1. Introduction
1.1. Motivation
The relationship between thermodynamics, information, and complexity has become increasingly central across physics, computation, and complex systems research. From disordered materials and glassy dynamics to information processing in physical substrates, it has become clear that energetic considerations alone are often insufficient to characterize the macroscopic behavior of systems with highly structured state spaces. In many such systems, the number and organization of long-lived macroscopic configurations plays a decisive role. Glassy materials, spin glasses, optimization landscapes, and certain classes of interacting many-body systems exhibit rugged free-energy landscapes with exponentially many metastable states. These features influence equilibration times, response functions, and stability properties, yet they are not explicitly represented in the standard thermodynamic state space. At the same time, parallel developments in the thermodynamics of information and computation have highlighted deep connections between physical resources, constraints, and informational structure. While foundational results such as Landauer’s principle establish minimal energetic costs for logically irreversible operations, they do not address how the
structure of a system’s state space influences equilibrium thermodynamics [
1,
2].
These observations motivate the exploration of extended thermodynamic descriptions capable of incorporating landscape-level structural information in a controlled and physically meaningful way.
1.2. Background and Related Work
The role of complexity-like quantities in physics has been explored from multiple perspectives. In the statistical mechanics of disordered systems, configurational complexity (also known as configurational entropy) quantifies the number of metastable macrostates and has proven essential for understanding glass transitions, ergodicity breaking, and aging phenomena [
3,
4]. Independently, generalized ensembles and constrained maximum-entropy formalisms have been developed to study systems subject to non-standard macroscopic constraints, including fixed magnetization, activity, or other collective observables. Such approaches are well established within equilibrium statistical mechanics and large deviation theory, where additional conjugate variables naturally emerge as Lagrange multipliers enforcing macroscopic constraints [
5].
More recently, thermodynamic geometry has been employed as a diagnostic tool to characterize phase transitions and stability properties by endowing the space of thermodynamic states with a metric structure. Extensions of this approach have been proposed in a variety of contexts, including systems with additional control parameters beyond the traditional variables [
6]. Despite these advances, there remains a gap between landscape-based notions of complexity and their systematic incorporation into thermodynamic frameworks. In particular, complexity is often discussed qualitatively or as an emergent property, rather than treated as a controlled macroscopic coordinate within equilibrium thermodynamics.
1.3. Scope and Claims of This Work
The purpose of the present work is to bridge this gap by introducing a constrained thermodynamic framework in which a well-defined complexity observable is treated as an additional macroscopic coordinate. Specifically, we consider equilibrium ensembles subject to simultaneous constraints on energy and configurational complexity, leading to an extended thermodynamic description with an associated conjugate variable. It is essential to clarify the scope of this contribution. This work does not propose a modification of the fundamental laws of thermodynamics, nor does it claim universality across all physical or computational systems. Instead, it should be understood as a phenomenological framework applicable to systems in which configurational complexity is a meaningful and well-defined quantity.
The framework developed here:
operates strictly within equilibrium statistical mechanics,
relies on explicit macroscopic constraints introduced via entropy maximization,
treats complexity as a landscape-level property rather than an algorithmic measure,
yields concrete, model-dependent predictions when applied to specific systems.
To demonstrate the utility of this approach, we present a worked example based on a mean-field glassy landscape, where configurational complexity can be computed explicitly and its thermodynamic consequences analyzed in detail.
1.4. Organization of the Paper
The remainder of this paper is structured as follows. In
Section 2, we provide a precise definition of the complexity variable used throughout this work and distinguish it from other notions of complexity.
Section 3 introduces the complexity-constrained thermodynamic formalism and derives the associated extended relations.
Section 4 constructs the corresponding complexity-biased equilibrium ensemble. In
Section 5, we present a concrete worked example illustrating the framework in a glassy system.
Section 6 discusses thermodynamic geometry in the extended state space.
Section 7 addresses the relation to computation and Landauer’s principle. Finally,
Section 8 and
Section 9 summarize the results, discuss limitations, and outline future directions.
2. Definition of the Complexity Variable
A central requirement for incorporating complexity into a thermodynamic framework is that the corresponding quantity be defined in a precise, operational, and physically interpretable manner. In this work, we adopt a definition of complexity that is firmly rooted in statistical physics and has been extensively studied in the context of disordered and glassy systems. Importantly, we restrict ourselves to a single, unambiguous notion of complexity throughout the manuscript.
2.1. Configurational Complexity in Disordered Systems
We define the complexity variable
C as the
configurational complexity (also known as the
complexity or
configurational entropy) of a thermodynamic system. For a system of
N degrees of freedom, configurational complexity is defined as
where
denotes the number of distinct metastable states (or free-energy minima) with energy density
E. This quantity characterizes the multiplicity of long-lived macroscopic configurations compatible with a given energetic constraint. Unlike the standard thermodynamic entropy, which counts microscopic configurations within a single equilibrium basin, the configurational complexity counts the number of distinct basins themselves. As such,
C encodes global information about the structure of the system’s energy landscape. Configurational complexity plays a central role in the statistical mechanics of spin glasses, structural glasses, and other systems with rugged free-energy landscapes. In these contexts, it is well established that the total entropy can be decomposed into a vibrational (intra-basin) contribution and a configurational (inter-basin) contribution, with the latter governing phenomena such as ergodicity breaking, aging, and glass transitions [
7,
8].
