Submitted:
04 January 2026
Posted:
05 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental
2.1. Catalyst Preparation
2.2. Catalyst Characterization and Activity Measurements
3. Results and Discussion
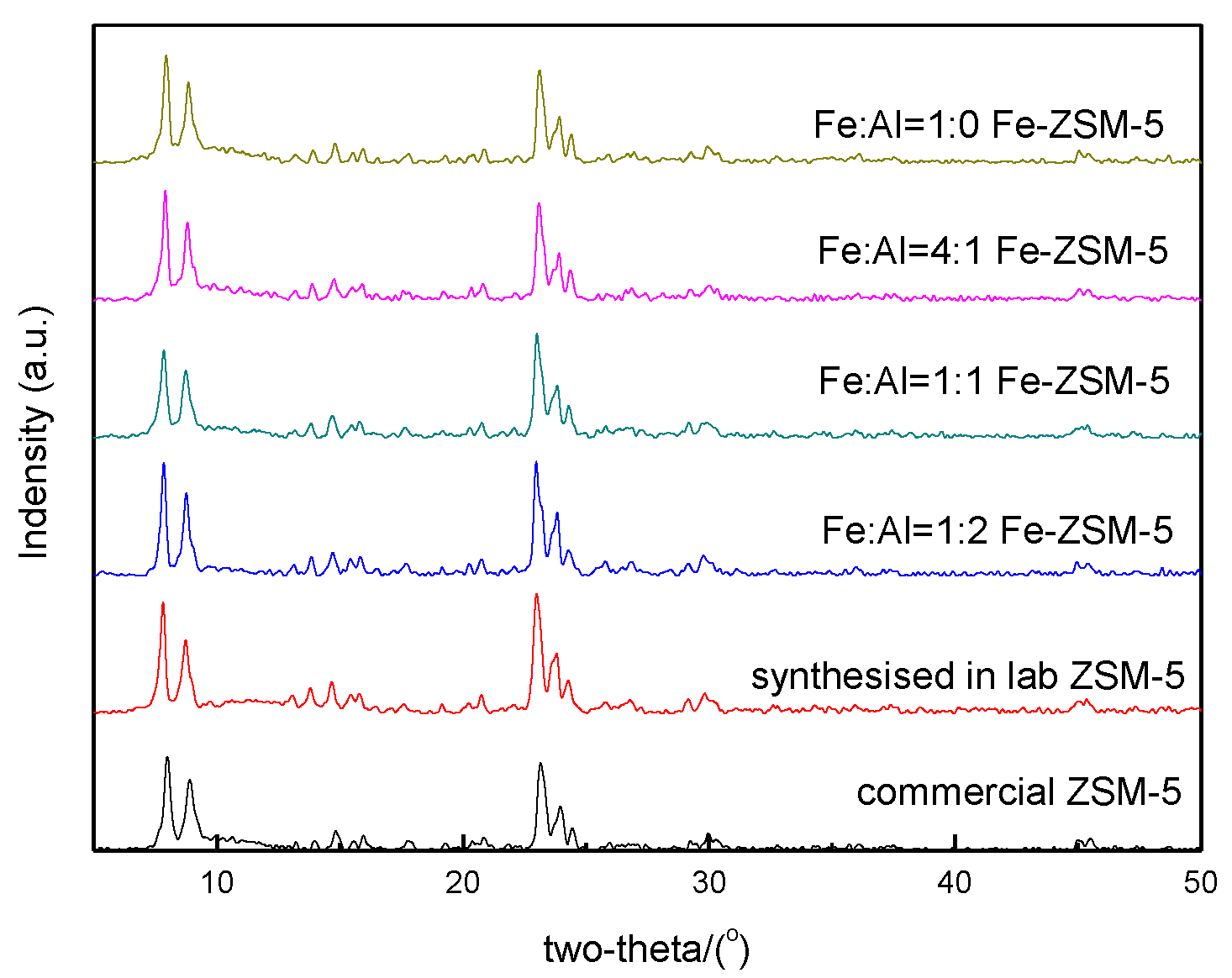
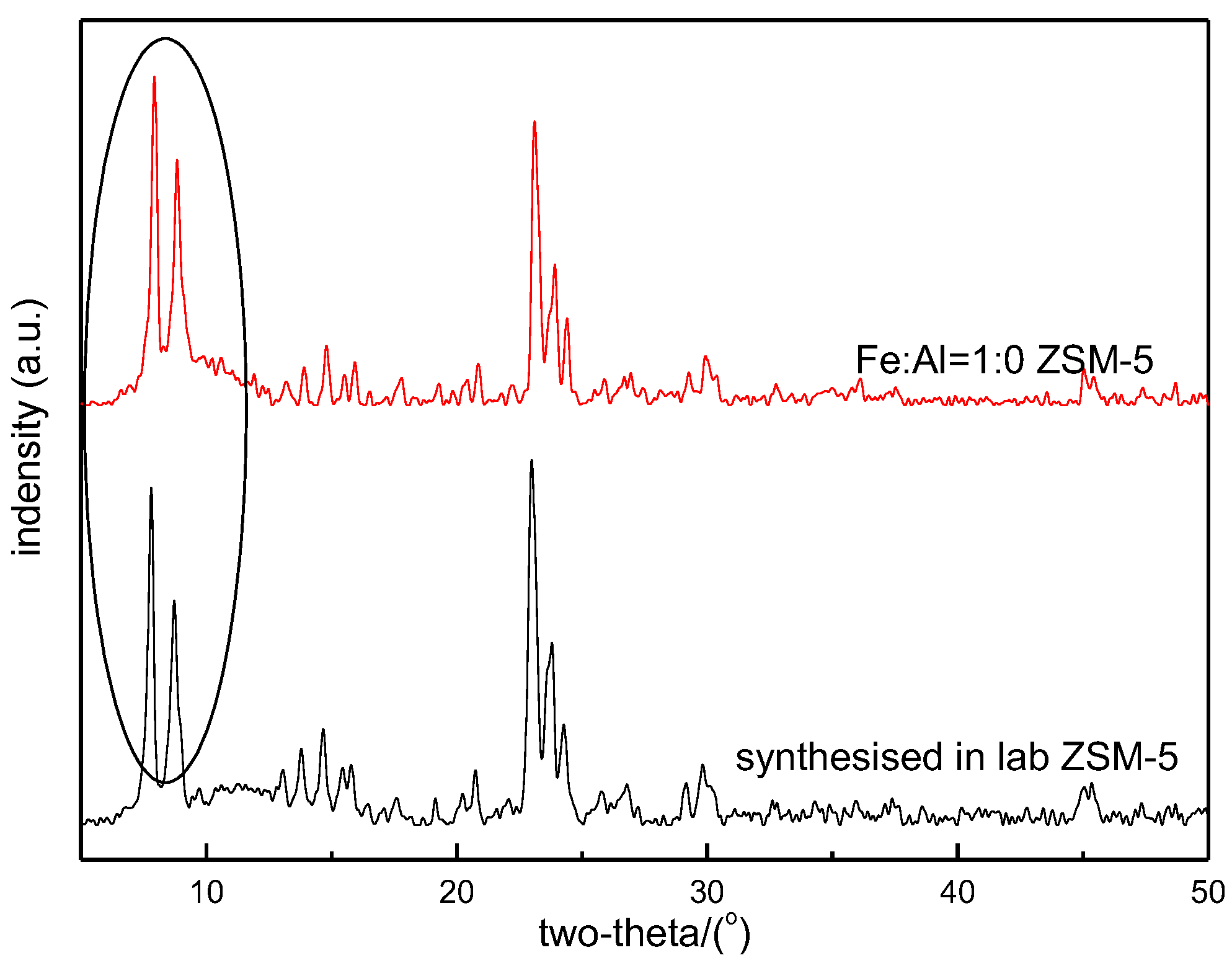
3.1. X-Ray Diffraction and Specific Surface Area
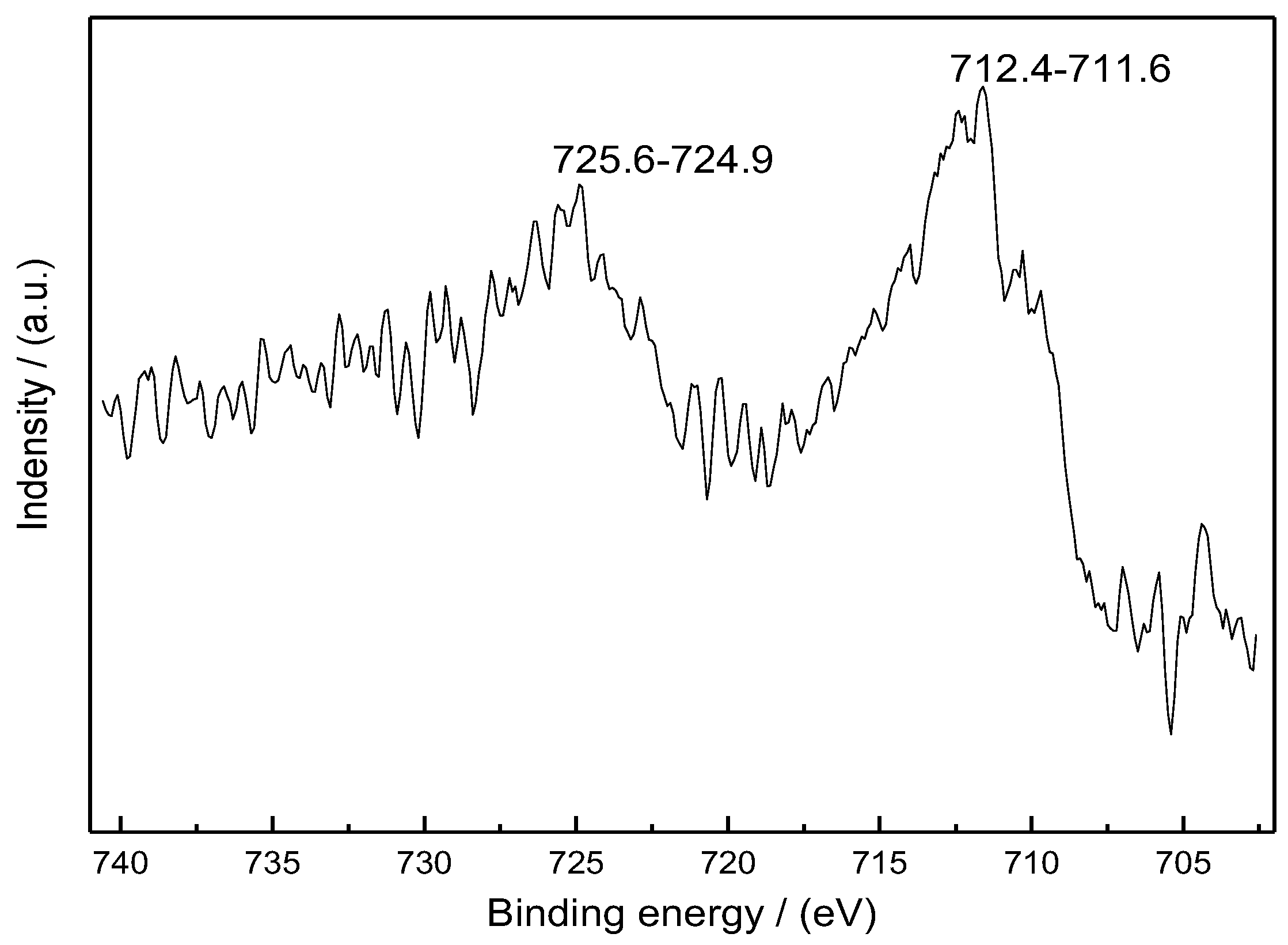
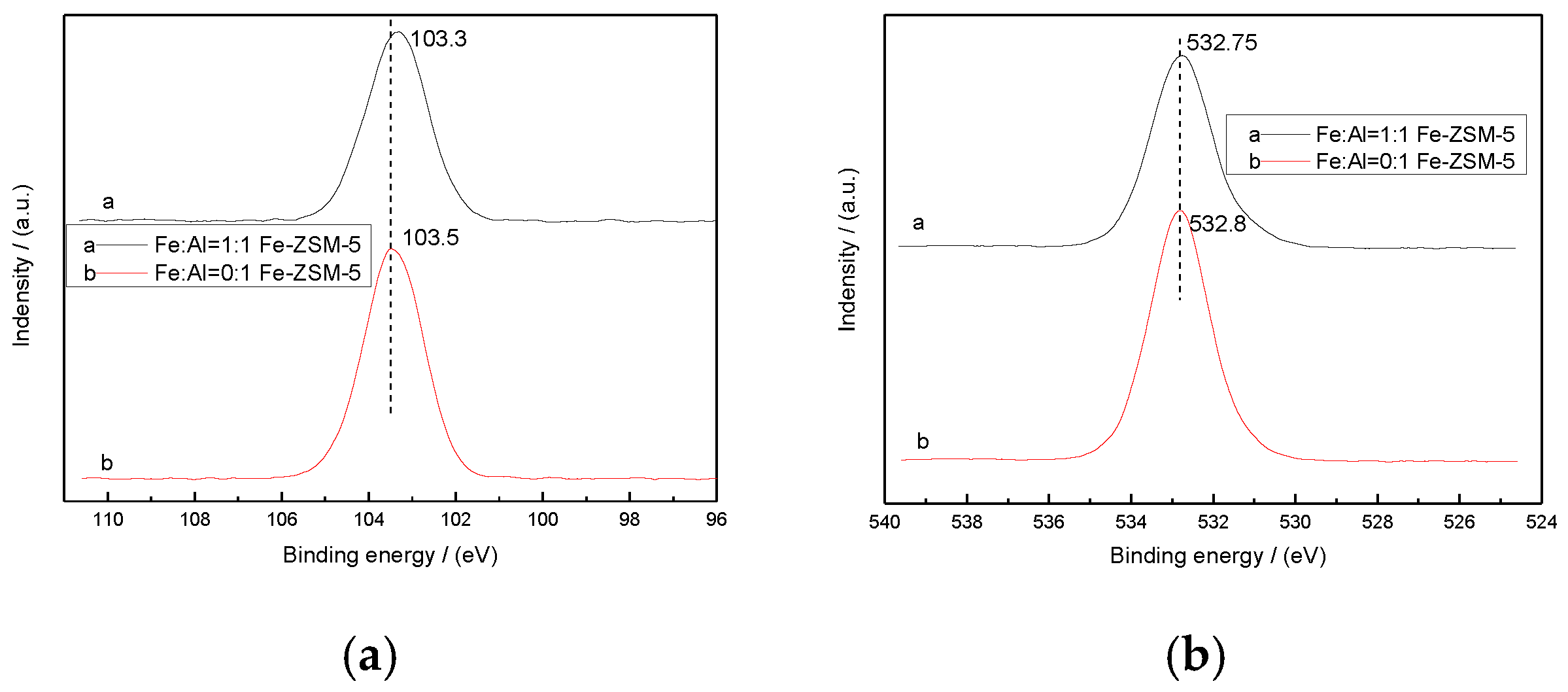
3.2. Scanning Electron Microscope and X-Ray Photoelectron Spectroscopy
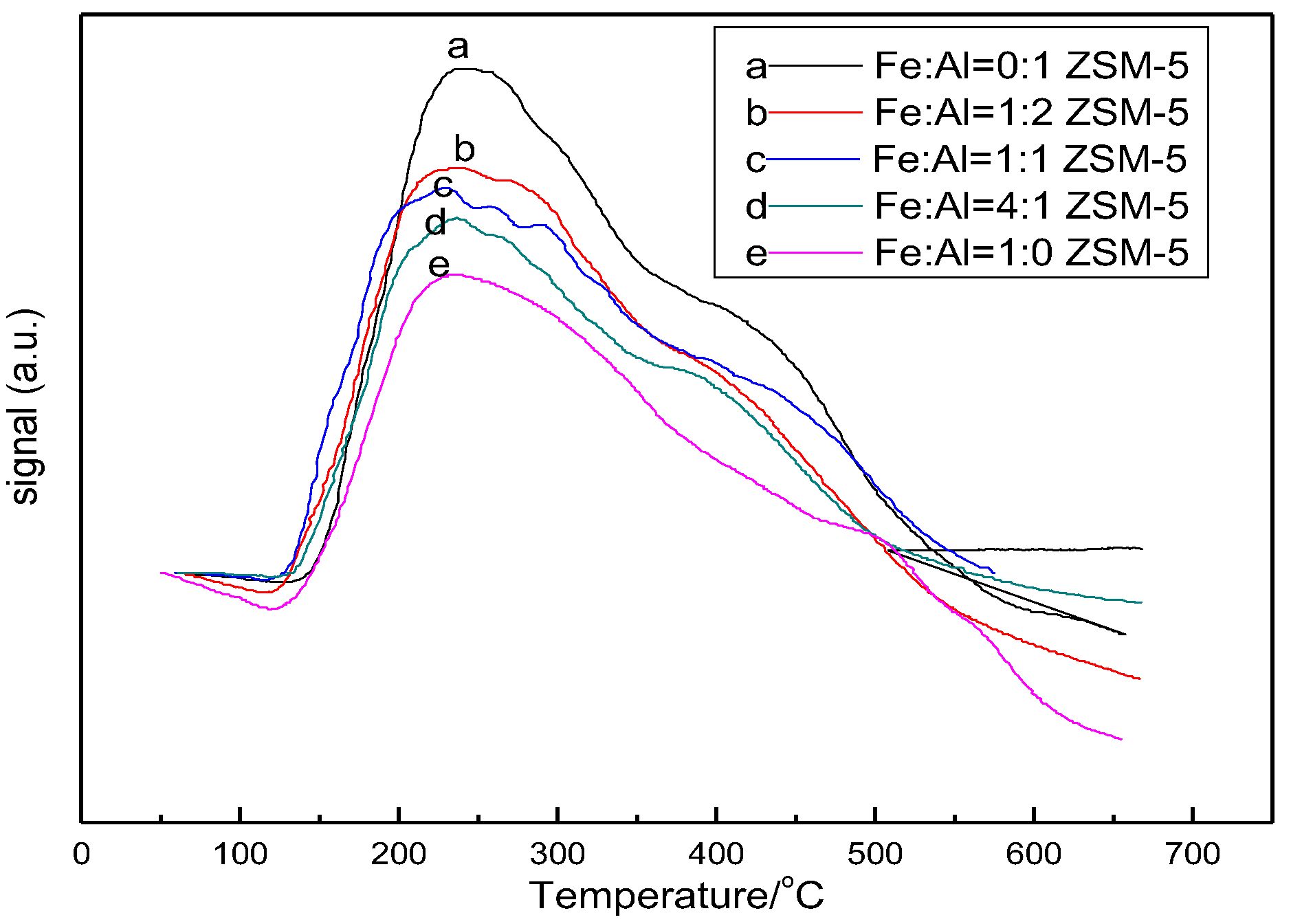
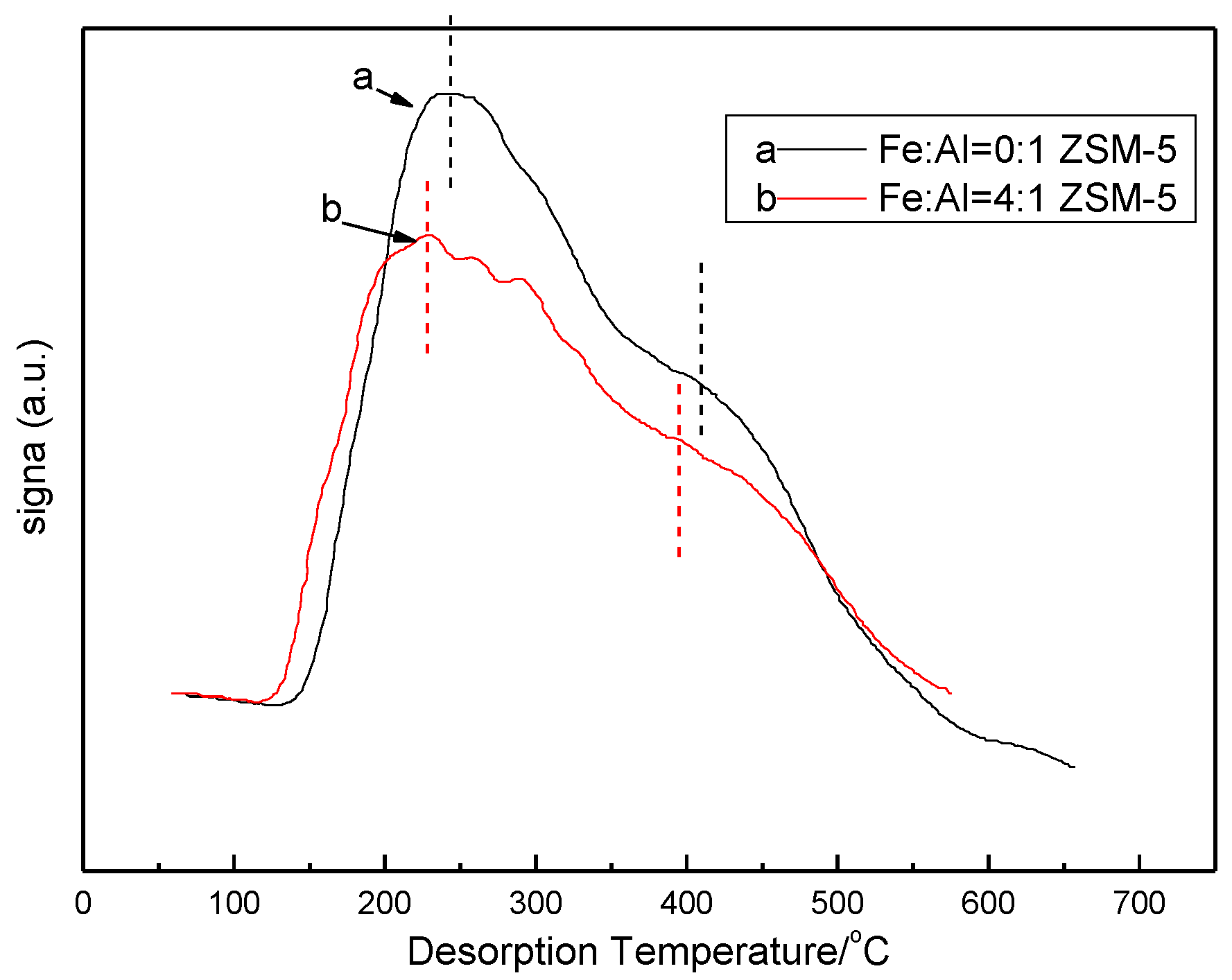
3.3. Temperature Programmed Desorption of Ammonia (NH3-TPD)
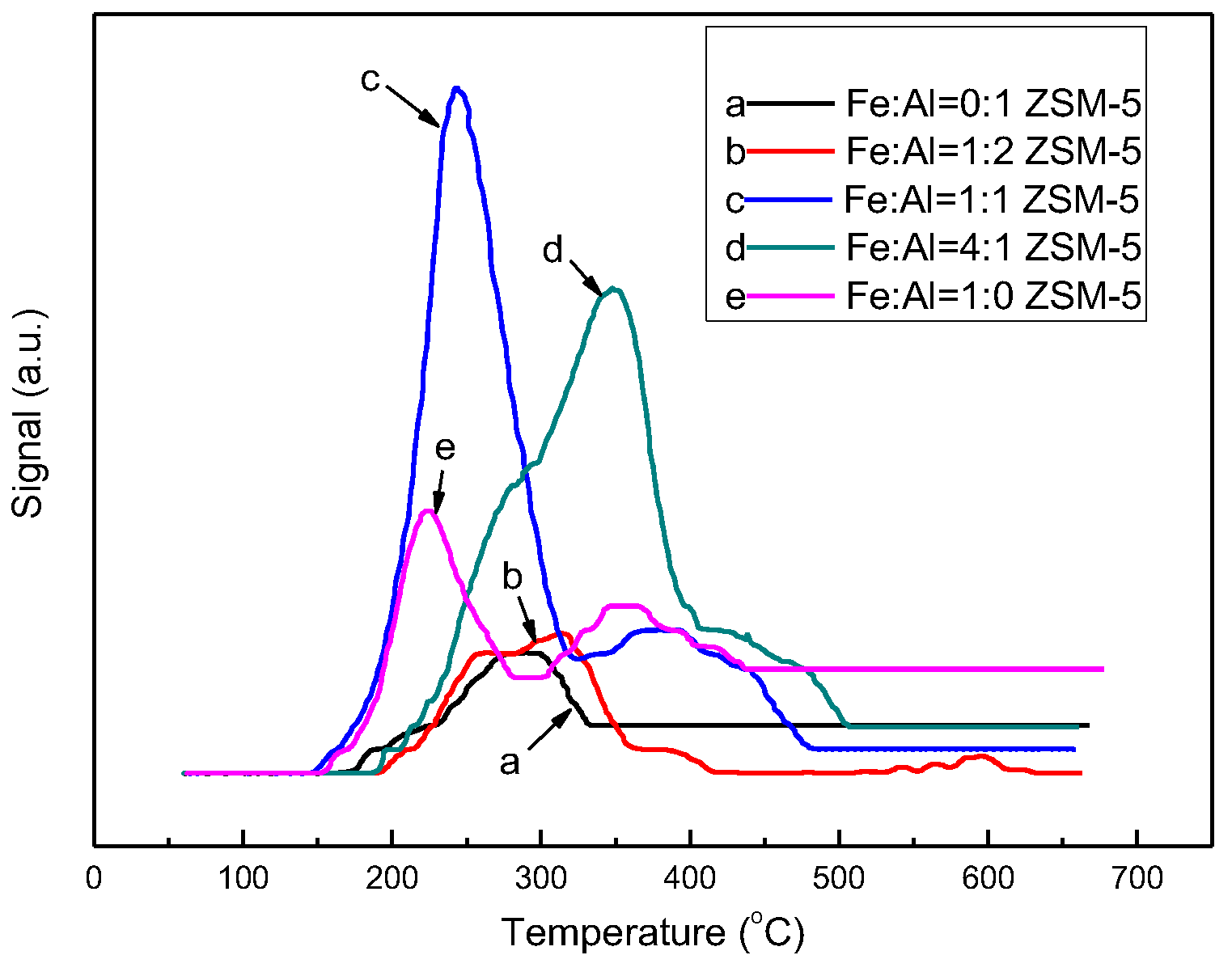
3.4. Temperature Programmed Desorption of Nitric Oxides (NO-TPD)
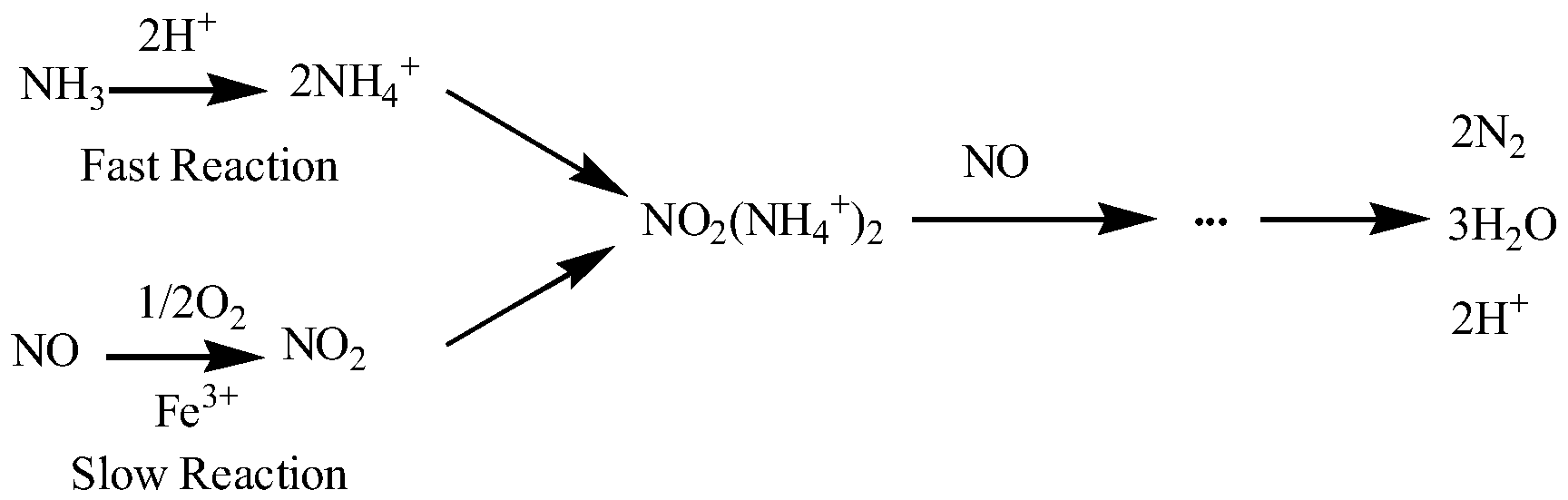
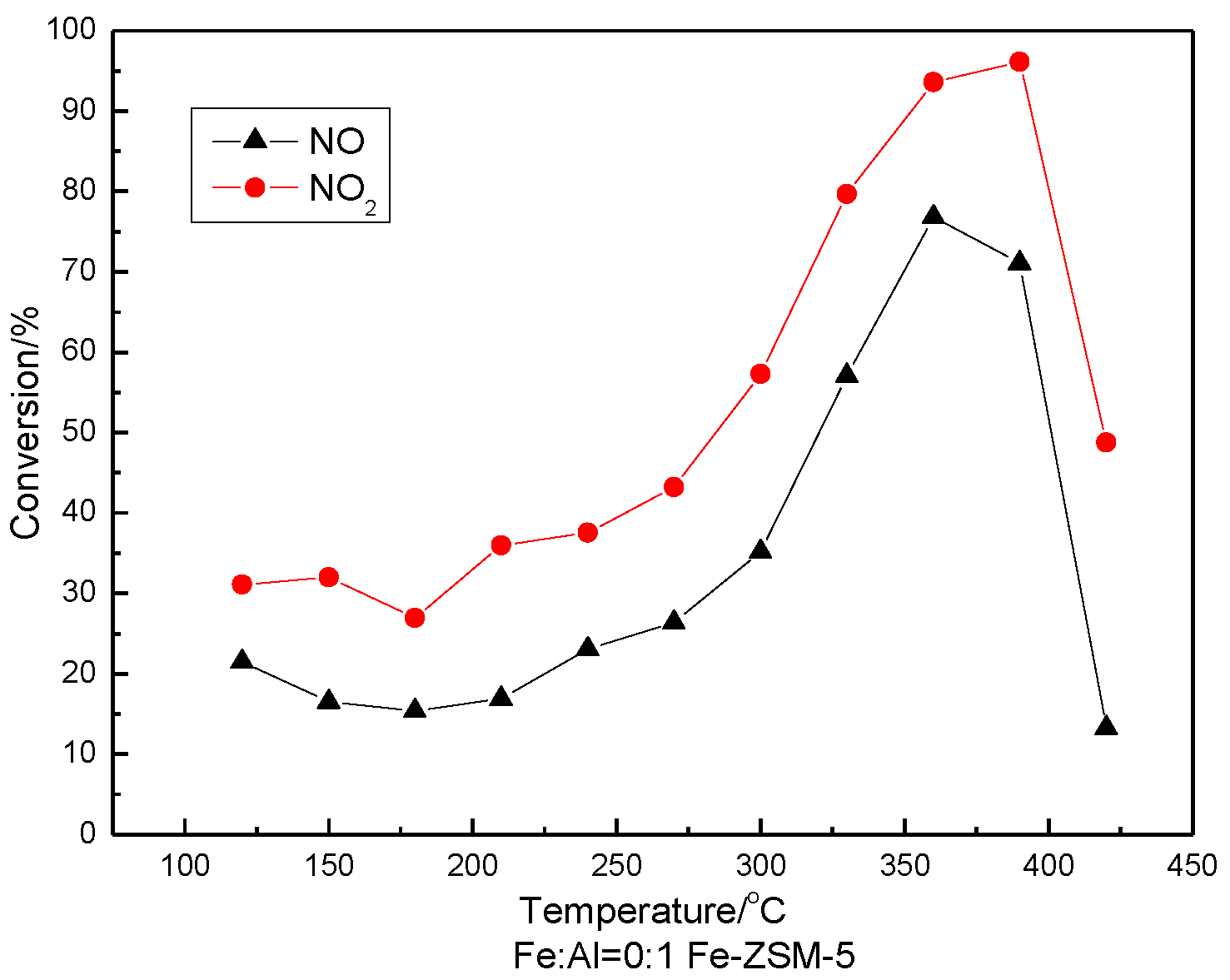
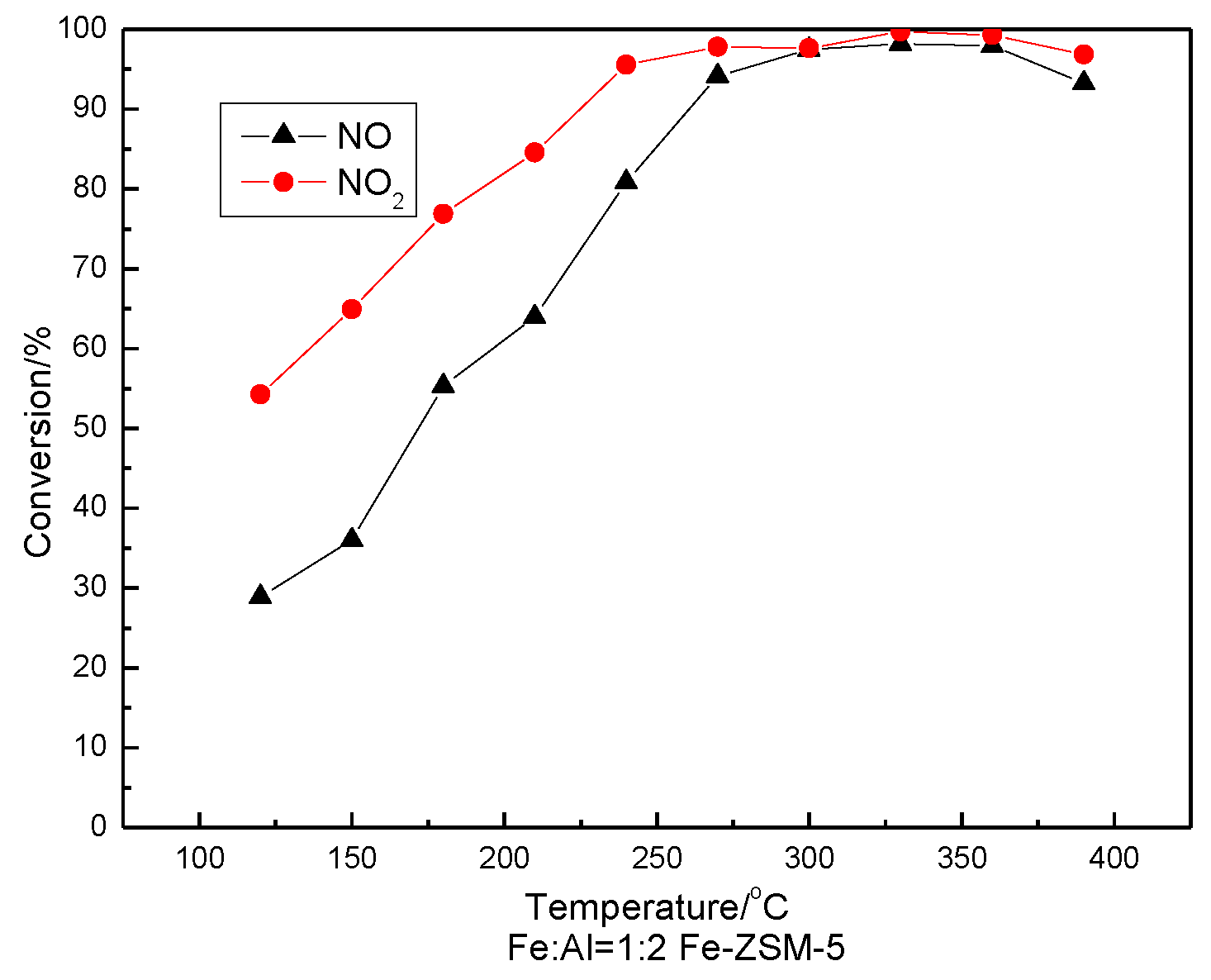
