Submitted:
04 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
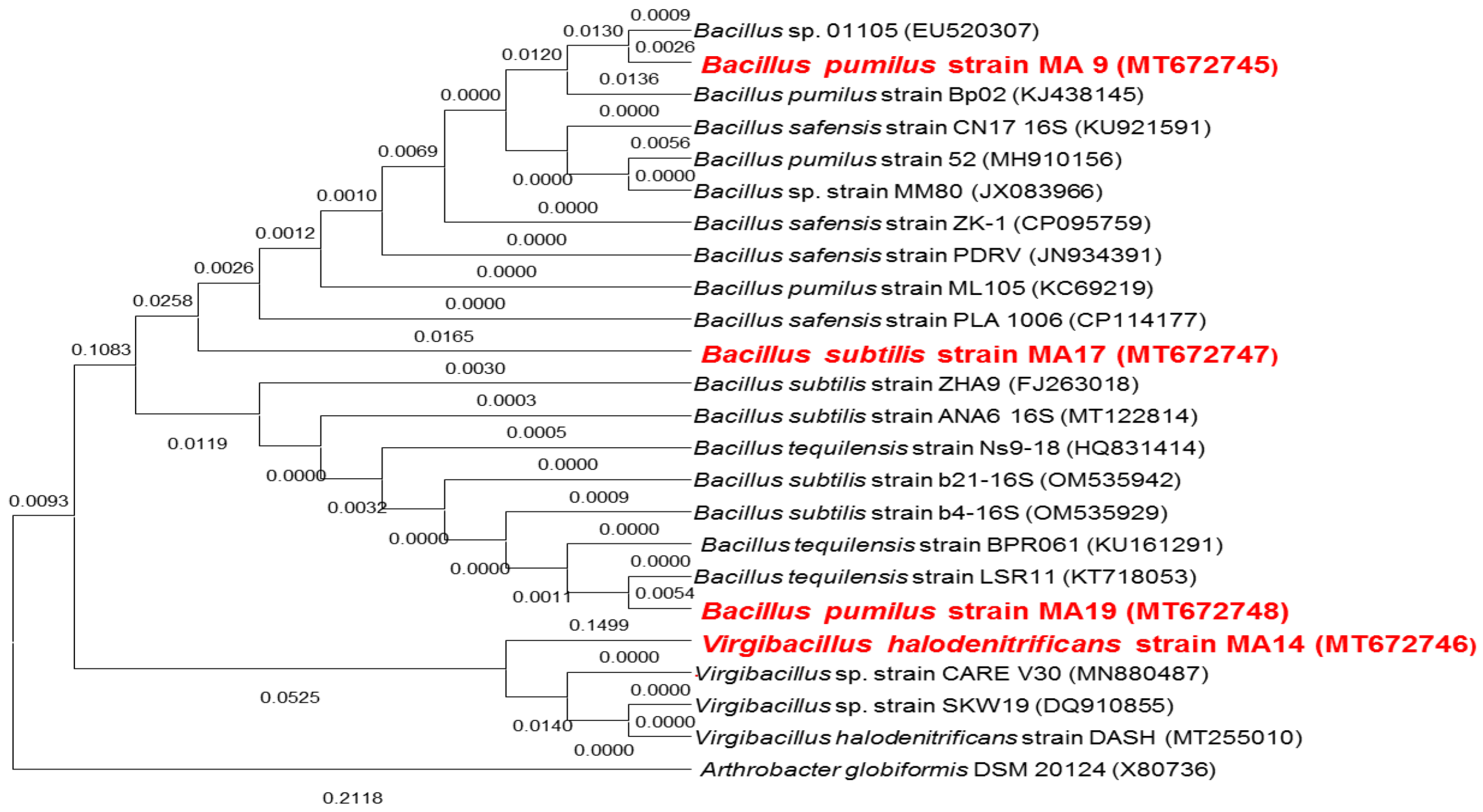
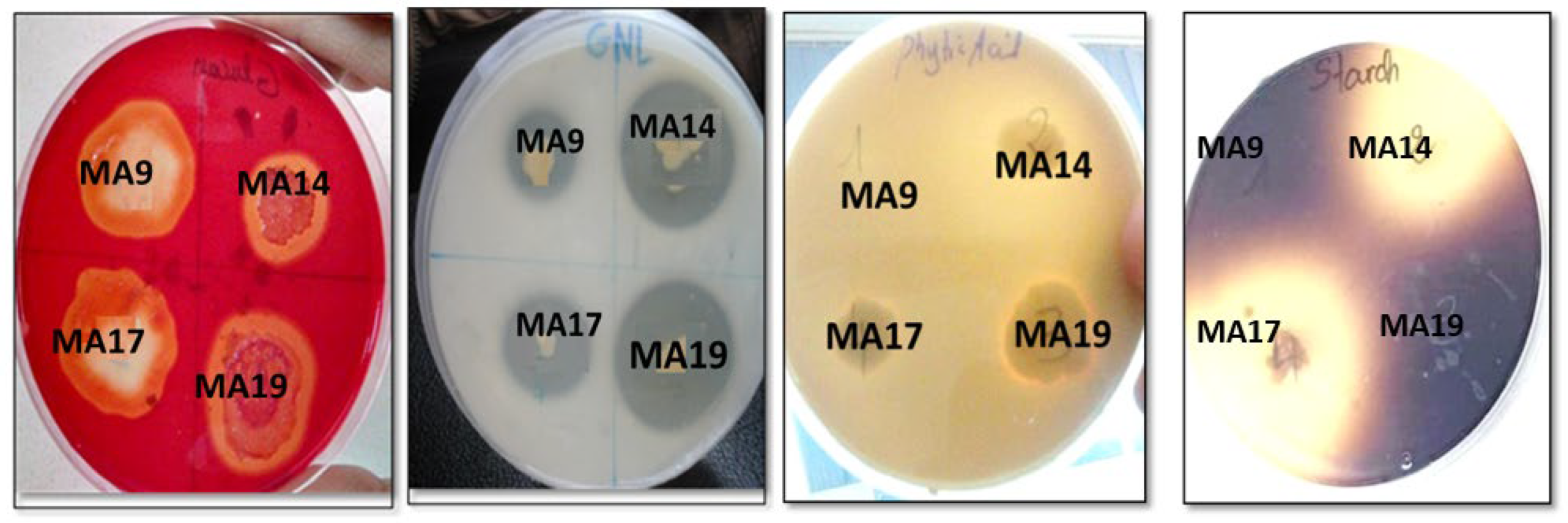
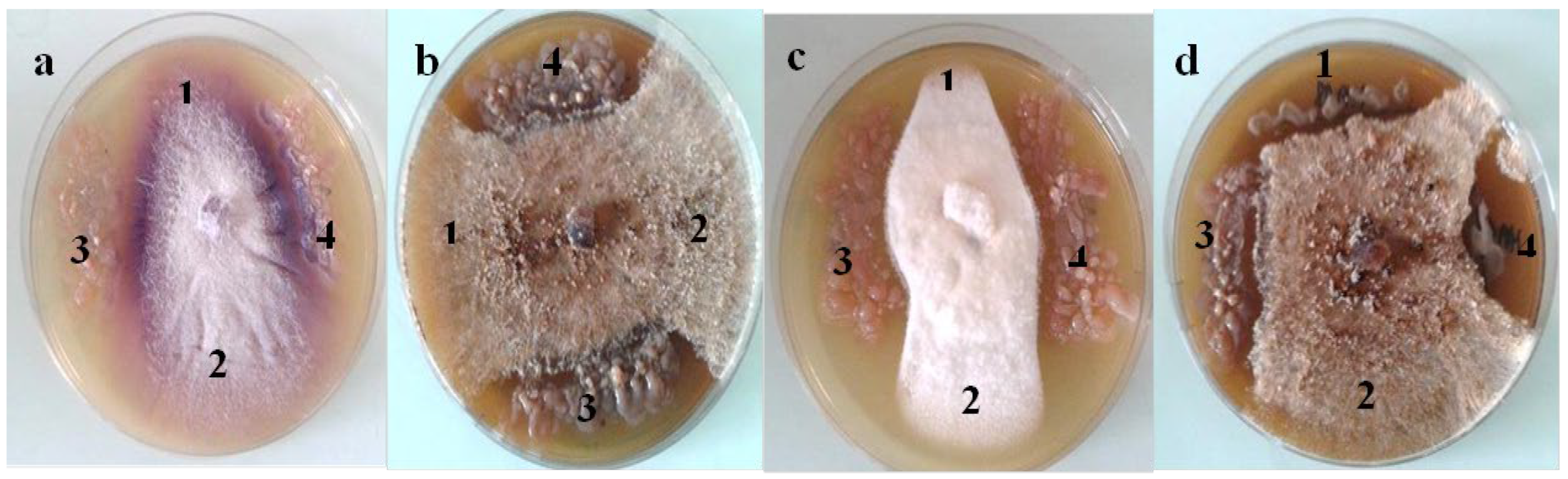
2.1. Biochemical Characterization of PGPB Strains
| Treatment | Total length (cm) | % Increase | Total dry mass (mg/plant) | % Increase |
|---|---|---|---|---|
| SN | 12,3333de | - | 53bc | - |
| BiopMA9 | 22b | 78 | 55,3333b | 42 |
| BiopMA14 | 13d | 05 | 46d | 18 |
| BiopMA17 | 24,3333a | 97 | 45,6667d | 17 |
| BiopMA19 | 24,3333a | 97 | 57b | 46 |
| SN+ salt | 7,6667 g | - | 24,3333f | - |
| BiopMA9 | 10f | 30.5 | 65a | 167 |
| BiopMA14 | 10,3333ef | 35 | 36e | 48 |
| BiopMA17 | 19c | 148 | 51,3333c | 111 |
| BiopMA19 | 14,3333d | 87 | 62a | 154.8 |
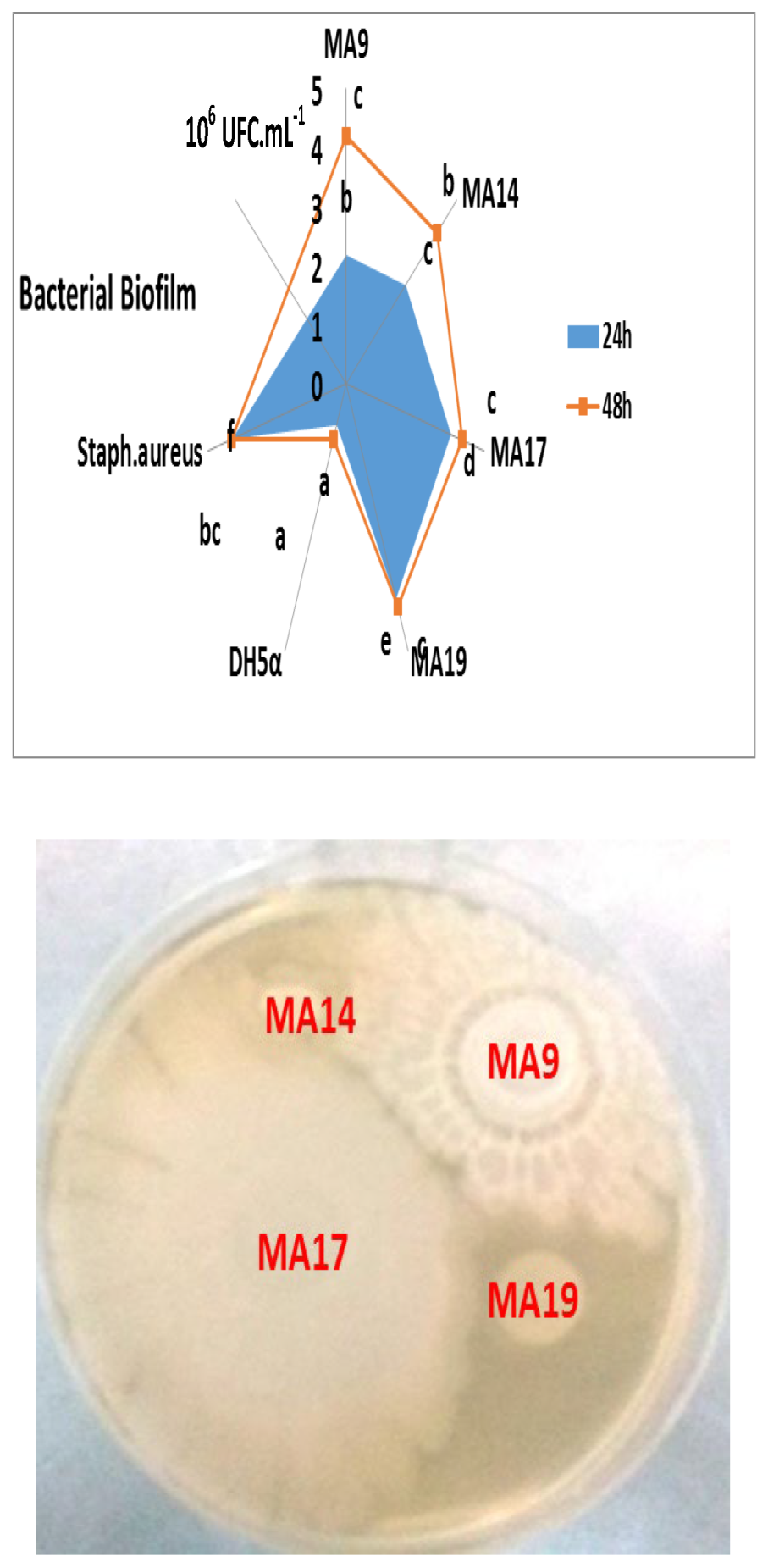
2.2. Biofilm and Exopolysaccharides Production in Vitro
