Submitted:
02 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
The Western honey bee, Apis mellifera, forms large colonies, which represent organisms in their own right, so-called superorganisms. Although the honey bee is one of the best studied species of insects, there are still contradictory explanations in use for characteristics as fundamental as colonial lifespan. In this review, considering colonies as potentially immortal is debunked due to a lack of conformity with biological convention. What has traditionally been referred to as honey bee colony represents a matrilineage of superorganisms. By proposing a refined colonial life cycle, the analogy between animals and superorganisms is taken further, thereby visualizing superorganismal traits in a new light. Zygotic, embryonic, fetal, juvenile and adult stages are identified during superorganismal ontogeny. An image of the honey bee superorganism emerges as viviparous with strictly sexual reproduction, which is engaged in various forms of maternal care. A multitude of pathways to sexual reproduction are analyzed. Since swarming precedes fertilization, it can be viewed as superorganismal autotomy rather than reproduction. While the prime swarm serves the function of allowing maternal superorganisms to survive reproduction, afterswarms are a means to produce more than one offspring per reproductive cycle. The implications of the findings for honey bee parasitology, monitoring and beekeeping are discussed.
Keywords:
Introduction
Parent or Offspring?
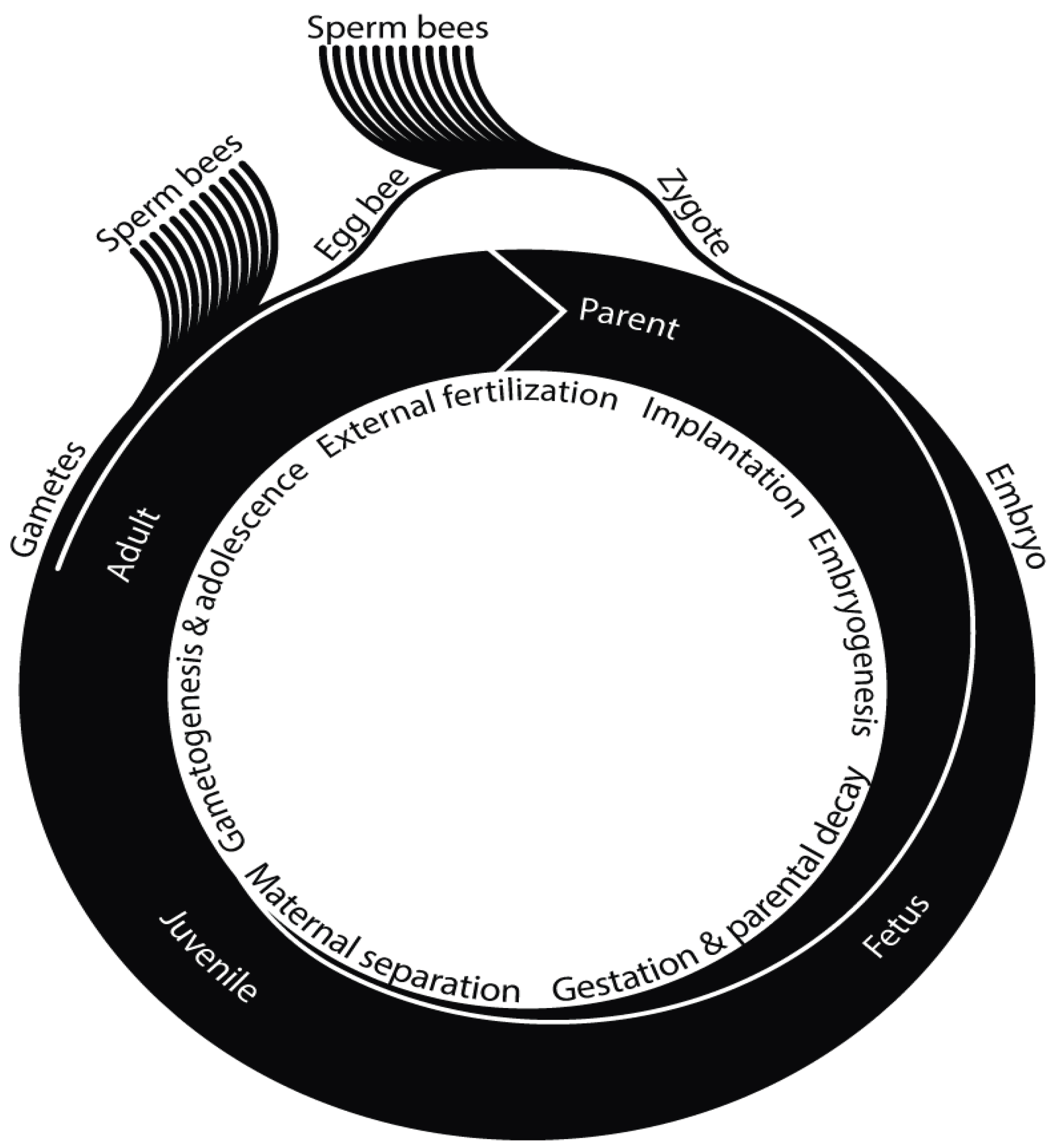
Life Cycle
Reproduction
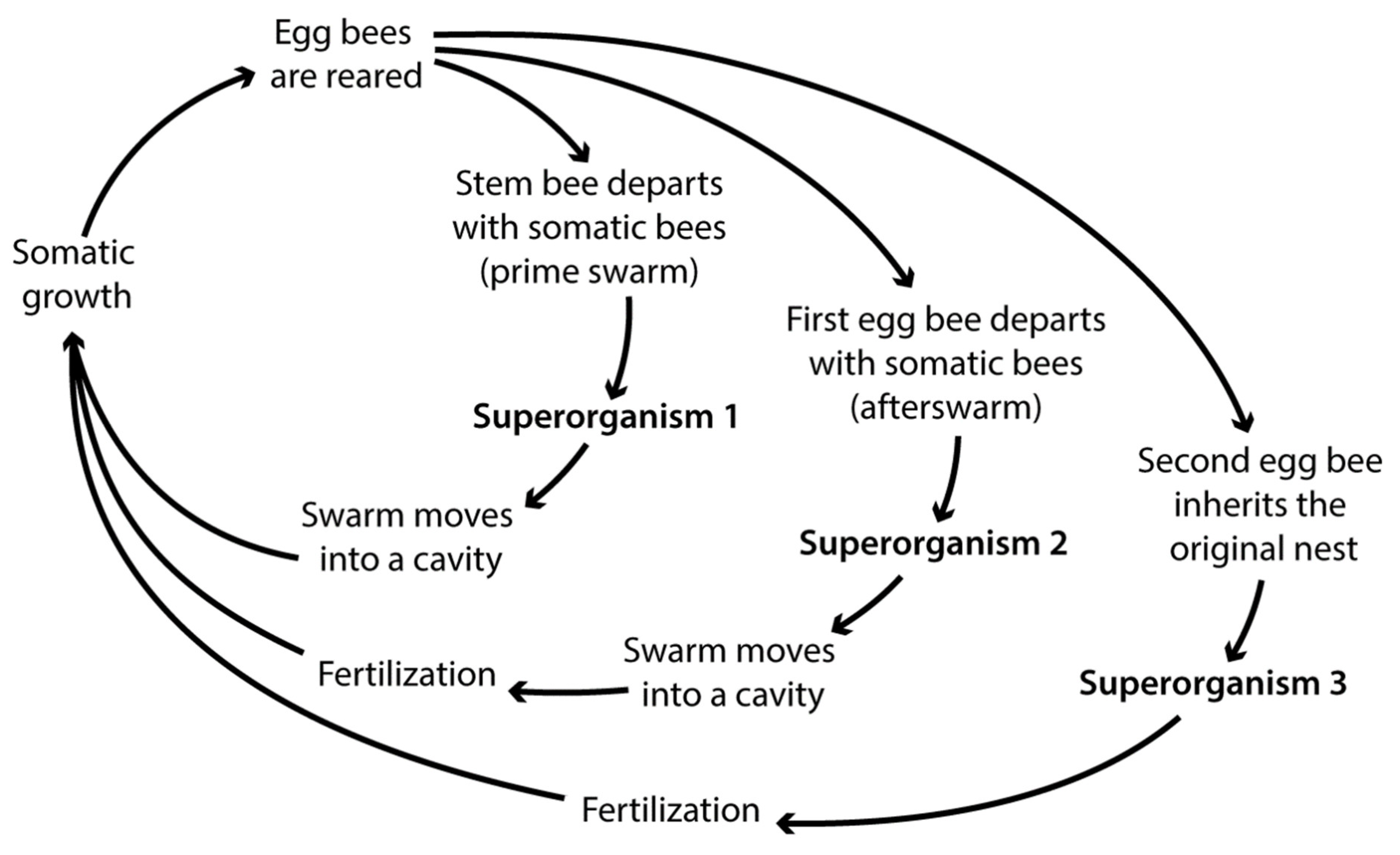
Swarming
Parental Care
Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Allsopp, MH; Hepburn, HR. Swarming, supersedure and the mating system of a natural population of honey bees (Apis mellifera capensis). J Apic Res 1997, 36, 41–48. [Google Scholar] [CrossRef]
- Boomsma, JJ; Gawne, R. Superorganismality and caste differentiation as points of no return: how the major evolutionary transitions were lost in translation. Biol Rev Camb Philos Soc 2018, 93, 28–54. [Google Scholar] [CrossRef]
- Butler C (1609) The Feminine Monarchie; Joseph Barnes: Oxford.
