1. Introduction
The Paris Agreement [
1], emphasizes the development of low- or zero-carbon solutions, with the ultimate goal of achieving carbon neutrality. As a result of this commitment, nations worldwide are striving to reduce their dependence on carbon-based fuels, thereby contributing to a significant reduction in environmental pollution [
2,
3].
Following a period of accelerated global coal consumption, the situation has stabilized in recent years as natural gas has gradually taken over as the primary energy source [
4]. In terms of consumption, the residential sector is a major user (often second only to the industrial sector), where natural gas remains the dominant fuel for heating and cooking
[5,6,7,8,9]. However, pollution-related challenges persist: conventional cooking systems fueled by natural gas emit carbon dioxide (CO2) as their primary pollutant [
10].
To meet climate commitments aimed at increasing the share of non-fossil energy, states must transition toward zero-carbon fuels. Hydrogen is therefore considered to hold major potential as a primary energy source, including in residential applications. A transitional solution increasingly explored today is the blending of hydrogen with natural gas, which represents an effective means of reducing harmful carbon dioxide (CO2) [
11,
12] emissions while improving the thermal performance of gas-fired appliances [
13]. A study conducted by Field and Derwent shows that, for instance in the United Kingdom, replacing natural gas with hydrogen in residential use could reduce annual CO₂ emissions by 76 million tons [
14].
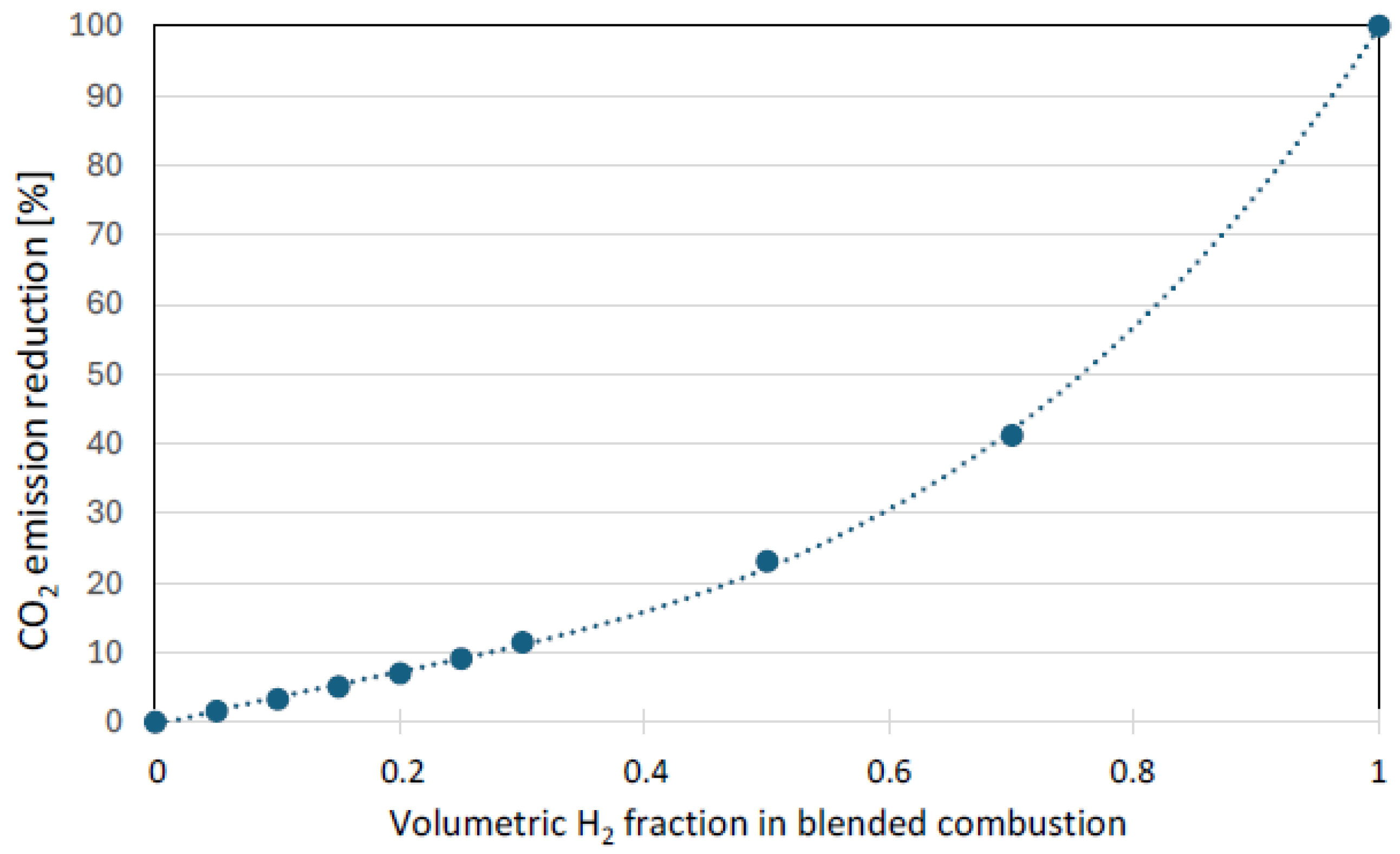
The use of hydrogen as an additive to natural gas in household applications, while offering benefits through reduced CO2 emissions (
Figure 1) and enhanced thermal efficiency, is governed by critical safety concerns. Hydrogen is recognized as an extremely flammable and explosive gas, with the highest rating of 4 on the NFPA 704 scale [
10]. Attention must therefore be paid to concentration limits. The most significant risk identified is the phenomenon of flame flashback, which can occur at relatively low concentrations in the gas network.
Di Lullo et al. [
15] and collaborators analyze the impact of blending blue hydrogen with natural gas in the existing infrastructure for transport and direct consumption, through a comprehensive evaluation of well-to-combustion (WTC) emissions. Their study shows that a mixture of 15% H₂ and 85% NG reduces combustion emissions by approximately 4–5%, but increases pipeline transport emissions by about 8% and decreases the energy capacity of the network by 11%. In the absence of CO₂ capture, total WTC emissions rise slightly (+2%), whereas scenarios with partial or full capture can maintain or even reduce emissions (up to −3.6% in the optimal scenario).
A previous review dedicated to the enhancement of domestic gas stoves [
16] also touched on the potential role of hydrogen addition; however, as the author explicitly noted, the analysis could not fully address hydrogen-enriched natural gas due to the limited scientific documentation available at that time. The rapid expansion of experimental and numerical studies in recent years now allows for a more comprehensive assessment of HENG behavior in residential appliances, filling the gap left by earlier reviews.
The purpose of this review is to synthesize current knowledge on the combustion characteristics of hydrogen-enriched natural gas, with particular emphasis on residential appliances. By integrating findings from experimental studies, computational analyses, and regulatory perspectives, the paper aims to clarify how hydrogen blending influences flame stability, pollutant emissions, thermal efficiency, and safety concerns in domestic-scale applications. To provide a clear and systematic overview, the selected literature has been organized into four categories: (i) studies addressing the phenomenon of flashback, (ii) investigations focused on efficiency, emissions, and temperature behavior, (iii) research examining flame geometry, and (iv) experimental works that do not fall neatly into the previous categories but contribute valuable insights. This structure highlights both the potential of hydrogen as a transitional fuel toward carbon neutrality and the technical challenges that must be addressed to enable its safe and efficient adoption in household energy systems.
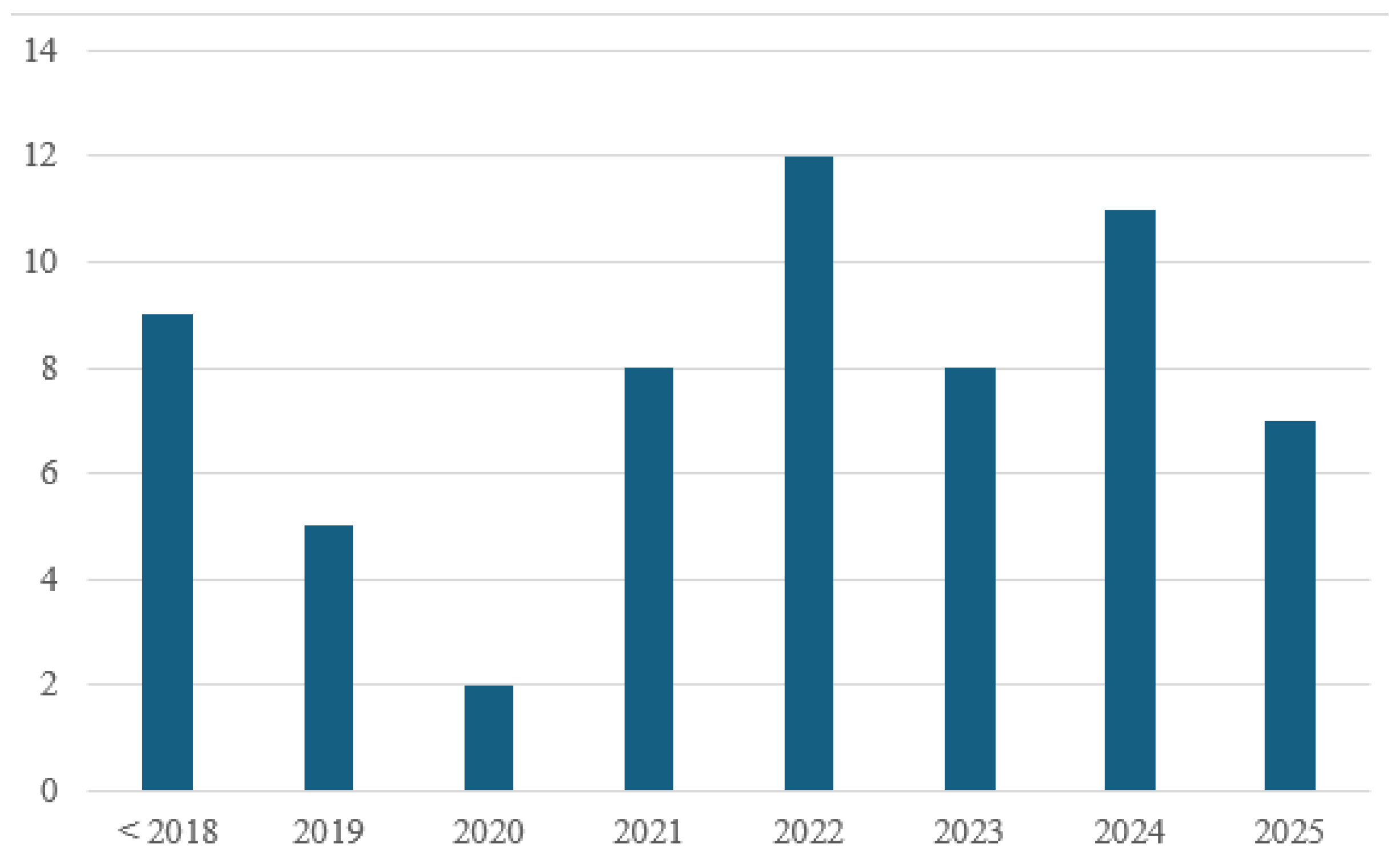
To ensure a comprehensive coverage of the topic, the literature review was conducted using the Web of Science database. The search strategy employed a combination of keywords related to hydrogen-enriched natural gas and residential combustion systems. Specifically, the following terms were used in various Boolean combinations: Topic – AND: hydrogen, natural gas, combustion, burner; Topic – NOT: engine, turbine, syngas, ammonia, catalytic, catalyst, reformer. A total of 450 results were shown, which were further filtered manually. A chart showing the year of publication for the selected articles is presented in
Figure 2. Papers that are published before 2018 consist mainly of research regarding the general interaction between hydrogen and natural gas, while the work published after 2018 consists mostly of research that considers also the implementation of HENG (hydrogen-enriched natural gas) as fuel for domestic applications.