2.3. Effect of PGPB on the Morphology and Physiological Parameters of PGPB
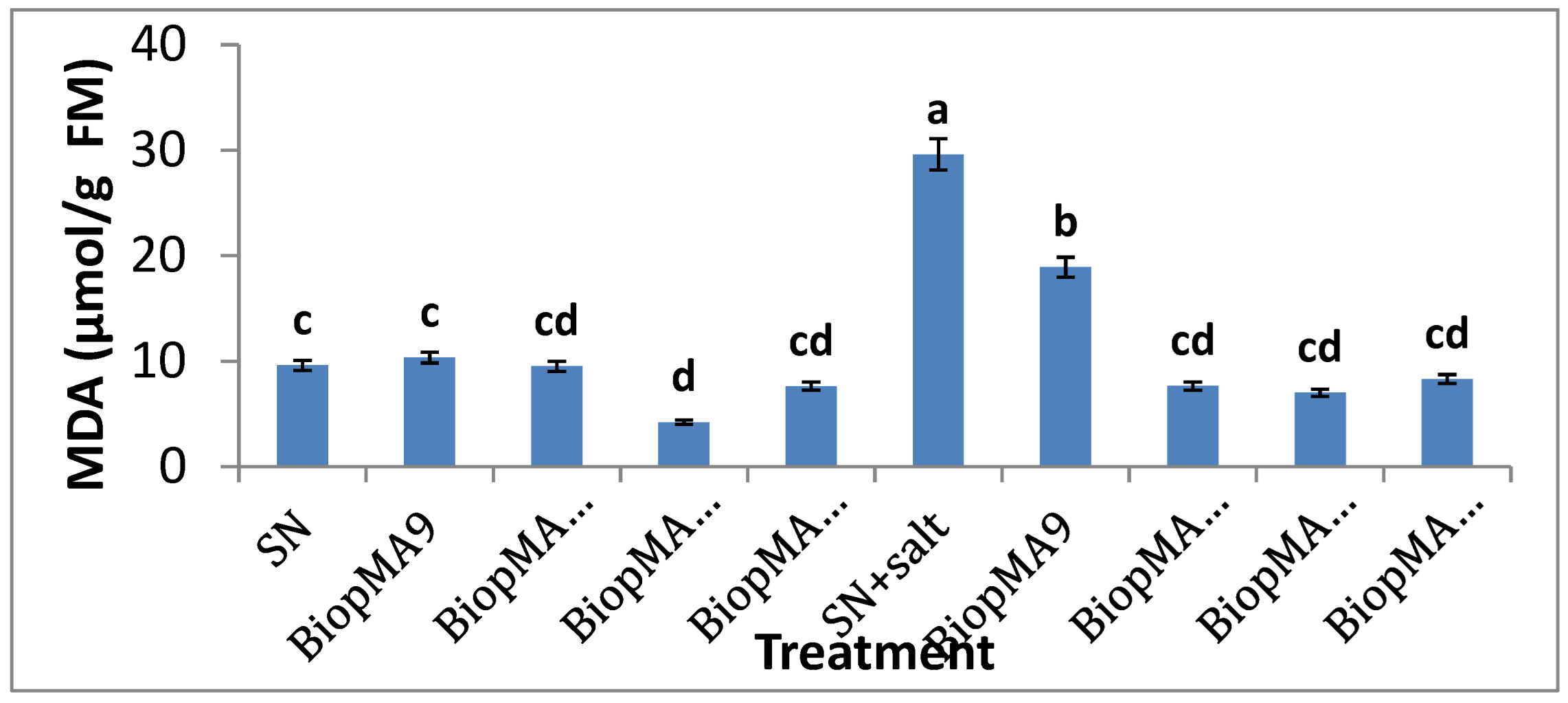
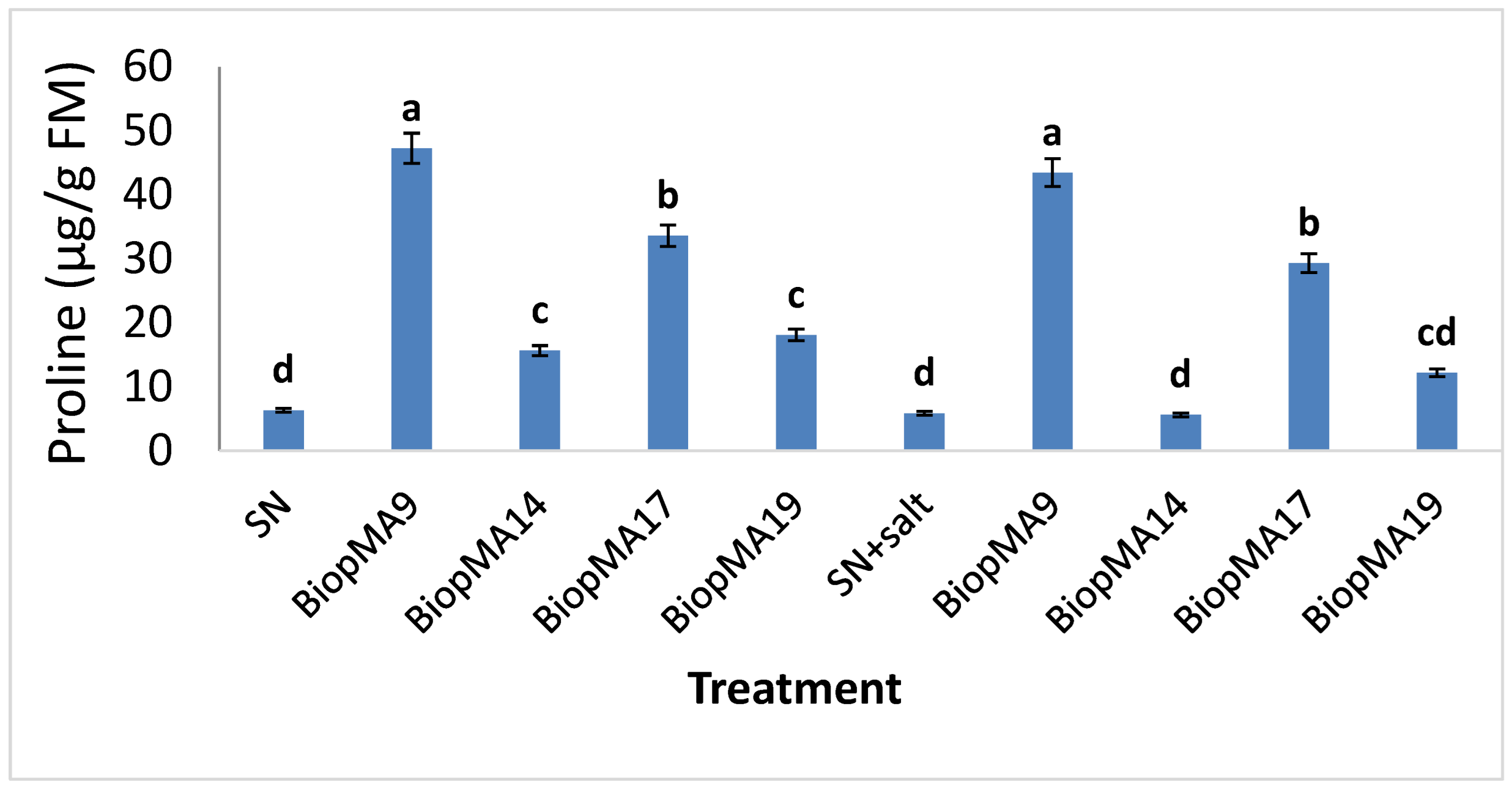
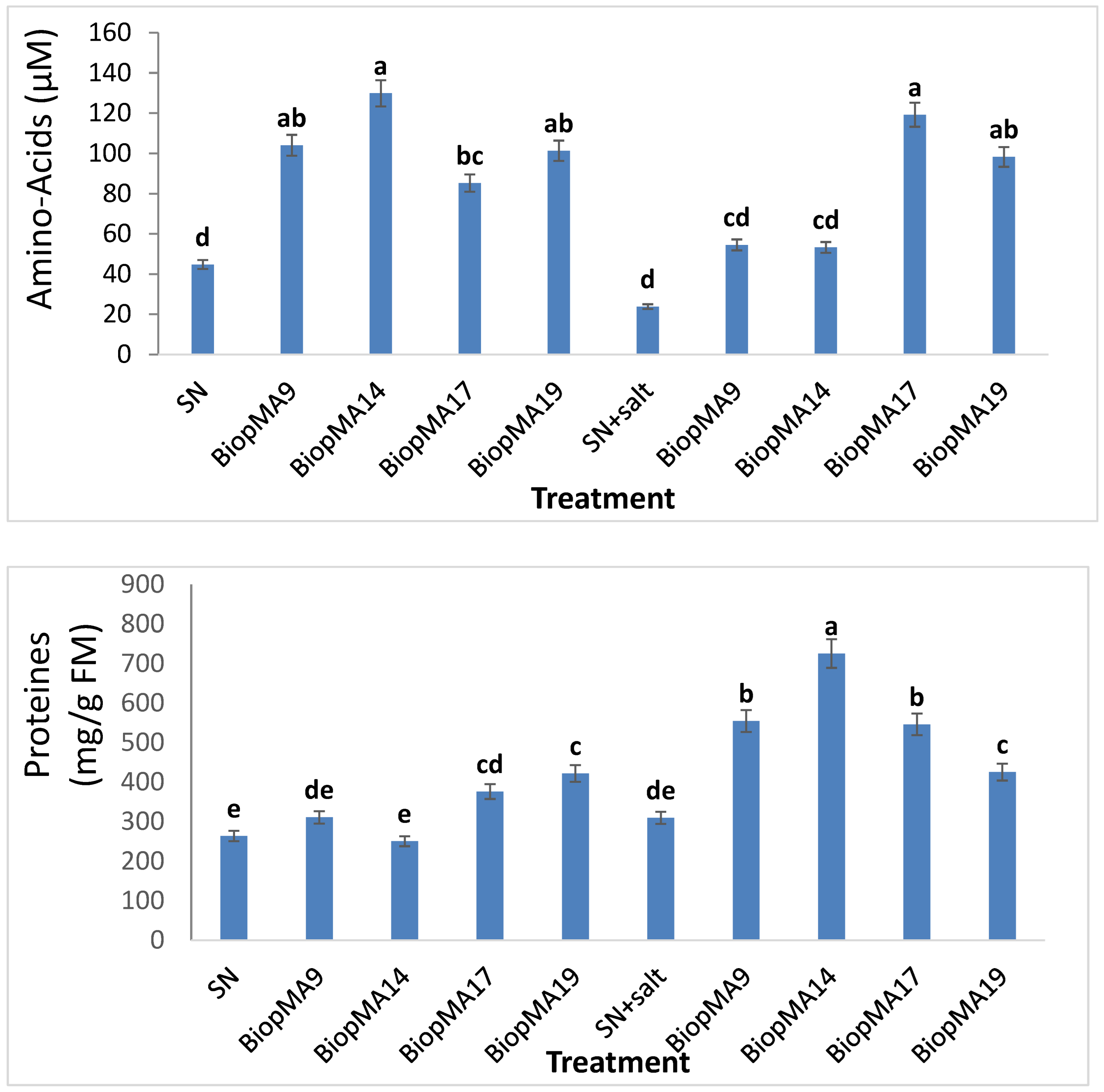
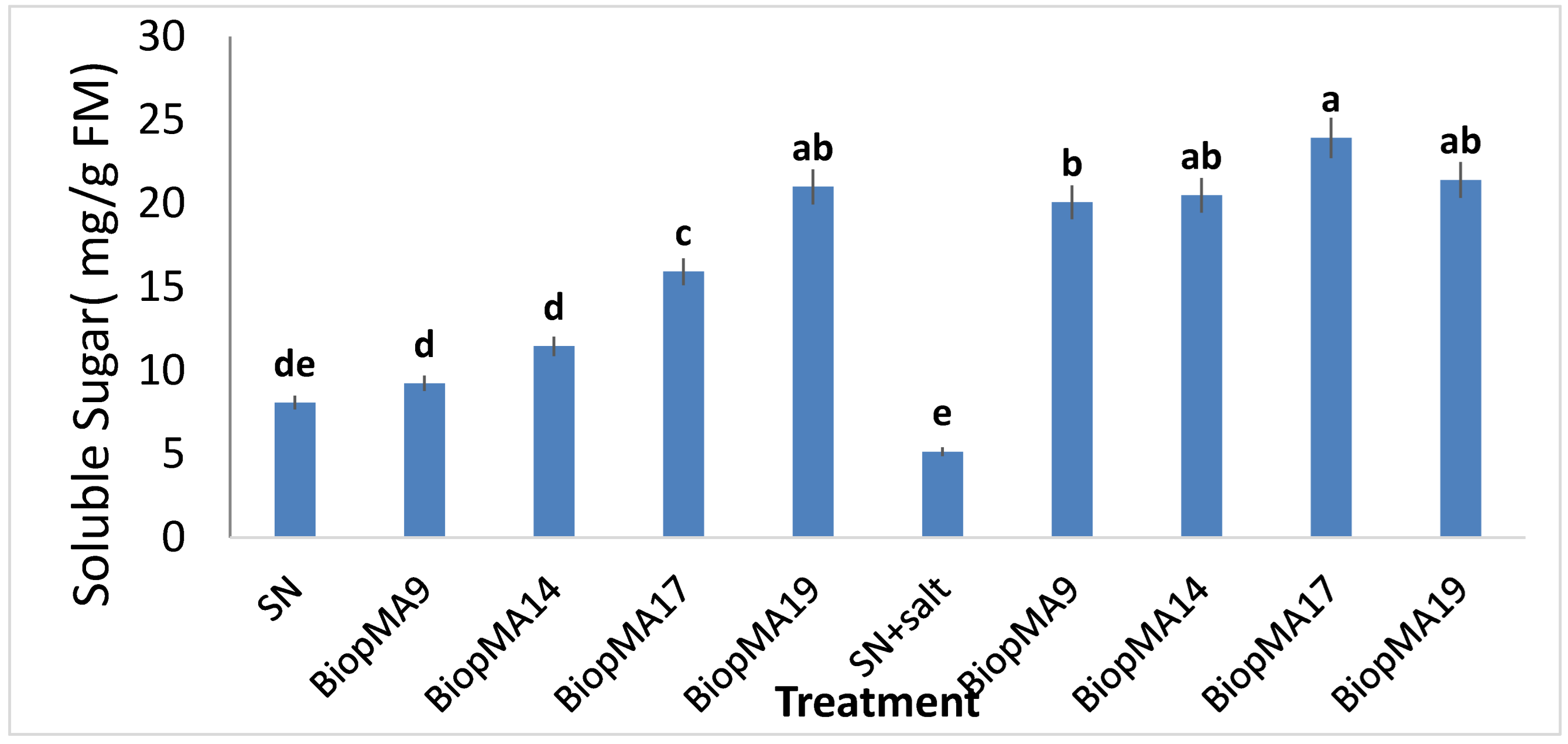
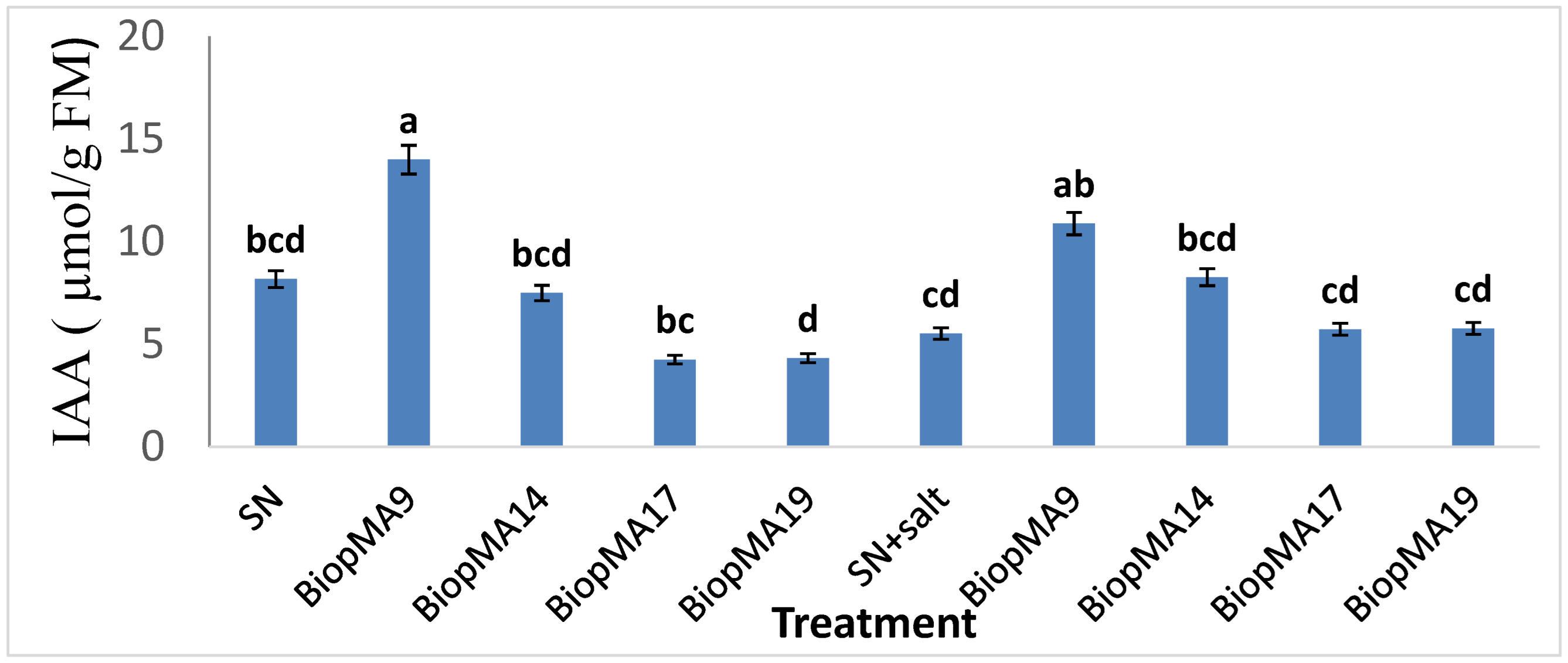
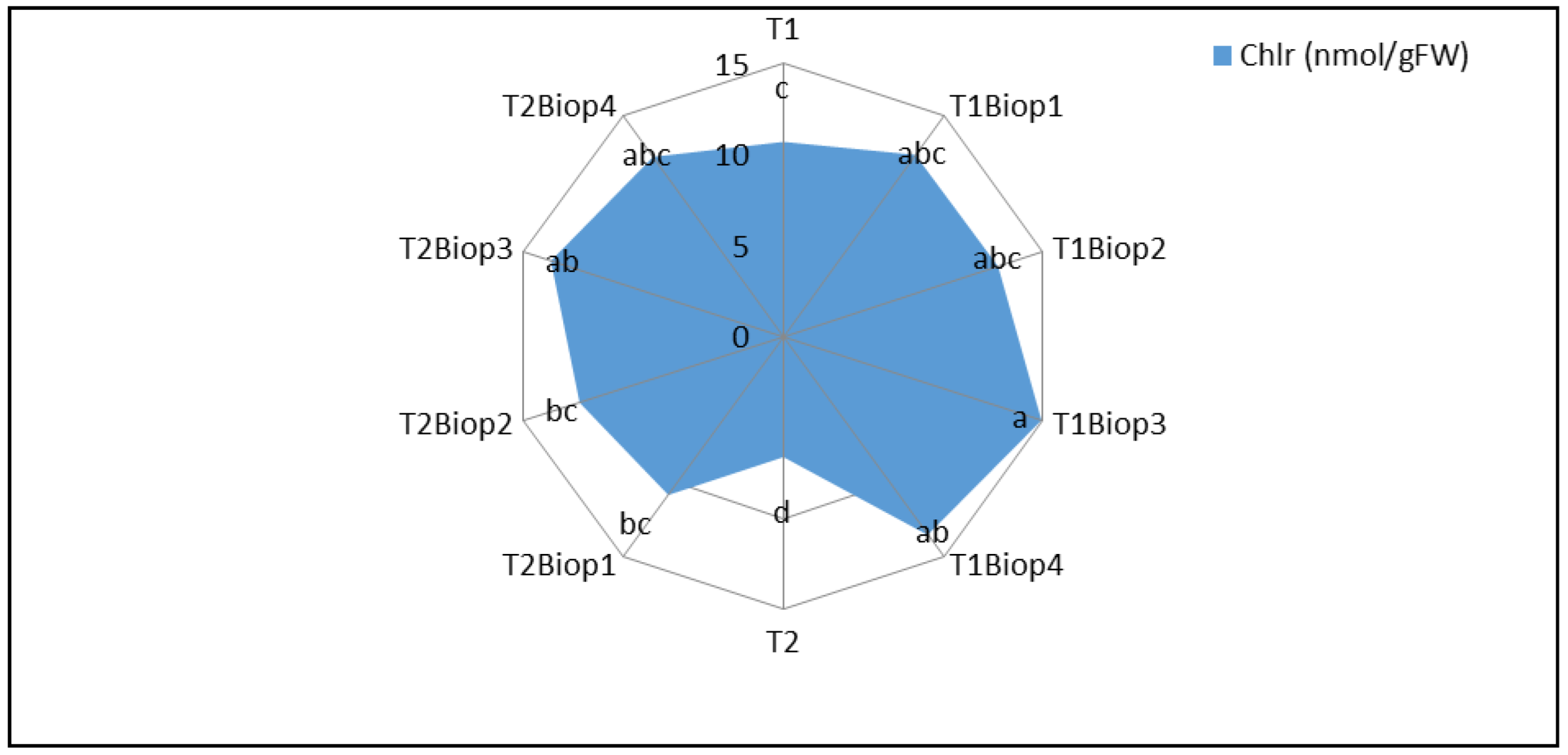
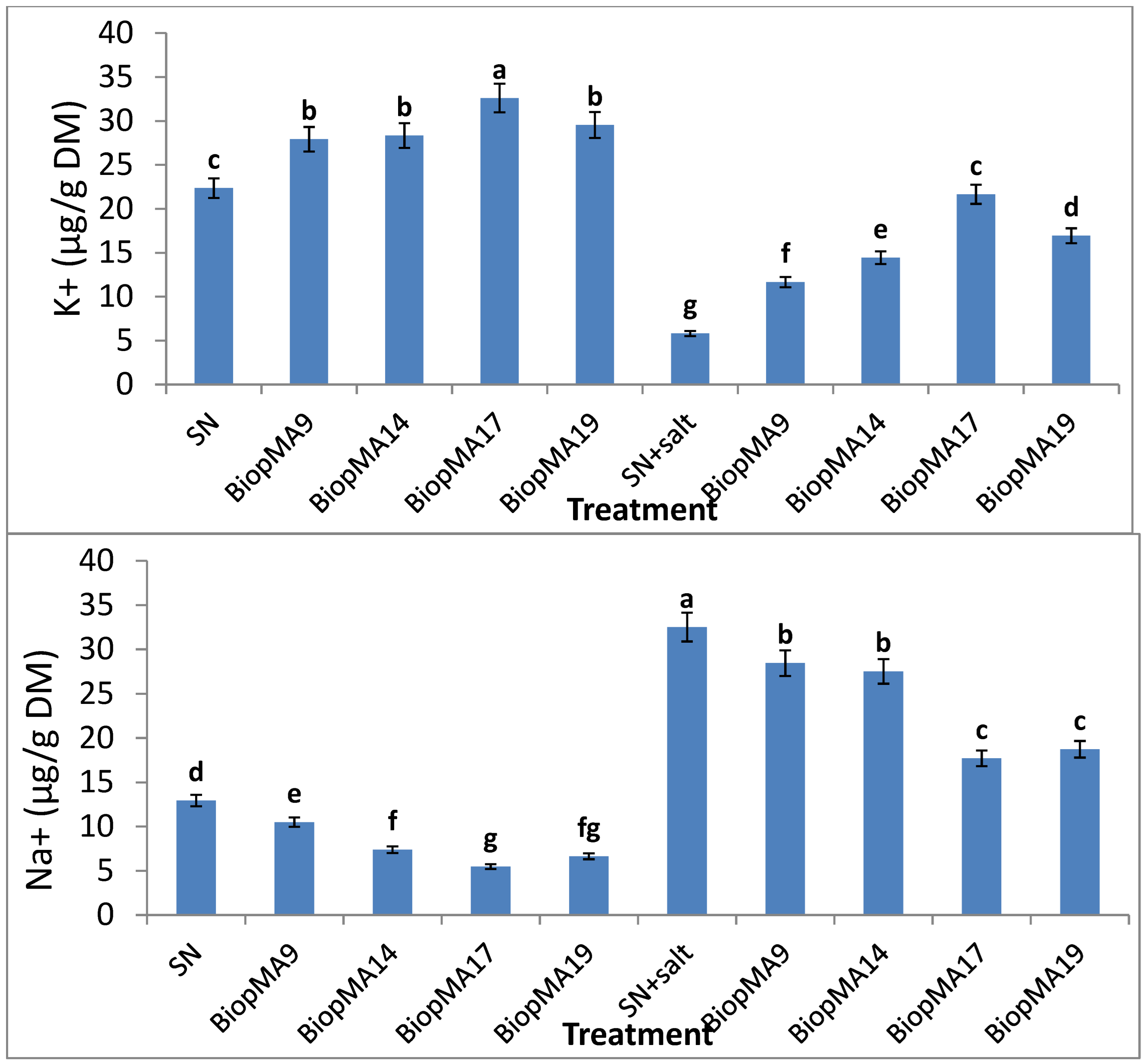
2.4. Effect of PGPB Biopriming on the Biochemical Parameters of Plants
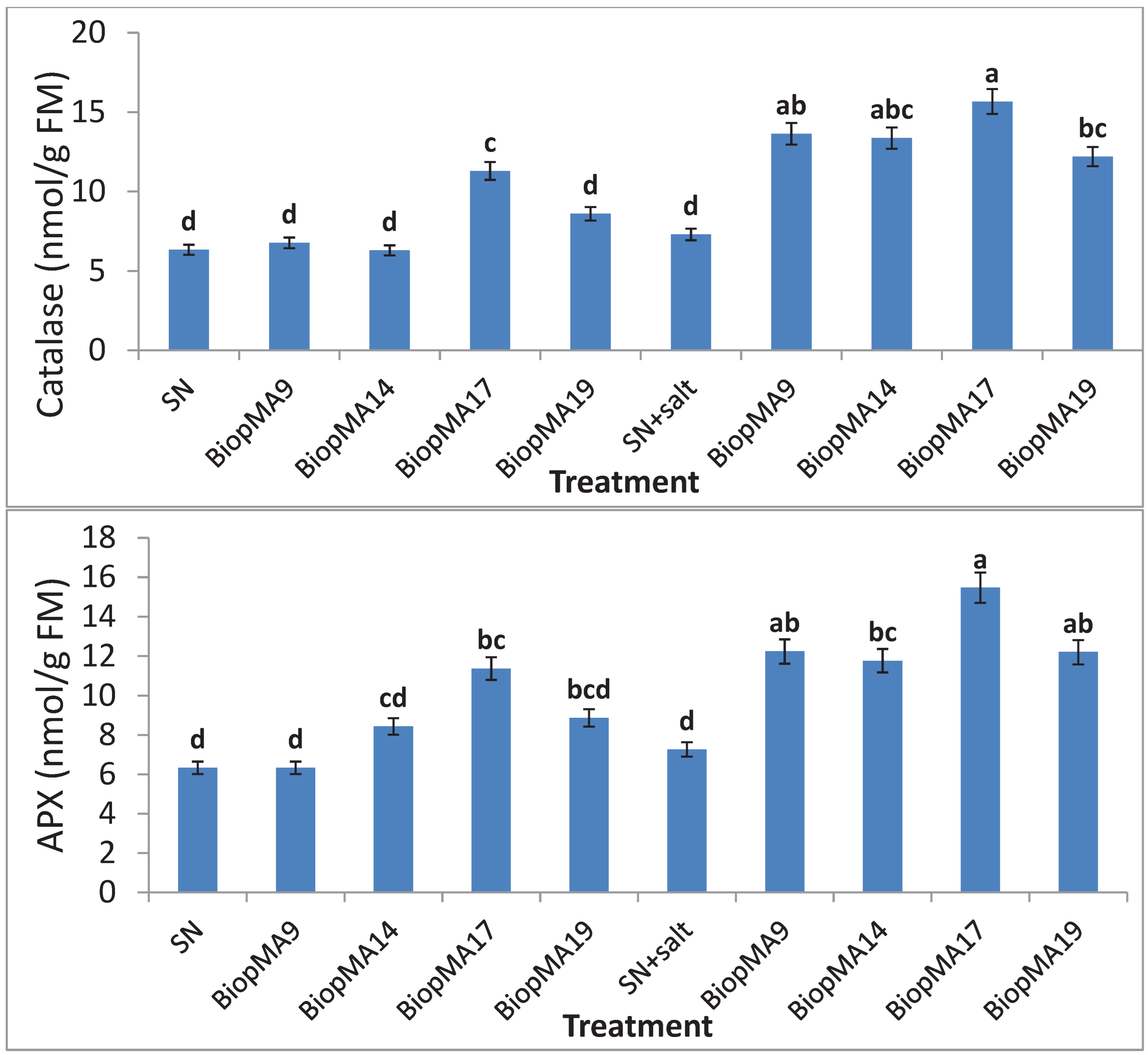
2.5. Expression of the Antioxidant Enzymatic System
3. Discussion
4. Materials and Methods
4.1. Microorganisms
4.2. Biofilm and Exopolysaccharides Production
4.3. Seed Biopriming with PGPB and Salt-Stress Treatment
4.4. Plant Analysis
4.4.1. Electrolyte Leakage and Percentage Change in Water Content
4.4.2. Lipid Peroxidation
4.4.3. Auxin Analysis
4.4.4. Biochemical Analysis
4.4.5. Studies on Antioxidative Enzymes of Plants
