Submitted:
21 January 2026
Posted:
23 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Bioactivity and Functional Properties of Chinese Chive (Allium tuberosum) and Its Fermented Products
2.1. Key Bioactive Components: A Chemical Inventory
- Organosulfur Compounds (OSCs): These are hallmark constituents of Allium plants and underpin their characteristic pungency. In Chinese chive (Allium tuberosum), the predominant S-alk(en)yl-L-cysteine sulfoxides are methiin (S-methyl-L-cysteine sulfoxide) and, to a lesser extent, isoalliin (S-1-propenyl-L-cysteine sulfoxide), whereas alliin (S-allyl-L-cysteine sulfoxide) occurs in lower amounts. Upon tissue disruption, alliinase converts these precursors into a mixture of thiosulfinates with strong antimicrobial activity; in A. tuberosum the major products are methyl- and 1-propenyl-derived thiosulfinates, and diallyl thiosulfinate (allicin) is not the dominant species[15,16].
2.2. Verified Biological Activities: Antimicrobial and Antioxidant Mechanisms
- Antimicrobial Activity: Studies have shown that Chinese chive extracts inhibit a variety of pathogenic microorganisms, including Gram-positive bacteria (e.g., Staphylococcus aureus, Bacillus subtilis) and Gram-negative bacteria (e.g., Escherichia coli, Salmonella), as well as some fungi[22,23]. One of its mechanisms of action is to disrupt the integrity of the microbial cell membrane, causing leakage of cellular contents, thereby inhibiting or killing the microorganisms[24].
- Antioxidant Activity: The high content of total phenols and flavonoids in Chinese chive is the material basis for its strong antioxidant capacity. Assays such as DPPH radical scavenging have demonstrated that Chinese chive has significant antioxidant activity, effectively scavenging excess free radicals in the body and mitigating oxidative stress damage[19,25]. Interestingly, one study found that the antioxidant activity of Chinese chive is even higher than that of the well-known garlic (A. sativum)[19].
- Other Activities: In addition to the functions mentioned above, Chinese chive also exhibits anti-inflammatory and immunomodulatory activities[19].
2.3. Enhanced Bioactivity Through Microbial Fermentation: Implications for Potential Complementarity
3. Black Soldier Fly (Hermetia illucens) Larvae: A Novel Source of Metabolically Active Fatty Acids
3.1. Unique Lipid Composition: Focus on Lauric Acid
3.2. Biological Roles of Medium-Chain Fatty Acids in Animal Nutrition
4. Regulation of Ruminant Metabolism: Mechanisms and Evidence
4.1. Effects of Bioactive Substances from Allium Plants on Rumen Fermentation and Methanogenesis
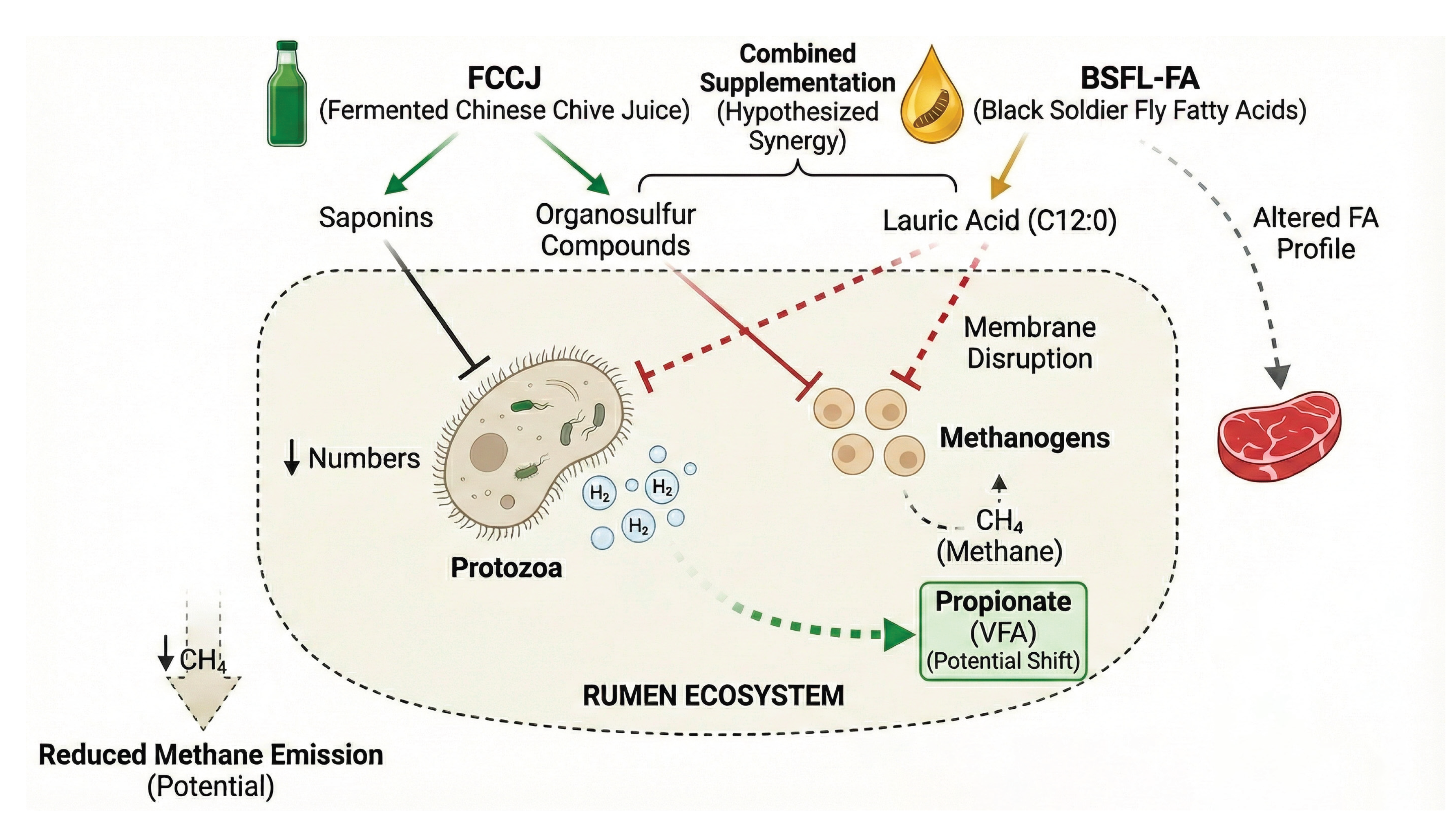
- Role of Saponins: The saponins in Chinese chive are effective antiprotozoal agents. Their mechanism of action involves forming irreversible complexes with sterols in the protozoal cell membrane, which disrupts membrane integrity and leads to cell lysis and death, a process known as "defaunation"[20,21]. Since protozoa are major predators of bacteria in the rumen, their removal can reduce the engulfment of beneficial bacteria, potentially increasing the efficiency of microbial protein synthesis and its flow to the lower digestive tract. More importantly, protozoa have a symbiotic relationship with methanogenic archaea and are one of the main providers of the hydrogen (H2) required by methanogens. Therefore, inhibiting protozoa can indirectly reduce substrate supply, thereby lowering methane production[36,37,38].
