1. Introduction
We discuss firstly, at present the Icy Jovian moons especially Europa, as fertile ground for the search of biosignatures. This is done in Secs. 2-6. In Sec. 7 we consider whether upcoming discoveries would suggest that in the future, life could emerge. This could help to answer the question of the first steps taken by the evolution of life on Earth, such as which were the first biomolecules that led to life as we know it. We emphasize that beyond the interpretation of the data gathered by the Galileo mission, there are preliminary works that point towards new detection of chemical entities, such as ammonia arising from the Europa ocean [
1]. Such suggestions open the door for further considerations of new chemical entities, as touched upon in this Review.
2. Probing for Chemical Elements and Their Isotopes on Icy Surfaces of the Jovian Moons
Potential biosignatures as, for instance, the recent report concerning a sample collected by NASA’s Perseverance Mars rover from an ancient dry riverbed in Jezero Crater, could preserve evidence of ancient microbial life, but like the biosignature proposal in this work, requires additional studies before such biosignatures could be accepted to be related to living organisms [
2]. Extending the isotopic fractionation from the rock record to astrobiology has been attempted by means of the index δ
34S, which had been used in the geologic record [
3].
We found it profitable to use isotopic fractionation excursions in astrobiology, but in previous research projects attention has been restricted exclusively to the icy surface of the ocean worlds (OWs), which are celestial bodies (planets, or satellites) that contain a hydrosphere with abundant water forming of oceans, often submerging dry land. In the case that concerns us in this review, the Jovian and other outer solar system moons, OWs are also called icy worlds, when the land has been totally submerged, unlike our own planet, where continents are present.
We have not entered in the wider context of isotopic fractionation taking place in planetary science, since that would clearly require a more extensive discussion. For example, the Life Detection Knowledge Base (LDKB) has been developed by a large group of researchers in order to test and evaluate strategies to search for evidence of life beyond Earth. Their emphasis has been on recognizing potential false-positive and false-negative results in the general case of planetology, rather than in the more restricted case of the icy surfaces of ocean worlds, which is exclusively the main topic to which this review has been dedicated [
5,
6]. The LDKB authors have done a careful analysis for applying life detection criteria to assess isotope abundance patterns as potential biosignatures. They have used previous work [
5] for creating a data base to put some order in planning future missions. With LDKB it will be easier to catalog reliable biosignatures, out of the many that are likely to be proposed after JUICE and Europa Clipper begin their measurements. At the same time, they have provided a repository for the acquisition of new data. These authors [
5] have introduced a useful language for highlighting reliable biosignatures, “high and low arguments”. For instance, isotopic discrimination during biological dissimilatory sulfate reduction can yield delta S values of sulfides that are 15-72%, lower than the reference values δ
34S (‰) of their sulfate source. In section 6 below we shall return to the clear example of such isotopic fractionation due to Wortmann and coworkers.
For the ongoing missions for the Jovian system [
7,
8], sulfur would have the most relevant excursions of isotopic fractionation for the detection of traces of biogenic activity, out of other biogenic elements: carbon, hydrogen, oxygen, nitrogen and phosphorous [
8]. Sulfur consists of four stable isotopes:
32S (95.02%),
33S (0.75%),
34S (4.21%),
34S (4.21%) and
36S (0.02%). Bacterial sulfate reduction (BSR) is a well-understood process, in which the bacterium involved links atoms of hydrogen with sulfur, taken away from dissolved sulfate (SO
4- 2) of seawater to form hydrogen sulfide (H
2S). This gas is enriched in
32S relative to the seawater source [
9,
10].
We consider the redistribution of the primordial isotopic mixtures as usual in terms of the sulfur delta parameter [
11]:
The value of δ34S is taken to be zero when the sample coincides with the corresponding value of the Canyon Diablo meteorite (CDM), a standard troilite (FeS) that was found in Phoenix, Arizona. The useful parameter δ34S allows a comparison of a sample (sa) with the standard (st) CDM. The relevant terms are the dominant sulfur isotope (32S) and the next in abundance (34S). In fact, the quotient (34S/32S)st is the average terrestrial fraction of the two most abundant isotopes of sulfur. In a given sample, we obtain a positive value of δ34S when the quotient (34S/32S) sa exceeds value of the standard CDM; otherwise, we obtain a negative value when the quotient (34S/32S) sa is less than the value of the standard CDM.
Analogously, the biogenic element chlorine has two stable isotopes [
12]:
35Cl (75.78%), 37Cl (24.22%),
The Cl isotope fractionation is discussed in terms of δ
37Cl, which is defined as we did above with the δ
34S parameter of sulfur. Measurements of the relative abundance of Cl isotopes is reported in the per mil notation (‰) vs. Standard Mean Ocean Chloride (SMOC). SMOC is thought to be isotopically homogeneous to within ± 0.15 ‰ [
13].
Interestingly, within our overall objective of searching for reliable indices of biogenicity, the bacterium
Azospira suillum shows a significant stable isotope fractionation excursion of –15 ‰ [
14,
15]. Chlorine has been identified on the icy surface of Europa [
16,
17]. While the model assumes the Europa’s seafloor initial composition to be chondritic Type II has been shown that water-rock cycling at the silicate seafloor is capable of producing a chloride-rich ocean [
18,
19,
20].
With the sulfur isotopes, it is possible to report three measurable delta values δ33S, δ34S and δ36S. The delta parameter, δ34S, has been highlighted for a very long time in the terrestrial rock record as a valuable index of biogenicity, and only more recently in the context of astrobiological discussions, as in our previous papers reported in this Review
. The support for δ
34S distributions, as indices of biogenicity has been extensively illustrated from sediments throughout the terrestrial rock record. Examples that clearly illustrate such support were to a large extent due to the distinguished Polish condensed matter physicist Manfred Schildowski and co-workers, mentioned above [
8]. These examples range throughout the whole evolution of life on Earth, for instance:
(i)Pyrites in banded iron formations (BIF) from the Isua Greenstone Belt dating from the Archaean in the time interval of 3.7-3.8 Gyr BP, which have been assigned δ
34S (‰) of – 4 ± 2 [
21].
(ii)A second example is from a rock formation in the Pilbara region of Western Australia, in which barite deposits of some 3.47 Gyr BP have been assigned values of δ
34S of -5 ± 7) [
22].
