Submitted:
13 January 2026
Posted:
15 January 2026
You are already at the latest version
Abstract
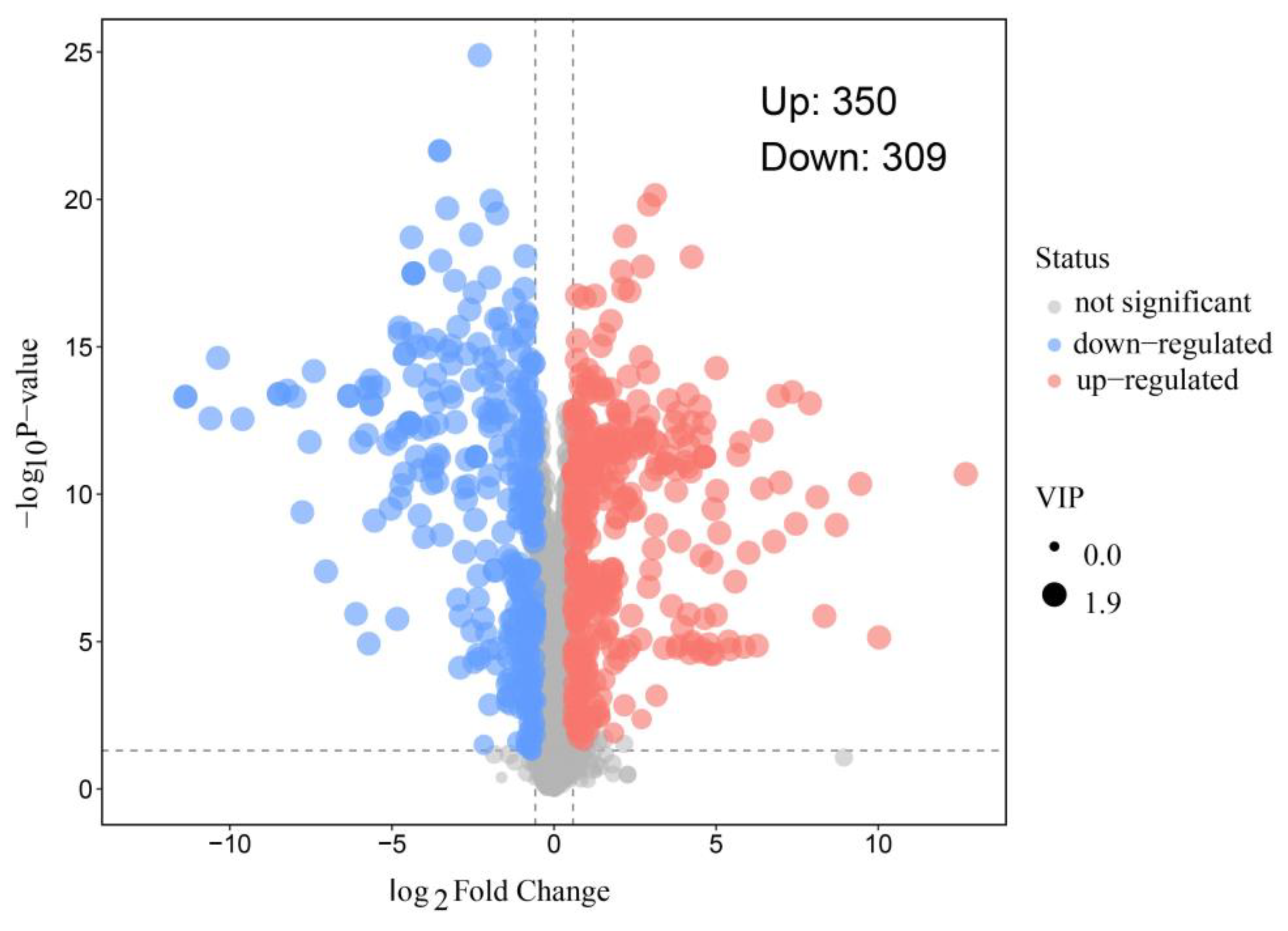
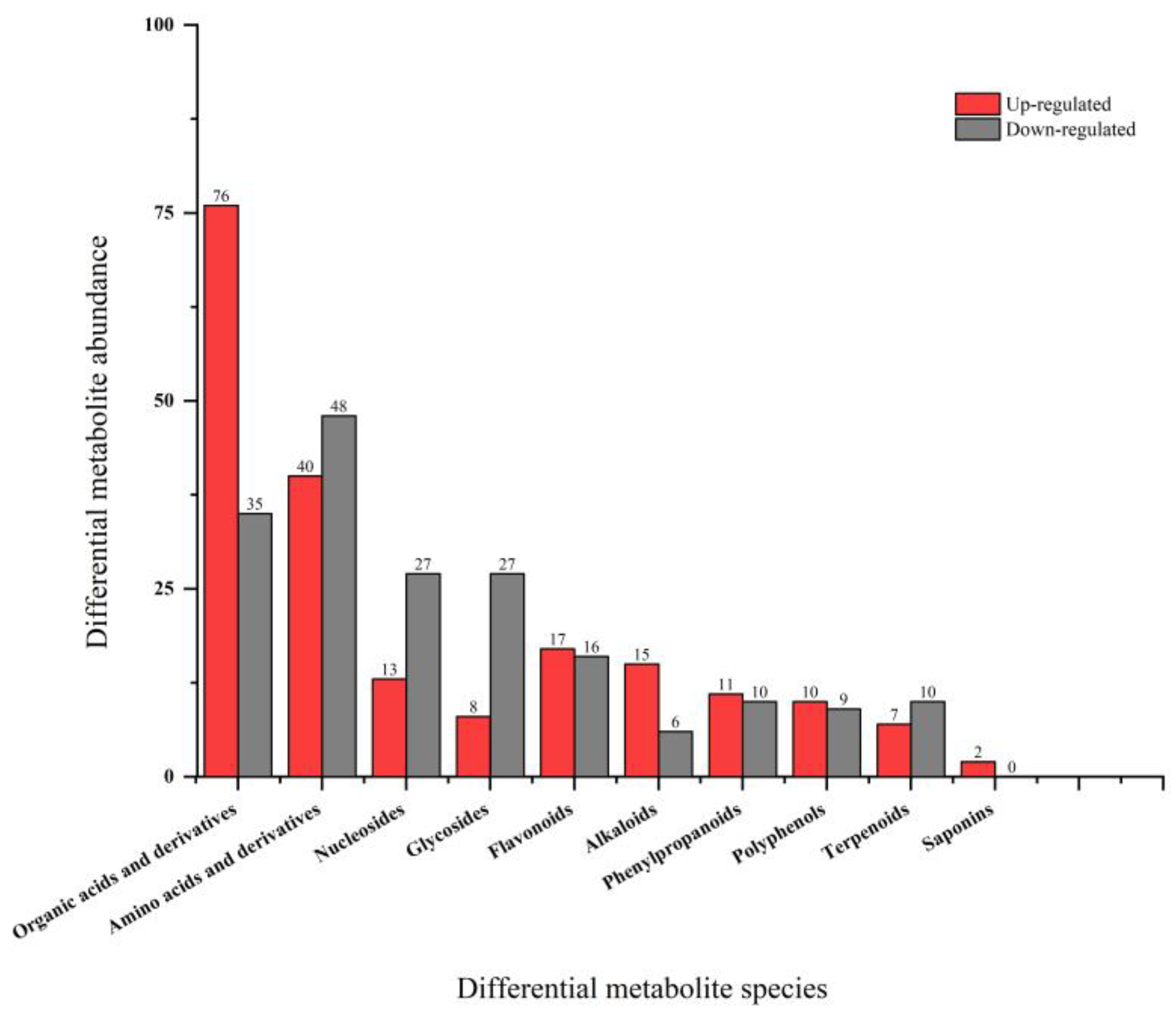
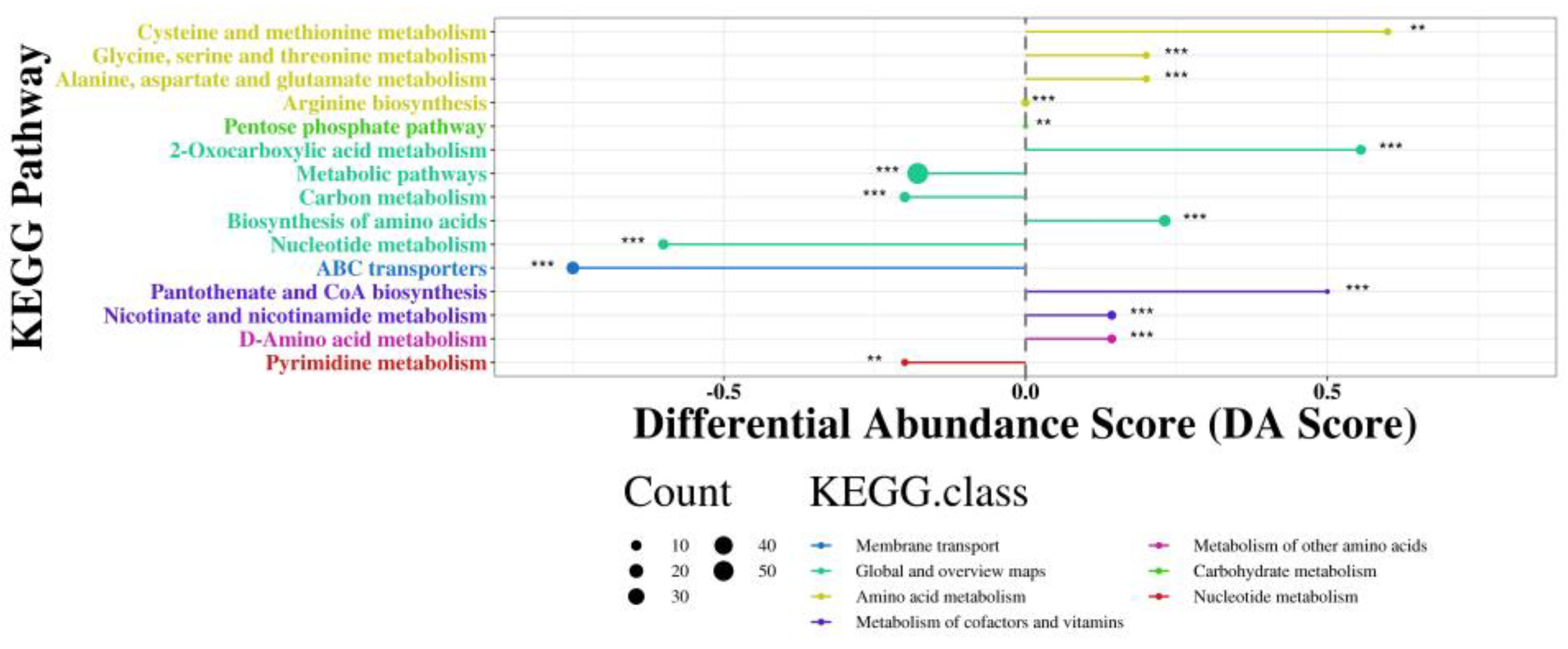
Astragalus membranaceus (AM) is a traditional medicinal and edible herb with well-documented immunomodulatory activities; however, its application in functional beverages is limited by the low bioavailability of its bioactive constituents. Probiotic fermentation has emerged as an effective strategy to enhance the nutritional and functional properties of herbal materials, yet the underlying metabolic mechanisms remain insufficiently understood. In this study, untargeted metabolomics based on ultra-high-performance liquid chromatography coupled with Orbitrap mass spectrometry (UHPLC-Orbitrap MS) was employed to comprehensively characterize metabolic alterations in AM aqueous extracts before and after fermentation with Pediococcus acidilactici (P. acidilactici) for 48 h. Multivariate statistical analyses combined with pathway enrichment analysis were used to identify differential metabolites and key metabolic pathways affected by fermentation. A total of 659 significantly altered metabolites were identified, including 350 upregulated and 309 downregulated metabolites after fermentation. These metabolites were mainly associated with organic acids, flavonoids, amino acid derivatives, nucleotides, and phenylpropanoids. Notably, fermentation markedly enhanced metabolites related to arginine biosynthesis, carbon metabolism, and nicotinate and nicotinamide metabolism, accompanied by a substantial accumulation of functional compounds such as lactate, phenyllactic acid, indolelactic acid, and nicotinamide adenine dinucleotide (NAD+). Overall, P. acidilactici fermentation induced extensive metabolic reprogramming of AM aqueous extracts, leading to the enrichment of multiple bioactive metabolites and the activation of key functional processes. These findings provide mechanistic insights into probiotic fermentation of medicinal and edible herbs and offer a scientific basis for the development of value-added fermented AM beverages with improved nutritional and functional properties.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals and Strain