- Congdon, JD; Vitt, LJ; King, WW. Geckos: adaptive significance and energetics of tail autotomy. Science 1974, 184, 1379–1380. [Google Scholar] [CrossRef]
- Danka, RG; Hellmich, RL; Rinderer, TE. Nest usurpation, supersedure and colony failure contribute to Africanization of commercially managed European honey bees in Venezuela. J Apic Res 1992, 31, 119–123. [Google Scholar] [CrossRef]
- Dong, S; Lin, T; Nieh, JC; Tan, K. Social signal learning of the waggle dance in honey bees. Science 2023, 379, 1015–1018. [Google Scholar] [CrossRef]
- Emberts, Z; Escalante, I; Bateman, PW. The ecology and evolution of autotomy. Biol Rev Camb Philos Soc 2019, 94, 1881–1896. [Google Scholar] [CrossRef]
- Fell, RD; Morse, RA. Emergency queen cell production in the honey bee colony. Insectes Soc 1984, 31, 221–237. [Google Scholar] [CrossRef]
- Fries, I; Imdorf, A; Rosenkranz, P. Survival of mite infested (Varroa destructor) honey bee (Apis mellifera) colonies in a Nordic climate. Apidologie 2006, 37, 564–570. [Google Scholar] [CrossRef]
- Goudie, F; Oldroyd, BP. Thelytoky in the honey bee. Apidologie 2014, 45, 306–326. [Google Scholar] [CrossRef]
- Gray, A; Adjlane, N; Arab, A; et al. Honey bee colony winter loss rates for 35 countries participating in the COLOSS survey for winter 2018–2019, and the effects of a new queen on the risk of colony winter loss. J Apic Res 2020, 59, 744–751. [Google Scholar] [CrossRef]
- Helanterä, H. An organismal perspective on the evolution of insect societies. In Front Ecol Evol; 2016. [Google Scholar] [CrossRef]
- Hepburn, HR. Reproductive cycling and hierarchical competition in Cape honeybees, Apis mellifera capensis Esch. Apidologie 1994, 25, 38–48. [Google Scholar] [CrossRef]
- Higham, TE; Russell, AP. Flip, flop and fly: modulated motor control and highly variable movement patterns of autotomized gecko tails. Biol Lett 2010, 6, 70–73. [Google Scholar] [CrossRef]
- Johnson, BR; Linksvayer, TA. Deconstructing the superorganism: social physiology, groundplans, and sociogenomics. Q Rev Biol 2010, 85, 57–79. [Google Scholar] [CrossRef] [PubMed]
- Kleckner, K; Ellis, JD. Reviewing evidence for varied colony structures in honey bees. In Insect Soc; 2025; pp. 1–14. [Google Scholar] [CrossRef]
- Kohl, PL; Rutschmann, B; Steffan-Dewenter, I. Population demography of feral honeybee colonies in central European forests. R Soc Open Sci 2022, 9, 220565. [Google Scholar] [CrossRef] [PubMed]
- Korpela, S; Aarhus, A; Fries, I; Hansen, H. Varroa jacobsoni Oud. in cold climates: population growth, winter mortality and influence on the survival of honey bee colonies. J Apic Res 1992, 31, 157–164. [Google Scholar] [CrossRef]
- Moritz, RFA; Southwick, EE. Bees as superorganisms; Springer-Verlag: Berlin, 1992. [Google Scholar]
- Moro, A; Albouy, V; Dickey, M; et al. A Protocol for Monitoring Populations of Free-Living Western Honey Bees in Temperate Regions. Bee World 2024, 101, 70–74. [Google Scholar] [CrossRef]
- Seeley, TD. Life history strategy of the honey bee, Apis mellifera. Oecologia 1978, 32, 109–118. [Google Scholar] [CrossRef]
- Seeley, TD. Adaptive significance of the age polyethism schedule in honeybee colonies. Behav Ecol Sociobiol 1982, 11, 287–293. [Google Scholar] [CrossRef]