(iii)A third example is given by pyrites in black shales from 2.7-2.8 Gyr BP from Western Australia that have a δ
34S of – 8 ± 7 [
23].
Finally, pyrites and other sulfides from the beginning of the Triassic Period from Central Europe, were characterized by δ
34S of – 40 ± 20 [
24]. We should underline that the above examples range from the earliest manifestation of life in the Archaean to a significantly recent geologic time, namely, the Triassic Period 251.9 Myr BP, when multicellular life was widespread.
In fact, both chemical elements, sulfur and chlorine on the surface of the Jovian moon Europa and whose origin is not of an external source, could have been cycled by oceanic dwelling microorganisms. This hypothesis will be available for testing after the arrival of the above-mentioned NASA and ESA missions, provided that sulfur fractionation, due to metabolic modifications that may have taken place underneath the icy shell render such phenomenon measurable. Fractionated sulfur isotopes may reach the upper side of Europa’s icy shell, since for some time we have been aware with telescopic observations of the existence of water vents arising from the Europan ocean. In other words, if microorganisms were responsible for sulfur fractionation in the Jovian OWs, their activity could be detectable from icy shell surficial traces with the sensitivity of the available instrumentation that has been incorporated in the payloads of JUICE and Clipper. To follow this approach for the identification of biosignatures on the icy surface of Europa and other OWs, some instrumental and technical challenges remain. These will be briefly outlined in
Section 4 below.
3. The Identification of Fractionation Due to Abiotic Processes
Thermochemical Processes is a first topic to keep in mind, regarding the Jovian icy moons from the point of view of stable isotope geochemistry. Our recent attempts have been restricted to anticipate how instrumentation could be used for the search for habitable ecosystems in the exploration of Europa and Ganymede [
25]: We have kept in mind the basic discussions how living processes can be distinguished from abiotic processes [
26]. Our previous discussion followed the standard approach [
27], as briefly described in this section.
In the fractionation of stable isotopes, there are two mutually exclusive regimes: for sulfate reduction in the range 60–80°C that should be compared with high temperature in the range of 150–200 °C. These higher temperatures are generally larger than the cases of biogenicity. However, the above two thermal regimes do overlap when aqueous sulfate can be reduced by organic compounds at temperature, close to the water boiling point [
28]. Abiotic fractionations are irrelevant for Europan ocean considerations, where abiotic sulfate reductions are larger than biogenic ones.
More specifically, experiments have only yielded fractionations of 10–20 ‰ for temperatures in the range of 100–200 °C. Even though there is some ocean-seafloor contact, hence raising the possibility of abiotic fractionation, due to the exposure to the temperatures of hydrothermal vents. However, we should keep in mind that, at the seafloor, the emergence of microorganisms would be subject to thermochemical sulfate reduction, but globally this would be largely negligible from the experimental evidence of fractionations being smaller than about 20 ‰ [
29].
When a chemical element is reduced, due to heat, rather than by microorganisms, we should insist in the use of the abbreviated phrase “thermochemical sulfate reduction (TSR)”. This adoption allows us to distinguish this abiotic phenomenon from the following independent sources of abiotic fractionation. Next in importance are the hydrothermal processes. First consider seafloor hydrothermal vent sulfides that are characteristic by low Δ
33 values, where the capital delta parameters are defined as follows:
The capital delta parameters measure deviations from mass-dependent relationships [
30], compared with biogenic sulfides. It should also be remembered that there are small contributions, or even null contributions, from microbial sulfate reduction into hydrothermal sulfides at sediment-free mid-oceanic ridge systems. Besides measurements of δ
x parameter (1), where x = 33, 34, 36 were carried out in four high temperature seafloor hydrothermal vents along sediment-free ridge systems. The sulfur systematics suggest isotopic exchange between sulfate and sulfide at T = 400
ο C. The systematics can decouple microbial sulfur cycling at ocean crust-seawater interfaces, even when δ
34 values lead to non-unique interpretations [
31].
Photochemical processes should also be taken into account. Returning to the above two thermal regimes, there is some overlap between them, but these cases are mostly irrelevant for our main interest that concerns the two OWs that are nearest to planet Jupiter. We should also keep in mind that at the seafloor, the possible origin of life would be subject to TSR, but this would tend to be true, since such fractionations have been verified to be smaller than about -20 ‰ [
32].
A careful experimental survey of SO2 photolysis caused by a series of different UV wavelengths have uncovered a relationship between such wavelengths and sulfur isotope fractionation in the geological record as in the above work of Farquhar and co-workers. These experiments have demonstrated that there is a given wavelength for which SO2 photolysis coincides with certain Archaean samples; other wavelengths in these experiments differ from the observed geological record. This can be used to retrieve information from the terrestrial atmosphere. To sum up, there is a certain wavelength-sensitive mass-independent abiotic sulfur isotope fractionation effect during SO2 photolysis.
It is interesting to highlight the fact that biotic processes that are characteristic of hydrothermal environments can simulate isotopic discrimination exhibited by sulfate-reducing enzymes. For instance, in hydrothermal systems, differences in δ
34S values between sulfate and sulfide of -14‰, to -21‰ have been shown to occur via geochemical (abiotic) processes [
33]. These values are too small to be relevant, for if observed, the magnitude of the δ
34S to be significant (δ
34S > -60 to -80 ‰) would in this case be suggestive of biogenic origin.
4. Fractionation from the Rock Record to the Jovian Moons
Our initial suggestion was to point out the relevance of transferring to the astrobiological context the successful application of geochemistry of the stable sulfur isotope fractionation from the early rock record on earth. Our motivation included in this work we have addressed several questions, the main ones being [
34]:
Why should sulfur isotopes be the main objective of the search for biosignatures?
Would it not be better to rather have a combination of sulfur and carbon isotope anomalies?
Sulfur is unique amongst the biogenic elements since in extraterrestrial material (lunar fines and meteorites) sulfur, rather than carbon, shows in isotopic fractionation a narrow range of values about zero per mil. An additional question in favor of sulfur isotopic fractionation is:
Can you accept some contribution of sulfur from Io, and still find the biogenic fraction in those sulfur deposits?
In this case, since we are assuming from the data that only biogenic processes alter the null values of the isotopic sulfur fractionation, the contribution from the exogenous (Io) sulfur, if it were the product of bacterial metabolism, would not give the significant biogenic signal, that would otherwise be produced by endogenous sulfur.
