1. Introduction
Systemic candidiasis continues to present substantial clinical challenges, with
Candida albicans (
C. albicans) remaining the leading causative agent and representing the majority of isolates yielded in surveillance studies [
1]. Once bloodstream dissemination occurs, outcomes among affected patients are often poor, with frequent mortality rates exceeding 40% [
2,
3,
4]. The expanded clinical use of azole antifungals, particularly fluconazole (FLZ), has further intensified concerns, as selective pressure has played a critical role in the emergence of drug-tolerant or resistant
C. albicans lineages [
5]. Despite increasing medical demand, few new antifungal classes have been introduced to the therapeutic landscape, highlighting a critical need for alternative agents and adjuvant strategies [
6,
7,
8].
Biofilm formation plays a central role in the pathogenicity of
C. albicans. Compared with planktonic cells, biofilm-associated cells exhibit reduced susceptibility to antifungal drugs, increased tolerance to oxidative and chemical stresses, and enhanced evasion of host immune defenses [
9]. Biofilm formation on mucosal surfaces such as the oral cavity or vagina contributes to recurrent infections and limited responses to standard therapy. Notably, biofilms show reduced sensitivity to drugs including amphotericin B and FLZ, providing a mechanistic basis for treatment failure in mucosal candidiasis [
10,
11].
Given the slow pace of antifungal drug discovery, exemplified by the approval of only isavuconazole in the past decade [
12], research efforts have increasingly shifted toward strategies that leverage the existing compounds. Combination therapy, widely adopted in oncology and antiviral research [
13,
14,
15], has similarly demonstrated potential in fungal infections. Several pharmacologic agents already in clinical use enhance the fungicidal activity of azoles and polyenes, including activity against resistant
C. albicans isolates [
16,
17,
18].
Natural compounds have emerged as promising antifungal adjuncts. Bioactive phytochemicals, including terpenes, phenolic compounds, alkaloids, and essential oils, have demonstrated the ability to impair
C. albicans biofilm development or disrupt mature biofilms, often through modulation of biofilm-associated gene expression [
19,
20,
21]. Some compounds, such as eugenol, cinnamaldehyde, thymol, and menthol, enhance FLZ sensitivity by altering membrane permeability and disrupting sterol biosynthesis [
22,
23,
24]. Likewise, berberine increases FLZ susceptibility by perturbing sterol biosynthesis and intensifying oxidative stress [
24,
25,
26,
27,
28].
Hinokitiol (HNK), a naturally occurring tropolone derivative first isolated in Japan in the early 20th century, has garnered considerable attention for its broad-spectrum antimicrobial activity against bacterial and fungal pathogens [
29,
30]. Owing to these properties, HNK has been incorporated into various quasi-drug formulations, including toothpastes, mouthwashes, and hair-care products. Beyond antimicrobial functions, accumulating evidence—including our previous findings—indicates that HNK induces apoptosis and suppresses proliferation of human oral squamous cell carcinoma cells [
31,
32,
33], highlighting potential relevance as a therapeutic agent in oral oncology. Additionally, experimental models of oral candidiasis suggest that loss of topical gel carriers may facilitate fungal adhesion to oral mucosa, implying that HNK itself warrants examination as a direct antifungal agent rather than solely as a formulation additive.
Candida-associated oral infections frequently occur among patients with systemic disease, denture use, salivary dysfunction, malignancy, or immunosuppression, and progression to invasive disease might occur when adequate control is not achieved [
34,
35,
36,
37,
38]. Recent evidence has also suggested a potential link between fungal invasion of the central nervous system and neurodegenerative pathology, including Alzheimer's disease [
39]. Given these concerns, identifying agents capable of suppressing early mucosal colonization might hold clinical significance.
On the basis of these considerations, direct antifungal activity of HNK and potential enhancement of standard antifungal efficacy were postulated. In this study, we aimed to evaluate the antifungal effects of HNK against C. albicans and examine potential interactions with FLZ, a first-line topical therapy for oral candidiasis.
2. Materials and Methods
2.1. Fungal Strain and Culture Conditions
In this study, the C. albicans ATCC 10261 strain was used and maintained in the laboratory. The strain was stored at −80°C in Bacto Peptone (Beckton, Dickinson and Company, Franklin Lakes, NJ, USA) containing 4% D-glucose (Sabouraud's medium; SM) and 10% glycerol until experiments were performed. C. albicans was cultured on Candida GS agar plates (Eiken Chemical Co., Ltd., Tokyo, Japan) at 37°C for 24 h. After incubation, the cells were collected and resuspended in Roswell Park Memorial Institute 1640 (RPMI 1640) medium containing 2.5% fetal bovine serum (FBS) for subsequent in vitro testing. All cultures were incubated at 37°C under aerobic conditions.
2.2. Antifungal Substances
FLZ, a first-generation triazole antifungal drug, was purchased from Meiji Seika Pharma Co., Ltd. (Tokyo, Japan) and dissolved in sterilized water at a concentration of 1.0 mg/mL for use in this study.
HNK (Tokyo Chemical Industry Co., Ltd., Tokyo, Japan) was prepared as a 100 mg/mL stock solution by dissolving it in dimethyl sulfoxide (DMSO; Kishida Chemical Co., Ltd., Osaka, Japan). The final HNK concentration in the culture medium, with a DMSO concentration of 0.1%, was achieved by adding the stock solution to the working medium at a 1:1000 ratio.
2.3. Determination of Minimum Inhibitory Concentration
A preliminary assay was conducted to determine the minimum inhibitory concentration (MIC) required for suppressing 50% of C. albicans growth (MIC₅₀) for HNK. Using the standard micro-broth dilution technique, six independent measurements yielded a consistent dose–response curve, with an MIC₅₀ of 6.3 µg/mL. To assess the potential synergistic antifungal interaction between HNK and FLZ on C. albicans, particularly regarding the yeast-to-hypha transition (dimorphism), HNK concentrations that did not independently inhibit fungal proliferation were selected. Employing HNK at or above its MIC₅₀ could mask the intrinsic fungicidal activity of FLZ; thereby, confounding assessment of combined effects. Clinical considerations also guided this selection. In the oral cavity, drug concentrations exceeding the MIC₅₀ are rapidly diluted by saliva and dispersed across mucosal surfaces. Consequently, evaluating the activity of sub-MIC₅₀ HNK levels provides a more realistic approximation of in vivo exposure. Subsequent experiments generated dose–response curves for FLZ administered alone and in combination with 3.2 µg/mL HNK. Triplicate measurements revealed clear synergism across FLZ concentrations ranging between 0.8 and 3.2 µg/mL. Based on these findings, all subsequent assays employed HNK at 3.2 µg/mL together with FLZ at 0.5 µg/mL.
2.4. Inhibitory Effect of HNK-Containing FLZ on C. albicans Biofilm Formation
In the study, we aimed to examine the combined pharmacological effects of HNK and FLZ on inhibiting
C. albicans biofilm formation. Crystal violet staining, frequently employed to assess yeast biofilm biomass, provides high sensitivity and accuracy in detecting substantial biofilm formation [
40]. Biofilm biomass, including both viable and nonviable cells, was visualized under crystal violet staining. The impact of combined HNK and FLZ treatment on
C. albicans biofilm development was assessed by a protocol adapted from the method described by Manoharan et al [
41]. Suspensions of
C. albicans were adjusted to a final density of 1 x 10
6 CFU/mL in RPMI 1640 medium. Aliquots of 200 μL were then dispensed into flat-bottom 96-well microplates and cultured at 37 °C for 24 h. Following incubation, nonadherent cells were eliminated by rinsing each well three times with sterile phosphate-buffered saline (PBS). Next, 200 μL of RPMI 1640 medium containing the test compounds at the indicated concentrations was added to each well and incubated at 37 °C for 24 h to evaluate drug-induced changes in biofilm mass. Biofilm biomass was quantified with crystal violet staining at a final concentration of 0.05%. Briefly, wells were rinsed three times with sterile PBS to eliminate nonadherent cells, stained with crystal violet for 15 min, and washed an additional three times with PBS. After the plates were allowed to air-dry, 120 μL of 95% ethanol was added to each well to dissolve the bound dye. Subsequently, 100 μL of the extracted solution was transferred to a fresh flat-bottom 96-well plate; absorbance was measured at 570 nm by a microplate reader (BioSpectrometer; Eppendorf, Hamburg, Germany). All experiments were conducted in triplicate.