4.5. Statistical Analysis
5. Conclusion
Supplementary Materials
Funding
Availability of Data and Materials
Competing Interests
Authors' Contributions
Acknowledgments
References
- Kaushal, M. and S.P. Wani, Rhizobacterial-plant interactions: strategies ensuring plant growth promotion under drought and salinity stress. Agriculture, Ecosystems & Environment, 2016. 231: p. 68-78. [CrossRef]
- Etesami, H. and B.R. Glick, Halotolerant plant growth–promoting bacteria: Prospects for alleviating salinity stress in plants. Environmental and Experimental Botany, 2020: p. 104124. [CrossRef]
- Gupta, A., et al., Mechanistic insights of plant growth promoting bacteria mediated drought and salt stress tolerance in plants for sustainable agriculture. International Journal of Molecular Sciences, 2022. 23(7): p. 3741.
- Gamalero, E. and B.R. Glick, Bacterial modulation of plant ethylene levels. Plant Physiology, 2015. 169(1): p. 13-22.
- Bernardes, M.F.F., et al., Impact of pesticides on environmental and human health. Toxicology studies-cells, drugs and environment, 2015: p. 195-233.
- Adesemoye, A.O. and J.W. Kloepper, Plant–microbes interactions in enhanced fertilizer-use efficiency. Applied microbiology and biotechnology, 2009. 85(1): p. 1-12.
- Zhu, Z., et al., Microsatellite markers linked to 2 powdery mildew resistance genes introgressed from Triticum carthlicum accession PS5 into common wheat. Genome, 2005. 48(4): p. 585-590. [CrossRef]
- Ahmadi, N., H. Mibus, and M. Serek, Characterization of ethylene-induced organ abscission in F1 breeding lines of miniature roses (Rosa hybrida L.). Postharvest Biology and Technology, 2009. 52(3): p. 260-266.
- Edwards, C., Environmental pollution by pesticides. Vol. 3. 2013: Springer Science & Business Media.