We have attempted to go from fractionation in the rock record to Galilean moons as follows: Besides long-term planning for the exploration of the moons of the Jovian system, additional search for biosignatures should focus firstly on two of the Galilean moons: Europa and Ganymede. Subsequently, Callisto would also deserve a separate mission too. Fortunately, both missions by ESA and NASA that are already on the way to the Jovian System are equipped with appropriate instrumentation [
35,
36]. In the short term, such instrumentation makes it feasible to explore the exoatmospheres of the Jovian OWs. Since generally Europa's surface composition determines the composition of its atmosphere and can yield information on surface components, most surficial trace species will be present in the extended atmosphere [
37].
In view of the many instruments that are in the payloads of the forthcoming missions to the Jovian system, with the δ
34S index of biogenicity, it is convenient to further comment on only a few of them that would be relevant from the point of view of the biogeochemical biosignatures. Firstly, there are mass spectrometers: In JUICE (the PEP Package with its NIM instrument); secondly, in the Europa Clipper we have MASPEX and SUDA. The NIM instrument is intended to measure the exospheres of the Jupiter satellites during their flybys [
38].
In the present context we could raise the question whether the bioastronomy of OW biosignatures is relevant, for we still do not have a reliable SETI signal, let us now consider some of its consequences. As a working hypothesis, we could assume that the evolution of life in the universe can be explained in terms of evolutionary forces that we experience today. In that case, once the living process has started, it will continue due to evolutionary pressures, as it was explained in detail in the Hawaii Bioastronomy conference [
39].
We can devise experiments addressing specifically the search for the evolution of microorganisms, especially from prokaryotes to eukaryotes, the latter being a first step towards the evolution of intelligence, as we have witnessed on earth. Indeed, several arguments militate in favor of the ubiquity of eukaryogenesis in habitable environments in other worlds, as argued in the San Juan Capistrano Conference No. 5 [
40]. We had argued that in the Drake Equation a factor f
e should be included, which denotes the fraction of planets or satellites, where eukaryogenesis occurs [
41].
For the distribution of life in the universe: one approach is the search for microorganisms to test for the onset of eukaryogenesis in the solar system. Alternatively, the direct approach is a major aim of bioastronomy, namely the time-honored SETI search of (intelligent) life in other solar systems, as proposed by Frank Drake, when in 1960 he set out to search for extraterrestrial intelligence as a legitimate and doable scientific endeavor [
42], which was subsequently widely accepted by the bioastronomy community, in terms of the Drake Equation. Sadly, as underlined at the beginning of this Section, this approach has still not produced a reliable signal, suggesting that alternatives should be followed up, such as searching for OW biosignatures as indicated in this Review.
5. The Future of Jovian Ocean World Exploration
In an astrobiological context of biosignatures from the Europan ocean, the minor S isotopic biosignatures are not corrections of an order of magnitude comparable with the biosignatures in the terrestrial cases, where there are definite large fractionation excursions of up to 72 ‰ that have been caused by
in-situ microbial sulfate reduction [
43]. Provided the result of the orbital Jovian measurement gave sufficient large fractionation excursions (> 50-60 ‰, or larger), the measurements of large δ
34S could be interpreted as a biosignature. We have insisted that measurements of the Jovian orbital missions ought to search for the presence of significant large values of δ
34S.
With the two ongoing probes for Europa or Ganymede, or with a subsequent lander on any of the OWs of the Jovian system, the isotopic abundances discussed in the present review should be complementary with other searches for life. Eventually, the measured anomalous isotopic abundances could be a first step towards an early identification of the existence of other life away from planet earth, possibly when JUICE and Clipper begin gathering data in the 2030s, or subsequently.
A Europa lander even as a mission concept, an eventual Europa lander remains an appealing project of NASA [
44]. This is not a new concept, since early proposals were made at the end of the Twentieth Century, in which a probe called a cryobot capable of penetration through the icy shall of the Jovian moon, provided with a miniature submarine probe (hydrobot) could attempt exploring the Europan ocean.
Such a proposal initiated at JPL, was appealing to some of us at that time [
45]; but at present, it is not clear whether a Europa lander would be capable of carrying a hydrobot in its payload and deliver it into the ocean. With our present technology and, especially with the current limited financial support, this possibility is definitely to be excluded in the short term, but it should not necessarily be discarded in the long term. Yet more recently some more advanced proposals have been tentatively explored with significant financial support, such as an extension of the cryobot miniature surficial probes such as PRIME, the comprehensive cryobot architecture for accessing Europa's ocean [
46].
To conclude with Europa landers, some recent more realistic proposal proposes to focus on a Europa lander instrument. It is suggested to couple a mass analyzer with sample introduction/laser ablation/desorption ion source (ORIGIN instrument).
This instrument is more like LMS with the laser ablation/desorption ion source [
47]. But the work of Pitesky and Hand summarize a further series of ideas that ought to be followed up after the “post-Europa Clipper” era.
6. Instrumentation Challenges, Uncertainties and Implicit Hypotheses
INSTRUMENTATION CHALLENGES. It is intuitive to consider especially the instruments on JUICE and Europa clipper but there remains the challenge to demonstrate how these instruments could determine the sulfur isotopic ratios and if the results would be accurate enough. For example: NIM on JUICE is expected to have only a mass resolution of around 800.
In the long term, it is uncertain whether penetrating instruments (cryobots) may not be an option to get further insights of the chemical nature of the ocean to constrain the distribution of chemical elements on the OWs icy surfaces.
UNCERTAINTIES. A mass resolution of around 800 is not sufficient to differentiate, e.g., between H2O2, 18O16O,34S, and H2S, which all have the nominal mass 34. Radiolysis/photolysis species could be expected in realistic observation scenarios and thus should be considered. The mass resolution of SUDA is even less than that of NIM. Only MASPEX has a sufficient resolution to allow delineation of the species on nominal mass 34. Our suggestion is only to be interpreted as a first step towards potential reliable biosignatures
IMPLICIT HYPOTHESES. Related is the question of how the (heavy isotope enriched) sulfate anions reach these mass spectrometers. For SUDA, which has the lowest mass resolution, it seems intuitive. It is less obvious for NIM or MASPEX.