Figure 1.
Illustrative free-energy landscape with multiple metastable states. Configurational complexity quantifies the logarithmic multiplicity of such basins rather than microscopic fluctuations within a single basin. Regions with a larger number of distinct minima correspond to higher configurational complexity.
Figure 1.
Illustrative free-energy landscape with multiple metastable states. Configurational complexity quantifies the logarithmic multiplicity of such basins rather than microscopic fluctuations within a single basin. Regions with a larger number of distinct minima correspond to higher configurational complexity.
2.2. Thermodynamic Properties of Configurational Complexity
The complexity
C defined in Equation (
1) possesses several properties that make it suitable as a thermodynamic macrovariable:
Extensivity: The total complexity scales linearly with system size for weakly interacting subsystems.
Additivity: For statistically independent subsystems, complexities are additive to leading order.
Physical Interpretability: C directly reflects the structure of the free-energy landscape and the number of metastable macrostates accessible to the system.
Because C depends on global properties of the energy landscape rather than on instantaneous microscopic fluctuations, it should be regarded as a macroscopic state variable, analogous in spirit to order parameters used in the thermodynamic description of phase transitions. While the counting of metastable states ultimately involves microscopic information, the resulting complexity is a property of macrostates, not individual microstates.
2.3. Distinction from Other Notions of Complexity
It is crucial to emphasize what the complexity variable C represents—and what it does not represent—in the present framework.
First, C is not algorithmic or computational complexity in the sense of computational complexity theory, such as time complexity, circuit depth, or complexity classes. While such notions are important in computer science and the theory of computation, they are not directly suitable as thermodynamic state variables, as they depend on external problem encodings and computational models rather than intrinsic physical properties.
Second, C is not Kolmogorov complexity or algorithmic information content. Those measures depend on description length relative to a universal Turing machine and are inherently non-thermodynamic.
Finally, C should not be conflated with the Shannon or Gibbs entropy of microscopic configurations. Instead, configurational complexity complements the usual entropy by quantifying the multiplicity of metastable macroscopic arrangements available to the system.
By committing to configurational complexity as defined above, we ensure that the extended thermodynamic framework developed in the following sections rests on a well-established and physically meaningful foundation.
3. Thermodynamics with a Complexity Constraint
3.1. Maximum Entropy with Multiple Macroscopic Constraints
The formal foundation of equilibrium thermodynamics lies in the principle of maximum entropy. Given a set of macroscopic constraints, the equilibrium ensemble is obtained by maximizing the Gibbs entropy subject to those constraints. This approach, originally articulated by Jaynes, provides a systematic and general route to constructing equilibrium statistical ensembles [
9]. In the present context, we consider systems for which, in addition to the mean energy, the configurational complexity introduced in
Section 2 constitutes a relevant macroscopic observable. We therefore seek the equilibrium distribution that maximizes the entropy
subject to the constraints
where
labels macroscopic configurations or metastable states,
is their energy, and
their configurational complexity.
Introducing Lagrange multipliers and to enforce the energy and complexity constraints, respectively, yields an equilibrium distribution of the generalized Gibbs form. The multiplier plays a role entirely analogous to other thermodynamic conjugate variables, such as chemical potentials or magnetic fields, and arises naturally from the constrained entropy maximization procedure.
3.2. Extended First Law as a Constrained Relation
Within this constrained framework, thermodynamic relations follow from standard Legendre-transform structure rather than from postulated modifications of fundamental laws. The internal energy becomes a function of entropy, volume, and complexity,
and its total differential takes the form
Here, the temperature
T and pressure
P retain their usual definitions, while
represents the conjugate variable associated with configurational complexity. Importantly, Equation (
7) should be interpreted as a constrained equilibrium relation rather than as a fundamental extension of the first law of thermodynamics. Its validity is limited to systems for which configurational complexity constitutes a well-defined and controllable macroscopic variable. The appearance of
reflects the energetic cost or benefit associated with changes in the multiplicity of metastable configurations. In physical terms, it quantifies how the internal energy responds to variations in the structure of the energy landscape at fixed entropy and volume.
3.3. Thermodynamic Potentials and Integrability
The presence of an additional macroscopic coordinate does not alter the formal structure of thermodynamics. Standard thermodynamic potentials can be defined via Legendre transformations, now extended to include complexity. For example, the generalized Helmholtz free energy is given by
Provided that the underlying entropy function is smooth and the equilibrium state is stable, the usual integrability conditions apply. In particular, mixed second derivatives of thermodynamic potentials commute, leading to generalized Maxwell relations involving complexity and its conjugate variable. These relations are a direct mathematical consequence of the assumed differentiability of the thermodynamic potentials and do not rely on any additional physical assumptions. It is important to emphasize that such relations are not presented here as novel thermodynamic laws. Rather, they serve as consistency conditions within the constrained ensemble framework. Their physical content becomes meaningful only when evaluated in concrete models, as demonstrated in
Section 5.