- Etesami, H. and D.K. Maheshwari, Use of plant growth promoting rhizobacteria (PGPRs) with multiple plant growth promoting traits in stress agriculture: Action mechanisms and future prospects. Ecotoxicology and environmental safety, 2018. 156: p. 225-246. [CrossRef]
- Majeed, A. and S. Siyyar, Salinity Stress Management in Field Crops: An Overview of the Agronomic Approaches, in Plant Ecophysiology and Adaptation under Climate Change: Mechanisms and Perspectives II. 2020, Springer. p. 1-16.
- Hadj Brahim, A., et al., Biopriming of Durum Wheat Seeds with Endophytic Diazotrophic Bacteria Enhances Tolerance to Fusarium Head Blight and Salinity. Microorganisms, 2022. 10(5): p. 970. [CrossRef]
- Lugtenberg, B.J., et al., Plant growth promotion by microbes. Molecular microbial ecology of the rhizosphere, 2013. 2: p. 561-573.
- Lugtenberg, B., et al., Microbial control of plant diseases. Molecular microbial ecology of the rhizosphere, Wiley-Blackwell Publishing, Oxford, 2013: p. 67-91.
- Pliego, C., F. Kamilova, and B. Lugtenberg, Plant growth-promoting bacteria: fundamentals and exploitation, in Bacteria in agrobiology: Crop ecosystems. 2011, Springer. p. 295-343.
- Glick, B.R., et al., Promotion of plant growth by ACC deaminase-producing soil bacteria, in New perspectives and approaches in plant growth-promoting Rhizobacteria research. 2007, Springer. p. 329-339.
- Dodd, I.C. and F. Pérez-Alfocea, Microbial amelioration of crop salinity stress. Journal of Experimental Botany, 2012. 63(9): p. 3415-3428. [CrossRef]
- Berg, G., et al., Biocontrol and osmoprotection for plants under salinated conditions. Molecular microbial ecology of the rhizosphere, 2013. 1: p. 561-573.
