Submitted:
16 January 2026
Posted:
16 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The importance of the Peripheral Nervous System Organization
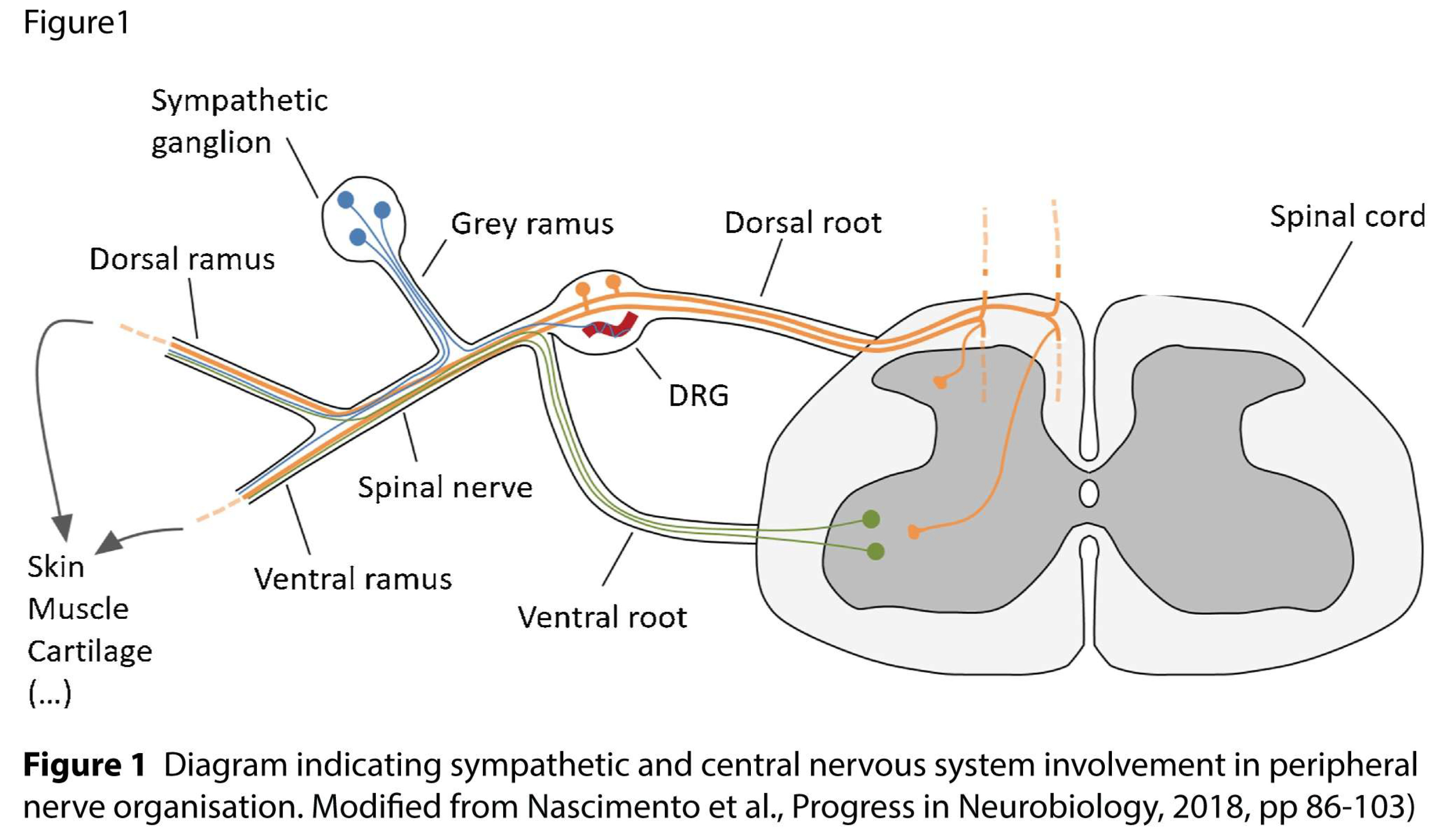
2.1. Dorsal Root Ganglia
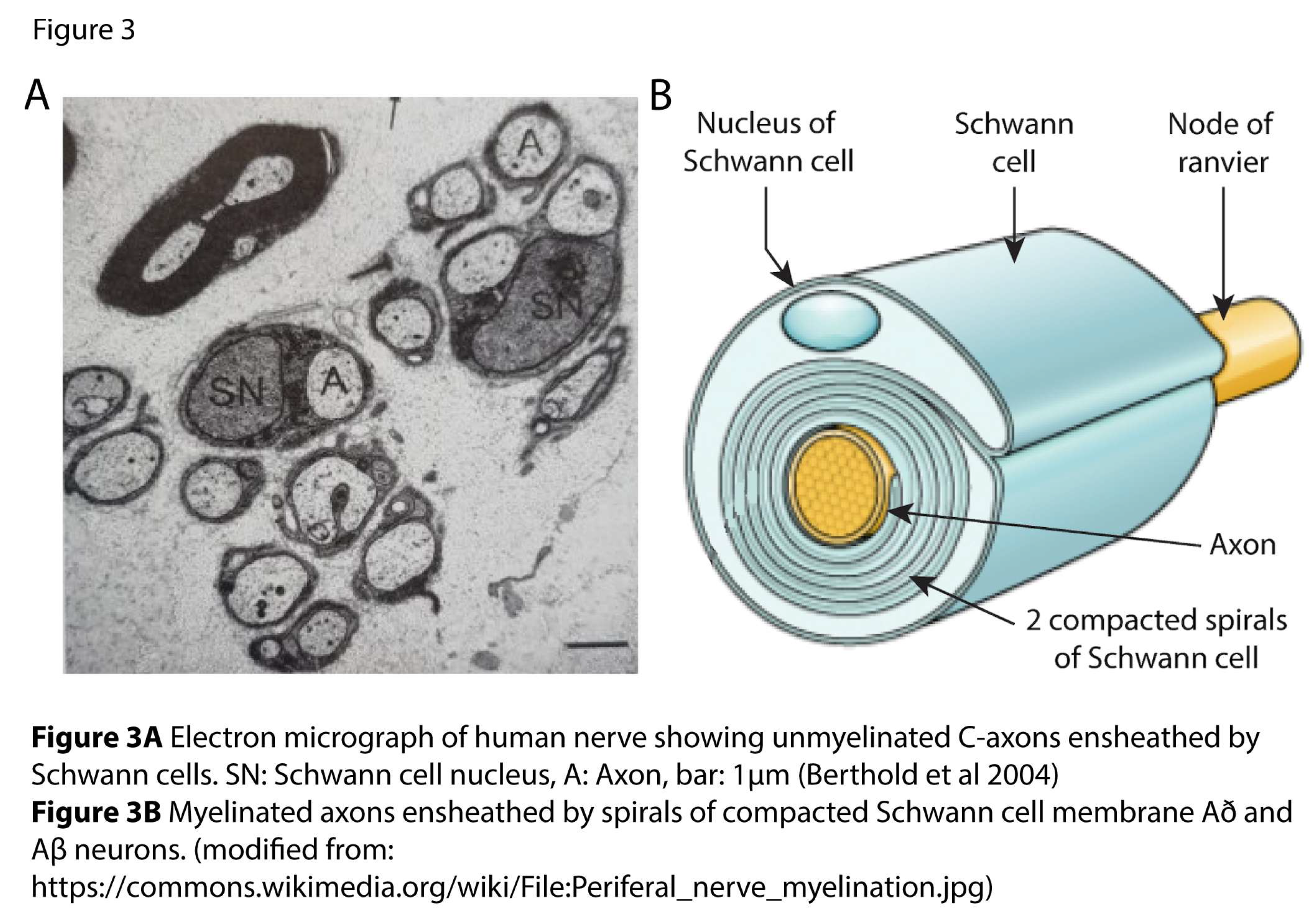
2.2. The Schwann Cell
3. Discussion of the Cytoskeleton
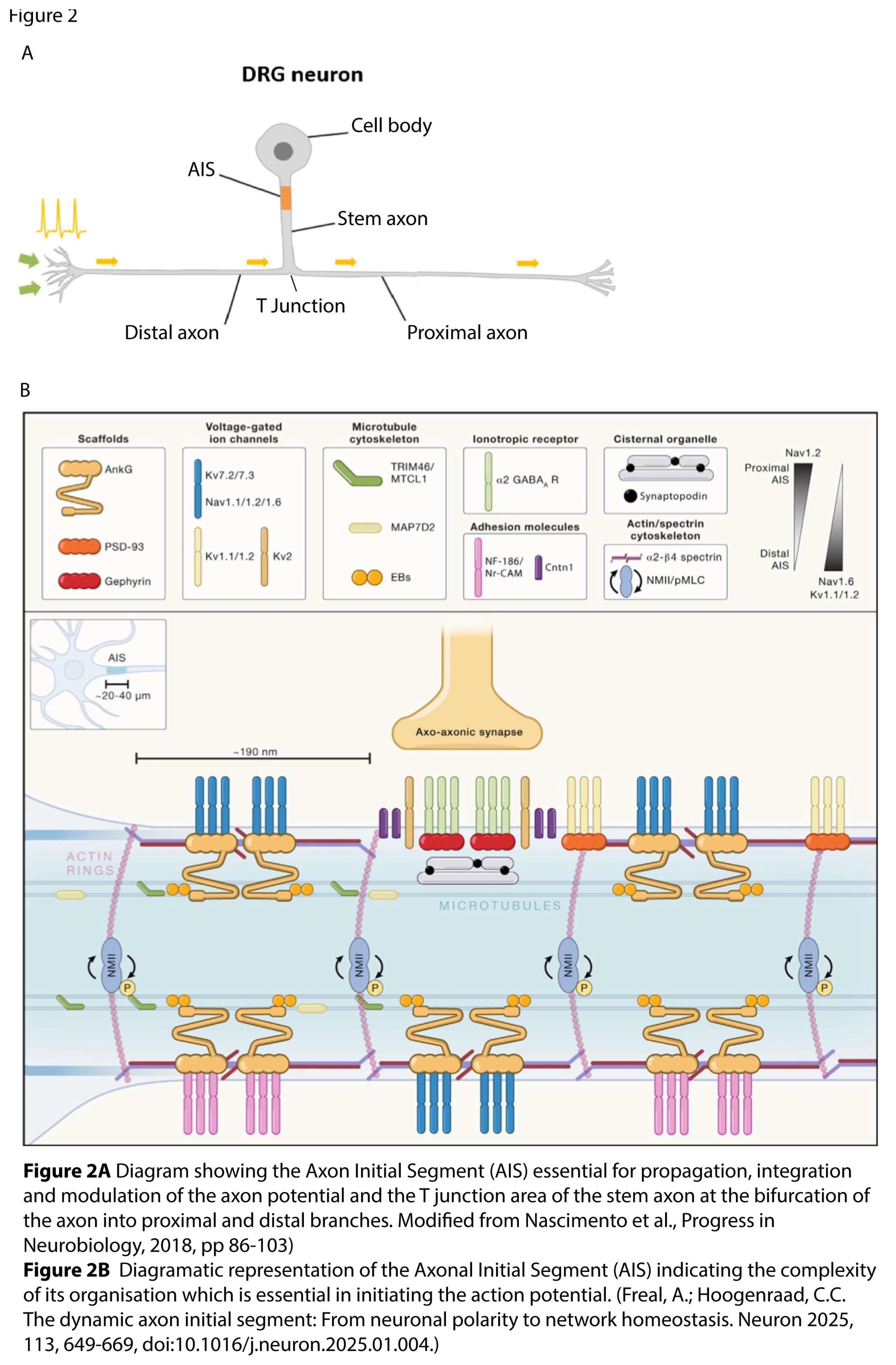
3.1. Axon Initial Segment
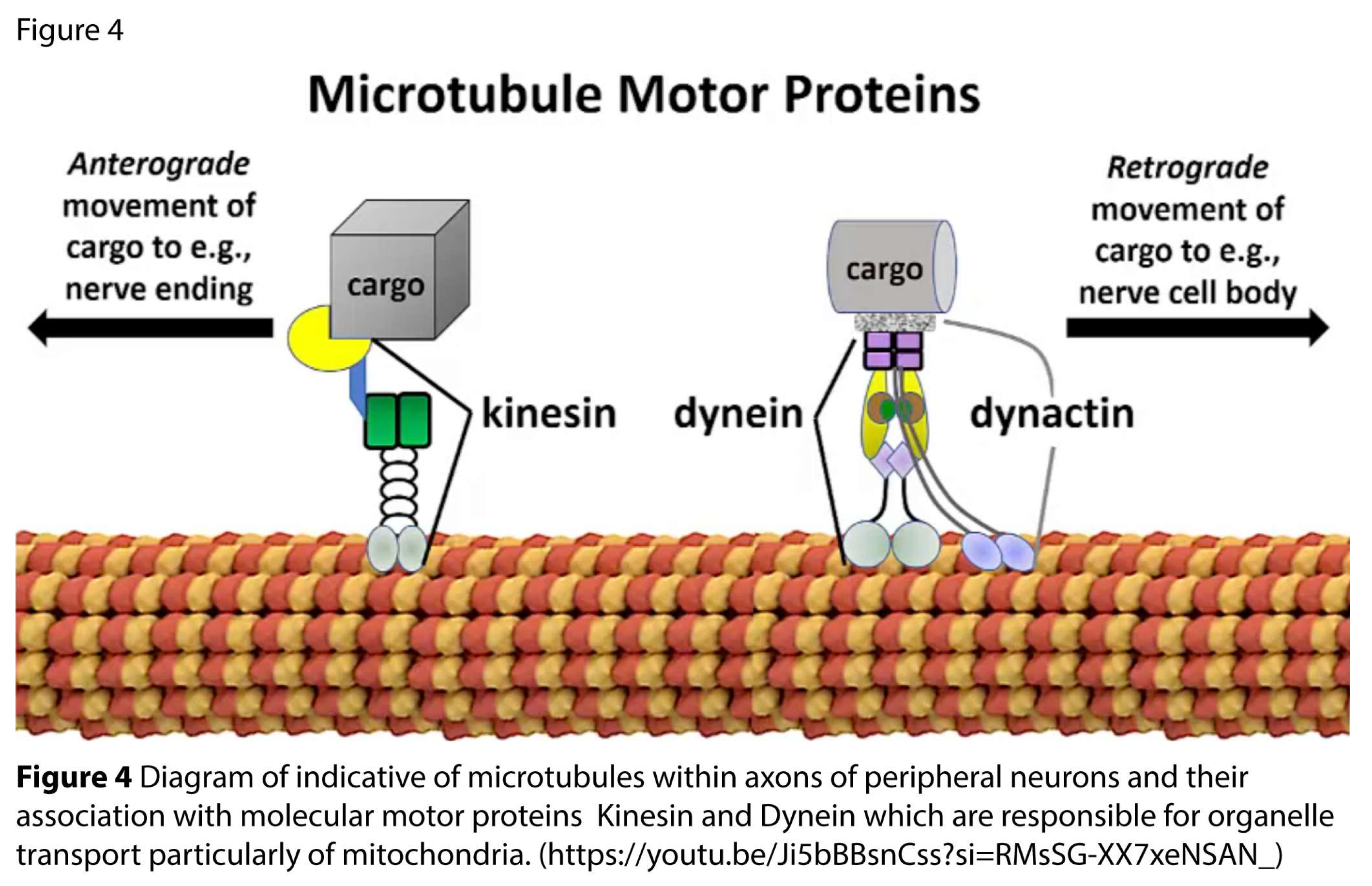
4.2. Fast Axonal Flow (FAF)
4. What Is Photobiomodulation?
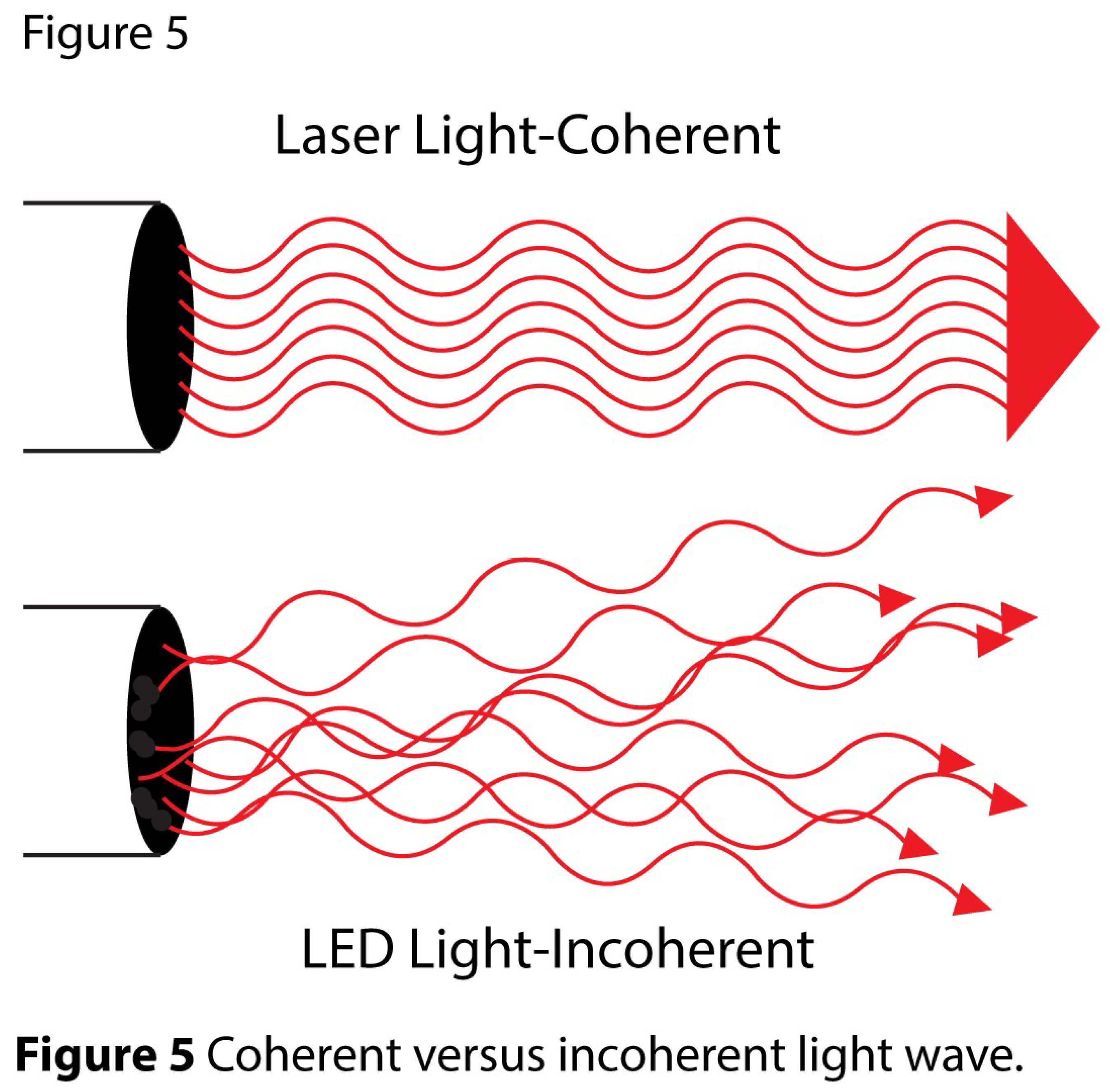
5. Experimental Studies of PBM
5.1. DRG Studies
5.1.1. Human DRG Study—In Vivo
5.1.2. Animal DRG Studies
5.1.3. Studies of DRG Neuron Cultures
5.1.4. Microtubule Depolymerization and Mitochondrial Membrane Potential (MMP)
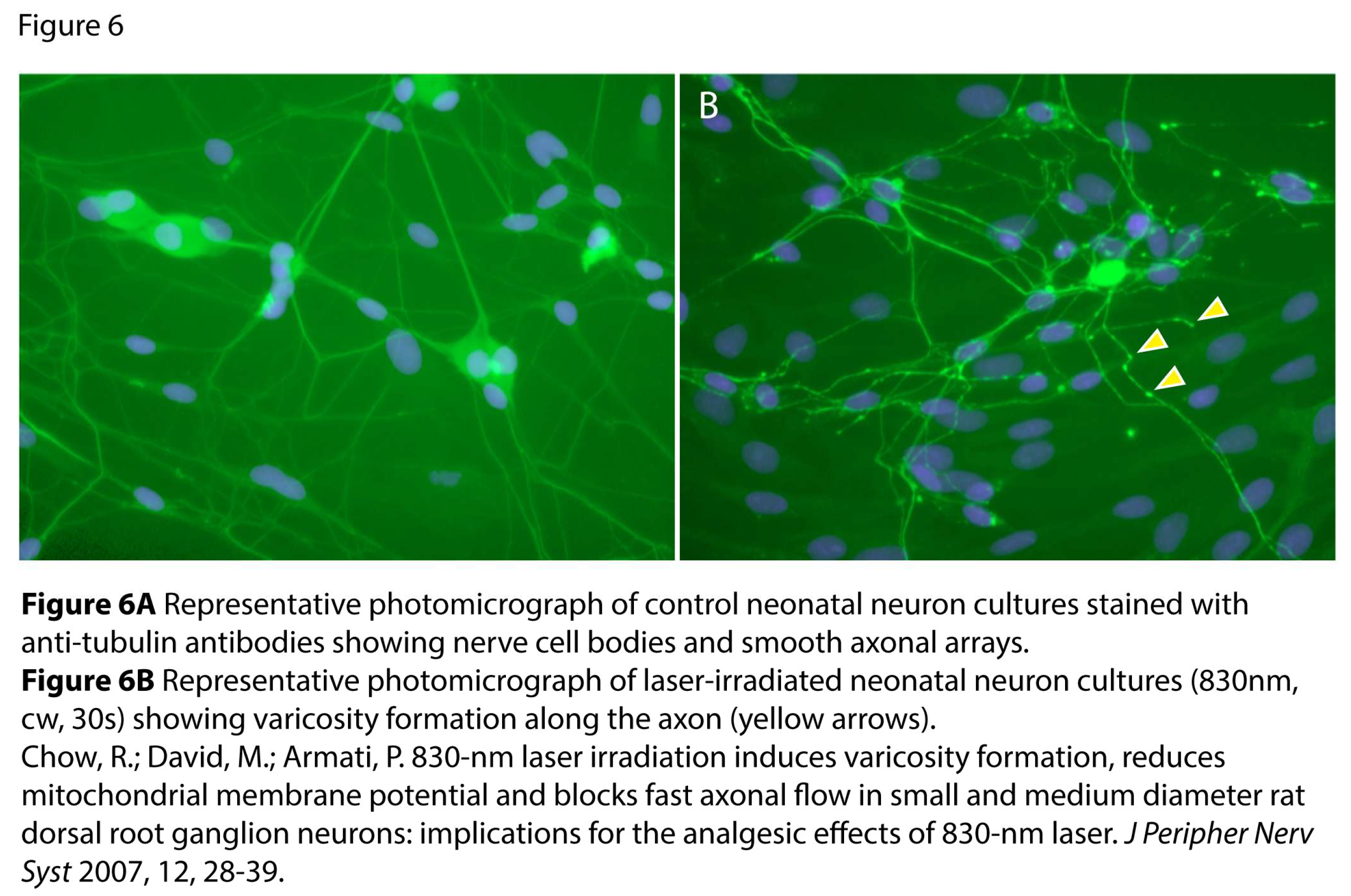
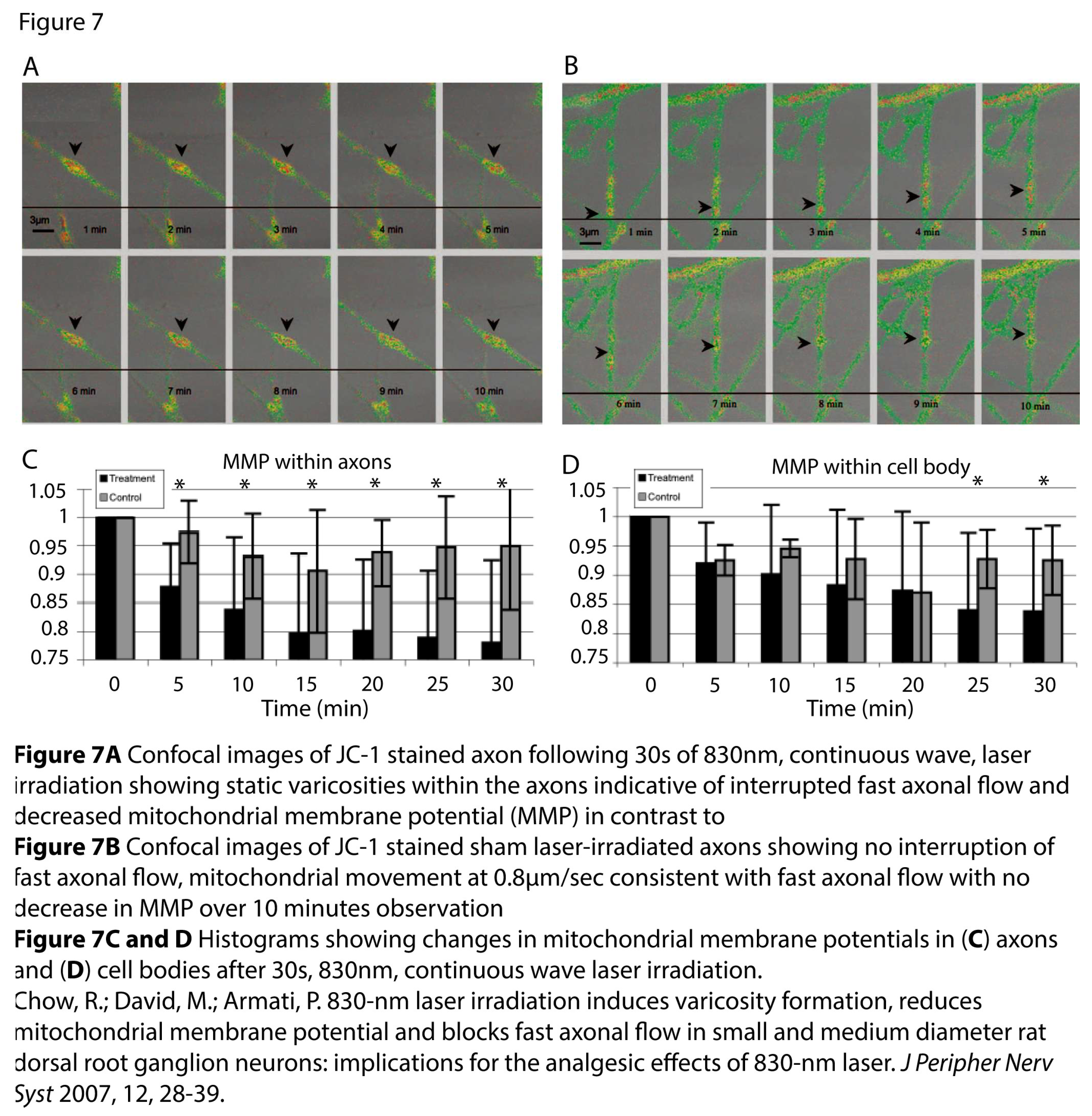
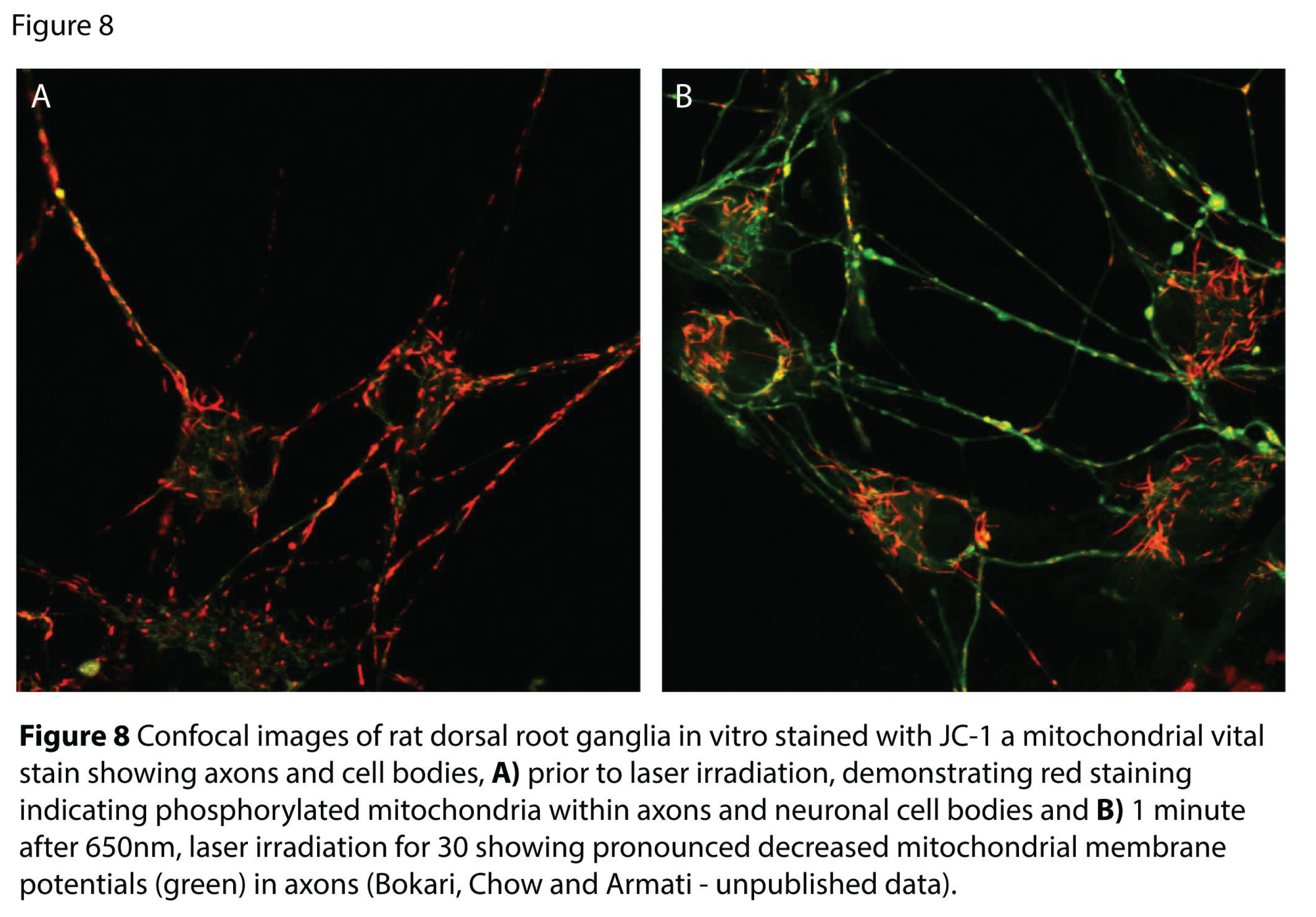
6. Depolarisation Blockade
7. PBM and Modulation of Action Potentials
7.1. PBM and Effects on Action Potentials
7.2. PBM-Induced Neural Blockade in Human Studies
7.2. PBM-Induced Neural Blockade in Animal Studies
8. Clinical Application of PBM
8.1. PBM in Medical Conditions
8.2. PBM in Dentistry
9. Discussion
9.1. The Cytoskeleton and Pharmacological Agents
9.2. Neuroplasticity and PBM
9.3. PBM in Prevention of Acute Pain Progressing to Chronic Pain
10. Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| DRG | Dorsal Root Ganglion |
| PBM | Photobiomodulation |
| AIS | Axon initial segment |
| LI | Laser irradiation |
| MMP | Mitochondrial membrane potential |
| FAF | Fast axonal flow |
References
- Hennessy, S.N.; Corcoran, G.D. Low-level laser therapy in osteoarthritic pain: A narrative review with an approach to integrated clinical use. Osteoarthr Cartil Open 2025, 7, 100685. [Google Scholar] [CrossRef]
- Khaleghi, A.; Salari, B.; Mafi, S.; Fekrazad, R. Photobiomodulation therapy in the management of myofascial pain dysfunction syndrome: A randomized triple-blind clinical trial. Photodiagnosis Photodyn Ther 2025, 56, 105239. [Google Scholar] [CrossRef] [PubMed]
- Cidral-Filho, F.; Donatello, N.N.; Lugtu, C.; Hewitson, A. Photobiomodulation on shoulder and neck pain and disability: A comprehensive review. Lasers Med Sci 2024, 39, 263. [Google Scholar] [CrossRef]
- Kulkarni, S.; Walsh, L.J.; Bhurani, Y.; George, R. Assessment of the onset of analgesia and length of analgesia following the use of PBM with different wavelengths: a clinical study. Lasers Med Sci 2024, 39, 236. [Google Scholar] [CrossRef]
- Liang, R.; George, R.; Walsh, L.J. Pulpal response following photo-biomodulation with a 904-nm diode laser: a double-blind clinical study. Lasers Med Sci 2016, 31, 1811–1817. [Google Scholar] [CrossRef]
- Nascimento, A.I.; Mar, F.M.; Sousa, M.M. The intriguing nature of dorsal root ganglion neurons: Linking structure with polarity and function. Prog Neurobiol 2018, 168, 86–103. [Google Scholar] [CrossRef] [PubMed]
- Fréal, A.; Hoogenraad, C.C. The dynamic axon initial segment: From neuronal polarity to network homeostasis. Neuron 2025, 113, 649–669. [Google Scholar] [CrossRef]
- Koike, T. Distinctive glial cells in the dorsal root ganglion: their morphology and functions. Anat Sci Int 2025, 100, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Moog, M.; Ballesteros, A.; Ernfors, P.; Swartz, K. Molecular and electrophysiological characterization of cutaneous Schwann cells. Proceedings of the Biophysical Society Annual Meeting 2025, 2025, 604a. [Google Scholar] [CrossRef]