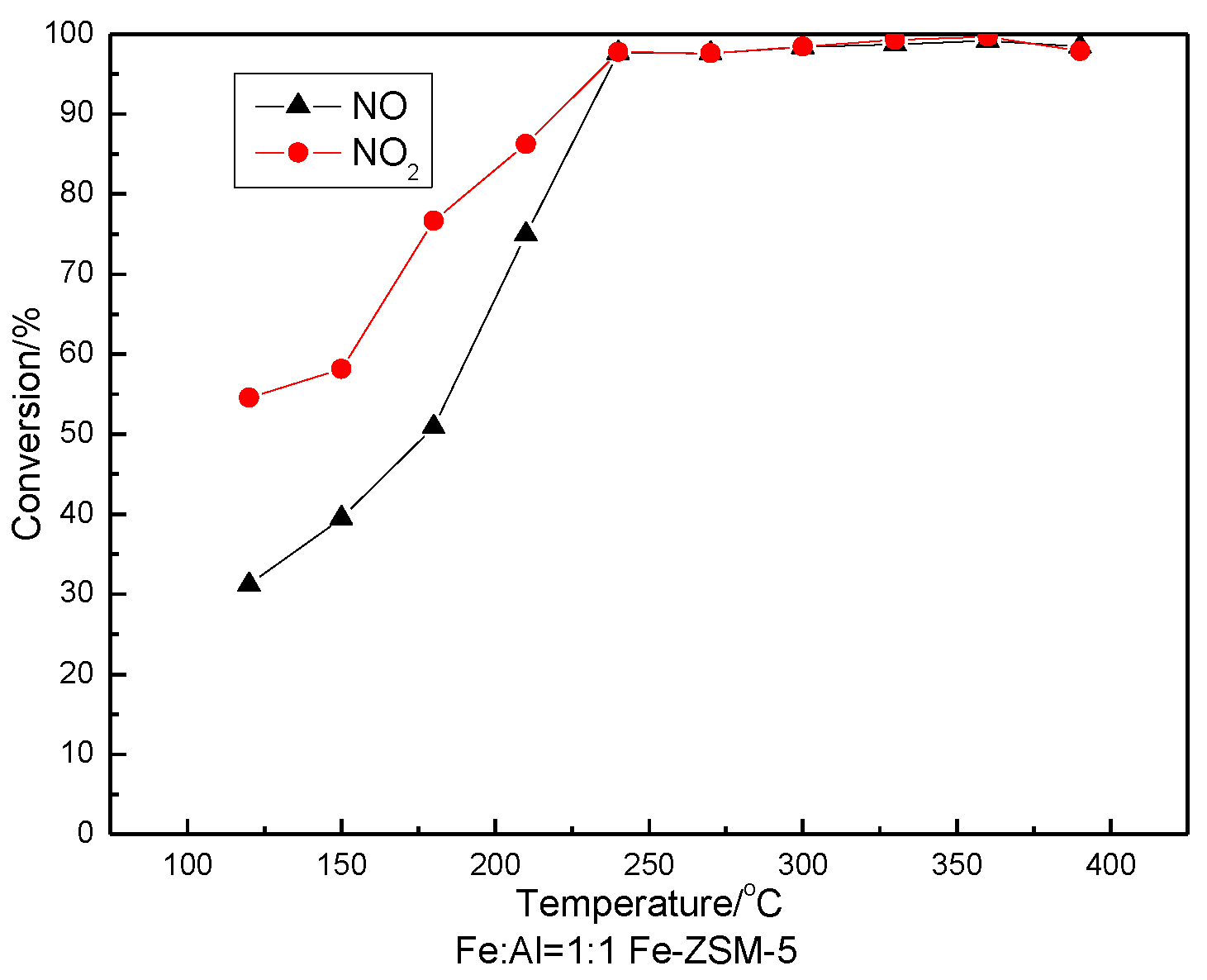
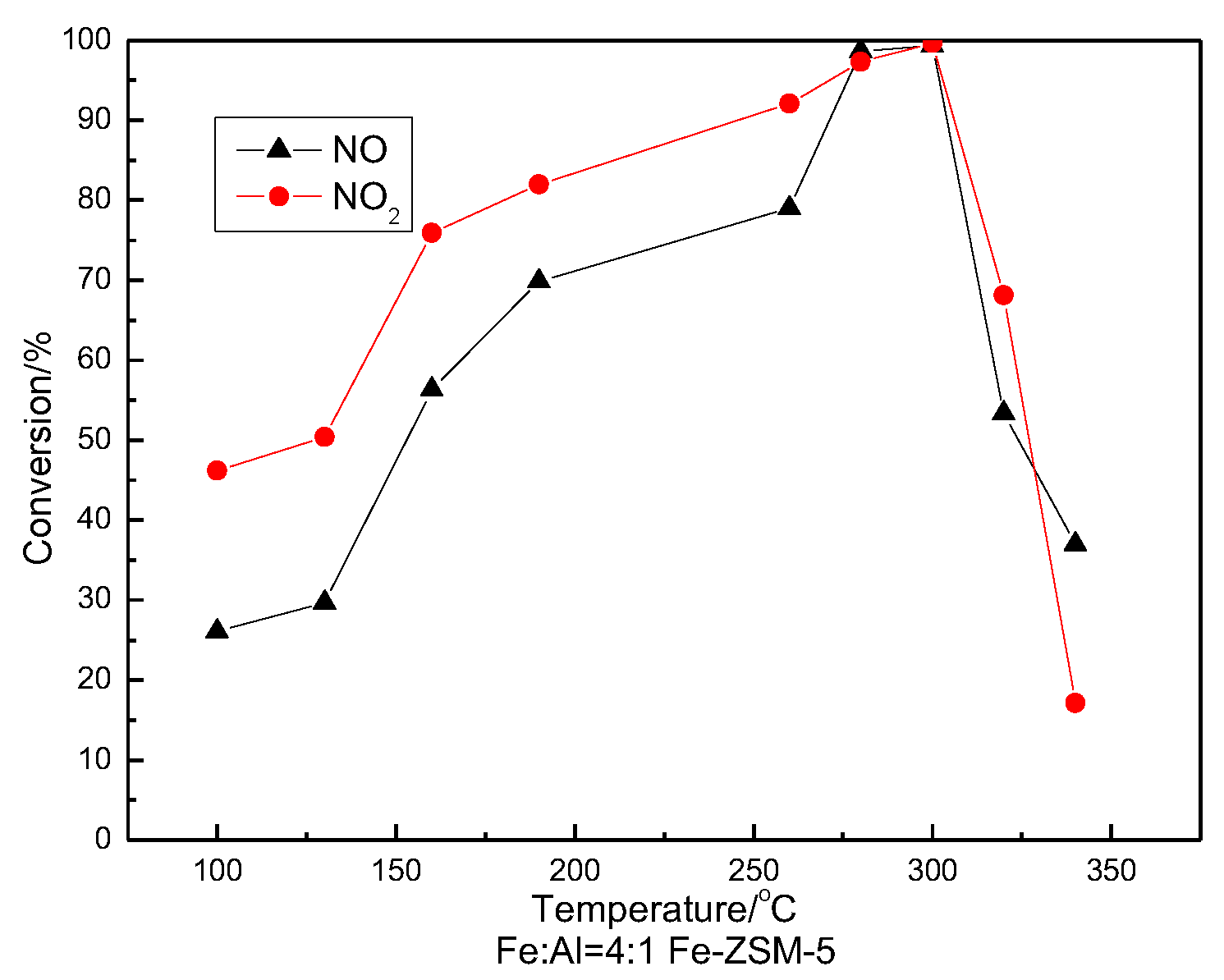
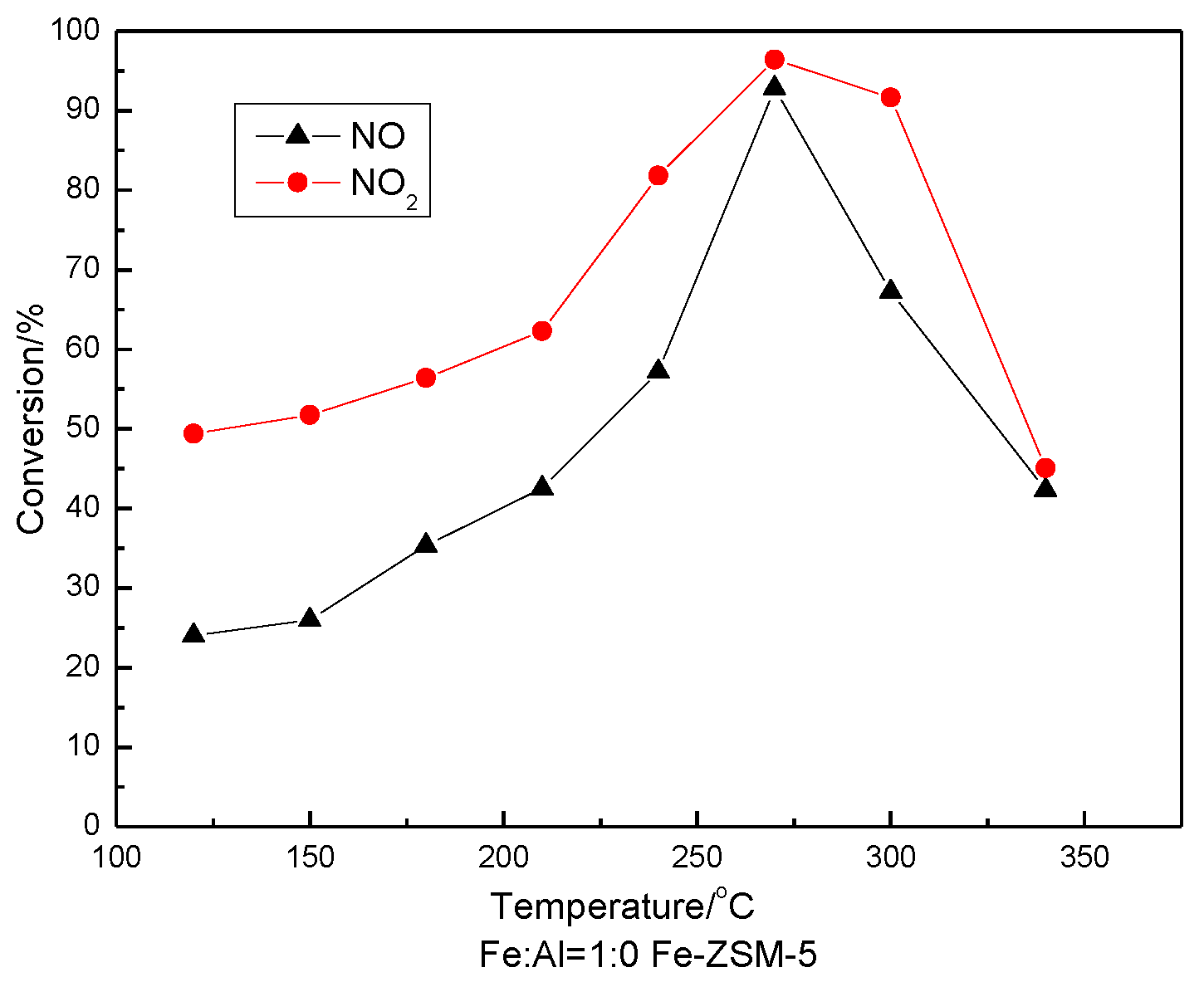
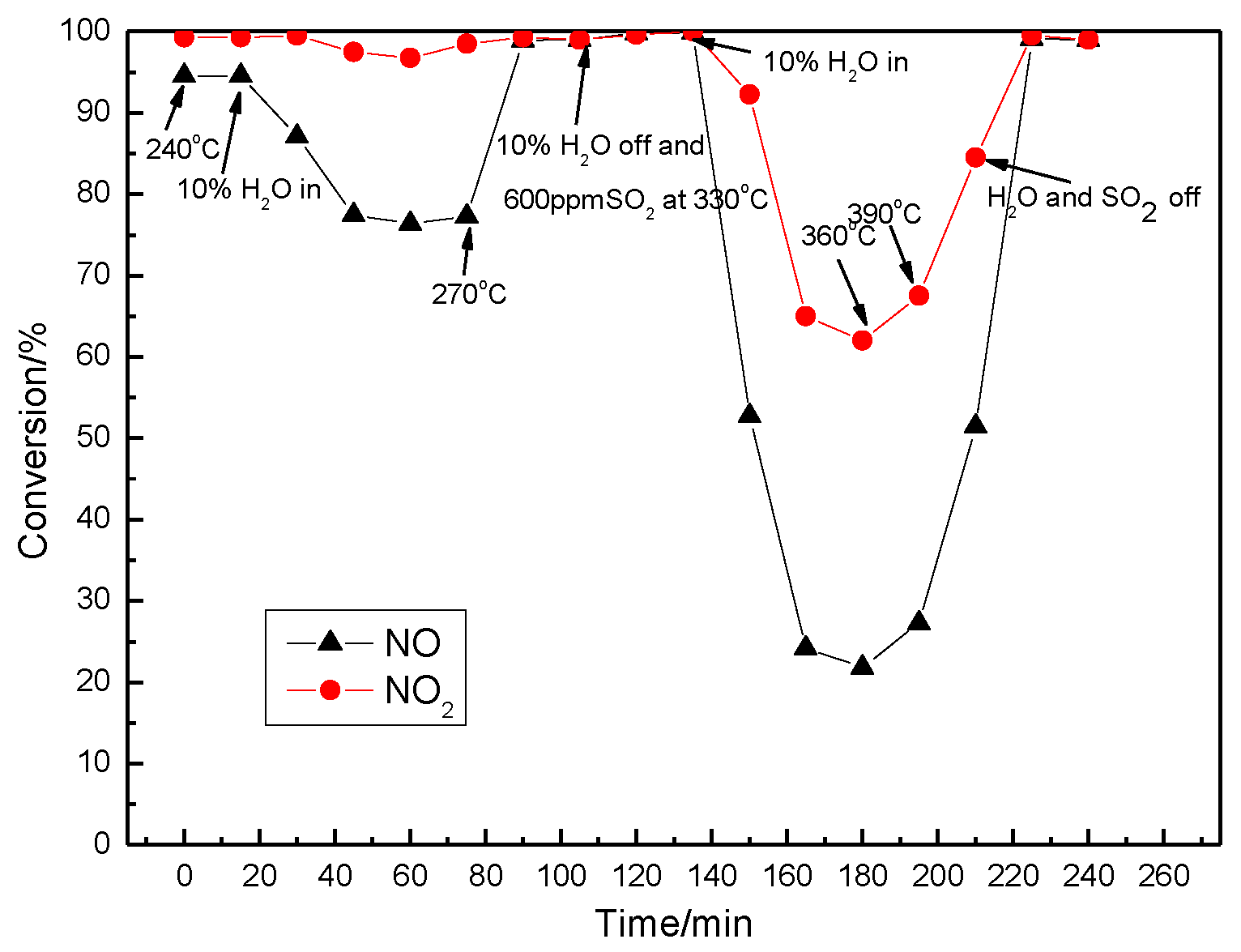
3.5. SCR Activity Testing
4. Conclusions
Funding
References
- Shi, X; Wang, Y; Shan, Y; et al. Investigation of the common intermediates over Fe-ZSM-5 in NH3-SCR reaction at low temperature by in situ DRIFTS[J]. Journal of Environmental Sciences 2020, 94(08), 32–39. [Google Scholar] [CrossRef] [PubMed]
- Sun, K Q; Xia, H A; Hensen, E; van, Santen R; Li, C. Chemistry of N2O decomposition on active sites with different nature: Effect of high-temperature treatment of Fe/ZSM-5 [J]. Journal of Catalysis 2006, 238, 186~195. [Google Scholar] [CrossRef]
- Pérez-Ramírez, J. Active iron sites associated with the reaction mechanism of N2O conversions over steam-actived FeMFI zeolites [J]. Journal of Catalysis 2004, 227, 512~522. [Google Scholar] [CrossRef]