2.5. Biofilm Inhibition Assay
A standardized
C. albicans suspension (100 µL) was added to each well of a sterile 96-well tissue-culture plate and incubated at 37°C for 24 h to establish biofilms. Following adhesion, the medium was aspirated, and wells were gently rinsed twice with PBS. Treatment solutions (100 µL per well) were then applied under the following conditions: HNK, 3.2 µg/mL; HNK, 3.2 µg/mL + FLZ 0.5 µg/mL; FLZ, 0.5 µg/mL; and an untreated control of RPMI 1640 medium alone. Plates were incubated statically at 37°C for an additional 24 h. After incubation, supernatants were removed, and wells were washed twice with PBS. To quantify metabolic activity, 200 µL of 2,3-bis-(2-methoxy-4-nitro-5-sulphenyl)-(2H)-tetrazolium-5-carboxanilide (XTT)/menadione solution (Sigma, USA) was added to each well in the dark, the plates were covered with aluminum foil to protect from light, and incubated statically at 37°C for 1 h. Subsequently, 100 µL of the supernatant from each well was transferred to a fresh sterile 96-well plate; absorbance was measured at 492 nm by a microplate reader. The percentage inhibition of biofilm formation was calculated as described by Koban et al. [
42].
2.6. Quantification of Intracellular Reactive Oxygen Species
Intracellular reactive oxygen species (ROS) were quantified by the oxidation-sensitive dye 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA; Sigma, USA). A standardized C. albicans suspension (100 µL) was dispensed into sterile black 96-well tissue-culture plates and incubated at 37°C for 24 h to permit adhesion. After incubation, the culture medium was removed, and wells were rinsed twice with sterile PBS. Wells were then treated with 100 µL of one of the following solutions: HNK 3.2 µg/mL; HNK 3.2 µg/mL + FLZ 0.5 µg/mL; FLZ 0.5 µg/mL; or RPMI 1640 medium as an untreated control. Plates were incubated statically at 37°C for an additional 24 h. Subsequently, 30 µL of DCFH-DA (10 µM) was added to each well, and the plates were incubated in the dark at 37°C for 40 min. The supernatant was carefully removed, and wells were washed twice with sterile PBS to eliminate excess extracellular dye. Finally, 200 µL of sterile RPMI 1640 medium was added to each well, and fluorescence was measured at an excitation wavelength of 488 nm and an emission wavelength of 522 nm by a microplate reader.
2.7. Morphological Analysis with the Light Microscope and Scanning Electron Microscope
To investigate the effect of HNK in combination with FLZ on the various morphological states of C. albicans, fungal cells were calculated in RPMI 1640 medium t 37°C for 24 h and subsequently examined using an all-in-one microplate (BZ-X800; Keyence Corporation, Tokyo, Japan) and a scanning electron microscope (SEM) (S-4800; Hitachi High-Tech Corporation, Tokyo, Japan). Regarding light microscopic observation, 10 μL of the C. albicans, suspension was analyzed under bright-field illumination at a magnification of ×200. C. albicans cultures were first incubated in RPMI 1640 at 37°C until an optical density of 1.2 at 600 nm (OD600) was reached for scanning electron microscopy. Thereafter, 100 μL of culture was inoculated into 10 mL of fresh RPMI 1640 medium containing HNK and FLZ or no treatment control. Aliquots of 1 mL were then dispensed onto glass coverslips placed in 24-well plates and incubated for an additional 24 h at 37 °C. Following incubation, cells were fixed overnight at 4 °C in 2.5% glutaraldehyde and rinsed with sterile phosphate-buffered saline. The specimens were subsequently dehydrated through a graded ethanol series (30–100%), with each step lasting 15 min, after which the coverslips were air-dried. After sputter-coating with a thin osmium layer, the samples were examined by SEM. Three randomly selected fields per specimen were imaged at ×2000 magnification.
2.8. Quantitative Real-Time Polymerase Chain Reaction Analysis of C. albicans Virulence-Related Gene Expression
Biofilms of
C. albicans were generated using sterile, enzyme-free 6-well tissue-culture plates. Each well contained 2 mL of a standardized fungal suspension and was incubated for 48 h to allow mature biofilm formation. Following incubation, the wells were gently washed three times with ice-cold sterile PBS to remove non-adherent (planktonic) cells. Two mL of one of the following test solutions was added to each well for treatment: HNK (3.2 µg/mL), HNK (3.2 µg/mL) combined with FLZ (0.5 µg/mL), or FLZ alone (4 µg/mL). Wells assigned to the negative control group contained 2 mL of blank RPMI 1640 medium. After 24 h of further incubation, the supernatant was carefully aspirated with a sterile, enzyme-free pipette tip. Adherent biofilms were then dislodged using a sterile biofilm scraper; the collected cells were centrifuged to pellet biomass. Pellets were washed once with diethylpyrocarbonate (DEPC)-treated water, centrifuged again, and supernatants discarded. Total ribonucleic acid (RNA) was isolated from each group using the NucleoSpin
® RNA Plant and Fungi kit (MACHEREY-NAGEL, Düren, Germany) according to the manufacturer’s instructions. RNA purity and concentration were assessed with a NanoDrop
™ 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). First-strand complementary deoxyribonucleic acid (cDNA) was synthesized from purified RNA using the ReverTra Ace
® qPCR RT Master Mix (Toyobo Co., Osaka, Japan). Primers (
Table 1) were designed to generate specific amplicons targeting the
HWP1 and
ALS3 genes. Quantitative real-time polymerase chain reaction (qRT-PCR) was performed on a StepOne
™ Real-Time PCR System (version 2; Applied Biosystems
®, Foster City, CA, USA) using Thunderbird
® Next SYBR
® qPCR Mix (Toyobo Co.) following the supplier’s protocol. The cycling program consisted of an initial denaturation at 95°C for 30 s, followed by 40 cycles of 95°C for 5 s and 60°C for 10 s. Relative gene expression was analyzed using the 2
−ΔΔCt method, with
EFB1 serving as the internal standard (Shaller M, Molecular Microbiology, 1998; Tsang PWK, PLOS ONE, 2012). Gene expression was expressed as relative values, setting the expression level of control samples to one.
2.9. Statistical Analysis
Data are expressed as means ± standard error (SE) from three independent experiments. Statistical significance of differences between means was calculated by Tukey’s test with GraphPad Prism software (version 7; GraphPad, San Diego, CA, USA). Statistical significance was set at p < 0.05, and significance levels were indicated as *p < 0.05 and **p < 0.01.
3. Results
3.1. HNK in Combination with FLZ Synergistically Inhibited the Cell Growth of C. albicans In Vitro
This study demonstrated a significant synergistic interaction between HNK and FLZ in inhibiting
C. albicans (
Table 2). Adding HNK at a concentration of 3.2 μg/mL notably reduced the MIC ranges of FLZ from over 4.0 to 0.5 μg/mL, indicating a potent synergistic effect against
C. albicans. The interaction between the two antifungal agents was assessed by the fractional inhibitory concentration index (FICI), based on the principles of Loewe additivity. This framework assumes a compound does not interact with itself, and comparisons are made under conditions yielding equivalent biological effects. The FICI was calculated using the formula:
FICI = FICIA + FICIB = (MIC Acomb) / (MIC Aalone) + (MIC Bcomb) / (MIC Balone)
Interpretation of the index follows established criteria: FICI ≤ 0.5 indicates synergy, 0.5 < FICI ≤ 1 indicates an additive effect, 1 < FICI ≤ 4 indicates no interaction, and FICI > 4 indicates antagonism.