- Role of Organosulfur Compounds: Organosulfur compounds (OSCs) from Allium plants like garlic and onion have been shown to have direct inhibitory activity against methanogenic archaea[15]. In vitro studies have shown that additives containing garlic extract can reduce methane production by 22% to 54% (These values are primarily derived from in vitro systems, and in vivo responses in sheep may differ). These compounds may act by interfering with key enzyme systems in the methanogenesis process.
- Effects on Fermentation Patterns: By selectively inhibiting certain microbial populations, Allium plant extracts can alter the production pattern of volatile fatty acids (VFAs). Several studies have reported that adding Allium plant extracts or their active components can lower the acetate-to-propionate ratio and increase the molar concentration of propionate. Propionate is the main precursor for gluconeogenesis, and an increase in its proportion means higher efficiency in converting feed energy into usable energy for the host. However, it is worth noting that high doses of additives can sometimes inhibit total VFA production or fiber digestibility, indicating that dose optimization is essential and that responses can be context-dependent[39,40].
4.2. Role of Black Soldier Fly Fatty Acids in the Rumen Ecosystem
- Mechanism of Action: As a medium-chain fatty acid, lauric acid also exerts strong antimicrobial effects in the rumen. It inhibits protozoa and some bacteria (especially Gram-positive bacteria) by disrupting the physical barrier of the microbial cell membrane[34].
- Methane Reduction: Lauric acid has been investigated as a rumen modifier with potential to reduce methane in some settings; however, reported responses vary by experimental model, basal diet, and dose, and consistent methane reduction is not universally observed in vivo. A meta-analysis of multiple studies confirmed that the addition of lauric acid significantly reduces in vitro methane production, mainly due to its inhibitory effect on methanogen and protozoa populations[37,53]. Because robust respiration-chamber measurements in sheep remain limited across published BSFL-FA studies, specific in vivo reduction magnitudes should be stated cautiously unless directly supported by sheep data[41].
- Effects on Fermentation Patterns: The effect of lauric acid on VFAs is clearly dose-dependent. At low to moderate inclusion levels, black soldier fly fat may increase total VFA concentration and the proportion of propionate in some in vitro systems; however, higher inclusion levels can inhibit fiber-degrading microbes and reduce total VFA production and dry matter digestibility, indicating a narrow and context-dependent dose window[42,43,44]. This phenomenon reveals that in practical applications, the dosage must be precisely controlled to find a balance between methane reduction and maintaining efficient fermentation (Table 3).
4.3. Downstream Effects on Host Metabolism and Product Quality
- Meat/Milk Quality: The biohydrogenation of dietary unsaturated fatty acids by rumen microbes determines the types of fatty acids ultimately absorbed by the small intestine. By modulating rumen microbes with additives, the biohydrogenation pathways can be altered, thereby affecting the fatty acid composition in tissues (muscle, fat) and milk[45]. For example, adding black soldier fly larvae to the diet of monogastric animals (poultry) resulted in a several-fold increase in the lauric and myristic acid content of their meat[46]. However, because medium-chain fatty acids (e.g., C12:0) in ruminants are transported via the portal vein and are preferentially oxidized in the liver rather than deposited in tissues, the extent of C12:0 enrichment in lamb or milk fat is limited and unlikely to reach the several-fold increases reported for monogastric species. Instead, BSFL-FA may modulate rumen fermentation and biohydrogenation patterns; however, meaningful enrichment of MUFAs/PUFAs in ruminant products generally requires co-supplementation with unsaturated fat sources or protection strategies.
- Muscle Growth: As some research suggests that flavonoids may play a role in muscle cell development, the flavonoid content of chives could potentially contribute to improved meat production, though this requires direct verification in livestock[47].