- Li, G., et al., Maize endophytic plant growth-promoting bacteria Peribacillus simplex can alleviate plant saline and alkaline stress. International Journal of Molecular Sciences, 2024. 25(20): p. 10870.
- Vimal, S.R., et al., PGPR: An Effective Bio-Agent in Stress Agricultural Management. 2016.
- Dar, Z.M., et al., Review on plant growth promoting rhizobacteria and its effect on plant growth. Journal of Pharmacognosy and Phytochemistry, 2018. 7(3): p. 2802-2804.
- Altuntaş, Ö. and İ.K. Kutsal, Use of Some Bacteria and Mycorrhizae as Biofertilizers in Vegetable Growing and Beneficial Effects in Salinity and Drought Stress Conditions. Physical Methods for Stimulation of Plant and Mushroom Development, 2018: p. 65.
- WANG, L., et al., Effects of NaCl on plant growth, root ultrastructure, water content, and ion accumulation in a halophytic seashore beach plum (Prunus maritima). Pak. J. Bot, 2018. 50(3): p. 863-869.
- Hniličková, H., et al., Effect of salt stress on growth, electrolyte leakage, Na+ and K+ content in selected plant species. Plant, Soil and Environment, 2019. 65(2): p. 90-96.
- Hajiboland, R. and A. Joudmand, The K/Na replacement and function of antioxidant defence system in sugar beet (Beta vulgaris L.) cultivars. Acta Agriculturae Scandinavica Section B–Soil and Plant Science, 2009. 59(3): p. 246-259. [CrossRef]
- Kärkönen, A. and K. Kuchitsu, Reactive oxygen species in cell wall metabolism and development in plants. Phytochemistry, 2015. 112: p. 22-32.
- Tsukagoshi, H., Control of root growth and development by reactive oxygen species. Current Opinion in Plant Biology, 2016. 29: p. 57-63.
- Das, K. and A. Roychoudhury, Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Frontiers in environmental science, 2014. 2: p. 53.
- Latef, A.A.H.A. and H. Chaoxing, Effect of arbuscular mycorrhizal fungi on growth, mineral nutrition, antioxidant enzymes activity and fruit yield of tomato grown under salinity stress. Scientia Horticulturae, 2011. 127(3): p. 228-233.
- Abd_Allah, E.F., et al., Endophytic bacterium Bacillus subtilis (BERA 71) improves salt tolerance in chickpea plants by regulating the plant defense mechanisms. Journal of Plant Interactions, 2018. 13(1): p. 37-44.
- Acosta-Motos, J.R., et al., Changes in growth, physiological parameters and the hormonal status of Myrtus communis L. plants irrigated with water with different chemical compositions. Journal of plant physiology, 2016. 191: p. 12-21.
- Rajasheker, G., et al., Role and regulation of osmolytes and ABA interaction in salt and drought stress tolerance, in Plant Signaling Molecules. 2019, Elsevier. p. 417-436.
- Wijayasinghe, Y.S., A. Tyagi, and N.K. Poddar, Regulation of cell volume by Osmolytes, in Cellular Osmolytes. 2017, Springer. p. 195-228.
- Ashraf, M., M. Shahbaz, and Q. Ali, Drought-induced modulation in growth and mineral nutrients in canola (Brassica napus L.). Pak J Bot, 2013. 45(1): p. 93-98.
- Allioui, N., et al., Two Novel Bacillus Strains (subtilis and simplex Species) with Promising Potential for the Biocontrol of Zymoseptoria tritici, the Causal Agent of Septoria Tritici Blotch of Wheat. BioMed Research International, 2021. 2021. [CrossRef]
- Zhou, Y., et al., The Effect of Salt-Tolerant Antagonistic Bacteria CZ-6 on the Rhizosphere Microbial Community of Winter Jujube (Ziziphus jujuba Mill.“Dongzao”) in Saline-Alkali Land. BioMed research international, 2021. 2021.
- Sun, Y., et al., The Inhibitory Effects of Ficin on Streptococcus mutans Biofilm Formation. BioMed Research International, 2021. 2021.
- Dimkpa, C., T. Weinand, and F. Asch, Plant–rhizobacteria interactions alleviate abiotic stress conditions. Plant, Cell & Environment, 2009. 32(12): p. 1682-1694. [CrossRef]
- Barnawal, D., et al., 1-Aminocyclopropane-1-carboxylic acid (ACC) deaminase-containing rhizobacteria protect Ocimum sanctum plants during waterlogging stress via reduced ethylene generation. Plant Physiology and Biochemistry, 2012. 58: p. 227-235.
- Singh, S.R., et al., Plant growth-promoting bacteria: an emerging tool for sustainable crop production under salt stress, in Bioremediation of Salt Affected Soils: An Indian Perspective. 2017, Springer. p. 101-131.
- Li, X., et al., A novel PGPR strain Kocuria rhizophila Y1 enhances salt stress tolerance in maize by regulating phytohormone levels, nutrient acquisition, redox potential, ion homeostasis, photosynthetic capacity and stress-responsive genes expression. Environmental and Experimental Botany, 2020: p. 104023.
- Ali, H., et al., Seed priming improves irrigation water use efficiency, yield, and yield components of late-sown wheat under limited water conditions. Turkish Journal of Agriculture and forestry, 2013. 37(5): p. 534-544. [CrossRef]
- Wu, S.S., Enhanced phytoremediation of salt-impacted soils using plant growth-promoting rhizobacteria (PGPR). 2009, University of Waterloo.
- Naidoo, C., Oxidative status and stress associated with cryopreservation of germplasm of recalcitrant-seeded species. 2012.
- Karuppanapandian, T., et al., Reactive oxygen species in plants: their generation, signal transduction, and scavenging mechanisms. Australian Journal of Crop Science, 2011. 5(6): p. 709.
- Khan, M. and S. Panda, Alterations in root lipid peroxidation and antioxidative responses in two rice cultivars under NaCl-salinity stress. Acta Physiologiae Plantarum, 2008. 30(1): p. 81.
- Li, H. and X. Jiang, Inoculation with plant growth-promoting bacteria (PGPB) improves salt tolerance of maize seedling. Russian Journal of Plant Physiology, 2017. 64(2): p. 235-241.
- Khan, M.A., et al., Halotolerant rhizobacterial strains mitigate the adverse effects of NaCl stress in soybean seedlings. BioMed research international, 2019. 2019. [CrossRef]
- Tao, J.-J., et al., The role of ethylene in plants under salinity stress. Frontiers in Plant Science, 2015. 6: p. 1059.
- Shrivastava, P. and R. Kumar, Soil salinity: a serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi journal of biological sciences, 2015. 22(2): p. 123-131.
- Ji, S.H., M.A. Gururani, and S.-C. Chun, Isolation and characterization of plant growth promoting endophytic diazotrophic bacteria from Korean rice cultivars. Microbiological research, 2014. 169(1): p. 83-98.
- Patten, C.L. and B.R. Glick, Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl. Environ. Microbiol., 2002. 68(8): p. 3795-3801.
- Xie, H., J. Pasternak, and B.R. Glick, Isolation and characterization of mutants of the plant growth-promoting rhizobacterium Pseudomonas putida GR12-2 that overproduce indoleacetic acid. Current Microbiology, 1996. 32(2): p. 67-71. [CrossRef]
- Choudhary, D.K. and A. Varma, Microbial-mediated induced systemic resistance in plants. 2016: Springer.
- Yoshiba, Y., et al., Regulation of levels of proline as an osmolyte in plants under water stress. Plant and Cell Physiology, 1997. 38(10): p. 1095-1102.
- Mohamed, H. and E. Gomaa, Effect of plant growth promoting Bacillus subtilis and Pseudomonas fluorescens on growth and pigment composition of radish plants (Raphanus sativus) under NaCl stress. Photosynthetica, 2012. 50(2): p. 263-272. [CrossRef]
- Ashraf, M., et al., Inoculating wheat seedlings with exopolysaccharide-producing bacteria restricts sodium uptake and stimulates plant growth under salt stress. Biology and Fertility of soils, 2004. 40(3): p. 157-162.