- Schwidder, M; KumarM, S; Klementiev, K; et al. Selective reduction of NO with Fe-ZSM-5 catalysts of low Fe content I. Relations between active site structure and catalytic performance [J]. Journal of Catalysis 2005, 231, 314~330. [Google Scholar] [CrossRef]
- Qi, G; Yang, R T. Ultra-active Fe/ZSM-5 catalyst for selective catalytic reduction of nitric oxide with ammonia [J]. Applied Catalysis. B Environmental 2005, 60, 13~22. [Google Scholar] [CrossRef]
- Yu-bo, ZHANG; Pan, WANG; Dan, YU. Evolution mechanism of active sites for NH3-selective catalytic reduction of NOₓ over Ce/Cu-doped Fe-ZSM-5 catalysts[J]. Journal of Central South University 2022, 29(7), 2239–2252. [Google Scholar]
- Long, R Q; Yang, R T. Selective catalytic oxidation (SCO) of ammonia to nitrogen over Fe-exchanged zeolites [J]. Journal of Catalysis 2001, 201, 145~152. [Google Scholar] [CrossRef]
- Krishna, K; Seijer, G B F; Bleek, C M; van den; et al. Selective catalytic reduction of NO with NH3 over Fe-ZSM-5 catalysts prepared by sublimation of FeCl3 at different temperatures [J]. Catalysis Letter 2003, 86, 121~132. [Google Scholar] [CrossRef]
- Long, R Q; Yang, R T. Temperature-programmed desorption/surface reaction (TPD/TPSR) study of Fe-exchanged ZSM-5 for selective catalytic reduction of nitric oxide by ammonia [J]. Journal of catalysis 1999, 198, 20~28. [Google Scholar] [CrossRef]
- Li, Z; Shen, L; Huang, W; et al. Kinetics of selective catalytic reduction of NO by NH3 on Fe-Mo/ZSM-5 catalyst [J]. Journal of Environmental Science 2007, 19, 1516~1519. [Google Scholar] [CrossRef]