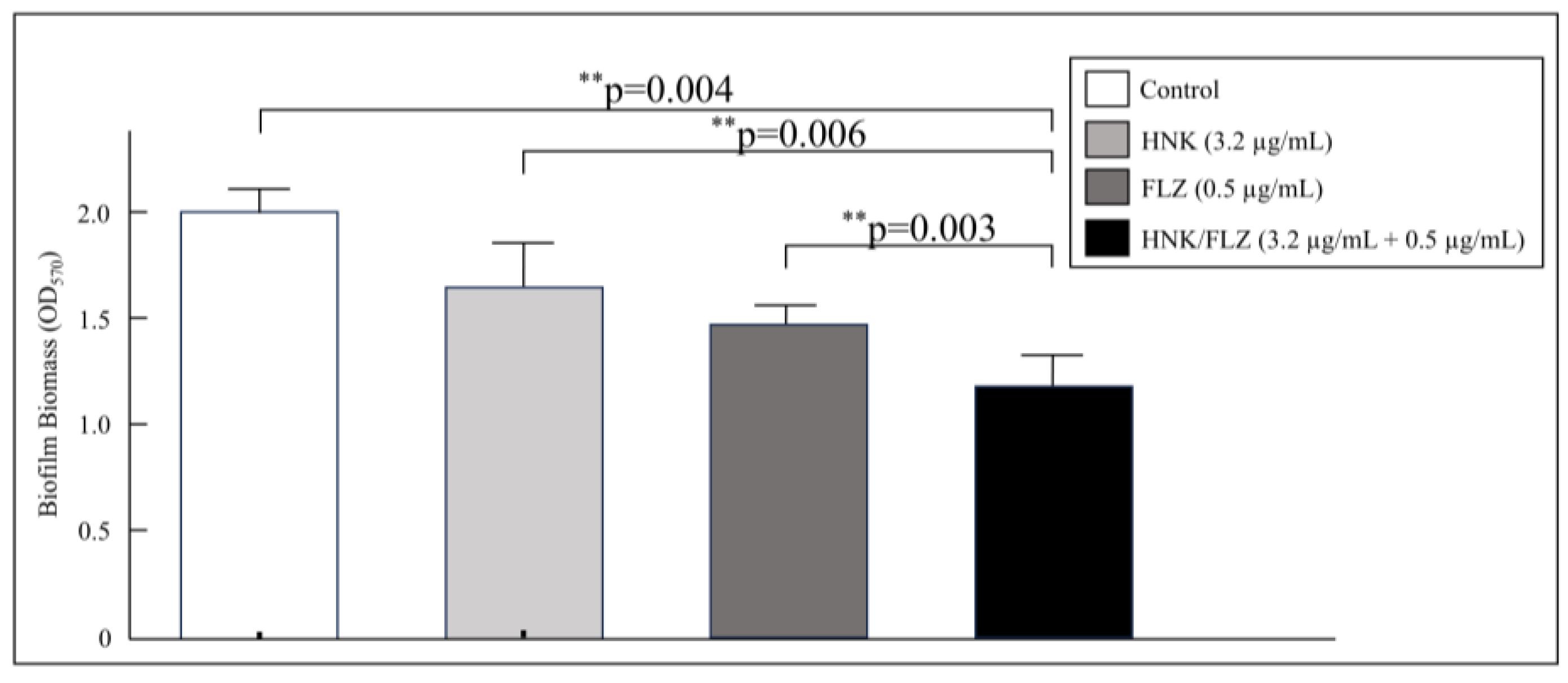
3.2. HNK in Combination with FLZ Inhibited Biofilm Biomass
Figure 1 illustrates the effect of combining HNK with FLZ on the biofilm biomass of
C. albicans. The combined treatment produced a substantial reduction in biofilm biomass compared to the control group, FLZ alone, or HNK alone (
Figure 1).
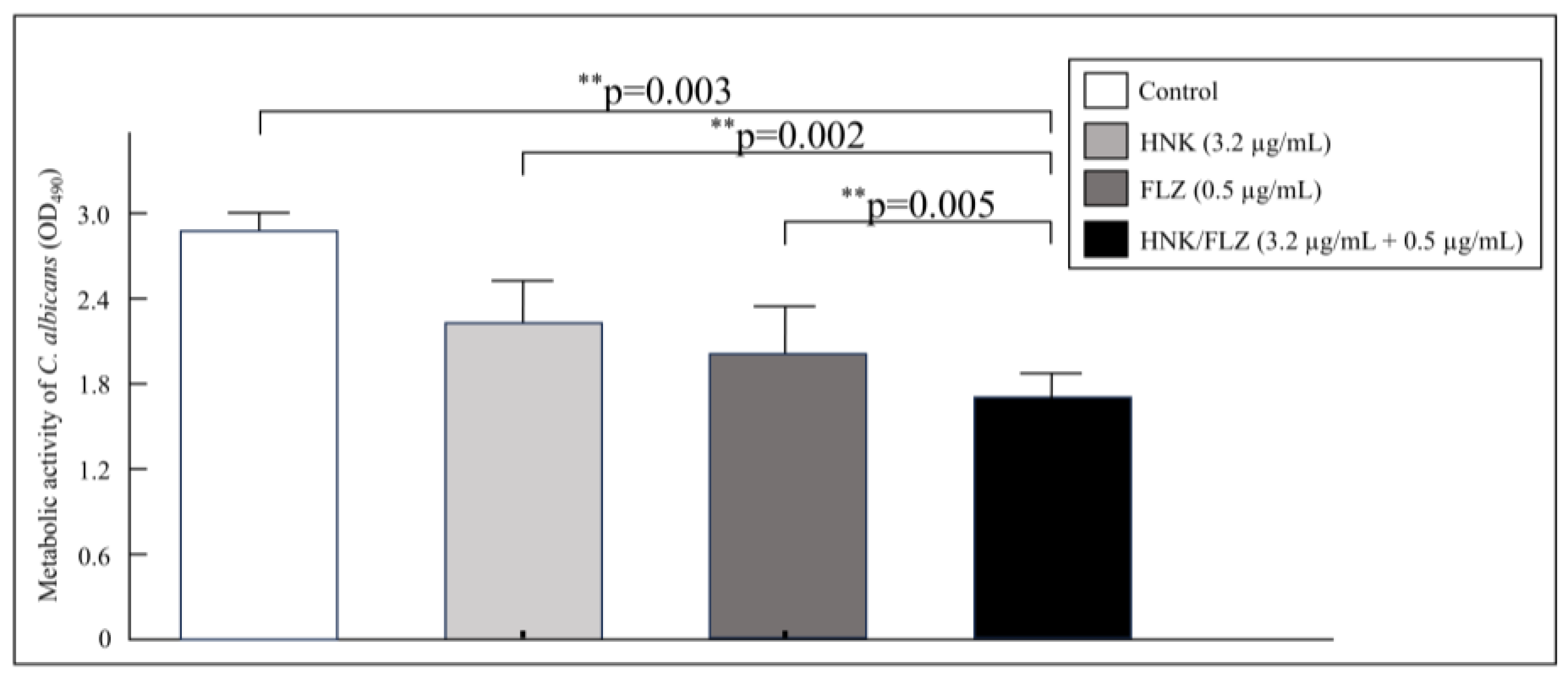
3.3. XTT Assay Evaluating the Inhibitory Effect on Biofilms
The impact of HNK combined with FLZ on both planktonic and biofilm-associated
C. albicans cells was quantitatively assessed by the XTT reduction assay. Biofilm inhibition was expressed as the percentage reduction in metabolic activity relative to untreated controls. Notably, treatment with either HNK or FLZ at their respective MIC values significantly suppressed metabolic activity, and further reductions were observed with three tested combination regimens. Among these, the co-administration of HNK at 3.2 µg/mL and FLZ at 0.5 µg/mL produced the most pronounced and reproducible inhibitory effect on biofilm viability (
Figure 2).
