Submitted:
06 January 2026
Posted:
07 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
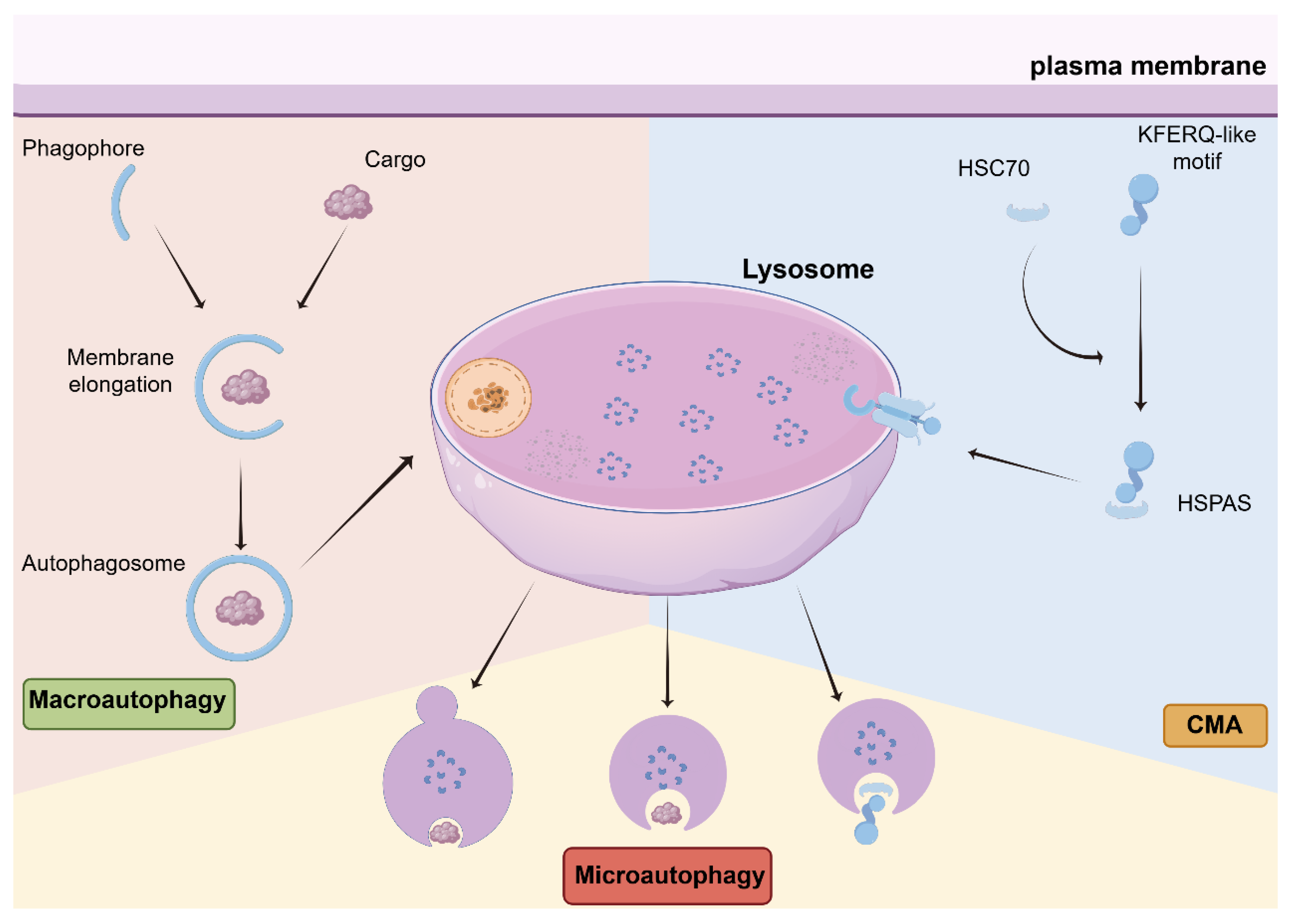
2. Autophagy as a Stage- and Context-Dependent Process in Cancer
3. Context-Dependent Roles of Key Autophagy Regulators in Cancer
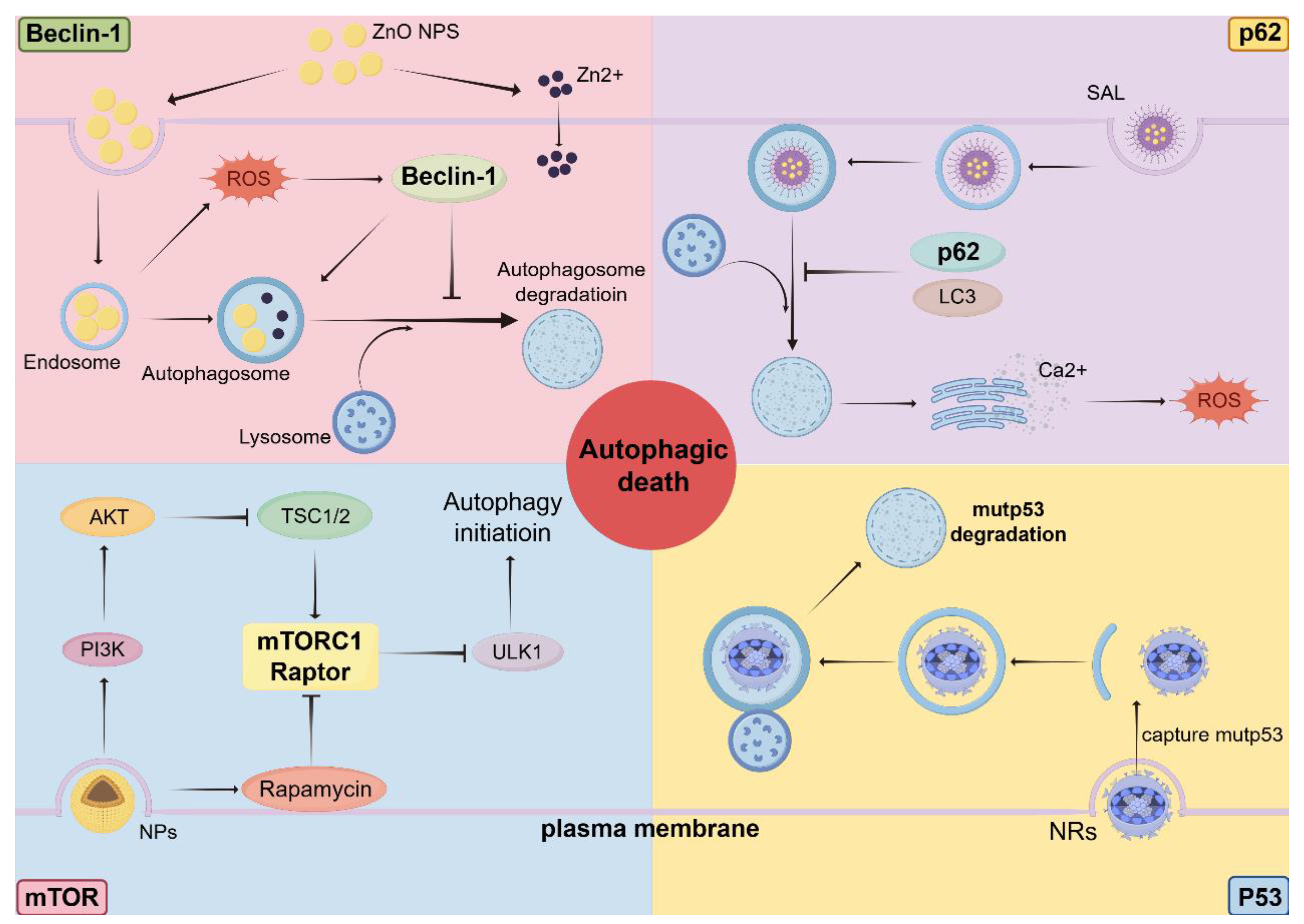
3.1. Beclin-1: Tumor Suppressor, Stress Adaptor, or Therapeutic Switch
3.2. p62/SQSTM1: Autophagy Flux Sensor and Oncogenic Signaling Hub
3.3. mTOR: Metabolic Gatekeeper Linking Autophagy and Therapeutic Vulnerability
3.4. p53: Genotype-Dependent Regulator of Autophagy and Therapy Response
3.5. Implications for Precision Autophagy Targeting
4. Tumor Microenvironment as a Determinant of Autophagy Dependency
4.1. Hypoxia and Metabolic Stress as Drivers of Autophagy Rewiring
4.2. Autophagy-Mediated Immune Evasion and Immunotherapy Response
4.3. Autophagy in Cancer-Associated Fibroblasts and Stromal Support
4.4. Therapeutic Stress, Adaptive Autophagy, and Resistance Mechanisms
4.5. Targeting TME-Driven Autophagy: Implications for Precision Therapy
5. Precision Nanomedicine for Spatiotemporal Modulation of Autophagy
5.1. Rationale for Nanotechnology-Based Autophagy Modulation
5.2. Targeted Nanocarriers for Modulating Beclin-1–Dependent Autophagy