- Mokoena, D.R.; Houreld, N.N.; Dhilip Kumar, S.S.; Abrahamse, H. Photobiomodulation at 660 nm Stimulates Fibroblast Differentiation. Lasers Surg Med 2020, 52, 671–681. [Google Scholar] [CrossRef]
- Etemadi, A.; Sadatmansouri, S.; Sodeif, F.; Jalalishirazi, F.; Chiniforush, N. Photobiomodulation Effect of Different Diode Wavelengths on the Proliferation of Human Gingival Fibroblast Cells. Photochem Photobiol 2021, 97, 1123–1128. [Google Scholar] [CrossRef]
- Shaikh-Kader, A.; Houreld, N.N.; Rajendran, N.K.; Abrahamse, H. Levels of Cyclooxygenase 2, Interleukin-6, and Tumour Necrosis Factor-alpha in Fibroblast Cell Culture Models after Photobiomodulation at 660 nm. Oxid Med Cell Longev 2021, 2021, 6667812. [Google Scholar] [CrossRef]
- y Cajal, S. Degeneration & regeneration of the nervous system; Hafner Publishing Company, 1959; Volume 1. [Google Scholar]
- Colom, B.; Poitelon, Y.; Huang, W.; Woodfin, A.; Averill, S.; Del Carro, U.; Zambroni, D.; Brain, S.D.; Perretti, M.; Ahluwalia, A.; et al. Schwann cell-specific JAM-C-deficient mice reveal novel expression and functions for JAM-C in peripheral nerves. Faseb j 2012, 26, 1064–1076. [Google Scholar] [CrossRef] [PubMed]
- Sundt, D.; Gamper, N.; Jaffe, D.B. Spike propagation through the dorsal root ganglia in an unmyelinated sensory neuron: a modeling study. J Neurophysiol 2015, 114, 3140–3153. [Google Scholar] [CrossRef] [PubMed]
- Costa, A.C.; Murillo, B.R.; Bessa, R.; Ribeiro, R.; Ferreira da Silva, T.; Porfírio-Rodrigues, P.; Martins, G.G.; Brites, P.; Kneussel, M.; Misgeld, T.; et al. Axon-specific microtubule regulation drives asymmetric regeneration of sensory neuron axons. Elife 2025, 13. [Google Scholar] [CrossRef]
- Vogl, A.W.; Lillie, M.A.; Piscitelli, M.A.; Goldbogen, J.A.; Pyenson, N.D.; Shadwick, R.E. Stretchy nerves are an essential component of the extreme feeding mechanism of rorqual whales. Curr Biol 2015, 25, R360–361. [Google Scholar] [CrossRef]
- Scherer, S.S.; Arroyo, E.J. Recent progress on the molecular organization of myelinated axons. J Peripher Nerv Syst 2002, 7, 1–12. [Google Scholar] [CrossRef]
- Purves, D.; Augustine, G.; Fitzpatrick, D.; Katz, L.; LaMantia, A.; McNamara, J.; Williams, S. Projections from the Cerebellum. In Neuroscience; Sinauer Associates, 2001. [Google Scholar]
- Hanani, M.; Spray, D.C. Emerging importance of satellite glia in nervous system function and dysfunction. Nature reviews 2020, 21, 485–498. [Google Scholar] [CrossRef]
- Jessen, K.R.; Mirsky, R. The Success and Failure of the Schwann Cell Response to Nerve Injury. Front Cell Neurosci 2019, 13, 33. [Google Scholar] [CrossRef]
- Kastriti, M.E.; Faure, L.; Von Ahsen, D.; Bouderlique, T.G.; Boström, J.; Solovieva, T.; Jackson, C.; Bronner, M.; Meijer, D.; Hadjab, S.; et al. Schwann cell precursors represent a neural crest-like state with biased multipotency. Embo j 2022, 41, e108780. [Google Scholar] [CrossRef]
- Furlan, A.; Adameyko, I. Schwann cell precursor: a neural crest cell in disguise? Dev Biol 2018, 444 Suppl 1, S25–s35. [Google Scholar] [CrossRef]
- Abdo, H.; Calvo-Enrique, L.; Lopez, J.M.; Song, J.; Zhang, M.D.; Usoskin, D.; El Manira, A.; Adameyko, I.; Hjerling-Leffler, J.; Ernfors, P. Specialized cutaneous Schwann cells initiate pain sensation. Science 2019, 365, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Freal, A.; Hoogenraad, C.C. Neuronal Cytoskeleton: Presynaptic Boutons as Hotspots for Activity-Dependent Microtubule Nucleation. Curr Biol 2019, 29, R1307–R1309. [Google Scholar] [CrossRef]
