatory agents, although controlled clinical studies remain necessary.
5.5. Wound Healing
Wound healing is a dynamic and multistep biological process involving hemostasis, inflammation, proliferation, and tissue remodeling. Impaired healing is commonly associated with conditions such as diabetes, infection, and excessive inflammation, necessitating advanced wound-care materials. ZnO nanoparticles have been increasingly incorporated into wound dressings due to their antimicrobial activity, biocompatibility, and ability to promote cellular regeneration.
Recent studies report that ZnO NP-functionalized polymeric scaffolds enhance fibroblast migration, proliferation, and angiogenesis, leading to accelerated wound closure in animal models without inducing significant inflammatory responses. Composite wound dressings containing ZnO NPs have demonstrated strong antibacterial activity against Staphylococcus aureus and Escherichia coli, alongside improved adhesion and proliferation of endothelial progenitor and keratinocyte cells. The release of Zn2+ ions is believed to stimulate cell migration and tissue regeneration, while ROS generation contributes to antimicrobial efficacy. Advanced hybrid systems combining ZnO nanostructures with hydrogels and graphene-based materials further enhance antibacterial performance through synergistic mechanisms. Collectively, these studies highlight the potential of ZnO nanoparticle-based materials as multifunctional platforms for wound-healing applications.
6. Conclusion
This review summarizes the diverse synthesis strategies, structural characteristics, and application domains of zinc oxide (ZnO) nanostructures, highlighting their versatility as multifunctional materials in contemporary nanotechnology. By integrating insights from materials science and biological research, the article provides a comprehensive perspective on how ZnO nanoparticles can be tailored for specific technological and biomedical applications. Particular attention has been given to synthesis-dependent properties, biocompatibility, and toxicity considerations, which are critical for responsible and effective deployment.
Recent advances in green and bio-mediated synthesis approaches have further increased interest in ZnO nanoparticles by offering environmentally sustainable routes with improved functional performance. In optoelectronic applications, especially as electron transport layers in dye-sensitized and perovskite solar cells, ZnO nanoparticles have demonstrated considerable potential, with performance enhancements achievable through doping, surface functionalization, and interface engineering. In parallel, growing research efforts have focused on the biological activity of ZnO nanoparticles, including their roles in antibacterial, anticancer, anti-inflammatory, drug delivery, and bio-imaging applications. These effects are largely attributed to controlled Zn2+ ion release, reactive oxygen species generation, and activation of apoptosis-related pathways, while drug-carrier systems have shown improved therapeutic bioavailability and efficacy.
Beyond healthcare and energy applications, emerging studies also highlight the potential of ZnO-based nanomaterials in agriculture, particularly as nano-fertilizers to enhance crop growth and productivity. Overall, ZnO nanoparticles exhibit a broad spectrum of applications across science and industry. Continued interdisciplinary research, with emphasis on safety assessment, standardization, and scalability, will be essential to fully realize their potential in sustainable technological and biomedical innovations.