2. Studies Regarding the Flashback Phenomenon
The transition toward hydrogen-enriched natural gas (HENG) raises important safety challenges, among which the flashback phenomenon is one of the most critical. Flashback occurs when the flame propagates upstream into the burner or mixing zone, and its likelihood increases significantly as hydrogen concentration rises due to the higher burning velocity and enhanced diffusivity of H₂. Understanding the mechanisms that govern flashback, ranging from flammability limits and burner wall temperature to preferential diffusion effects, is essential for ensuring the safe operation of domestic and industrial appliances. This chapter reviews the main analytical, experimental, and numerical studies that investigate flashback behavior in HENG systems, highlighting the key parameters that influence stability and the implications for burner design, appliance certification, and hydrogen integration limits.
Analytical and experimental studies have shown that the flashback phenomenon occurs in domestic gas-fired appliances when the volumetric concentration of hydrogen is between 20% and 25% for cooking devices [
10,
17,
18], while in the case of a boiler, the safety limit identified was up to 40%[
19,
20,
21].
The study conducted by Miao et al. [22] investigated the flammability limits of hydrogen-enriched natural gas using a constant-volume combustion chamber and optical visualization techniques. The results showed that as the hydrogen fraction increases, the flammability limits widen significantly: the lower limit remains nearly constant at around 4–5% by volume, whereas the upper limit increases from approximately 16% for natural gas to over 70% for pure hydrogen.
Aniello’s study [23] analyzes the substitution of methane with hydrogen in commercial premixed burners and the resulting effects on blow-off and flashback limits. The results indicate that adding H₂ enhances blow-off resistance but simultaneously increases the thermal load on burner walls and promotes flashback. It was demonstrated that simply comparing the flow velocity at the burner exit with the laminar burning velocity is insufficient to predict flashback. The determining factor is the thermal state of the burner, specifically the temperature of the metallic surface. Transient analyses revealed a direct correspondence between the onset of flashback and the attainment of a critical wall temperature for a given mixture composition. The main conclusion is that although hydrogen improves blow-off stability, it restricts the operating range due to the increased risk of overheating and flashback, requiring a redesign of burners to ensure compatibility with hydrogen-rich mixtures.
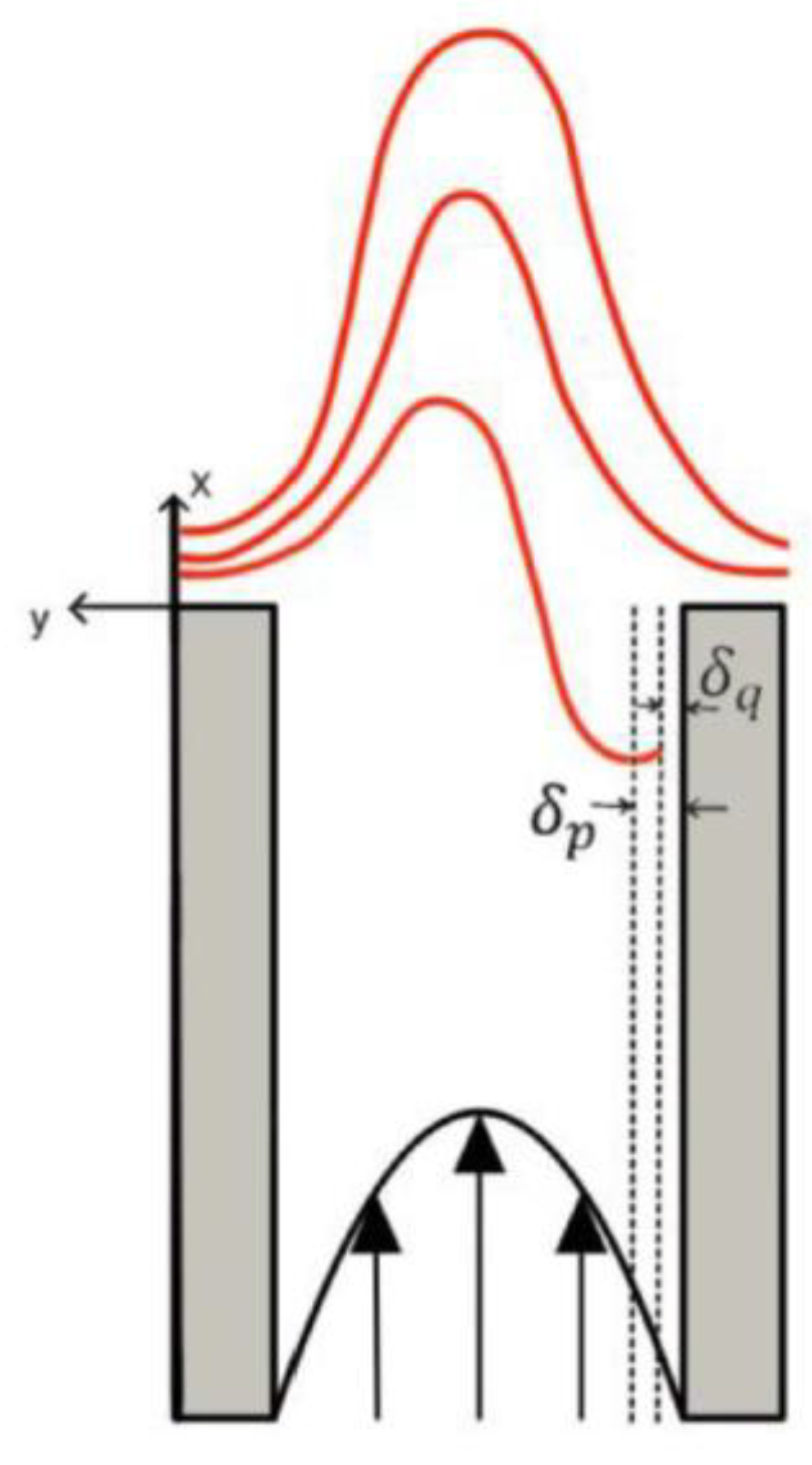
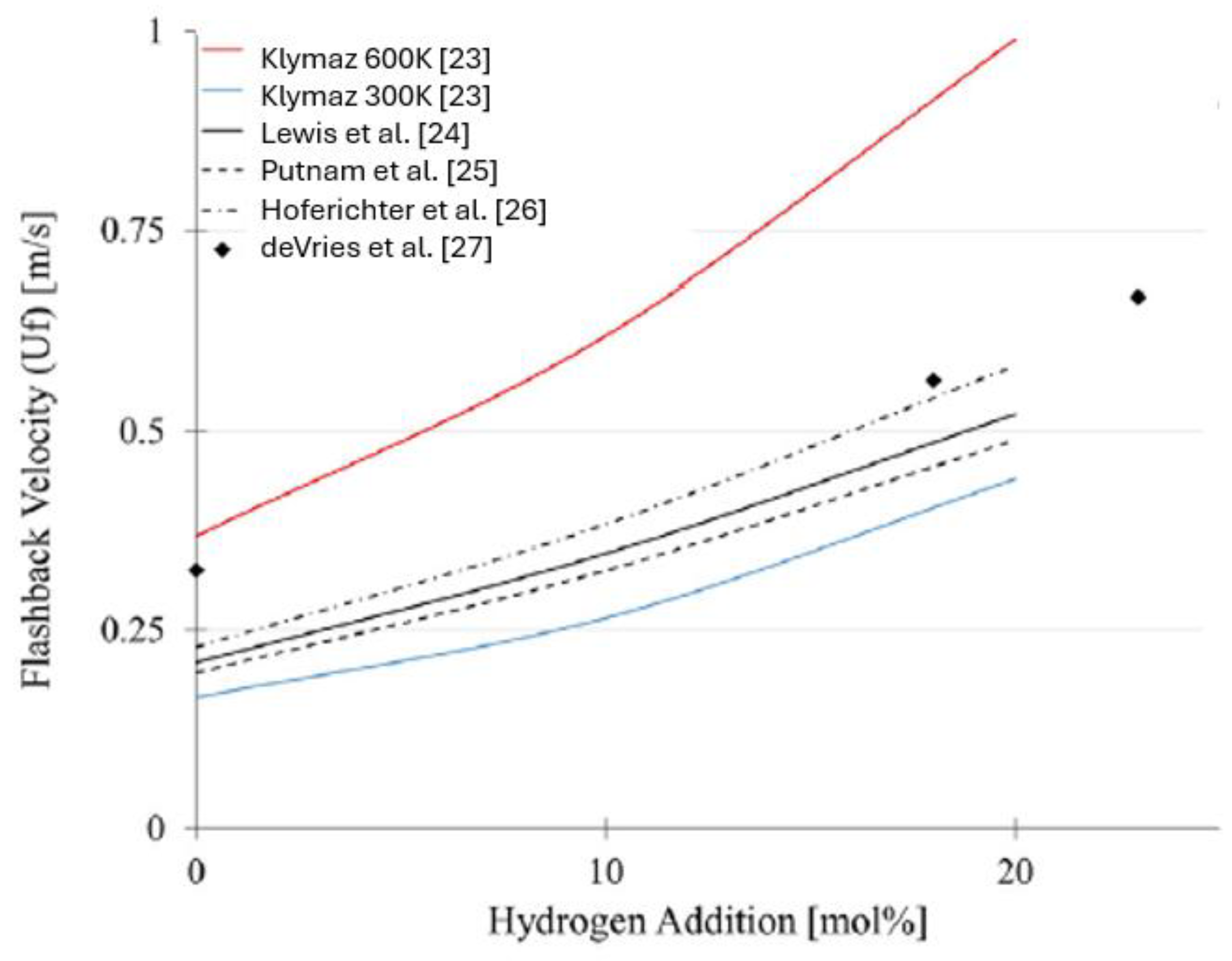
Klymaz [24] highlights that burner wall temperature plays a critical role: at 600 K the risk of flashback is significantly higher than at 300 K, due to modifications in the thermal boundary layer (
Figure 3).
Enrique et al. [29] and Fruzza et al. [
30,
31] numerically investigate the influence of hydrogen enrichment on premixed flames, focusing on flashback limits and flame behavior. Their results reveal the existence of two distinct flashback regimes: a symmetric regime for low H₂ content (up to 75% H₂[
30,
31]) and an asymmetric regime (
Figure 4) for high concentrations (>87.5%[
30,
31]). The critical threshold is defined by an effective Lewis number (the ratio of thermal to mass diffusivity) of 0.5, below which preferential diffusion of hydrogen dominates flame dynamics. The main conclusion is that flashback risk in hydrogen-containing flames is governed not only by burning velocity but also by diffusion effects, requiring careful consideration in burner design.
The study by Cuoci et al. [
32] focused on predicting flashback limits in hydrogen–methane–air mixtures using numerical simulations. It concluded that conventional flashback models based on the critical velocity gradient become inadequate for H₂ fractions exceeding 50%, highlighting the need to develop new models that incorporate preferential diffusion effects to ensure safe operation of such systems. The authors also examined the influence of hole distribution, specifically the spacing between them. It was observed that depending on mixture composition, this spacing has different effects. For fuel-rich mixtures (ϕ > 0.50), modifying the hole spacing significantly affects the specific flashback power, whereas for lean mixtures (ϕ = 0.50), the flashback limits remain unchanged.