- Pellegrini, L.; Wetzel, A.; Granno, S.; Heaton, G.; Harvey, K. Back to the tubule: microtubule dynamics in Parkinson’s disease. Cell Mol Life Sci 2017, 74, 409–434. [Google Scholar] [CrossRef]
- Koltzenburg, M.; Bennett, D.L.; Shelton, D.L.; McMahon, S.B. Neutralization of endogenous NGF prevents the sensitization of nociceptors supplying inflamed skin. The European journal of neuroscience 1999, 11, 1698–1704. [Google Scholar] [CrossRef]
- Anders, J.J.; Lanzafame, R.J.; Arany, P.R. Low-level light/laser therapy versus photobiomodulation therapy. Photomed Laser Surg 2015, 33, 183–184. [Google Scholar] [CrossRef]
- Lam, T.S.; Abergel, R.P.; Meeker, C.A.; Castel, J.C.; Dwyer, R.M.; Uitto, J. Laser Stimulation of Collagen Synthesis in Human Skin Fibroblasts. Lasers in Life Sciences 1986, 1, 61–77. [Google Scholar]
- Young, S.; Dyson, M.; Bolton, P. Effect of light on calcium uptake by macrophages. Laser Therapy 1990, 2, 53–57. [Google Scholar] [CrossRef]
- Honmura, A.; Yanase, M.; Obata, J.; Haruki, E. Therapeutic effect on GaAlAs Diode Laser Irradiation on Experimentally Induced Inflammation in Rats. Lasers in Surgery & Medicine 1992, 12, 441–449. [Google Scholar]
- Holanda, V.M.; Chavantes, M.C.; Wu, X.; Anders, J.J. The mechanistic basis for photobiomodulation therapy of neuropathic pain by near infrared laser light. Lasers Surg Med 2017, 49, 516–524. [Google Scholar] [CrossRef]
- Chen, Y.-J.; Wang, Y.-H.; Wang, C.-Z.; Ho, M.-L.; Kuo, P.-L.; Huang, M.-H.; Chen, C.-H. Effect of Low Level Laser Therapy on Chronic Compression of the Dorsal Root Ganglion. PloS one 2014, 9, e89894. [Google Scholar] [CrossRef] [PubMed]
- de Sousa, M.V.P.; Kawakubo, M.; Ferraresi, C.; Kaippert, B.; Yoshimura, E.M.; Hamblin, M.R. Pain management using photobiomodulation: Mechanisms, location, and repeatability quantified by pain threshold and neural biomarkers in mice. J Biophotonics 2018, 11, e201700370. [Google Scholar] [CrossRef]
- Jenkins, M.W.; Buzza, A.; Skubal, A.C.; Moffitt, M.A.; Anders, J.J. Transient Selective Neural Inhibition via PBM. Photobiomodul Photomed Laser Surg 2024, 42, 574–576. [Google Scholar] [CrossRef]
- Chen, M.; Shimada, K.; Fujita, K.; Ishii, J.; Hirata, T.; Fujisawa, H. Neurite elongation from cultured dorsal root ganglia is inhibited by Ga-Al-As diode laser irradiation. Laser Life Sci 1993, 5, 237–242. [Google Scholar]
- Wesselmann, U.; Kerns, J.; Rymer, W. Laser effects in myelinated and nonmyelinated fibres in the rat peroneal nerve: a quantitative ultrastructural analysis. Exp Neurol 1994, 129, 257–265. [Google Scholar] [CrossRef]
- Wesselmann, U.; Kerns, J.; Rymer, W. Laser effects on myelinated and non-myelinated axons in rat peroneal nerve. Soc Neurosci Abstr 1992, 18, 134. [Google Scholar]
- Wesselmann, U.; Lin, S.; Rymer, W. Effects of Q-switched Nd:YAG laser irradiation on neural impulse propagation: II. Dorsal Roots and Peripheral Nerves. Physiol Chem Phys Med NMR 1991, 23, 81–100. [Google Scholar] [PubMed]
- Wesselmann, U.; Lin, S.; Rymer, W. Effects of Q-switched Nd:YAG laser irradiation on neural impulse propagation: I. Spinal Cord. Physiol Chem Phys Med NMR 1991, 23, 67–80. [Google Scholar] [PubMed]
- Wesselmann, U.; Lin, S.; Rymer, W. Selective decrease of small sensory neurons in lumbar dorsal root ganglia labeled with horseradish peroxidase after Nd:YAG laser irradiation of the tibial nerve in the rat. Exp Neurol 1991, 111, 251–262. [Google Scholar]
