Submitted:
29 December 2025
Posted:
30 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Historical Background
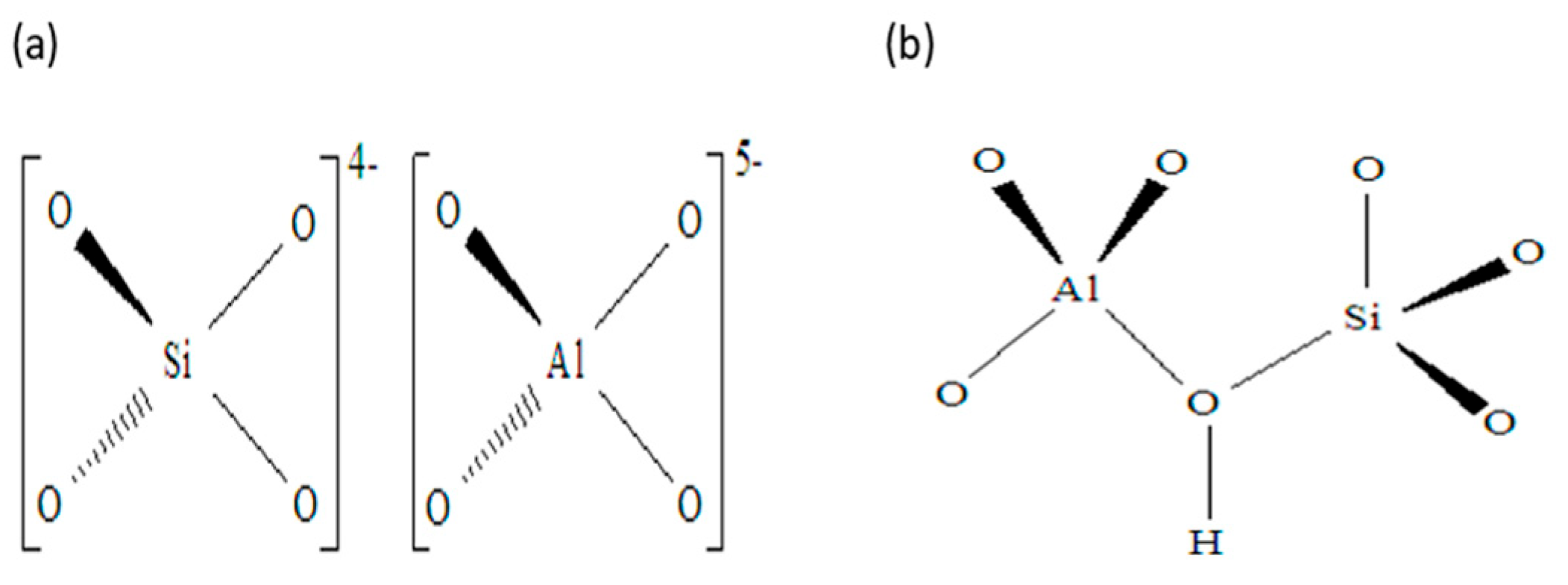
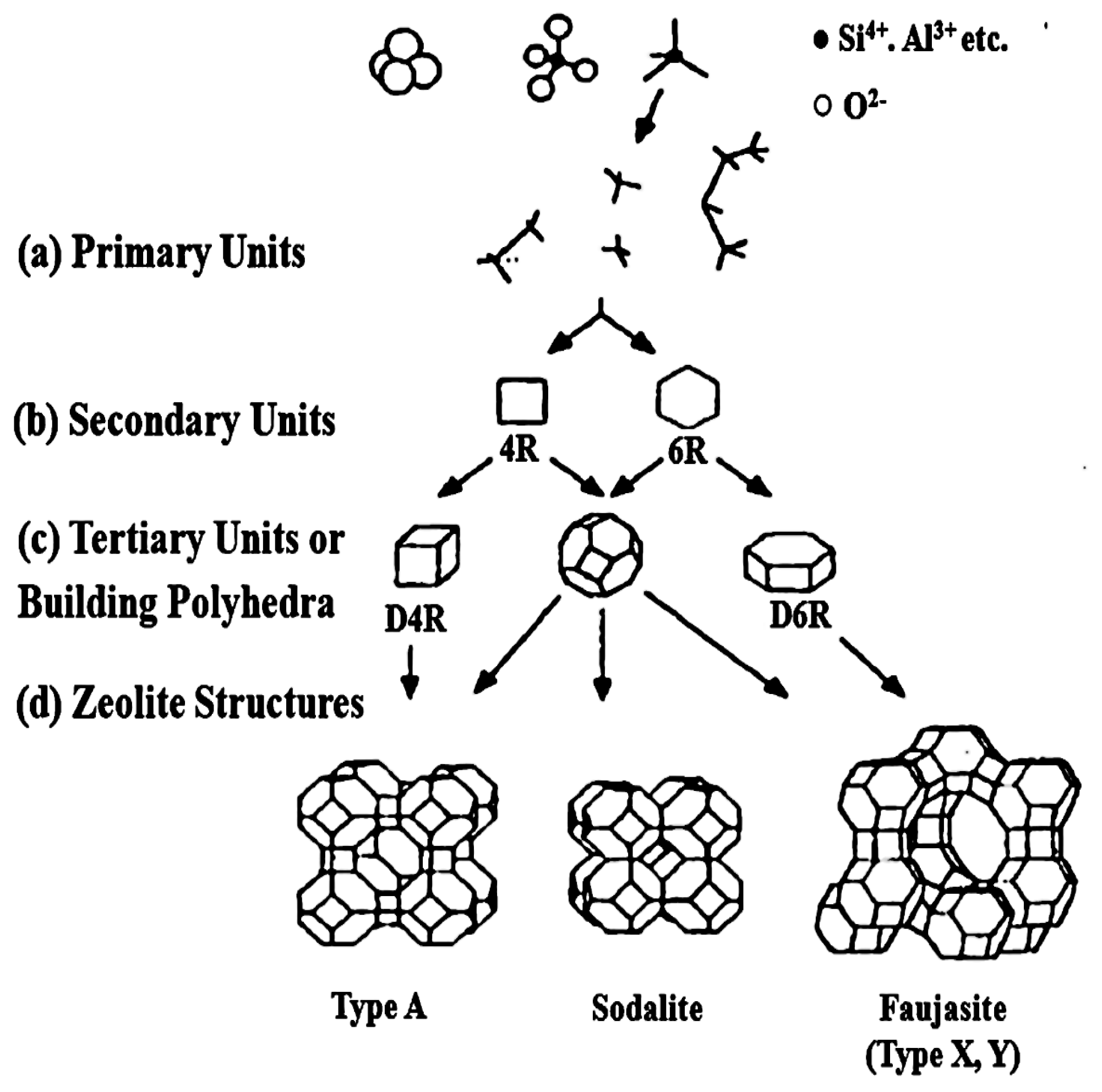
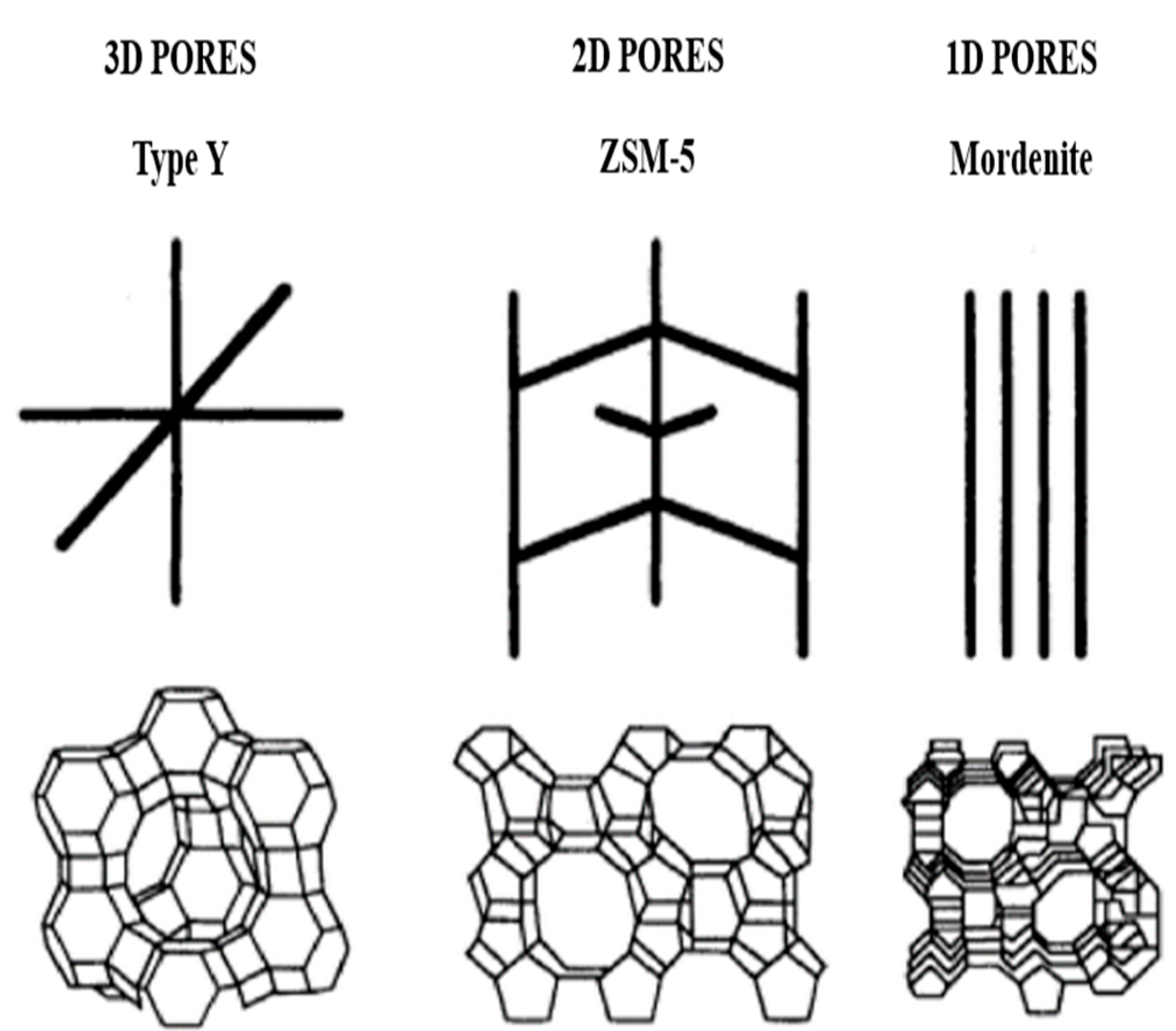
1.2. Description of Zeolite
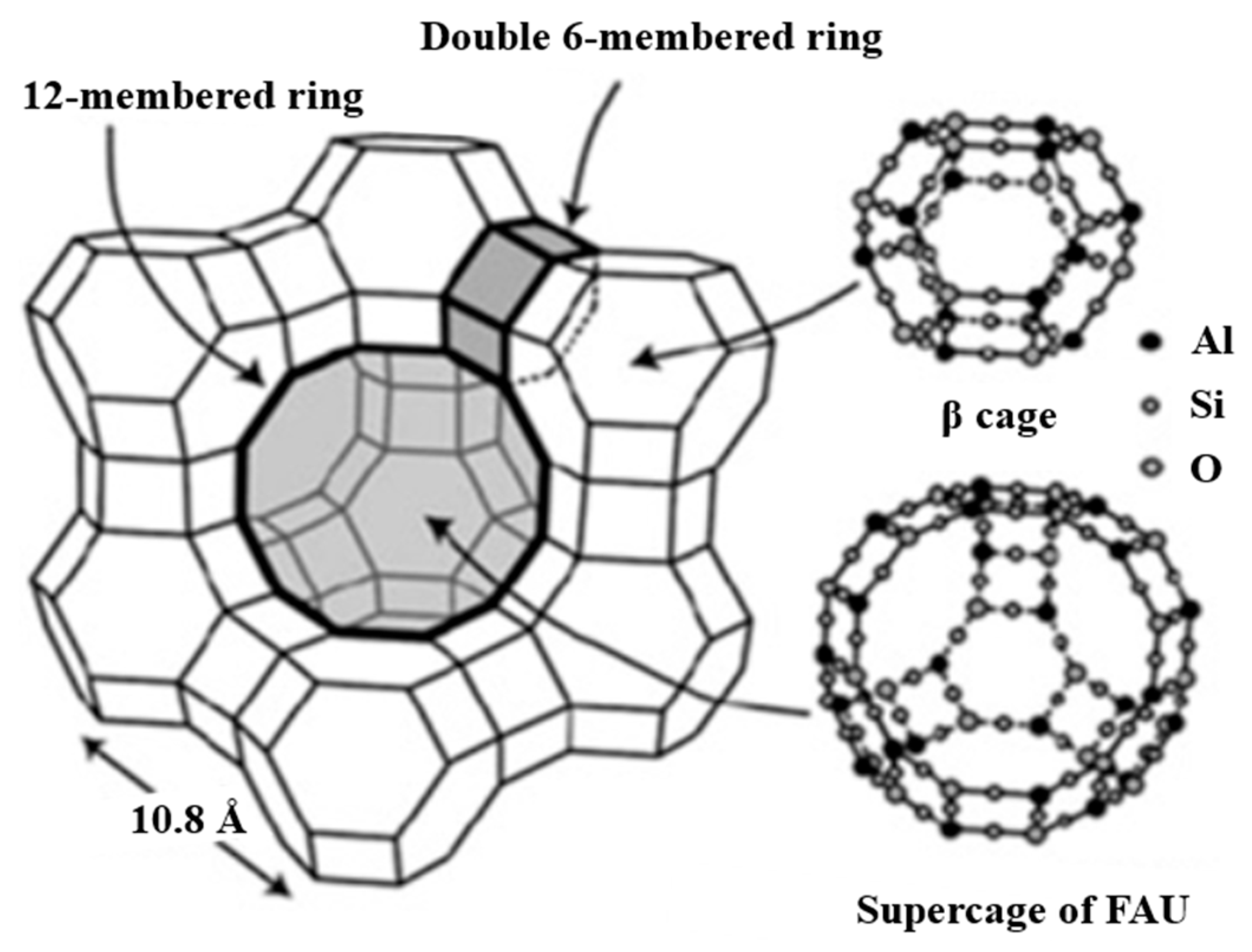
1.3. Zeolite Y
2. Types of Post-Synthesis Modifications
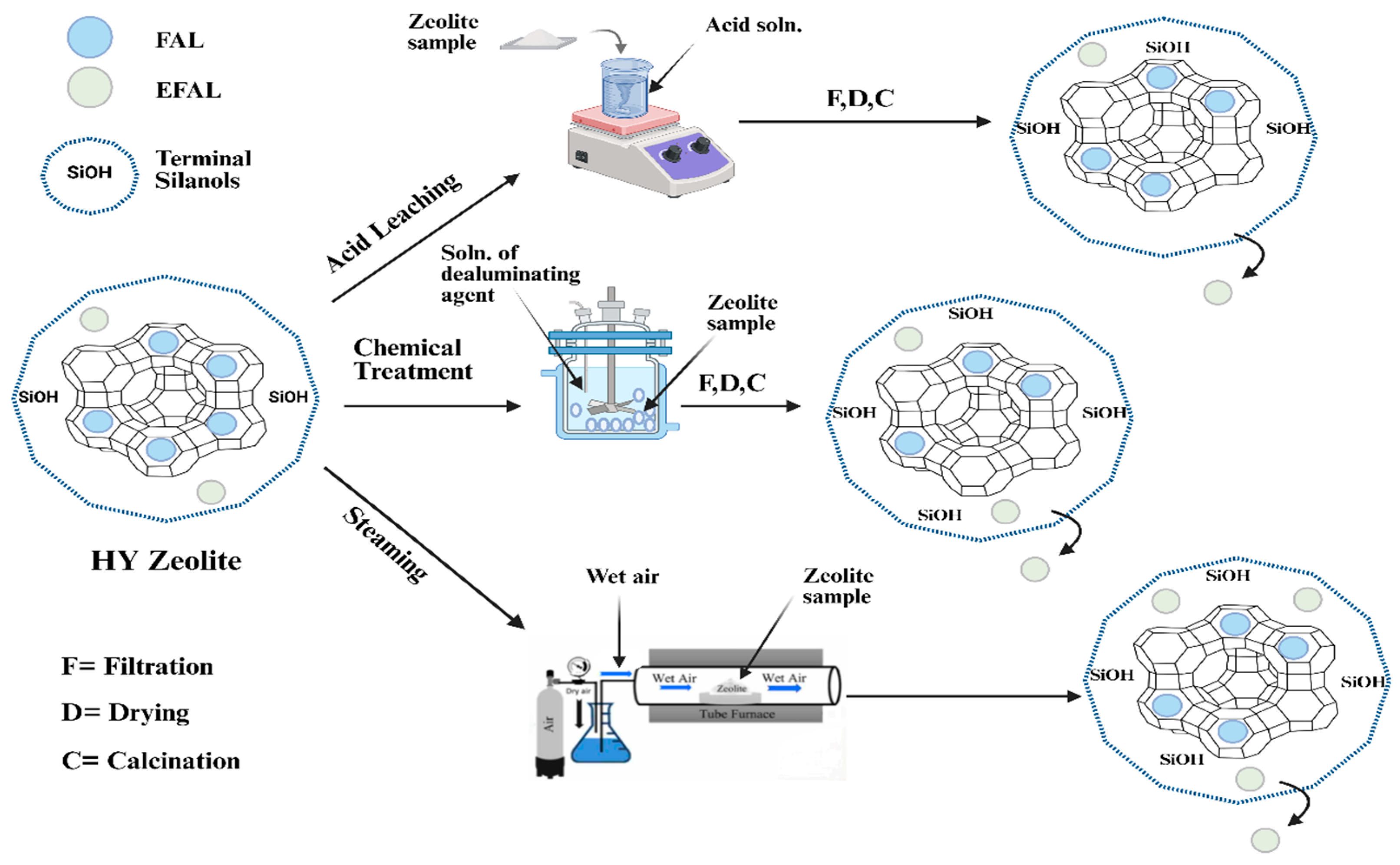
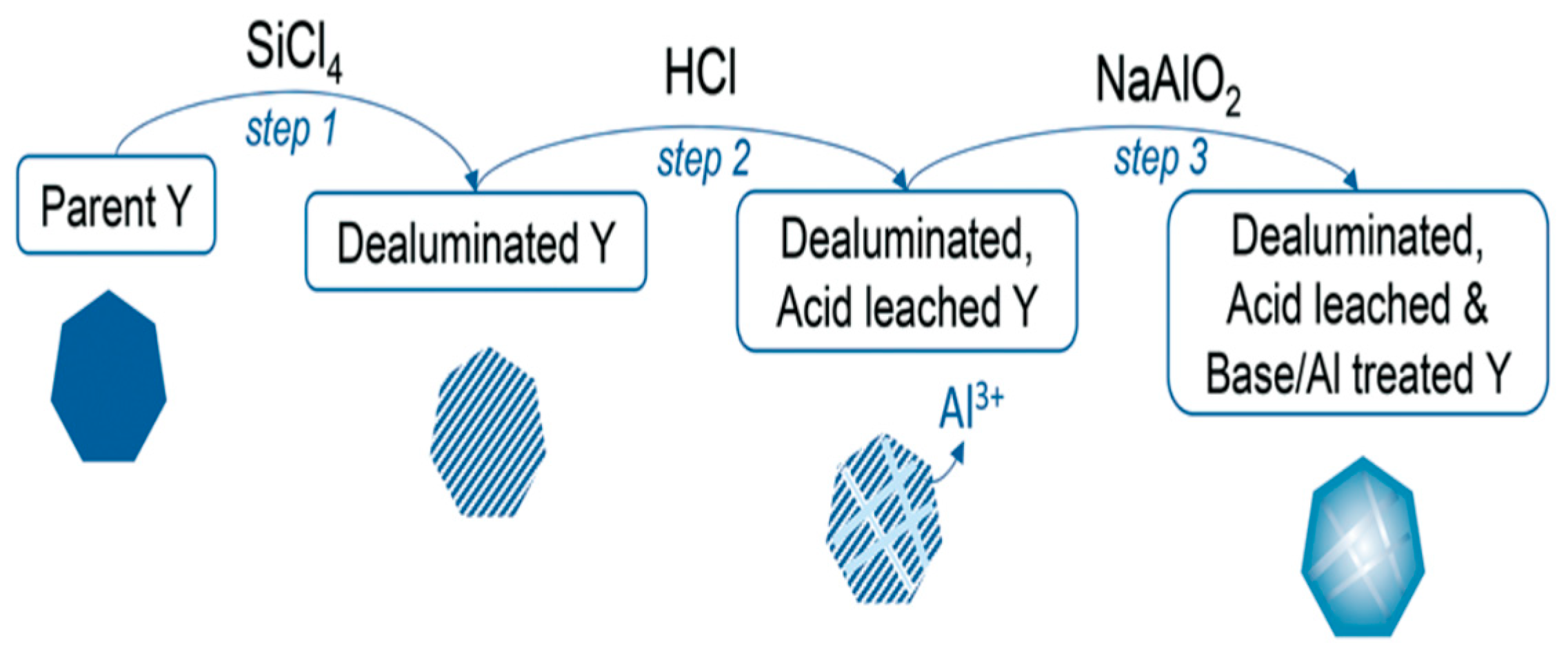
2.1. Dealumination Techniques
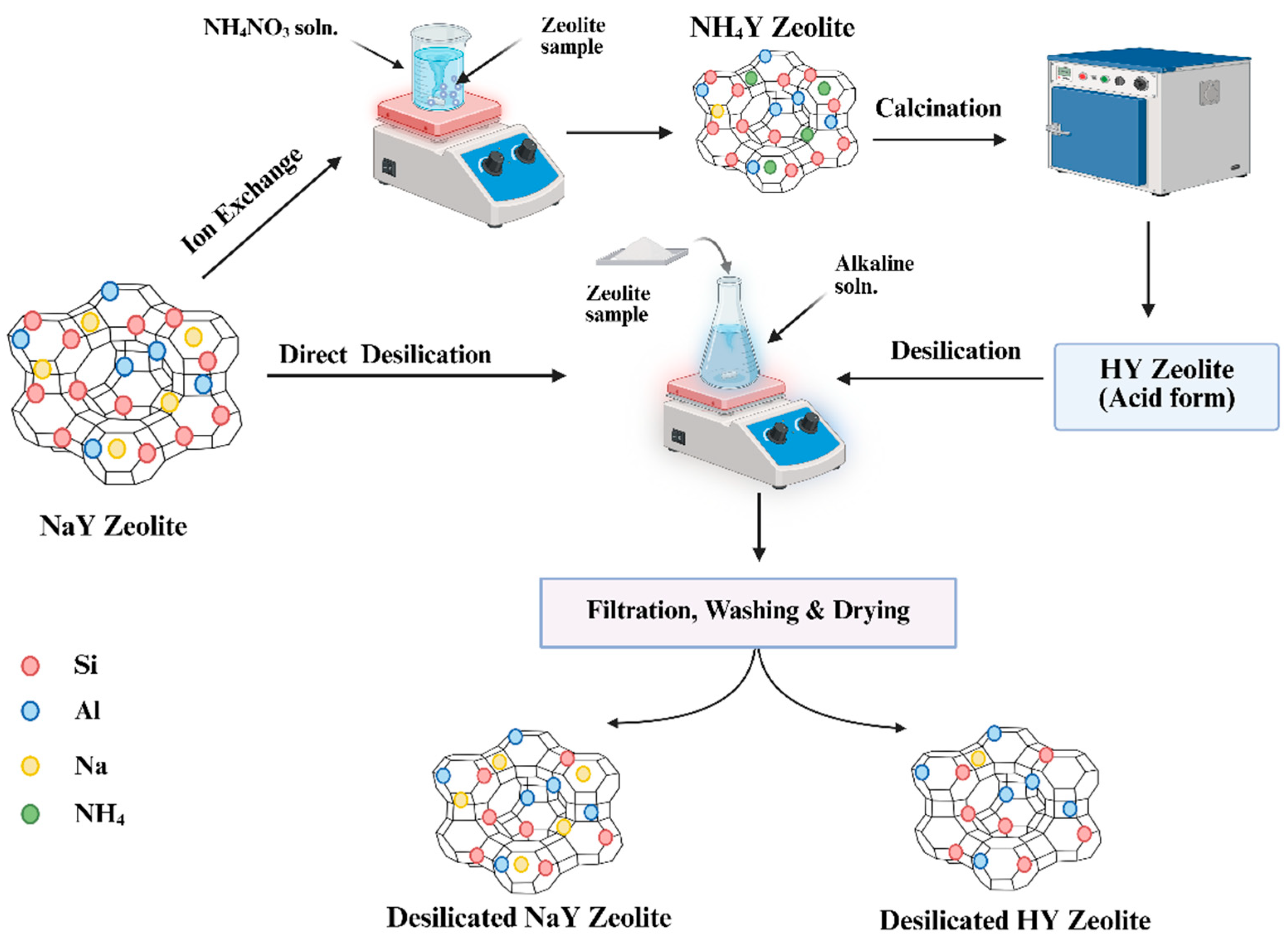
2.2. Desilication
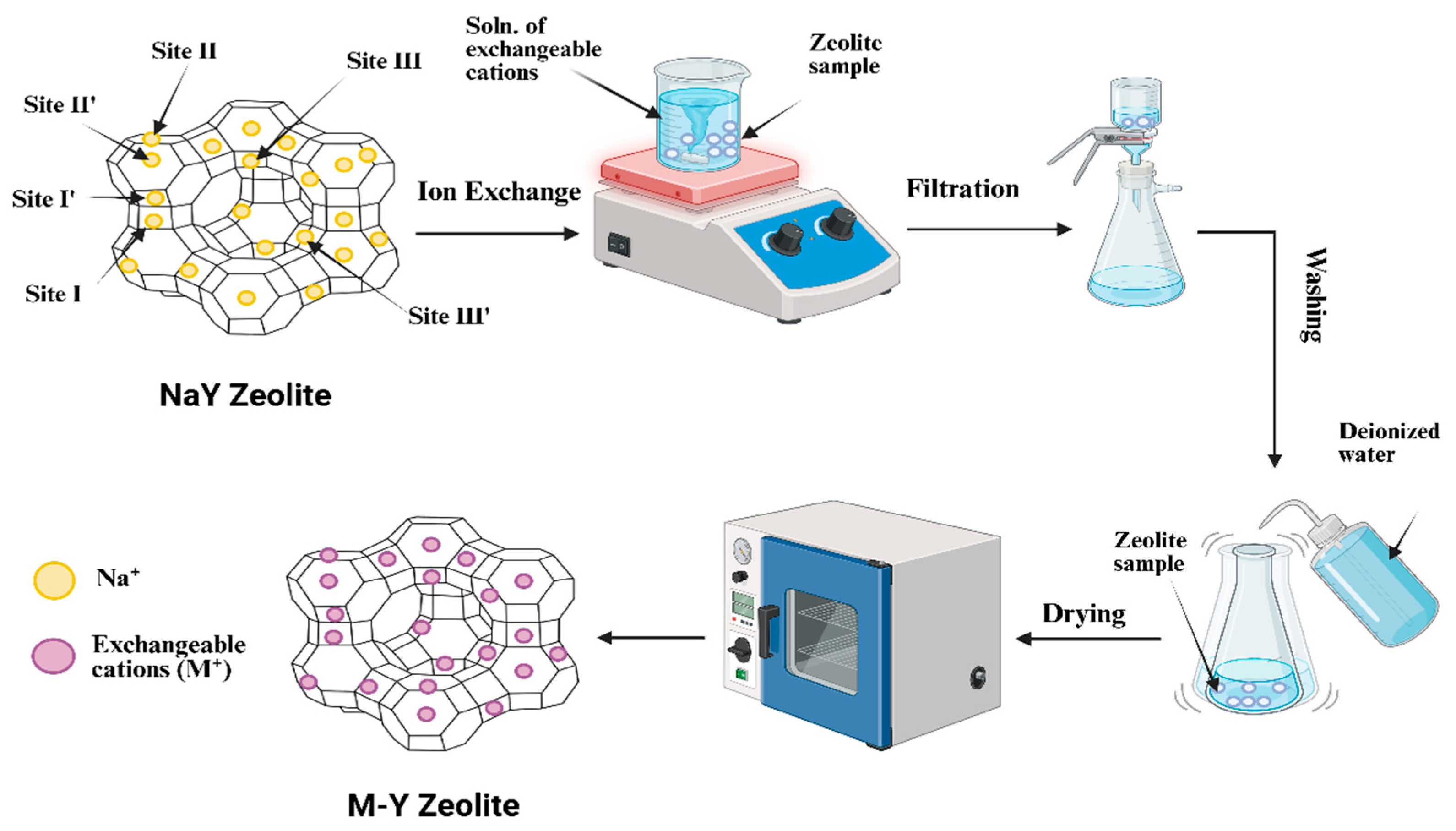
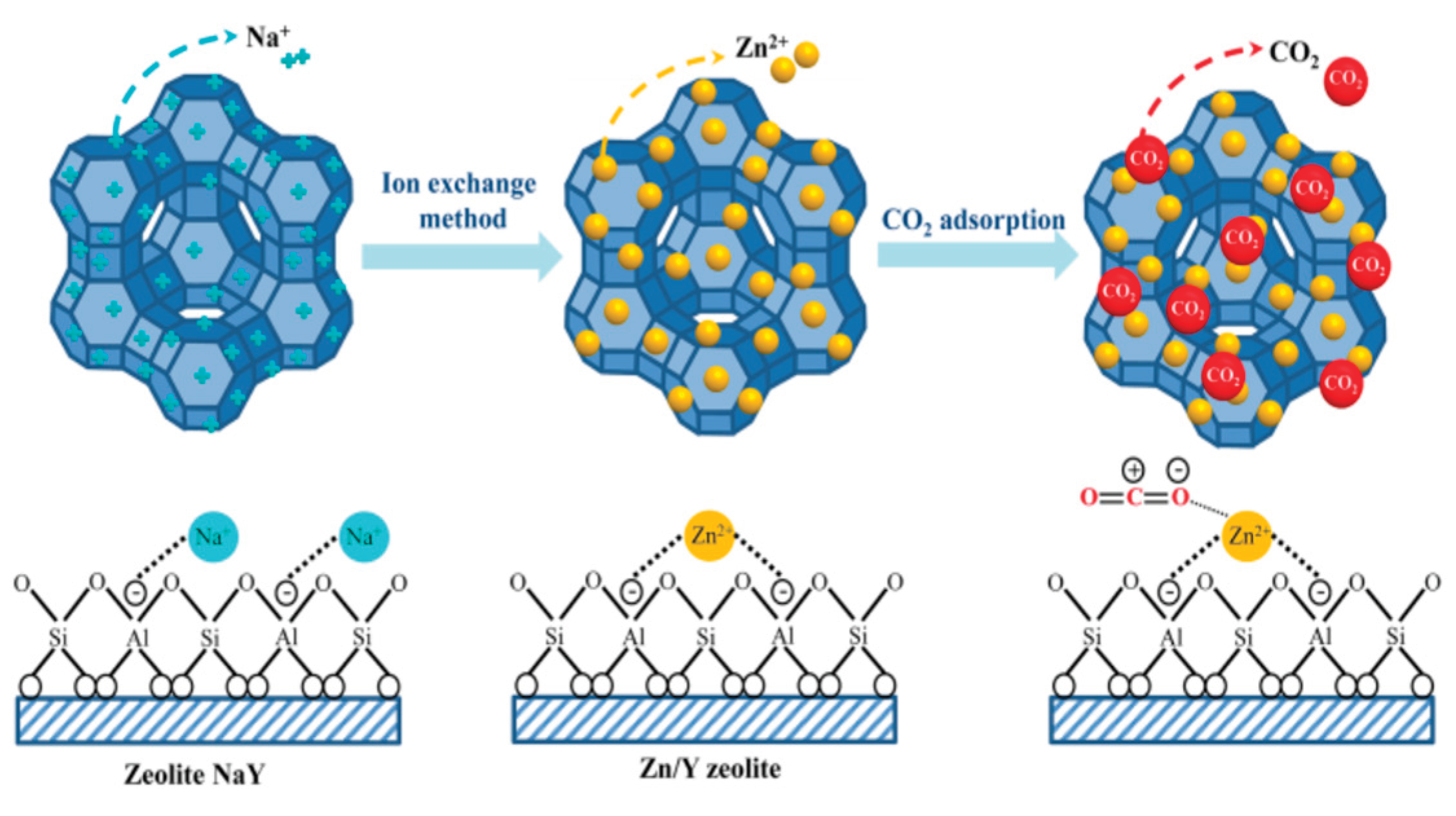
2.3. Ion Exchange
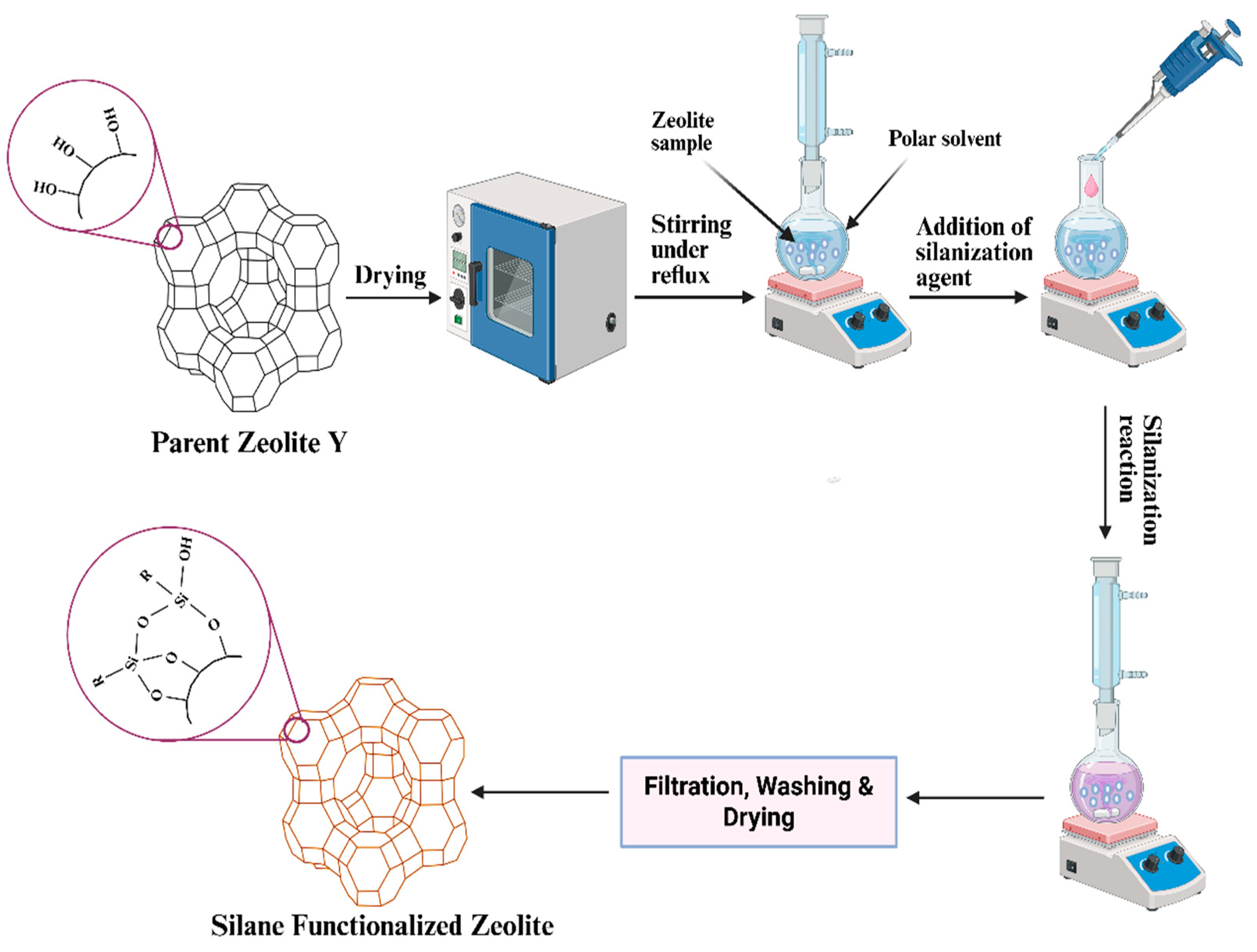
2.4. Surface Functionalization
3. Impact of Post-Synthesis Modifications on Zeolite Y Properties