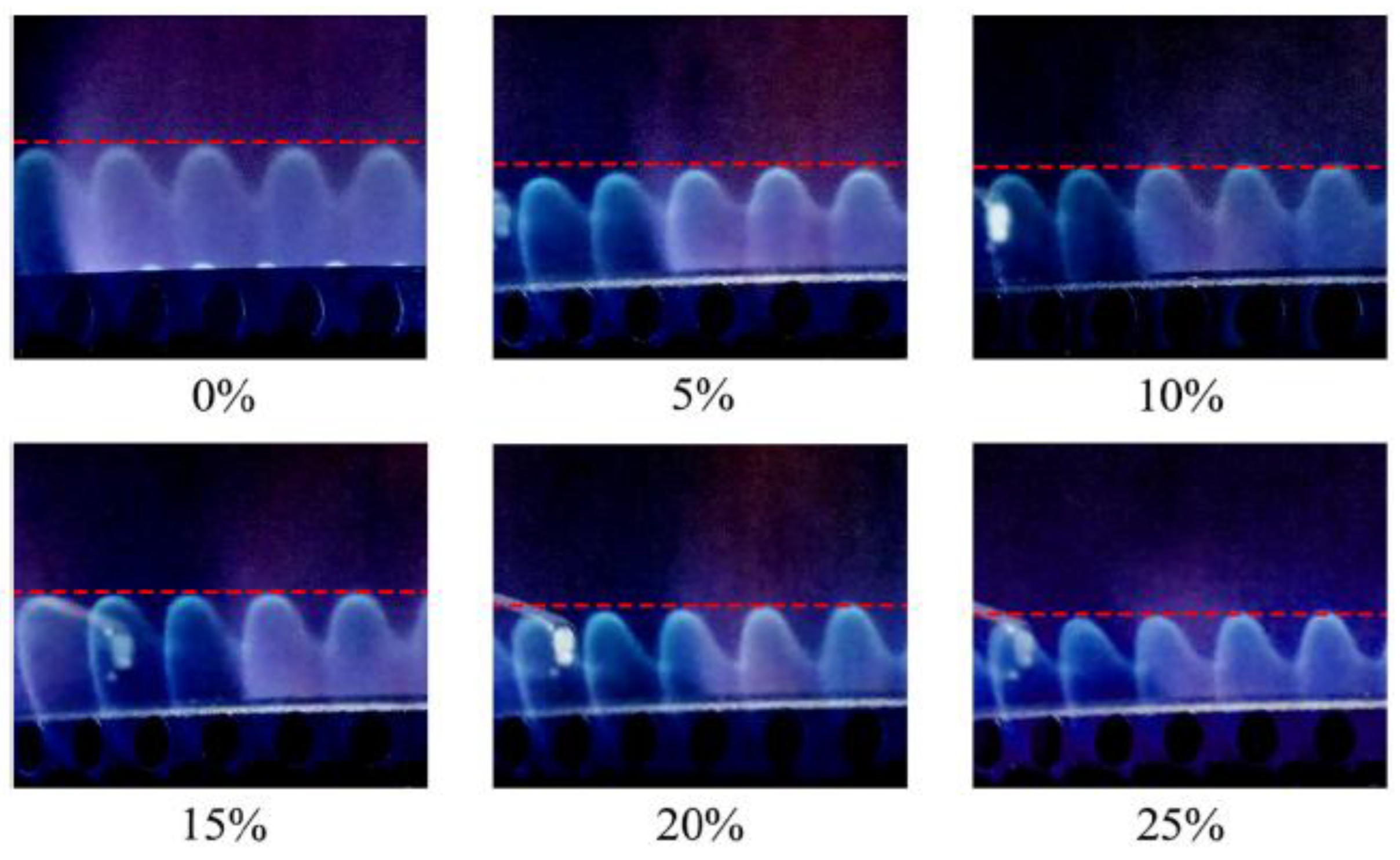
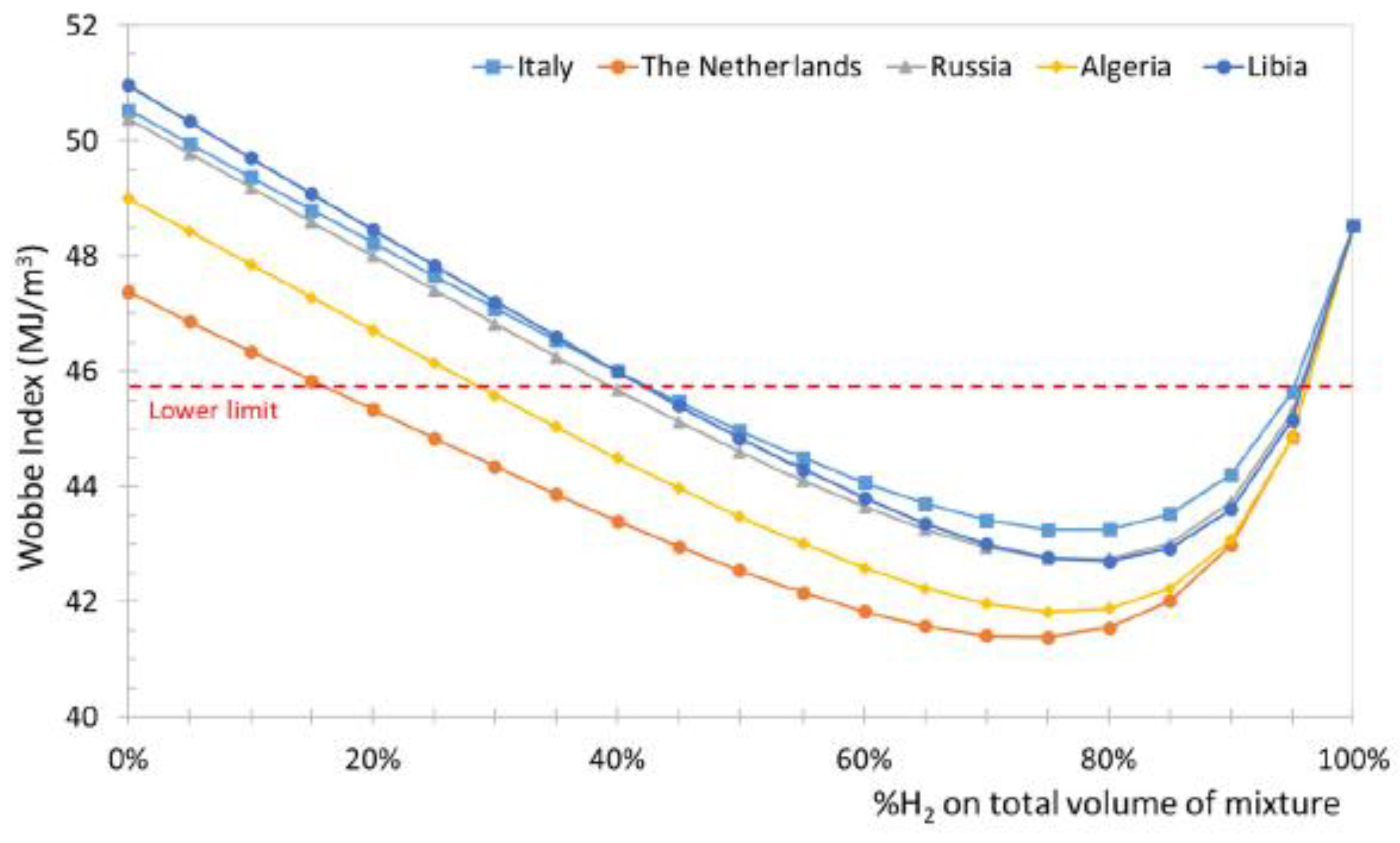
Leicher [33] also emphasizes that relying solely on the Wobbe index is insufficient for assessing the impact of H₂, and that adapting combustion technologies and certification procedures is essential for the safe integration of H₂-NG blends in the residential and commercial sectors. The Wobbe Index is the main metric used to assess the interchangeability of gaseous fuels, linking heating value to density and indicating how much energy a burner receives under fixed conditions. For hydrogen–natural gas blends, it helps determine whether existing appliances can operate safely without modification. Although moderate hydrogen levels (≈20–30%, see
Figure 5) typically keep the Wobbe Index within acceptable limits, the index alone cannot predict changes in flame speed, flashback risk, or NOx formation, so it must be complemented by detailed combustion analysis. Gases whose Wobbe index differs by no more than 5–10% are considered interchangeable for most gas-fired applications. However, Xie et al. [34] demonstrated that the Wobbe index lose applicability for HENG blends with hydrogen fractions above 20%.
The overarching conclusion is that flammability data obtained for methane–hydrogen mixtures can be used to characterize the behavior of hydrogen-enriched natural gas, and the extension of these limits highlights both the potential for more stable combustion and the increased explosion risks at high H₂ concentrations [
22,
35,
36,
37,
38,
39].
3. Investigations Focused on Efficiency, Emissions, and Temperature Behavior
3.1. General Studies
Understanding how hydrogen enrichment influences combustion behavior is essential for evaluating the feasibility of hydrogen–natural gas blends in practical applications. General studies in the literature examine fundamental parameters such as laminar burning velocity, adiabatic flame temperature, radical formation, and thermal output, providing a baseline for predicting system-level performance. These investigations also assess how hydrogen affects efficiency, emissions, and flame structure across a range of conditions and appliance types. Together, they establish the foundational trends (hotter and faster flames, lower carbon-based emissions, and shifting operational limits) that guide subsequent analyses of burner design, appliance compatibility, and safe integration of hydrogen into existing gas infrastructures.
Ren et al. [40] analyzed the effect of hydrogen addition on the combustion characteristics of the main alkanes present in natural gas (CH₄, C₂H₆, C₃H₈, C₄H₁₀). Three combustion mechanisms were compared against experimental data to identify the most suitable one (GRI Mech 3.0, USC Mech 2.0, San Diego). The results showed that both the laminar burning velocity and the adiabatic flame temperature increase with rising hydrogen fraction, with the strongest effect observed for methane. A strong correlation was also identified between the increased concentration of active radicals (H, O, OH) and the intensification of flame-propagation speed.
The study by Nosek et al. [
41] focused on the numerical analysis of hydrogen-enriched natural gas mixtures (up to 45%) using a mathematical model. The results confirmed that the mixtures remain compatible for use (Wobbe Index preserved), but revealed an inverse correlation: at constant volumetric flow rate, increasing H₂ concentration leads to a significant reduction in thermal power output (by 44.9% at 45% H₂), requiring fuel-flow adjustments to maintain performance. Environmentally, a 25% reduction in CO₂ emissions was confirmed for a mixture containing 25% H₂.
Hasche’s results [
42] show that increasing the H₂ fraction significantly raises the adiabatic flame temperature from ~2350 K for methane–oxygen to ~3100 K for pure hydrogen–oxygen. At the same time, the laminar burning velocity increases more than threefold from ~316 cm/s for methane–oxygen to ~954 cm/s for hydrogen–oxygen, reaching up to 2.7 m/s at higher inlet temperatures (700 K). Experimentally, it was observed that flame length decreases with hydrogen addition (at 35% H₂ the reduction was ~0.5 mm compared to pure methane). Spectroscopic investigations revealed an increase in OH* radical concentration and an intensification of oxidation reactions, explaining the higher burning velocity and more compact thermal profiles.