3.4. Physical Interpretation and Domain of Validity
The complexity-constrained thermodynamic description developed above is applicable under specific conditions. First, the system must admit a clear separation between microscopic fluctuations within metastable states and macroscopic transitions between distinct metastable basins. Second, the relevant timescales must permit equilibration within basins, even if transitions between basins are slow or suppressed. Under these conditions, configurational complexity acts as a collective coordinate characterizing the global organization of the energy landscape. The conjugate variable can then be interpreted as an external control parameter that biases the system toward regions of configuration space with higher or lower metastable multiplicity. Outside this regime—particularly in strongly driven or far-from-equilibrium systems—the present framework is not expected to apply directly. Extensions to non-equilibrium settings would require additional dynamical considerations and are left for future work.
4. Complexity-Biased Equilibrium Ensemble
4.1. Derivation of the Ensemble
The thermodynamic relations derived in
Section 3 acquire concrete meaning once an explicit equilibrium ensemble is constructed. As shown there, the appropriate ensemble follows directly from the principle of maximum entropy subject to simultaneous constraints on the mean energy and the mean configurational complexity.
Carrying out the constrained entropy maximization described in
Section 3 yields an equilibrium probability distribution of the form
where
and
are the Lagrange multipliers enforcing the energy and complexity constraints, respectively. The normalization factor
defines the partition function of the complexity-biased equilibrium ensemble.
Equations (
9) and (
10) are a straightforward generalization of the canonical ensemble. No modification of the underlying principles of equilibrium statistical mechanics is required. The additional exponential weight reflects the presence of an explicit macroscopic constraint on configurational complexity.
4.2. Interpretation of the Partition Function
The partition function
introduced in Equation (
10) should be understood as the normalization constant of a generalized Gibbs ensemble. It encodes the statistical properties of a system whose equilibrium states are biased according to their configurational complexity.
Importantly, the presence of the complexity-dependent term does not imply that the system is coupled to a physical “complexity reservoir.” Rather,
is a control parameter arising from constrained entropy maximization, analogous to chemical potentials or external fields. Fixed
corresponds to studying the response of the system under a prescribed bias toward configurations with higher or lower metastable multiplicity, while fixed
corresponds to selecting ensembles with a prescribed average complexity. Such biased ensembles are well known in statistical physics and appear naturally in the study of conditioned equilibrium states and large deviation phenomena. In this sense, the complexity-biased ensemble belongs to a broader class of tilted or constrained ensembles that remain fully consistent with equilibrium statistical mechanics [
10,
11].
4.3. Thermodynamic Observables and Response Functions
Once the partition function
is specified, thermodynamic observables follow by standard differentiation. In particular, the average energy and average complexity are given by
Fluctuations and response functions are obtained from second derivatives. Of particular interest is the complexity susceptibility,
which quantifies the sensitivity of the system’s configurational structure to changes in the complexity bias. Peaks or divergences in
signal regimes in which small changes in
produce large reorganizations of the energy landscape.
Cross-response functions, such as , encode couplings between energetic and structural degrees of freedom and provide direct insight into how complexity constraints modify equilibrium thermodynamics.
4.4. Physical Realization and Limitations
The complexity-biased ensemble should be regarded as an effective equilibrium description applicable when configurational complexity can be controlled or selected indirectly. In numerical simulations, such ensembles can be realized via reweighting techniques, umbrella sampling, or explicit biasing of metastable states. In experimental systems, approximate realizations may arise through controlled preparation protocols or slow annealing procedures that favor specific regions of the energy landscape. As with any constrained ensemble, the present framework assumes that the system equilibrates under the imposed constraints. In situations where transitions between metastable states are dynamically suppressed on experimental timescales, the ensemble description may break down. These limitations are intrinsic to equilibrium statistical mechanics and do not represent a special feature of the complexity bias.
5. Worked Example: Mean-Field Glassy Landscape
To demonstrate that the complexity-constrained thermodynamic framework developed above yields concrete and nontrivial results, we now apply it to a schematic glassy system in which configurational complexity can be computed explicitly. The purpose of this section is not to exhaustively analyze a particular model, but rather to show how complexity enters equilibrium thermodynamics in a controlled and calculable way.
5.1. Model Definition
We consider a mean-field system characterized by a rugged free-energy landscape containing an exponential number of metastable states. Such models arise naturally in the statistical mechanics of spin glasses and structural glasses and admit a well-defined configurational complexity. Let
label metastable states with free-energy density
. The number of metastable states with free-energy density
f scales as
where
is the configurational complexity density. For concreteness, we assume that
is a smooth, concave function defined over a finite interval
, as commonly found in mean-field glass models [
8,
12].