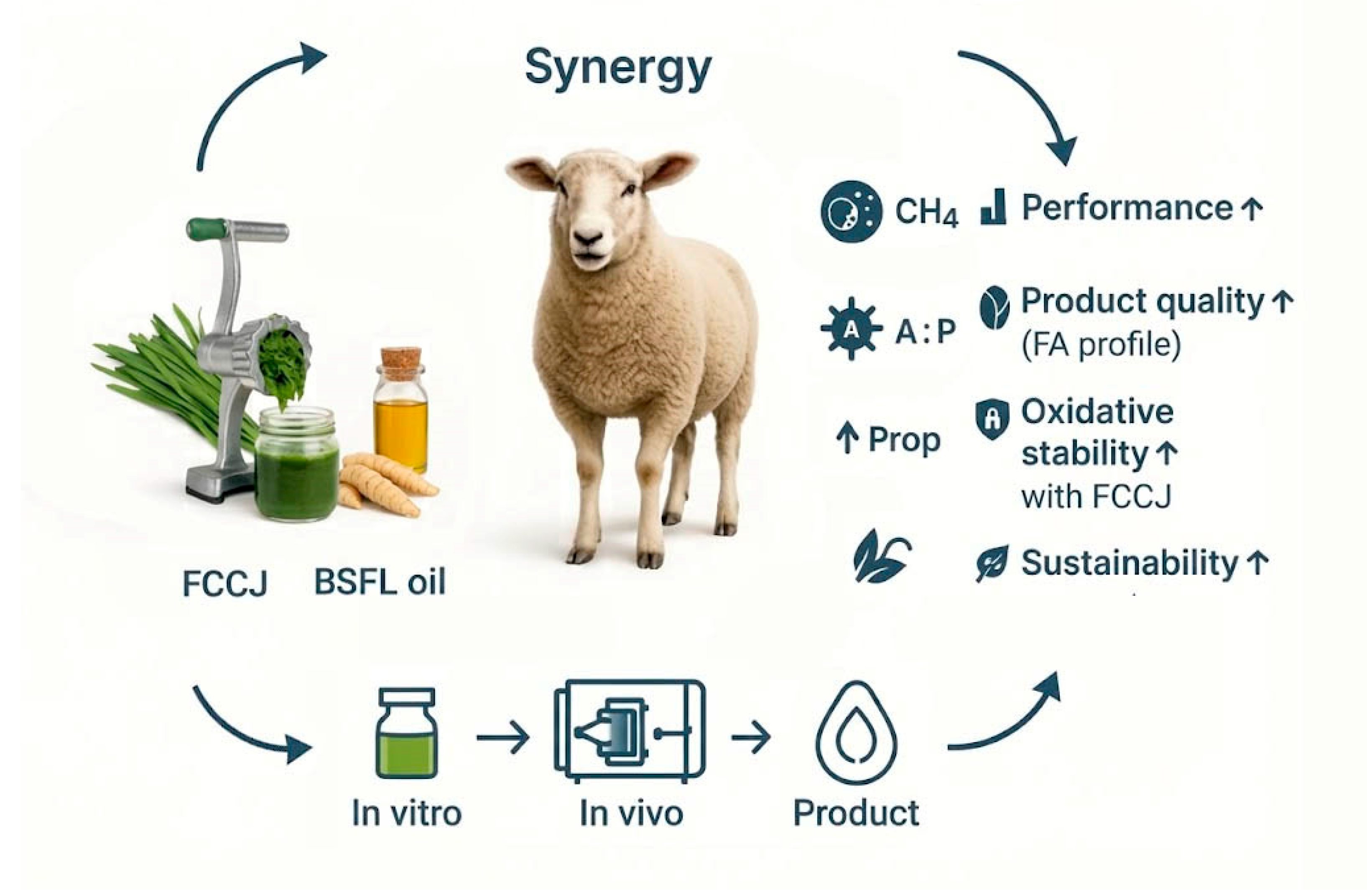
5. Hypothesized Complementary Interactions: Combining Fermented Chinese Chive Juice and Black Soldier Fly–Derived Fatty Acids
5.1. Potential Mechanisms for Complementary Regulation of Rumen Microecology
- Complementary Antimicrobial Spectrum: FCCJ provides a complex "cocktail" of antimicrobial compounds, including organosulfur compounds, flavonoids, saponins, and organic acids produced during fermentation. In contrast, BSFL-FA provides a high concentration of a single potent antimicrobial substance—lauric acid. The active substances from these two sources have different chemical structures and modes of action (e.g., enzyme inhibition vs. cell membrane disruption), potentially targeting different microbial populations. This could create a broader and more robust antimicrobial spectrum than any single component, more effectively inhibiting harmful or inefficient microbes in the rumen.
-
Multi-pronged Methane Reduction Strategy: This combination presents a multi-faceted attack on the methanogenesis process, which may be more effective and less prone to microbial adaptation than single-mechanism inhibitors.
- Dual Defaunation Effect: The saponins in FCCJ and the lauric acid in BSFL-FA both effectively inhibit protozoa, cutting off a major hydrogen source for methanogens from two angles.
- Direct Inhibition of Methanogens: The organosulfur compounds in FCCJ and the lauric acid in BSFL-FA can directly act on methanogenic archaea through different mechanisms to inhibit their activity.
- Competition for Hydrogen Utilization: Both additives are likely to promote the production of propionate, and the synthesis of propionate is itself a hydrogen-consuming pathway. This creates competition for metabolic hydrogen with the methanogenesis process, thereby directing hydrogen towards a pathway more beneficial for the host's energy utilization.
5.2. Potential Complementary Effects on Health and Product Quality
- Potential Antioxidant-Mediated Protection of Lipids: This is a plausible complementary mechanism that warrants validation. Given its lauric-acid–rich and largely saturated profile, BSFL-FA is more likely to increase the contribution of medium-chain saturated fatty acids (e.g., C12:0, C14:0) to the absorbed lipid pool. If the goal is to enrich MUFAs/PUFAs in tissues or milk, co-supplementation with unsaturated fat sources (e.g., flaxseed or fish oil) or protection strategies is generally required. In that context, the potent antioxidants in FCCJ may help protect these more oxidation-prone PUFAs by improving oxidative stability of meat and milk fat[48]. At the same time, the potent antioxidants like flavonoids in FCCJ, after being absorbed by the animal, can exert systemic antioxidant effects in the body. Thus, a "protection and enhancement" model is formed: BSFL-FA is responsible for optimizing the fatty acid profile, while the antioxidants from FCCJ protect these newly formed, more easily oxidized valuable fatty acids. This potential interaction may go beyond a simple addition in the rumen and directly affects the economic value and consumer acceptance of the final product.