Another implicit hypothesis is whether all the sulfur on the icy surfaces of the ocean worlds, including specific case of Europa, is exogenic. There remains the question whether the sulfur that could emanate from hydrothermal vents could reach the lower side of the icy shell and eventually contaminate the exogenic sulfur, whose source could be due exogenic, possibly emanating from the Jovian moon Io’s significant volcanic eruptions.
Tentatively there has been a report of the detection of a characteristic NH
3 absorption feature at 2.20 ± 0.02 µm on Europa’s icy surface by careful studies of the NIMS Spectrometer. that suggests NH
3-hydrate and NH
4-chloride are the most likely candidates. The implication is that there has been emplacement from the underground (or shallow subsurface). The paper suggests transportation to the surface via cryovolcanism during recent geological past [
1]. The presence of ammoniated compounds implies a thinner ice shell [
49] and a thicker, chemically reduced, high-pH subsurface ocean on Europa [
50]. Besides reporting the detection of NH
3-bearing components, this study also presents the first evidence of a N-bearing species on Europa, an element that is intimately related to life as we know it on earth, especially for the synthesis of more complex biomolecules such as amino acids, nucleotides and phospholipids.
7. Discovering Evidence for Future Life on Ocean Moons
There are in fact two quite different approaches for finding chemical entities on planets, or their satellites. The first is the attempt, as in the above Secs 2-6, to discover chemical phenomena suggesting that life is currently existing on the planet or their moons, or whether life existed there in the past. The second approach, to be reviewed in the present
Section 7, consists in seeking chemical phenomena that are rather primitive, but with published models, can predict a path towards life. In the previous sections, we discussed evidence for past or present life on Icy Jovian moons, especially Europa, as fertile ground for the search of biosignatures. Here, instead, we consider whether upcoming discoveries from the current missions to the Jovian system or later, would suggest that life could emerge. This could help to answer questions about the early stages of life’s emergence on Earth.
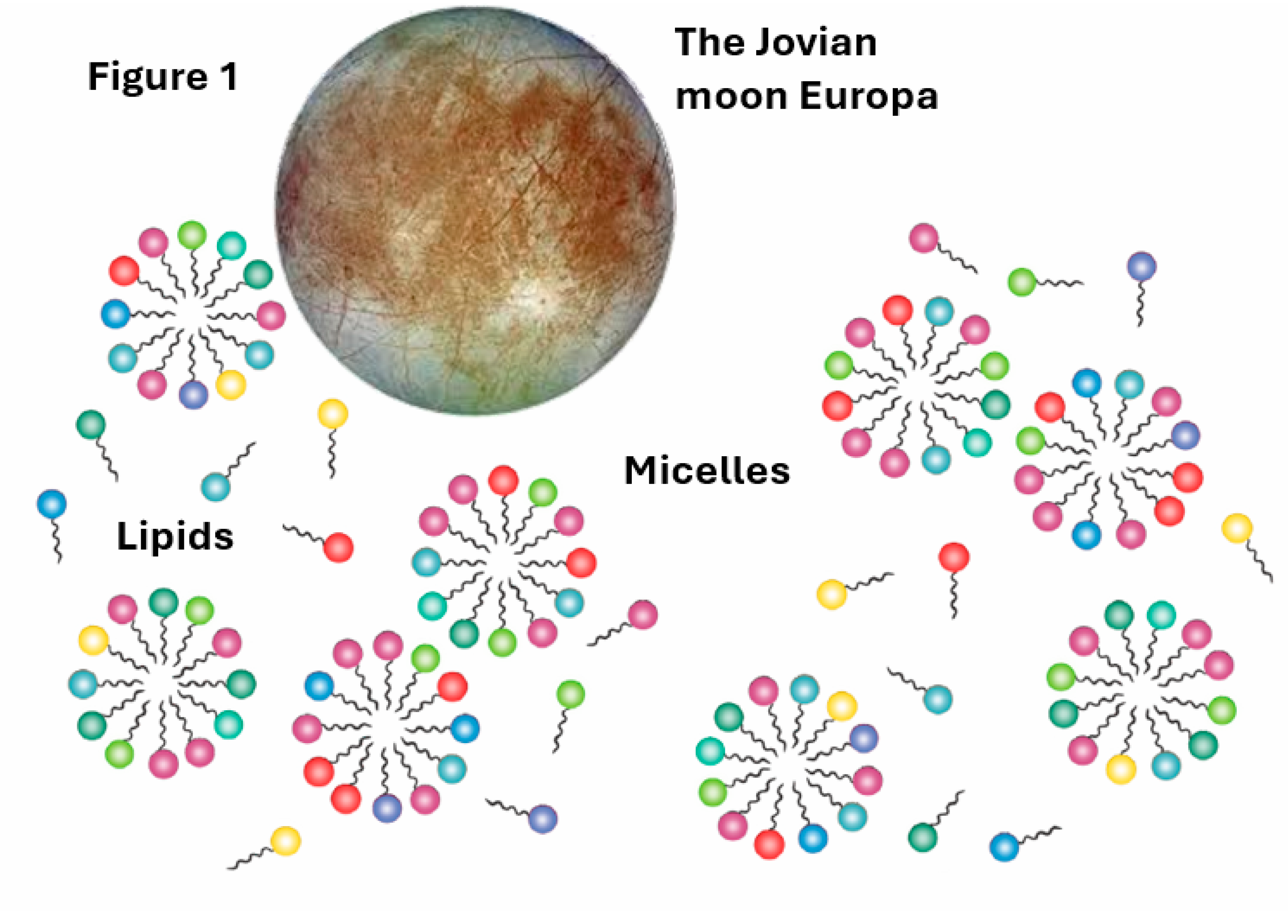
In line with the Lipid World hypothesis [
51], which is realized and quantitated by the GARD model, a group of amphiphile aggregates, e.g., micelles or vesicles, may be protocell precursors [
52]. The most important finding portrayed by computer simulations show that some of these aggregates can undergo self-reproduction [
53,
54] (
Figure 1).