2.2. Preparation and Fermentation of Astragalus membranaceus Extract
2.3. Metabolite Extraction
2.4. UHPLC-Orbitrap MS Analysis
2.5. Data Processing and Statistical Analysis
3. Results and Discussion
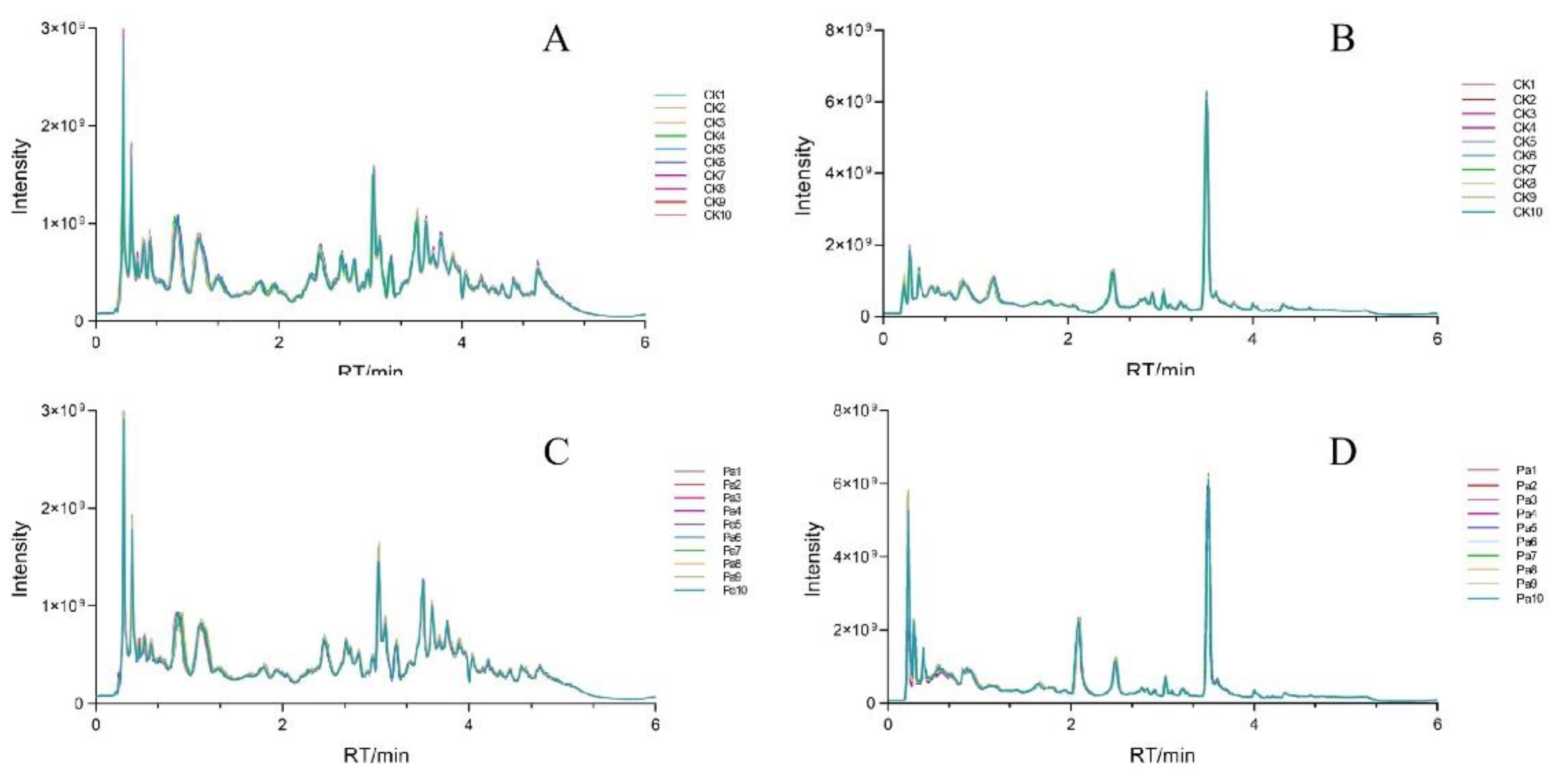
3.1. UHPLC–Orbitrap MS-Based Metabolic Profiling of Astragalus membranaceus Extracts
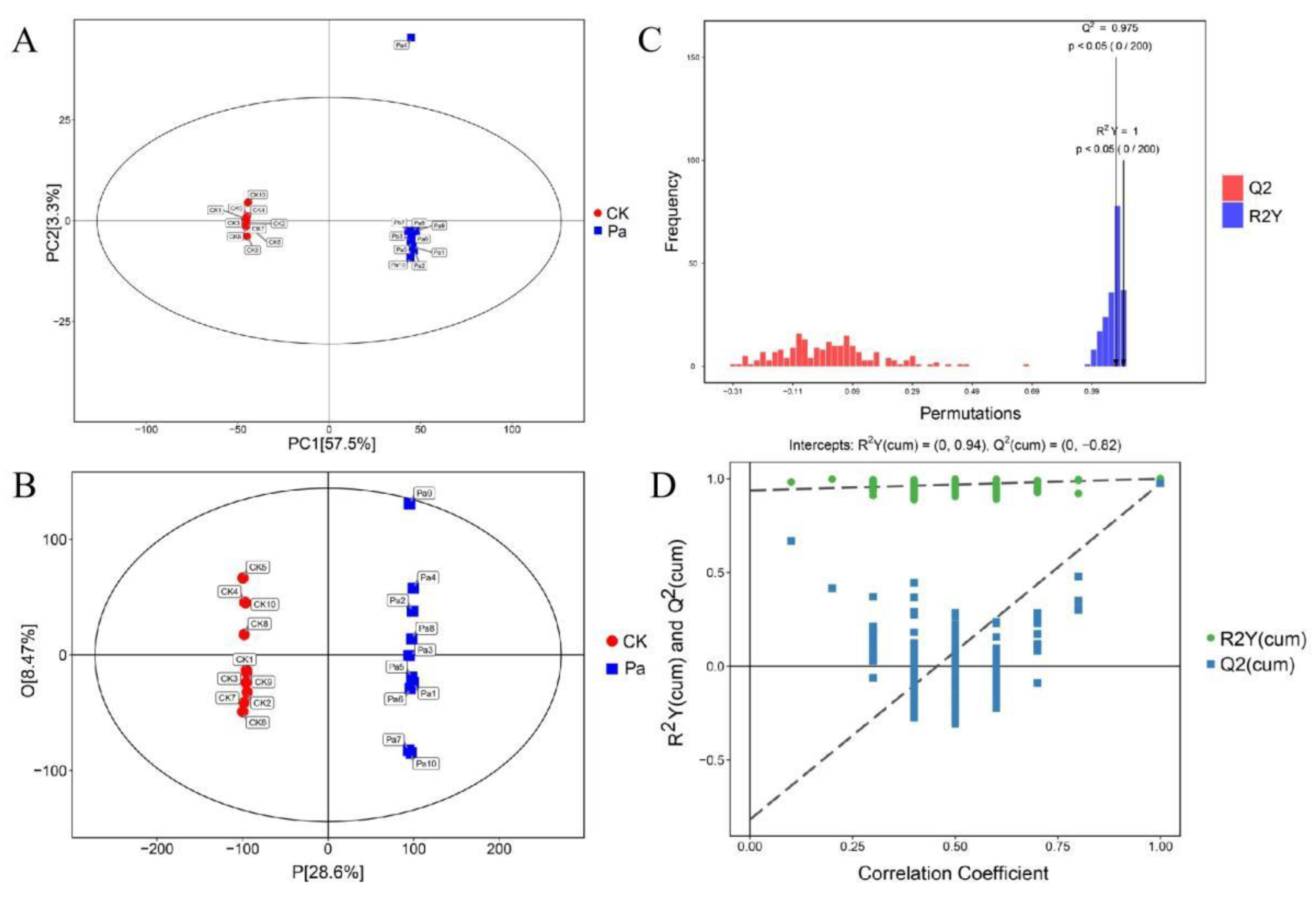
3.2. Multivariate Statistical Analysis
3.3. Analysis of Differential Metabolites
3.3.1. Organic Acids and Their Derivatives
3.3.2. Flavonoid Compounds
3.3.3. Alkaloids
3.3.4. Phenylpropanoids
3.3.5. Polyphenols
3.4. Metabolic Pathway Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, B.; Zhang, Q.; Liu, Z.; Su, Y.; Mu, Y.; Sun, S.; Chen, G. Root-associated microbiomes are influenced by multiple factors and regulate the growth and quality of Astragalus membranaceus (fisch) Bge. var. mongholicus (Bge.) Hsiao. Rhizosphere 2022, 24. [Google Scholar] [CrossRef]
- Durazzo, A.; Nazhand, A.; Lucarini, M.; Silva, A.M.; Souto, S.B.; Guerra, F.; Severino, P.; Zaccardelli, M.; Souto, E.B.; Santini, A. Astragalus (Astragalus membranaceus Bunge): botanical, geographical, and historical aspects to pharmaceutical components and beneficial role. Rendiconti Lincei. Scienze Fisiche e Naturali 2021, 32, 625–642. [Google Scholar] [CrossRef]
- Fu, J.; Wang, Z.; Huang, L.; Zheng, S.; Wang, D.; Chen, S.; Zhang, H.; Yang, S. Review of the Botanical Characteristics, Phytochemistry, and Pharmacology of Astragalus membranaceus (Huangqi). Phytotherapy Research 2014, 28, 1275–1283. [Google Scholar] [CrossRef]
- Dong, M.; Li, J.; Yang, D.; Li, M.; Wei, J. Biosynthesis and Pharmacological Activities of Flavonoids, Triterpene Saponins and Polysaccharides Derived from Astragalus membranaceus. Molecules 2023, 28. [Google Scholar] [CrossRef]
- Chen, T.; Xie, L.; Shen, M.; Yu, Q.; Chen, Y.; Xie, j. Recent advances in Astragalus polysaccharides: Structural characterization, bioactivities and gut microbiota modulation effects. Trends in Food Science & Technology 2024, 153. [Google Scholar]
