Submitted:
05 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Theoretical Basis
- When → modulation overcomes arousal → less
- When → arousal dominates → greater
- High = rapid changes (young neuroplasticity)
- Low = slow changes (adult neuroplasticity)
Methodological Note
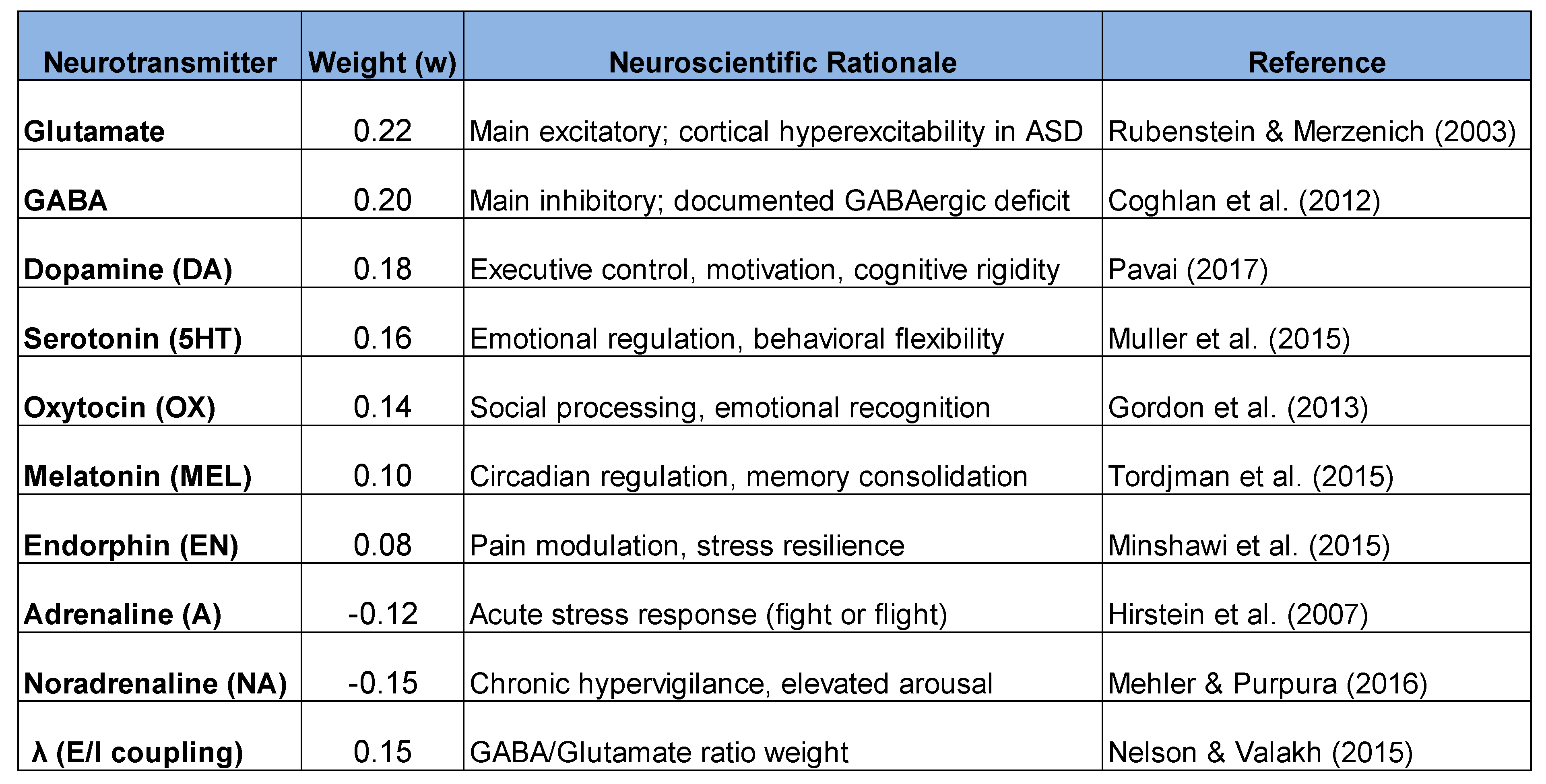
- GABA and glutamate are the major sensory modulators in ASD.