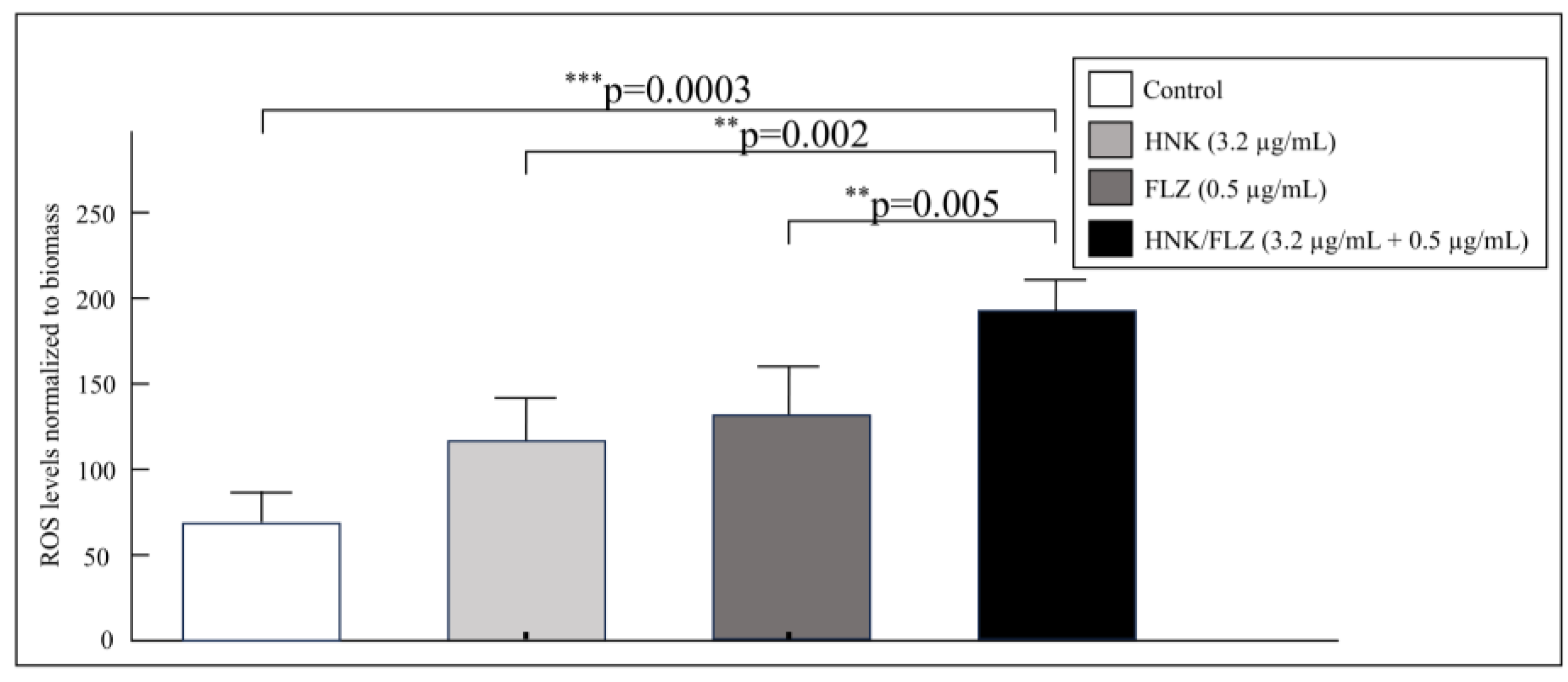
3.4. Quantification of Intracellular Reactive Oxygen Species
Using DCFH-DA as a fluorescent probe, intracellular ROS accumulation was evaluated in
C. albicans biofilms at both early and mature stages following treatment with varying concentrations of HNK and FLZ, either alone or in combination. Across all tested concentrations, fluorescence intensity was significantly elevated compared to the untreated control, indicating enhanced intracellular ROS generation. Combination therapy with HNK and FLZ at high, medium, and low doses produced fluorescence levels comparable to the respective MIC values of each agent administered individually. Notably, the combination of HNK at 3.2 μg/mL and FLZ at 0.5 μg/mL yielded the greatest and most consistent increase in ROS fluorescence in biofilms (
Figure 3). These findings suggest that this specific combination could markedly amplify oxidative stress; thereby, promoting fungal cell damage and death.
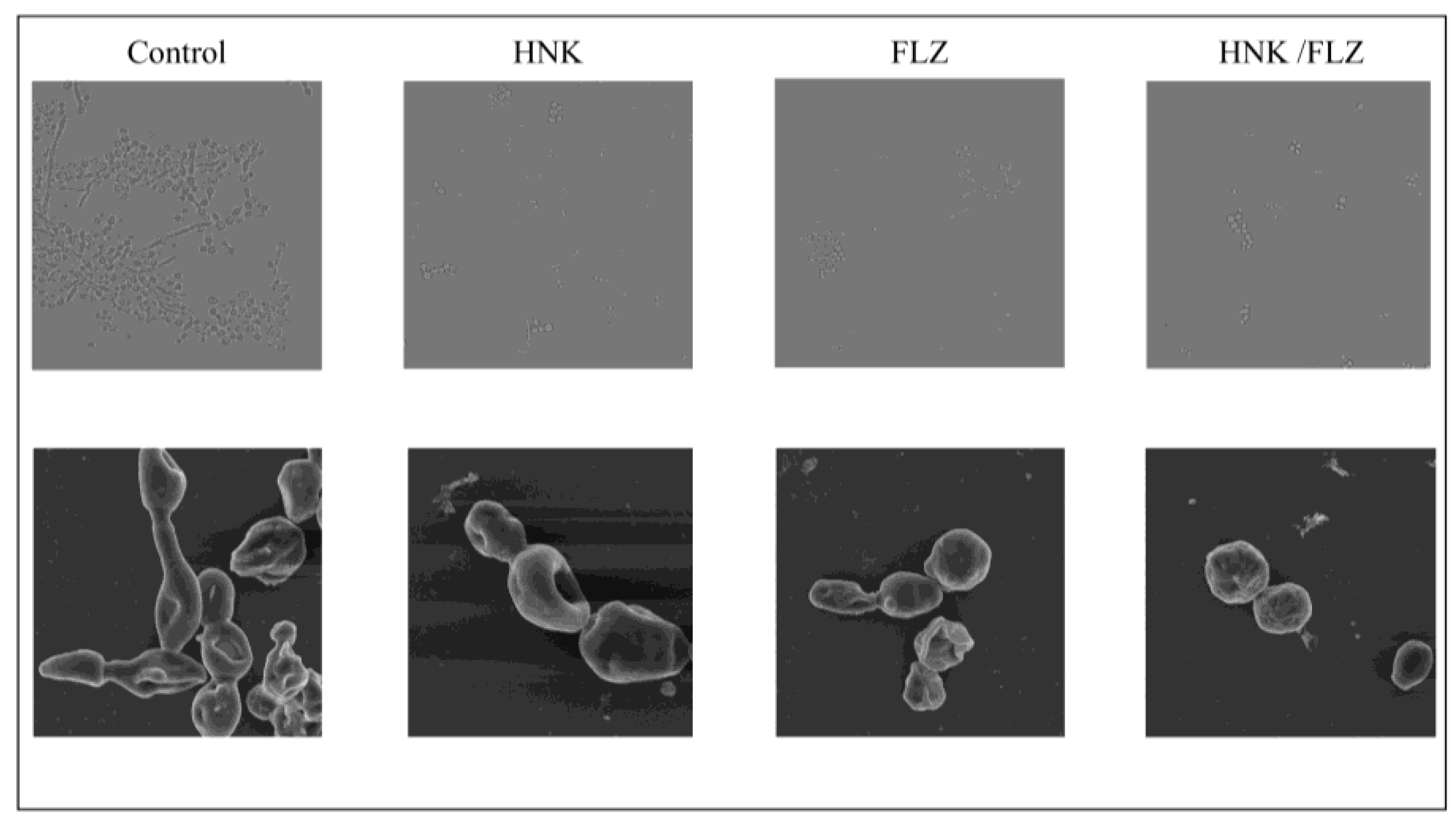
3.5. Effect of HNK and FLZ on C. albicans Biofilm Formation by Morphology
Hyphal development was evaluated by fluorescence microscopy. In drug-resistant
C. albicans, exposure to HNK combined with FLZ markedly suppressed filamentation compared with FLZ alone and the untreated control (
Figure 4). In the combination group, cells remained exclusively in the yeast form, without detectable hyphal structures. In contrast, the control group exhibited extensive hyphal networks, whereas FLZ-treated and HNK-treated groups retained residual hyphal clusters, although to a much lesser extent than the control group. These findings suggest that the dual-drug regimen could synergistically inhibit the transition from yeast to hyphae.
