Submitted:
14 January 2026
Posted:
15 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Ethnopharmacology and Phytochemical Diversity of the Verbesina Genus
2.1. Ethnopharmacology and Ecological Context
2.1.1. Chemotaxonomy and Global Distribution
2.1.2. Mexican Ethnobotany: The Rationale for Anti-AMR
2.1.3. Ecological Resilience and Chemical Defense
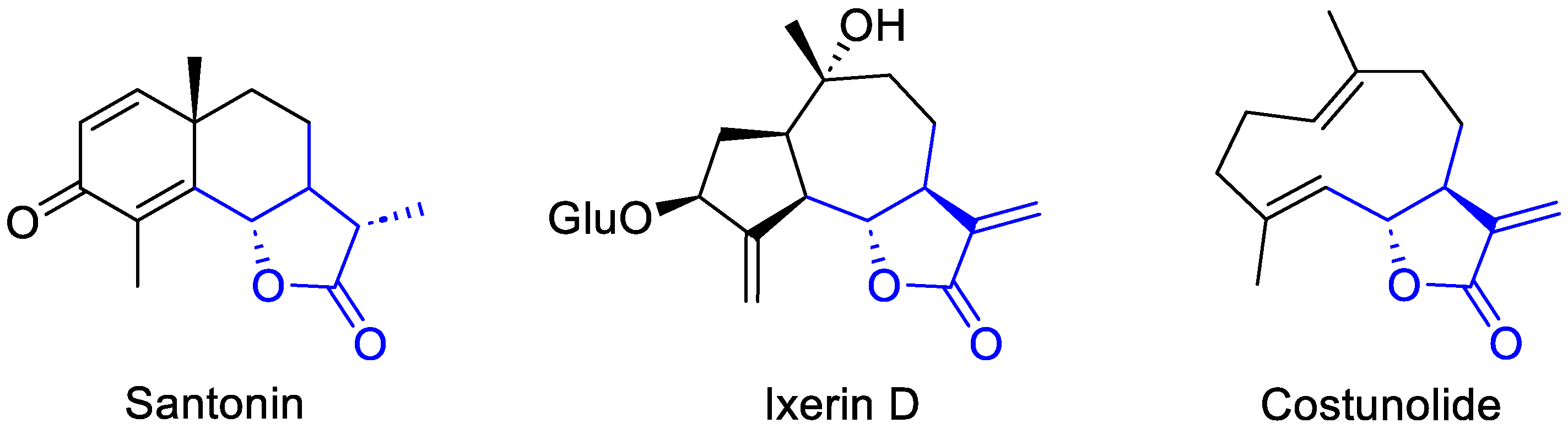
2.2. The Core Chemical Landscape: Sesquiterpene Lactones
2.2.1. Sesquiterpene Lactones: Structure, Chemotaxonomy, and Stereochemistry
| Compound Name (Skeleton) | Structural Descriptor | Source Species | Biological Role |
|---|---|---|---|
| Verbesindiol (Eudesmane - Diol) |
Parent core; 4R,5S,6R,7S,10R configuration; Structural standard. |
V. virginica, V. eggersii |
Potent against prostate cancer cells. |
| CDE (Eudesmane Cinnamate) | 4β-cinnamoyloxy, 1β,3α-dihydroxyeudesm-7,8-ene; Alkylating pharmacophore. | V. persicifolia | Mild mitochondrial uncoupler. Patented for obesity/diabetes/cancer treatment. |
| Arbusculin derivative (Eudesmane Lactone) |
(1R,4S,4aS,7R)-1-methyl-7-(prop-1-en-2-yl)octahydro-2H-1,4a-(epoxymethano)naphthalen-4-yl acetate. | V. lanata | Against the protozoan L. major |
| Rupestrol (Pentahydroxy Eudesmane) |
Highly oxygenated enantio-eudesmane with C-9 oxygenation; isolated as cinnamate esters. | V. rupestris | Confirms complex stereochemistry/chemotaxonomic links. |
| Rupestrinol (Enantio-Eudesmane) |
Epimeric sesquiterpene isolated as cinnamate esters. | V. rupestris | Part of the complex enantio-eudesmane structural series. |
| Eudesmane Esters (Eudesmane Cinnamate) |
Phenolic acid (cinnamate/coumarate) conjugated eudesmanes. | V. virginica | Upholds the chemical signature of SLs conjugated with phenolic acids. |
| SL Acetylated Derivatives (Eudesmane/ Germacrane) | Compounds with acetate substituents. |
V. persicifolia | Acetylation shifts cytotoxicity mechanism to Complex II inhibition (Mitochondrial). |
| Germacrene-Cinnamate | 6β-cinnamoyloxy-1β-hydroxy-10α-methoxy-3-oxo-germacra-4,5Z-ene. | V. negrensis | Antibacterial against S. aureus. |
| Zempoaline C (Elemanolide) |
Elemanolide skeleton with fused α,β-unsaturated-γ-lactone moiety. | V. seattonii | Deploys alkylating pharmacophore via diverse skeletal type. |
| Verocephol (Amorphane Lactol) |
Unique γ-lactol; first reported amorphane skeleton. | V. sphaerocephala | Marks high biosynthetic diversity (C15 precursors). |
| Cadinenes (Sesquiterpenoid) |
New Cadinene-type SLs confirmed by X-ray. | V. sphaerocephala | Structural diversity increases selective lead probability. |
| Parthenin Analog (SL) | SLs induce cytotoxicity via Michael addition to -SH groups. | (General SL mechanism) | α-methylene-γ-lactone is the alkylating pharmacophore. |
| V. macrophylla EO (Sesquiterpenes) | Essential Oil dominated by C15 Sesquiterpenes (Germacrene D). | V. macrophylla | Antimicrobial/anti-inflammatory action with very low host toxicity |
| Anthelmintic Action (Sesquiterpenes / Phenolics) | Essential oil shows 100% adulticidal activity against Habronema muscae. | V. alternifolia | Causes degenerative changes to parasite morphology (SEM confirmed). |
2.2.2. Sesquiterpenoid and Monoterpenoid Diversity
2.2.3. Non-SL Chemical Arsenal and Biosynthetic Complexity
Guanidine Alkaloids: The Dual Challenge of Toxicity and Therapy:
Phenolic Compounds, Flavonoids, and Supportive Metabolites:
Triterpenoids, Alkaloids, and Specialized Defense Structures:
Non-Traditional Bioactive Classes and Technological Application
3. Cytotoxic Activity of Verbesina Metabolites: Focus on IC50 Profiles