- Dopamine plays a central role in executive functions.
- Oxytocin influences social skills.
- Serotonin influences rigidity and mood.
- Adrenaline and noradrenaline increase externalized symptoms.
- Glutamate - Excitatory structural neurotransmitter , “accelerator” :
- 2.
- GABA - Structural inhibitory neurotransmitter “brake” :
- 3.
- Dopamine → executive control and reward-based learning
- 4.
- Oxytocin → social
- 5.
- Serotonin → emotional regulation
- 6.
- Endorphins → tolerance to discomfort
- 7.
- Melatonin → circadian stability
- 8.
- Norepinephrine → hypervigilance
- 9.
- Adrenaline → fight or flight
- Strength recognized in the literature
- Direct impact on the autistic phenotype
- Correlation with sensory, executive, and social symptoms
- Contemporary models of ASD ( imbalance, arousal model, predictive coding)
- a)
-
Glutamate
- Main excitatory neurotransmitter of the central nervous system.
- • Responsible for 80-90% of excitatory synaptic transmission in the cortex.
-
In ASD, elevated glutamate levels are associated with:
- Cortical hyperexcitability
- Sensory overload
- Difficulties in attentional filtering
- Increased susceptibility to epileptic seizures (comorbidity in ~30% of cases)
- • In proper balance with GABA, it allows for efficient information processing; in excess, it generates “neural noise” and dysregulation.
- b)
- Dopamine
- c)
-
• Regulates motivation, reward, and goal setting.
- It acts as a “selector” of behaviors, facilitating focus, learning, and reinforcement.
- At balanced levels, it sustains concentration and decision-making; in excess, it generates impulsivity; in deficiency, apathy and slowness.
- d)
- Serotonin
- e)
- • Stabilizes mood, modulates anxiety, and regulates impulsivity.
- f)
- • Acts on the feeling of inner security and stress tolerance.
- g)
- • Contributes to cognitive flexibility and smooth transitions between mental states.
- h)
- Melatonin
- i)
-
• Synchronizes the sleep–wake cycle and regulates circadian rhythms.
- It influences memory, information consolidation, and the quality of neuronal rest.
- It indirectly affects sensory sensitivity, as adequate sleep reduces hyper-reactivity.
- j)
- Oxytocin
- k)
- • Modulates social bonds, emotional recognition, and sense of belonging.
- l)
- • Increases trust and reduces the perception of threat in social interactions.
- m)
- • Is fundamental for pro-social behaviors and emotional reading.
- n)
- Norepinephrine
- o)
-
• Adjusts alertness, vigilance, and responsiveness to the environment.
- It acts as an “attention tuner,” increasing focus on relevant cues.
- Very high levels produce anxiety; low levels impair sustained attention.
- p)
- Endorphins
- q)
- • Modulate physical and emotional pain, providing a feeling of relief and well-being.
- r)
- • Facilitate resilience, reduce reactivity to stress, and strengthen disposition.
- s)
- • Act as a natural emotional “buffer”.
- t)
- GABA
- u)
- • Main inhibitory neurotransmitter of the nervous system.
- v)
- • Reduces hyperexcitability and modulates sensory overload.
- w)
- • Important for calmness, filtering stimuli, and controlling physiological anxiety.
- x)
- Adrenaline
- y)
- • Activates rapid coping responses (fight or flight).
- z)
- • Increases energy, processing speed, and motor reactions.
- aa)
- • In excess, it produces hyperalertness; in balance, it aids motivation and readiness.