5.3. Nanomedicine-Enabled Control of mTOR Signaling and Autophagy Flux
5.4. Nanoparticle-Based Strategies Targeting p53–Autophagy Interplay
5.5. Responsive and Multifunctional Nanocarriers for TME-Adapted Autophagy Modulation
5.6. Challenges and Perspectives for Clinical Translation
6. Integrating Autophagy Targeting with Tumor Microenvironment Modulation and Combination Therapy
6.1. Autophagy Modulation to Overcome Therapy Resistance
6.2. Enhancing Radiotherapy Efficacy Through Stromal and Microenvironmental Autophagy Targeting
6.3. Autophagy and Immunotherapy: Balancing Immune Activation and Immune Evasion
6.4. Nanomedicine-Enabled Combination Strategies Targeting Autophagy and the TME
6.5. Clinical Considerations and Future Integration Strategies
7. Challenges and Future Perspectives
7.1. Defining and Measuring Autophagy Dependency in Patients
7.2. Managing Spatial and Temporal Heterogeneity of Autophagy
7.3. Balancing Anti-Tumor Efficacy with Immune Preservation
7.4. Translational and Clinical Trial Challenges
7.5. Future Directions: Toward Context-Aware Autophagy Therapy
8. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, Z.; Klionsky, D.J. Eaten alive: a history of macroautophagy. Nat. Cell. Biol. 2019, 12, 814-822.
- Nakatogawa, H. Mechanisms governing autophagosome biogenesis. Nat. Rev. Mol. Cell. Biol. 2020, 21, 439-458.