3.1. Anticancer Potential Across Cell Lines
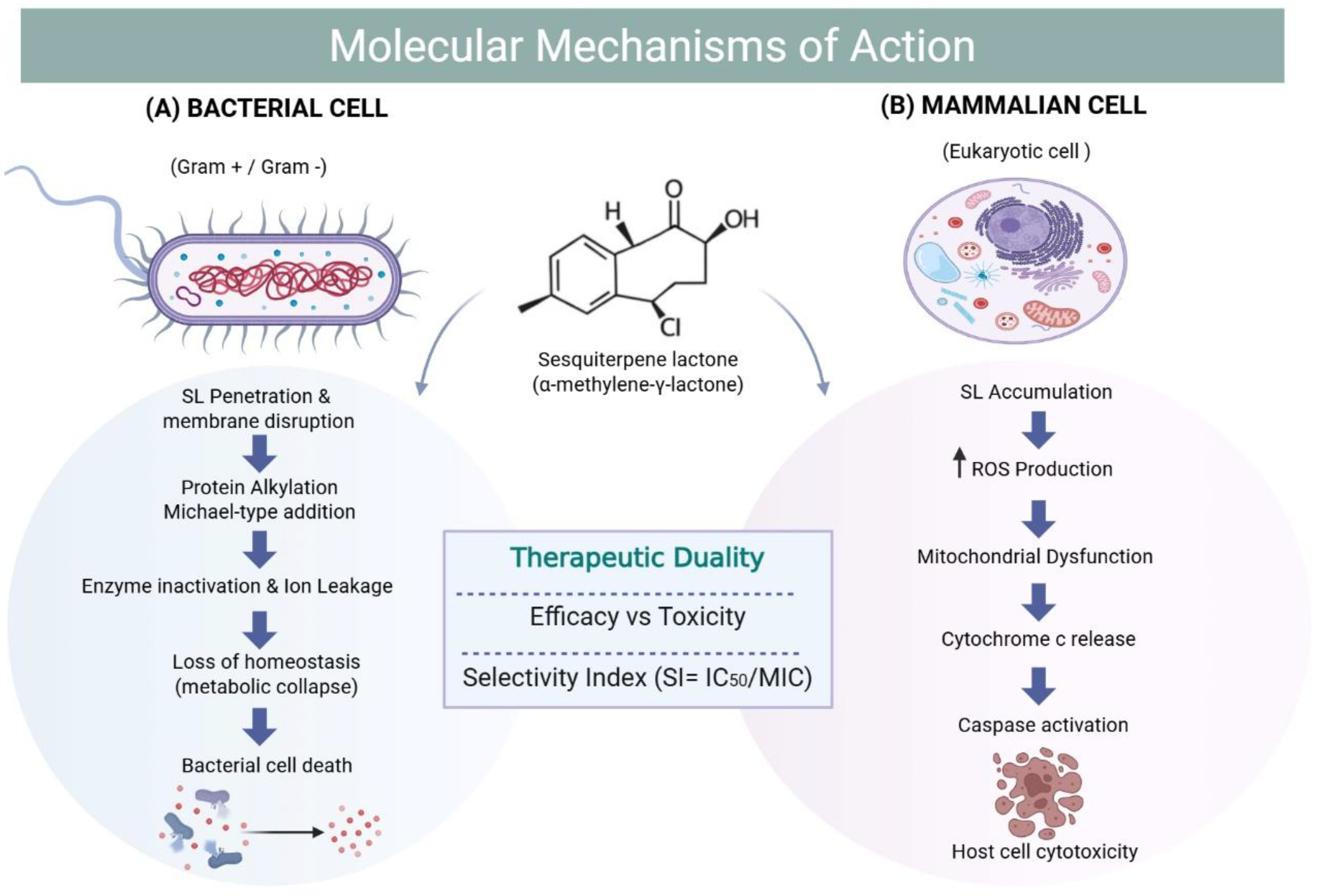
3.2. Molecular Mechanisms of Cytotoxicity
3.2.1. Oxidative Stress and the Apoptotic Cascade
3.2.2. Scaffold Dependency and Mitochondrial Targeting
3.2.3. Long-Term Cytotoxic Profile
3.3. Toxicity to Non-Cancerous Cells
4. Antimicrobial Efficacy and Anti-AMR Mechanisms
4.1. Activity Against Clinically Relevant Pathogens
4.2. Molecular Mechanisms of Antimicrobial Action
4.2.1. The Core Mechanistic Hypothesis: Alkylation and Membrane Disruption
4.2.2. Specialized Mechanisms of Non-SL Metabolites
4.2.3. Ecological and Biosynthetic Context
4.3. Combating Resistance: New Approaches
4.3.1. Host-Mediated and Anti-Inflammatory Strategies
4.3.2. Non-Infectious Therapeutic Applications (Chronic Disease and SAR)
4.3.3. Technological Valorization and Biocontrol Applications
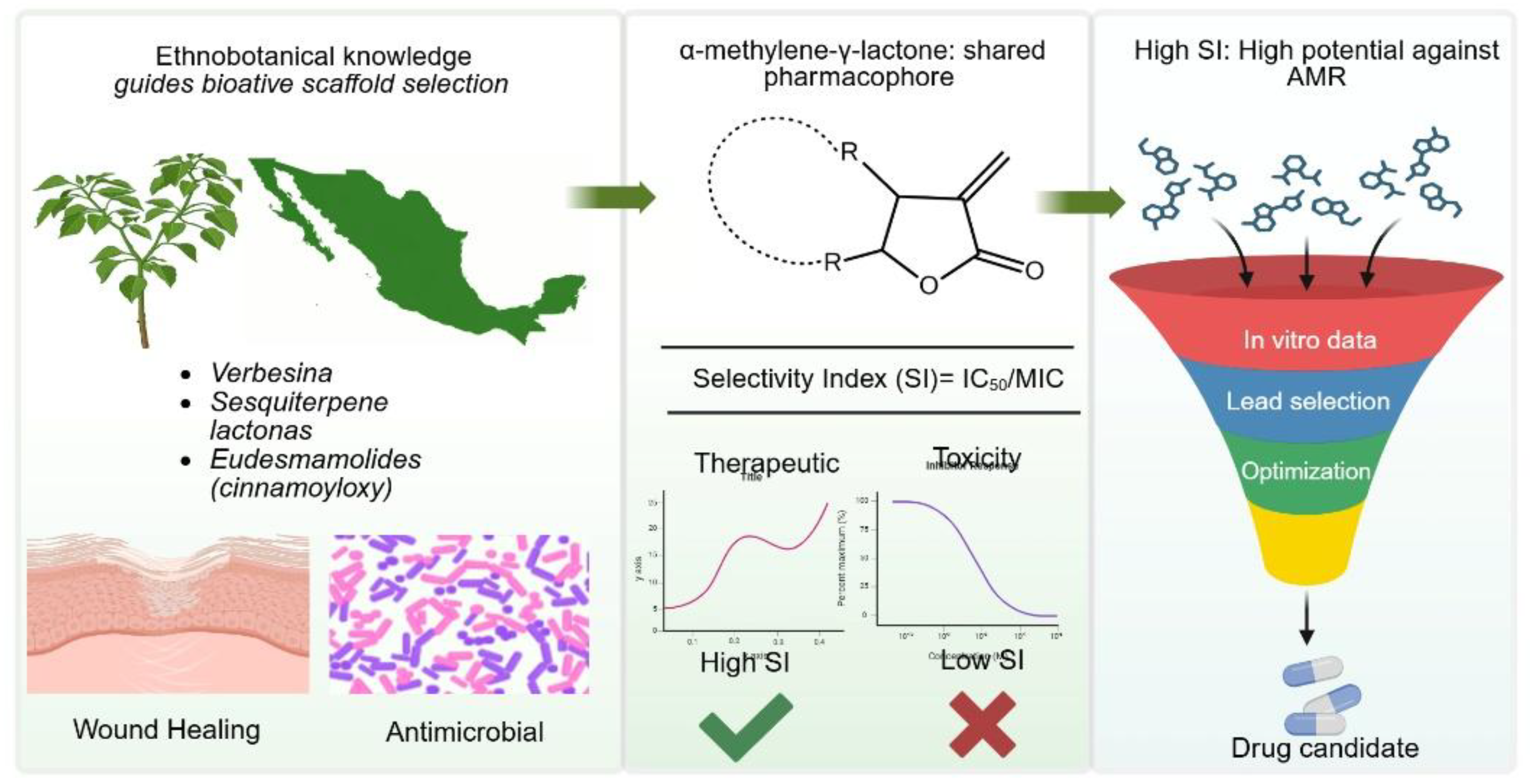
5. Critical Analysis: The Therapeutic Index and Drug Feasibility
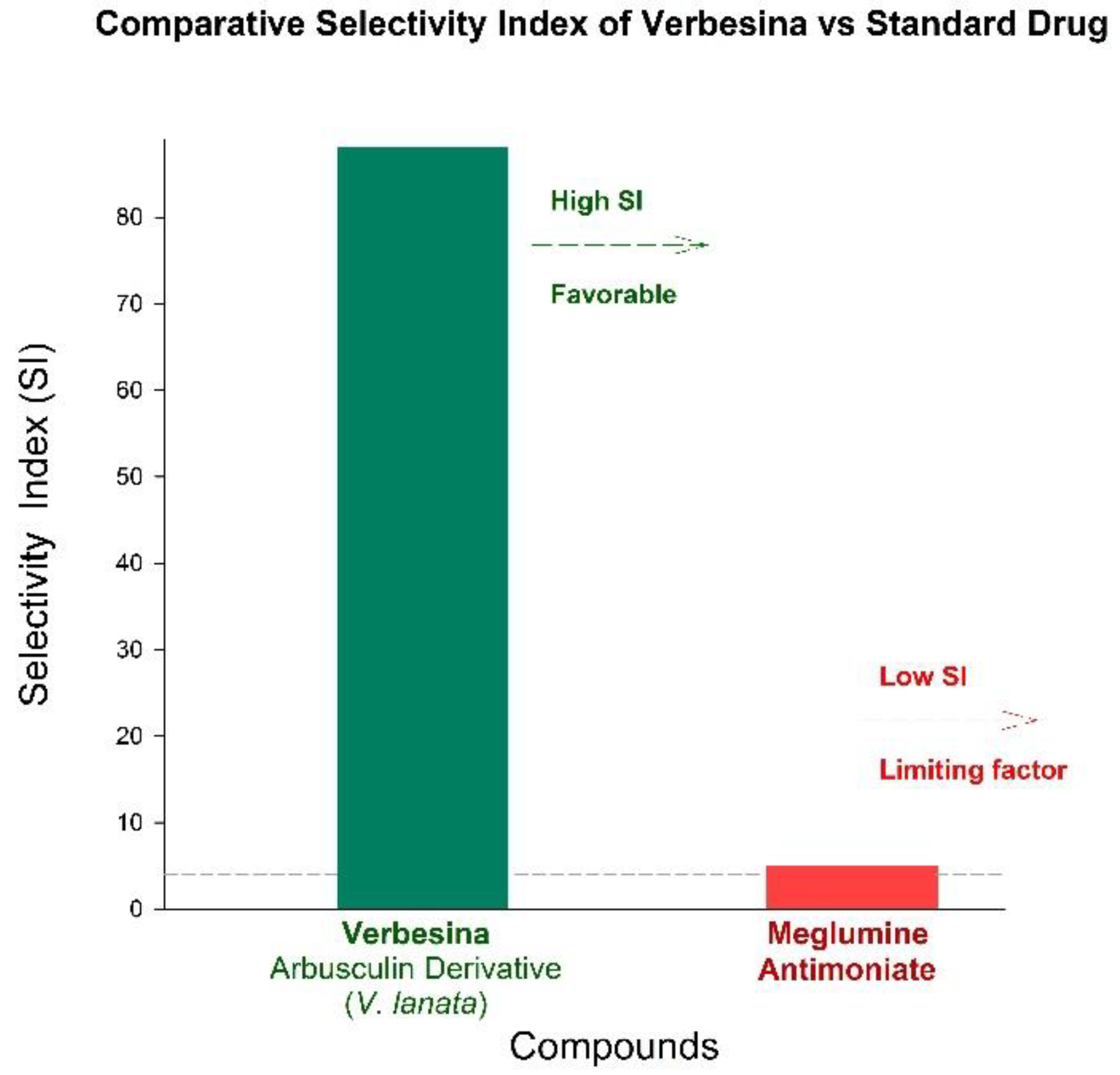
5.1. Calculation and Interpretation of the Selectivity Index (SI)
5.1.1. Quantitative Evidence for a Favorable Selectivity Index
5.1.2. The Duality Challenge and Evidence for Mitigation
5.1.3. The Need for Rigorous Validation
5.2. Structure-Activity Relationship (SAR)
5.2.1. Foundational Structural Requirements and Stereochemical Precision
5.2.2. Functional Group Dependence and Mechanistic Shift
5.2.3. SAR Challenges and Translational Optimization
6. Discussion and Translational Direction
6.1. The Imperative for Chemical Optimization and Analogue Development
6.2. Standardization and Bioassay-Guided Isolation
- Addressing Chemical Variability: The measured IC50 and MIC values are susceptible to ontogenetic and geographical chemical variation, as the relative percentage of key sesquiterpenes can fluctuate significantly depending on the plant’s maturity [68]. Future work must implement high-resolution analytical standards like HPLC profiling to define standardized chemotypes and minimize this variability, thereby ensuring the reproducibility of the SI calculation.
- Genetic Anchoring: The successful assembly of the complete plastome for V. alternifolia provides a crucial genetic reference point, which should be leveraged to map and minimize the impact of genetic and chemotypic variability when working with new accessions [16].
- Targeted Toxicity Assessment: Rigorous SI calculation requires toxicity profiles (IC50 on non-cancerous lines) for every potent isolate to bridge the gap between high efficacy and unassessed safety [55]. Furthermore, the scarcity of information on many ethnobotanically important species, such as V. crocata, demands comprehensive characterization of active principles and toxicity assessments to elaborate safe pharmaceutical products [23].
- Biotechnology for Consistency: To circumvent the challenges of field-collected material, the successful biosynthesis of key triterpenoids like lupeol in V. encelioides cell cultures offers a high-value pathway for the scale-up and standardization of non-toxic support molecules [94]. This difficulty has prompted translational research into novel process engineering. A specific process patent demonstrates the development of an optimized hexane extraction method for V. persicifolia that dramatically increases the yield and purity of the lead eudesmane 4CDE (up to 3 g/kg of fresh material), successfully solving the scalability challenge for this high-value scaffold [104].
7. Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grichar, W.J.; Sestak, D.C. Control of Golden Crownbeard (Verbesina encelioides) in Peanut (Arachis hypogaea) with Postemergence Herbicides. Peanut Sci. 1998, 25, 39–43. [Google Scholar] [CrossRef]
- Panero, J.L.; Jansen, R.K.; Clevinger, J.A. Phylogenetic relationships of subtribe Ecliptinae (Asteraceae: Heliantheae) based on chloroplast DNA restriction site data. Am. J. Bot. 1999, 86, 413–427. [Google Scholar] [CrossRef] [PubMed]
- Inderjit; Asakawa, C.; Dakshini, K.M.M. Allelopathic potential of Verbesina encelioides root leachate in soil. Can. J. Bot. 2000, 77, 1419–1424. [Google Scholar] [CrossRef]
- Chappelka, A.H.; Neufeld, H.S.; Davison, A.W.; Somers, G.L.; Renfro, J.R. Ozone Injury on Cutleaf Coneflower (Rudbeckia laciniata) and Crown-Beard (Verbesina occidentalis) in Great Smoky Mountains National Park. Environ. Pollut. 2003, 125, 53–59. [Google Scholar] [CrossRef]
- Grichar, W.J.; Jordan, D.L.; Prostko, E.P. Weed control and peanut (Arachis hypogaea L.) response to formulations of imazapic. Crop Prot. 2012, 36, 31–36. [Google Scholar] [CrossRef]
- Evans, B.; Borowicz, V. Verbesina alternifolia Tolerance to the Holoparasite Cuscuta gronovii and the Impact of Drought. Plants 2013, 2, 635–649. [Google Scholar] [CrossRef] [PubMed]
- Carmona-Chit, E.; Carrillo-González, R.; González-Chávez, M.d.C.A.; Vibrans, H.; Yáñez-Espinosa, L.; Delgado-Alvarado, A. Riparian plants on mine runoff in Zimapan, Hidalgo, Mexico: Useful for phytoremediation? Int. J. Phytoremediation 2016, 18, 861–868. [Google Scholar] [CrossRef]
- Sharma, R.; Chandel, V.; Rishi, N. A new variant of Croton yellow vein mosaic virus naturally infecting wild sunflower in India. VirusDis. 2018, 29, 513–519. [Google Scholar] [CrossRef]
- Rivera, P.; Villaseñor, J.L.; Terrazas, T.; Panero, J.L. The importance of the Mexican taxa of Asteraceae in the family phylogeny. J. Syst. Evol. 2021, 59, 935–952. [Google Scholar] [CrossRef]
- Kaur, A.; Kaur, S.; Jandrotia, R.; Singh, H.P.; Batish, D.R.; Kohli, R.K.; Rana, V.S.; Shakil, N.A. Parthenin—A Sesquiterpene Lactone with Multifaceted Biological Activities: Insights and Prospects. Molecules 2021, 26, 5347. [Google Scholar] [CrossRef]
- Kaur, K.; Sidhu, M.C.; Ahluwalia, A.S. A Systematic Review of an Invasive Plant Species: Verbesina encelioides (Cav.) Benth. & Hook. f. Ex A. Gray. Int. J. Pharm. Sci. Res. 2022, 13, 3824–3832. [Google Scholar] [CrossRef]
- Velasco-Ramírez, A.P.; Velasco-Ramírez, A.; Hernández-Herrera, R.M.; Ceja-Esquivez, J.; Velasco-Ramírez, S.F.; Ramirez-Anguiano, A.C.; Torres-Morán, M.I. The Impact of Aqueous Extracts of Verbesina sphaerocephala and Verbesina fastigiata on Germination and Growth in Solanum lycopersicum and Cucumis sativus Seedlings. Horticulturae 2022, 8, 652. [Google Scholar] [CrossRef]
- Velasco-Ramírez, A.P.; Velasco-Ramírez, A.; Hernández-Herrera, R.M.; Murguia-Lizalde, D.M.; Velasco-Ramírez, S.F. Extractos de Verbesina sphaerocephala y Verbesina fastigiata promueven el desarrollo en maceta de Fragaria ananassa. Agroind. Sci. 2022, 12, 29–37. [Google Scholar] [CrossRef]
- Mehal, K.K.; Kaur, A.; Singh, H.P.; Batish, D.R. Investigating the phytotoxic potential of Verbesina encelioides: effect on growth and performance of co-occurring weed species. Protoplasma 2023, 260, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.-X.; Zhao, H.-Y.; Peng, C.; Liu, F.; Xiong, L. Eudesmane-type sesquiterpenoids: Structural diversity and biological activity. Heliyon 2024, 10, e35270. [Google Scholar] [CrossRef]
- Tomasello, S.; Manzo, E.; Karbstein, K. Comparative plastome assembly of the yellow ironweed (Verbesina alternifolia) using Nanopore and Illumina reads. Front. Plant Sci. 2024, 15, 1429494. [Google Scholar] [CrossRef]