- Chen, K.; Zhang, M.; Gao, B.; Hasan, A.; Li, J.; Bao, Y.o.; Fan, J.; Yu, R.; Yi, Y.; Ågren, H.; Wang, Z.; Liu, H.; Ye, M.; Qiao, X. Characterization and protein engineering of glycosyltransferases for the biosynthesis of diverse hepatoprotective cycloartane-type saponins in Astragalus membranaceus. Plant Biotechnology Journal 2023, 21, 698–710. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Liu, L.; Gao, C.; Chen, W.; Vong, C.T.; Yao, P.; Yang, Y.; Li, X.; Tang, X.; Wang, S.; Wang, Y. Astragali Radix (Huangqi): A promising edible immunomodulatory herbal medicine. Journal of Ethnopharmacology 2020, 258. [Google Scholar] [CrossRef] [PubMed]
- Sheng, X.; Yang, L.; Huang, B.; Lin, G.; Wang, Y.; Wu, X.; Lin, R. Efficacy of Astragalus Membranaceus (Huang Qi) for Cancer-Related Fatigue: A Systematic Review and Meta-Analysis of Randomized Controlled Studies. Integrative Cancer Therapies 2025, 24. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Z.; Chen, L.; Dong, Q.; Yang, D.-H.; Zhang, Q.; Zeng, J.; Wang, Y.; Liu, X.; Cui, Y.; Li, M.; Luo, X.; Zhou, C.; Ye, M.; Li, L.; He, Y. Astragali radix (Huangqi): a time-honored nourishing herbal medicine. Chinese Medicine 2024, 19. [Google Scholar] [CrossRef]
- Wang, C.; Huang, M.; Guo, B.; Zhou, X.; Cui, Z.; Xu, Y.; Ren, Y. Severe enterovirus A71 infection is associated with dysfunction of T cell immune response and alleviated by Astragaloside A. Virologica Sinica 2025, 40, 451–461. [Google Scholar] [CrossRef]
- Liu, C.H.; Tsai, C.H.; Li, T.C.; Yang, Y.W.; Huang, W.S.; Lu, M.K.; Tseng, C.H.; Huang, H.C.; Chen, K.F.; Hsu, T.S.; Hsu, Y.T.; Tsai, C.H.; Hsieh, C.L. Effects of the traditional Chinese herb Astragalus membranaceus in patients with poststroke fatigue: A double-blind, randomized, controlled preliminary study. Journal of Ethnopharmacology 2016, 194, 954–962. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.H.; Tsai, W.J.; Loke, S.H.; Wu, T.S.; Chiou, W.F. Astragalus membranaceus flavonoids (AMF) ameliorate chronic fatigue syndrome induced by food intake restriction plus forced swimming. Journal of Ethnopharmacology 2009, 122, 28–34. [Google Scholar] [CrossRef]