Wojtowicz et al. [
43] conducted a study on several types of household appliances operating on natural gas, evaluating combustion quality, ignition behavior, flame stability, and thermal efficiency for different hydrogen concentrations added to the base fuel (10%, 15%, and 23%). The study found that hydrogen fractions up to 23% should not have any negative impact on appliance safety. In terms of efficiency, a maximum decrease of 5% was recorded for cooking appliances, a value that is unlikely to be noticeable to the end user.
The main conclusion is that hydrogen addition in premixed methane–oxygen flames leads to hotter, faster, and more compact flames, with reduced CO₂ and CO emissions but an increased potential for NOx formation [44]. This highlights the need for optimizing the equivalence ratio and burner design to ensure safe and efficient operation.
3.2. Studies Focused on Cooktop Burners
Cooktop burners represent one of the most widespread domestic applications of natural gas, making them a critical test case for assessing the compatibility of hydrogen-enriched fuels. Because these appliances operate with open flames, relatively small burner geometries, and direct user interaction, their performance is highly sensitive to changes in flame temperature, burning velocity, and heat-transfer characteristics introduced by hydrogen addition. Recent studies have therefore focused on understanding how hydrogen affects flame structure, thermal efficiency, pollutant emissions, and operational stability in domestic stoves. The following works examine these aspects through experimental measurements, numerical simulations, and burner-design optimization, providing insight into the blending limits and design adaptations required for safe and efficient hydrogen use in household cooking appliances.
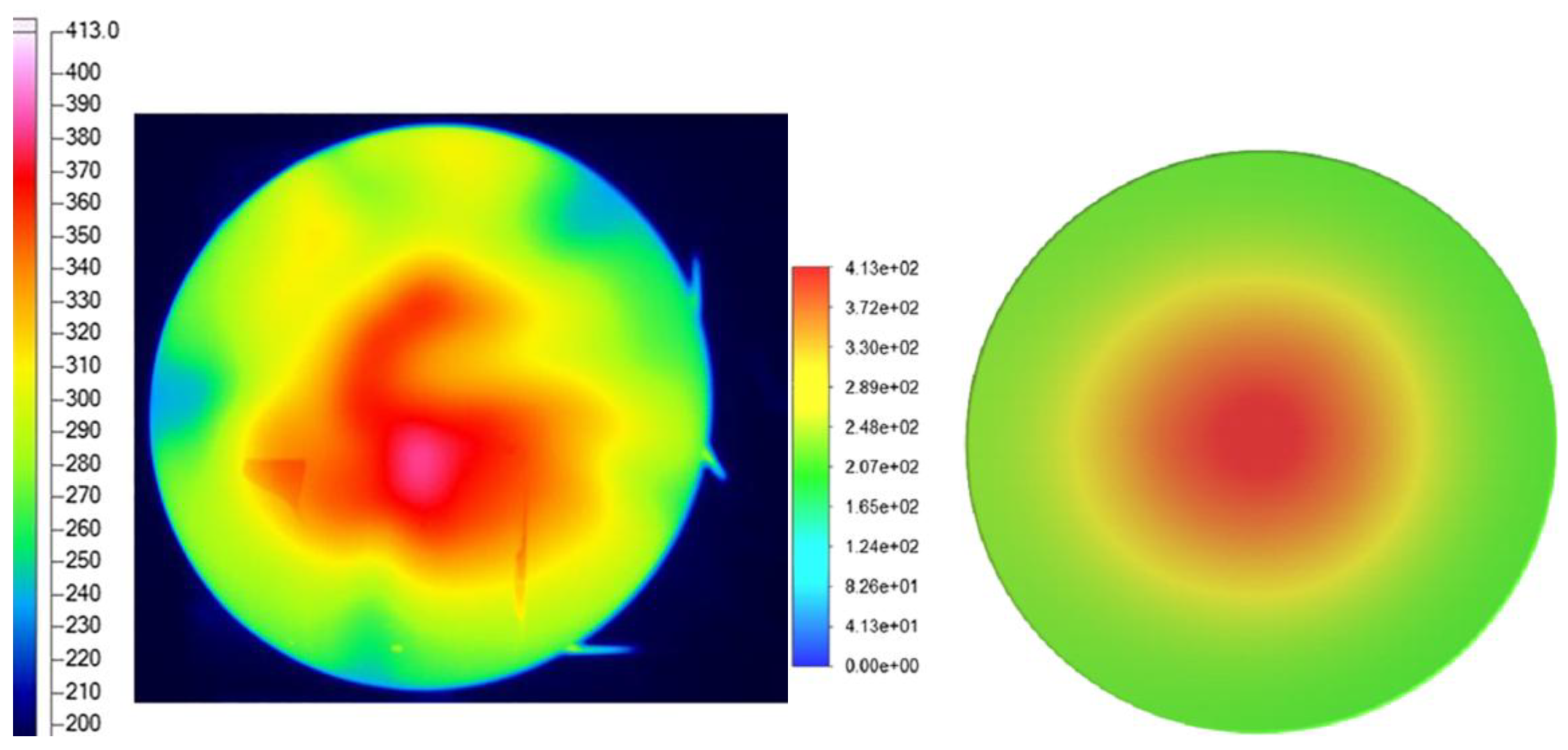
Liu et al. [
10] demonstrated both experimentally and numerically that, for a domestic cooking appliance, a 15% hydrogen fraction in natural gas leads to a 6.7% decrease in the average combustion-zone temperature (
Figure 6), while CO emissions are reduced by 25% (
Figure 7).
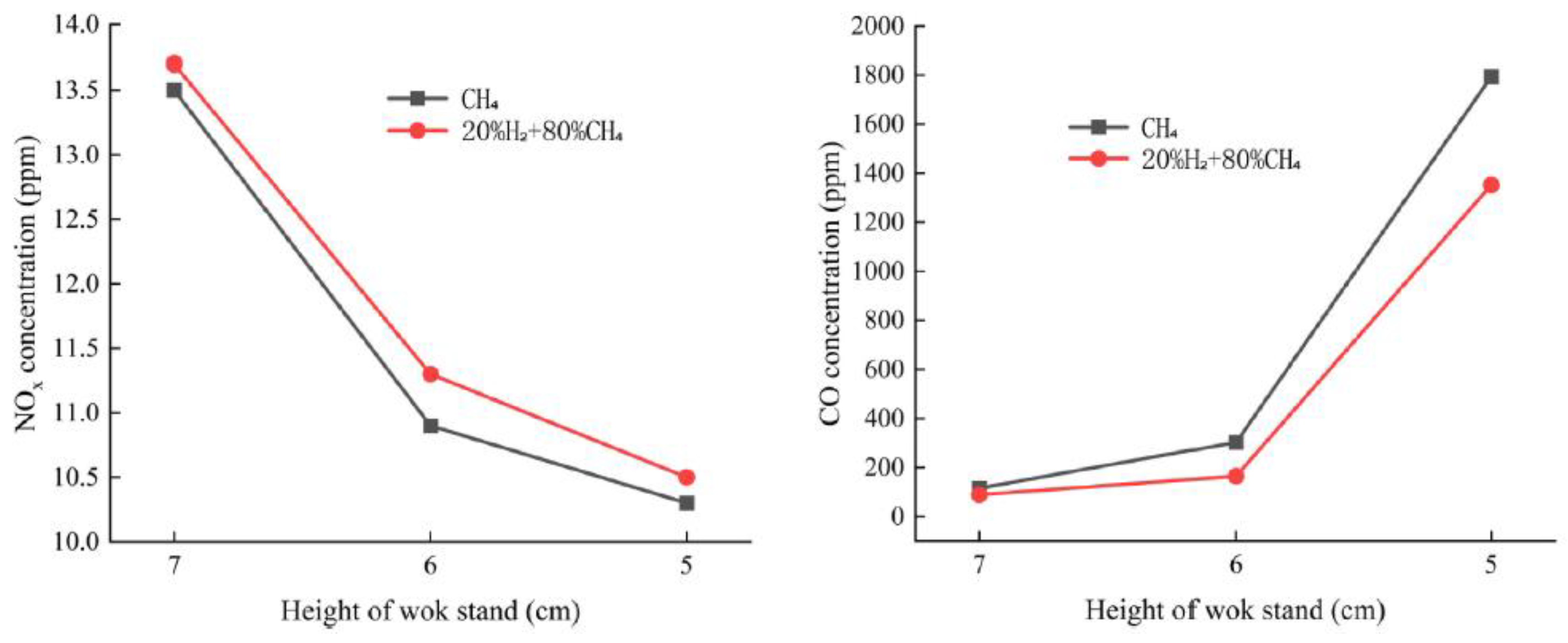
They also experimentally investigated the behavior of a household stove fueled with an 80% natural-gas / 20% hydrogen mixture, as well as geometric optimization through adjusting the wok-support height (
Figure 8) [45]. The results showed that the flame becomes shorter but hotter, which initially reduces heat-transfer efficiency and increases NOx emissions, while CO emissions decrease. Lowering the wok support from 7 cm to 6 cm increased thermal efficiency, reduced NOx emissions, and caused only a marginal rise in CO.
The main conclusion is that moderate geometric adjustments (such as support height) allow the advantages of hydrogen combustion to be exploited while maintaining performance and meeting emission standards.
The study by Z. Fang et al. [
46] analyzes the performance of three types of domestic burners supplied with CH₄/H₂ mixtures containing up to 25% hydrogen. The results show that the flame becomes shorter and more intense as the hydrogen fraction increases (
Figure 14), and the average efficiency increases slightly for TRPGS (typical round-port gas stove) and SSPGS (swirling strip-port gas stove). For RPMGS (radiant porous-media gas stove), efficiency increases up to 15% H₂ and then decreases. CO emissions decrease significantly at 25% H₂, while NOx emissions remain nearly constant. Combustion remains stable up to 25% H₂, although the reduced distance between the flame and burner surface may affect component durability.
Ahemat’s study [
47] comparatively investigates emissions generated by butane, methane, and methane–hydrogen mixtures in domestic stove burners, using EN 30-1-1-compliant experiments and CFD simulations. The main objective is to evaluate CO and CO₂ levels after combustion and to determine how hydrogen affects combustion performance. The results show that butane produces the highest emissions (CO ≈ 0.30 ppm), methane generates lower values (≈ 0.005% CO), and the methane–hydrogen mixture further reduces CO to 0.13 ppm. Additionally, butane combustion reaches maximum temperatures of approximately 2254 K, explaining its higher emissions. The overall conclusion is that adding 50% hydrogen to methane improves combustion efficiency and significantly reduces carbon-based emissions, supporting the transition toward cleaner and more efficient cooking systems.
Chen et al. [
48] propose an optimized porous-medium domestic burner for natural-gas/hydrogen mixtures, analyzing acceptable blending limits through experimental and numerical methods. The results show that above 45% H₂, flashback occurs in the injector and mixture uniformity decreases. Two combustion regimes (immersed and surface) appear in the porous zone, with transitions that cause sudden increases in CO emissions and decreases in efficiency and NOx emissions. The conclusion is that, to balance safety, emissions, and performance, the hydrogen fraction should not exceed 35%
3.3. Studies Focused on Boilers
Boilers represent a key application area for assessing the feasibility of hydrogen–natural gas blends, as their operating conditions, combustion chamber geometry, and heat-transfer requirements differ significantly from those of cooktop burners. Compared with domestic stoves, residential boilers have been less extensively studied in the context of hydrogen enrichment, resulting in a limited pool of experimental data. For this reason, several semi-industrial and industrial boiler studies are also included in this section: although they operate at higher thermal loads, their combustion behavior, emission trends, and stability characteristics remain relevant for understanding the broader implications of hydrogen addition. Collectively, these investigations provide insight into how hydrogen affects flame temperature, pollutant formation, thermal efficiency, and operational safety across a range of boiler configurations. The following studies therefore contribute to defining practical blending limits and identifying the design and control strategies needed to ensure reliable and low-emission operation in both residential and industrial heating systems.