- Wesselmann, U.; Rymer, W.; Lin, S. Effect of pulsed infrared lasers on neural condition and axoplasmic transport in sensory nerves. In Proceedings of the Laser Surgery: Advanced Characterization, Therapeutics, and Systems II, 1990; pp. 319–333. [Google Scholar]
- Lin, S.-F.; Wesselmann, U.; Rymer, W.Z. Pulsed laser radiation effects on the distribution of conduction velocities in sciatic nerve of the rat. In Proceedings of the Laser Surgery:Advanced Characterization, Therapeutics and Systems II. SPIE Proceedings, Belingham, WA, 1990; pp. 334–342. [Google Scholar]
- Wesselmann, U.; Rymer, W.Z. Retrograde Horseradish Peroxidase Transport in Motor Axons after Nd:YAG Laser Irradiation of the Tibial Nerve in Rats. Experimental Neurology 1993, 119, 147–152. [Google Scholar] [CrossRef]
- Chow, R.; David, M.; Armati, P. 830-nm laser irradiation induces varicosity formation, reduces mitochondrial membrane potential and blocks fast axonal flow in small and medium diameter rat dorsal root ganglion neurons: implications for the analgesic effects of 830-nm laser. J Peripher Nerv Syst 2007, 12, 28–39. [Google Scholar] [CrossRef]
- Bokhari, L.; Chow, R.; Yan, W.; Armati, P. The effects of 650nm continuous wave laser irradiation on rat dorsal root ganglion neurons and sciatic nerve: relevance to neural mechanisms of low-level laser therapy for chronic pain. In Proceedings of the 18th International Society for Lasers in Medicine and Surgery (ISLMS), Tokyo, Japan, 1 December 2009, 2009. [Google Scholar]
- Kudoh, C.; Inomata, K.; Okajima, K.; Motegi, M.; Ohshiro, T. Effects of 830nm Gallium Aluminium Garsenide diode laser radiation on rat saphenous nerve sodium-potassium-adenosine triphosphatase activity: a possible pain attenuation mechanism examined. Laser Ther 1989, 1, 63–67. [Google Scholar]
- Motzkin, J.C.; Basbaum, A.I.; Crowther, A.J. Neuroanatomy of the nociceptive system: From nociceptors to brain networks. Int Rev Neurobiol 2024, 179, 1–39. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Chow, R.; Armati, P.J. Inhibitory effects of visible 650-nm and infrared 808-nm laser irradiation on somatosensory and compound muscle action potentials in rat sciatic nerve: implications for laser-induced analgesia. J Peripher Nerv Syst 2011, 16, 130–135. [Google Scholar] [CrossRef]
- Chan, A.; Armati, P.; Moorthy, A.P. Pulsed Nd: YAG laser induces pulpal analgesia: a randomized clinical trial. J Dent Res 2012, 91, 79S–84S. [Google Scholar] [CrossRef] [PubMed]
- Chow, R.; Armati, P.; Laakso, E.L.; Bjordal, J.M.; Baxter, G.D. Inhibitory effects of laser irradiation on peripheral mammalian nerves and relevance to analgesic effects: a systematic review. Photomed Laser Surg 2011, 29, 365–381. [Google Scholar] [CrossRef]
- Walker, J. Temporary suppression of clonus in humans by brief photostimulation. Brain Res 1985, 340, 109–113. [Google Scholar] [CrossRef]
- Baxter, G.; Allen, J.; Bell, A. The effect of low-energy density laser irradiation upon human median nerve conduction latencies. J Physiol 1991, 435, 63. [Google Scholar]
- Baxter, G.; Bell, A.; Allen, J.; Ravey, J. Laser mediated increase in median nerve conduction latencies. Irish J Med Sci 1991, 160, 145. [Google Scholar]
- Baxter, G.D.; Allen, A.J.; Bell, J.; Ravey, J.; Diamantopoulos, C. Effect of laser (830nm) upon conduction in the median nerve. Amer Soc Laser Med Surg Abstr 1991, 3, 79. [Google Scholar]