- = 0.0 → Totally “invisible” autism (impossible in practice)
- = 0.3 → Low phenotypic expression (intense masking)
- = 0.6 → Moderate expression (authenticity with regulation)
- = 1.0 → Maximum expression (no modulation or regulation)
- Risk polymorphisms ( e.g. , CHD8, SHANK3, CNTNAP2)
- Genomic architecture (de novo variants, SNPs )
- Structural neural connectivity (white matter volume, synaptic density)
- = 0.5 → ASD level 1 (minimal support, “classic Asperger’s”)
- = 0.7 → ASD level 1-2 (moderate support)
- = 1.0 → ASD level 3 (substantial support)
- High (2.5–3.5) → High neuroplasticity (children, adolescents)
- Average (1.8–2.2) → Adult neuroplasticity (20–45 years)
- Low (1.0–1.5) → Reduced neuroplasticity (elderly, >60 years)
- (Prefrontal dopamine)
- (Serotonin)
- (Melatonin)
- (Oxytocin)
- (Endorphin)
- (Sensory inhibition)
- = 0.2 → Low-activity modulating systems (dysregulation)
- = 0.5 → Intermediate modulation (initial training)
- = 0.8 → Optimized modulating systems (years of practice)
- (Epinephrine — acute stress response)
- (Norepinephrine — chronic hypervigilance)
- = 0.1 → Low arousal (calm, regulated)
- = 0.4 → Moderate arousal (high but controllable alertness)
- = 0.8 → High arousal (fight or flight, frequent meltdowns )