- Zhang, X.; Miao, Q.; Pan, C.; Yin, J.; Wang, L.; Qu, L.; Yin, Y.; Wei, Y. Research advances in probiotic fermentation of Chinese herbal medicines. iMeta 2023, 2. [Google Scholar] [CrossRef]
- Yang, H.Y.; Han, L.; Lin, Y.Q.; Li, T.; Wei, Y.; Zhao, L.H.; Tong, X.L. Probiotic Fermentation of Herbal Medicine: Progress, Challenges, and Opportunities. The American Journal of Chinese Medicine 2023, 51, 1105–1126. [Google Scholar] [CrossRef]
- An, Z.; Ye, T.; Yu, J.; Wu, H.; Niu, P.; Wei, X.; Liu, H.; Fang, H. A novel concoction method of Chinese medicinal and edible plants: probiotic fermentation, sensory and functional composition analysis. Sustainable Food Technology 2025, 3, 822–836. [Google Scholar] [CrossRef]
- Liu, W.; Luo, X.; Qiu, S.; Huang, W.; Su, Y.; Li, L. Determining the changes in metabolites of Dendrobium officinale juice fermented with starter cultures containing Saccharomycopsis fibuligera FBKL2.8DCJS1 and Lactobacillus paracasei FBKL1.3028 through untargeted metabolomics. BMC Microbiology 2023, 23. [Google Scholar] [CrossRef]
- Xiong, W.; Jiang, X.; He, J.; Liu, X.; Zhu, Y.; Liu, B.; Huang, Y. Probiotic Fermentation of Kelp Enzymatic Hydrolysate Promoted its Anti-Aging Activity in D-Galactose-Induced Aging Mice by Modulating Gut Microbiota. Molecular Nutrition & Food Research 2023, 67. [Google Scholar]
- Li, Y.L.; Liu, L.; Huang, X.C.; Huang, H.; Jiang, B.Y.; Guo, R.X.; Zhang, K.; Du, B.; Li, P. Probiotic fermentation of Polygonatum plant polysaccharides converting fructans to glucans with enhanced anti-obesity activity. International Journal of Biological Macromolecules 2025, 320. [Google Scholar] [CrossRef] [PubMed]
- Rao, H.; Lin, L.; Zhao, M. Probiotic fermentation affects the chemical characteristics of coix seed-chrysanthemum beverage: Regulatory role in sensory and nutritional qualities. Food Bioscience 2024, 58. [Google Scholar] [CrossRef]
- Cheng, H.; Du, Y.; Hu, J.; Cao, J.; Zhang, G.; Ling, J. New flavonoid and their anti-A549 cell activity from the bi-directional solid fermentation products of Astragalus membranaceus and Cordyceps kyushuensis. Fitoterapia 2024, 176. [Google Scholar] [CrossRef]