- Baxter, G.C.; Walsh, D.M.; Allen, J.M.; Lowe, A.S.; Bell, A.J. Effects of low intensity infrared laser irradiation upon conduction in the human median nerve in vivo. Exp Physiol 1994, 79, 227–234. [Google Scholar] [CrossRef]
- Lowe, A.; Baxter, G.; Walsh, D.; Allen, J. Effect of low-intensity laser irradiation (830nm) upon skin temperature and antidromic conduction latencies in human median nerve: relevance of radiant exposure. Laser Surg Med 1994, 14, 40–46. [Google Scholar] [CrossRef]
- Walsh, D.; Baxter, G.; Allen, J. The effect of 820nm laser irradiation upon conduction in the frog (Rana Temporaria) sciatic nerve in vitro. Laser Therapy 1995, 7, 5–10. [Google Scholar] [CrossRef]
- Snyder-Mackler, L.; Bork, C.E. Effect of helium-neon laser irradiation on peripheral sensory nerve latency. Phys Ther 1988, 68, 223–225. [Google Scholar] [CrossRef] [PubMed]
- Cambier, D.; Blom, K.; Witvrouw, E.; Ollevier, G.; De Muynck, M.; Vanderstraeten, G. The influence of low intensity infrared laser irradiation on conduction characteristics of peripheral nerve: A randomised, controlled, double blind study on the sural nerve. Laser Med Sci 2000, 15, 195–200. [Google Scholar] [CrossRef]
- Hadian, M.; Moghagdam, B. The effects of low power laser on electrophysiological parameters of sural nerve in normal subjects: a comparison between 670 and 780nm wavelengths. Acta Med Iran 2003, 41, 138–142. [Google Scholar]
- Vinck, E.; Coorevits, P.; Cagnie, B.; De Muynck, M.; Vanderstraeten, G.; Cambier, D. Evidence of changes in sural nerve conduction mediated by light emitting diode irradiation. Lasers Med Sci 2005, 20, 35–40. [Google Scholar] [CrossRef]
- Nelson, A.; Friedman, M. Somatosensory trigeminal evoked potential amplitudes following low-level laser and sham irradiation over time. Laser Ther 2001, 13, 60–64. [Google Scholar] [CrossRef]
- Safavi-Farokhi, Z.; Bakhtiary, A.H. The effect of infrared laser on sensory radial nerve electrophysiological parameters. Electromyogr Clin Neurophysiol 2005, 45, 353–356. [Google Scholar]
- Suganthirababu, P.; Prathap, L.; Alagesan, J.; Kumaresan, A.; Srinivasan, V.; D., G. Neurophysiological effect of low level laser therapy on Ulnar Nerve. Biomedicine 2021, 3, 543–546. [Google Scholar] [CrossRef]
- Telemeco, T.A.; Schrank, E.C. The Effect of Light Therapy on Superficial Radial Nerve Conduction Using a Clustered Array of Infrared Super luminous Diodes and Red Light Emitting Diodes. J Lasers Med Sci 2013, 4, 17–24. [Google Scholar]
- Orchardson, R.; Whitters, C. Effect of HeNe and pulsed Nd:YAG laser irradiation on intradental nerve responses to mechanical stimulation of dentine. Laser Surg Med 2000, 26, 241–249. [Google Scholar] [CrossRef]
- Tsuchiya, D.; Kawatani, M.; Takeshige, C. Laser irradiation abates neuronal responses to nociceptive stimulation of rat-paw skin. Brain Res Bull 1994, 34, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Mezawa, S.; Iwata, K.; Naito, K.; Kamogawa, H. The possible analgesic effect of soft-laser irradiation on heat nociceptors in the cat tongue. Archs Oral Biol 1988, 33, 693–694. [Google Scholar] [CrossRef]
- Kasai, S.; Kono, T.; Sakamoto, T.; Mito, M. Effects of low-power laser irradiation on multiple unit discharges induced by noxious stimuli in the anesthetized rabbit. J Clin Laser Med Surg 1994, 12, 221–224. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Hamba, M.; Matsumoto, K.; Tachibana, H. Effect of irradiation by semiconductor laser on responses evoked in trigeminal caudal neurons by tooth pulp stimulation. Laser Surg Med 1993, 13, 605–610. [Google Scholar] [CrossRef]