- Russell, R.C.; Guan, K.L. The multifaceted role of autophagy in cancer. EMBO. J. 2022, 41, e110031. [CrossRef]
- White, E. The role for autophagy in cancer. J. Clin. Invest. 2015, 12, 42-46.
- Debnath, J.; Gammoh, N.; Ryan, K.M. Autophagy and autophagy-related pathways in cancer. Nat. Rev. Mol. Cell. Biol. 2023, 24, 560-575.
- Shi, Z.; Hu, C.L.; Zheng, X.G.; Sun, C.; Li, Q. Feedback loop between hypoxia and energy metabolic reprogramming aggravates the radioresistance of cancer cells. Exp. Hematol. Oncol. 2024, 13, 55. [CrossRef]
- Levy, J.M.; Thorburn, A. Targeting autophagy during cancer therapy to improve clinical outcomes. Pharmacol. Ther. 2011, 131, 130-141. [CrossRef]
- Pérez-Hernández, M.; Arias, A.; Martínez-García, D.; Pérez-Tomás, R.; Quesada, R.; Soto-Cerrato, V. Targeting Autophagy for Cancer Treatment and Tumor Chemosensitization. Cancers. 2019, 11, 1599.
- Zou, Z.; Yuan, Z.; Zhang, Q.; Long. Z.; Chen, J.; Tang, Z.; Zhu, Y.L.; Chen, S.P.; Xu, J.; Yan, M.; et al. Aurora kinase A inhibition-induced autophagy triggers drug resistance in breast cancer cells. Autophagy. 2012, 8, 1798-1810. [CrossRef]
- Kenific, C.M.; Debnath, J. Cellular and metabolic functions for autophagy in cancer cells. Trends. Cell. Biol. 2015, 25, 37-45.