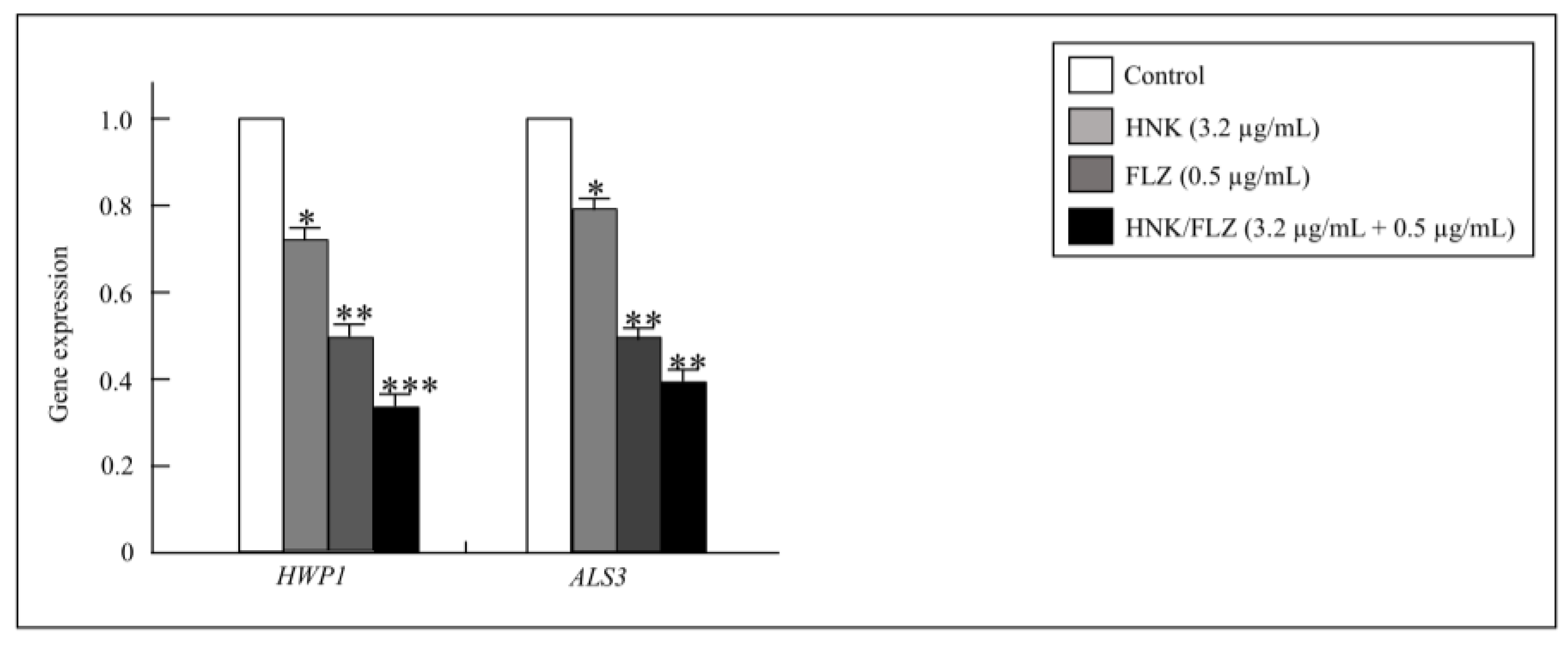
3.6. Expression Analysis of ALS3 and HWP1 in C. albicans Biofilm Formation by RT-PCR
Given the inhibitory effects of HNK and FLZ on
C. albicans biofilm formation, transcriptional profiles of biofilm-associated genes were examined by RT-PCR. The analysis demonstrated a marked reduction in
ALS3 and
HWP1 expression following treatment. Downregulation observed at combined concentrations was comparable to that achieved at the respective MIC levels of HNK or FLZ individually. Importantly, the combination of HNK (3.2 μg/mL) and FLZ (0.5 μg/mL) produced the strongest suppressive effect, resulting in greater transcriptional inhibition than either compound alone at its MIC concentration (
Figure 5).
4. Discussion
C. albicans is frequently exist as a normal resident microorganism on human epithelial surfaces, including the skin and the vaginal, gastrointestinal, and oropharyngeal mucosa. However, under conditions such as immunosuppression or microbiota imbalance, it can transition into an opportunistic pathogen leading to clinical manifestations that span from mild mucosal involvement to severe, potentially fatal disseminated candidiasis. A major contributor to its pathogenicity is biofilm formation, which provides protection against antifungal agents and host immune defenses, which is associated with a significant disease burden and increased fatality, most notably in immunosuppressed populations [
46,
47]. Biofilm formation also markedly increases resistance to first-line antifungal agents, complicating clinical management [
48]. In the context of rising antifungal resistance, especially in biofilm-associated
C. albicans infections, the use of combined antifungal agents has gained recognition as a valuable approach to treatment. The synergistic interaction between HNK and FLZ observed in this study highlights the potential of combination therapy to enhance antifungal activity.
The need for alternative strategies targeting biofilm formation and maintenance is increasingly recognized. Approaches such as novel antifungal agents and rational drug combinations are essential for reducing the burden of biofilm-associated infections [
46,
49]. The demonstrated efficacy of the HNK–FLZ combination in this study underscores the therapeutic potential of such strategies and suggests their ability to overcome limitations inherent to current antifungal regimens.
HNK exhibits potent antimicrobial activity by suppressing
C. albicans biofilm formation through downregulation of
HWP1 and
ALS3, genes critical for hyphal development, adhesion, biofilm maturation, and immune evasion. Experimental evidence supports the inhibitory effect of HNK on biofilm formation [
50]. Low-dose combination regimens, such as HNK with FLZ, can effectively disrupt fungal biofilms. These strategies highlight the therapeutic potential of sub-inhibitory drug concentrations to interfere with biofilm-associated resistance pathways and restore antifungal susceptibility.
The marked reduction in MIC values observed with the combined use of HNK and FLZ demonstrates a robust synergistic interaction. This finding suggests that low-dose combination therapy can effectively potentiate biofilm disruption; thereby, enhancing antifungal efficacy. Additionally, accumulating evidence indicates that oxidative stress serves as a pivotal mechanism underlying antifungal activity against
C. albicans, particularly within biofilms. Previous reports showed that D319 induces ROS-dependent apoptosis through suppression of isocitrate lyase activity, which leads to impaired mitochondrial function and ultimately culminating in cell death [
51]. It has been shown that (+)-medioresinol increasing intracellular generation of ROS, which subsequently induces apoptosis via mitochondria-associated pathways [
52]. Likewise, the antifungal properties of nanoparticles are attributed to their ability to elevate oxidative stress, impair cell wall structure, and trigger ROS-dependent cellular death [
53]. Collectively, these findings are consistent with our results and indicate that the enhanced antifungal activity of the HNK-FLZ combination is likely driven, at least in part, by the promotion of oxidative stress, which in turn diminishes fungal viability and contributes to improved therapeutic effectiveness. These observations were further corroborated by SEM analysis findings. Strains exposed to the two-drug combination formed biofilms composed almost entirely of yeast cells with minimal filamentous structures. In contrast, both the FLZ-treated group and the untreated control exhibited markedly greater filamentation (
Figure 4).