- Zhou, H; Liu, J; Zhang, Y; et al. Hydrothermal aging alleviates hydrocarbon poisoning effects on high-silica Cu-SSZ-16catalysts for NH3-SCR[J]. Journal of Environmental Sciences 2025, 158(12), 13–23. [Google Scholar] [CrossRef] [PubMed]
- Delahay, G; Valade, D; Guzmán-Vargas, A; et al. Selective catalytic reduction of nitric oxide with ammonia on Fe-ZSM-5 catalysts prepared by different methods [J]. Applied Catalysis B: Environmental 2005, 55, 149~155. [Google Scholar] [CrossRef]
- Kustov, A L; Hansen, T W; Kustova, M; et al. Selective catalytic reduction of NO by ammonia using mesoporous Fe-containing HZSM-5 and HZSM-12 zeolite catalysts: an option for automotive applications [J]. Applied Catalysis B: Environmental 2007, 76, 311~319. [Google Scholar] [CrossRef]
- Wang, X; Chi, R; Gu, L; et al. Effect of water vapor on low temperature SCR performances over Cu and Mn-based catalysts:A comparison study[J]. Journal of Rare Earths 2025, 43(08), 1661–1667. [Google Scholar] [CrossRef]
- Taboada, J B; Oveweg, A R; Crajé, M W J; et al. Systematic variation of 57Fe and Al content in isomorphously substituted and characterization [J]. Microporous and Mesoporous Materials 2004, 75, 237~246. [Google Scholar] [CrossRef]
- Long, R Q; Yang, R T. Reaction mechanism of selective catalytic reduction of NO with NH3 over Fe-ZSM-5 catalyst [J]. Journal of Catalysis 2002, 207, 224~231. [Google Scholar] [CrossRef]
- Berlier, G; Spoto, G; Bordiga, S; et al. Evolution of extraframework iron species in Fe silicalite 1. Effect of Fe content, activation temperature, and interaction with redox agents [J]. Journal of Catalysis 2002, 208, 64~82. [Google Scholar]