- Mehal, K.K.; Sharma, A.; Kaur, A.; Kalia, N.; Kohli, R.K.; Singh, H.P.; Batish, D.R. Modelling the ecological impact of invasive weed Verbesina encelioides on vegetation composition across dryland ecosystems of Punjab, northwestern India. Environ. Monit. Assess. 2023, 195, 725. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Pérez, A.R.; Hernández-Herrera, R.M.; Velasco-Ramírez, S.F.; Velasco-Ramírez, A.; Ramírez-Anguiano, A.C.; Velasco-Ramírez, A.P. Effect of Botanical Extracts from Verbesina sphaerocephala on the Growth and Yield of Tomato Under Greenhouse Conditions. Acta Agrobot. 2025, 78, 201390. [Google Scholar] [CrossRef]
- Verma, V.; Rao, L.; Chaudhary, M.; Jain, S.; Pinapati, K.K.; Dey, A.; Sharma, S.; Srivastava, N. Spirilloxanthin from Verbesina encelioides Flower Extract has Neuroprotective Benefits in Scopolamine-Induced Memory Impaired Rats that Resemble Alzheimer’s Disease. Mol. Neurobiol. 2025, 62, 12602–12615. [Google Scholar] [CrossRef] [PubMed]
- Herz, W.; Kumar, N. Aromatic and other constituents of four Verbesina species: Structure and stereochemistry of verbesindiol. Phytochemistry 1981, 20, 247–250. [Google Scholar] [CrossRef]
- Mora, F.D.; Alpan, L.; McCracken, V.J.; Nieto, M. Chemical and Biological Aspects of the Genus Verbesina. Nat. Prod. J. 2013, 3, 140–150. [Google Scholar] [CrossRef]
- García-Bores, A.M.; Álvarez-Santos, N.; López-Villafranco, M.E.; Jácquez-Ríos, M.P.; Aguilar-Rodríguez, S.; Grego-Valencia, D.; Espinosa-González, A.M.; Estrella-Parra, E.A.; Hernández-Delgado, C.T.; Serrano-Parrales, R.; González-Valle, M. R.; Benítez-Flores, J. C. Verbesina crocata: A Pharmacognostic Study for the Treatment of Wound Healing. Saudi J. Biol. Sci. 2020, 27, 3113–3124. [Google Scholar] [CrossRef]
- Heyerdahl-Viau, I.; López-Naranjo, F.; Córdova-Moreno, R.; Urbina-Pastrana, A.; Martínez-Núñez, J.M. A Brief Review of Verbesina crocata (Cav.) Less., a Scantly Studied Medicinal Plant. Res. J. Pharmacogn. 2023, 10, 51–56. [Google Scholar] [CrossRef]
- Bohlmann, F.; Zdero, C.; Robinson, H.; King, R.M. Two New Eudesmane Cinnamates from Verbesina sordescens. Phytochemistry 1982, 21, 1663–1664. [Google Scholar] [CrossRef]
- Albalawi, M.A.D.; Bashir, N.A.O.; Tawfik, A. Anticancer and Antifolate Activities of Extracts of Six Saudi Arabian Wild Plants Used in Folk Medicine. J. Life Sci. 2015, 9, 334–340. [Google Scholar] [CrossRef]
- Farshori, N.N. Verbesina encelioides-Induced Cytotoxicity and Mitochondria-Mediated Apoptosis in Human Colon Cancer Cells through ROS Generation. Biologia 2021, 76, 2711–2720. [Google Scholar] [CrossRef]
- Oelrichs, P.B.; Vallely, P.J.; MacLeod, J.K.; Lewis, I.A.S. Isolation of Galegine from Verbesina enceloiodes. J. Nat. Prod. 1981, 44, 754–755. [Google Scholar] [CrossRef]
- Indrayanto, G.; Putra, G.S.; Suhud, F. Chapter Six - Validation of in-vitro bioassay methods: Application in herbal drug research. In Profiles of Drug Substances, Excipients and Related Methodology; Elsevier: Amsterdam, The Netherlands, 2021; Volume 46, pp. 273–307. [Google Scholar] [CrossRef]
- Villaseñor, J.L.; Ortiz, E. La Familia Asteraceae en la Flora del Bajío y de Regiones Adyacentes. Acta Bot. Mex. 2012, 100, 259–291. [Google Scholar] [CrossRef]
- Van Auken, O.W.; Leonard, W.J. Why is Verbesina virginica (Frostweed, Asteraceae) Not Found in Grasslands? Phytologia 2016, 98, 76–88. https://www.phytologia.org/uploads/2/3/4/2/23422706/98_1_76-88vanauken_verbesina_in_grasslands_rev_12-29-15.pdf.