Mayrohfer et al. [
21] investigated the use of CH₄/H₂ mixtures (0–40%) in heating systems and reported a 1.2% increase in efficiency and a 16.6% reduction in CO₂ emissions at 40% H₂, correlated with a decrease in flue-gas temperature from 582 °C to 565 °C. By analyzing two combustion modes (staged-air combustion and flameless combustion) the authors showed that NOx emissions can be kept below the TA-Luft [49] limit of 350 mg/m³: in staged-air combustion this threshold is met for burner powers above 135 kW, while in flameless combustion the values are even lower (60–200 mg/m³, depending on power and hydrogen fraction).
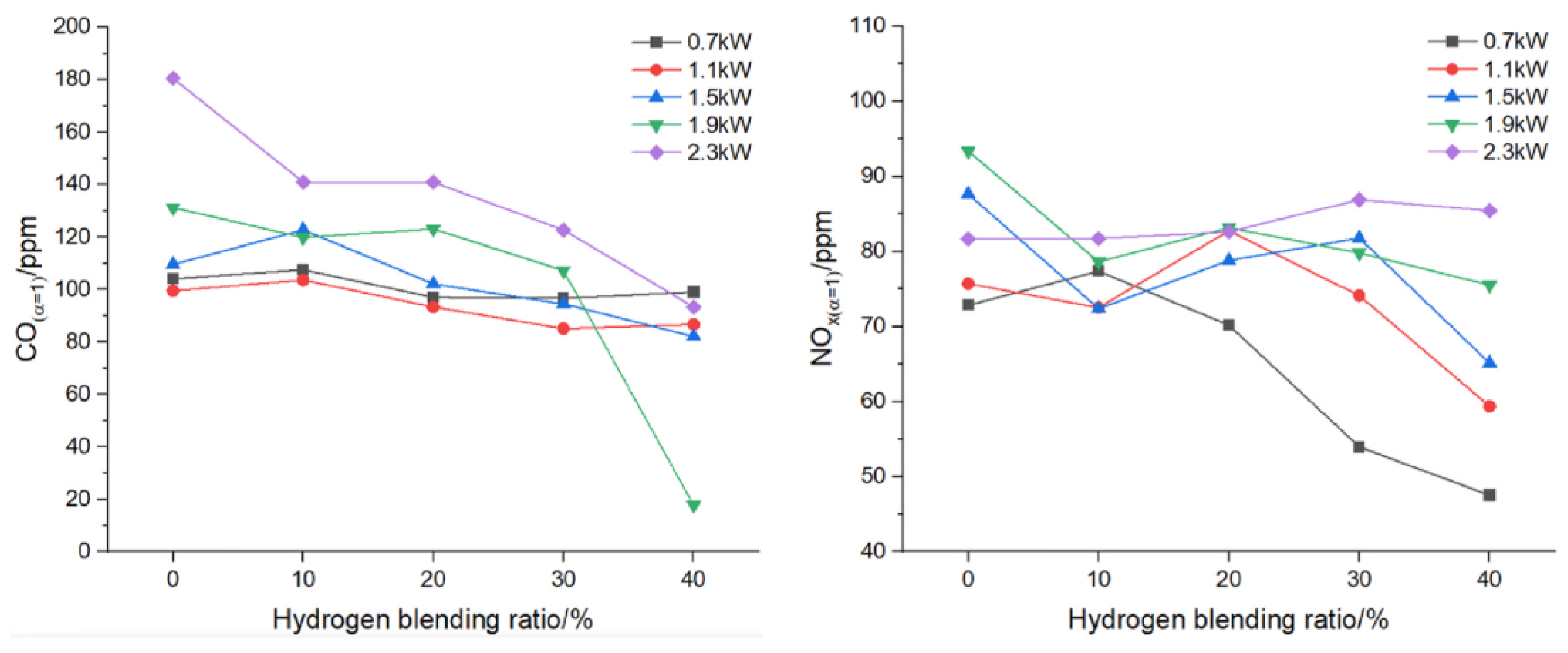
For a residential boiler, Zhan et al. [
19] observed that at the same thermal input, flame temperature decreases for low H₂ concentrations (10–20%) and then increases as the mixture becomes richer in hydrogen; at constant power, increasing the hydrogen fraction leads to a clear rise in temperature. CO emissions decrease slightly between 0.7–1.5 kW and more significantly at 1.9–2.3 kW, while NO variations remain small up to 20% H₂, becoming noticeable only at higher concentrations (
Figure 9).
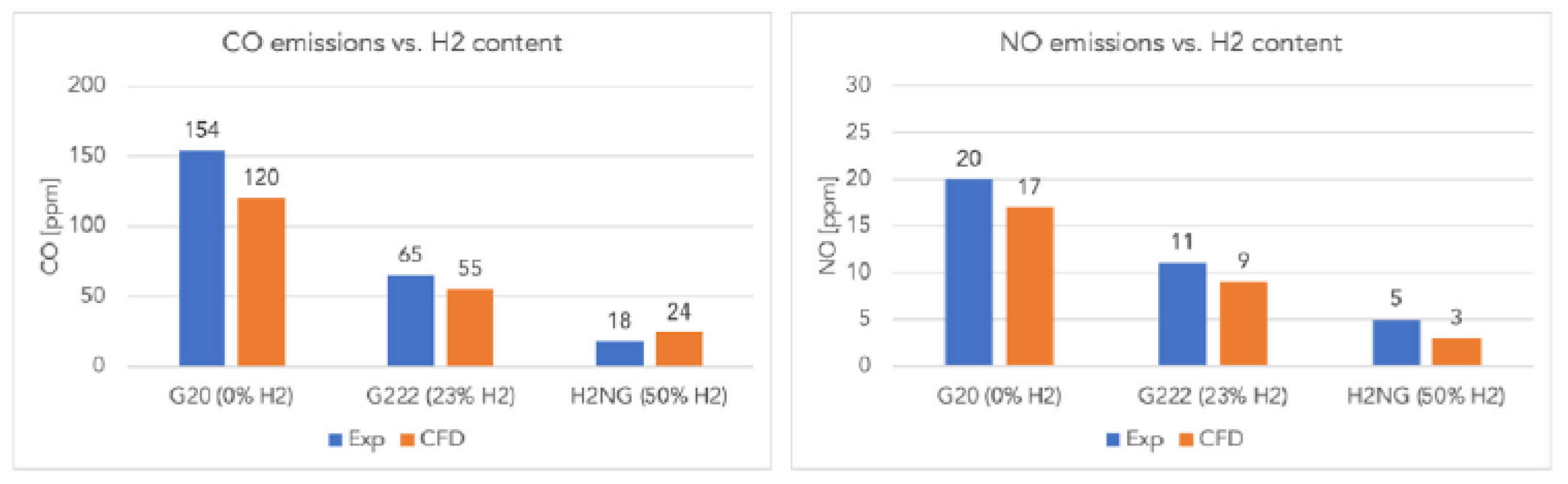
Similar results are reported by Lamioni [
50], who shows through CFD simulations validated experimentally that hydrogen addition significantly reduces pollutant emissions in condensing boilers: CO decreases from 154 ppm (pure CH₄) to 18 ppm (50% H₂), and NO from 20 ppm to 5 ppm. Moreover, 23% H₂ reduces CO₂ emissions by about 8.7%, and 50% H₂ by 24.2%, due to the more “diluted” combustion conditions (
Figure 10).
Consistent with Zhan, Siqi et al. [
20] tested CH₄/H₂ mixtures up to 40% and observed both an increase in temperature with hydrogen fraction and a more uniform temperature distribution in the combustion chamber. The study also reports a pronounced local increase in NO emissions near the injection zone; however, the minimum NO level is obtained at 20% H₂ (only a 14.7% increase compared to pure CH₄), while at 30% H₂ emissions rise by 47%, indicating an optimal limit around 20%.
Bălanescu et al. [
51] analyzed the variation of adiabatic flame temperature as a function of hydrogen fraction and excess-air coefficient in a 25 kW condensing boiler. The results indicate that increasing both the H₂ fraction and the excess air significantly reduces flame temperature, contributing to lower CO₂ emissions and improved energy efficiency. Specifically, increases of up to 49% in condensate production, 47% in boiler energy savings (
Figure 11), and a 56.15% reduction in CO₂ emission intensity were reported.
The study by Schiro et al. [
52] shows that enriching natural gas with hydrogen reduces CO₂ emissions and increases the amount of condensable water, which can enhance the efficiency of condensing boilers. However, hydrogen addition decreases volumetric heating value and requires higher fuel flow rates, while high concentrations introduce risks of flashback and undesired ignition. The main conclusion is that current boilers can operate safely and efficiently with approximately 20–23% H₂, but higher proportions require design modifications to components and condensate-handling systems.
Wahiba et al. [
53] investigated the performance of domestic heating systems fueled with hydrogen-enriched natural gas (HENG) and pure hydrogen. Their results show that thermal efficiency increases with hydrogen fraction due to the higher water-vapor content in the flue gases. However, excess air reduces exhaust-gas temperature and condensation efficiency. The best performance is achieved under stoichiometric combustion with pure hydrogen, though this condition increases pollutant-formation risks and requires well-controlled lean combustion.
Jankowski [
54] evaluated the thermal performance and emissions of condensing boilers fueled with natural-gas/hydrogen mixtures up to 50% H₂. The results show that hydrogen addition enhances convective heat transfer, flue-gas radiation, and energy recovery from water-vapor condensation (up to +6%). Overall efficiency increased by about 1.6 percentage points at 50% H₂, without significant exhaust-gas losses. Pollutant emissions dropped sharply: NOx decreased to 2.7 mg/m³ and CO to 9.9 mg/m³, well below regulatory limits (33.5 mg/m³).
4. Research Examining Flame Geometry
Flame geometry is a key indicator of combustion behavior, strongly influencing heat transfer, stability, pollutant formation, and the overall performance of gas-fired appliances. Because hydrogen addition alters fundamental flame properties (such as burning velocity, diffusivity, and thermal release) its impact is often first observed through changes in flame height, shape, and attachment. Understanding these geometric transformations is therefore essential for evaluating the safe integration of hydrogen into domestic and small-scale combustion systems. The studies reviewed in this chapter investigate how hydrogen concentration, burner design, and operating conditions affect flame morphology across boilers, cooktop burners, and oven systems, providing insight into both the benefits and the operational constraints associated with hydrogen-enriched fuels.