References
- Mohandas, A.; Sudheesh Kumar, P. T.; Raja, B.; Lakshmanan, V. K.; Jayakumar, R. Exploration of Alginate Hydrogel/Nano Zinc Oxide Composite Bandages for Infected Wounds. Int. J. Nanomed. 2015, 10 (Suppl. 1), 53–66. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Khan, M.; et al. Zinc Oxide Nanoparticles in Agriculture: Applications and Implications for Crop Improvement and Soil Health. J. Agric. Food Chem. 2023, 71(15), 5678–5690. [Google Scholar]
- Wiegand, C.; Hipler, U. C.; Boldt, S.; Strehle, J.; Wollina, U. Skin-Protective Effects of a Zinc Oxide–Functionalized Textile and Its Relevance for Atopic Dermatitis. Clin. Cosmet. Investig. Dermatol. 2013, 6, 115–121. [Google Scholar]
- Selvakumari, D.; Deepa, R.; Mahalakshmi, V.; Subhashini, P.; Lakshminarayan, N. Synthesis and Characterization of Zinc Oxide Nanoparticles and Their Antimicrobial Activity against Bacillus subtilis and Escherichia coli. J. Eng. Appl. Sci. 2015, 10(12), 5418–5421. [Google Scholar]
- Daeschlein, G. Antimicrobial and Antiseptic Strategies in Wound Management. Int. Wound J. 2013, 10 (Suppl. 1), 9–14. [Google Scholar] [CrossRef]
- Ghorbani, H. R.; Mehr, F. P.; Pazoki, H.; Rahmani, B. M. Synthesis of ZnO Nanoparticles by Precipitation Method. Orient. J. Chem. 2015, 31(2), 1219–1221. [Google Scholar] [CrossRef]
- Chhabra, H.; Deshpande, R.; Kanitkar, M.; Jaiswal, A.; Kale, V. P.; Bellare, J. R. A Nano Zinc Oxide–Doped Electrospun Scaffold Improves Wound Healing in a Rodent Model. RSC Adv. 2016, 6(2), 1428–1439. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, B.; Jiang, H.; Wang, C.; Wang, H.; Wang, X. A Strategy for ZnO Nanoparticles as Anticancer Agents. Biomaterials 2011, 32(7), 1906–1914. [Google Scholar] [CrossRef]
- Hernández-Alonso, M. D.; Fresno, F.; Suárez, S.; Coronado, J. M. Development of Alternative Photocatalysts to TiO2: Challenges and Opportunities. Energy Environ. Sci. 2009, 2(12), 1231–1257. [Google Scholar] [CrossRef]
- Nomura, K.; Ohta, H.; Ueda, K.; Kamiya, T.; Hirano, M.; Hosono, H. Thin-Film Transistor Fabricated in Single-Crystalline Transparent Oxide Semiconductor. Science 2003, 300(5623), 1269–1272. [Google Scholar] [CrossRef]
- Kumar, M.; Singh, R.; et al. Toxicity and Environmental Impact of Zinc Oxide Nanoparticles: A Review. Environ. Sci.: Nano 2023, 10(3), 456–468. [Google Scholar]
- Ilves, M.; Palomäki, J.; Vippola, M.; Lehto, M.; Savolainen, K.; Savinko, T.; Alenius, H. Topically Applied ZnO Nanoparticles Suppress Allergen-Induced Skin Inflammation but Induce IgE Production in an Atopic Dermatitis Mouse Model. Part. Fibre Toxicol. 2014, 11(1), 38. [Google Scholar] [CrossRef]
- Premanathan, M.; Karthikeyan, K.; Jeyasubramanian, K.; Manivannan, G. Selective Toxicity of ZnO Nanoparticles toward Gram-Positive Bacteria and Cancer Cells through Apoptosis. Nanomedicine 2011, 7(2), 184–192. [Google Scholar] [CrossRef] [PubMed]
- Ostrovsky, S.; Kazimirsky, G.; Gedanken, A.; Brodie, C. Selective Cytotoxic Effect of ZnO Nanoparticles on Glioma Cells. Nano Res. 2009, 2(11), 882–890. [Google Scholar] [CrossRef]
- Nagajyothi, P. C.; Cha, S. J.; Yang, I. J.; Sreekanth, T. V. M.; Kim, K. J.; Shin, H. M. Antioxidant and Anti-Inflammatory Activities of ZnO Nanoparticles Synthesized Using Polygala tenuifolia Root Extract. J. Photochem. Photobiol. B 2015, 146, 10–17. [Google Scholar] [CrossRef]