In this setting, the complexity associated with a metastable state is given by
in accordance with the definition introduced in
Section 2.
5.2. Complexity-Biased Partition Function
Using the complexity-biased ensemble constructed in
Section 4, the partition function can be written as a sum over metastable states,
In the thermodynamic limit
, this sum is dominated by a saddle point and can be expressed as an integral over free-energy densities,
The exponent defines an effective large-deviation function,
whose maximum determines the dominant contribution to the partition function.
5.3. Saddle-Point Structure and Equilibrium Complexity
The equilibrium free-energy density
is obtained by solving the saddle-point condition
Equation (
19) shows explicitly how the complexity bias
shifts the equilibrium selection of metastable states. For
, the usual equilibrium condition of the unconstrained ensemble is recovered. For
, the system is biased toward regions of the energy landscape with higher or lower configurational multiplicity.
The equilibrium configurational complexity is then given by
and depends parametrically on both temperature and complexity bias.
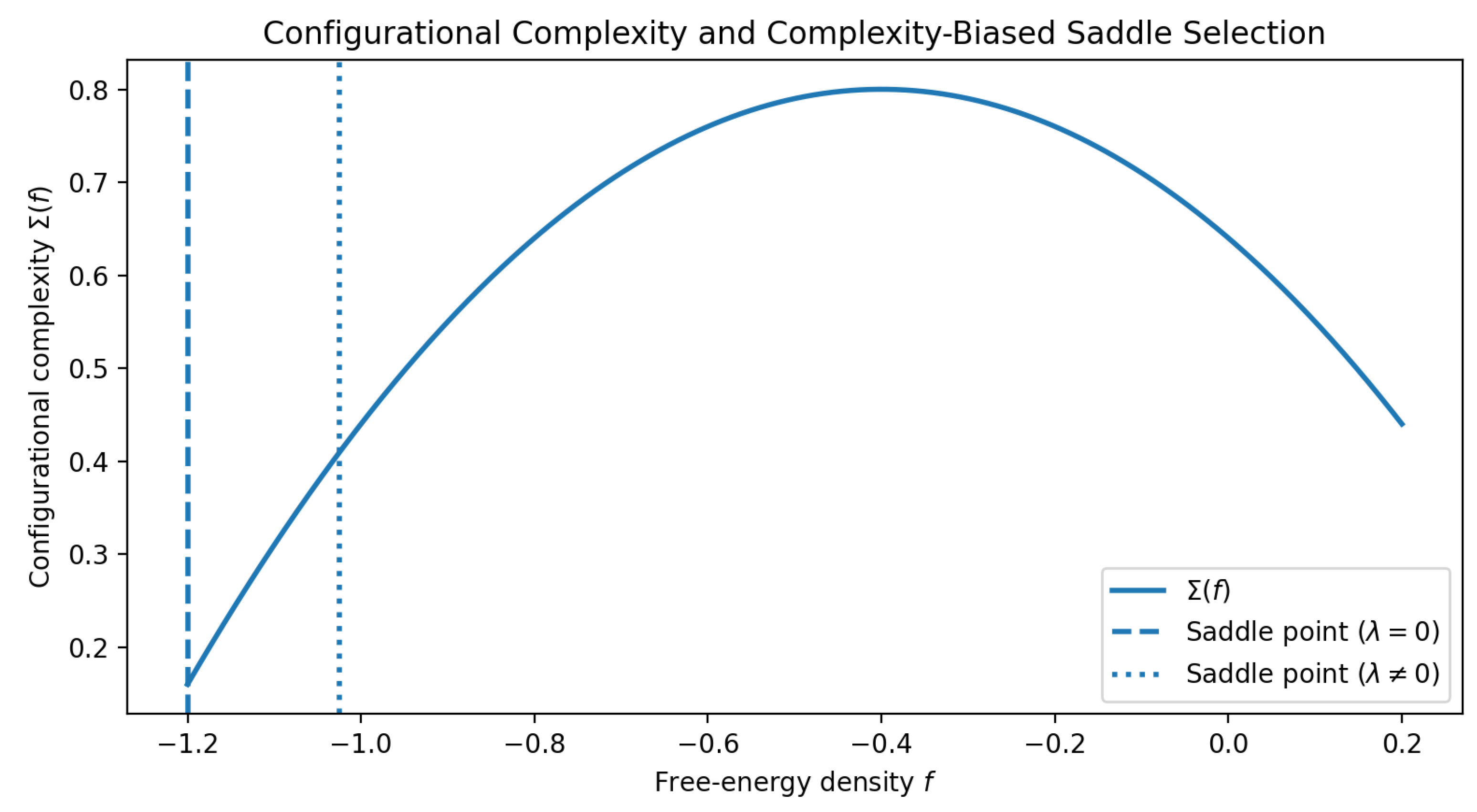
Figure 2.