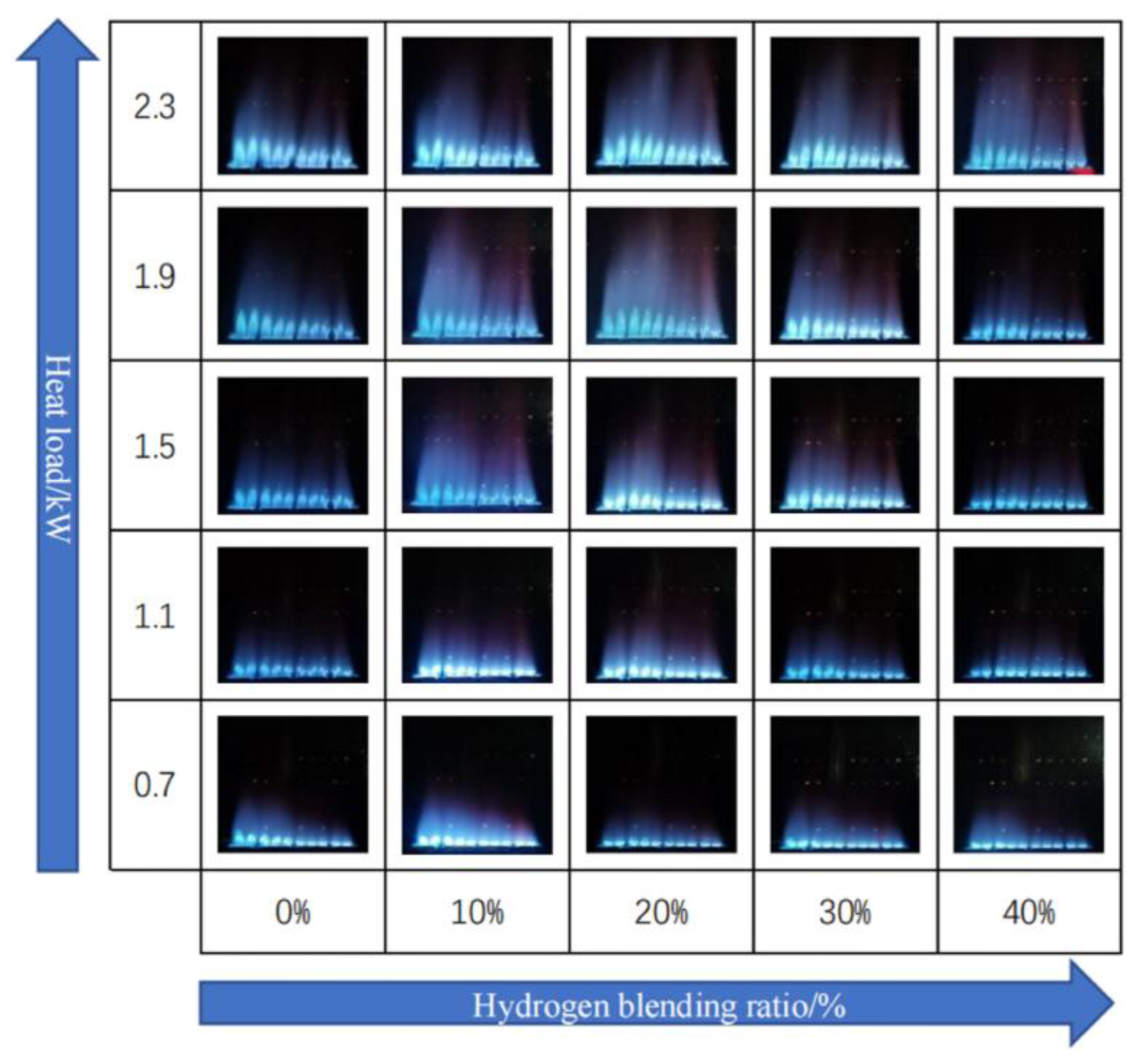
Zhan et al. [
19] studied the impact of hydrogen concentrations ranging from 0–40% by volume in a domestic boiler operating within a thermal power range of 0.7 to 2.3 kW. They reported an initial increase followed by a decrease in flame height as the hydrogen fraction increased, for the same thermal input. Conversely, for a constant hydrogen concentration, increasing the thermal power resulted in a significant rise in flame height (
Figure 12).
Feith et al. [
55] investigated the influence of burner-head geometry on flame characteristics and combustion performance in household cooking appliances operating with natural gas–hydrogen mixtures (30% H₂ by volume). By testing six different burner configurations (
Figure 13), the authors demonstrated that hydrogen addition leads to shorter and flatter flames, as well as a shift in flame color toward blue, indicating more complete and efficient combustion. Their results showed that burner heads with multiple orifices arranged both laterally and on the upper surface generate more stable and compact flames, with width-to-height ratios between 0.27 and 0.67, underscoring the importance of burner-head design in optimizing the use of hydrogen-based alternative fuels.
The study conducted by Zhao [
35] evaluated the performance of an oven burner supplied with grid natural gas blended with hydrogen. The results showed that hydrogen addition reduces ignition time and CO emissions, but above 25% H₂ the risk of flashback during ignition becomes significant. Under steady-state operation, the flame becomes shorter and stabilizes closer to the burner surface when the hydrogen fraction reaches 10%, leading to localized overheating of components. However, beyond this 10% threshold, the burner temperature does not increase substantially further. NOx emissions remained practically unchanged, while the cyclic operating behavior of the oven produced complex temporal variations in emissions. The main conclusion is that hydrogen addition can improve ignition and reduce CO emissions, but safety constraints require that the hydrogen fraction should not exceed approximately 25% in residential applications.
Figure 14.
Flame of TRPGS with different hydrogen addition [
46].
Figure 14.
Flame of TRPGS with different hydrogen addition [
46].
Restrepo et al. [
56] examined the flame morphology of non-premixed natural gas–hydrogen mixtures, varying the hydrogen content from 0 to 100% and the air preheating temperature between 25 °C and 400 °C at a constant thermal input of 0.8 kW. Results showed that visible flame height consistently decreases with increasing hydrogen fraction, while air preheating produces a non-linear effect for blends up to 75% H₂ and a linear height increase for pure hydrogen. Overall, hydrogen addition up to 50% does not significantly alter visible flame morphology compared to pure natural gas.
5. Other Studies
Beyond the core research areas addressed in previous sections, a broader set of experimental and numerical investigations provides additional insight into how hydrogen enrichment influences combustion behavior across diverse heating and industrial systems. These studies examine aspects that do not fit neatly into the categories of residencial appliences, yet they offer valuable complementary evidence regarding the practical implications of hydrogen addition. The works reviewed here explore heating performance, fuel consumption, NOx formation mechanisms, turbulence effects, model validation, and the combined influence of hydrogen with oxygen enrichment or swirl-stabilized flow. Although many of these investigations involve semi-industrial or industrial-scale equipment, their findings remain relevant for understanding the fundamental combustion trends that also govern residential appliances. Collectively, they highlight both the benefits, such as improved stability, reduced carbon-based emissions, and enhanced heat-transfer rates, and the challenges, including increased NOx formation and higher component temperatures, that accompany the use of hydrogen-enriched natural gas in real-world combustion systems.
Fatih et al. [
57] evaluated the heating time required to raise 5 kg of water to 60 °C. Two identical burners were used: one supplied with natural gas and the other with three gas cylinders containing natural-gas mixtures with 10%, 20%, and 30% hydrogen. For pure natural gas, the measured heating time was 632 s. Adding 10% hydrogen increased the heating time by 13.4% (717 s). Increasing the hydrogen fraction to 20% and 30% did not lead to a linear rise in heating time, resulting instead in increases of only 15.8% (732 s) and 16.7% (738 s). The study also showed that using a mixture containing 20% hydrogen can reduce natural-gas consumption by 8% and improve burner efficiency by up to 44.4%.
Sun et al. [
58] confirm that the use of hydrogen-enriched natural gas in industrial boilers involves a practical trade-off between thermal efficiency and NOx control. Numerical simulations (up to 30% H₂) and field tests (up to 9.7% H₂) on a 2 t/h steam boiler show that simultaneously increasing the hydrogen fraction and the excess air lowers the theoretical combustion temperature, thereby reducing NOx emissions (e.g., a 5% decrease in tests, from 118 mg/m³ to 112 mg/m³, with an even stronger reduction in simulations). However, this strategy also increases flue-gas temperature, raising heat losses and causing a slight drop in overall boiler efficiency (less than 1% in tests). CO emissions remained below regulatory limits, confirming compliance with testing standards.
Schwarz et al. [
59] experimentally investigated hydrogen-enriched natural gas combustion at semi-industrial scale, focusing on NOx formation. Their results show that increasing the hydrogen fraction raises the peak furnace temperature and elongates the flame, improving energy efficiency (up to +5.5%). However, NOx emissions increased sharply, reaching +167% compared to natural gas, confirming their sensitivity to the higher temperatures induced by hydrogen.
Salci et al. [
60] modeled and experimentally tested the combustion of natural gas mixed with hydrogen in a 150 kW industrial burner. Up to 20% H₂ reduced CO₂ emissions by about 5.8%, improved flame stability, and increased flame temperature, velocity, and pressure. However, NO emissions rose with hydrogen concentration due to intensified OH radical formation and higher temperatures. Maintaining low NOx levels requires operating at higher excess-air coefficients (λ = 1.3–1.4). The study concludes that diffusion-flame industrial burners can safely operate with up to 20% H₂, offering real potential for industrial decarbonization.
Yamei et al. [
61] studied natural-gas/hydrogen combustion in an industrial furnace and highlighted four key findings. First, CFD simulations validated experimentally show flame-temperature increases of +47 K, +52 K, +72 K, and +91 K for 15%, 30%, 45%, and 60% H₂, respectively, while furnace outlet temperature remains nearly constant. Second, internal turbulence intensifies with hydrogen content, altering both flame height and width. Third, NO emissions rise significantly: +155% in the chamber and +146% at the outlet for 60% H₂. Finally, CO₂ and CO emissions decrease by 37.5% and 70%, respectively.
Kislinger [
62] conducted a comparative analysis of methane and hydrogen combustion in a semi-industrial furnace equipped with a low-swirl burner, using CFD simulations and experimental measurements. The results show that achieving the same sample temperature (~1060 °C) requires less fuel input with hydrogen (70 kW) than with methane (94 kW). Moreover, convective heat transfer is more intense with hydrogen, leading to a heating process approximately 13% faster.
Weidinger [
63] analyzed, through experimentally validated CFD simulations, how increasing hydrogen content in natural gas affects NOx emissions from an industrial forced-draft burner operating at 358 kW and an excess-air ratio of 1.2. The authors show that the Steady Diffusion Flamelet model accurately predicts emissions for mixtures with up to 70% H₂, while for 100% hydrogen the most accurate model is the Flamelet Generated Manifold, with only 1.3% deviation from measurements. An important observation is the increase in burner-surface and flame-tube temperatures (around 920–930 K depending on operating point), which promotes thermal NOx formation and may affect equipment integrity.
Zouhaier [
64] experimentally investigated how oxygen enrichment (20–50%) and hydrogen addition (up to 15%) modify the stability, structure, and emissions of a natural-gas flame in a coaxial burner. The results show that both enrichments extend the flame-existence domain and eliminate blow-off tendencies, producing a more stable flame. Aerodynamically, increasing oxygen reduces oxidizer flow rate and significantly decreases axial velocity in the outer jet, while hydrogen addition increases fuel-jet velocity (up to ~15.6 m/s), reducing the velocity gradient between the two jets and improving mixing. PIV profiles show that transverse velocities increase when oxygen decreases (from 50% to 20%), intensifying turbulence in the internal mixing zone. Regarding emissions, CO decreases, CO₂ increases, and NOx rises sharply (from ~90 ppm to ~331 ppm) with increasing oxygen content due to higher flame temperatures.