- Schlie, K.; Spowart, J.E.; Hughson, L.R.; Townsend, K.N.; Lum, J.J. When Cells Suffocate: Autophagy in Cancer and Immune Cells under Low Oxygen. Int. J. Cell. Biol. 2011, 470597. [CrossRef]
- Xia, H.; Green, D.R.; Zou, W. Autophagy in tumour immunity and therapy. Nat. Rev. Cancer. 2021, 21, 281-297. [CrossRef]
- Gao, Z.; Chen, J.F.; Fu, X.T.; Ding, Z.B. Advances in autophagy regulation of macrophages involved in the construction of tumor microenvironment. Chin. J. Clin. Med. 2021, 28, 894-899.
- Yamamoto, K.; Venida, A.; Yano, J.; Biancur, D.E.; Kakiuchi, M.; Gupta, S.; Sohn A.S.W.; Mukhopadhyay, S.; Lin E.Y.; Parker S.J.; et al. Autophagy promotes immune evasion of pancreatic cancer by degrading MHC-I. Nature. 2020, 581, 100-105.
- Mirnezami, R.; Nicholson, J.; Darzi, A. Preparing for precision medicine. N. Engl. J. Med. 2012, 366, 489-491.
- Mosele, M.F.; Westphalen, C.B.; Stenzinger, A.; Barlesi, F.; Bayle, A.; Bièche, I.; Bonastre, J.; Castro, E.; Dienstmann, R.; Krämer, A.; et al. Recommendations for the use of next-generation sequencing (NGS) for patients with advanced cancer in 2024: a report from the ESMO Precision Medicine Working Group. Ann. Oncol. 2024, 35, 588-606. [CrossRef]
- Wang, Y.; Sun, S.; Zhang, Z.; Shi, D. Nanomaterials for Cancer Precision Medicine. Adv. Mater. 2018, 30, e1705660.
- Zhao, G.Y.; Yousefi, F.; Tsukamoto, I.; Moran, S.; Behfar, A.; Evans, C.; Zhao, C.F. A therapeutic-grade purified exosome system alleviates osteoarthritis by regulating autophagy through the BCL2-Beclin1 axis. J. Nanobiotechnology. 2025, Online ahead of print. [CrossRef]
- Rahman, M.A.; Jalouli, M.; Bhajan, S. K.; Al-Zharani, M.; Harrath, A.H. A Comprehensive Review of Nanoparticle-Based Drug Delivery for Modulating PI3K/AKT/mTOR-Mediated Autophagy in Cancer. Int. J. Mol. Sci. 2025, 26, 1868. [CrossRef]
- Kong, N.; Tao, W.; Ling, X.; Wang, J.; Xiao, Y.; Shi, S.; Ji, X.Y.; Shajii, A.; Gan, S.T.; Kim, N.T.; et al. Synthetic mRNA nanoparticle-mediated restoration of p53 tumor suppressor sensitizes p53-deficient cancers to mTOR inhibition. Sci. Transl. Med. 2019, 11, eaaw1565.
- Lu, Y.Y.; Fang, Y.Y.; Wang, S.S.; Guo, J., Song, J.L.; Zhu, L.; Lin, Z.K.; Wang, R.; Zhang, S.Y.; Qiu, W.S.; et al. Cepharanthine sensitizes gastric cancer cells to chemotherapy by targeting TRIB3-FOXO3-FOXM1 axis to inhibit autophagy. Phytomedicine. 2024, 135, 156161. [CrossRef]
- Koschade, S.E.; Klann, K., Shaid, S.; Vick, B.; Stratmann, J.A.; Thölken, M.; Meyer L.M.; Nguyen, T.D.; Campe, J.; Moser, L.M.; et al. Translatome proteomics identifies autophagy as a resistance mechanism to on-target FLT3 inhibitors in acute myeloid leukemia. Leukemia. 2022, 36, 2396-2407.
- Jin, S. p53, Autophagy and tumor suppression. Autophagy. 2005, 1, 171-173.
- Wang, Y.; Gan, G.; Wang, B.; Wu, J.; Cao, Y.; Zhu, D.; Xu, Y.; Wang, X.N.; Han, H.X.; Li, X.L.; et al. Cancer-associated Fibroblasts Promote Irradiated Cancer Cell Recovery Through Autophagy. EBioMedicine. 2017, 17, 45-56.
- Chen, Y.; Zhang, X.; Yang, H.; Liang, T.; Bai, X. The"Self-eating" of cancer-associated fibroblast: A potential target for cancer. Biomed. Pharmacother. 2023, 163, 114762.