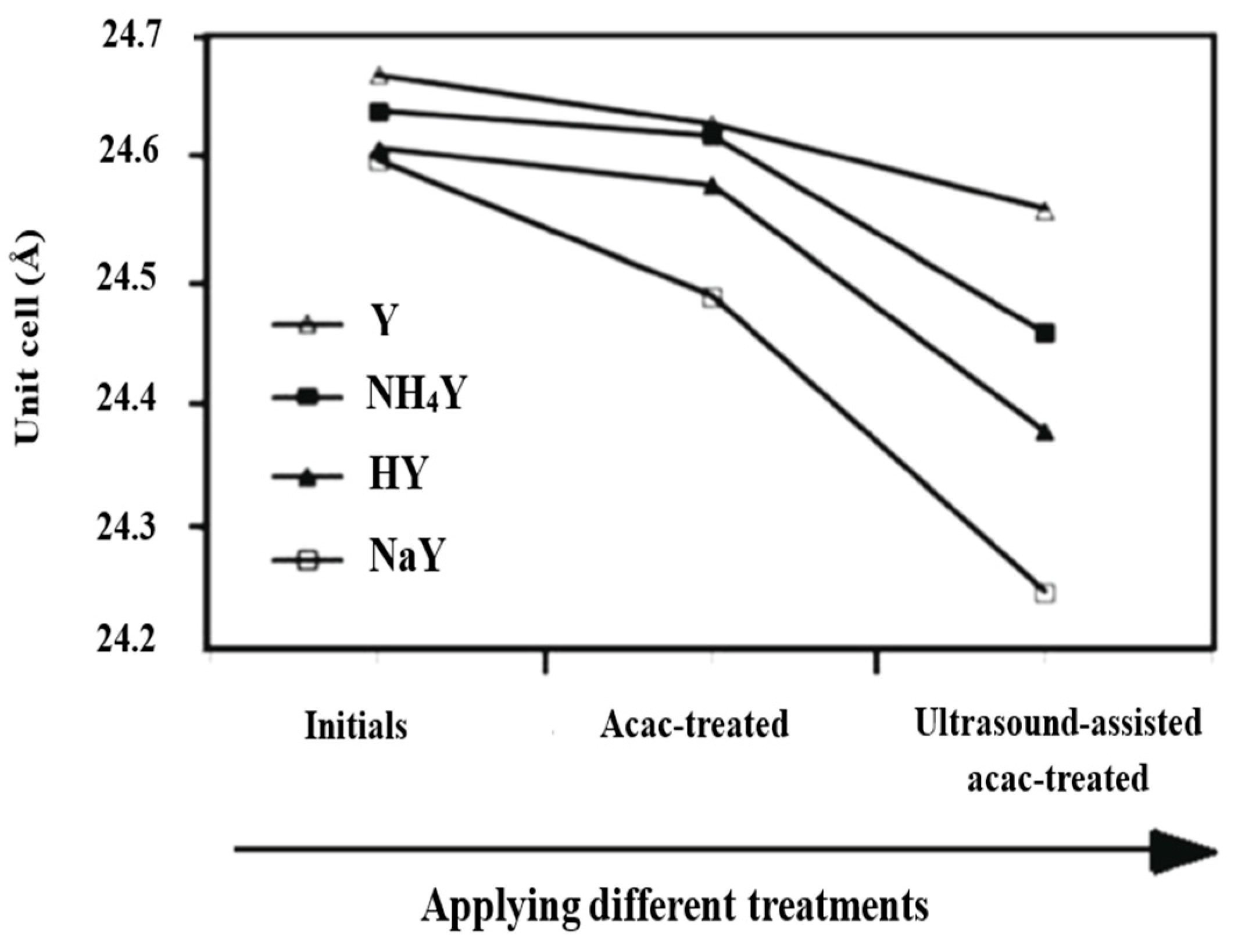
3.1. Structural Changes
3.2. Physicochemical Properties
4. Limitations and Future Prospects
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| FAU | Faujasite framework |
| USY | Ultra-Stable Y Zeolite |
| EFAl | Extra-Framework aluminum |
| AFS | Ammonium fluosilicate |
| EDTA | Ethylene diamine tetraacetic acid |
| AHFS | Ammonium hexafluorosilicate |
| NaOH | Sodium hydroxide |
| TAAOH | Tetraalkylammonium hydroxide |
| TPA | Tetrapropylammonium |
| CTA | Cetyltrimethylammonium |
| OMM | Ordered mesoporous material |
| CTAB | Cetyltrimethylammonium bromide |
| TPAOH | Tetrapropylammonium hydroxide |
| TBAOH | Tetrabutylammonium hydroxide |
| LPIE | Liquid-phase ion exchange |