- Kaur, K.; Ramanpreet; Gupta, R.C.; Kumari, S. Cyto-Morphological Studies of Some Dicot Plants from Rajasthan (India). Cytologia 2015, 80, 353–362. [Google Scholar] [CrossRef]
- Domínguez-Barradas, C.; Cruz-Morales, G.E.; González-Gándara, C. Plantas de uso medicinal de la Reserva Ecológica "Sierra de Otontepec", municipio de Chontla, Veracruz, México. CienciaUAT 2015, 9, 41–52. [Google Scholar] [CrossRef]
- Perez G., R.M.; Perez G., S.; Zavala, M.A.; Perez G., S.C. Effect of Agarista mexicana and Verbesina persicifolia on Blood Glucose Level of Normoglycaemic and Alloxan-diabetic Mice and Rats. Phytother. Res. 1996, 10, 351–353. [Google Scholar] [CrossRef]
- Dalla Via, L.; Mejia, M.; García-Argáez, A.N.; Braga, A.; Toninello, A.; Martínez-Vázquez, M. Anti-Inflammatory and Antiproliferative Evaluation of 4β-Cinnamoyloxy,1β,3α -Dihydroxyeudesm-7,8-ene from Verbesina persicifolia and Derivatives. Bioorg. Med. Chem. 2015, 23, 5816–5828. [Google Scholar] [CrossRef]
- Martínez Alfaro, M.Á.; Evangelista Oliva, V.; Mendoza Cruz, M.; Morales Garcia, G.; Toledo Olazcoaga, G.; Wong León, A. Catálogo de plantas útiles de la Sierra Norte de Puebla, México. Cuadernos IBUNAM 27. Universidad Nacional Autónoma de México, Instituto de Biología, 2001. https://cepe-taxco.bibliotecas.unam.mx:84/cgi-bin/koha/opac-detail.pl?biblionumber=2513.
- Rodríguez-Valdovinos, K.Y.; Salgado-Garciglia, R.; Vázquez-Sánchez, M.; Álvarez-Bernal, D.; Oregel-Zamudio, E.; Ceja-Torres, L.F.; Medina-Medrano, J.R. Quantitative Analysis of Rutin by HPTLC and In Vitro Antioxidant and Antibacterial Activities of Phenolic-Rich Extracts from Verbesina sphaerocephala. Plants 2021, 10, 475. [Google Scholar] [CrossRef]
- López-Rosas, C.A.; González-Periañez, S.; Pawar, T.J.; Zurutuza-Lorméndez, J.I.; Ramos-Morales, F.R.; Olivares-Romero, J.L.; Saavedra Vélez, M.V.; Hernández-Rosas, F. Anticonvulsant Potential and Toxicological Profile of Verbesina persicifolia Leaf Extracts: Evaluation in Zebrafish Seizure and Artemia salina Toxicity Models. Plants 2025, 14, 1078. [Google Scholar] [CrossRef]
- Mora, F.D.; Alpan, L.; De Tommasi, N.; McCracken, V.J.; Nieto, M. New Antibacterial Germacrene from Verbesina negrensis. Planta Med. 2013, 79, 707–710. [Google Scholar] [CrossRef]
- Veras, B.O.d.; Oliveira, J.R.S.d.; Lima, V.L.M.d.M.; Navarro, D.M.d.A.F.; Aguiar, J.C.R.d.O.F.d.; Mourad, G.M.d.M.; Silva, J.W.d.; Assis, C.R.D.d.; Gorlach-Lira, K.; Assis, P.A.C.d.; Barbosa, J.I.d.S.; Melo, M.R.C.S.d.; Oliveira, M.B.M.d.; Silva, M.V.d.; Lopes, A.C.S. The essential oil of the leaves of Verbesina macrophylla (Cass.) S.F.Blake has antimicrobial, anti-inflammatory and antipyretic activities and is toxicologically safe. J. Ethnopharmacol. 2021, 265, 113248. [Google Scholar] [CrossRef]
- Al-Sodany, Y.M.; Issa, A.A.; Kahil, A.A.; Ali, E.F. Diversity of Soil Cyanobacteria in Relation to Dominant Wild Plants and Edaphic Factors at Western Saudi Arabia. Annu. Res. Rev. Biol. 2018, 26, 1–14. [Google Scholar] [CrossRef]
- Essinger, J.F., Jr.; Gan, L.; Behrens, N.J. Stable Liquid Formulations and Methods of Using the Same. U.S. Patent Application 20200260722 A1, 20 August 2020. https://portal.unifiedpatents.com/patents/patent/US-11109588-B2.
- Grichar, W.J.; Sestak, D.C. Herbicide Systems for Golden Crownbeard (Verbesina encelioides) Control in Peanut. Peanut Sci. 2000, 27, 23–26. [Google Scholar] [CrossRef]
- Van Nuland, M.E.; Haag, E.N.; Bryant, J.A.M.; Read, Q.D.; Klein, R.N.; Douglas, M.J.; Gorman, C.E.; Greenwell, T.D.; Busby, M.W.; Collins, J.; LeRoy, J.T.; Schuchmann, G.; Schweitzer, J.A.; Bailey, J.K. Fire Promotes Pollinator Visitation: Implications for Ameliorating Declines of Pollination Services. PLoS ONE 2013, 11, e79853. [Google Scholar] [CrossRef] [PubMed]
- Aschenbrenner, A.-K.; Horakh, S.; Spring, O. Linear Glandular Trichomes of Helianthus (Asteraceae): Morphology, Localization, Metabolite Activity and Occurrence. AoB PLANTS 2013, 5, plt028. [Google Scholar] [CrossRef]
- Sultana, S.; Ali, M.; Mir, S.R. Chemical Constituents From the Aerial Roots of Ficus benghalensis L., Leaves of Nyctanthes arbor-tristis L. and Roots of Verbesina encelioides (Cav.) Benth. et Hook. f. J. Pharm. Biosci. 2018, 6, 16–26. [Google Scholar] [CrossRef]
- Bonchev, G.N.; Vassilevska-Ivanova, R. Fingerprinting the genetic variation and intergeneric hybrid dynamics in the family Asteraceae (genera Helianthus, Echinaceae, Tagetes and Verbesina) using iPBS markers. Biologia 2019, 75, 457–464. [Google Scholar] [CrossRef]
- Wu, Q.-X.; Shi, Y.-P.; Jia, Z.-J. Eudesmane Sesquiterpenoids from the Asteraceae Family. Nat. Prod. Rep. 2006, 23, 699–734. [Google Scholar] [CrossRef]
- Gardner, P.D.; Park, G.J.; Albers, C.C. α- and β-Verbesinol. Sesquiterpene Alcohols of the cis-Decalin Series. J. Am. Chem. Soc. 1961, 83, 1511–1512. [Google Scholar] [CrossRef]
- Bohlmann, F.; Lonitz, M. Neue Eudesman-Derivate und andere Sesquiterpene aus Verbesina-Arten [New Eudesmane Derivatives and Other Sesquiterpenes from Verbesina Species]. Chem. Ber. 1978, 111, 254–263. [Google Scholar] [CrossRef]
- Jakupovic, J.; Ellmauerer, E.; Jia, Y.; Bohlmann, F.; Dominguez, X.A.; Schmeda-Hirschmann, G. Further Eudesmane Derivatives from Verbesina Species. Planta Med. 1987, 53, 39–42. [Google Scholar] [CrossRef]
- Bohlmann, F.; Grenz, M.; Gupta, R.K.; Dhar, A.K.; Ahmed, M.; King, R.M.; Robinson, H. Eudesmane Derivatives from Verbesina Species. Phytochemistry 1980, 19, 2391–2397. [Google Scholar] [CrossRef]
- Amaro-Luis, J.M.; Ramírez, I.; Delgado-Méndez, P.; Jorge, Z.D. Eudesmane Derivatives from Verbesina turbacensis. J. Braz. Chem. Soc. 2002, 13, 352–357. [Google Scholar] [CrossRef]
- Box, V.G.S.; Chan, W.R.; Taylor, D.R. The Structure of Rupestrol a New Sesquiterpenoid from Verbesina rupestris (Urb.) Blake. Tetrahedron Lett. 1971, 12, 4371–4374. [Google Scholar] [CrossRef]
- Box, V.G.S.; Bardouille, V.; Chan, W.R. Enantio-Eudesmane Sesquiterpenes from Verbesina rupestris. Phytochemistry 1977, 16, 987–990. [Google Scholar] [CrossRef]
- Ramseyer, J.; Thuerig, B.; De Mieri, M.; Schärer, H.-J.; Oberhänsli, T.; Gupta, M.P.; Tamm, L.; Hamburger, M.; Potterat, O. Eudesmane Sesquiterpenes from Verbesina lanata with Inhibitory Activity against Grapevine Downy Mildew. J. Nat. Prod. 2017, 80, 3296–3304. [Google Scholar] [CrossRef]
- Herz, W.; Kumar, N.; Blount, J.F. Crystal Structure and Stereochemistry of Verbesindiol. J. Org. Chem. 1982, 47, 1785–1786. [Google Scholar] [CrossRef]
- Banerjee, S.; Jakupovic, J.; Bohlmann, F.; King, R.M.; Robinson, H. A Rearranged Eudesmane and Further Verbesindiol Derivatives from Verbesina eggersii. Phytochemistry 1985, 24, 1106–1108. [Google Scholar] [CrossRef]
- Martínez, M.; de Vivar, A.R.; Ortega, A.; Quintero, M. de L.; García, C.; Fronczek, F.R. Eudesmane Triols from Verbesina virgata. Phytochemistry 1983, 22, 979–982. [Google Scholar] [CrossRef]
- Xu, W.H.; Liang, Q.; Liang, Z.S.; Li, X.C. Two New Eudesmane Derivatives from Verbesina virginica. Chin. Chem. Lett. 2010, 21, 693–695. [Google Scholar] [CrossRef]
- Bruno-Colmenárez, J.; Amaro-Luis, J.M.; Ramírez, I.; Delgado-Méndez, P.; Díaz de Delgado, G. Absolute Configuration of Two Eudesmane Derivatives from Verbesina turbacensis H.B.K. J. Chem. Crystallogr. 2010, 40, 110–115. [Google Scholar] [CrossRef]
- Vazquez Cadanedo, A.; Díaz-T, E.; Jankowski, C.K.; Dako, E.; Doiron, J.; Jiménez-E, M. On the Structure of Triolcinnamate from Verbesina persicifolia. Indian J. Chem. B 2015, 54, 1342–1345. https://www.researchgate.net/publication/283489292_On_the_structure_of_triolcinnamate_from_Verbesina_persicifolia.