- Aita, V.M.; Liang, X.H.; Murty, V.V.; Pincus, D.L.; Yu. W.; Cayanis, E.; Kalachikov, S.; Gilliam, T.C.; Levine, B. Cloning and genomic organization of beclin 1, a candidate tumor suppressor gene on chromosome 17q21. Genomics. 1999, 59, 59-65. [CrossRef]
- Sun, P.H.; Zhu, L.M.; Qiao, M.M.; Zhang, Y.P.; Jiang, S.H.; Wu, Y.L.; Tu, S.P. The XAF1 tumor suppressor induces autophagic cell death via upregulation of Beclin-1 and inhibition of Akt pathway. Cancer Lett. 2011, 310, 170-180.
- Kaur, S.; Changotra, H. The beclin 1 interactome: Modification and roles in the pathology of autophagy-related disorders. Biochimie. 2020, 175, 34-49.
- Wang, S.; Ma, D.; Yang, M.; Zhang, Y; Wang, S.; Zhou, W. Arsenic trioxide-based nanoparticles for enhanced chemotherapy by activating pyroptosis. Acta. Pharm. Sin. B. 2025, 15, 6001-6018. [CrossRef]
- Li, Z.H.; Yin, X.W.; Lyu, C.Y.; Wang, J.Y.; Liu, K.; Cui, S.Y.; Ding, S.M.; Wang, Y.Y.; Wang, J.X.; Guo, D.L.; et al. Zinc Oxide Nanoparticles Trigger Autophagy in the Human Multiple Myeloma Cell Line RPMI8226: an In Vitro Study. Biol. Trace. Elem. Res. 2024, 203, 913-926. [CrossRef]
- Wen, J.; Chen, H.; Ren, Z.; Zhang, P.; Chen, J.; Jiang, S. Ultrasmall iron oxide nanoparticles induced ferroptosis via Beclin1/ATG5-dependent autophagy pathway. Nano Converg. 2021, 8, 10. [CrossRef]
- Wang, Y, Lin, Y.X.; Qiao, Z.Y.; An, H.W.; Qiao, S.L.; Wang, L.; Rajapaksha R.P.Y.; Wang, H. Self-assembled autophagy-inducing polymeric nanoparticles for breast cancer interference in-vivo. Adv. Mater. 2015, 27, 2627-2634.
- Puissant, A.; Fenouille, N.; Auberger, P. When autophagy meets cancer through p62/SQSTM1. Am. J. Cancer Res. 2012, 2, 397-413.
- Wei, H.; Wang, C.; Croce, C.M.; Guan J.L. p62/SQSTM1 synergizes with autophagy for tumor growth in vivo. Genes. Dev. 2014, 28, 1204-1216.
- Islam, M.A.; Sooro, M.A.; Zhang, P. Autophagic Regulation of p62 is Critical for Cancer Therapy. Int. J. Mol. Sci. 2018, 19, 1405. [CrossRef]
- Kim, Y.C.; Guan, K.L. mTOR: a pharmacologic target for autophagy regulation. J. Clin. Invest. 2015, 125, 25-32.
- Glaviano, A.; Foo, A.S.C.; Lam, H.Y.; Yap, K.C.H.; Jacot, W.; Jones, R.H.; Eng, H.; Nair, M.G.; Makvandi, P.; Geoerger, B.; et al. PI3K/AKT/mTOR signaling transduction pathway and targeted therapies in cancer. Mol. Cancer. 2023, 22, 138. [CrossRef]
- Ramirez, J.A.Z.; Romagnoli, G.G.; Kaneno, R. Inhibiting autophagy to prevent drug resistance and improve anti-tumor therapy. Life Sci. 2021, 265,118745. [CrossRef]
- Woo, H.N.; Chung, H.K.; Ju, E.J.; Jung. J.; Kang, H.W.; Lee, S.W.; Seo, M.H.; Lee, J.S.; Lee, J.S.; Park, H.J.; et al. Preclinical evaluation of injectable sirolimus formulated with polymeric nanoparticle for cancer therapy. Int. J. Nanomedicine. 2012, 7, 2197-208.
- Jia. L.; Hao. S.L.; Yang, W.X. Nanoparticles induce autophagy via mTOR pathway inhibition and reactive oxygen species generation. Nanomedicine (London). 2020, 15, 1419-1435.