| SSIE | Solid-state ion exchange |
| VOC | Volatile organic compound |
| APTES | 3-Aminopropyltriethoxysilane |
| APDEMS | 3-aminopropyl(diethoxy)methylsilane |
| MMM | Mixed matrix membrane |
| AEAPTMS | N-[3-(trimethoxysilyl)propyl]ethylenediamine () |
| PSA | Pressure swing adsorption |
| OTS | N-octadecyltrichlorosilane |
| TSA | Temperature swing adsorption |
| XRD | X-ray diffraction |
| SEM | Scanning electron microscopy |
| TEM | Transmission electron microscopy |
| BET | Brunauer–Emmett–Teller surface area analysis |
| NMR | Nuclear magnetic resonance spectroscopy |
| Raman | Raman spectroscopy |
| FTIR | Fourier transform infrared spectroscopy |
| XRF | X-ray fluorescence spectroscopy |
| TGA | Thermogravimetric analysis |
| NH₃-TPD | Ammonia temperature-programmed desorption |
| EPR | Electron paramagnetic resonance spectroscopy |
References
- F. Cronstedt, Rön och beskrifning om en obekant bärg art, som kallas Zeolites. 1756.
- M. A. Damour, “Sur quelques minéraux connus sous le nom de quartz résinite,” Ann. Mines, vol. 3, pp. 191–202, 1840.
- H. Eichhorn, “Ueber die einwirkung verdünnter salzlösungen auf silicate,” Ann Phys, vol. 181, no. 9, pp. 126–133, 1858.
- H. de S. Claire-Deville, “Reproduction de la levyne,” Comptes Rendus, vol. 54, no. 1862, pp. 324–327, 1862.
- G. Friedel, “Sur quelques proprietés nouvelles des zéolithes,” Bulletin de Minéralogie, vol. 19, no. 3, pp. 94–118, 1896.
- M. F. Grandjean, “Étude optique de l’absorption des vapeurs lourdes par certaines zéolithes,” CR Acad. Sci, vol. 149, pp. 866–868, 1909.
- Weigel and E. Steinhoff, “IX. Die Aufnahme organischer Flüssigkeitsdämpfe durch Chabasit,” Zeitschrift für Kristallographie-Crystalline Materials, vol. 61, no. 1–6, pp. 125–154, 1924.
- L. Pauling, “The structure of some sodium and calcium aluminosilicates,” Proceedings of the National Academy of Sciences, vol. 16, no. 7, pp. 453–459, 1930.
- L. Pauling, “XXII. The Structure of Sodalite and Helvite.,” Zeitschrift für Kristallographie-Crystalline Materials, vol. 74, no. 1–6, pp. 213–225, 1930.
- W. H. Taylor, “I. The structure of analcite (NaAlSi2O6· H2O,” Zeitschrift für Kristallographie-Crystalline Materials, vol. 74, no. 1–6, pp. 1–19, 1930.