- Dalla Via, L.; García-Argáez, A.N.; Braga, A.; Martínez-Vázquez, M.; Grancara, S.; Martinis, P.; Agostinelli, E.; Toninello, A. An Eudesman Derivative from Verbesina persicifolia D.C. as a Natural Mild Uncoupler in Liver Mitochondria. A New Potential Anti-obesity Agent? Curr. Pharm. Des. 2014, 20, 253–261. [Google Scholar] [CrossRef]
- Arciniegas, A.; Pérez-Castorena, A.L.; Villaseñor, J.L.; Romo de Vivar, A. Cadinenes and other metabolites from Verbesina sphaerocephala A. Gray. Biochem. Syst. Ecol. 2020, 93, 104183. [Google Scholar] [CrossRef]
- Ortega, A.; Maldonado, E.; Fronczek, F.R.; Delord, T.J.; Chiari, G. Elemanolides from Verbesina seattonii. Phytochemistry 1985, 24, 1755–1760. [Google Scholar] [CrossRef]
- Selener, M.G.; Elso, O.; Grosso, C.; Borgo, J.; Clavin, M.; Malchiodi, E.L.; Cazorla, S.I.; Flavia, F.; Sülsen, V.P. Anti-Trypanosoma cruzi Activity of Extracts from Argentinean Asteraceae Species. Iran. J. Pharm. Res. 2019, 18, 1854–1861. [Google Scholar] [CrossRef]
- Bohlmann, F.; Lonitz, M. Neue Sesquiterpene aus Verbesina occidentalis. Phytochemistry 1978, 17, 453–455. [Google Scholar] [CrossRef]
- Salmón, M.; Soriano-Garcia, M.; Toscano, R.A.; Cárdenas, J.; Miranda, R.; Vargas, F.; Angeles, E. Verocephol, a Unique Amorphane Sesquiterpene γ-Lactol. J. Org. Chem. 1985, 50, 4171–4172. [Google Scholar] [CrossRef]
- Albuquerque, M.R.J.R.; Canuto, K.M.; Pessoa, O.D.L.; Nunes, E.P.; Nascimento, R.F.; Silveira, E.R. Essential Oil Composition of Verbesina diversifolia DC. Flavour Fragr. J. 2006, 21, 634–636. [Google Scholar] [CrossRef]
- Bezerra, L.D.A.; Mangabeira, P.A.O.; de Oliveira, R.A.; Costa, L.C.D.B.; Da Cunha, M. Leaf Blade Structure of Verbesina macrophylla (Cass.) F. S. Blake (Asteraceae): Ontogeny, Duct Secretion Mechanism and Essential Oil Composition. Plant Biol. 2018, 20, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Mora, F.D.; Rojas, Y.L.; González, V.; Velasco, J.; Díaz, T.; Ríos, N.; Rojas-Fermin, L.B.; Carmona, J.; Silva, B.; Nieto, M. Chemical Composition and in vitro Antibacterial Activity of the Essential Oil of Verbesina negrensis from the Venezuelan Andes. Nat. Prod. Commun. 2015, 10, 1309–1310. [Google Scholar] [CrossRef]
- Box, V.G.S.; Chan, W.R. Terpenoids from Verbesina rupestris. Phytochemistry 1975, 14, 583. [Google Scholar] [CrossRef]
- Ogungbe, I.V.; Crouch, R.A.; Haber, W.A.; Setzer, W.N. Phytochemical Investigation of Verbesina turbacensis Kunth: Trypanosome Cysteine Protease Inhibition by (-)-Bornyl Esters. Nat. Prod. Commun. 2010, 5, 1161–1166. [Google Scholar] [CrossRef] [PubMed]
- Caballero-Gallardo, K.; Alvarez-Ortega, N.; Olivero-Verbel, J. Cytotoxicity of Nine Medicinal Plants from San Basilio de Palenque (Colombia) on HepG2 Cells. Plants 2023, 12, 2686. [Google Scholar] [CrossRef]
- Fawzy, M.A.; Hifney, A.F.; Issa, A.A.; Gareib, G. Phytochemical Constituents and Allelopathic Effects of Some Medicinal Plants Extract on the Soil Algal Diversity. J. Agric. Sci. Technol. A 2013, 3, 1000–1009. https://qikan.cqvip.com/Qikan/Article/Detail?id=48303743&from=Qikan_Search_Index.
- Botta, B.; Carmignani, M.; Volpe, A.R.; Botta, M.; Corelli, F.; Delle Monache, G. Novel Hypotensive Agents from Verbesina Caracasana: Structure, Synthesis and Pharmacology. Curr. Med. Chem. 2003, 10, 1845–1862. [Google Scholar] [CrossRef]
- Eichholzer, J.V.; Lewis, I.A.S.; MacLeod, J.K.; Oelrichs, P.B.; Vallely, P.J. Galegine and a new dihydroxyalkylacetamide from Verbesina enceloiodes. Phytochemistry 1982, 21, 97–99. [Google Scholar] [CrossRef]
- Delle Monache, G.; Delle Monache, F.; Carmignani, M.; Bonnevaux, S.C.; Espinal, R.; De Luca, C.; Botta, B. Guanidine Derivatives Having Hypotensive Activity, Composition Containing Them and Process for Obtaining Them. U.S. Patent 5,059,624, 22 October 1991. https://worldwide.espacenet.com/patent/search/family/011261768/publication/EP0330629A2?q=EP330629A2.