Illustrative configurational complexity function and complexity-biased saddle-point selection. The unbiased equilibrium saddle point () and a biased saddle point () correspond to different dominant regions of the energy landscape, demonstrating how complexity bias modifies equilibrium selection.
Figure 2.
Illustrative configurational complexity function and complexity-biased saddle-point selection. The unbiased equilibrium saddle point () and a biased saddle point () correspond to different dominant regions of the energy landscape, demonstrating how complexity bias modifies equilibrium selection.
5.4. Response Functions and Structural Transitions
Differentiating the saddle-point condition with respect to
yields the complexity susceptibility,
provided that
is twice differentiable.
Divergences or sharp peaks in occur when the curvature of the complexity function approaches zero, signaling structural instabilities in the energy landscape. Such behavior is well known in mean-field glass models and is associated with transitions between regimes dominated by few deep minima and those dominated by exponentially many shallow states. This explicit calculation illustrates that the complexity-constrained thermodynamic framework leads to concrete, model-dependent predictions that go beyond formal consistency relations.
5.5. Discussion
The present example demonstrates how configurational complexity enters equilibrium thermodynamics as a genuine macroscopic coordinate. The complexity bias modifies the saddle-point structure of the partition function, leading to measurable changes in equilibrium properties and response functions. While the specific form of is model-dependent, the structure of the analysis is general and can be applied to a wide class of systems with rugged energy landscapes. This highlights the usefulness of treating configurational complexity as a thermodynamic variable within a constrained ensemble framework.
6. Thermodynamic Geometry with a Complexity Coordinate
Thermodynamic geometry provides a powerful framework for analyzing stability and structural changes in equilibrium systems by endowing the space of thermodynamic states with a metric structure. In this section, we extend this approach to include configurational complexity as an additional thermodynamic coordinate and evaluate its implications within the complexity-constrained framework developed above.
6.1. Extended Thermodynamic State Space
In conventional thermodynamic geometry, the space of equilibrium states is parametrized by extensive variables such as entropy and volume, or equivalently by their conjugate intensive variables. In the present framework, the thermodynamic state space is augmented by the configurational complexity
C, yielding an extended set of coordinates
or, in the dual representation,
where
is the complexity-conjugate variable introduced in
Section 3.
The presence of the additional coordinate enlarges the thermodynamic manifold, allowing geometric diagnostics to probe structural properties of the energy landscape that are inaccessible in the standard formulation.
6.2. Metric Definition
Following the Ruppeiner formalism, the thermodynamic metric is defined as the Hessian of the entropy with respect to the extensive variables,
Equivalently, in the energy representation, one may use the Weinhold metric defined by the Hessian of the internal energy with respect to extensive variables. In either representation, the inclusion of complexity introduces additional metric components involving second derivatives with respect to
C. These components encode correlations between entropy fluctuations, energetic fluctuations, and structural fluctuations associated with changes in the number of metastable states.
6.3. Curvature as a Diagnostic of Structural Transitions
The scalar curvature
R associated with the thermodynamic metric serves as a coordinate-invariant diagnostic of interaction strength and instability in the underlying system. In numerous physical systems, divergences or sharp features in
R have been shown to coincide with phase transitions or critical points. Within the complexity-extended framework, curvature contributions involving derivatives with respect to
C probe the sensitivity of the system to changes in landscape multiplicity. In particular, near regimes where the configurational complexity function
develops vanishing curvature, the corresponding thermodynamic curvature is expected to exhibit pronounced features. This behavior can be seen explicitly in the worked example of
Section 5. There, the complexity susceptibility diverges when the second derivative of
approaches zero. Since the thermodynamic metric components include second derivatives of the entropy and free energy with respect to
C, such divergences naturally propagate into the curvature scalar. The resulting geometric singularities thus provide a complementary characterization of complexity-driven structural transitions.
6.4. Interpretation and Limitations
The geometric perspective reinforces the interpretation of configurational complexity as a genuine thermodynamic coordinate. Curvature anomalies in the extended state space signal reorganizations of the energy landscape rather than conventional energetic phase transitions. This distinction is particularly relevant in glassy systems, where dramatic changes in dynamics and structure occur without sharp discontinuities in standard thermodynamic observables. At the same time, it is important to emphasize that thermodynamic geometry does not introduce new physics by itself. Its utility lies in providing a compact and coordinate-invariant summary of response functions and stability properties derived from the underlying thermodynamic potentials. Consequently, the geometric analysis presented here should be viewed as a diagnostic tool rather than as an independent theoretical framework.
7. Relation to Computation and Landauer’s Principle
The introduction of configurational complexity as a thermodynamic coordinate naturally invites comparison with concepts from the thermodynamics of computation. In this section, we clarify the relationship between the present equilibrium framework and established results such as Landauer’s principle, and we delineate the conceptual boundaries between landscape complexity and algorithmic notions of computational cost.