- Potential Promotion of Muscle Growth and Meat Quality: FCCJ's flavonoids may promote muscle cell growth, while BSFL-FA can influence intramuscular fat deposition, which is crucial for meat tenderness and flavor. Combining these effects may contribute to improvements in muscle development and meat quality; however, this remains speculative without controlled in vivo validation.
- Enhanced Health and Immune Function: The anti-inflammatory and antioxidant properties of FCCJ, combined with the antimicrobial activity of both additives, can provide a comprehensive health support system for the animals, potentially enhancing the resistance of sheep to environmental stress (such as weaning, transport) and diseases.
6. Conclusion and Future Research Perspectives
6.1. Evidence Synthesis and Hypothesis Confirmation
6.2. Key Knowledge Gaps and Future Research Directions
- Lack of Direct In Vivo Validation: Currently, systematic in vivo studies on directly feeding Chinese chive (fermented or not) to ruminants like sheep are extremely scarce. Its efficacy in ruminants is mainly inferred from knowledge of its active components and from poultry research. This is the weakest link in the current theoretical framework.
- Dose-Effect Relationship and Optimal Ratio: Both FCCJ and BSFL-FA are complex mixtures, and the interactions between their components (additive, synergistic, antagonistic, or unrelated) are difficult to predict. Determining the optimal dosage and ratio for both individual and combined use is key to achieving the desired effects and avoiding potential negative impacts at high doses (such as inhibiting digestibility).
- Long-term Effects and Adaptation: Long-term feeding trials covering the entire production cycle are needed to assess the persistence of their effects and to determine whether rumen microbes will adapt to this composite additive.
- Diet Interaction Effects: The effectiveness of the additives may be significantly influenced by the composition of the basal diet (e.g., high-concentrate vs. high-forage diets), requiring research tailored to different feeding systems.
- Economic Feasibility Assessment: A comprehensive cost-benefit analysis must be conducted, considering the costs of raw material acquisition and processing (Chinese chive fermentation, black soldier fly farming, and oil extraction) against the economic benefits of practical application (e.g., weight gain, feed conversion ratio, product added value).
6.3. Roadmap for Product Development and In Vivo Validation
- Step One: In Vitro Screening and Preliminary Mechanism Exploration: Use rumen simulation techniques (such as the RUSITEC system) to screen combinations of FCCJ and BSFL fat (or its purified oil) at different ratios and dosages. By measuring methane production, VFA profiles, ammonia-N concentration, and the abundance of key microbes (such as protozoa, methanogens, and fiber-degrading bacteria), the synergistic mechanisms can be initially verified and promising formulations identified.
-
Step Two: Systematic In Vivo Feeding Trials: Use sheep as the model animal to design rigorously controlled in vivo feeding trials. Comprehensively evaluate the combined effects of the composite additive on the following indicators:
- Production Performance: Feed intake, average daily gain, feed conversion ratio.
- Rumen Fermentation and Digestion: Rumen fluid pH, VFAs, ammonia-N, microbial community composition, apparent nutrient digestibility.
- Environmental Impact: Accurately measure methane emissions using respiration chambers.
- Health Status: Blood biochemical parameters, immune and antioxidant indicators.
- Product Quality: Detailed analysis of the fatty acid and amino acid profiles of lamb and sheep's milk, assessment of meat quality parameters such as tenderness and color, and oxidative stability (shelf-life) tests.
- Step Three: Technology Optimization and Industrial Application: Optimize the fermentation process for Chinese chive and the farming and oil extraction techniques for black soldier flies to reduce costs and improve the yield and stability of active components. Develop standardized composite additive products with stable properties that are easy to store and feed (e.g., powders, granules, microcapsules). Strengthen collaboration with feed companies and farms to promote the translation and application of research findings.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zapata, O.; Cervantes, A.; Barreras, A.; Monge-Navarro, F.; González-Vizcarra, V.M.; Estrada-Angulo, A.; Urías-Estrada, J.D.; Corona, L.; Zinn, R.A.; Martínez-Alvarez, I.G. Effects of Single or Combined Supplementation of Probiotics and Prebiotics on Ruminal Fermentation, Ruminal Bacteria and Total Tract Digestion in Lambs. Small Ruminant Research 2021, 204, 106538. [Google Scholar] [CrossRef]