- Sleep quality
- Cortisol levels (stress)
- Immediate social context
- Metabolic state
- Fundamental perceptual differences
- Unique processing patterns
- Stable sensory preferences
- Estimates of and derived from longitudinal studies with repeated measures of the ADI-R (Autism Diagnostic Interview-Revised).
- Correlation with salivary cortisol variability in autistic adults.
-
Even with = 1.0 (perfect modulators) and = 0 (no arousal), there will always be:
- ○
- Basic perceptual differences
- ○
- Unique processing patterns
- ○
- Persistent sensory preferences
- ○
- Fundamental “autistic essence”
- = 0.05 → Very low variance (rarely noticeable)
- = 0.10 → Typical variance (subtle traits persist)
- = 0.15 → High variance (base always visible)
- When → the function tends to 1 → reduces
- When → the function tends 0, it increases
- When → the function = 0.5 → intermediate regulation
Integrated Abstract
- defines the “ceiling”
- defines “response speed”
- defines the “neurochemical balance”
- The sigmoid function translates this balance into a probability of regulation
- It asserts that there is always “underlying autism”
- Diagnosed with ASD level 1 at age 40
- History of recurrent burnout, chronic anxiety, masked social difficulties
- Developed unconscious compensatory strategies throughout life
- Sought specialized therapy after diagnosis
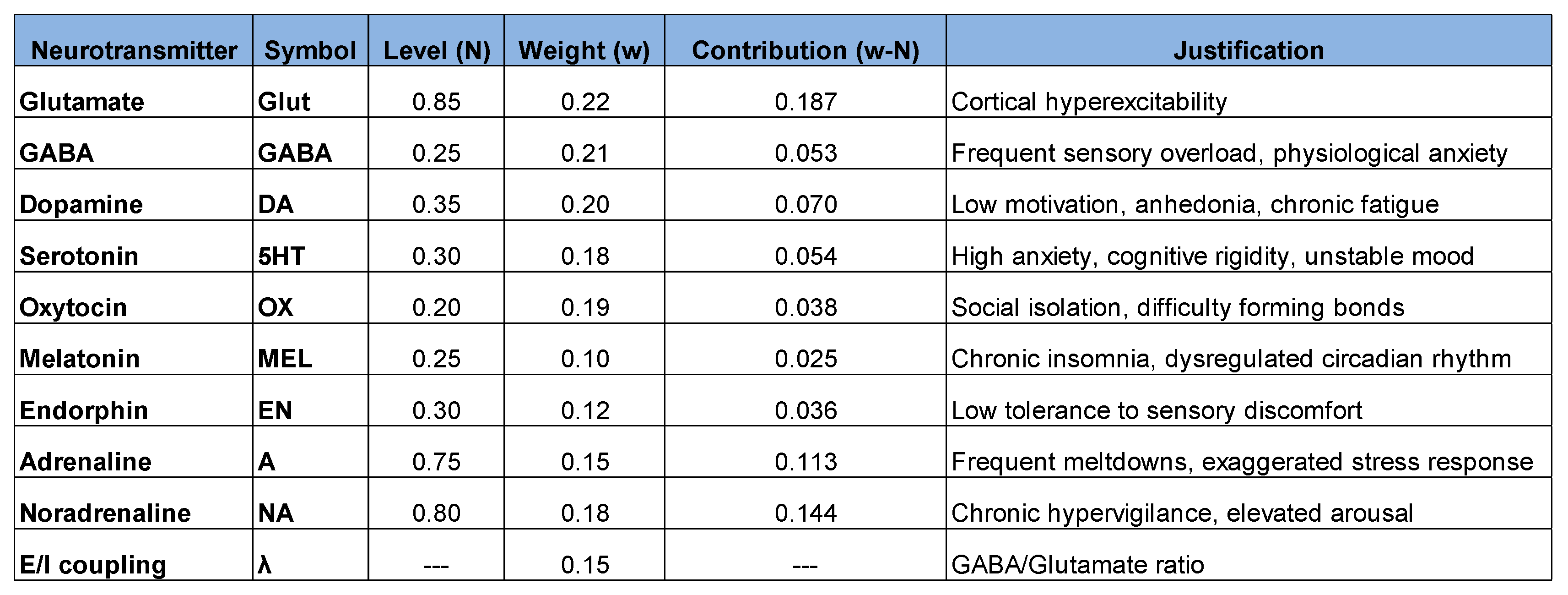
- Very high glutamate (0.85) → Severe cortical hyperexcitability
- Very low GABA (0.25) → Critical failure in sensory filtering
- Ratio(0.294) → Excitation-inhibition imbalance (ideal: 0.8-1.0)
- Arousal (0.257) → Nervous system in constant reactive mode
- Heavy masking reduces external visibility
- Maximum internal suffering (burnout, anxiety, insomnia)
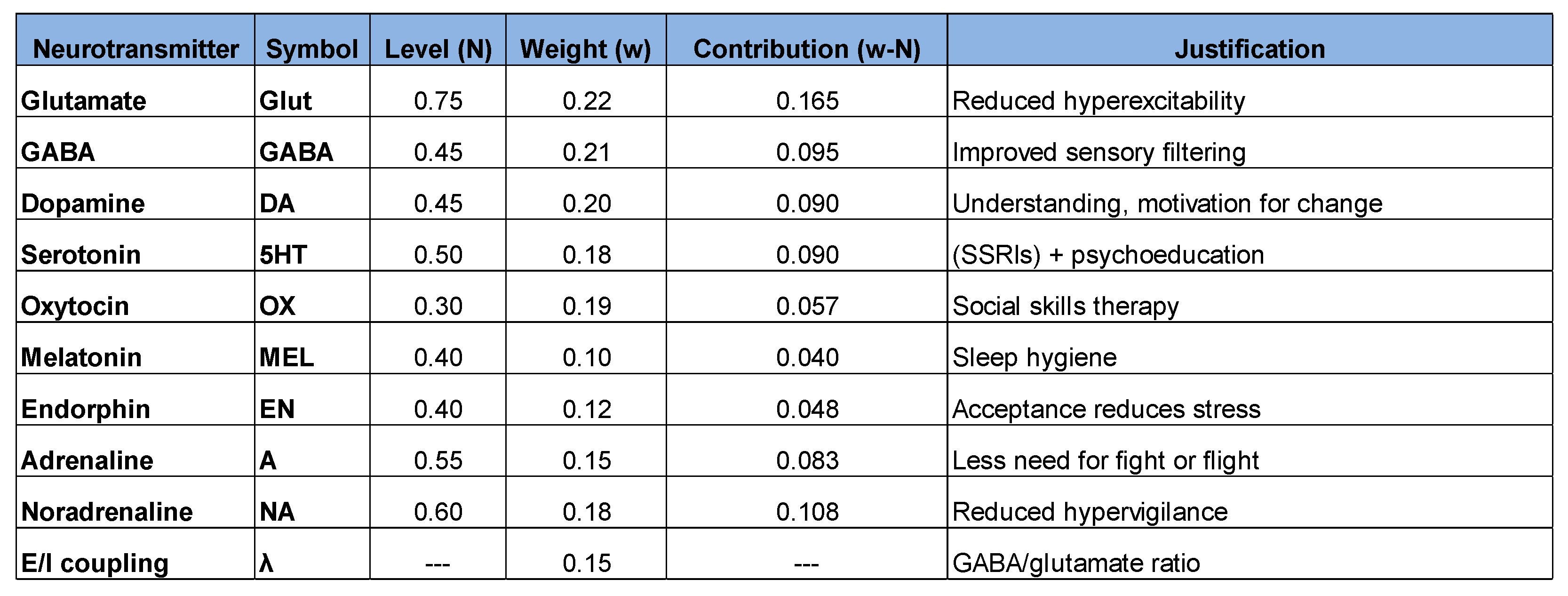
- Reduced glutamate (0.85 → 0.75) → Less hyperexcitability
- Increased GABA (0.25 → 0.45) → Improved sensory filtering
- Ratio (0.294 → 0.600) → Balance sheet recovering
- Reduced arousal (0.257 → 0.191) → Less hypervigilance
- Apparent paradox: It decreased even with increased authenticity
- Explanation: The system is more regulated, reducing internal suffering for patient “P”
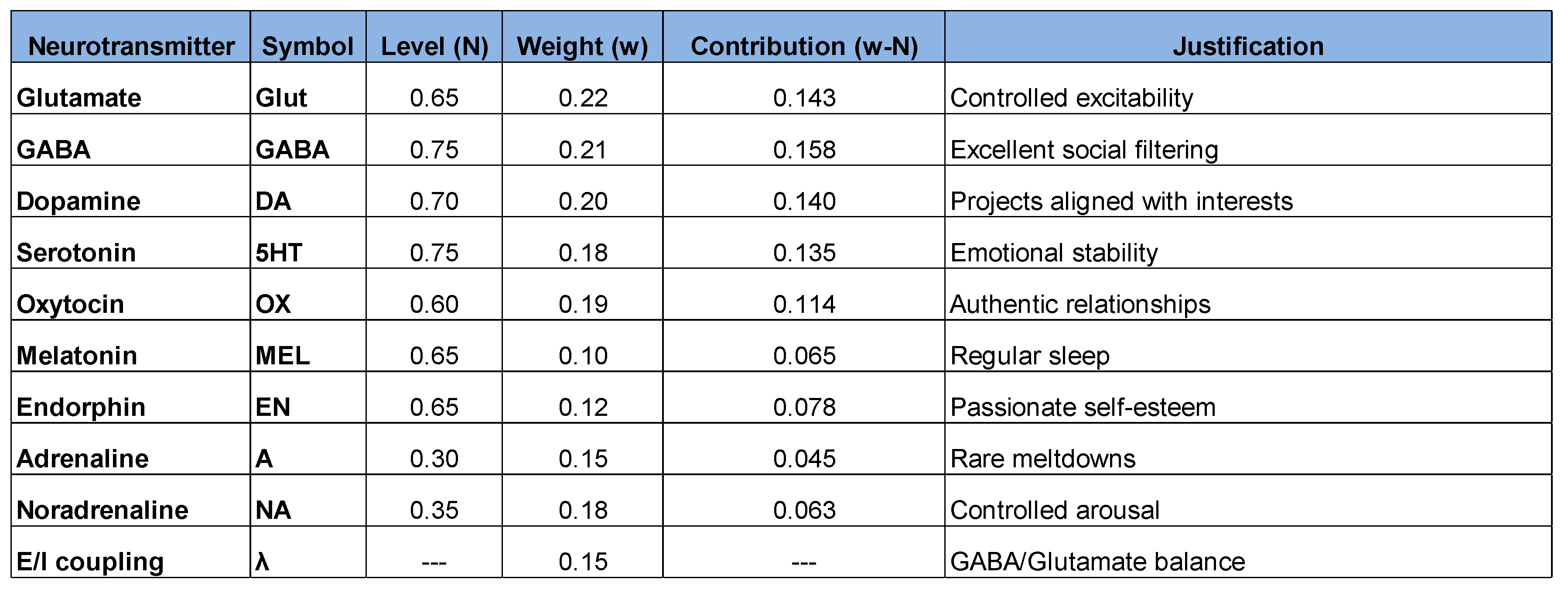
- Normalized glutamate (0.65) → Controlled excitability
- High GABA (0.75) → Excellent sensory filtering
- Ideal ratio (1.154) → Optimal excitation-inhibition balance
- Minimum arousal (0.108) → Calm nervous system
- Full authenticity + maximum well-being
- Smaller than at the beginning, but with a very high quality of life.