- Xu. J.; Patel, N.H.; Gewirtz, D.A. Triangular Relationship between p53, Autophagy, and Chemotherapy Resistance. Int. J. Mol. Sci. 2020, 21, 8991.
- Liu, Y.; Su, Z.; Tavana, O.; Gu, W.; Understanding the complexity of p53 in a new era of tumor suppression. Cancer Cell. 2024, 42, 946-967.
- Huang, X.; Cao. Z.; Qian, J.; Ding, T.; Wu, Y.; Zhang, H.; Zhong, S.Q.; Wang, X.L.; Ren, X.G.; Zhang, W.; et al. Nanoreceptors promote mutant p53 protein degradation by mimicking selective autophagy receptors. Nat. Nanotechnol. 2024, 19, 545-553. [CrossRef]
- Camp, E.R.; Wang, C.; Little, E.C.; Watson, P.M.; Pirollo, K.F.; Rait. A.; Cole, D.J.; Chang, E.H.; Watson, D.K. Transferrin receptor targeting nanomedicine delivering wild-type p53 gene sensitizes pancreatic cancer to gemcitabine therapy. Cancer Gene Ther. 2013, 20, 222-228.
- Bilotta, M.T.; Antignani, A.; Fitzgerald, D.J. Managing the TME to improve the efficacy of cancer therapy. Front. Immunol. 2022, 13, 954992.
- Chen, F.; Zhuang, X.; Lin, L.; Yu, P.; Wang, Y.; Shi, Y.; Hu, G.; Sun, Y. New horizons in tumor microenvironment biology: challenges and opportunities. BMC Med. 2015, 13, 45.
- Roma-Rodrigues, C.; Mendes, R.; Baptista, P.V.; Fernandes, A.R. Targeting Tumor Microenvironment for Cancer Therapy. Int. J. Mol. Sci. 2019, 20.
- Ashraf, R.; Kumar, S. Mfn2-mediated mitochondrial fusion promotes autophagy and suppresses ovarian cancer progression by reducing ROS through AMPK/mTOR/ERK signaling. Cell. Mol. Life Sci. 2022, 79, 573. [CrossRef]
- Xanthopoulou, E.; Lamprou, I.; Mitrakas, A.G.; Michos, G.D.; Zois, C.E.; Giatromanolaki, A.; Harris, A.; Koukourakis M. Autophagy Blockage Up-Regulates HLA-Class-I Molecule Expression in Lung Cancer and Enhances Anti-PD-L1 Immunotherapy Efficacy. Cancers. 2024, 16, 3272.
- Sari, D.; Gozuacik, D.; Akkoc, Y. Role of autophagy in cancer-associated fibroblast activation, signaling and metabolic reprograming. Front. Cell Dev. Biol. 2024, 11, 1274682.
- Liang, L.; Fu, J.; Wang, S.; Cen, H.; Zhang, L.; Mandukhail, S.R.; Du, L.R.; Wu, Q.N.; Zhang, P.Q.; Yu, X.Y. MiR-142-3p enhances chemosensitivity of breast cancer cells and inhibits autophagy by targeting HMGB1. Acta Pharm. Sin. B. 2020, 10, 1036-1046.
- Carew, J.S.; Nawrocki, S.T.; Kahue, C.N.; Zhang, H.; Yang. C.; Chung, L.; Houghton, J.A.; Giles, P.H.F.J.; Cleveland, J.L. Targeting autophagy augments the anticancer activity of the histone deacetylase inhibitor SAHA to overcome Bcr-Abl-mediated drug resistance. Blood. 2007, 110, 313-322. [CrossRef]
- König, I.R.; Fuchs, O.; Hansen, G.; Mutius, E.; Kopp, M.V. What is precision medicine? Eur. Respir. J. 2017, 50, 1700391.
- Awad, N. S.; Paul, V.; AlSawaftah, N.M.; Haar G.T.; Allen, T.M.; Pitt, W.G.; Husseini, G.A. Ultrasound-Responsive Nanocarriers in Cancer Treatment: A Review. ACS Pharmacol. Transl. Sci. 2021, 4, 589-612.
- Fulton, M. D.; Najahi-Missaoui, W. Liposomes in Cancer Therapy: How Did We Start and Where Are We Now. Int. J. Mol. Sci. 2023, 24, 6615.