The numerical simulation study of turbulent natural-gas/H₂ combustion in a swirl-stabilized chamber [
65] highlights the significant impact of hydrogen addition on thermal behavior and emissions. As the molar fraction of H₂ increases, both the average and peak temperatures in the combustion chamber rise, and the thermal field becomes more uniform. CO emissions decrease substantially, while NO emissions increase, particularly at high H₂ concentrations and high primary-air ratios. Introducing secondary air lowers the reaction-zone temperature and limits NO formation even during pure-hydrogen combustion. Overall, controlling the primary-air fraction and employing secondary air are essential for optimizing NG/H₂ combustion in terms of efficiency and emissions.
The study conducted by Du et al. [
66] investigated the combustion behavior of natural-gas/hydrogen mixtures in swirl burners using experiments and CFD simulations. The results showed that the swirl angle and the length of the recirculation zone directly influence flame stability, temperature distribution, and pollutant emissions: increasing the swirl angle to 45° led to more stable combustion with more uniform temperatures and reduced CO and NO emissions, while extending the recirculation-zone length to 12 mm reduced NO emissions by up to 36% compared with 6 mm.
6. Conclusions
This review consolidates current findings on the behavior of hydrogen-enriched natural gas in residential combustion systems, revealing both the opportunities and the technical challenges associated with hydrogen integration.
i. Flashback remains the primary safety constraint.
Hydrogen’s high burning velocity and diffusivity significantly increase flashback risk, with domestic appliances typically tolerating 20–25% H₂ and boilers up to ~40% under controlled conditions. Studies consistently show that burner wall temperature, preferential diffusion effects, and mixture composition play decisive roles, underscoring the need for improved predictive models and burner redesigns.
ii. Efficiency and emissions exhibit clear but non-linear trends.
Hydrogen addition generally produces hotter, faster, and more compact flames, improving thermal efficiency and reducing CO and CO₂ emissions. However, NOx formation tends to rise with increasing hydrogen fraction due to elevated flame temperatures, unless mitigated through excess air, staged combustion, or optimized burner geometry.
iii. Flame geometry is highly sensitive to hydrogen content.
Across boilers, cooktop burners, and oven systems, hydrogen consistently shortens and intensifies the flame, alters attachment behavior, and increases the risk of component overheating. Burner design, particularly orifice distribution, swirl intensity, and recirculation-zone length, plays a critical role in stabilizing H₂-enriched flames.
iv. Additional experimental studies confirm broader system-level impacts.
Research on heating performance, condensate production, oxygen enrichment, and swirl-stabilized combustion shows that hydrogen can enhance efficiency and reduce carbon-based emissions, but may require higher fuel flow rates, improved cooling, and stricter control of air distribution to maintain safe operation.
v. Practical blending limits for current residential appliances lie around 20–30% H₂.
Most studies converge on the conclusion that existing domestic systems can safely and efficiently operate with moderate hydrogen fractions, while higher concentrations demand targeted design modifications, updated certification procedures, and more advanced combustion-control strategies.
Overall, hydrogen-enriched natural gas represents a viable transitional pathway toward low-carbon residential energy systems, provided that safety constraints, especially flashback and NOx formation, are addressed through burner optimization, improved modeling, and updated regulatory frameworks. Continued research is essential to refine blending limits, develop hydrogen-ready appliance designs, and support the broader decarbonization of residential heating and cooking.
Author Contributions
Conceptualization, ; methodology, T.S., C.I., and T.P.; software, T.S. and C.I.; validation, T.S. and C.I.; formal analysis, T.S. and C.I.; investigation, T.S. and C.I.; resources, T.S. and C.I.; data curation, T.S. and C.I.; writing—original draft preparation, T.P. and C.I.; writing—review and editing, T.P.; visualization, T.S. and I.C.; supervision, T.P.; project administration, T.P.; funding acquisition, T.P. All authors have read and agreed to the published version of the manuscript.
Funding
Please add: “This research received no external funding” or “This research was funded by NAME OF FUNDER, grant number XXX” and “The APC was funded by XXX”. Check carefully that the details given are accurate and use the standard spelling of funding agency names at
https://search.crossref.org/funding. Any errors may affect your future funding.
Data Availability Statement
The original contributions presented in this study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author(s).
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
CFD – Computational Fluid Dynamics
CH₄ – Methane
CO – Carbon Monoxide
CO₂ – Carbon Dioxide
H₂ – Hydrogen
HENG – Hydrogen Enriched Natural Gas
NFPA – National Fire Protection Association
NG – Natural Gas
NOx – Nitrogen Oxides
OH* – Excited Hydroxyl Radical
RPMGS – Radiant Porous Media Gas Stove
SSPGS – Swirling Strip Port Gas Stove
TRPGS – Typical Round Port Gas Stove
WTC – Well to Combustion
WI – Wobbe Index (Fuel Interchangeability Index)
References
- United Nations. Paris Agreement, United Nations Framework Convention on Climate Change, 2015. Available online: https://unfccc.int/process-and-meetings/the-paris-agreement/the-paris-agreement.
- Wright, M.L.; Lewis, A.C. Emissions of NOx from blending of hydrogen and natural gas in space heating boilers. Elementa: Science of the Anthropocene 2022, 10(1), 00114. [Google Scholar] [CrossRef]
- Franco, A.; Rocca, M. Industrial Decarbonization through Blended Combustion of Natural Gas and Hydrogen. Hydrogen 2024, 5, 519–539. [Google Scholar] [CrossRef]
- British Petroleum report, Statistical Review of World Energy, 2021, 70th edition. Available online: https://www.energyinst.org/statistical-review.
- Quinn, A. K.; Bruce, N.; Puzzolo, E.; Dickinson, K.; Sturke, R.; Jack, D. W.; Mehta, S.; Shankar, A.; Sherr, K.; Rosenthal, J. P. An Analysis of Efforts to Scale up Clean Household Energy for Cooking around the World, Energy Sustain. Dev. 2018, 46, 1−10. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, W.; Yang, T.; Yu, Y.; Liu, C.; Li, B. Numerical and Experimental Investigation on Heat Transfer Enhancement by Adding Fins on the Pot in a Domestic Gas Stove. Energy 2022, 239, 122439. [Google Scholar] [CrossRef]
- Zhao, N.; Li, B.; Li, H.; Ahmad, R.; Peng, K.; Chen, D.; Yu, X.; Zhou, Y.; Dong, R.; Wang, H.; Ju, X.; Ibrahim, Z.A.M. Field-Based Measurements of Natural Gas Burning in Domestic Wall-Mounted Gas Stove and Estimates of Climate, Health and Economic Benefits in Rural Baoding and Langfang Regions of Northern China. Atmospheric Environment 2020, 229, 117454. [Google Scholar] [CrossRef]
- Popkova, E. G.; Sergi, B. S. Energy Efficiency in Leading Emerging and Developed Countries. Energy 2021, 221, 119730. [Google Scholar] [CrossRef]
- Acharya, R. H.; Sadath, A. C. Energy Poverty and Economic Development: Household-Level Evidence from India. Energy and Buildings 2019, 183, 785−791. [Google Scholar] [CrossRef]
- Liu, X.; Zhu, G.; Asim, T.; Mishra, R. Combustion characterization of hybrid methane-hydrogen gas in domestic swirl stoves. Fuel 2023, 333(2), 126413. [Google Scholar] [CrossRef]
- Elasu, J.; Ntayi, J.; Adaramola, M. S.; Buyinza, F. Drivers of Household Transition to Clean Energy Fuels: A Systematic Review of Evidence. Renew, Sustain. Energy Transit. 2023, 3, 100047. [Google Scholar] [CrossRef]
- Lebel, E. D.; Finnegan, C. J.; Ouyang, Z.; Jackson, R. B. Methane and NOx Emissions from Natural Gas Stoves, Cooktops, and Ovens in Residential Homes. Environ. Sci. Technol. 2022, 56(4), 2529−2539. [Google Scholar] [CrossRef]
- Dries, H. William D., The use of the natural-gas pipeline infrastructure for hydrogen transport in a changing market structure. International Journal of Hydrogen Energy 32, 1381–1386. [CrossRef]
- Field, R.A.; Derwent, R.G. Global warming consequences of replacing natural gas with hydrogen in the domestic energy sectors of future low-carbon economies in the United Kingdom and the United States of America. International Journal of Hydrogen Energy 2021, 46(58), 30190–30203. [Google Scholar] [CrossRef]
- Giovanni, D.L.; Abayomi, O. O.; Amit, K. Blending blue hydrogen with natural gas for direct consumption: Examining the effect of hydrogen concentration on transportation and well-to combustion greenhouse gas emissions. International Journal of Hydrogen Energy 2021, 46(36), 19202–19216. [Google Scholar] [CrossRef]
- Wenxue, G.; Yingjie, H.; Rongsong, Y.; Wentao, Y.; Mingchang, Y.; Qingwei, M.; Lin, Y.; Yan, W. Comprehensive Review on Thermal Performance Enhancement of Domestic Gas Stoves. ACS Omega 2023, 8(30), 26663–26684. [Google Scholar] [CrossRef]
- Zhao, Y.; Mcdonell, V.; Samuelsen, S. Influence of hydrogen addition to pipeline natural gas on the combustion performance of a cooktop burner. International Journal of Hydrogen Energy 2019, 44. [Google Scholar] [CrossRef]
- Sun, M.; Huang, X.; Hu, Y.; Lyu, S. Effects on the performance of domestic gas appliances operated on natural gas mixed with hydrogen. Energy 2021, 244, 122557. [Google Scholar] [CrossRef]
- Xinyi, Z.; Zhiguang, C.; Qin, Chaokui. Effect of hydrogen-blended natural gas on combustion stability and emission of water heater burner. Case Studies in Thermal Engineering 2022, 37, 102246. [Google Scholar] [CrossRef]
- Li, S.; Li, X.; Jin, H.; Liu, Y.; Wu, Y. A Numerical Simulation Study on the Combustion of Natural Gas Mixed with Hydrogen in a Partially Premixed Gas Water Heater. Energies 2024, 17, 4069. [Google Scholar] [CrossRef]
- Mayrhofer, M.; Koller, M.; Seemann, P.; Prieler, R.; Hochenauer, C. Assessment of natural gas / hydrogen blends as an alternative fuel for industrial heat treatment furnaces. International Journal of Hydrogen Energy 2021, 46, 21672–21686. [Google Scholar] [CrossRef]
- Haiyan, M.; Lin, L.; Zuohua, H. Flammability limits of hydrogen-enriched natural gas. International Journal of Hydrogen Energy 2011, 36(11)(2011), 6937–6947. [Google Scholar] [CrossRef]