- Tangyu, M.; Muller, J.; Bolten, C.J.; Wittmann, C. Fermentation of plant-based milk alternatives for improved flavour and nutritional value. Applied Microbiology and Biotechnology 2019, 103(23-24), 9263–9275. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Song, Q.; Wang, M.; Ren, J.; Liu, S.; Zhao, S. Comparative genomics analysis of Pediococcus acidilactici species. Journal of Microbiology 2021, 59, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Ahn, H.; Lee, H.J. Investigation of Bacteriocin Production Ability of Pediococcus acidilactici JM01 and Probiotic Properties Isolated From Tarak, a Conventional Korean Fermented Milk. Food Science & Nutrition 2025, 13. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, L.; Zhang, B.; Bao, J. pH shifting adaptive evolution stimulates the low pH tolerance of Pediococcus acidilactici and high L-lactic acid fermentation efficiency. Bioresource Technology 2025, 416. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.; Jantama, K.; Prasitpuriprecha, C.; Wansutha, S.; Phosriran, C.; Yuenyaow, L.; Cheng, K.-C.; Jantama, S.S. Harnessing Fermented Soymilk Production by a Newly Isolated Pediococcus acidilactici F3 to Enhance Antioxidant Level with High Antimicrobial Activity against Food-Borne Pathogens during Co-Culture. Foods 2024, 13. [Google Scholar] [CrossRef]
- Zhao, M.; Liu, K.; Zhang, Y.; Li, Y.; Zhou, N.; Li, G. Probiotic characteristics and whole-genome sequence analysis of Pediococcus acidilactici isolated from the feces of adult beagles. Frontiers in Microbiology 2023, 14. [Google Scholar]
- Sharma, A.; Noda, M.; Sugiyama, M.; Kumar, B.; Kaur, B. Application ofPediococcus acidilacticiBD16 (alaD+) expressing L-alanine dehydrogenase enzyme as a starter culture candidate for secondary wine fermentation. Biotechnology & Biotechnological Equipment 2021, 35, 1643–1661. [Google Scholar]
- Chaichana, N.; Boonsan, J.; Dechathai, T.; Suwannasin, S.; Singkhamanan, K.; Wonglapsuwan, M.; Pomwised, R.; Surachat, K. Comparative genomic analysis of the Pediococcus genus reveals functional diversity for fermentation and probiotic applications. Computational and Structural Biotechnology Journal 2025, 27, 4597–4614. [Google Scholar] [CrossRef]