- Shimoyama, M.; Fukuda, Y.; Shimoyama, N.; IIjima, K.; Mizuguchi, T. Effect of HeNe laser irradiation on synaptic transmission of the superior cervical ganglion in the rat. J Clin Laser Med Surg 1992, 10, 337–342. [Google Scholar] [CrossRef]
- Sato, T.; Kawatani, M.; Takeshige, C.; Matsumoto, I. Ga-Al-As laser irradiation inhibits neuronal activity associated with inflammation. Acupunct Electrother Res 1994, 19, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Jimbo, K.; Noda, K.; Suzuki, H.; Yoda, K. Suppressive effects of low-power laser irradiation on bradykinin evoked action potentials in cultured murine dorsal root ganglia cells. Neurosci Lett 1998, 240, 93–96. [Google Scholar] [CrossRef]
- Kono, T.; Kasai, S.; Sakamoto, T.; Mito, M. Cord dorsum potentials suppressed by low power laser irradiation on a peripheral nerve in the cat. J Clin Laser Med Surg 1993, 11, 115–118. [Google Scholar] [CrossRef]
- Miura, A.; Kawatani, M. Effects of diode laser irradiation on sensory ganglion cells from the rat. Pain Res 1996, 11, 175–183. [Google Scholar] [CrossRef]
- Arber, S.; Boskov, D.; Rymer, W. Effects of CO2 and HeNe laser irradiation on rat sciatic nerve in vitro. In Proceedings of the Laser Tissue Interaction - P Soc Photo-Opt Ins SPIE, Los Angeles, California, USA, 1990; pp. 196–204. [Google Scholar]
- Tsuchiya, D.; Kawatani, M.; Takeshige, C.; Sato, T.; Matsumoto, I. Diode laser irradiation selectively diminishes slow component of axonal volleys to dorsal roots from the saphenous nerve in the rat. Neurosci Lett 1993, 161, 65–68. [Google Scholar] [CrossRef]
- Kasai, S.; Kono, T.; Yasuhiro, Y.; Kotani, H.; Sakamoto, T.; Mito, M. Effect of low-power laser irradiation on impulse conduction in anaesthetized rabbits. J Clin Laser Med Surg 1996, 14, 107–109. [Google Scholar] [CrossRef]
- Kao, M.-C.; Lin, F.-Y.; Chiu, H.C. Laser effect on somatosensory potential of the peripheral nerve. Amercian Society for Laser Medicine & Surgery Abstracts 1989, 30–31. [Google Scholar]
- Shimoyama, N.; Iijima, K.; Shimoyama, M.; Mizuguchi, T. The effects of helium-neon laser on formalin-induced activity of dorsal horn neurons in the rat. J Clin Laser Med Surg 1992, 10, 91–94. [Google Scholar] [CrossRef]
- Uta, D.; Ishibashi, N.; Konno, T.; Okada, Y.; Kawase, Y.; Tao, S.; Kume, T. Near-Infrared Photobiomodulation of the Peripheral Nerve Inhibits the Neuronal Firing in a Rat Spinal Dorsal Horn Evoked by Mechanical Stimulation. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Buzza, A.; Tapas, K.; Zhuo, J.; Anders, J.J.; Lewis, S.J.; Jenkins, M.W.; Moffitt, M. Selective neural inhibition via photobiomodulation alleviates behavioral hypersensitivity associated with small sensory fiber activation. Lasers Surg Med 2024, 56, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Chow, R.T.; Barnsley, L.B.; Heller, G.Z. The effect of 300mW, 830nm laser on chronic neck pain: a double-blind, randomized, placebo-controlled study. Pain 2006, 124, 201–210. [Google Scholar] [CrossRef]
- Stausholm, M.B.; Naterstad, I.F.; Joensen, J.; Lopes-Martins, R.A.B.; Saebo, H.; Lund, H.; Fersum, K.V.; Bjordal, J.M. Efficacy of low-level laser therapy on pain and disability in knee osteoarthritis: systematic review and meta-analysis of randomised placebo-controlled trials. BMJ Open 2019, 9, e031142. [Google Scholar] [CrossRef]