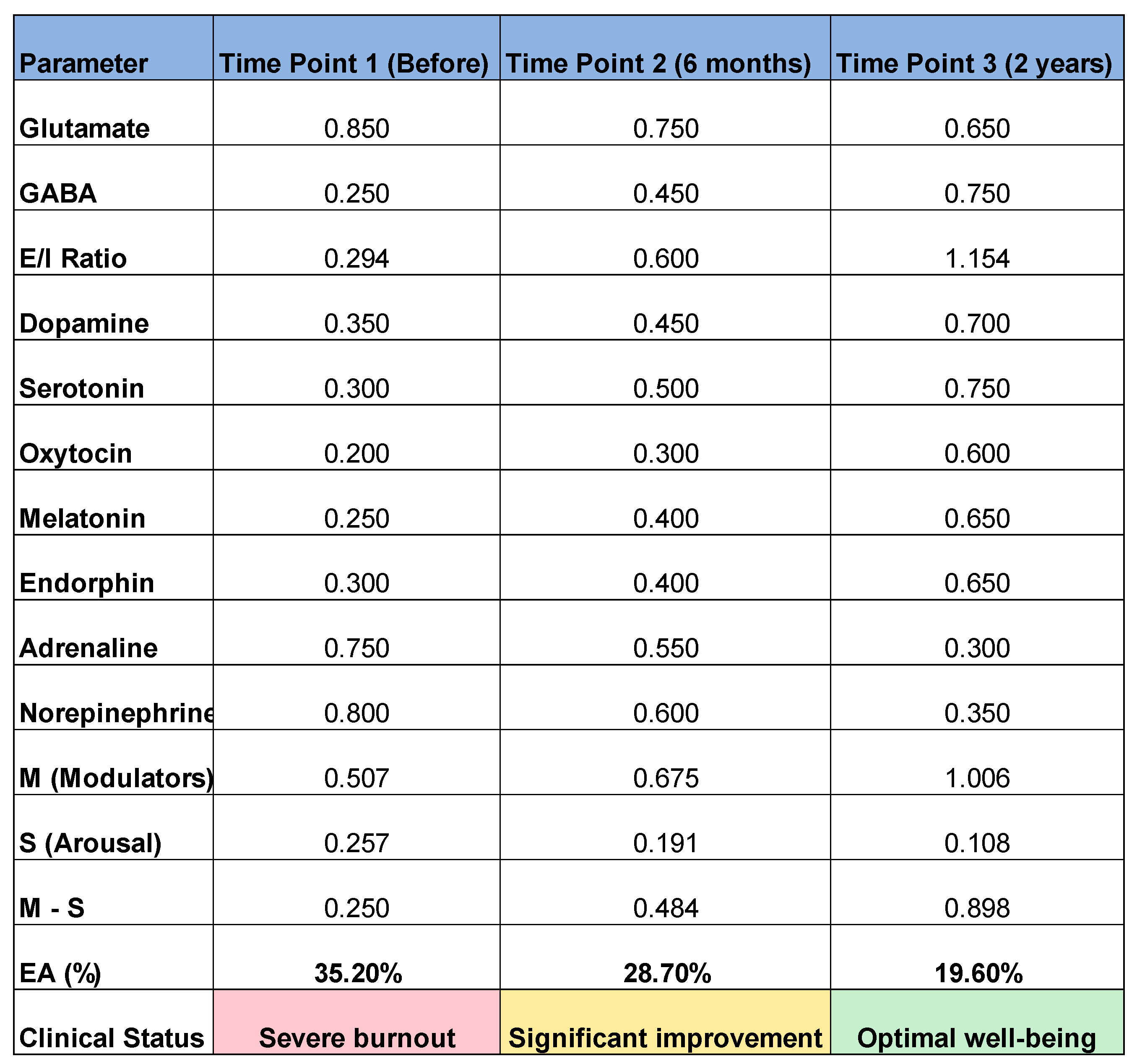
- Stopped wasting energy on masking
- Optimized modulating neurotransmitters
- Reduced chronic arousal
Limitations of the Current Model
- Bimodal functions: to capture profiles traditionally labeled as “low-functioning” vs. “high-functioning”
- Mixture models: Representing subgroups with distinct neurochemical dynamics.