- Ren, H.; Vahjen, W.; Dadi, T.; Saliu, E.-M.; Boroojeni, F.G.; Zentek, J. Synergistic Effects of Probiotics and Phytobiotics on the Intestinal Microbiota in Young Broiler Chicken. Microorganisms 2019, 7, 684. [Google Scholar] [CrossRef]
- Tseten, T.; Sanjorjo, R.A.; Kwon, M.; Kim, S.-W. Strategies to Mitigate Enteric Methane Emissions from Ruminant Animals. Journal of Microbiology and Biotechnology 2022, 32, 269. [Google Scholar] [CrossRef]
- Ahmed, E.; Yano, R.; Fujimori, M.; Kand, D.; Hanada, M.; Nishida, T.; Fukuma, N. Impacts of Mootral on Methane Production, Rumen Fermentation, and Microbial Community in an in Vitro Study. Frontiers in veterinary science 2021, 7, 623817. [Google Scholar] [CrossRef] [PubMed]
- Barbu, I.A.; Ciorîță, A.; Carpa, R.; Moț, A.C.; Butiuc-Keul, A.; Pârvu, M. Phytochemical Characterization and Antimicrobial Activity of Several Allium Extracts. Molecules 2023, 28, 3980. [Google Scholar] [CrossRef]
- Mandey, J.S.; Sompie, F.N. Phytogenic Feed Additives as An Alternative to Antibiotic Growth Promoters in Poultry Nutrition. In Advanced Studies in the 21st Century Animal Nutrition; Babinszky, L., Oliveira, J., Santos, E.M., Eds.; IntechOpen: Rijeka, 2021; ISBN 978-1-83969-404-2. [Google Scholar]
- Yabuuchi, Y.; Tani, M.; Matsushita, Y.; Otsuka, H.; Kobayashi, Y. Effects of Lauric Acid on Physical, Chemical and Microbial Characteristics in the Rumen of Steers on a High Grain Diet. Animal Science Journal 2007, 78, 387–394. [Google Scholar] [CrossRef]
- Gadzama, I.U. Black Soldier Fly Larvae as Animal Feed. Bulgarian Journal of Animal Husbandry/Životnov Dni Nauki 2025, 62. [Google Scholar] [CrossRef]
- Embaby, M.G.; Günal, M.; AbuGhazaleh, A. Effect of Rumen Culture Adaptation to Origanum Vulgare L. Essential Oil on Rumen Methane and Fermentation. American Journal of Animal and Veterinary Sciences 2019, 14. [Google Scholar] [CrossRef]
- Foggi, G.; Terranova, M.; Conte, G.; Mantino, A.; Amelchanka, S.L.; Kreuzer, M.; Mele, M. In Vitro Screening of the Ruminal Methane and Ammonia Mitigating Potential of Mixtures of Either Chestnut or Quebracho Tannins with Blends of Essential Oils as Feed Additives. Italian Journal of Animal Science 2022, 21, 1520–1532. [Google Scholar] [CrossRef]
- Palangi, V.; Lackner, M. Management of Enteric Methane Emissions in Ruminants Using Feed Additives: A Review. Animals 2022, 12, 3452. [Google Scholar] [CrossRef]
- Abd El Tawab, A.M.; Liu, Q.; Xu, G.; Han, X. Feed Additives Strategies to Control Methanogenesis in Ruminants, Review. Archiva Zootechnica 2024, 27, 90–125. [Google Scholar] [CrossRef]
- Gao, Q.; Li, X.-B.; Sun, J.; Xia, E.-D.; Tang, F.; Cao, H.-Q.; Xun, H. Isolation and Identification of New Chemical Constituents from Chinese Chive (Allium Tuberosum) and Toxicological Evaluation of Raw and Cooked Chinese Chive. Food and Chemical Toxicology 2018, 112, 400–411. [Google Scholar] [CrossRef]
- Kalasee, B.; Mittraparp-arthorn, P. Phytochemicals, Antioxidant, and Antibacterial Activities of Fresh and Dried Chinese Chive (Allium Tuberosum Rottler) Leaf Extracts. ASEAN Journal of Scientific and Technological Reports 2023, 26, 1–10. [Google Scholar] [CrossRef]
- Cascajosa-Lira, A.; Andreo-Martinez, P.; Prieto, A.I.; Banos, A.; Guillamon, E.; Jos, A.; Camean, A.M. In Vitro Toxicity Studies of Bioactive Organosulfur Compounds from Allium Spp. with Potential Application in the Agri-Food Industry: A Review. Foods 2022, 11, 2620. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Cai, J.; Ren, Y.; Xu, Y.; Liu, H.; Zhao, Y.; Chen, X.; Liu, Z. Antimicrobial Activity, Chemical Composition and Mechanism of Action of Chinese Chive (Allium Tuberosum Rottler) Extracts. Frontiers in Microbiology 2022, 13, 1028627. [Google Scholar] [CrossRef]
- Chiryapkin, A.S.; Zolotykh, D.S.; Pozdnyakov, D.I. Review of the Biological Activity of Flavonoids: Quercetin and Kaempferol. Juvenis scientia 2023, 9, 5–20. [Google Scholar] [CrossRef]
- Dai, X.; Jia, C.; Lu, J.; Yu, Z. Metabolism of Phenolic Compounds and Antioxidant Activity in Different Tissue Parts of Post-Harvest Chive (Allium Schoenoprasum L.). Antioxidants 2024, 13, 279. [Google Scholar] [CrossRef] [PubMed]
- Khalid, N.; Ahmed, I.; Latif, M.S.Z.; Rafique, T.; Fawad, S.A. Comparison of Antimicrobial Activity, Phytochemical Profile and Minerals Composition of Garlic Allium Sativum and Allium Tuberosum. Journal of the Korean Society for Applied Biological Chemistry 2014, 57, 311–317. [Google Scholar] [CrossRef]
- Wina, E.; Muetzel, S.; Becker, K. The Impact of Saponins or Saponin-Containing Plant Materials on Ruminant Production A Review. Journal of agricultural and food chemistry 2005, 53, 8093–8105. [Google Scholar] [CrossRef]
- Addisu, S.; Assefa, A. Role of Plant Containing Saponin on Livestock Production; a Review. Advances in Biological Research 2016, 10, 309–314. [Google Scholar]