- Delle Monache, G.; Botta, B.; Delle Monache, F.; Espinal, R.; De Bonnevaux, S.C.; De Luca, C.; Botta, M.; Corelli, F.; Dei, D.; Gacs-Baitz, E.; Carmignani, M. Caracasandiamide, a Truxinic Hypotensive Agent from Verbesina caracasana. Bioorg. Med. Chem. Lett. 1996, 6, 233–238. [Google Scholar] [CrossRef]
- Delle Monache, G.; Botta, B.; Delle Monache, F.; Espinal, R.; De Bonnevaux, S.C.; De Luca, C.; Botta, M.; Corelli, F.; Carmignani, M. Caracasanamide, a Novel Hypotensive Agent from Verbesina caracasana. Bioorg. Med. Chem. Lett. 1992, 2, 415–418. [Google Scholar] [CrossRef]
- Delle Monache, G.; Botta, B.; Delle Monache, F.; Espinal, R.; De Bonnevaux, S.C.; De Luca, C.; Botta, M.; Corelli, F.; Carmignani, M. Novel Hypotensive Agents from Verbesina caracasana. 2. Synthesis and Pharmacology of Caracasanamide. J. Med. Chem. 1993, 36, 2956–2963. [Google Scholar] [CrossRef]
- Carmignani, M.; Volpe, A.R.; Delle Monache, F.; Botta, B.; Espinal, R.; De Bonnevaux, S.C.; De Luca, C.; Botta, M.; Corelli, F.; Tafi, A.; Ripanti, G.; Delle Monache, G. Novel Hypotensive Agents from Verbesina caracasana. 6. Synthesis and Pharmacology of Caracasandiamide. J. Med. Chem. 1999, 42, 3116–3125. [Google Scholar] [CrossRef] [PubMed]
- Delle Monache, G.; Volpe, A.R.; Delle Monache, F.; Vitali, A.; Botta, B.; Espinal, R.; De Bonnevaux, S.C.; De Luca, C.; Botta, M.; Corelli, F.; Camignani, M. Further Hypotensive Metabolites from Verbesina caracasana. Bioorg. Med. Chem. Lett. 1999, 9, 3249–3254. [Google Scholar] [CrossRef]
- Carmignani, M.; Volpe, A.R.; Botta, B.; Espinal, R.; De Bonnevaux, S.C.; De Luca, C.; Botta, M.; Corelli, F.; Tafi, A.; Sacco, R.; Delle Monache, G. Novel Hypotensive Agents from Verbesina caracasana. 8. Synthesis and Pharmacology of (3,4-Dimethoxycinnamoyl)-N1-agmatine and Synthetic Analogs. J. Med. Chem. 2001, 44, 2950–2958. [Google Scholar] [CrossRef]
- Compagnone, R.S.; Bermudez, J.; Ibáñez, G.; Díaz, B.; Garrido, M.R.; Israel, A.; Suárez, A.I. New Guanidine Alkaloids from the Leaves of Verbesina peraffinis. Nat. Prod. Commun. 2008, 3, 511–514. [Google Scholar] [CrossRef]
- Rodríguez-Valdovinos, K.Y.; Salgado-Garciglia, R.; Hernández-García, A.; Saavedra-Molina, A.; del Río-Torres, R.E.N.; López-Meza, J.E.; Monribot-Villanueva, J.L.; Guerrero-Analco, J.A.; Medina-Medrano, J.R. Antioxidant and Antifungal Activities and Characterization of Phenolic Compounds Using Ultra-High Performance Liquid Chromatography and Mass Spectrometry (UPLC-MS) of Aqueous Extracts and Fractions from Verbesina sphaerocephala Stems. Plants 2024, 13, 2791. [Google Scholar] [CrossRef]
- Glennie, C.W.; Jain, S.C. Flavonol 3,7-Diglycosides of Verbesina enceloiodes. Phytochemistry 1980, 19, 157–158. [Google Scholar] [CrossRef]
- Abbas, F.A.; El Sayed, Z.I.; Dora, G.A.; Ateya, A.-M.; Samy, S. Phytochemical and Biological Studies of Verbesina encelioides (Cav.) Benth. and Hook. Asian J. Phytomed. Clin. Res. 2016, 4, 108–120. http://www.ajpcrjournal.com/article/PHYTOCHEMICAL%20AND%20BIOLOGICAL%20STUDIES%20OF%20VERBESINA_ENCELIOIDES%20(CAV.)%20BENTH%20AND%20HOOK.pdf.
- Wagner, H.; Iyengar, M.A.; Seligmann, O.; Herz, W. Rhamnocitrin-3-Glucuronid in Verbesina myricephala. Phytochemistry 1974, 13, 493–494. [Google Scholar] [CrossRef]
- Bohlmann, F.; Zdero, C. Neue Terpen-Inhaltsstoffe aus Verbesina-Arten. Phytochemistry 1976, 15, 1310–1311. [Google Scholar] [CrossRef]
- Farber, C.; Sanchez, L.; Kurouski, D. Confirmatory Non-Invasive and Non-Destructive Identification of Poison Ivy Using a Hand-Held Raman Spectrometer. RSC Adv. 2020, 10, 21530–21534. [Google Scholar] [CrossRef]
- Burkey, K.O.; Neufeld, H.S.; Souza, L.; Chappelka, A.H.; Davison, A.W. Seasonal Profiles of Leaf Ascorbic Acid Content and Redox State in Ozone-Sensitive Wildflowers. Environ. Pollut. 2006, 143, 427–434. [Google Scholar] [CrossRef]
- Verma, V.; Chaudhary, M.; Srivastava, N. Antioxidative Properties of Isolated Saponins of Verbesina encelioides (Cav.) Benth. & Hook. f. ex Gray and SEM Studies of Synthesized Green Nanoparticles for Acne Management. Plant Sci. Today 2019, 6, 575–582. [Google Scholar] [CrossRef]
- Xu, W.-H.; Jacob, M.R.; Agarwal, A.K.; Clark, A.M.; Liang, Z.-S.; Li, X.-C. Verbesinosides A−F, 15,27-Cyclooleanane Saponins from the American Native Plant Verbesina virginica. J. Nat. Prod. 2009, 72, 1022–1027. [Google Scholar] [CrossRef] [PubMed]
- Cerda-Garcia-Rojas, C.M.; Zamorano, G.; Chávez, M.I.; Catalán, C.A.N.; Joseph-Nathan, P. 1H and 13C NMR Study of Copteroside E Derivatives. Magn. Reson. Chem. 2000, 38, 494–499. [Google Scholar] [CrossRef]
- Singh, R.; Jain, R. Biopotentialities of Verbesina encelioides Cell Cultures. Indian J. Plant Physiol. 2009, 13, 224–230. https://www.semanticscholar.org/paper/BIOPOTENTIALITIES-OF-VERBESINA-ENCELIOIDES-CELL-Singh-Jain/cfc158b31d2d1dd7db47d5690a8c583b0f788dc6.
- Christensen, L.P.; Lam, J. Acetylenes and Related Compounds in Heliantheae. Phytochemistry 1991, 30, 11–49. [Google Scholar] [CrossRef]
- Bohlmann, F.; Zdero, C. Zur konfiguration von α- und β-Verbesinolcumarat. Phytochemistry 1979, 18, 1751–1752. [Google Scholar] [CrossRef]
- Pergomet, J.L.; Larghi, E.L.; Kaufman, T.S.; Bracca, A.B.J. First Total Synthesis of the Only Known 2-Isopropyliden-2H-Benzofuran-3-one Isolated from V. luetzelburgii. RSC Adv. 2017, 7, 5242–5250. [Google Scholar] [CrossRef]
- Divya Ramakrishnan, C.K.; Doss, D.; Vijayabharathi, A. Biochemical and Antimicrobial Characterization of an Underexploited Medicinal Plant Verbesina encelioides. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 3407–3416. [Google Scholar] [CrossRef]
- Sindhu, R.K.; Vasudeva, N.; Sharma, S.K. Pharmacognostical and Preliminary Phytochemical Investigations on Verbesina encelioides Benth. Roots. J. Herb. Med. Toxicol. 2010, 4, 113–118. https://www.researchgate.net/publication/239524726_Pharmacognostical_and_preliminary_phytochemical_investigations_on_Verbesina_encelioides_Benth_Roots.