7.1. Landauer’s Principle and Its Domain of Validity
Landauer’s principle establishes a lower bound on the work dissipated during logically irreversible operations, stating that the erasure of one bit of information incurs a minimum energetic cost of
[
1]. This bound arises fundamentally from entropy reduction and applies to processes that are intrinsically non-equilibrium in nature. Crucially, Landauer’s principle does not depend on the complexity of a computation or the structure of the system’s state space. Instead, it reflects the thermodynamic cost of logical irreversibility, independent of how a given logical operation is implemented physically. As such, Landauer’s bound is universal within its domain of applicability. The framework developed in this work addresses a different aspect of physical systems. It operates strictly within equilibrium statistical mechanics and concerns the thermodynamic consequences of constraining or biasing the multiplicity of metastable macrostates. No universal lower bounds analogous to Landauer’s limit are implied or claimed here.
7.2. Landscape Complexity Versus Algorithmic Complexity
Configurational complexity, as defined in
Section 2, is a property of the physical energy landscape of a system. It quantifies the number of long-lived macroscopic configurations compatible with given thermodynamic constraints. This notion is intrinsic to the physical system and independent of any external computational task. By contrast, algorithmic or computational complexity refers to the resources required to solve a problem within a specified computational model, typically measured in terms of time, memory, or circuit depth. These quantities depend on problem encoding, algorithm choice, and abstract computational assumptions, and they do not constitute thermodynamic state variables. While both notions involve the term “complexity,” they operate at fundamentally different conceptual levels. The present framework deliberately restricts itself to landscape complexity, which admits a clear thermodynamic interpretation. Any connection between landscape complexity and algorithmic hardness is necessarily indirect and model-dependent.
7.3. Complexity Constraints and Energetic Overheads
Although no universal energetic bound associated with complexity is proposed, the constrained ensemble framework developed here provides a systematic way to analyze how structural constraints on configuration space influence equilibrium energetics. In particular, biasing a system toward regions of higher or lower configurational complexity modifies its equilibrium free energy and response functions, as demonstrated explicitly in
Section 5. In physical implementations of computation or optimization, such structural constraints may manifest as additional energetic overheads beyond the minimal Landauer cost. These overheads arise not from logical irreversibility, but from the need to control or select among exponentially many metastable configurations. Their magnitude is inherently system-specific and depends on the details of the energy landscape and the preparation protocol. From this perspective, configurational complexity provides a useful language for discussing why certain physical computations or optimization processes are energetically costly, even when implemented reversibly in principle. The framework presented here offers a route to quantifying such effects without conflating them with fundamental limits on computation.
7.4. Outlook on Computation-Inspired Extensions
While the present work focuses exclusively on equilibrium thermodynamics, it opens the door to future investigations connecting landscape complexity with computational processes in more dynamical settings. Extensions to non-equilibrium thermodynamics, stochastic thermodynamics, or driven systems may allow more direct links between configurational complexity and computational performance to be explored. Such developments would require explicit modeling of dynamics, protocols, and time-dependent constraints, and therefore lie beyond the scope of the current study. The results presented here should be viewed as a foundational equilibrium framework upon which such extensions may be built.
8. Discussion and Outlook
8.1. Summary of Results
In this work, we have developed a constrained equilibrium thermodynamic framework in which configurational complexity is treated as an explicit macroscopic coordinate. By defining complexity in terms of the logarithmic density of metastable states, we ensured that the additional variable admits a clear physical interpretation rooted in the statistical mechanics of disordered systems. Starting from the principle of maximum entropy, we constructed a complexity-biased equilibrium ensemble characterized by a conjugate control parameter . Within this framework, standard thermodynamic relations follow without modification of the underlying formalism, and the extended first-law-like relation emerges naturally as a constrained equilibrium identity. The utility of the framework was demonstrated through a worked example based on a mean-field glassy landscape. In this setting, complexity bias modifies the saddle-point structure of the partition function, leading to nontrivial responses in equilibrium observables and well-defined susceptibility signatures. The associated thermodynamic geometry further highlighted how structural transitions in the energy landscape manifest as geometric features in the extended state space.
Taken together, these results show that configurational complexity can be incorporated into equilibrium thermodynamics in a precise, operational, and model-dependent manner.
8.2. Conceptual Implications
The framework developed here clarifies the role of complexity in thermodynamics by distinguishing between landscape-level structural properties and algorithmic or computational notions of complexity. Treating configurational complexity as a thermodynamic coordinate does not imply a universal energetic cost of complexity, nor does it introduce new fundamental laws. Instead, it provides a systematic language for analyzing how the organization of configuration space influences equilibrium behavior. From this perspective, complexity emerges as a collective descriptor of macroscopic structure, analogous to order parameters used in the study of phase transitions. Its conjugate variable quantifies the sensitivity of the system’s energetics to changes in the multiplicity of metastable states, offering a controlled way to probe structural reorganizations that may not be visible through conventional thermodynamic observables alone.
8.3. Limitations
Several limitations of the present approach should be emphasized. First, the framework is restricted to equilibrium statistical mechanics and assumes that relevant metastable states can be meaningfully enumerated or characterized. Systems far from equilibrium, or those dominated by strong dynamical constraints, fall outside the scope of the present analysis.