- J. W. McBain, “The Sorption of Gases and Vapours by Solids.,” J Phys Chem, vol. 37, no. 1, pp. 149–150, 2002.
- R. M. Barrer, “The sorption of polar and non-polar gases by zeolites,” Proc R Soc Lond A Math Phys Sci, vol. 167, no. 930, pp. 392–420, 1938.
- R. M. Barrer, “Migration in crystal lattices,” Transactions of the Faraday Society, vol. 37, pp. 590–599, 1941.
- R. M. Barrer and E. A. D. White, “286. The hydrothermal chemistry of silicates. Part II. Synthetic crystalline sodium aluminosilicates,” Journal of the Chemical Society (Resumed), pp. 1561–1571, 1952.
- R. M. Barrer and D. W. Riley, “34. Sorptive and molecular-sieve properties of a new zeolitic mineral,” Journal of the Chemical Society (Resumed), pp. 133–143, 1948.
- R. M. Barrer, “33. Synthesis of a zeolitic mineral with chabazite-like sorptive properties,” Journal of the Chemical Society (Resumed), pp. 127–132, 1948.
- R. M. Barrer and D. J. Robinson, “The structures of the salt-bearing aluminosilicates, Species P and Q,” Zeitschrift für Kristallographie, vol. 135, no. 5–6, pp. 374–390, 1972.
- R. M. Barrer, “Preparation of some crystalline hydrogen zeolites,” Nature, vol. 164, no. 4159, pp. 112–113, 1949.
- R. M. Barrer and P. J. Denny, “201. Hydrothermal chemistry of the silicates. Part IX. Nitrogenous aluminosilicates,” Journal of the Chemical Society (Resumed), pp. 971–982, 1961.
- R. M. Barrer, P. J. Denny, and E. M. Flanigen, “Molecular sieve adsorbents,” Feb. 28, 1967, Google Patents.
- R. M. Barrer and H. Villiger, “Probable structure of zeolite Ω,” Journal of the Chemical Society D: Chemical Communications, no. 12, pp. 659–660, 1969.
- Martínez and J. Pérez-Pariente, “Zeolites and ordered porous solids: fundamentals and applications,” Valencia, Spain: FEZA, 2011.
- S. I. Zones, “Translating new materials discoveries in zeolite research to commercial manufacture,” Microporous and Mesoporous materials, vol. 144, no. 1–3, pp. 1–8, 2011.
- L. Gómez-Hortigüela and B. Bernardo-Maestro, Insights into the chemistry of organic structure-directing agents in the synthesis of zeolitic materials. Springer, 2018.
- R. M. Milton, “Molecular Sieve Science and Technology,” pp. 1–10, Jul. 1989. [CrossRef]
- E. M. Flanigen and J. A. Rabo, “A tribute to Robert Mitchell Milton, zeolite pioneer (1920-2000)-Obituary,” 2001, ELSEVIER SCIENCE BV PO BOX 211, 1000 AE AMSTERDAM, NETHERLANDS.
- R. M. Milton, “Molecular sieve adsorbents,” Apr. 14, 1959, Google Patents.
- W. Breck, W. G. Eversole, R. M. Milton, T. B. Reed, and T. L. Thomas, “Crystalline zeolites. I. The properties of a new synthetic zeolite, type A,” J Am Chem Soc, vol. 78, no. 23, pp. 5963–5972, 1956.
- W. Breck, W. G. Eversole, and R. M. Milton, “New synthetic crystalline zeolites,” J Am Chem Soc, vol. 78, no. 10, pp. 2338–2339, 1956.
- T. B. Reed and D. W. Breck, “Crystalline zeolites. II. Crystal structure of synthetic zeolite, type A,” J Am Chem Soc, vol. 78, no. 23, pp. 5972–5977, 1956.
- M. Flanigen, J. C. Jansen, and H. van Bekkum, Introduction to zeolite science and practice. Elsevier, 1991.
- M. Flanigen, R. W. Broach, and S. T. Wilson, “Zeolites in industrial separations and catalysis. Zeolites in Industrial Separation and Catalysis. 1–26,” 2010.
- V. Verdoliva, M. Saviano, and S. De Luca, “Zeolites as acid/basic solid catalysts: recent synthetic developments,” Catalysts, vol. 9, no. 3, p. 248, 2019.
- Pines, The chemistry of catalytic hydrocarbon conversions. Elsevier, 2012.
- J. Weitkamp, “Zeolites and catalysis,” Solid State Ion, vol. 131, no. 1–2, pp. 175–188, 2000.
- Primo and H. Garcia, “Zeolites as catalysts in oil refining,” Chem Soc Rev, vol. 43, no. 22, pp. 7548–7561, 2014.
- L. B. McCusker and C. Baerlocher, “Zeolite structures,” in Studies in Surface Science and Catalysis, vol. 157, Elsevier, 2005, pp. 41–64.
- C. H. Bartholomew and R. J. Farrauto, Fundamentals of industrial catalytic processes. John Wiley & Sons, 2011.
- Kamlesh Sahu, “STUDIES ON THE SYNTHESIS AND CHARACTERIZATION OF ZEOLITES AND THEIR APPLICATION AS A CATALYST,” International Journal for Research Publication and Seminar, vol. 15, pp. 357–364, Sep. 2024. [CrossRef]
- S. Qiu et al., “Zeolite-Based Materials for Catalytic Oxidation of Volatile Organic Compounds,” 2024, John Wiley and Sons Inc. [CrossRef]
- Q. Lang, P. Lu, X. Yang, and V. Valtchev, “Zeolites for the environment,” Green Carbon, vol. 2, pp. 12–32, Mar. 2024. [CrossRef]
- W. Zhang et al., “Recent Progress on the Synthesis and Applications of Zeolites from Industrial Solid Wastes,” Catalysts, vol. 14, p. 734, Oct. 2024. [CrossRef]
- M. A. den Hollander, M. Wissink, M. Makkee, and J. A. Moulijn, “Gasoline conversion: reactivity towards cracking with equilibrated FCC and ZSM-5 catalysts,” Appl Catal A Gen, vol. 223, no. 1–2, pp. 85–102, 2002.
- K. Sun, J. Lu, L. Ma, Y. Han, Z. Fu, and J. Ding, “A comparative study on the catalytic performance of different types of zeolites for biodiesel production,” Fuel, vol. 158, pp. 848–854, 2015.
- P. K. Bajpai, “Synthesis of mordenite type zeolite,” Zeolites, vol. 6, no. 1, pp. 2–8, 1986.
- V. Komvokis, L. X. L. Tan, M. Clough, S. S. Pan, and B. Yilmaz, “Zeolites in fluid catalytic cracking (FCC),” Zeolites in Sustainable Chemistry: Synthesis, Characterization and Catalytic Applications, pp. 271–297, 2016.
- W. Li, J. Zheng, Y. Luo, C. Tu, Y. Zhang, and Z. Da, “Hierarchical Zeolite Y with Full Crystallinity: Formation Mechanism and Catalytic Cracking Performance,” Energy and Fuels, vol. 31, pp. 3804–3811, Apr. 2017. [CrossRef]
- R. Simancas et al., “Recent progress in the improvement of hydrothermal stability of zeolites,” Jun. 2021, Royal Society of Chemistry. [CrossRef]
- J. Zang, H. Yu, G. Liu, M. Hong, J. Liu, and T. Chen, “Research Progress on Modifications of Zeolite Y for Improved Catalytic Properties,” Jan. 2023, MDPI. [CrossRef]
- Y. D. G. Edañol, K. A. S. Usman, S. Jr. C. Buenviaje, M. E. Mantua, and L. Jr. M. Payawan, “Utilizing Silica from Rice Hull for the Hydrothermal Synthesis of Zeolite Y,” KIMIKA, vol. 29, pp. 17–21, Jun. 2018. [CrossRef]
- S. Jafari, H. Asilian Mahabady, and H. Kazemian, “Gold-nano particles supported on Na-Y and H-Y types zeolites: Activity and thermal stability for CO oxidation reaction,” Catal Letters, vol. 128, pp. 57–63, Mar. 2009. [CrossRef]
- B. Meng et al., “Synthesis of USY zeolite with a high mesoporous content by introducing Sn and enhanced catalytic performance,” Ind Eng Chem Res, vol. 59, no. 13, pp. 5712–5719, 2020.
- J. Scherzer and A. P. Humphries, “Dealumination of faujasite-type zeolites using ion exchange resins,” Apr. 23, 1985, Google Patents.
- T. Yoshioka, K. Iyoki, Y. Yanaba, T. Okubo, and T. Wakihara, “Dealumination of RHO zeolite by acid treatment and recrystallization with organic pore filler,” Journal of the Ceramic Society of Japan, vol. 132, no. 2, pp. 45–49, 2024.
- P. Huifang, W. Xiaofeng, Z. Yewen, and S. Zhihong, “The dealumination process of acid attack and coking behaviour in ultrastable Y zeolites,” in Studies in Surface Science and Catalysis, vol. 88, Elsevier, 1994, pp. 223–231.