- Kocevski, D.; Du, M.; Kan, J.; Jing, C.; Lačanin, I.; Pavlović, H. Antifungal Effect of Allium Tuberosum, Cinnamomum Cassia, and Pogostemon Cablin Essential Oils and Their Components against Population of Aspergillus Species. Journal of food science 2013, 78, M731–M737. [Google Scholar] [CrossRef] [PubMed]
- Restuana, S.D.; Herla, R.; Dwi, S.; Dera, R.T.S.; Sisilia, F.Y. Antimicrobial Effects of Chive Extracts against Bacteria Pathogen and Lactobacillus Acidophilus. In Proceedings of the IOP Conference Series: Earth and Environmental Science; IOP Publishing, 2018; Vol. 205, p. 012049. [Google Scholar]
- Benfield, A.H.; Henriques, S.T. Mode-of-Action of Antimicrobial Peptides: Membrane Disruption vs. Intracellular Mechanisms. Frontiers in Medical Technology 2020, 2, 610997. [Google Scholar] [CrossRef]
- Dai, X.; Jia, C.; Lu, J.; Yu, Z. The Dynamics of Bioactive Compounds and Their Contributions to the Antioxidant Activity of Postharvest Chive (Allium Schoenoprasum L.). Food Research International 2023, 174, 113600. [Google Scholar] [CrossRef]
- Kothari, D.; Lee, W.-D.; Jung, E.S.; Niu, K.-M.; Lee, C.H.; Kim, S.-K. Controlled Fermentation Using Autochthonous Lactobacillus Plantarum Improves Antimicrobial Potential of Chinese Chives against Poultry Pathogens. Antibiotics 2020, 9, 386. [Google Scholar] [CrossRef]
- Lee, W.-D.; Kothari, D.; Moon, S.-G.; Kim, J.; Kim, K.-I.; Ga, G.-W.; Kim, Y.-G.; Kim, S.-K. Evaluation of Non-Fermented and Fermented Chinese Chive Juice as an Alternative to Antibiotic Growth Promoters of Broilers. Animals 2022, 12, 2742. [Google Scholar] [CrossRef]
- Sun, H.; Chen, D.; Cai, H.; Chang, W.; Wang, Z.; Liu, G.; Deng, X.; Chen, Z. Effects of Fermenting the Plant Fraction of a Complete Feed on the Growth Performance, Nutrient Utilization, Antioxidant Functions, Meat Quality, and Intestinal Microbiota of Broilers. Animals 2022, 12, 2870. [Google Scholar] [CrossRef]
- Dos Reis, J.H.; Gebert, R.R.; Fortuoso, B.F.; Dos Santos, D.S.; Souza, C.F.; Baldissera, M.D.; Tavernari, F. de C.; Boiago, M.M.; Paiano, D.; Da Silva, A.S. Selenomethionine as a Dietary Supplement for Laying Hens: Impacts on Lipid Peroxidation and Antioxidant Capacity in Fresh and Stored Eggs. Journal of Food Biochemistry 2019, 43, e12957. [Google Scholar] [CrossRef]
- Ewald, N.; Vidakovic, A.; Langeland, M.; Kiessling, A.; Sampels, S.; Lalander, C. Fatty Acid Composition of Black Soldier Fly Larvae (Hermetia Illucens)–Possibilities and Limitations for Modification through Diet. Waste management 2020, 102, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Cattaneo, A.; Meneguz, M.; Dabbou, S. The Fatty Acid Composition of Black Soldier Fly Larvae: The Influence of Feed Substrate and Applications in the Feed Industry. Journal of Insects as Food and Feed 2023, 10, 533–558. [Google Scholar] [CrossRef]
- Suryati, T.; Julaeha, E.; Farabi, K.; Ambarsari, H.; Hidayat, A.T. Lauric Acid from the Black Soldier Fly (Hermetia Illucens) and Its Potential Applications. Sustainability 2023, 15, 10383. [Google Scholar] [CrossRef]
- Addeo, N.F.; Tucciarone, I.; Bovera, F.; Vozzo, S.; Secci, G.; Parisi, G. Fatty Acid Profile of Black Soldier Fly Larvae and Frass as Affected by Different Growing Substrates. Journal of Insects as Food and Feed 2024, 10, 1437–1451. [Google Scholar] [CrossRef]
- Nitbani, F.O.; Tjitda, P.J.P.; Nitti, F.; Jumina, J.; Detha, A.I.R. Antimicrobial Properties of Lauric Acid and Monolaurin in Virgin Coconut Oil: A Review. ChemBioEng Reviews 2022, 9, 442–461. [Google Scholar] [CrossRef]
- Huang, L.; Gao, L.; Chen, C. Role of Medium-Chain Fatty Acids in Healthy Metabolism: A Clinical Perspective. Trends in Endocrinology & Metabolism 2021, 32, 351–366. [Google Scholar] [CrossRef]
- Luan, J.; Feng, X.; Yang, D.; Yang, M.; Zhou, J.; Geng, C. Effects of Medium-chain Fatty Acids (MCFAs) on in Vitro Rumen Fermentation, Methane Production, and Nutrient Digestibility under Low-and High-concentrate Diets. Animal Science Journal 2023, 94, e13818. [Google Scholar] [CrossRef]
- Yanza, Y.R.; Szumacher-Strabel, M.; Jayanegara, A.; Kasenta, A.M.; Gao, M.; Huang, H.; Patra, A.K.; Warzych, E.; Cieślak, A. The Effects of Dietary Medium-chain Fatty Acids on Ruminal Methanogenesis and Fermentation in Vitro and in Vivo: A Meta-analysis. Journal of animal physiology and animal nutrition 2021, 105, 874–889. [Google Scholar] [CrossRef] [PubMed]
- Park, T.; Mao, H.; Yu, Z. Inhibition of Rumen Protozoa by Specific Inhibitors of Lysozyme and Peptidases in Vitro. Frontiers in microbiology 2019, 10, 2822. [Google Scholar] [CrossRef] [PubMed]
- Kekana, M.R.; Luseba, D.; Muyu, M.C. Effects of Garlic Supplementation on in Vitro Nutrient Digestibility, Rumen Fermentation, and Gas Production. South African Journal of Animal Science 2021, 51, 271–279. [Google Scholar] [CrossRef]
- Salem, A.Z.M.; Ronquillo, M.; Camacho, L.M.; Cerrillo, S.M.A.; Domínguez, I.A.; Bórquez, J.L. Beneficial Effects of Plant Extracts in Ruminant Nutrition: A Review. Indian Journal of Animal Sciences 2012, 82, 1117–1121. [Google Scholar] [CrossRef]