To gain deeper insight into how the HNK-FLZ combination suppresses C.
albicans biofilm formation, we analyzed the transcriptional profiles of representative genes associated with adhesion, hyphal morphogenesis, and stress-related responses. RT-PCR analysis demonstrated a pronounced suppression of
ALS3 and
HWP1 following combination treatment. Notably, the three tested concentration pairs exhibited inhibitory profiles comparable to the MIC levels of each agent individually, with HNK at 3.2 μg/mL combined with FLZ at 0.5 μg/mL producing the most substantial reduction in gene expression. This downregulation exceeded that achieved by either compound alone at its respective MIC concentration. The observed transcriptional repression aligns with the established roles of these genes in fungal pathogenicity. The adhesion-associated gene
ALS3 is essential for initial attachment and structural stability of biofilms [
54,
55,
56].
HWP1, a hypha-specific gene, is critical for filamentous growth, a defining feature of mature biofilm architecture [
56,
57]. Collectively, these findings indicate that the HNK–FLZ combination disrupts multiple virulence pathways—adhesion, hyphal morphogenesis, and stress tolerance—providing a mechanistic basis for its enhanced antibiofilm efficacy against
C. albicans.
The present findings provide new insight into elucidating the molecular mechanisms underlying resistance in
C. albicans and indicate that combinational treatment approaches, specifically those influencing specific gene regulatory pathways, could offer an effective option for the control of fungal infections. The synergistic suppression of key genes involved in biofilm formation and virulence by HNK and FLZ highlights a potential therapeutic pathway for addressing the multifaceted challenge of antifungal resistance. These findings warrant further investigation into the specific molecular interactions triggered by the combination treatment to identify novel targets that could strengthen antifungal interventions [
58,
59]. In line with current research trends [
60], a growing consensus supports the value of combination approaches against fungal biofilms. Recent reviews [
61] have emphasized that understanding biofilm biology and identifying actionable molecular targets are essential for developing more sophisticated antifungal therapies [
62]. This work contributes to this evolving discourse by offering new insights into biofilm-mediated resistance mechanisms and underscoring the potential of innovative therapeutic strategies to advance the management of fungal infections.
5. Conclusions
To confront the increasing problem of antifungal resistance, particularly of biofilm-forming C. albicans, we carried out a set of in vitro investigations to assess the effects of combined treatment with HNK-FLZ. The results demonstrated clear synergistic activity against established biofilms. Biofilm imaging using fluorescein isothiocyanate–concanavalin A and quantitative assessment of gene expression via qRT-PCR revealed that this combination disrupted biofilm architecture and markedly downregulated key genes involved in fungal adhesion and hyphal development. The results indicate that combination-based therapeutic strategies for defining molecular targets may markedly enhance antifungal treatment efficacy and represent a viable approach to addressing the challenges posed by antifungal resistance. Furthermore, our findings provide initial experimental evidence supporting the potential clinical use of HNK alongside antimicrobial agents for the treatment of infections associated with drug-resistant C. albicans.
Author Contributions
Conceptualization, H.Y. and R.K.; methodology, K.K.; software, K.K.; validation, H.Y., R.K. and K.K.; formal analysis, H.Y.; investigation, H.Y.; resources, R.K.; data curation, R.K.; writing—original draft preparation, H.Y.; writing—review and editing, T.Y.; visualization, H.Y.; supervision, H.Y.; project administration, R.K.; funding acquisition, H.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by a Grant-in-Aid for Scientific Research (C) (Grant No. JP23K16240) from the Japan Society for the Promotion of Science (JSPS), funded by the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) of Japan. The APC was also covered by this grant.
Institutional Review Board Statement
“Not applicable” for studies not involving humans or animals.
Informed Consent Statement
“Not applicable. ”for studies not involving humans.
Data Availability Statement
The original contributions presented in this study are included in the article. Furthermore, inquiries can be directed to the corresponding authors.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Abbreviations
The following abbreviations are used in this manuscript:
| HNK |
hinokitiol |
| MIC |
minimum inhibitory concentration |
| PBS |
Phosphate-buffered saline |
| FLZ |
fluconazole |
| ROS |
Intracellular reactive oxygen species |
| C. albicans |
Candida albicans |
| FICI |
fractional inhibitory concentration index |
| DMSO |
dimethyl sulfoxide |
References
- Chin, V.K.; Lee, T.Y.; Rusliza, B.; Chong, P.P. Dissecting Candida albicans infection from the perspective of C. albicans virulence and omics approaches on host-pathogen interaction: a review. Int. J. Mol. Sci. 2016, 17, 1643. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.A.; Diekema, D.J. Epidemiology of invasive candidiasis: a persistent public health problem. Clin. Microbiol. Rev. 2007, 20, 133–163. [Google Scholar] [CrossRef]
- Gow, N.A.R; Yadav, B. Microbe Profile: Candida albicans: a shape-changing,opportunistic pathogenic fungus of humans. Microbiol. 2017, 163, 1145–1147. [Google Scholar] [CrossRef] [PubMed]
- Robbins, N.; Caplan, T.; Cowen, L.E. Molecular evolution of antifungal drug resistance. Annu. Rev. Microbiol. 2017, 71, 753–775. [Google Scholar] [CrossRef]
- Kohli, A.; Smriti, N.F.N.; Mukhopadhyay, K.; Rattan, A.; Prasad, R. Invitro low-level resistance to azoles in Candida albicans is associated with changes in membrane lipid fluidity and asymmetry. Antimicrob. Agents Chemother. 2002, 46, 1046–1052. [Google Scholar] [CrossRef]
- Khanna, I. Drug discovery in pharmaceutical industry: productivity challenges and trends. Drug Discov. Today 2012, 17, 1088–1102. [Google Scholar] [CrossRef]
- Wang, C.H.; Yu, J.; Cai, Y.X.; Zhu, P.P.; Liu, C.Y.; Zhao, A.C.; Lü, R.H.; Li, M.J.; Xu, F.X; Yu, M.D. Characterization and functional analysis of 4-coumarate: CoA ligase genes in mulberry. PLoS One 2016, 11, e0155814. [Google Scholar] [CrossRef]
- Fisher, M.C.; Alastruey-Izquierdo, A.; Berman, J.; Bicanic, T.; Bignell, E.M.; Bowyer, P.; Bromley, M.; Brüggemann, R.; Garber, G.; Cornely, O.A; et al. Tackling the emerging threat of antifungal resistance to human health. Nat. Rev. Microbiol. 2022, 20, 557–571. [Google Scholar] [CrossRef]