- DNA methylation in SHANK3 and MECP2 genes
- Histone modifications in response to the environment
- Long-term plasticity (LTP/LTD)
- Elevation of pro-inflammatory cytokines (, )
- Chronic microglial activation
- Blood-brain barrier dysfunction
- Ordinary differential equations ( ODEs )
- Simulation of therapeutic interventions over time
- Prediction of individualized trajectories
- Validation in pediatric cohorts (2-12 years) to test generalization.
- Integration with wearable data (heart rate, skin conductance) for continuous monitoring of
- Development of a clinical application for personalized calculation and intervention suggestions.
- Longitudinal neuroimaging studies correlating changes in and with structural alterations (gray matter volume, synaptic density)
Conclusion
References
- ASHWOOD, P. et al. Elevated plasma cytokines in autism spectrum disorders provide evidence of immune dysfunction and are associated with impaired behavioral outcome. Brain, Behavior, and Immunity, [sl], v. 25, no. 1, p. 40-45, 2011. [CrossRef]
- BOUVARD, MP et al. Evidence for dysregulated endogenous opioids in ASD. Biological Psychiatry, [sl], v. 37, no. 11, p. 770-779, 1995. [CrossRef]
- CARLSSON, ML Hypothesis: is infantile autism a hypoglutamatergic disorder? Relevance of glutamate-serotonin interactions for pharmacotherapy. Journal of Neural Transmission, [sl], v. 105, p. 525-535, 1998. [CrossRef]
- CHUGANI, DC et al. Whole blood serotonin levels in autism spectrum disorder: a systematic review. Journal of Autism and Developmental Disorders, [sl], v. 44, p. 2060-2063, 2014. [CrossRef]
- COGHLAN, S. et al. Altered GABAergic signaling in autism spectrum disorders. Frontiers in Human Neuroscience, [sl], v. 6, p. 200, 2012. [CrossRef]
- ECKER, C. The neurobiology of autism: structural brain alterations and connectivity. Brain, Oxford, vol. 140, no. 1, p. 205-227, 2017. [CrossRef]
- FRISTON, K. A theory of cortical responses. Philosophical Transactions of the Royal Society B: Biological Sciences, London, vol. 360, n. 1456, p. 815-836, 2005. [CrossRef]
- FRISTON, K. The free-energy principle: a unified brain theory? Nature Reviews Neuroscience, London, vol. 11, no. 2, p. 127-138, 2010. [CrossRef]
- FUJIYOSHI, K. et al. Multimodal prediction of autism spectrum disorder diagnosis using plasma metabolites and machine learning. JAMA Network Open, [sl], v. 5, no. 12, e2248981, 2022. [CrossRef]
- GESCHWIND, DH; LEVITT, P. Autism spectrum disorders: developmental disconnection syndromes. Current Opinion in Neurobiology, [sl], v. 17, no. 1, p. 103-111, 2007. [CrossRef]
- GORDON, I. et al. Oxytocin enhances brain function in children with autism. PNAS - Proceedings of the National Academy of Sciences, [sl], v. 110, no. 52, p. 20953-20958, 2013. [CrossRef]
- GRINGRAS, P. et al. Melatonin in children with autism spectrum disorders: endogenous and pharmacokinetic studies. Sleep Medicine, [sl], v. 22, p. 10-14, 2016. [CrossRef]
- HIRSTEIN, W. et al. Autonomic nervous system arousal and sensory sensitivity in ASD. Brain and Cognition, [sl], v. 65, no. 3, p. 267-274, 2007. [CrossRef]
- HORDER, J. et al. Glutamate and GABA in autism spectrum disorder—a translational magnetic resonance spectroscopy study in man and rodent models. Translational Psychiatry, [sl], v. 8, p. 106, 2018. [CrossRef]