- Myo, N.Z.; Kamwa, R.; Khurajog, B.; Pupa, P.; Sirichokchatchawan, W.; Hampson, D.J.; Prapasarakul, N. Industrial production and functional profiling of probiotic Pediococcus acidilactici 72 N for potential use as a swine feed additive. Scientific Reports 2025, 15. [Google Scholar] [CrossRef]
- Yan, Y.; Zou, M.; Tang, C.; Ao, H.; He, L.; Qiu, S.; Li, C. The insights into sour flavor and organic acids in alcoholic beverages. Food Chemistry 2024, 460. [Google Scholar] [CrossRef]
- Yang, J.; Yu, J.; Chen, Y.; Xu, A.; Yang, C.; Li, J.; Wu, F.; Li, X.; Bi, J.; Xiang, B.; Jiang, K. Hyperoside, a dietary flavonoid, protects against endometritis via gut microbiota-dependent production of hydroxyphenyllactic acid and the gut–uterus axis. Food & Function 2026. [Google Scholar]
- Parappilly, S.J.; Radhakrishnan, E.K.; George, S.M. Antibacterial and antibiofilm activity of human gut lactic acid bacteria. Brazilian Journal of Microbiology 2024, 55, 3529–3539. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, Y.; Guo, M.; Kong, Y.; Fan, X.; Sun, S.; Du, C.; Gong, H. Antifungal mechanisms of phenyllactic acid against Mucor racemosus: Insights from spore growth suppression, and proteomic analysis. Food Chemistry 2025, 475. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Bi, M.; Li, S.; Yang, S.; Zhang, Y.; Zhao, L.; Jiang, X.; Yu, T. Inhibition of Listeria monocytogenes biofilm formation by phenyllactic acid in pasteurized milk is associated with suppression of the Agr system. International Journal of Food Microbiology 2026, 444. [Google Scholar] [CrossRef]
- Liu, T.; Wei, X.; Chen, Y.; Liu, L.; Wu, X. Proteomic investigation of the antibacterial and anti-biofilm mechanisms of phenyllactic acid against Shigella flexneri and its application in tofu preservation. Food Bioscience 2025, 72. [Google Scholar] [CrossRef]
- Qian, X.; Li, Q.; Zhu, H.; Chen, Y.; Lin, G.; Zhang, H.; Chen, W.; Wang, G.; Tian, P. Bifidobacteria with indole-3-lactic acid-producing capacity exhibit psychobiotic potential via reducing neuroinflammation. Cell Reports Medicine 2024, 5. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.T.; Nguyen, H.T. Environmental stress for improving the functionality of lactic acid bacteria in malolactic fermentation. The Microbe 2024, 4. [Google Scholar] [CrossRef]