- Korada, H.Y.; Arora, E.; Maiya, G.A.; Rao, S.; Hande, M.; Shetty, S.; Gundmi, S.; Anche, P.; Amravadi, S. Effectiveness of Photobiomodulation Therapy on Neuropathic Pain, Nerve Conduction and Plantar Pressure Distribution in Diabetic Peripheral Neuropathy - A Systematic Review. Curr Diabetes Rev 2023, 19, e290422204244. [Google Scholar] [CrossRef]
- Haslerud, S.; Magnussen, L.H.; Joensen, J.; Lopes-Martins, R.A.; Bjordal, J.M. The efficacy of low-level laser therapy for shoulder tendinopathy: a systematic review and meta-analysis of randomized controlled trials. Physiother Res Int 2015, 20, 108–125. [Google Scholar] [CrossRef]
- de Lara Quagliotto, G.; Manchope, M.P.; Hilario, R.; Zubeldia, V.; Stacheslki, R.A.; de Carvalho, A.R.; Buzanello, M.R.; Bertolini, G.R.F. Photobiomodulation associated with physical exercise in shoulder impingement syndrome. Systematic review with meta-analysis. Photochem Photobiol 2025. [Google Scholar] [CrossRef] [PubMed]
- de Andrade, A.L.; Bossini, P.S.; Parizotto, N.A. Use of low level laser therapy to control neuropathic pain: A systematic review. J Photochem Photobiol B 2016, 164, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Argenta, P.A.; Ballman, K.V.; Geller, M.A.; Carson, L.F.; Ghebre, R.; Mullany, S.A.; Teoh, D.G.; Winterhoff, B.J.; Rivard, C.L.; Erickson, B.K. The effect of photobiomodulation on chemotherapy-induced peripheral neuropathy: A randomized, sham-controlled clinical trial. Gynecol Oncol 2017, 144, 159–166. [Google Scholar] [CrossRef]
- Clijsen, R.; Brunner, A.; Barbero, M.; Clarys, P.; Taeymans, J. Effects of low-level laser therapy on pain in patients with musculoskeletal disorders: a systematic review and meta-analysis. Eur J Phys Rehabil Med 2017, 53, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.J.; Liao, C.D.; Hong, J.P.; Hsu, W.C.; Wu, C.W.; Chen, H.C. Effects of laser therapy on chronic low back pain: A systematic review and meta-analysis of randomized controlled trials. Clin Rehabil 2022, 36, 289–302. [Google Scholar] [CrossRef]
- Huang, Z.; Ma, J.; Chen, J.; Shen, B.; Pei, F.; Kraus, V.B. The effectiveness of low-level laser therapy for nonspecific chronic low back pain: a systematic review and meta-analysis. Arthritis Res Ther 2015, 17, 360. [Google Scholar] [CrossRef]
- Ibarra, A.M.C.; Biasotto-Gonzalez, D.A.; Kohatsu, E.Y.I.; de Oliveira, S.S.I.; Bussadori, S.K.; Tanganeli, J.P.C. Photobiomodulation on trigeminal neuralgia: systematic review. Lasers Med Sci 2021, 36, 715–722. [Google Scholar] [CrossRef]
- Mukhtar, R.; Fazal, M.U.; Saleem, M.; Saleem, S. Role of low-level laser therapy in post-herpetic neuralgia: a pilot study. Lasers Med Sci 2020, 35, 1759–1764. [Google Scholar] [CrossRef]
- Navarro-Ledesma, S.; Carroll, J.; Burton, P.; Ana, G.M. Short-Term Effects of Whole-Body Photobiomodulation on Pain, Quality of Life and Psychological Factors in a Population Suffering from Fibromyalgia: A Triple-Blinded Randomised Clinical Trial. Pain Ther 2023, 12, 225–239. [Google Scholar] [CrossRef]
- Soriano, F.; Campana, V.; Moya, M.; Gavotto, A.; Simes, J.; Soriano, M.; Soriano, R.; Spitale, L.; Palma, J. Photobiomodulation of pain and inflammation in microcrystalline arthropathies: experimental and clinical results. Photomed Laser Surg 2006, 24, 140–150. [Google Scholar] [CrossRef]
- Orchardson, R.; Peacock, J.M.; Whitters, C.J. Effects of pulsed Nd:YAG laser radiation on action potential conduction in.
- nerve fibres inside teeth in vitro. Journal of Dentistry 1998, 26, 421–426. [CrossRef] [PubMed]
- Díaz, L.; Restelli, L.; Valencia, E.; Atalay, D.I.; Abarca, J.M.; Gil, A.C.; Fernández, E. Effectiveness of low-level laser therapy on temporomandibular disorders. A systematic review of randomized clinical trials. Photodiagnosis Photodyn Ther 2025, 53, 104558. [Google Scholar] [CrossRef] [PubMed]
- Candido-do-Prado, L.G.; Ribeiro-Silva, V.H.A.; Simoes-Barbosa, A.F.; Mazzi-Chaves, J.F.; Magri, L.V. Differential effectiveness of photobiomodulation in muscular and articular temporomandibular disorders: a systematic review and critical appraisal. Lasers Med Sci 2025, 40, 487. [Google Scholar] [CrossRef]
- Shukla, D.; Muthusekhar, M.R. Efficacy of low-level laser therapy in temporomandibular disorders: A systematic review. Natl J Maxillofac Surg 2016, 7, 62–66. [Google Scholar] [CrossRef]
- Sarangi, P.; Suman, S.; Satapathy, S.K.; Meher, R.; Das, A.; Pradeep, D.M. The Pros and Cons of Dental Laser Therapy in Conservative Dentistry and Endodontics-A Systematic Review. J Pharm Bioallied Sci 2024, 16, S3083–s3085. [Google Scholar] [CrossRef]
- Khemiss, M.; Dammak, N.; Lajili, O.; Yacoub, S.; Ben Khelifa, M. Efficacy of laser therapy on primary burning mouth syndrome: a systematic review. J Oral Facial Pain Headache 2024, 38, 17–31. [Google Scholar] [CrossRef] [PubMed]
- Sonesson, M.; De Geer, E.; Subraian, J.; Petrén, S. Efficacy of low-level laser therapy in accelerating tooth movement, preventing relapse and managing acute pain during orthodontic treatment in humans: a systematic review. BMC Oral Health 2016, 17, 11. [Google Scholar] [CrossRef]
- Scheuer, T. Regulation of sodium channel activity by phosphorylation. Semin Cell Dev Biol 2011, 22, 160–165. [Google Scholar] [CrossRef]
- Ross, G. Photobiomodulation Therapy: A Possible Answer to the Opioid Crisis. Photobiomodul Photomed Laser Surg 2019, 37, 667–668. [Google Scholar] [CrossRef]
- Poste, G.; Papahadjopoulos, D.; Nicholson, G. Local anaesthetics affect transmembrane cytoskeletal control of mobility and distribution of cell surface receptors. P Nat Acad Sci USA 1975, 72, 4430–4434. [Google Scholar] [CrossRef]
- Nicolson, G.; JR., S.; Poste, G. Effects of local anaesthetics on cell morphology and membrane-associated cytoskeletal organization in BALB3/3T3. J Cell Biol 1976, 68, 395–402. [Google Scholar] [CrossRef]
- Tanelian, D.L.; Markin, V.S. Biophysical and functional consequences of receptor-mediated nerve fiber transformation. Biophys J 1997, 72, 1092–1108. [Google Scholar] [CrossRef]
- Haschke, R.H.; Byers, M.R.; Fink, B.R. Effects of lidocaine on rabbit brain microtubular protein. J Neurochem 1974, 22, 837–843. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, Y.; Muto, E.; Mashimo, T.; Iwane, A.H.; Yoshiya, I.; Yanagida, T. Direct inhibition of microtubule-based kinesin motility by local anesthetics. Biophys J 2000, 78, 940–949. [Google Scholar] [CrossRef] [PubMed]
- Chow, R.T.; Johnson, M.I.; Lopes-Martins, R.A.; Bjordal, J.M. Efficacy of low-level laser therapy in the management of neck pain: a systematic review and meta-analysis of randomised placebo or active-treatment controlled trials. Lancet 2009, 374, 1897–1908. [Google Scholar] [CrossRef]
- Glazov, G.; Yelland, M.; Emery, J. Low-level laser therapy for chronic non-specific low back pain: a meta-analysis of randomised controlled trials. Acupunct Med 2016, 34, 328–341. [Google Scholar] [CrossRef]
- Naterstad, I.F.; Joensen, J.; Bjordal, J.M.; Couppe, C.; Lopes-Martins, R.A.B.; Stausholm, M.B. Efficacy of low-level laser therapy in patients with lower extremity tendinopathy or plantar fasciitis: systematic review and meta-analysis of randomised controlled trials. BMJ Open 2022, 12, e059479. [Google Scholar] [CrossRef] [PubMed]
- Bjordal, J.; Johnson, M.; Iverson, V.; Aimbire, F.; Lopes-Martins, R. Photoradiation in acute pain: A systematic review of possible mechanisms of action and clinical effects in randomized placebo-controlled trials. Photomed Laser Surgery 2006, 24, 158–168. [Google Scholar] [CrossRef]
- Hu, Y.; Ma, J.; Chen, B.; Pang, J.; Liang, W.; Wu, W. The Duration of Chronic Pain Can Affect Brain Functional Changes of the Pain Matrix in Patients with Chronic Back Pain: A Resting-State fMRI Study. J Pain Res 2024, 17, 1941–1951. [Google Scholar] [CrossRef]
- Gazerani, P. The neuroplastic brain: current breakthroughs and emerging frontiers. Brain Res 2025, 1858, 149643. [Google Scholar] [CrossRef]
- Glare, P.; Aubrey, K.R.; Myles, P.S. Transition from acute to chronic pain after surgery. Lancet 2019, 393, 1537–1546. [Google Scholar] [CrossRef]
- Shubayev, V.I.; Myers, R.R. Axonal transport of TNF-alpha in painful neuropathy: distribution of ligand tracer and TNF receptors. J Neuroimmunol 2001, 114, 48–56. [Google Scholar] [CrossRef]
- Murwani, R.; Armati, P. Peripheral nerve fibroblasts as a source of IL-6, TNFalpha and IL-1 and their modulation by IFNgamma. Journal of Neurological Sciences 1998, 161, 99–109. [Google Scholar] [CrossRef]
- Fregnan, F.; Muratori, L.; Simoes, A.R.; Giacobini-Robecchi, M.G.; Raimondo, S. Role of inflammatory cytokines in peripheral nerve injury. Neural Regen Res 2012, 7, 2259–2266. [Google Scholar] [CrossRef] [PubMed]
- Guptarak, J.; Wanchoo, S.; Durham-Lee, J.; Wu, Y.; Zivadinovic, D.; Paulucci-Holthauzen, A.; Nesic, O. Inhibition of IL-6 signaling: A novel therapeutic approach to treating spinal cord injury pain. Pain 2013, 154, 1115–1128. [Google Scholar] [CrossRef] [PubMed]
- Lindwall, C.; Kanje, M. Retrograde axonal transport of JNK signaling molecules influence injury induced nuclear changes in p-c-Jun and ATF3 in adult rat sensory neurons. Mol Cell Neurosci 2005, 29, 269–282. [Google Scholar] [CrossRef] [PubMed]
- Sorkin, A.; Von Zastrow, M. Signal transduction and endocytosis: close encounters of many kinds. Nat Rev Mol Cell Biol 2002, 3, 600–614. [Google Scholar] [CrossRef]
- Harorli, O.T.; Hatipoglu, M.; Erin, N. Effect of Photobiomodulation on Secretion of IL-6 and IL-8 by Human Gingival Fibroblasts In Vitro. Photobiomodul Photomed Laser Surg 2019, 37, 457–464. [Google Scholar] [CrossRef]
- Mojarad, N.; Janzadeh, A.; Yousefifard, M.; Nasirinezhad, F. The role of low level laser therapy on neuropathic pain relief and interleukin-6 expression following spinal cord injury: An experimental study. J Chem Neuroanat 2018, 87, 60–70. [Google Scholar] [CrossRef]
- Pallotta, R.C.; Bjordal, J.M.; Frigo, L.; Leal Junior, E.C.; Teixeira, S.; Marcos, R.L.; Ramos, L.; Messias Fde, M.; Lopes-Martins, R.A. Infrared (810-nm) low-level laser therapy on rat experimental knee inflammation. Lasers Med Sci 2012, 27, 71–78. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira Martins, D.; Martinez dos Santos, F.; Evany de Oliveira, M.; de Britto, L.R.; Benedito Dias Lemos, J.; Chacur, M. Laser therapy and pain-related behavior after injury of the inferior alveolar nerve: possible involvement of neurotrophins. J Neurotrauma 2013, 30, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Yazdani, S.O.; Golestaneh, A.F.; Shafiee, A.; Hafizi, M.; Omrani, H.A.; Soleimani, M. Effects of low level laser therapy on proliferation and neurotrophic factor gene expression of human schwann cells in vitro. J Photochem Photobiol B 2012, 107, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Belanger, P.; West, C.R.; Brown, M.T. Development of pain therapies targeting nerve growth factor signal transduction and the strategies used to resolve safety issues. J Toxicol Sci 2018, 43, 1–10. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