- Prachumchai, R.; Suntara, C.; Kanakai, N.; Cherdthong, A. Inclusion of Black Soldier Fly Larval Oil in Ruminant Diets Influences Feed Consumption, Nutritional Digestibility, Ruminal Characteristics, and Methane Estimation in Thai-Indigenous Steers. Journal of Animal Physiology and Animal Nutrition 2025. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Chen, S.; Paengkoum, S.; Taethaisong, N.; Meethip, W.; Surakhunthod, J.; Wang, Q.; Thongpea, S.; Paengkoum, P. Effects of Black Soldier Fly (Hermetia Illucens L., BSF) Larvae Addition on in Vitro Fermentation Parameters of Goat Diets. Insects 2024, 15, 343. [Google Scholar] [CrossRef]
- Prachumchai, R.; Cherdthong, A. Black Soldier Fly Larva Oil in Diets with Roughage to Concentrate Ratios on Fermentation Characteristics, Degradability, and Methane Generation. Animals 2023, 13, 2416. [Google Scholar] [CrossRef]
- Anele, U.Y.; Crumel, X.; Olagunju, L.; Compart, D.P. Effects of Yucca Schidigera Based Feed Additive on in Vitro Dry Matter Digestibility, Efficiency of Microbial Production, and Greenhouse Gas Emissions of Four Dairy Diets. Dairy 2022, 3, 326–332. [Google Scholar] [CrossRef]
- Toral, P.G.; Monahan, F.J.; Hervás, G.; Frutos, P.; Moloney, A.P. Modulating Ruminal Lipid Metabolism to Improve the Fatty Acid Composition of Meat and Milk. Challenges and Opportunities. Animal 2018, 12, s272–s281. [Google Scholar] [CrossRef]
- Marín, A.L.M.; Gariglio, M.; Trocino, A.; Schiavone, A. Lauric Acid Content in Intramuscular Fat Is a Reliable Indicator of Black Soldier Fly Larvae Meal Consumption in Muscovy Ducks. Heliyon 2024, 10. [Google Scholar] [CrossRef]
- North, M.K.; Dalle Zotte, A.; Hoffman, L.C. The Use of Dietary Flavonoids in Meat Production: A Review. Animal Feed Science and Technology 2019, 257, 114291. [Google Scholar] [CrossRef]
- Gadeyne, F.; De Neve, N.; Vlaeminck, B.; Fievez, V. State of the Art in Rumen Lipid Protection Technologies and Emerging Interfacial Protein Cross-linking Methods. European Journal of Lipid Science and Technology 2017, 119, 1600345. [Google Scholar] [CrossRef]
- Putnik, P.; Gabrić, D.; Roohinejad, S.; Barba, F.J.; Granato, D.; Mallikarjunan, K.; Lorenzo, J.M.; Kovačević, D.B. An Overview of Organosulfur Compounds from Allium Spp.: From Processing and Preservation to Evaluation of Their Bioavailability, Antimicrobial, and Anti-Inflammatory Properties. Food chemistry 2019, 276, 680–691. [Google Scholar] [CrossRef]
- Marefati, N.; Ghorani, V.; Shakeri, F.; Boskabady, M.; Kianian, F.; Rezaee, R.; Boskabady, M.H. A Review of Anti-Inflammatory, Antioxidant, and Immunomodulatory Effects of Allium Cepa and Its Main Constituents. Pharmaceutical biology 2021, 59, 285–300. [Google Scholar] [CrossRef]
- Upadhyay, R.K. Nutritional and Therapeutic Potential of Allium Vegetables. J. Nutr. Ther 2017, 6, 18–37. [Google Scholar] [CrossRef]
- Xie, K.; Wang, Z.; Wang, Y.; Wang, C.; Chang, S.; Zhang, C.; Zhu, W.; Hou, F. Effects of Allium Mongolicum Regel Supplementation on the Digestibility, Methane Production, and Antioxidant Capacity of Simmental Calves in Northwest China. Animal Science Journal 2020, 91, e13392. [Google Scholar] [CrossRef] [PubMed]
- Martins, L.F.; Cueva, S.F.; Lage, C.F.A.; Ramin, M.; Silvestre, T.; Tricarico, J.; Hristov, A.N. A Meta-Analysis of Methane-Mitigation Potential of Feed Additives Evaluated in Vitro. Journal of dairy science 2024, 107, 288–300. [Google Scholar] [CrossRef]
| Compound Class | Specific Examples | Key Biological Activity | References |
|---|---|---|---|
| Organosulfur Compounds | Methiin, Isoalliin; methyl/1-propenyl thiosulfinates | Broad-spectrum antimicrobial, antiviral, anti-inflammatory | [49] |
| Polyphenols (Flavonoids) | Quercetin, Kaempferol | Strong antioxidant, free radical scavenging, anti-inflammatory, promotes muscle cell proliferation | [50] |
| Saponins | Steroidal/Triterpenoid Saponins | Antiprotozoal, cholesterol-lowering, immunomodulatory | [51] |
| Fatty Acid | Common Name | BSFL Fat (%) | Coconut Oil (%) | Palm Kernel Oil (%) |
|---|---|---|---|---|
| C12:0 | Lauric acid | 40 – 60 | 45 – 53 | 46 – 48 |
| C14:0 | Myristic acid | 6 – 10 | 16 – 21 | 15 – 17 |
| C16:0 | Palmitic acid | 12 – 22 | 7 – 10 | 8 – 9 |
| C18:1 | Oleic acid | 10 – 25 | 5 – 10 | 14 – 15 |
| C18:2 | Linoleic acid | 2.5 – 12.5 | 1 – 3 | 2 – 3 |
| Additive Category | Parameter | Experimental Model | Typical Dose / Concentration | Effect | Notes / Limitations | Key Refs |
|---|---|---|---|---|---|---|
| Allium Bioactives | Methane (CH₄) | In vitro (rumen batch culture: garlic juice/powder—fermentation & gas production); in vivo (Simmental calves: Allium mongolicum Regel) | In vitro: garlic juice 0.5–1.0 mL/100 mL (≈5–10 mL/L) and/or garlic powder (as tested); In vivo: 200–800 mg/kg BW/day | Reduced | In vivo CH₄ reduction demonstrated in calves; some in vitro Allium studies infer methane-mitigation potential from fermentation shifts, although direct CH₄ measurement is available in some gas-production systems. | [39,52] |