Second, the quantitative predictions obtained within the complexity-constrained ensemble are inherently model-dependent. The specific form of the configurational complexity function plays a central role, and different physical systems may exhibit qualitatively different behavior. As such, the framework should be viewed as a tool for structured analysis rather than as a universal theory.
Finally, the treatment of complexity as a macroscopic coordinate relies on a separation of timescales between intra-state equilibration and inter-state transitions. When this separation breaks down, the applicability of the equilibrium ensemble description becomes limited.
8.4. Future Directions
Despite these limitations, the present framework opens several promising avenues for future research. Extensions to non-equilibrium settings, such as driven or slowly annealed systems, may allow the role of configurational complexity to be explored dynamically. Incorporating explicit time dependence or stochastic dynamics could provide further insight into how landscape structure influences relaxation and dissipation. Another natural direction is the application of the complexity-constrained ensemble to specific physical systems, including structural glasses, optimization-inspired physical models, and interacting networks with rugged energy landscapes. In such contexts, empirical or numerical estimates of configurational complexity could be combined with the present formalism to yield testable predictions.
Finally, while the present work deliberately avoids conflating landscape complexity with algorithmic complexity, future studies may explore controlled links between the two in well-defined models. Establishing such connections would require careful attention to dynamics, encoding, and physical implementation, and remains an open challenge.
9. Conclusions
In this paper, we have developed a constrained equilibrium thermodynamic framework in which configurational complexity is treated as an explicit macroscopic coordinate. By grounding the definition of complexity in the well-established statistical mechanics of disordered systems, we ensured that the extended description remains physically meaningful and operationally well defined. Starting from the principle of maximum entropy, we showed that imposing simultaneous constraints on energy and configurational complexity leads naturally to a generalized equilibrium ensemble characterized by a conjugate complexity control parameter. Within this setting, standard thermodynamic relations and integrability conditions remain intact, and the resulting extended relations should be understood as constrained equilibrium identities rather than as modifications of fundamental laws. The framework was illustrated through a worked example based on a mean-field glassy landscape, where configurational complexity can be computed explicitly. In this example, complexity bias alters the equilibrium saddle-point structure, gives rise to nontrivial response functions, and produces clear signatures of structural transitions. An extension to thermodynamic geometry further demonstrated how landscape-level reorganizations manifest as geometric features in an enlarged thermodynamic state space. Importantly, the present work does not propose a universal energetic cost of complexity, nor does it seek to replace established results in the thermodynamics of computation, such as Landauer’s principle. Instead, it provides a systematic equilibrium language for analyzing how the organization of configuration space influences thermodynamic behavior in systems with rugged energy landscapes.
We expect that this complexity-constrained thermodynamic perspective will prove useful in the study of disordered materials, optimization-inspired physical models, and other complex systems where landscape structure plays a dominant role. By making complexity an explicit and controllable element of equilibrium thermodynamics, the framework developed here offers a foundation for future investigations into both equilibrium and non-equilibrium manifestations of structural complexity in physical systems.
Author Contributions
Conceptualization, F.N.; methodology, F.N.; formal analysis, F.N.; investigation, F.N.; writing—original draft preparation, F.N.; writing—review and editing, F.N.; visualization, F.N.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study.
Acknowledgments
Not applicable.
Conflicts of Interest
The author declares no conflicts of interest.
Appendix A. Derivation of the Complexity-Constrained Ensemble
In this appendix, we provide a detailed derivation of the complexity-biased equilibrium ensemble used throughout the main text. The construction follows directly from the principle of maximum entropy with multiple macroscopic constraints.
We consider a set of macroscopic configurations or metastable states indexed by
, each characterized by an energy
and a configurational complexity
. The Gibbs entropy is defined as
The equilibrium distribution is obtained by maximizing
S subject to the normalization constraint
as well as constraints on the mean energy and mean complexity,
Introducing Lagrange multipliers
,
, and
to enforce these constraints yields the variational functional
Setting the functional derivative with respect to
to zero gives
which leads directly to the complexity-biased distribution and partition function presented in
Section 4.
—
Appendix B. Thermodynamic Properties of Configurational Complexity
Configurational complexity, defined as the logarithmic density of metastable states, possesses several properties that justify its treatment as a thermodynamic macrovariable.
Appendix B.1. Extensivity and Additivity
For systems composed of weakly interacting subsystems
A and
B, the total number of metastable states approximately factorizes,
implying additivity of the total complexity,
This property ensures that configurational complexity scales linearly with system size in the thermodynamic limit.
Appendix B.2. Relation to Entropy
Configurational complexity should be distinguished from the standard thermodynamic entropy. While entropy counts microscopic configurations within a given metastable basin, complexity counts the number of such basins themselves. In glassy systems, the total entropy can often be decomposed into a vibrational and a configurational contribution, but only the latter is considered here.