- Son, K.H.; Hong, J.H.; Lee, J.W. Carbon nanotubes as cancer therapeutic carriers and mediators. Int. J. Nanomedicine. 2016, 11, 5163-5185.
- Hari, S.K.; Gauba, A.; Shrivastava, N.; Tripathi, R.M.; Jain, S.K.; Pandey, A.K. Polymeric micelles and cancer therapy: an ingenious multimodal tumor-targeted drug delivery system. Drug Deliv. Transl. Res. 2023, 13, 135-163. [CrossRef]
- Zhao, M.X.; Zhu, B.J. The Research and Applications of Quantum Dots as Nano-Carriers for Targeted Drug Delivery and Cancer Therapy. Nanoscale Res. Lett. 2016, 11, 207.
- Liu, L.; Tu, B.; Sun, Y.; Liao, L.L.; Lu, X.L.; Liu, E.G.; Huang, Y.Z. Nanobody-based drug delivery systems for cancer therapy. J. Control Release. 2025, 381, 113562.
- Steichen, S.D.; Caldorera-Moore, M.; Peppas, N.A. A review of current nanoparticle and targeting moieties for the delivery of cancer therapeutics. Eur. J. Pharm. Sci. 2013, 48, 416-427.
- Xiang, J.; Zhao, R.; Wang, B.; Sun, X.; Guo, X.; Tan, S.; Liu, W.J. Advanced Nano-Carriers for Anti-Tumor Drug Loading. Front. Oncol. 2021, 11, 758143. [CrossRef]
- Dai, X.; Tan, C. Combination of microRNA therapeutics with small-molecule anticancer drugs: mechanism of action and co-delivery nanocarriers. Adv. Drug Deliv. Rev. 2015, 81, 184-197.
- Wagner, A.J.; Ravi, V.; Ganjoo, K.N.; Tine, V.B.A.; Riedel, R.F.; Chugh. R. ABI-009 (nab-sirolimus) in advanced malignant perivascular epithelioid cell tumors (PEComa): Preliminary efficacy, safety, and mutational status from AMPECT, an open label phase II registration trial. American Society of Clinical Oncology; 2019.
- Elimam, H.; El-Say, K.M.; Cybulsky, A.V.; Khalil, H.; Regulation of Autophagy Progress via Lysosomal Depletion by Fluvastatin Nanoparticle Treatment in Breast Cancer Cells. ACS omega. 2020, 5, 15476-15486. [CrossRef]
- Liu, Z.; Lv, X.; Xu, L.; Liu, X.; Zhu, X.; Song, E.; et al. Zinc oxide nanoparticles effectively regulate autophagic cell death by activating autophagosome formation and interfering with their maturation. Part. Fibre. Toxicol. 2020, 17, 46. [CrossRef]
| Nanocarrier | Key features and associated benefits |
|---|---|
| Liposomes | Liposomes exhibit several advantages, such as biocompatibility, biodegradability, and the ability to reduce drug side effects. Liposomes enable controlled drug release while preventing rapid metabolism and clearance [55]. |
| Carbon nanotubes | CNTs have emerged as highly efficient drug delivery carriers and non-invasive therapeutic agents for the administration of anticancer drugs as well as for gene delivery applications, Furthermore, CNTs can serve as a medium for photothermal and photodynamic therapies, enabling precise treatment through the direct destruction of cancer cells while minimizing collateral damage to normal tissues [56]. |
| Polymeic micelles | Polymeric micelles are multifunctional nanomaterials that enhance delivery of poorly soluble drugs (e.g., cancer therapeutics and gene vectors) by modulating release profiles and concentrating agents at target sites. Their stimuli-responsive properties further enable targeted delivery to specific cancer locations [57]. |
| Quantum dots | Quantum dots are nanoscale semiconductor crystals with high brightness and photostability, making them promising for oncology applications. They serve as novel delivery vehicles, enabling combined tumor treatment and imaging through complexes with tumor-targeting drugs [58] |
| Albumin-drug conjugates | Albumin is the most abundant protein in plasma. It has good biocompatibility, no cytotoxicity, no immunogenicity, and biodegradability, which makes it an ideal material for preparing nanoparticles. In addition, albumin can enhance drug targeting, reduce the toxicity of free drugs, and enhance the water solubility of hydrophobic drugs [59]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