- Donlan, R.M.; Costerton, J.W. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin. Microbiol. Rev. 2002, 15, 167–193. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, T.; Silva, S.; Henriques, M. Candida tropicalis biofilm’s matrix–involvement on its resistance to amphotericin B. Diagn. Microbiol. Infect. Dis. 2015, 83, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Taff, H.T.; Mitchell, K.F.; Edward, J.A.; Andes, D.R. Mechanisms of Candida biofilm drug resistance. Future Microbiol. 2013, 8, 1325–1337. [Google Scholar] [CrossRef] [PubMed]
- Van Daele, R.; Spriet, I.; Wauters, J.; Maertens, J.; Mercier, T.; Van Hecke, S.; Brüggemann, R. Antifungal drugs: what brings the future? Med. Mycol 2019, 57, S328–S343. [Google Scholar] [CrossRef]
- Lehar, J.; Krueger, A.S.; Avery, W.; Heilbut, A.M.; Johansen, L.M.; Price, E.R.; Rickles, R.J.; Short, G.F., 3rd; Staunton, J.E.; Jin, X; et al. Synergistic drug combinations tend to improve therapeutically relevant selectivity. Nat. Biotechnol. 2009, 27, 659–666. [Google Scholar] [CrossRef]
- Ahmad, A.; Wani, M.Y.; Khan, A.; Manzoor, N.; Molepo, J. Synergistic interactions of eugenol-tosylate and its congeners with fluconazole against Candida albicans. PLoS One 2015, 10, e0145053. [Google Scholar] [CrossRef]
- Shrestha, S.K.; Fosso, M.Y.; Garneau-Tsodikova, S. A combination approach to treating fungal infections. Sci. Rep. 2015, 5, 17070. [Google Scholar] [CrossRef]
- Li, X.; Wu, X.; Gao, Y.; Hao, L.; Sun, S. Apoptosis-linked antifungal effect of ambroxol hydrochloride by cystolic calcium concentration disturbance in resistant Candida albicans. Sci. China Life Sci. 2019, 62, 1601–1604. [Google Scholar] [CrossRef]
- Rajasekharan, S.K.; Lee, J.H.; Lee, J. Aripiprazole repurposed as an inhibitor of biofilm formation and sterol biosynthesis in multidrug-resistant Candida albicans. Int. J. Antimicrob. Agents 2019, 54, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Yan, H.; Lu, M.; Wang, D.; Sun, S. Antifungal activity of ribavirin used alone or in combination with fluconazole against Candida albicans is mediated by reduced virulence. Int. J. Antimicrob. Agents 2020, 55, 105804. [Google Scholar] [CrossRef] [PubMed]
- Bersan, S.M.; Galvao, L.C.; Goes, V.F.; Sartoratto, A.; Figueira, G.M.; Rehder, V.L.; Alencar, S.M.; Duarte, R.M.; Rosalen, P.L; Duarte, M.C. Action of essential oils from Brazilian native and exotic medicinal species on oral biofilms. BMC Complement Altern. Med. 2014, 14, 451. [Google Scholar] [CrossRef]
- Rodrigues de Araújo, A.; Iles, B.; de Melo Nogueira, K.; Dias, J.D.N.; Plácido, A.; Rodrigues, A.; Albuquerque, P.; Silva-Pereira, I.; Socodatto, R.; Portugal, C.C; et al. de Souza de Almeida Leite, J. R., Antifungal and anti-inflammatory potential of eschweilenol C-rich fraction derived from Terminalia fagifolia Mart. J. Ethnopharmacol 2019, 240, 111941. [CrossRef]
- Fabri, R.L.; Freitas, J.C.O.; Lemos, A.S.O.; Campos, L.M.; Diniz, I.O.M.; Pinto, N.C.C.; Silva, T.P.; Palazzi, C.; Marchesini, P.; Monteiro, C; et al. Spilanthol as a promising antifungal alkylamide for the treatment of vulvovaginal candidiasis. Med. Mycol 2021, 59, 1210–1224. [Google Scholar] [CrossRef]
- Ahmad, A.; Khan, A.; Akhtar, F.; Yousuf, S.; Xess, I.; Khan, L.A.; Manzoor, N. Fungicidal activity of thymol and carvacrol by disrupting ergosterol biosynthesis and membrane integrity against Candida. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 41–50. [Google Scholar] [CrossRef]
- Khan, M.S.; Ahmad, I. Antibiofilm activity of certain phytocompounds and their synergy with fluconazole against Candida albicans biofilms. J. Antimicrob. Chemother. 2012, 67, 618–621. [Google Scholar] [CrossRef]
- Pemmaraju, S.C.; Pruthi, P.A.; Prasad, R.; Pruthi, V. Candida albicans biofilm inhibition by synergistic action of terpenes and fluconazole. Indian J. Exp. Biol. 2013, 51, 1032–1037. [Google Scholar]
- Park, K.S.; Kang, K.C.; Kim, J.H.; Adams, D.J.; Johng, T.N.; Paik, Y.K. Differential inhibitory effects of protoberberines on sterol and chitin biosyntheses in Candida albicans. J. Antimicrob. Chemother. 1999, 43, 667–674. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, Y.; Yan, L.; Liang, R.M.; Dai, B.D.; Tang, R.J.; Gao, P.H; Jiang, Y.Y. Proteomic analysis reveals a synergistic mechanism of fluconazole and berberine against fluconazole-resistant Candida albicans: endogenous ROS augmentation. J. Proteome Res. 2009, 8, 5296–5304. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Chen, Z.; Chen, X.; Cao, L.; Liu, P.; Sun, S. The combination of minocycline and fluconazole causes synergistic growth inhibition against Candida albicans: an in vitro interaction antifungal antibacterial agents. FEMS Yeast Res. 2010, 10, 885–893. [Google Scholar] [CrossRef]
- De Cremer, K.; Lanckacker, E.; Cools, T.L.; Bax, M.; De Brucker, K.; Cos, P.; Cammue, B.P.; Thevissen, K. Artemisinins, new fluconazole potentiators resulting in increased activity against Candida albicans biofilms. Antimicrob. Agents Chemother. 2015, 59, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Fotopoulou, T.; Ćirić, A.; Kritsi, E.; Calhelha, R.C.; Ferreira, I.C.; Soković, M.; Zoumpoulakis, P.; Koufaki, M. Antimicrobial/antibiofilm activity and cytotoxic studies of b-thujaplicin derivatives. Arch. Pharm. Chem. Life Sci. 2016, 349, 698–709. [Google Scholar] [CrossRef]
- Kim, D.J.; Lee, M.W.; Choi, J.S.; Lee, S.G.; Park, J.Y.; Kim, S.W. Inhibitory activity of hinokitiol against biofilm formation in fluconazole-resistant Candida species. PLoS One 2017, 12, e0171244. [Google Scholar] [CrossRef] [PubMed]
- Kanda, R.; Yoshimatsu, H.; Lyu, X. Investigation of antibacterial effects of hinokitiol on Lactobacillus casei. J. Osaka Dent. Univ. 2025, 59, 7–11. [Google Scholar]
- Tsuji, N.; Yoshimatsu, H.; Kanda, R.; Hashimoto, N.; Maeda, H. Investigation of antibacterial effects of hinokitiol on Fusobacterium nucleatum. WJARR 2025, 25, 103–108. [Google Scholar] [CrossRef]
- Yoshimatsu, H.; Kanda, R.; Xinghui, L.; Kano, K. Hinokitiol exhibits potent anticancer effects in human oral squamous cell carcinoma by inducing apoptosis and inhibition of cell migration. J. Oral Maxillofac. Surg. Med. Pathol. 2025, 37. [Google Scholar] [CrossRef]
- Vila, T.; Sultan, A.S.; Montelongo-Jauregui, D; Jabra-Rizk, M.A. Oral candidiasis: a disease of opportunity. J. Fungi 2020, 6, 15. [Google Scholar] [CrossRef] [PubMed]
- Perić, M.; Miličić, B.; Kuzmanović Pfićer, J.; Živković, R.; Arsić Arsenijević, V. A systematic review of denture stomatitis: predisposing factors, clinical features, etiology, and global Candida spp. distribution. J. Fungi 2024, 10, 328. [Google Scholar] [CrossRef]