- Seeley, TD. Honeybee ecology; Princeton University Press, 1985. [Google Scholar]
- Seeley, TD. Life-history traits of wild honey bee colonies living in forests around Ithaca, NY, USA. Apidologie 2017, 48, 743–754. [Google Scholar] [CrossRef]
- Smith, ML; Ostwald, MM; Seeley, TD. Honey bee sociometry: tracking honey bee colonies and their nest contents from colony founding until death. Insect Soc 2016, 63, 553–563. [Google Scholar] [CrossRef]
- Szathmáry, E. Toward major evolutionary transitions theory 2.0. PNAS 2015, 112, 10104–10111. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, WM. Ants: Their Structure, Development and Behavior; Columbia University Press: New York, 1910. [Google Scholar]
- Wilson, EO. The insect societies; Belknap Press: Cambridge, MA, 1971. [Google Scholar]
| Cavity 1 | Cavity 2 | Cavity 3 | |
| Year 1 before reproduction | Mother queen and her workers | Not occupied | Not occupied |
| Year 2 before reproduction | Daughter queen and her workers | Mother queen and her new workers | Daughter queen and her workers |
| Year 2 before reproduction for potentially immortal colonies | Parent | Offspring | Offspring |
| Year 2 before reproduction for mortal colonies | Offspring | Parent | Offspring |
| Conventional terminology | Alternative terminology |
| Drone | Sperm bee (gamete) |
| Virgin queen | Egg bee (gamete) |
| Mated queen | Fertilized egg bee (zygote) |
| Laying queen | Stem bee |
| Worker | Somatic bee |
| Colony/society | Superorganism |
| Zygote | Fertilized egg bee |
| Embryo | Stem bee with laid eggs and later larvae and pupae |
| Fetus | Stem bee with laid eggs, larvae, pupae and somatic bees performing a subset of the required functions |
| Juvenile | Stem bee with somatic bees performing all functions (optional: laid eggs, larvae, pupae) |
| Adult | Stem bee with somatic bees performing all functions, sperm bees and/or egg bees (optional: laid eggs, larvae, pupae) |
| Parent | Stem bee with somatic bees performing all functions and at least one fertilized egg bee (optional: laid eggs, larvae, pupae, sperm bees, egg bees) |
| Pathway to reproduction: | supersedure | after departure of prime swarm in original nest | via afterswarm | spontaneous after loss of stem bee |
| Provided nest: | established or newly founded | established | newly founded | established or newly founded |
| Provided stores: | all | fraction | all | all |
| Maternal care by: | entire or fraction of superorganism | fraction of superorganism | fraction of superorganism | entire or fraction of superorganism |
| Postnatal maternal care: | potentially high | low | low | very low or potentially high when reproduction occurs late in the season |
| Survivorship curve: | type I in established nest or II in newly founded nest | type I | type II | type I in established nest or II in newly founded nest |
| Fate of maternal superorganism: | reproduction entails obligate death | may survive reproduction | may survive reproduction | reproduction initiated by impending death |
| Possible number of offspring per reproductive cycle: | 1 (replacement of maternal superorganism) | 1 (may lead to multiplication) | 2 or more (may lead to multiplication) | 1 (replacement of maternal superorganism) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