| Protozoa | Mechanistic + applied rumen evidence from saponin-containing plant materials (review-based synthesis) | Varies by saponin source/preparation (not standardized across studies) | Reduced | Defaunation mechanism is well-established for saponins; direct protozoa-count data for Chinese chive-derived preparations remain limited. | [20,21] | |
| Total VFA | In vitro (batch culture: garlic); in vivo (Simmental calves: Allium mongolicum Regel) | As above (in vitro garlic preparations; in vivo 200–800 mg/kg BW/day) | Variable / dose-dependent | Often maintained or increased at moderate inclusion; response depends on dose and basal diet. | [39,52] | |
| Acetate:Propionate | In vitro (batch culture: garlic); in vivo (Simmental calves: Allium mongolicum Regel) | As above | Reduced | Consistent with a shift of H₂ use toward propionate as an alternative sink. | [39,52] | |
| Ammonia-N | In vitro (batch culture: garlic preparations) | Garlic powder or garlic juice 0.5 mL/100 mL (per study designs) | Reduced | Ammonia reduction is reported in vitro with garlic preparations; in vivo ammonia response for Allium mongolicum is not explicit in the abstract. | [39] | |
| Lauric Acid / BSFL Fat | Methane (CH₄) | Meta-analysis (MCFA; in vitro & in vivo) + in vitro gas production (BSFL oil; BSF larvae meal) + in vivo (Thai-indigenous steers; methane estimation) | Oil: 1–4% DM (in vivo) / 2–6% DM (in vitro); Larvae meal: 5–15% DM (in vitro) | Reduced (consistent in vitro); variable (in vivo) | Meta-analysis shows CH₄ decreases with increasing MCFA dose; steer trial reports a linear decline in CH₄ estimate with 1–4% oil; diet and dose influence in vivo magnitude. | [37,41,42,43,53] |
| Protozoa | Meta-analysis (MCFA; in vitro & in vivo) and in vivo (Thai-indigenous steers) | Oil: 1–4% DM | Reduced | Protozoa respond negatively (dose-related) to MCFA; steer trial reports protozoa decreasing with increasing BSFL oil inclusion. | [37,41] | |
| Total VFA | In vivo (steers) + in vitro (goat-diet fermentation wi[37,41th larvae meal; BSFL oil with R:C ratios) | Oil: 1–4% DM (in vivo) / 2–6% DM (in vitro); Larvae meal: 5–15% DM (in vitro) | Dose-dependent | Moderate inclusion can maintain fermentation; higher larvae meal (e.g., 15% DM) may reduce CH₄ but also depress VFA in vitro. | [41,42,43] | |
| Acetate:Propionate | In vivo (steers) and in vitro (BSFL oil with R:C ratios) | Oil: 1–4% DM (in vivo) / 2–6% DM (in vitro) | Reduced / no effect | Often associated with higher propionate proportion; steers showed higher propionic acid at 2% oil in one sampling time. | [41,43] | |
| Dry Matter Digestibility | Meta-analysis (MCFA; digestibility outcomes) + in vivo (steers: BSFL oil) + in vivo (steers: lauric acid, high-grain diet) | Oil: 1–4% DM (in vivo; quadratic response); Lauric acid: high-grain steer model (dose per study); higher MCFA doses reduce fibre digestibility (meta-analysis) | Dose-dependent (can decrease at higher dose) | Meta-analysis reports fibre digestibility declines with increasing MCFA dose; steer trial with BSFL oil shows a quadratic DM/OM digestibility response; lauric acid evidence is from a high-grain steer model. | [7,37,41] |
| Target Area | Mechanism of Fermented Chinese Chive Juice (FCCJ) | Mechanism of Black Soldier Fly Fatty Acids (BSFL-FA) | Expected Outcome (Hypothesized) |
|---|---|---|---|
| Rumen Microecology | Provides a complex of antimicrobial substances (OSCs, flavonoids, saponins, and organic acids) to modulate the microbial community structure.† | Provides a high concentration of lauric acid, potently inhibiting Gram-positive bacteria and protozoa.† | A broader and more potent antimicrobial effect, effectively optimizing the rumen microbial ecosystem. |
| Methane Reduction | Saponins for defaunation†; OSCs may directly inhibit methanogens.‡ | Lauric acid for defaunation† and may directly inhibit methanogens.‡ | Potential additive (or synergistic) inhibition of methane production through multiple targets and pathways, resulting in a more lasting and significant effect. |
| Product Quality | Flavonoids may promote muscle growth‡; antioxidants protect tissues.† | Modulates rumen biohydrogenation†; may alter product fatty-acid composition and may require co-supplementation with unsaturated fat sources or protection strategies.‡ | Improved meat/milk quality via microbial modulation and antioxidant protection, including enhanced oxidative stability and potential nutritional value. |
| Animal Health | Antioxidant†, may exert anti-inflammatory‡ and immunomodulatory effects.‡ | Antimicrobial†, antiprotozoal†; may improve gut health.‡ | Comprehensive enhancement of the animal's stress resistance and disease resilience. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.