- Nadig, S.D.; Ashwathappa, D.T.; Manjunath, M.; Krishna, S.; Annaji, A.G.; Shivaprakash, P.K. A relationship between salivary flow rates and Candida counts in patients with xerostomia. J. Oral Maxillofac. Pathol. 2017, 21, 316. [Google Scholar] [CrossRef]
- Pagheh, A.S.; Kardan, F.; Ghojoghi, A.; Sebzari, A.R.; Erfaninejad, M.; Askari, P.; Aghili, S.R.; Nazar, E.; Ziaee, M. Exploring oral candidiasis among cancer patients undergoing chemotherapy in eastern Iran. Iran. J. Microbiol. 2024, 16, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Lalla, R.V.; Latortue, M.C.; Hong, C.H.; Ariyawardana, A.; D'Amato-Palumbo, S.; Fischer, D.J.; Martof, A.; Nicolatou-Galitis, O.; Patton, L.L.; Elting, L.S.; et al. A systematic review of oral fungal infections in medically compromised patients. Support. Care. Cancer. 2010, 18, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; et al. Hematogenous Candida albicans invasion of the brain induces transient cerebritis and memory impairment — implications for neurodegeneration. Nat Commun. 2018, 9, 1–12. [Google Scholar]
- Kulisova, M.; Matatkova, O.; Branyik, T.; Zelenka, J.; Drabova, L.; Kolouchova, I.J. Detection of microscopic filamentous fungal biofilms choosing the suitable methodology. J. Microbiol. Methods 2023, 205, 106676. [Google Scholar] [CrossRef]
- Manoharan, R.K.; Lee, J.H.; Kim, Y.G.; Lee, J. Alizarin and chrysazin inhibit biofilm and hyphal formation by candida albicans. Front. Cell Infect. Microbiol. 2017, 7, 447. [Google Scholar] [CrossRef]
- Koban, I.; Matthes, R.; Hubner, N.O.; Welk, A.; Sietmann, R.; Lademann, J.; Kramer, A.; Kocher, T. XTT assay of ex vivo saliva biofilms to test antimicrobial influences. GMS Krankenhhyg Interdiszip 2012, 7, Doc06. [Google Scholar] [CrossRef] [PubMed]
- Samaranayake, Y.H.; Dassanayake, R.S.; Jayatilake, J.A.; Cheung, B.P.; Yau, J.Y.; Yeung, K.W.; Samaranayake, L.P. Phospholipase B enzyme expression is not associated with other virulemce attributes in Candida albicans isolates from patients with human immunodeficiency virus infection. J. Med. Microbiol. 2005, 54, 583–593. [Google Scholar] [CrossRef] [PubMed]
- Naglik, J.R.; Fostira, F.; Ruprai, J.; Staab, J.F.; Challacombe, S.J.; Sundstrom, P. Candida albicans HWP1 gene expression and host antibody responses in colonization and disease. J. Med. Microbiol. 2006, 55, 1323–1327. [Google Scholar] [CrossRef] [PubMed]
- Uppuluri, P.; Dinakaran, H.; Thomas, D.P.; Chaturvedi, A.K.; Lopez-Ribot, J.L. Characteristics of Candida albicans biofilms grown in a synthetic urine medium. J. Clin. Microbiol. 2009, 47, 4078–4083. [Google Scholar] [CrossRef]
- Atiencia-Carrera, M.B.; Cabezas-Mera, F.S.; Vizuete, K.; Debut, A.; Tejera, E.; MaChado, A. Evaluation of the biofilm life cycle between Candida albicans and Candida tropicalis. Front. Cell Infect. Microbiol. 2022, 12, 953168. [Google Scholar] [CrossRef]
- Yu, J.; Wang, F.; Shen, Y.; Yu, F.; Qiu, L.; Zhang, L.; Chen, Y.; Yuan, Q.; Zhang, H.; Sun, Y.; et al. Inhibitory effect of ficin on Candida albicans biofilm formation and pre-formed biofilms. BMC Oral. Health 2022, 22, 350. [Google Scholar] [CrossRef]
- Zhu, J.; Chu, W.; Luo, J.; Yang, J.; He, L.; Li, J. Dental materials for oral microbiota dysbiosis: an update. Front. Cell Infect. Microbiol. 2022, 12, 900918. [Google Scholar] [CrossRef]
- Atiencia-Carrera, M.B.; Cabezas-Mera, F.S.; Tejera, E.; MaChado, A. Prevalence of biofilms in Candida spp. bloodstream infections: a meta-analysis. PLoS One 2022, 17, e0263522. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.J.; Lee, M.W.; Choi, J.S.; Lee, S.G.; Park, J.Y.; Kim, S.W. Inhibitory activity of hinokitiol against biofilm formation in fluconazole-resistant Candida species. PLoS One 2017, 12, e0171244. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.H.; Hwang, I.S.; Liu, Q.H.; Woo, E.R.; Lee, D.G. (+)-Medioresinol leads to intracellular ROS accumulation and mitochondria-mediated apoptotic cell death in Candida albicans. Biochimie 2012, 94, 1784–1793. [Google Scholar] [CrossRef]
- Tian, W.; Li, F.; Wu, S.; Li, G.; Fan, L.; Qu, X.; Jia, X.; Wang, Y. Efficient capture and T2 magnetic resonance assay of candida albicans with inorganic nanoparticles: role of nanoparticle surface charge and fungal cell wall. ACS Biomater Sci. Eng. 2019, 5, 3270–3278. [Google Scholar] [CrossRef]
- Ding, Y.; Zhang, K.; Yin, Y.; Wu, J. D319 induced antifungal effects through ROS-mediated apoptosis and inhibited isocitrate lyase in Candida albicans. Biochim. Biophys. Acta, Gen. Subj. 2022, 1866, 130050. [Google Scholar] [CrossRef]
- Liu, Y.; Filler, S.G. Candida albicans ALS3, a multifunctional adhesin and invasion. Eukaryot. Cell 2011, 10, 168–173. [Google Scholar] [CrossRef]
- Roudbarmohammadi, S.; Roudbary, M.; Bakhshi, B.; Katiraee, F.; Mohammadi, R.; Falahati, M. ALS1 and ALS3 gene expression and biofilm formation in Candida albicans isolated from vulvovaginal candidiasis. Adv. BioMed. Res. 2016, 5, 105. [Google Scholar] [CrossRef]
- Murad, A.M.; Leng, P.; Straffon, M.; Wishart, J.; Macaskill, S.; MacCallum, D.; Schnell, N.; Talibi, D.; Marechal, D.; Tekaia, F.; et al. NRG1 represses yeast-hypha morphogenesis and hypha-specific gene expression in Candida albicans. EMBO J. 2001, 20, 4742–4752. [Google Scholar] [CrossRef] [PubMed]
- Salehipour, K.; Aboutalebian, S.; Charsizadeh, A.; Ahmadi, B.; Mirhendi, H. Differentiation of Candida albicans complex species isolated from invasive and non-invasive infections using HWP1 gene size polymorphism. Curr. Med. Mycol 2021, 7, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Romo, J.A.; Pierce, C.G.; Chaturvedi, A.K.; Lazzell, A.L.; McHardy, S.F.; Saville, S.P.; Lopez-Ribot, J.L. Development of anti-virulence approaches for candidiasis via a novel series of small-molecule inhibitors of Candida albicans filamentation. mBio 2017, 8, e01991–17. [Google Scholar] [CrossRef] [PubMed]
- Wall, G.; Montelongo-Jauregui, D.; Vidal Bonifacio, B.; Lopez-Ribot, J.L.; Uppuluri, P. Candida albicans biofilm growth and dispersal: contributions to pathogenesis. Curr. Opin. Microbiol. 2019, 52, 1–6. [Google Scholar] [CrossRef]
- Pohl, C.H. Recent advances and opportunities in the study of candida albicans polymicrobial biofilms. Front. Cell Infect. Microbiol. 2022, 12, 836379. [Google Scholar] [CrossRef]
- Chen, H.; Zhou, X.; Ren, B.; Cheng, L. The regulation of hyphae growth in Candida albicans. Virulence 2020, 11, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.R.; Yan, L.; Lv, Q.Z.; Zhou, M.; Sui, X.; Cao, Y.B.; Jiang, Y.Y. Molecular genetic techniques for gene manipulation in Candida albicans. Virulence 2014, 5, 507–520. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).