—
Appendix C. Alternative Notions of Complexity and Rationale for Exclusion
The term “complexity” is used in many contexts across physics, information theory, and computer science. In this appendix, we briefly summarize alternative notions and clarify why they are not adopted in the present framework.
Appendix C.1. Algorithmic and Computational Complexity
Algorithmic complexity measures, such as time complexity, circuit depth, or complexity classes, depend on problem encoding, computational models, and algorithm choice. These quantities are not intrinsic properties of a physical system and therefore cannot serve as thermodynamic state variables.
Appendix C.2. Kolmogorov Complexity
Kolmogorov complexity quantifies the length of the shortest description of an object relative to a universal Turing machine. While conceptually powerful, it is non-computable in general and lacks a direct thermodynamic interpretation.
Appendix C.3. Information-Theoretic Entropies
Shannon and Gibbs entropies quantify uncertainty over microstates or symbols. While related to thermodynamics, they do not capture the multiplicity of metastable macrostates and therefore address a different level of description than configurational complexity.
—
Appendix D. Additional Analysis of the Mean-Field Example
In this appendix, we provide supplementary analysis related to the worked example in
Section 5.
Appendix D.1. Stability Conditions
The saddle-point solution obtained from Equation (
28) in the main text is stable provided that
which corresponds to concavity of the effective large-deviation function. This condition is violated near points where the curvature of the configurational complexity function
vanishes, signaling structural transitions.
Appendix D.2. Finite-Size Considerations
For finite systems, the sharp saddle-point selection is replaced by a smooth crossover. Fluctuations in both energy and complexity scale as , and susceptibility peaks are rounded. These effects do not alter the qualitative conclusions of the thermodynamic-limit analysis but are relevant for numerical simulations.
—
Appendix E. Relation to Large Deviation Theory
The complexity-biased ensemble introduced in this work is formally equivalent to a tilted ensemble in large deviation theory. In this language, configurational complexity acts as an extensive observable whose fluctuations are exponentially suppressed in the system size.
The scaled cumulant generating function associated with complexity,
encodes all equilibrium cumulants of
C. Non-analytic behavior of
corresponds to dynamical or structural phase transitions in the large deviation sense.
This connection provides an alternative mathematical perspective on the constrained ensemble framework and further supports its consistency within equilibrium statistical mechanics.
References
- Landauer, Rolf. Irreversibility and heat generation in the computing process. IBM Journal of Research and Development 1961, 5, 183–191. [Google Scholar] [CrossRef]
- Bennett, Charles H. The thermodynamics of computation—a review. International Journal of Theoretical Physics 1982, 21, 905–940. [Google Scholar] [CrossRef]
- Kirkpatrick, T. R.; Thirumalai, D. Dynamics of the structural glass transition and the p-spin–interaction spin-glass model. Physical Review Letters 1987, 58, 2091–2094. [Google Scholar] [CrossRef] [PubMed]
- Berthier, Ludovic; Biroli, Giulio. Theoretical perspective on the glass transition and amorphous materials. Reviews of Modern Physics 2011, 83, 587–645. [Google Scholar] [CrossRef]
- Touchette, Hugo. The large deviation approach to statistical mechanics. Physics Reports 2009, 478, 1–69. [Google Scholar] [CrossRef]
- Ruppeiner, George. Riemannian geometry in thermodynamic fluctuation theory. Reviews of Modern Physics 1995, 67, 605–659. [Google Scholar] [CrossRef]
- Mézard, Marc; Parisi, Giorgio; Virasoro, Miguel. Spin Glass Theory and Beyond; World Scientific: Singapore, 1987. [Google Scholar]
- Monasson, Rémi. Structural glass transition and the entropy of the metastable states. Physical Review Letters 1995, 75, 2847–2850. [Google Scholar] [CrossRef] [PubMed]
- Jaynes, Edwin T. Information theory and statistical mechanics. Physical Review 1957, 106, 620–630. [Google Scholar] [CrossRef]
- Evans, Denis J.; Morriss, Gary. Statistical mechanics of nonequilibrium liquids. Cambridge University Press, 2008. [Google Scholar]
- Garrahan, Juan P.; Lesanovsky, Igor. Thermodynamics of quantum jump trajectories. Physical Review Letters 2010, 104, 160601. [Google Scholar] [CrossRef] [PubMed]
- Cavagna, Andrea. Supercooled liquids for pedestrians. Physics Reports 2009, 476, 51–124. [Google Scholar] [CrossRef]
- Weinhold, Frank. Metric geometry of equilibrium thermodynamics. The Journal of Chemical Physics 1975, 63, 2479–2483. [Google Scholar] [CrossRef]
- Janyszek, Henryk; Mrugała, Ryszard. Riemannian geometry and stability of ideal quantum gases. Journal of Physics A: Mathematical and General 1989, 22, 1–14. [Google Scholar] [CrossRef]
- Bennett, Charles H. Notes on Landauer’s principle, reversible computation, and Maxwell’s Demon. Studies in History and Philosophy of Modern Physics 2003, 34, 501–510. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).