- L. Buchori and D. D. Anggoro, “Modification of zeolite Y as a catalyst in the production of dimethyl ether from methanol dehydration,” Journal of Engineering Research, 2022.
- Maghfirah, Y. Susanti, A. T. N. Fajar, R. R. Mukti, and G. T. M. Kadja, “The role of tetraalkylammonium for controlling dealumination of zeolite y in acid media,” Mater Res Express, vol. 6, Jun. 2019. [CrossRef]
- M.-C. Silaghi, “Ab initio molecular modelling of the dealumination and desilication mechanisms of relevant zeolite frameworks,” 2014.
- Y. Kamimura, T. Kodaira, H. Yamada, N. Hiyoshi, and A. Endo, “Direct Visualization of the Dealumination Process on Zeolite Y: How Was the Mesoporous Architecture Formed?,” Chemistry of Materials, vol. 37, no. 8, pp. 2735–2748, Apr. 2025. [CrossRef]
- S. Abdulridha et al., “An efficient microwave-assisted chelation (MWAC) post-synthetic modification method to produce hierarchical Y zeolites,” Microporous and Mesoporous Materials, vol. 311, p. 110715, 2021.
- W. Lutz et al., “Investigations of the mechanism of dealumination of zeolite Y by steam: Tuned mesopore formation versus the Si/Al ratio,” in Studies in Surface Science and Catalysis, Elsevier Inc., 2004, pp. 1411–1417. [CrossRef]
- W. Lutz, “Zeolite Y: Synthesis, modification, and properties—A case revisited,” Advances in Materials Science and Engineering, vol. 2014, no. 1, p. 724248, 2014.
- Pliekhov et al., “Study of water adsorption on EDTA dealuminated zeolite Y,” Microporous and Mesoporous Materials, vol. 302, Aug. 2020. [CrossRef]
- J. Pérez-Ramírez, C. H. Christensen, K. Egeblad, C. H. Christensen, and J. C. Groen, “Hierarchical zeolites: enhanced utilisation of microporous crystals in catalysis by advances in materials design,” Chem Soc Rev, vol. 37, no. 11, pp. 2530–2542, 2008.
- C. Manrique, R. Solano, C. Mendoza, S. Amaya, and A. Echavarría, “Hierarchical submicrosized Y zeolites prepared by sequential desilication–dealumination post-synthesis modification and their catalytic performance in vacuum gas oil hydrocracking,” New Journal of Chemistry, vol. 48, no. 14, pp. 6188–6200, 2024.
- K. Qiao et al., “An efficient modification of ultra-stable Y zeolites using citric acid and ammonium fluosilicate,” Appl Petrochem Res, vol. 4, pp. 373–378, 2014.
- X. Li et al., “Combined modification of ultra-stable Y zeolites via citric acid and phosphoric acid,” Appl Petrochem Res, vol. 4, pp. 343–349, 2014.
- K. Qiao et al., “Modification of USY zeolites with malic–nitric acid for hydrocracking,” Appl Petrochem Res, vol. 6, pp. 353–359, 2016.
- K. Qiao, L. Wei, R. Feng, Z. Yan, Z. Zhang, and X. Gao, “Preparation and characterization of hierarchical USY by post-treatment,” Appl Petrochem Res, vol. 5, pp. 313–319, 2015.
- M. Hosseini, M. A. Zanjanchi, B. Ghalami-Choobar, and H. Golmojdeh, “Ultrasound-assisted dealumination of zeolite y,” Journal of Chemical Sciences, vol. 127, pp. 25–31, 2015. [CrossRef]
- S. Esmaeili, M. A. Zanjanchi, H. Golmojdeh, and F. Mizani, “Increasing the adsorption capabilitiy of mordenite and Y zeolites via post-synthesis chemical/physical treatments in order to remove cationic dyes from polluted water,” Water and Environment Journal, vol. 34, pp. 117–130, Feb. 2020. [CrossRef]
- C. S. Triantafillidis and N. P. Evmiridis, “Dealuminated H− Y zeolites: influence of the number and type of acid sites on the catalytic activity for isopropanol dehydration,” Ind Eng Chem Res, vol. 39, no. 9, pp. 3233–3240, 2000.
- Agostini et al., “In situ XAS and XRPD parametric rietveld refinement to understand dealumination of Y zeolite catalyst,” J Am Chem Soc, vol. 132, pp. 667–678, Jan. 2010. [CrossRef]
- K. S. Yoo, S. Gopal, and P. G. Smirniotis, “Enhancement of n-hexane cracking activity over modified ZSM-12 zeolites,” Ind Eng Chem Res, vol. 44, no. 13, pp. 4562–4568, 2005.
- V. Yakimov et al., “Dealumination of Nanosized Zeolites Y,” Petroleum Chemistry, vol. 59, pp. 540–545, May 2019. [CrossRef]
- D. Verboekend, M. Milina, S. Mitchell, and J. Pérez-Ramírez, “Hierarchical zeolites by desilication: Occurrence and catalytic impact of recrystallization and restructuring,” Cryst Growth Des, vol. 13, no. 11, pp. 5025–5035, 2013.
- D. Liang, Y. Liu, R. Zhang, Q. Xie, and L. Zhang, “A Review on the Influence Factors in the Synthesis of Zeolites and the Transformation Behavior of Silicon and Aluminum During the Process,” 2024, Taylor and Francis Ltd. [CrossRef]
- R. García, M. Falco, and U. Sedran, “Impact of the Desilication Treatment of y Zeolite on the Catalytic Cracking of Bulky Hydrocarbon Molecules,” Top Catal, vol. 59, pp. 268–277, Feb. 2016. [CrossRef]
- G. Dashtpeyma, S. R. Shabanian, J. Ahmadpour, and M. Nikzad, “Effect of desilication of NaY zeolite on sulfur content reduction of gasoline model in presence of toluene and cyclohexene,” Chemical Engineering Research and Design, vol. 178, pp. 523–539, Feb. 2022. [CrossRef]
- Y. Han, K. Larmier, M. Rivallan, and G. D. Pirngruber, “Generation of mesoporosity in H–Y zeolites by basic or acid/basic treatments: Towards a guideline of optimal Si/Al ratio and basic reagent,” Microporous and Mesoporous Materials, vol. 365, Feb. 2024. [CrossRef]
- D. Verboekend, T. C. Keller, S. Mitchell, and J. Pérez-Ramírez, “Hierarchical FAU-and LTA-type zeolites by post-synthetic design: a new generation of highly efficient base catalysts,” Adv Funct Mater, vol. 23, no. 15, pp. 1923–1934, 2013.
- R. Zhang et al., “Using ultrasound to improve the sequential post-synthesis modification method for making mesoporous Y zeolites,” Front Chem Sci Eng, vol. 14, pp. 275–287, 2020.
- C. Pagis, A. R. M. Prates, N. Bats, A. Tuel, and D. Farrusseng, “High-silica hollow Y zeolite by selective desilication of dealuminated NaY crystals in the presence of protective Al species,” CrystEngComm, vol. 20, no. 11, pp. 1564–1572, 2018.
- Mandela, W. Trisunaryanti, T. Triyono, M. Koketsu, and D. A. Fatmawati, “Hydrochloric Acid and/or Sodium Hydroxide-modified Zeolite Y for Catalytic Hydrotreating of α-Cellulose Bio-Oil,” Indonesian Journal of Chemistry, vol. 21, no. 4, pp. 787–796.
- D. Verboekend, G. Vilé, and J. Pérez-Ramírez, “Mesopore formation in usy and beta zeolites by base leaching: Selection criteria and optimization of pore-directing agents,” Cryst Growth Des, vol. 12, pp. 3123–3132, Jun. 2012. [CrossRef]
- D. Verboekend and J. Pérez-Ramírez, “Desilication mechanism revisited: Highly mesoporous all-silica zeolites enabled through pore-directing agents,” Chemistry - A European Journal, vol. 17, pp. 1137–1147, Jan. 2011. [CrossRef]
- D. Verboekend, G. Vilé, and J. Pérez-Ramírez, “Hierarchical Y and USY zeolites designed by post-synthetic strategies,” Adv Funct Mater, vol. 22, no. 5, pp. 916–928, 2012.
- C. F. Imbachi-Gamba, J. P. Cano-Restrepo, and A. L. Villa, “Effect of the Desilication Conditions on the Synthesis of Hierarchical Zeolite Y,” in 2019 North American Catalysis Society Meeting, NAM, 2019.
- W. Q. Jiao, W. H. Fu, X. M. Liang, Y. M. Wang, and M. Y. He, “Preparation of hierarchically structured y zeolite with low Si/Al ratio and its applications in acetalization reactions,” RSC Adv, vol. 4, pp. 58596–58607, Oct. 2014. [CrossRef]
- M. Gackowski, Ł. Kuterasiński, J. Podobiński, and J. Datka, “Hydroxyl groups of exceptionally high acidity in desilicated zeolites Y,” ChemPhysChem, vol. 19, no. 24, pp. 3372–3379, 2018.