- Padmavathy, N.; Vijayaraghavan, R. Enhanced Bioactivity of ZnO Nanoparticles—An Antimicrobial Study. Sci. Technol. Adv. Mater. 2008, 9(3), 035004. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.; Mehta, P.; et al. Zinc Oxide Nanoparticles as a Promising Agent for Cancer Therapy: Mechanisms and Recent Advances. J. Cancer Res. Ther. 2023, 19(2), 345–356. [Google Scholar]
- Augustine, R.; Dominic, E. A.; Reju, I.; Kaimal, B.; Kalarikkal, N.; Thomas, S. Electrospun Polycaprolactone Membranes Incorporated with ZnO Nanoparticles as Skin Substitutes with Enhanced Fibroblast Proliferation and Wound Healing. RSC Adv. 2014, 4(93), 51528–51536. [Google Scholar] [CrossRef]
- Rasmussen, J. W.; Martinez, E.; Louka, P.; Wingett, D. G. Zinc Oxide Nanoparticles for Selective Destruction of Tumor Cells and Potential for Drug Delivery Applications. Expert Opin. Drug Deliv. 2010, 7(9), 1063–1077. [Google Scholar] [CrossRef]
- Mitra, S.; Subia, B.; Patra, P.; Chandra, S.; Debnath, N.; Das, S.; Banerjee, R.; Kundu, S. C.; Pramanik, P.; Goswami, A. Porous ZnO Nanorods for Targeted Delivery of Doxorubicin: In Vitro and In Vivo Response. J. Mater. Chem. 2012, 22(45), 24145–24154. [Google Scholar] [CrossRef]
- Dhoke, S. K. Synthesis of Nano-ZnO by Chemical Method and Its Characterization. Results Chem. 2023, 5, 100792. [Google Scholar] [CrossRef]
- Sharma, A.; Gupta, S.; et al. Green Synthesis of Zinc Oxide Nanoparticles Using Plant Extracts and Their Biomedical Applications. J. Nanomater. 2023, Article ID 1234567. [Google Scholar]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Kaus, N. H. M.; Ann, L. C.; Bakhori, S. K. M.; Hasan, H.; Mohamad, D. Review on Zinc Oxide Nanoparticles: Antibacterial Activity and Toxicity Mechanism. Nano-Micro Lett. 2015, 7(3), 219–242. [Google Scholar] [CrossRef]
- Smijs, T. G.; Pavel, S. Titanium Dioxide and Zinc Oxide Nanoparticles in Sunscreens: Focus on Their Safety and Effectiveness. Nanotechnol. Sci. Appl. 2011, 4, 95–112. [Google Scholar] [CrossRef] [PubMed]
- Nakada, T.; Hirabayashi, Y.; Tokado, T.; Ohmori, D.; Mise, T. Novel Device Structure for Cu(In,Ga)Se2 Thin-Film Solar Cells Using Transparent Conducting Oxide Contacts. Sol. Energy 2004, 77(6), 739–747. [Google Scholar] [CrossRef]
- Sharma, V.; Anderson, D.; Dhawan, A. Zinc Oxide Nanoparticles Induce Oxidative DNA Damage and Apoptosis in Human Cells. Apoptosis 2012, 17(8), 852–870. [Google Scholar] [CrossRef] [PubMed]
- Verma, P.; Kumar, S.; et al. Zinc Oxide Nanoparticles in Wound Healing: Mechanisms and Applications. Nanomedicine 2023, 45, 102567. [Google Scholar]
- Salem, W.; Leitner, D. R.; Zingl, F. G.; Schratter, G.; Prassl, R.; Goessler, W.; Schild, S. Antibacterial Activity of Silver and Zinc Nanoparticles against Vibrio cholerae and Enterotoxic Escherichia coli. Int. J. Med. Microbiol. 2015, 305(1), 85–95. [Google Scholar] [CrossRef]
- Wang, Z. L. Zinc Oxide Nanostructures: Growth, Properties, and Applications. J. Phys.: Condens. Matter 2004, 16(25), R829–R858. [Google Scholar] [CrossRef]
- Liang, Y.; Wang, M.; Zhang, Z.; Ren, G.; Liu, Y.; Wu, S.; Shen, J. Fabrication of Porous ZnO Nanostructures for Photocatalytic Degradation of Organic Pollutants. Chem. Eng. J. 2019, 378, 122043. [Google Scholar] [CrossRef]
- Zhang, Y.; Nayak, T. R.; Hong, H.; Cai, W. Biomedical Applications of Zinc Oxide Nanomaterials. Curr. Mol. Med. 2013, 13(10), 1633–1645. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |