Submitted:
15 January 2026
Posted:
16 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Materials
Methods
Yeast Strain and Erlenmeyer Flask Culture
Continuous Fermentation in Bioreactor
Biomass Calculation
Dilution Rate and Retention Time
Harvest Rate and Feed Rate of Substrates
KEX2 Protease Characterisation and Quantification
3. Results
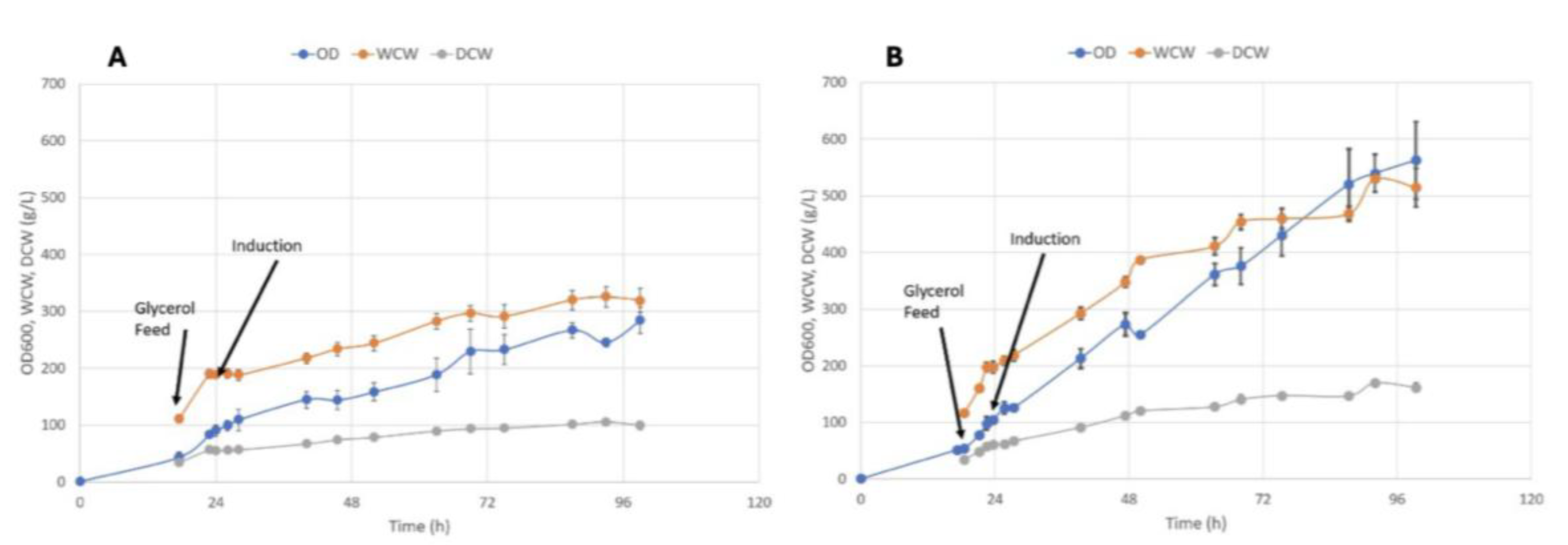
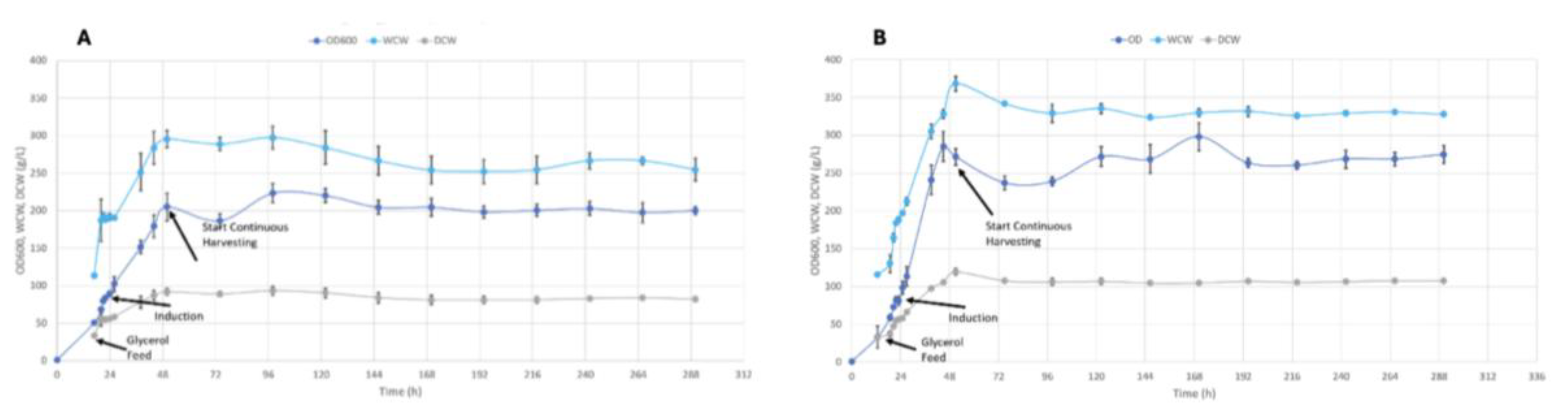
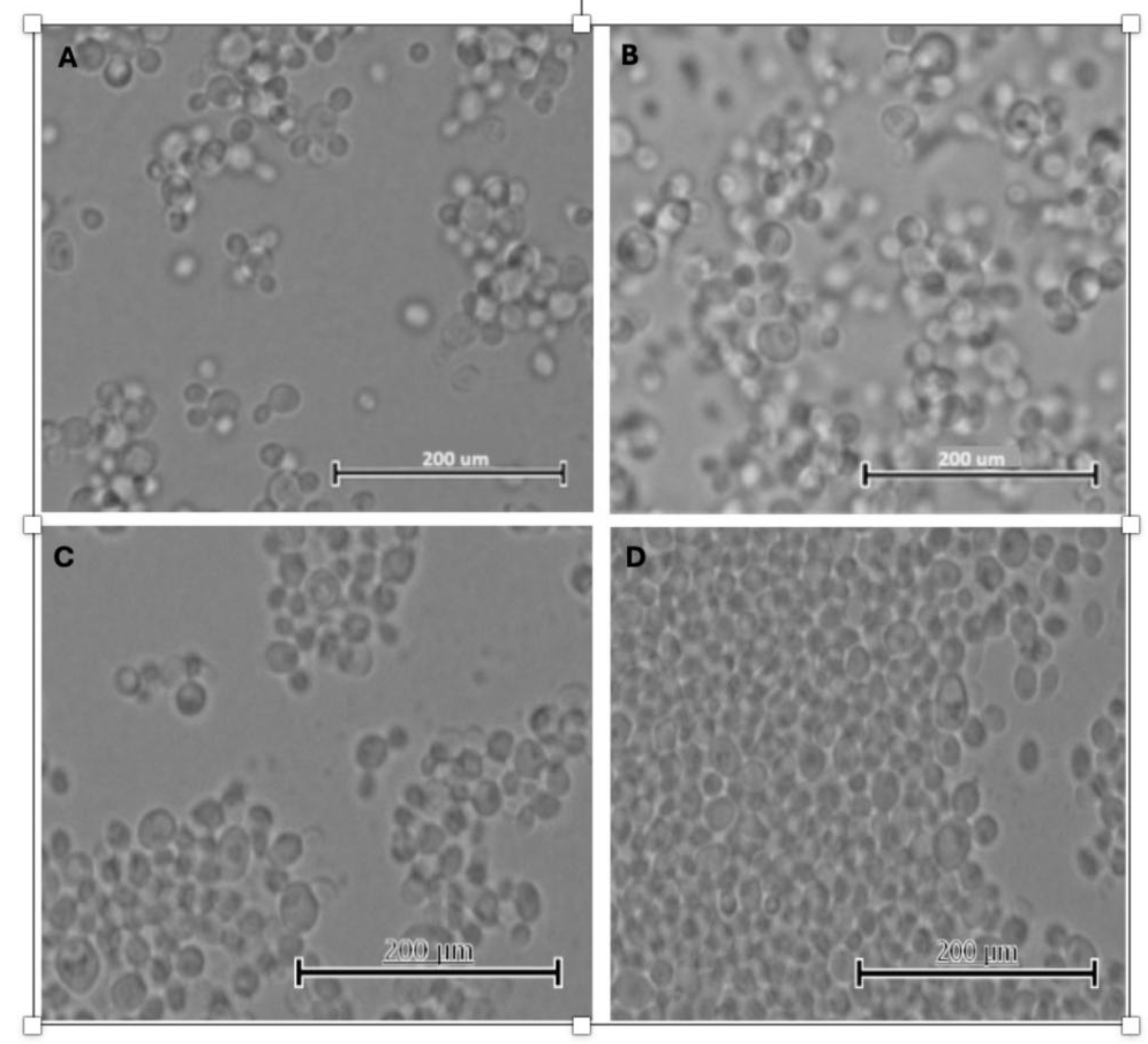
3.1. Biological Performance
3.2. Productivity Analysis
3.3. Techno-Economic Evaluation
Industrial-Scale Simulation and Economic Assessment
Scenario 2: 400 L Fed-Batch vs 40 L Continuous Mixed Induction
Yield-to-Cost Efficiency
4. Discussion
Biological and Process Synergy
Techno-Economic Implications
Industrial Feasibility and Scalability
Limitations and Future Directions
5. Conclusions
Recommendations for Industry
Future Directions
- Optimizing sorbitol-to-methanol ratios across different strains and target proteins to maximize yield and cost-efficiency [5].
- Extending continuous cultivation beyond 10–15 days to evaluate long-term genetic stability, process consistency, and product quality attributes under prolonged operation [8].
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Wen, L.; Menghao, C. Ad v Ancing Recombinant Protein Expression in K Oma Gataella Phaffii : Opportunities and Challenges. FEMS Yeast Res 2025, 25. [Google Scholar] [CrossRef]
- Bustos, C.; Quezada, J.; Veas, R.; Altamirano, C.; Braun-galleani, S.; Fickers, P.; Berrios, J. Advances in Cell Engineering of the Komagataella Phaffii Platform for Recombinant Protein Production; 2022. [Google Scholar]
- Niu, H.; Jost, L.; Pirlot, N.; Sassi, H.; Daukandt, M.; Rodriguez, C.; Fickers, P. A Quantitative Study of Methanol/Sorbitol Co-Feeding Process of a Pichia Pastoris Mut+/PAOX1-LacZ Strain. Microb Cell Fact 2013, 12, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Nesbeth, D.; Keshavarz-Moore, E. Sorbitol/Methanol Mixed Induction Reduces Process Impurities and Improves Centrifugal Dewatering in Pichia Pastoris Culture. Enzyme Microb Technol 2019, 130, 109366. [Google Scholar] [CrossRef] [PubMed]
- Carly, F.; Niu, H.; Delvigne, F.; Fickers, P. Influence of Methanol/Sorbitol Co-Feeding Rate on PAOX1 Induction in a Pichia Pastoris Mut+ Strain in Bioreactor with Limited Oxygen Transfer Rate. J Ind Microbiol Biotechnol 2016, 43, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Narang, A. P AOX1 Expression in Mixed-Substrate Continuous Cultures of Komagataella Phaf Fi i ( Pichia Pastoris ) Is Completely Determined by Methanol Consumption Regardless of the Secondary Carbon Source; 2023; pp. 1–10. [Google Scholar] [CrossRef]
- Vogl, T.; Glieder, A.; Sturmberger, L.; Fauland, P.C.; Hyden, P.; Fischer, J.E.; Schmid, C.; Thallinger, G.G.; Geier, M. Methanol Independent Induction in Pichia Pastoris by Simple Derepressed Overexpression of Single Transcription Factors; 2018; pp. 1037–1050. [Google Scholar] [CrossRef]
- Nieto-Taype, M.A.; Garcia-Ortega, X.; Albiol, J.; Montesinos-Seguí, J.L.; Valero, F. Continuous Cultivation as a Tool Toward the Rational Bioprocess Development With Pichia Pastoris Cell Factory. Front Bioeng Biotechnol 2020, 8, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Herwig, C.; Slouka, C.; Neubauer, P.; Delvigne, F. Editorial : Continuous Biomanufacturing in Microbial Systems. 2021, 9, 7–9. [Google Scholar] [CrossRef] [PubMed]
- Niazi, S.K. Continuous Manufacturing of Recombinant Drugs : Comprehensive Analysis of Cost Reduction Strategies, Regulatory Pathways, and Global Implementation. 2025, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Linova, M.Y.; Sripada, S.A.; Menegatti, S.; Woodley, J.M. Effect of Cell Retention Techniques in Komagataella Phaffii Lab-Scale Continuous Processes. 2025, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Weiss, F.; Requena-moreno, G.; Pichler, C.; Valero, F.; Glieder, A.; Garcia-ortega, X. Scalable Protein Production by Komagataella Phaffii Enabled by ARS Plasmids and Carbon Source-Based Selection; 2024; pp. 1–16. [Google Scholar]
- Bernat-camps, N.; Ebner, K.; Schusterbauer, V.; Fischer, J.E.; Nieto-taype, M.A.; Valero, F.; Glieder, A.; Garcia-ortega, X. Enabling Growth-Decoupled Komagataella Phaf Fi i Recombinant Protein Production Based on the Methanol-Free P DH Promoter; 2023; pp. 1–16. [Google Scholar] [CrossRef]
- Soltani, M.K.; Arjmand, S.; Omid, S.; Siadat, R.; Bagheri, A. Hansenula Polymorpha Methanol Metabolism Genes Enhance Recombinant Protein Production in Komagataella Phaffi. 2024.
- Badger, N.; Boylu, R.; Ilojianya, V.; Erguvan, M.; Amini, S. A Cradle-to-Gate Life Cycle Assessment of Green Methanol Production Using Direct Air Capture. Energy Advances 2024, 3, 2311–2327. [Google Scholar] [CrossRef]
- Kumar, H.; Sharma, R.; Malik, A.K.; Sharma, A.K.; Kumar, P.; Singh, D. Advancements in Carbon Capture and Utilization Technologies_ Transforming CO2 into Valuable Resources for a Sustainable Carbon Economy. Next Energy 2026, 10, 100476. [Google Scholar] [CrossRef]
- de la Cal, Ana; Martin, A. Sustainable Methanol Sourcing the Methanol Economy to Advance Net Zero Chemicals. Available online: https://globalimpactcoalition.com/sustainable-methanol/ (accessed on 25 November 2025).
- Rahimi, A.; Nezamedin, S.; Karimi, A.; Aghdasinia, H. Enhancing the e Ffi Ciency of Recombinant Hepatitis B Surface Antigen Production in Pichia Pastoris by Employing Continuous Fermentation. Biochem Eng J 2019, 141, 112–119. [Google Scholar] [CrossRef]
- Fernandes, B.D.; Mota, A.; Teixeira, J.A.; Vicente, A.A. Continuous Cultivation of Photosynthetic Microorganisms: Approaches, Applications and Future Trends. Biotechnol Adv 2015, 33, 1228–1245. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Mehmet, Æ.C.L.Æ.; Meagher, M.M. Optimization of Cell Density and Dilution Rate in Pichia Pastoris Continuous Fermentations for Production of Recombinant Proteins; 2004; pp. 330–334. [Google Scholar] [CrossRef]
- Azadi, S.; Mahboubi, A.; Naghdi, N.; Solaimanian, R.; Alireza, S. Evaluation of Sorbitol-Methanol Co-Feeding Strategy on Production of Recombinant Human Growth Hormone in Pichia Pastoris. 2017, 16, 1555–1564. [Google Scholar] [PubMed]
- Jaswal, A.S.; Elangovan, R.; Mishra, S. Optimization of Dilution Rate and Mixed Carbon Feed for Continuous Production of Recombinant Plant Sucrose : Sucrose 1 - Fructosyltransferase in Komagataella Phaffii. Bioprocess Biosyst Eng 2024, 47, 1499–1514. [Google Scholar] [CrossRef] [PubMed]
- Steimann, T.; Wegmann, J.; Espinosa, M.I.; Blank, L.M.; Büchs, J.; Mann, M.; Magnus, J.B. Avoiding Overflow Metabolite Formation in Komagataella Phaffii Fermentations to Enhance Recombinant Protein Production. J Biol Eng 2024, 0. [Google Scholar] [CrossRef] [PubMed]
- Innovations in Cell Culture New Flexsafe Bag Family 12.
- Challener, C.A. The Future Is Continuous: Accelerating the Shift in Biomanufacturing. Available online: https://www.pharmasalmanac.com/articles/the-future-is-continuous-accelerating-the-shift-in-biomanufacturing (accessed on 25 November 2025).
- Drobnjakovic, M.; Hart, R. Current Challenges and Recent Advances on the Path towards Continuous Biomanufacturing; 2023; pp. 1–23. [Google Scholar] [CrossRef]
- Khanal, O.; Lenhoff, A.M. Developments and Opportunities in Continuous Biopharmaceutical Manufacturing ABSTRACT. MAbs 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Arulrajah, P.; Elina, A.; Dilara, L.; Subhashree, S.; Dirk, P.; Botz, W.; Heins, A.L. Scale-down Bioreactors—Comparative Analysis of Configurations. Bioprocess Biosyst Eng 2025, 48, 1619–1635. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, Donna; Zhang, Yi; Mootrey, Karen; Davis, Keith; Gonzalez, Lunedt; Richards, M. PAT Monitoring and Control Roadmap. Available online: https://www.biophorum.com/webinar/pat-monitoring-and-control-roadmap/ (accessed on 25 November 2025).
- Strube, J. Continuous Biomanufacturing in Upstream and Downstream Processing. Physical Sciences Reviews 2022, 3167–3222. [Google Scholar] [CrossRef]
- Odum, J. The Challenges of Downstream Operations in the World of Continuous Biomanufacturing. Biopharm Int 2025, 38, 16–18. [Google Scholar]
| Parameter | Fed-batch Methanol |
Fed-batch Mixed |
Continuous Methanol |
Continuous Mixed |
| Yield (mg) | 466.8 ±80 | 1115.6 ±240 | 1154.9 ±870 | 9932.7 ±2980 |
| Fold-change vs. Fed-batch Methanol | 1.0 | 2.4 | 2.5 | 21.3 |
| Per-day yield (mg/day) | 93.4 | 223.1 | 76.9 | 662.2 |
| Total harvested volume (L) | 2 ±0.5 | 2 ±0.3 | 8.2 ±0.8 | 13.5 ±0.6 |
| Total induction volume (L) | 0.6 | 0.6 | 4.6 | 5.9 |
| Total NH₄OH volume (L) | 0.26 ±0.03 | 0.3 ±0.03 | 1 ±0.3 | 1.5 ±0.3 |
| Total antifoam (mL) | 5 ±3 | 5 ±3 | 5 ±3 | 5 ±3 |
| Cultivation Mode | Induction Strategy | Yield per Batch (mg) | Process Time (days) |
| Fed-batch | Pure Methanol | 466.8 | 5 |
| Fed-batch | Mixed (Methanol + Sorbitol) | 1,078.9 | 5 |
| Continuous | Pure Methanol | 1,154.9 | 15 |
| Continuous | Mixed (Methanol + Sorbitol) | 9,932.7 | 15 |
| Method | Material Cost (USD) |
| Fed-batch (Methanol) | 131.17 |
| Fed-batch (Mixed) | 136.99 |
| Continuous (Methanol) | 329.02 |
| Continuous (Mixed) | 479.04 |
| Method | Yield (mg per batch) | Material Cost (USD) | Yield-to-material Cost Ratio |
| Fed-batch (Methanol) | 466.8 | 131.17 | 3.56 |
| Fed-batch (Mixed) | 1078.9 | 136.99 | 7.88 |
| Continuous (Methanol) | 1154.9 | 329.02 | 3.51 |
| Continuous (Mixed) | 9932.7 | 479.04 | 20.73 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