We now intend to discuss how the instrumentation currently included in Europa Clipper’s payload can support the Lipid World hypothesis. The Clipper’s Mass Spectrometer for Planetary EXploration (MASPEX) can generally measure volatile and organic molecules in Europa’s sputtered and radiolytically processed exosphere as well as ocean plumes, at very high sensitivity and resolution [
57]. This is the best shot at detecting amphiphilic organics, beginning with fatty acids, and ending with rather complex lipid-like molecules that can aggregate into micelles and vesicles. The SUrface Dust Analyzer (SUDA), which is a dust/ice grain impact time-of-flight mass spectrometer, analyzes ice grains/ejecta and is particularly strong at salts and organics in grains, which has importance for reporting counterions, and salinity that set aggregation behavior i.e. critical micelle concentration (CMC) [
58] (
Figure 2).
Taken together, both technologies can also help find chain-length distributions and headgroup diversity that can define amphiphile populations rather than random trace organics. The above information allows determining a whole spectrum relevant to protocellular chemistry. Careful analyses of the data and discovering amphiphiles in non-equilibrium ratios should provide further robustness. All in all, the data collected could provide support for the idea that ocean worlds could harbor amphiphile mixtures, or populations that would lead to a lipid-first origin of life. Results from the current mission could also guide future landers, including sample-return missions.
To demonstrate reproduction or heredity, or infer specific structures (e.g., micelles or vesicles) directly there is a need to show high similarity of composition and that they are progeny of a composome (attractor-like reproducing state predicted by the GARD model) reproducing with only a few mutations. Such groups may be considered protospecies, hence may be viewed as a seed of life, capable of undergoing natural selection, the prerequisite for Darwinian evolution.
A central requirement for evaluating compositional heredity in Europa samples, in future endeavors, is the ability to resolve the molecular composition of lipid micelles after controlled growth–dilution cycles, allowing detection of composomes. This necessitates instrumentation capable of handling microliter-scale melted-ice aliquots, separating complex amphiphile mixtures, and quantifying their relative abundances with sufficient resolution to distinguish non-random chemical organization from stochastic assembly. Space-qualified microfluidic capillary electrophoresis (CE) platforms, most notably the “Chemical Laptop”, a fully automated CE–LIF (capillary electrophoresis - laser-induced fluorescence) system for high-resolution organic separations [
59,
60], and the more recent Microfluidic Organic Analyzer (MOA) [
61], which integrates a programmable microfluidic handler with a precision CE wafer and is explicitly targeted to ocean-world missions, provide robust separation of amphiphiles by charge and size, converting each micelle into a measurable electrophoretic composition vector. Complementary detection can be achieved using miniature laser-desorption mass spectrometry (LDMS) [
62], in which micelles are disrupted and their components resolved by mass-to-charge ratio, yielding quantitative stoichiometric spectra. Front-end sample preparation—including melting, filtration, derivatization, reagent addition, and aliquoting—is supported by monolithic lab-on-chip processors such as SPLIce [
63], while auxiliary techniques such as contactless conductivity microfluidics for ionic and amphiphile profiling [
64] and digital microfluidics for controlled mixing, splitting, and serial growth–dilution cycles [
65] provide additional operational flexibility. Together, these microfluidic and spectrometric technologies form a realistic, flight-deployable analytical architecture in which micellar compositions—whether random or convergent—can be reconstructed across multiple aliquots and compared statistically to the compositional clustering expected for GARD-type composomes.
In parallel, new technologies are being utilized in terrestrial settings that have, with modification, potential to be employed in extraterrestrial settings as well. One such technology, termed CytoFLEX nano flow cytometer, is currently in use to profile extracellular vesicles [
66,
67] and lipid nanoparticles (LNPs) [
68,
69]. While at this point in time, the CytoFLEX nano flow cytometer is better suited for > 80 nm micelle profiling, in the near future this might be honed down to also suit smaller sizes. Providing it is further fine-tuned to a space-qualified nanoscale cytometer, the advantage of such instrumentation would be direct detection of single-particle biosignatures as opposed to bulk composition detection with CE-LIF. Although the sensitivity for micelles is borderline, the next step in evolution, i.e. fusion of micelles to much larger vesicular protocell precursors (0.1-10µm) [
53], and that both micelles and vesicles can show the emergence of protospecies, could be significantly supported by the above-mentioned technology (
Figure 3).
8. Conclusion
We have suggested that the search for biosignatures on Europa’s icy surface should aim at the search for a High Argument signal, in the LDKB terminology, by identifying a definite S-isotopic excursion. Such potential biosignatures, if detected, should be supplemented soon by independent evidence to produce complementary evidence of truly a biogenic origin. In other words, the ongoing Jovian Orbital Missions in due course could search for large values of δ34S for the possible detection of meaningful values.
This could occur either orbitally, or eventually by means of a lander. Thus, such results would be a first step for the detection of a possible biosignature, to be confirmed by independent measurements. Some possibilities have been explored by looking into the early stages of life on earth. The astrobiological experimentation described in Sec 7 would be useful for discovering chemical entities on oceanic moons, protospecies that could be evidence for on the surfaces of Europa and Ganymede that would be a route to future life’s emergence.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study.