- Li, H; Ren, W; Xue, H; et al. Significantly enhanced low-temperature activity and SO2/H2O tolerance of Mn-Ce-Ox/TiO2 catalysts prepared by a facile citric acid assisted impregnation method[J]. Journal of Rare Earths 2025, 43(06), 1195–1204. [Google Scholar] [CrossRef]
- Brundle, C R; Chuang, T J; Wandelt, K. Core and valence level photoemission studies of iron-oxide surfaces and oxidation of iron [J]. Surface Science 1997, 68, 459~468. [Google Scholar] [CrossRef]
- Udovic, T J; Dumesic, J A. Preparation and characterization of magnetite surfaces on metallic iron substrates [J]. Journal of Catalysis 1984, 89, 303~313. [Google Scholar] [CrossRef]
- Fabrizioli, P; Bürgi, T; Burgener, M; et al. Synthesis, structural and chemical properties of iron oxide-silica aerogels. Journal of Materials Chemistry 2002, 12, 619~630. [Google Scholar]
- Chen, H Y; Sachtler, W M H. Activity and durability of Fe/ZSM-5 catalysts for lean burn NOx reduction in the presence of water vapor [J]. Catalysis Today 1998, 42, 73~83. [Google Scholar] [CrossRef]
- Long, R Q; Yang, R T. Characterization of Fe-ZSM-5 catalyst for selective catalytic reduction of nitric oxide by ammonia [J]. Journal of Catalysis 2000, 194, 80~90. [Google Scholar] [CrossRef]
- Malpartida, I; Marie, O; Bazin, P; et al. The NO/NOx ratio effect on the NH3-SCR efficiency of a commercial automotive Fe-zeolite catalyst studied by operando IR-MS [J]. Applied Catalysis B: Environmental 2012, 113-114, 52~60. [Google Scholar] [CrossRef]
- Colombo, M; Nova, I; Tronconi, E. A comparative study of the NH3-SCR reaction over a Cu-zeolite and a Fe-zeolite catalyst [J]. Catalysis Today 2010, 151, 223~230. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