- Sandhya, V., et al., Effect of plant growth promoting Pseudomonas spp. on compatible solutes, antioxidant status and plant growth of maize under drought stress. Plant Growth Regulation, 2010. 62(1): p. 21-30. [CrossRef]
- Leigh, R.A., N. Ahmad, and R.G.W. Jones, Assessment of glycinebetaine and proline compartmentation by analysis of isolated beet vacuoles. Planta, 1981. 153(1): p. 34-41.
- Upadhyay, S.K., et al., Impact of PGPR inoculation on growth and antioxidant status of wheat under saline conditions. Plant Biology, 2012. 14(4): p. 605-611.
- Ashraf, M. and M. Foolad, Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environmental and experimental botany, 2007. 59(2): p. 206-216.
- Mohammadi, M., et al., Study the effect of 24-epibrassinolide application on the Cu/Zn-SOD expression and tolerance to drought stress in common bean. Physiology and Molecular Biology of Plants, 2020: p. 1-16. [CrossRef]
- Yuan, L., et al., Effects of 24-epibrassinolide on nitrogen metabolism in cucumber seedlings under Ca (NO3) 2 stress. Plant Physiology and Biochemistry, 2012. 61: p. 29-35.
- Mansour, M.M.F., Nitrogen Containing Compounds and Adaptation of Plants to Salinity Stress. Biologia Plantarum, 2000. 43(4): p. 491-500.
- AM, B.N., Oxidative stress markers and antioxidant potential of wheat treated with phytohormones under salinity stress. Journal of Stress Physiology & Biochemistry, 2011. 7(4).
- Nasir, F.A., et al., Free amino acids content in some halophytes under salinity stress in arid environment, Jordan. CLEAN–Soil, Air, Water, 2010. 38(7): p. 592-600.
- Parida, A.K., et al., Physiological, anatomical and metabolic implications of salt tolerance in the halophyte Salvadora persica under hydroponic culture condition. Frontiers in Plant Science, 2016. 7: p. 351.
- Kandasamy, S., et al., Understanding the molecular basis of plant growth promotional effect of Pseudomonas fluorescens on rice through protein profiling. Proteome Science, 2009. 7(1): p. 47.
- Yaghubi, K., et al., Potassium silicate alleviates deleterious effects of salinity on two strawberry cultivars grown under soilless pot culture. Scientia Horticulturae, 2016. 213: p. 87-95.
- Bano, A. and M. Fatima, Salt tolerance in Zea mays (L). following inoculation with Rhizobium and Pseudomonas. Biology and Fertility of Soils, 2009. 45(4): p. 405-413.
- Nadeem, S.M., et al., Variation in growth and ion uptake of maize due to inoculation with plant growth promoting rhizobacteria under salt stress. Soil Environ, 2006. 25(2): p. 78-84.
- Hamdia, M.A.E.-S., M. Shaddad, and M.M. Doaa, Mechanisms of salt tolerance and interactive effects of Azospirillum brasilense inoculation on maize cultivars grown under salt stress conditions. Plant Growth Regulation, 2004. 44(2): p. 165-174.
- Karlidag, H., et al., Plant growth-promoting rhizobacteria mitigate deleterious effects of salt stress on strawberry plants (Fragaria× ananassa). Hortscience, 2013. 48(5): p. 563-567.
- Babu, M., D. Singh, and K. Gothandam, The effect of salinity on growth, hormones and mineral elements in leaf and fruit of tomato cultivar PKM1. J Anim Plant Sci, 2012. 22(1): p. 159-164.
- Sofo, A., et al., Ascorbate peroxidase and catalase activities and their genetic regulation in plants subjected to drought and salinity stresses. International Journal of Molecular Sciences, 2015. 16(6): p. 13561-13578.
- Bianco, C. and R. Defez, Medicago truncatula improves salt tolerance when nodulated by an indole-3-acetic acid-overproducing Sinorhizobium meliloti strain. Journal of experimental botany, 2009. 60(11): p. 3097-3107. [CrossRef]
- Kohler, J., F. Caravaca, and A. Roldán, An AM fungus and a PGPR intensify the adverse effects of salinity on the stability of rhizosphere soil aggregates of Lactuca sativa. Soil Biology and Biochemistry, 2010. 42(3): p. 429-434. [CrossRef]
- El-Esawi, M.A., et al., Azospirillum lipoferum FK1 confers improved salt tolerance in chickpea (Cicer arietinum L.) by modulating osmolytes, antioxidant machinery and stress-related genes expression. Environmental and Experimental Botany, 2019. 159: p. 55-65.
- Zhang, N., et al., Effects of different plant root exudates and their organic acid components on chemotaxis, biofilm formation and colonization by beneficial rhizosphere-associated bacterial strains. Plant and soil, 2014. 374(1-2): p. 689-700.
- Meliani, A. and A. Bensoltane, Biofilm-mediated heavy metals bioremediation in PGPR Pseudomonas. J Bioremediat Biodegrad, 2016. 7(370): p. 2.
- Paulo, E.M., et al., An alternative method for screening lactic acid bacteria for the production of exopolysaccharides with rapid confirmation. Food Science and Technology, 2012. 32(4): p. 710-714.
- LUTTS, S., J.M. KINET, and J. BOUHARMONT, NaCl-induced Senescence in Leaves of Rice ( Oryza sativa L.) Cultivars Differing in Salinity Resistance. Annals of Botany, 1996. 78(3): p. 389-398.
- Zhang, H.-X. and E. Blumwald, Transgenic salt-tolerant tomato plants accumulate salt in foliage but not in fruit. Nature biotechnology, 2001. 19(8): p. 765.
- Hodges, D.M., et al., Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta, 1999. 207(4): p. 604-611. [CrossRef]
- Andreae, W. and M. Van Ysselstein, Studies on 3-indoleacetic acid metabolism. VI. 3-Indoleacetic acid uptake and metabolism by pea roots and epicotyls. Plant physiology, 1960. 35(2): p. 225.
- Bates, L.S., R.P. Waldren, and I. Teare, Rapid determination of free proline for water-stress studies. Plant and soil, 1973. 39(1): p. 205-207.
- Shi, H.-T., et al., Increasing nitric oxide content in Arabidopsis thaliana by expressing rat neuronal nitric oxide synthase resulted in enhanced stress tolerance. Plant and Cell Physiology, 2012. 53(2): p. 344-357. [CrossRef]
- Shi, H., et al., Analysis of natural variation in bermudagrass (Cynodon dactylon) reveals physiological responses underlying drought tolerance. PLoS One, 2012. 7(12): p. e53422.
- Cheng, Z., E. Park, and B.R. Glick, 1-Aminocyclopropane-1-carboxylate deaminase from Pseudomonas putida UW4 facilitates the growth of canola in the presence of salt. Canadian Journal of Microbiology, 2007. 53(7): p. 912-918. [CrossRef]
- Bradford, M.M., A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical biochemistry, 1976. 72(1-2): p. 248-254.
- Irigoyen, J., D. Einerich, and M. Sánchez-Díaz, Water stress induced changes in concentrations of proline and total soluble sugars in nodulated alfalfa (Medicago sativd) plants. Physiologia plantarum, 1992. 84(1): p. 55-60.
- Arnon, D.I., Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant physiology, 1949. 24(1): p. 1.
- Ferus, P. and M. Arkosiova, Variability of chlorophyll content under fluctuating environment. Acta fytotechnica et zootechnica, 2001. 4(2): p. 123-125.
- Chamkouri, N., M. Torabpour, and F. Ghafarizadeh, Microwave assisted digestion coupled with inductively coupled plasma optical emission spectrometry for determining element concentrations in halophytes. Journal of Basic Research in Medical Sciences, 2015. 2(1): p. 49-56.
- Aebi, H., [13] Catalase in vitro, in Methods in enzymology. 1984, Elsevier. p. 121-126.
- Nakano, Y. and K. Asada, Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant and cell physiology, 1981. 22(5): p. 867-880. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