Acknowledgments
One of the authors (JCF) would like to thank Dr Atish Dabholkar, the International Atomic Agency and UNESCO for hospitality at The Abdus Salam International Centre for Theoretical Physics, Trieste, Italy. The other authors (DL, RY) acknowledge grant from the Minerva Foundation.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| BIF |
Banded Iron Formation Before the present. |
| BP |
Before the present. |
| BSR |
Bacterial sulfate reduction. |
| CE |
Capillary electrophoresis. |
| CDM |
Canyon Diablo meteorite. |
| CMC |
Critical Micelle Concentration |
| CytoFLEX |
Nano flow cytometer. |
| ESA |
European Space Agency. |
| GARD Model: |
Graded Autocatalysis Replication Domain. |
| LIF |
Laser-induced fluorescence detection. |
| LDKB |
Life Detection Knowledge Base. |
| LDMS |
Laser-desorption mass spectrometry. |
| LIMS |
Laser ablation ionization mass spectrometry. |
| LMS |
Laser-Abation Time-of-Flight Mass Spectrometer. |
| LNPs |
Lipid nanoparticles. |
| MASPEX |
MAss SPectrometer for Planetary EXploration/Europa (Europa Clipper). |
| MOA |
Microfluidic Organic Analyzer. |
| MSR |
Microbial sulfate reducers. |
| NASA |
National Aeronautics Space Administration. |
| NIM |
Neutral Ion Mass Spectrometer (JUICE). |
| NIMS |
Near Infrared Mapping Spectrometer (Galileo). |
| ORIGIN |
ORganics Information Gathering Instrument. |
| OW |
Ocean world. |
| PEP |
Particle Environment Package (JUICE). |
| PRIME |
Probe using Radioisotopes for Icy Moons Exploration. |
| SETI |
Search for Extraterrestrial Intelligence. |
| SIG |
Stable Isotope Geochemistry. |
| SMOC |
Standard mean ocean chloride. |
| SPLIce |
Sample Processor for Life on Icy Worlds. |
| SRM |
Sulfate reducing microorganisms. |
| SUDA |
SUrface Dust Analyzer (Europa Clipper). |
| TSR |
Thermochemical sulfate reduction. |
References
- Emran, A. Europa’s Composition as Evidenced by Spectral Analysis of Galileo NIMS Data. Intended for Astrophysical Journal Letters.. 2025. [Google Scholar]
- Hurowitz, JA; Tice, MM; Allwood, ACet. Redox-driven mineral and organic associations in Jezero Crater, Mars. Nature 2025, 645, 332–340. [Google Scholar] [CrossRef]
- Schidlowski, M; Hayes, JM; Kaplan, IR. Isotopic inferences of ancient biochemistries: carbon, sulfur, hydrogen and nitrogen. In Earths Earliest Biosphere, Its Origin and Evolution; Schopf, JW, Ed.; Princeton University Press: Princeton, NJ, 1983; pp. 149–186. [Google Scholar]
- Davila, A; Hoehler, T; Parenteau, N. Life Detection Knowledge Base: Taxonomy of Potential Biosignatures. Astrobiology 2025, 25(7), 464–473. [Google Scholar] [CrossRef]
- Des Marais, DJ; Caro, T; Dhingra, R; Fox, AC; Galloway, T. Stable Isotope Abundance Patterns as Potential Biosignatures. Astrobiology 2025, 25(8), 537–549. [Google Scholar] [CrossRef]
- Pohorille, A.; Lau, G; Gliniewiez, S; et al. The life detection knowledge base: A community tool for knowledge management and representation. Astrobiology 2025, 25((71), 454–463. [Google Scholar] [CrossRef]
- Grasset, O; Dougherty, MK; Coustenis, A. JUpiter ICy Moons Explorer (JUICE): An ESA Mission to Orbit Ganymede and to Characterize the Jupiter System. Planet Space Sci 2013, 78, 1–21. [Google Scholar] [CrossRef]
- Phillips, CB; Pappalardo, RT. Europa Clipper Mission Concept. Eos Transactions. American Geophys Union 2014, 95(20), 165–167. [Google Scholar] [CrossRef]
- Ryu, J; Zierenberg, R; Dahlgren, RA. Sulfur biogeochemistry and isotopic fractionation in shallow groundwater and sediments of Owens Dry Lake, California. Chemical Geology 2006, 229, 257–72. [Google Scholar] [CrossRef]
- Schopf, JW. Cradle of Life; Princeton University Press: Princeton, New Jersey, USA, 2009; p. 179. [Google Scholar]
- Kaplan, IR. Stable isotopes as a guide to biogeochemical processes. Proceedings of the Royal Society of London. Series B 1975, 189, 183–211. [Google Scholar] [CrossRef]
- Coplen, TB; Böhlke, JK; De Bièvre, P. Isotope-abundance variations of selected elements (IUPAC Technical Report). Pure and Applied Chemistry 2002, 74(10), 1987–2017. [Google Scholar] [CrossRef]
- Kaufmann, RS; Long, A; Campbell, DJ. Chlorine isotope distribution in formation waters, Texas and Louisiana. Am Assoc Petrol Geol Bull 1988, 72, 839–844. [Google Scholar]
- Coates, JD; Achenbach, LA. Microbial perchlorate reduction: rocket-fueled metabolism. Nature Reviews Microbiology 2004, 2(7), 569–580. [Google Scholar] [CrossRef]
- Coleman, ML; Ader, M; Chaudhuri, S; Coates, JD. Microbial isotopic fractionation of perchlorate chlorine. Appl Environ Microbiol 2003, 69, 4997–5000. [Google Scholar] [CrossRef]
- Ligier, N; Poulet, F; Carter, J. VLT/SINFONI Observations of Europa: New Insights into the Surface Composition. The Astronomical Journal 2016, 151(6), 163. [Google Scholar] [CrossRef]
- Trumbo, S; Brown, ME; Hand, K. Sodium Chloride on the Surface of Europa. Science Advances 2019, 5((6)). [Google Scholar] [CrossRef] [PubMed]
- Glein, CR; Baross, JA; Waite, JH, Jr. The pH of Enceladus’ocean. Geochimica et Cosmochimica Acta 2015, 162, 202–219. [Google Scholar] [CrossRef]
- Zolotov, MY. An oceanic composition on early and today's Enceladus. Geophys Res Lett 2007, 34, L23203. [Google Scholar] [CrossRef]