- Kushwaha, H.B.; Malik, C.P. Nanofabrication of Silver Nanoparticles From the Stem and Leaf Extract of Verbesina encelioides. Natl. Acad. Sci. Lett. 2012, 35, 555–563. [Google Scholar] [CrossRef]
- Ramírez-Herrera, A.D.; Ceja-Buenrostro, J. de J.; Miranda-Torres, A.; Reynoso-Marín, F.J. Verbesina crocata como agente biosintético de nanopartículas de plata/cloruro de plata y su evaluación en plántulas de rábano. Tópicos Investig. Cienc. Tierra Mater. 2025, 12, 38–45. [Google Scholar] [CrossRef]
- Shaffer, C.V.; Cai, S.; Peng, J.; Robles, A.J.; Hartley, R.M.; Powell, D.R.; Du, L.; Cichewicz, R.H.; Mooberry, S.L. Texas Native Plants Yield Compounds with Cytotoxic Activities against Prostate Cancer Cells. J. Nat. Prod. 2016, 79, 531–540. [Google Scholar] [CrossRef]
- Universidad Nacional Autónoma de México. Process for obtaining eudesmane sesquiterpene 4a-cinnamoiloxy-1b,3a-dihydroxyeudesm-7,8-eno(4CDE). MX 2013014284 A. 5 June 2015. https://worldwide.espacenet.com/patent/search/family/054261219/publication/MX2013014284A?q=MX2013014284A.
- Al-Oqail, M.M.; Siddiqui, M.A.; Al-Sheddi, E.S.; Saquib, Q.; Musarrat, J.; Al-Khedhairy, A.A.; Farshori, N.N. Verbesina encelioides: Cytotoxicity, Cell Cycle Arrest, and Oxidative DNA Damage in Human Liver Cancer (HepG2) Cell Line. BMC Complement. Altern. Med. 2016, 16, 126. [Google Scholar] [CrossRef]
- Ezzat, S.M.; Salama, M.M.; Mahrous, E.A.; Maes, L.; Pan, C.-H.; Abdel-Sattar, E. Antiprotozoal Activity of Major Constituents from the Bioactive Fraction of Verbesina encelioides. Nat. Prod. Res. 2016, 31, 676–680. [Google Scholar] [CrossRef]
- Abdel-Wahab, A.; Abdel-Maogood, S.Z.; Mahdy, O.A. Scanning Electron Microscopic Changes in Habronema muscae after In Vitro Exposure to Plant Extract (Verbesina alternifolia). J. Egypt. Soc. Parasitol. 2017, 47, 47–54. https://jesp.journals.ekb.eg/article_77965_1c54bbae5f06ac169e35f7a1a9176f73.pdf. [CrossRef]
- Bhati-Kushwaha, H.; Malik, C.P. Biopotential of Verbesina encelioides (Stem and Leaf Powders) in Silver Nanoparticle Fabrication. Turk. J. Biol. 2013, 37, 645–654. [Google Scholar] [CrossRef]
- Wankhade, S.; Rai, M.K. A Product and Process of Extraction of Asteraceae Plant Material for Treatment of Dermatophyte Infections. IN 3395/MUM/2010. 14 December 2010. https://patentscope.wipo.int/search/en/detail.jsf?docId=IN211588671.
- Bezerra, L.A.; Carvalho, L.P.; Mello, É. de O.; Taveira, G.B.; de Melo, E.J.T.; Gomes, V.M.; da Cunha, M. Fungicidal and trypanocidal activity of aqueous extracts of leaves and inflorescences of Verbesina macrophylla (Asteraceae). Acta Sci. Biol. Sci. 2024, 46, e71523. [Google Scholar] [CrossRef]
- Oka, Y. Nematicidal Activity of Verbesina encelioides against the Root-Knot Nematode Meloidogyne javanica and Effects on Plant Growth. Plant Soil 2012, 355, 311–322. [Google Scholar] [CrossRef]
- Lahlali, M.; Manaut, N.; Loudiki, M.; Douma, M. First Assessment of the Anti-Cyanobacterial Potentialities of the Invasive Weed Verbesina encelioides Against Microcystis aeruginosa Growth. Desalination Water Treat. 2023, 310, 150–156. [Google Scholar] [CrossRef]
- Yaseen, R.; Branitzki-Heinemann, K.; Moubasher, H.; Setzer, W.N.; Naim, H.Y.; von Köckritz-Blickwede, M. In Vitro Testing of Crude Natural Plant Extracts from Costa Rica for Their Ability to Boost Innate Immune Cells against Staphylococcus aureus. Biomedicines 2017, 5, 40. [Google Scholar] [CrossRef] [PubMed]
- Corelli, F.; Dei, D.; Delle Monache, G.; Botta, B.; De Luca, C.; Carmignani, M.; Volpe, A.R.; Botta, M. Synthesis and Preliminary Pharmacological Evaluation of Analogues of Caracasanamide, a Hypotensive Natural Product. Bioorg. Med. Chem. Lett. 1996, 6, 653–658. [Google Scholar] [CrossRef]
- Sindhu, R.K.; Vasudeva, N.; Sharma, S.K. Hypolipidemic Potential of Ethanolic Extract of Verbesina encelioides Benth. Roots. Acta Pol. Pharm. 2011, 68, 301–304. https://www.ptfarm.pl/wydawnictwa/czasopisma/acta-poloniae-pharmaceutica/110/-/13479.
- Banasthali University. Anti-Termite Herbal Composition And Process For Preparation Thereof. IN 2010/MUM/3395. 14 December 2017. https://patentimages.storage.googleapis.com/33/1b/a1/98dbb5da2bfd4d/WO2011027362A2.pdf.
- Carr, M.E.; Bagby, M.O. Tennessee Plant Species Screened for Renewable Energy Sources. Econ. Bot. 1987, 41, 78–85. [Google Scholar] [CrossRef]
| Category | Compound / Extract |
Efficacy (Target & Value) |
Toxicity (Cell Line & Value) |
Selectivity Index (SI) |
|---|---|---|---|---|
| I. Confirmed High Selectivity (SI >> 1) | ||||
| Anti-Protozoal Lead [47] | Arbusculin Derivative (V. lanata) |
L. major amastigotes, 4.9 - 25.3 μM |
NMM, 432.5 - 620.7 μM |
Up to 88.2 |
| Non-Cytotoxic Triterpenes [106] | Pseudotaraxasterol Derivatives (from V. encelioides) |
Protozoa (Antiprotozoal activity), 32.2 - 48.2 μg/mL |
HELF, (MRC-5) (CC50), > 64 μg/mL |
>1.3−2.0 |
| Host-Immune Booster [113] | V. oerstediana Acetone Bark Extract |
S. aureus, Significantly boosts killing at 10 μg/mL |
Neutrophils (Viability), Non-cytotoxic at 10 μg/mL |
High (Qualitative) |
| II. Efficacy & Duality Challenges | ||||
| Efficacy Benchmark (Germacrene) [38] |
Germacrene SL (V. negrensis) |
S. aureus / E. faecalis (MIC), 64 μg/mL |
CC50 on non-cancerous line: > 64 μg/mL | ≈1 (Implied) |
| Duality Case (Mild Uncoupler) [62] | CDE (from V. persicifolia) | Undocumented | RLM (Ceff), 20-100 μM (Mild Uncoupling) | ≈1 (Implied) |
| Cytotoxicity Benchmark (Extract) [73] |
V. turbacensis Hydroethanolic Extract |
(Low Activity assumed) | HepG2 cells (IC50), 334.6 μg/mL | High (Qualitative) |
| Toxicity Benchmark (Isolated Toxin) [87] |
Galegine (Alkaloid) (from V. encelioides) | (Toxicity focus) | Hep G2 cells (IC50), 10.9 μg/mL | Very Low SI (Implied) |
| Extract Duality (Allelopathy) [3] |
V. encelioides Root Leachate |
Inhibition (Full Strength), -25.7% (RRG) | Stimulation (Low Dilution), +36.5% (RRG) |
Concentration- Dependent |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