- Aniello, A.; Poinsot, T.; Selle, L.; Schuller, T. Hydrogen substitution of natural-gas in premixed burners and implications for blow-off and flashback limits. International Journal of Hydrogen Energy 2022, 47(77), 33067–33081. [Google Scholar] [CrossRef]
- Tahsin, B.K.; Emre, B.; Dilay, G.; Mehmet, K.; Barış, Y.; Christophe, A.; İskender, G. Numerical investigations on flashback dynamics of premixed methane-hydrogen-air laminar flames. International Journal of Hydrogen Energy 2022, 47(59), 25022–25033. [Google Scholar] [CrossRef]
- Lewis, B.; von Elbe, G. Stability and structure of burner flames. J Chem Phys 1943, 11(2), 75–97. [Google Scholar] [CrossRef]
- Putnam, A.A.; Jensen, R.A. Application of dimensionless numbers to flash-back and other combustion phenomena. Symposium on Combustion and Flame, and Explosion Phenomena 1948, 3(1), 89–98. [Google Scholar] [CrossRef]
- Hoferichter, V.; Hirsch, C.; Sattelmayer, T. Prediction of boundary layer flashback limits of laminar premixed jet flames; power for land, sea, and air: Turbo expo, 2018; p. 51050. [Google Scholar] [CrossRef]
- de Vries, H.; Mokhov, A.V.; Levinsky, H.B. The impact of natural gas/hydrogen mixtures on the performance of end-use equipment: interchangeability analysis for domestic appliances. Appl Energy 2017, 208, 1007, 19. [Google Scholar] [CrossRef]
- Enrique, F.M.; Andrea, A.; Thierry, S.; Laurent, S. Predicting flashback limits in H2 enriched CH4/air and C3H8/air laminar flames. Combustion and Flame 2023, 258(2), 113055. [Google Scholar] [CrossRef]
- Filippo, F.; Rachele, L.; Leonardo, T.; Chiara, G. Flashback of H2-enriched premixed flames in perforated burners: Numerical prediction of critical velocity. International Journal of Hydrogen Energy 2023, 48(81), 31790–31801. [Google Scholar] [CrossRef]
- Filippo, F.; Rachele, L.; Alessandro, M.; Maria, V.S.; Chiara, G. Flashback propensity due to hydrogen blending in natural gas: Sensitivity to operating and geometrical parameters. Fuel 2024, 362, 130838. [Google Scholar] [CrossRef]
- Cuoci, A.; Frassoldati, A.; Cozzi, F. Numerical predictions of flashback limits of H2-enriched methane/air premixed laminar flames. Proceedings of the Combustion Institute 2024, 40, 105696. [Google Scholar] [CrossRef]
- Leicher, J.; Schaffert, J.; Cigarida, H.; Tali, E.; Burmeister, F.; Giese, A.; Albus, R.; Görner, K.; Carpentier, S.; Milin, P. The Impact of Hydrogen Admixture into Natural Gas on Residential and Commercial Gas Appliances. Energies 2022, 15, 777. [Google Scholar] [CrossRef]
- Xie, Y.; Qin, C.; Chen, Z.; Duan, P.; Guo, S. The Impact of Hydrogen Addition to Natural Gas on Flame Stability. Int. J. Hydrog. Energy 2022, 47(84), 35851−35863. [Google Scholar] [CrossRef]
- Zhao, Y.; Mcdonell, V.; Samuelsen, S. Experimental assessment of the combustion performance of an oven burner operated on pipeline natural gas mixed with hydrogen. Int J Hydrogen Energy 2019, 44, 26049–62. [Google Scholar] [CrossRef]
- Jones, D.; Al-Masry, W.; Dunnill, C. Hydrogen-enriched natural gas as a domestic fuel: An analysis based on flash-back and blow-off limits for domestic natural gas appliances within the UK. Sustainable Energy, Fuels 2017, 2. [Google Scholar] [CrossRef]
- Makaryan, I. A.; Sedov, I. V.; Salgansky, E. A.; Arutyunov, A. V.; Arutyunov, V. S. A Comprehensive Review on the Prospects of Using Hydrogen-Methane Blends: Challenges and Opportunities. Energies 2022, 15(6), 2265. [Google Scholar] [CrossRef]
- Yilmaz, H.; Schröder, L.; Hillenbrand, T.; Brüggemann, D. Effects of Hydrogen Addition on Combustion and Flame Propagation Characteristics of Laser Ignited Methane/Air Mixtures. Int. J. Hydrog Energy 2023, 48(45), 17324−17338. [Google Scholar] [CrossRef]
- Yang, X.; Wang, T.; Zhang, Y.; Zhang, H.; Wu, Y.; Zhang, J. Hydrogen Effect on Flame Extinction of Hydrogen-Enriched Methane/ Air Premixed Flames: An Assessment from the Combustion Safety Point of View. Energy 2022, 239, 122248. [Google Scholar] [CrossRef]
- Fei, R.; Huaqiang, C.; Longkai, X.; Weiwei, H.; Mingyan, G. Effect of hydrogen addition on the laminar premixed combustion characteristics the main components of natural gas. Journal of the Energy Institute 2019, 92(4), 1178–1190. [Google Scholar] [CrossRef]
- Nosek, R.; Zvada, B.; Ďurčanský, P.; et al. Numerical Analysis of Hydrogen-Enriched Natural Gas on Combustion and Emission Characteristics. Arab J Sci Eng 2025, 50, 13745–13754. [Google Scholar] [CrossRef]
- Anna, H.; Ali, N.; Hartmut, K.; Sven, E. Experimental and numerical assessment of the effects of hydrogen admixtures on premixed methane-oxygen flames. Fuel 2023, 352, 128964. [Google Scholar] [CrossRef]
- Wojtowicz, Robert. An analysis of the effects of hydrogen addition to natural gas on the work of gas appliances. Nafta-Gaz 2019, 75, 465–473. [Google Scholar] [CrossRef]
- Xin, Y.; Wang, K.; Zhang, Y.; Zeng, F.; He, X.; Takyi, S.A.; Tontiwachwuthikul, P. Numerical Simulation of Combustion of Natural Gas Mixed with Hydrogen in Gas Boilers. Energies 2021, 14, 6883. [Google Scholar] [CrossRef]
- Liu, X.Y.; Huang, G.L.; Zhen, H.S.; Wei, Z.L. Experimental testing and geometric optimization of a domestic cooker burning 80%Natural-Gas+20%Hydrogen. International Journal of Hydrogen Energy 2024, 85, 281–286. [Google Scholar] [CrossRef]
- Zhicong, F.; Shuhao, Z.; Xiaomei, H.; Yelong, H.; Qian, X. Performance of three typical domestic gas stoves operated with methane-hydrogen mixture. Case Studies in Thermal Engineering 2023, 41, 102631. [Google Scholar] [CrossRef]
- Ahmet, T. Ö.; Dinçer, A.; Ugur, A. Investigation of the emission values of butane, methane and methane-hydrogen mixtures used in household stove burners. International Journal of Hydrogen Energy 2025, 141, 88–98. [Google Scholar] [CrossRef]
- Yiyu, C.; Jie, N.; Wenjie, L.; Liwen, L.; Taiming, H.; Yingkai, S.; Zhongmin, W.; Bo, Y. Experimental analysis and modeling on the blending limit of domestic burner with porous media for hydrogen enriched natural gas. International Journal of Hydrogen Energy 2024, 88, 1321–1331. [Google Scholar] [CrossRef]
-
Bundesministerium für Umwelt, Naturschutz und Reaktorsicherheit, Technische Anleitung zur Reinhaltung der Luft (TA Luft); 2022.
- Rachele, L.; Cristiana, B.; Marco, F.; Leonardo, T.; Chiara, G. Impact of H2-enriched natural gas on pollutant emissions from domestic condensing boilers: numerical simulations of the combustion chamber. International Journal of Hydrogen Energy 2023, 48(51), 19686–19699. [Google Scholar] [CrossRef]
- Bălănescu, D.T.; Homutescu, V.M. Effects of hydrogen-enriched methane combustion on latent heat recovery potential and environmental impact of condensing boilers. Applied Thermal Engineering 2021, 197, 117411. [Google Scholar] [CrossRef]
- Fabio, S.; Anna, S.; Alberto, B. Modelling and analyzing the impact of hydrogen enriched natural gas on domestic gas boilers in a decarbonization perspective. Carbon Resources Conversion 2020, 3, 122–129. [Google Scholar] [CrossRef]
- Wahiba, Y.; Michela, L. Performance analysis of domestic boilers fuelled with hydrogen-enriched natural gas blends and pure hydrogen. Energy 2025, 322, 135536. [Google Scholar] [CrossRef]
- Rimar, M.; Kizek, J.; Varga, A.; Fedák, M.; Jablonský, G. The influence of hydrogen concentration in natural gas on heat flows in a thermal aggregate. MM Science Journal 2022. [Google Scholar] [CrossRef]
- Fatih, S.; Merve, O.; Nader, J.; Ibrahim, D. Effect of burner head geometry on flame dispersion in gas stoves with hydrogen and natural gas blends. Process Safety and Environmental Protection 2024, 183, 1135–1151. [Google Scholar] [CrossRef]
- Restrepo, A.; Viana, M.; Colorado, A.; Amell, A. Experimental investigation of hydrogen enriched natural gas diffusion reactions using preheated air in a hot coflow burner. International Journal of Hydrogen Energy 2021 48. [CrossRef]
- Fatih, S.; Merve, O.; Nader, J.; Ibrahim, D. Experimental investigation for combustion performance of hydrogen and natural gas fuel blends. International Journal of Hydrogen Energy 2023, 48(88), 34476–34485. [Google Scholar] [CrossRef]
- Sun, C.; Wang, T.; Wang, P.; Zhang, Y.; Cui, C.; Lu, Y.; Liu, W.; Zhang, Y.; Zhang, Y. Numerical Simulation and Field Experimental Study of Combustion Characteristics of Hydrogen-Enriched Natural Gas. Processes 2024, 12, 1325. [Google Scholar] [CrossRef]
- Schwarz, Stefan; Daurer, Georg; Gaber, Christian; Demuth, Martin; Hochenauer, Christoph. Experimental investigation of hydrogen enriched natural gas combustion with a focus on nitrogen oxide formation on a semi-industrial scale. International Journal of Hydrogen Energy 2024, 63, 173–183. [Google Scholar] [CrossRef]
- Onur, S.; Semiha, Ö. Modeling of hydrogen blending natural gas combustion characteristics and emission analyses in industrial burners. International Journal of Hydrogen Energy 2025, 144, 782–797. [Google Scholar] [CrossRef]
- Lan, Y.; Wang, Z.; Xu, J.; Yi, W. The Impact of Hydrogen on Flame Characteristics and Pollutant Emissions in Natural Gas Industrial Combustion Systems. Energies 2024, 17, 4959. [Google Scholar] [CrossRef]
- Christian, K.; Georg, D.; Stefan, S.; Martin, D.; Christian, G.; Christoph, H. CFD study of hydrogen combustion effects on the heat-up characteristics of steel samples using a low-swirl burner: A comparative analysis with methane. Applied Thermal Engineering 2025, 261, 125105. [Google Scholar] [CrossRef]
- Fabian, W.; Georg, A.; Daniel, D.; Christoph, H. The impact of hydrogen on forced-draft gas burners: A numerical investigation of nitrogen oxide emissions. International Journal of Hydrogen Energy 2025, 143, 582–595. [Google Scholar] [CrossRef]
- Zouhaier, R.; Mohamed, A. M.; Jean-Charles, S.; Sassi, B. N. Experimental study of natural gas flame enriched by hydrogen and oxygen in a coaxial burner. Applied Thermal Engineering 2016, 108, 287–295. [Google Scholar] [CrossRef]
- Pinto, B.M.; Pacheco, G.P.; Mendes, M.A.A.; Coelho, P.J. Numerical Simulation of Natural Gas/Hydrogen Combustion in a Novel Laboratory Combustor. Appl. Sci. 2025, 15, 7123. [Google Scholar] [CrossRef]
- Wei, D.; Shuiqing, Z.; Huichong, Q.; Jianguo, Z.; Yutao, F. Experiment and numerical study of the combustion behavior of hydrogen-blended natural gas in swirl burners. Case Studies in Thermal Engineering 2022, 39, 102468. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).