- Glein, CR; Shock, L. A geochemical model of non-ideal solutions in the methane–ethane–propane–nitrogen–acetylene system on Titan. Geochimica et Cosmochimica Acta 2010, 115, 217–240. [Google Scholar] [CrossRef]
- Monster, J; Appel, P.W.U.; Thode, H.G. Sulfur isotope studies on Early Archaean sediments from Isua, West Greenland: Implications for the antiquity of bacterial sulfate reduction. Geochimica Cosmochimica Acta 1979, 43, 405–413. [Google Scholar] [CrossRef]
- Shen, Y.; Buick, R.; Canfield, D. E. Isotopic evidence for microbial sulphate reduction in the Early Archaean era. Nature 2001, 410, 77–81. [Google Scholar] [CrossRef]
- Donnelly, T. H.; et al. A reconnaissance study of stable isotope ratios in Archaean rocks from the Yilgarn Block, Western Australia. Journal of the Geological Society of Australia 1977, 24(7-8), 409–420. [Google Scholar] [CrossRef]
- Goodwin, A.M; Monster, J; Thode, HG. Carbon and sulfur isotope abundances in Archean iron-formations and early Precambrian life. Econ Geol 1976, 71, 870–891. [Google Scholar] [CrossRef]
- Chela-Flores, J. Habitability of Ganymede. In Ganymede (Cambridge Planetary Science); Volwerk, M, McGrath, M, Jia, X, Spohn, T., Eds.; Cambridge University Press: Cambridge; Cambridge Planetary Science (28): Series, 2025. [Google Scholar]
- Chela-Flores, J. Instrumentation for the search for habitable ecosystems in the future exploration of Europa and Ganymede. International Journal of Astrobiology 2010, 9(2), 101–108. [Google Scholar] [CrossRef]
- Machel, HG; Krouse, HR; Sassen, R. Products and distinguishing criteria of bacterial and thermochemical sulfate reduction. Applied Geochemistry 1995, 10, 373–389. [Google Scholar] [CrossRef]
- Krouse, HR; Viau, CA; Eliuk, LS; Ueda, A; Halas, S. Chemical and isotopic evidence of thermochemical sulfate reduction by light-hydrocarbon gases in deep carbonate reservoirs. Nature 1998, 333, 415–419. [Google Scholar] [CrossRef]
- Kiyosu, Y; Krouse, HR. The role of organic acid in the abiogenic reduction of sulfate and the sulfur isotope effect. Geochemical Journal 1990, 24, 21–27. [Google Scholar] [CrossRef]
- Gao, X; Thiemens, MH. Systematic study of sulfur isotopic composition in iron meteorites and the occurrence of excess 33S and 36S. Geochimica Cosmochimica Acta 1991, 55, 2671–2679. [Google Scholar] [CrossRef]
- Ono, S; Shanks, WC; Rouxel, O; Rumble, D. S-33 constraints on the seawater sulfate contribution in modern seafloor hydrothermal vent sulfides. Geochimica Cosmochimica Acta 2007, 71, 1170–1182. [Google Scholar] [CrossRef]
- Farquhar, J; Savarino, J; Airieau, S; Thiemens, MH. Observation of wavelength-sensitive mass-independent sulfur isotope effects during SO2 photolysis: Implications for the early atmosphere. Journal of Geophysical Research 2001, 106, 1–11. [Google Scholar] [CrossRef]
- Kokh, MA; Assayag, N; Mounic, S; et al. Multiple sulfur isotope fractionation in hydrothermal systems in the presence of radical ions and molecular sulfur. Geochim Cosmochim Acta 2020, 285, 100–128. [Google Scholar] [CrossRef]
- Chela-Flores, J. The sulphur dilemma: are there biosignatures on Europa’s icy and patchy surface? International Journal of Astrobiology 2006, 5(1), 17–22. [Google Scholar] [CrossRef]
- Grasset, O; Dougherty, MK; Coustenis, A; Bunce, EJ; Erd, C; Titov, D; Blanc, M; Coates, A; Drossart, P; Fletcher, LN; Hussmann, N; Jaumann, H; Krupp, R; Lebreton, JP; Prieto-Ballesteros, O; Tortora, P; Tosi, F; Van Hoolst, T. JUpiter ICy moons Explorer (JUICE): An ESA mission to orbit Ganymede and to characterize the Jupiter system. Planetary and Space Science 2013, 78, 1–21. [Google Scholar] [CrossRef]
- Pappalardo, RT; Buratti, BJ; Korth, H; Senske, DA; Blaney, DL; Blankenship, DD; Burch, J.; Christensen, PR; Kempf, S; Kivelson, MG; Mazarico, E; Retherford, KD; Turtle, EP; Westlake, JH; Paczkowski, BG; Ray, TL; Kampmeier, J; Craft, KL; Howell, SM; Hay, HCFC. Science Overview of the Europa Clipper Mission. Space Science Reviews For payload instruments cf. Table 1. 2024, 220(4). [Google Scholar] [CrossRef]
- Chela-Flores, J. Miniaturized instrumentation for biosignatures on the ocean worlds in the solar system. Frontiers in Space Technology 2021. [Google Scholar] [CrossRef]
- Föhn, M; Galli, A; Vorburger, A; Tulej, M; Lasi, D; Riedo, A; Fausch, RG; Althaus, M; Brüngger, S; Fahrer, P; Gerber, M; Lüthi, M; Munz, HP; Oeschger, S; Piazza, D; Wurz, P. Description of the Mass Spectrometer for the Jupiter Icy Moons Explorer Mission. IEEE 14 2021. [Google Scholar]
- Chela-Flores, J. Testing the Drake Equation in the Solar System. In Bioastronomy '99 A New Era in Astronomy; San Francisco, Proceedings of the meeting at Hapuna Beach Prince Hotel, Kohala Coast, Hawaii, USA, Lemarchand, G.A., Meech, K., Eds.; Astronomical Society of the Pacific Conference Series, 2-6 August 2000; Volume 213, pp. 402–410. [Google Scholar]
- 40; Chela-Flores, J. Habitability of Europa: possible degree of evolution of Europan biota. Europa Ocean Conference, Capistrano Conference, Nov. 12–14; San Juan Capistrano Research Institute: San Juan Capistrano, CA, 1996; No. 5, pp. 21–21a. Available online: https://users.ictp.it/~chelaf/ss29.html.