- Li, X.; Qi, T.; Zhou, L.; Lin, P.; Chen, Q.; Li, X.; He, R.; Yang, S.; Liu, Y.; Qi, F. Isoliquiritigenin alleviates abnormal sarcomere contraction and inflammation in myofascial trigger points via the IL-17RA/Act1/p38 pathway in rats. Phytomedicine 2025, 145. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.; Yang, Z.; Gao, Q.; Deng, Q.; Li, L.; Chen, H. Protective Effects of Isoliquiritigenin and Licochalcone B on the Immunotoxicity of BDE-47: Antioxidant Effects Based on the Activation of the Nrf2 Pathway and Inhibition of the NF-κB Pathway. Antioxidants 2024, 13. [Google Scholar] [CrossRef]
- Lim, Y.J.; Lim, B.; Kim, H.Y.; Kwon, S.-J.; Eom, S.H. Deglycosylation patterns of isoflavones in soybean extracts inoculated with two enzymatically different strains of lactobacillus species. Enzyme and Microbial Technology 2020, 132. [Google Scholar] [CrossRef]
- Al-Zharani, M.; Mubarak, M.; Almuqri, E.; Rudayni, H.; Aljarba, N.; Yaseen, K.; Albatli, S.; Alkahtani, S.; Nasr, F.; Al-Doaiss, A.; Al-eissa, M. Catechin (epigallocatechin-3-gallate) supplement restores the oxidation: antioxidation balance through enhancing the total antioxidant capacity in Wistar rats with cadmium-induced oxidative stress. Journal of Nutritional Science 2025, 14. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, M.; Guzman, L.; Olloquequi, J.; Cano, A.; Fortuna, A.; Vazquez-Carrera, M.; Verdaguer, E.; Auladell, C.; Ettcheto, M.; Camins, A. Licochalcone A prevents cognitive decline in a lipopolysaccharide-induced neuroinflammation mice model. Molecular Medicine 2025, 31. [Google Scholar] [CrossRef]
- Xu, L.; Zhou, S.; Li, J.; Yu, W.; Gao, W.; Luo, H.; Fang, X. The Anti-Inflammatory Effects of Formononetin, an Active Constituent of Pueraria montana Var. Lobata, via Modulation of Macrophage Autophagy and Polarization. Molecules 2025, 30. [Google Scholar] [CrossRef]
- Guo, J.; Zhang, P.; Yang, X.; Wang, X.; Cao, X.; Zhao, J. Homoplantaginin exerts therapeutic effects on intervertebral disc degeneration by alleviating TNF-α-induced nucleus pulposus cell senescence and inflammation. Frontiers in Pharmacology 2025, 16. [Google Scholar] [CrossRef] [PubMed]