- HULL, L. et al. Development and validation of the Camouflaging Autistic Traits Questionnaire (CAT-Q). Journal of Autism and Developmental Disorders, [sl], v. 49, p. 819-833, 2019. [CrossRef]
- HUTT, J.; HOSEIN, A.; GHANIZADEH, A. The arousal regulation model of autism spectrum disorder. Neuroscience & Biobehavioral Reviews, Oxford, vol. 73, p. 1-20, 2017. [CrossRef]
- KLABUNDE, M. et al. Arousal and ASD: a systematic review of autonomic reactivity. Journal of Autism and Developmental Disorders, [sl], v. 45, p. 3075-3086, 2015. [CrossRef]
- LIVINGSTON, LA et al. Quantifying compensatory strategies in adults with and without diagnosed autism. Molecular Autism, [sl], v. 11, p. 15, 2020. [CrossRef]
- MCKAY, M.D.; BECKMAN, RJ; CONOVER, WJ A comparison of three methods for selecting values of input variables in the analysis of output from a computer code. Technometrics, [sl], v. 42, no. 1, p. 55-61, 2000. [CrossRef]
- MEHLER, MF; PURPURA, DP The locus coeruleus–noradrenergic system in autism spectrum disorder. Brain Research, [sl], v. 1642, p. 149-156, 2016. [CrossRef]
- MINSHAWI, NF et al. Pain reactivity and opioid modulation in autism spectrum disorder. Neuroscience & Biobehavioral Reviews, [sl], v. 51, p. 1-10, 2015. [CrossRef]
- MULLER, CL et al. Serotonin in autism spectrum disorder: from biomarker to treatment. Neuroscience & Biobehavioral Reviews, [sl], v. 55, p. 1-24, 2015. [CrossRef]
- NARAYANAN, A. et al. Altered striatal dopamine in autism spectrum disorder. Molecular Psychiatry, [sl], v. 22, no. 11, p. 1512-1512, 2017. [CrossRef]
- NELSON, SB; VALAKH, V. Excitation–inhibition imbalance in autism. Neuron, [sl], v. 87, no. 4, p. 684-698, 2015. [CrossRef]
- PAVAL, D. Dopaminergic contributions to autism spectrum disorder. Brain Research, [sl], v. 1664, p. 1-10, 2017. [CrossRef]
- RUBENSTEIN, JLR; MERZENICH, MM Model of autism: increased ratio of excitation/inhibition in key neural systems. Genes, Brain and Behavior, [sl], v. 2, no. 5, p. 255-267, 2003. [CrossRef]
- SALTELLI, A. et al. Variance based sensitivity analysis of model output. Design and estimator for the total sensitivity index. Computer Physics Communications, [sl], v. 181, no. 2, p. 259-270, 2010. [CrossRef]
- SAPEY-TRIOMPHE, L. et al. Cortical GABA levels and sensory over-reactivity in autism spectrum disorder. NeuroImage, [sl], v. 204, p. 116183, 2019. [CrossRef]
- SINHA, P. et al. Autism as a disorder of prediction. Nature Neuroscience, [sl], v. 17, no. 11, p. 1431-1433, 2014. [CrossRef]
- TORDJMAN, S. et al. Sleep disturbances in autism spectrum disorder: role of melatonin. Journal of Autism and Developmental Disorders, [sl], v. 45, p. 3060-3074, 2015. [CrossRef]
- UDDIN, LQ Salience network dysfunction in autism spectrum disorder: a predictive coding perspective. Neuroscience & Biobehavioral Reviews, Oxford, vol. 52, p. 1-15, 2015. [CrossRef]
- VARGAS, DL et al. Neuroglial activation and neuroinflammation in the brain of patients with autism. Annals of Neurology, [sl], v. 57, no. 1, p. 67-81, 2005. [CrossRef]
- YOUNG, L.J.; BARRETT, CE Oxytocin and autism spectrum disorders: a review. Journal of Neuroendocrinology, [sl], v. 22, no. 11, p. 1130-1135, 2010. [CrossRef]
- ZHENG, Z. et al. Altered glutamate and GABA concentrations in autism spectrum disorder: a meta-analysis of ¹H-MRS studies. Molecular Psychiatry, [sl], v. 27, no. 3, p. 1846-1856, 2022. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