- Chela-Flores, J. A search for extraterrestrial eukaryotes: physical and paleontological aspects. Origins of Life and Evolution of Biosphere 1998, 28(4-6), 583–96. [Google Scholar] [CrossRef] [PubMed]
- Drake, F; Sobel, D. Is Anyone Out There? The Scientific Search for Extraterrestrial Intelligence; Dalacorte Press: New York, 1992; pp. ix–xv. [Google Scholar]
- Wortmann, UG; Bernasconi, SM; Böttcher, ME. Hypersulfidic deep biosphere indicates extreme sulfur isotope fractionation during single-step microbial sulfate reduction. Geology 2001, 29(7), 647–650. [Google Scholar] [CrossRef]
- Pitesky, JKP; Hand, CB. The Europa Lander Mission Concept: Situ Exploration of an Ocean World; AGU Fall Meeting: San Francisco, 2020; pp. 0511–P101. [Google Scholar]
- Horvath, J; Carsey, F; Cutts, J; Chela-Flores, J. Searching for ice and ocean biogenic activity on Europa and Earth. In Instruments, Methods and Missions for Investigation of Extraterrestrial Microorganisms Proc. SPIE 3111; Hoover, RB, Ed.; Society of Photo-Optical Instrumentation Engineers (SPIE): Bellingham, Washington, USA, 1997; pp. 490–500. [Google Scholar]
- Hockman, BJ; Smith, MW; Howell, SM. PRIME: Probe using Radioisotopes for Icy Moons Exploration- A Comprehensive Cryobot Architecture for Accessing Europa's Ocean. The Astrobiology Science Conference (AbSciCon) Bibcode 2022absc.conf50204H, held in Atlanta, Georgia, US, 2022. [Google Scholar]
- Ligterink, NFW; Grimaudo, V; Moreno-García, P; Lukmanov, R; Tulej, M; Leya, I; Lindner, R; Wurz, P; Cockell, CS; Ehrenfreund, P; Riedo, A. ORIGIN: a Novel and Compact Laser Desorption – Mass Spectrometry System for Sensitive in situ Detection of Amino Acids on Extraterrestrial Surfaces. Scientific Reports 2020, 10, 9641. [Google Scholar] [CrossRef]
- Emran, A. Europa’s Composition as Evidenced by Spectral Analysis of Galileo NIMS Data. Intended for Astrophysical Journal Letters.. 2025. [Google Scholar]
- Spohn, T; Schubert, G. Oceans in the icy Galilean satellites of Jupiter? Icarus 2003, 161(2), 456–467. [Google Scholar] [CrossRef]
- Hand, KP; Chyba, CF; Priscu, JC. Astrobiology and the Potential for Life on Europa. In Europa; Pappalardo, Robert T., McKinnon, William B., Khurana, Krishan K., Eds.; with the assistance of René Dotson with 85 collaborating authors. University of Arizona Press: Tucson; The University of Arizona space science series ISBN: 9780816528448, 2009; p. 589. [Google Scholar]
- Segré, D.; Ben-Eli, D.; Deamer, D W; Lancet, D. The lipid world. Origins of Life and Evolution of the Biosphere 2001, 31, 119–145. [Google Scholar] [CrossRef]
- Kahana, A.; Lancet, D. Self-reproducing catalytic micelles as nanoscopic protocell precursors. Nature Reviews Chemistry 2021, 5(12), 870–878. [Google Scholar] [CrossRef] [PubMed]
- Segré, D.; Ben-Eli, D.; Lancet, D. Compositional genomes: Prebiotic information transfer in mutually catalytic noncovalent assemblies. Proc. Natl. Acad. Sci. U.S.A 2000, 97(8), 4112–4117. [Google Scholar] [CrossRef] [PubMed]
- Lancet, D.; Zidovetzki, R.; Markovitch, O. Systems protobiology: origin of life in lipid catalytic networks. Journal of The Royal Society Interface 2018, 15(144), 20180159. [Google Scholar] [CrossRef]
- Kahana, A; Segev, L; Lancet, D. Attractor dynamics drives self-reproduction in protobiological catalytic networks. Cell ReportsPhysical Science 2023, 4(5). [Google Scholar] [CrossRef]
- Lancet, D; Yaniv, R. Reproducing lipid micelles permit early Darwinian evolution. Scientia 1302 2025. [Google Scholar] [CrossRef]
- Waite, JH; Burch, JL; Brockwell, TG; et al. MASPEX-Europa: The Europa Clipper Neutral Gas Mass Spectrometer Investigation. Space Sci Rev 2024, 220, 30. [Google Scholar] [CrossRef]
- Kempf, S; Tucker, S; Altobelli, N; et al. SUDA: A SUrface Dust Analyser for Compositional Mapping of the Galilean Moon Europa. Space Sci Rev 2025, 221, 10. [Google Scholar] [CrossRef]
- Kehl, F; Drevinskas, T; Creamer, J. Providing Enhanced Migration Time Reproducibility with a High-Voltage-Compatible Flow Sensor for Capillary Electrophoresis. Anal Chem 2022, 94, 5734−5740. [Google Scholar] [CrossRef]
- Oborny, NJ; Kehl, F; Cretu, V. A radiation tolerant laser-induced fluorescence detection system for a potential Europa Lander mission. Acta Astronautica 2021, 186, 465–472. [Google Scholar] [CrossRef]
- Butterworth, AL; Golozar, M; Estlack, Z. Integrated high performance microfluidic organic analysis instrument for planetary and space exploration. Lab Chip 2024, 24(9), 2551–2560. [Google Scholar] [CrossRef]
- Arevalo, R; Willhite, L; Bardyn, A.; et al. Laser desorption mass spectrometry with an Orbitrap analyser for in situ astrobiology. Nat Astron 2023, 7, 359–365. [Google Scholar] [CrossRef]
- Harandi, B; Ng, S; Liddell, LC; Gentry, DM; Santa Maria, SR. Fluidic-Based Instruments for Space Biology Research in CubeSats. Frontiers in Space Technologies 2022, 3, 1–9. [Google Scholar] [CrossRef]
- MacKenzie, SM; Neveu, M; Davila, AF. Science Objectives for Flagship-Class Mission Concepts for the Search for Evidence of Life at Enceladus. Astrobiology 2022, 22(6), 685–712. [Google Scholar] [CrossRef]
- Jensen, EC; Stockton, AM; Chiesl, TN. Digitally programmable microfluidic automaton for multiscale combinatorial mixing and sample processing. Lab Chip 2013, 13(2), 288–96. [Google Scholar] [CrossRef] [PubMed]
- Cao, L; Wang, H; Zhu, J. Exploring exosome profiling via CytoFLEX Nano flow cytometer: Approaches and applications. View 2025, 6(6), 20250068. [Google Scholar] [CrossRef]
- Hu, Q; Wu, J; Chen, X. See the Unseen: Rapid, Multiparametric Single-Particle Analysis of Extracellular Vesicles With Nano-Flow Cytometry. Proteomics 2025, e70078. [Google Scholar] [CrossRef]
- Yu, H; Song, S. Application of nano-flow cytometry for the detection of extracellular vesicles and lipid nanoparticles. Journal of Chinese Pharmaceutical Sciences 2025, 34(10), 943–953. [Google Scholar] [CrossRef]
- Gao, W; An, K; Ma, Y. Resolving the mRNA Encapsulation-Release Trade-off via Compensatory Forces in Engineered Ioni zable Lipids. Adv. Matere 12235 2025. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).