- Alghamdi, H.A.; Alghamdi, S.S.; Al-Zahrani, M.H.; Trivilegio, T.; Bahattab, S.; AlRoshody, R.; Alhaidan, Y.; Alghamdi, R.A.; Matou-Nasri, S. Reticuline and Coclaurine Exhibit Vitamin D Receptor-Dependent Anticancer and Pro-Apoptotic Activities in the Colorectal Cancer Cell Line HCT116. Current Issues in Molecular Biology 2025, 47. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.Y.; Lin, T.H.; Chen, C.Y.; He, Y.H.; Huang, W.C.; Hsieh, C.Y.; Chen, Y.H.; Chang, W.C. Stephania tetrandra and Its Active Compound Coclaurine Sensitize NSCLC Cells to Cisplatin through EFHD2 Inhibition. Pharmaceuticals 2024, 17. [Google Scholar] [CrossRef]
- Lin, Z.; Li, S.; Guo, P.; Wang, L.; Zheng, L.; Yan, Z.; Chen, X.; Cheng, Z.; Yan, H.; Zheng, C.; Zhao, C. Columbamine suppresses hepatocellular carcinoma cells through down-regulation of PI3K/AKT, p38 and ERK1/2 MAPK signaling pathways. Life Sciences 2019, 218, 197–204. [Google Scholar] [CrossRef]
- Gao, D.; Liu, T.; Gao, J.; Xu, J.; Gou, Y.; Pan, Y.; Li, D.; Ye, C.; Pan, R.; Huang, L.; Xu, Z.; Lian, J. De Novo Biosynthesis of Vindoline and Catharanthine in Saccharomyces cerevisiae. BioDesign Research 2022, 2022. [Google Scholar] [CrossRef]
- Ko, W.; Kim, K.W.; Liu, Z.; Dong, L.; Yoon, C.S.; Lee, H.; Kim, Y.C.; Oh, H.; Lee, D.S.; Kim, S.C. Macluraxanthone B inhibits LPS-induced inflammatory responses in RAW264.7 and BV2 cells by regulating the NF-κB and MAPK signaling pathways. Immunopharmacology and Immunotoxicology 2021, 44, 67–75. [Google Scholar] [CrossRef]
- Zhu, L.X.; Zhang, H.Z.; Zhang, C.; Zhou, C.H. Comprehensive insights into emodin compounds research in medicinal chemistry. Bioorganic & Medicinal Chemistry 2025, 128. [Google Scholar]
- Antonio, J.M.; Liu, Y.; Suntornsaratoon, P.; Jones, A.; Ambat, J.; Bala, A.; Kanattu, J.J.; Flores, J.; Bandyopadhyay, S.; Upadhyay, R.; Bhupana, J.N.; Su, X.; Li, W.V.; Gao, N.; Ferraris, R.P. Lacticaseibacillus rhamnosus GG-driven remodeling of arginine metabolism mitigates gut barrier dysfunction. American Journal of Physiology-Gastrointestinal and Liver Physiology 2025, 329, G162–G185. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