- M. Gackowski, J. Podobiński, E. Broclawik, and J. Datka, “IR and NMR Studies of the status of Al and acid sites in desilicated zeolite Y,” Molecules, vol. 25, no. 1, p. 31, 2019.
- M. Zendehdel, M. Kooti, and M. M. Amini, “Dispersion and solid state ion exchange of VCl3, CrCl 3·6H2O, MnCl2·4H2O and CoCl2·6H2O onto the surface of NaY zeolite using microwave irradiation,” Journal of Porous Materials, vol. 12, pp. 143–149, Apr. 2005. [CrossRef]
- T. Alam, Y. K. Krisnandi, W. Wibowo, D. A. Nurani, D. U. C. Rahayu, and H. Haerudin, “Synthesis and characterization hierarchical HY zeolite using template and non template methods,” in AIP Conference Proceedings, American Institute of Physics Inc., Oct. 2018. [CrossRef]
- S. M. Seo, D. J. Moon, J. An, H.-K. Jeong, and W. T. Lim, “Time-dependent Ni2+-ion exchange in zeolites y (FAU, si/Al= 1.56) and their single-crystal structures,” The Journal of Physical Chemistry C, vol. 120, no. 50, pp. 28563–28574, 2016.
- Z. Ayad, H. Q. Hussein, and B. A. Al-Tabbakh, “Synthesis and characterization of high silica HY zeolite by basicity reduction,” in AIP Conference Proceedings, American Institute of Physics Inc., Mar. 2020. [CrossRef]
- Y. K. Krisnandi et al., “Synthesis and characterization of crystalline NaY-Zeolite from Belitung Kaolin as catalyst for n-Hexadecane cracking,” Crystals (Basel), vol. 9, 2019. [CrossRef]
- Q. Dong, R. Li, and H. Jiao, “Zeolite Catalysts for Selective Hydrocracking of Polycyclic Aromatic Hydrocarbons – Structures and Mechanisms,” ChemCatChem, Nov. 2024. [CrossRef]
- Y. Khalaf, B. Sherhan, and Z. Shakour, “Hydroisomerization of n-Heptane in a Fixed-Bed Reactor Using a Synthesized Bimetallic Type-HY Zeolite Catalyst,” Engineering and Technology Journal, vol. 40, pp. 1–13, Sep. 2022. [CrossRef]
- S. M. Seo, H. S. Kim, D. Y. Chung, J. M. Suh, and W. T. Lim, “The Effect of Co2+-ion exchange time into zeolite y (FAU, Si/Al = 1.56): Their Single-Crystal Structures,” Bull Korean Chem Soc, vol. 35, pp. 243–249, Jan. 2014. [CrossRef]
- S. Shrestha and P. K. Dutta, “Photochemical water oxidation by manganese oxides supported on zeolite surfaces,” ChemistrySelect, vol. 1, no. 7, pp. 1431–1440, 2016.
- H. S. Kim, D. J. Moon, H. Y. Yoo, J. S. Park, M. Park, and W. T. Lim, “A crystallographic study of Sr2+ and K+ ion-exchanged zeolite Y (FAU, Si/Al = 1.56) from binary solution with different mole ratio of Sr2+ and K+,” Journal of Porous Materials, vol. 27, pp. 63–71, Feb. 2020. [CrossRef]
- Y. Khabzina, C. Laroche, C. Pagis, and D. Farrusseng, “Monovalent and bivalent cations exchange isotherms for faujasites X and y,” Physical Chemistry Chemical Physics, vol. 19, pp. 17242–17249, 2017. [CrossRef]
- Wu and A. Navrotsky, “Synthesis and thermodynamic study of transition metal ion (Mn2+, Co2+, Cu2+, and Zn2+) exchanged zeolites A and y,” Physical Chemistry Chemical Physics, vol. 18, pp. 10116–10122, Apr. 2016. [CrossRef]
- T. Izquierdo, R. Juan, B. Rubio, and C. Gómez-Giménez, “No removal in the selective catalitic reduction process over Cu and Fe exchanged type Y zeolites synthesized from coal fly ash,” Energy Sources, Part A: Recovery, Utilization, and Environmental Effects, vol. 38, no. 9, pp. 1183–1188, 2016.
- C. Wang, E. Xing, J. Zheng, Y. Luo, and X. Shu, “Effects of Pressured Steaming on the Migration of La3+ Ions and the Performance of As-Prepared LaY Zeolite,” Ind Eng Chem Res, vol. 60, no. 47, pp. 17268–17277, 2021.
- Dastanian and F. Seyedeyn-Azad, “Desulfurization of gasoline over nanoporous nickel-loaded Y-type zeolite at ambient conditions,” Ind Eng Chem Res, vol. 49, no. 22, pp. 11254–11259, 2010.
- X. Chen, B. Shen, H. Sun, and G. Zhan, “Ion-exchanged zeolites Y for selective adsorption of methyl mercaptan from natural gas: experimental performance evaluation and computational mechanism explorations,” Ind Eng Chem Res, vol. 56, no. 36, pp. 10164–10173, 2017.
- X. Chen, B. X. Shen, H. Sun, G. X. Zhan, and Z. Z. Huo, “Adsorption and Its Mechanism of CS2 on Ion-Exchanged Zeolites y,” Ind Eng Chem Res, vol. 56, pp. 6499–6507, Jun. 2017. [CrossRef]
- P. Tobarameekul, S. Sangsuradet, and P. Worathanakul, “Comparative Study of Zn Loading on Advanced Functional Zeolite NaY from Bagasse Ash and Rice Husk Ash for Sustainable CO2 Adsorption with ANOVA and Factorial Design,” Atmosphere (Basel), vol. 13, Feb. 2022. [CrossRef]
- Djeffal, M. Benbouzid, B. Boukoussa, H. Sekkiou, and A. Bengueddach, “CO2 adsorption properties of ion-exchanged zeolite y prepared from natural clays,” Mater Res Express, vol. 4, Mar. 2017. [CrossRef]
- R. S. Pillai, G. Sethia, and R. V Jasra, “Sorption of CO, CH4, and N2 in alkali metal ion exchanged zeolite-X: grand canonical Monte Carlo simulation and volumetric measurements,” Ind Eng Chem Res, vol. 49, no. 12, pp. 5816–5825, 2010.
- B. Silva et al., “Reutilization of Cr-Y zeolite obtained by biosorption in the catalytic oxidation of volatile organic compounds,” J Hazard Mater, vol. 192, no. 2, pp. 545–553, 2011.
- Cauvel, D. Brunel, F. Di Renzo, P. Moreau, and F. Fajula, “Functionalization of Y zeolites with organosilane reagents,” in Studies in Surface Science and Catalysis, vol. 94, Elsevier, 1995, pp. 286–293.
- W. Peng et al., “Grafting of R4N+-Bearing Organosilane on Kaolinite, Montmorillonite, and Zeolite for Simultaneous Adsorption of Ammonium and Nitrate,” Int J Environ Res Public Health, vol. 19, no. 19, p. 12562, 2022.
- R. E. Kukwa and S. E. Dann, “Grafted zeolites for the removal of metal cations from crude oil hydrotreatment extract,” Desalination Water Treat, vol. 153, pp. 136–144, Jun. 2019. [CrossRef]
- H. Sanaeepur, A. Kargari, and B. Nasernejad, “Aminosilane-functionalization of a nanoporous Y-type zeolite for application in a cellulose acetate based mixed matrix membrane for CO 2 separation,” RSC Adv, vol. 4, no. 109, pp. 63966–63976, 2014.
- S. C. Lu, T. Wichidit, T. Narkkun, K. L. Tung, K. Faungnawakij, and C. Klaysom, “Aminosilane-Functionalized Zeolite Y in Pebax Mixed Matrix Hollow Fiber Membranes for CO2/CH4 Separation,” Polymers (Basel), vol. 15, Jan. 2023. [CrossRef]
- B. Zhang, K. Zhang, Z. Duan, J. Zhu, and J. Gao, “Y Zeolites Modified by Organosilane for Toluene Adsorption under High Humidity Condition,” Am J Analyt Chem, vol. 14, pp. 451–466, 2023. [CrossRef]
- L. Yang et al., “Facilely Grafting Small Amine Molecules into Co-Exchanged Zeolite for Enhancing CO2 Adsorption,” Ind Eng Chem Res, vol. 63, pp. 5863–5870, Apr. 2024. [CrossRef]
- Y. Li et al., “Grafting of functional organic ligand on zeolite Y for efficient CH4/N2 separation,” Sep Purif Technol, vol. 354, p. 128719, 2025.
- C. Wang et al., “Preparation of amino functionalized hydrophobic zeolite and its adsorption properties for chromate and naphthalene,” Minerals, vol. 8, Apr. 2018. [CrossRef]
- M. S. Rahaman et al., “Hydrophobic functionalization of HY zeolites for efficient conversion of glycerol to solketal,” Appl Catal A Gen, vol. 592, Feb. 2020. [CrossRef]
- H.-T. Vu, F. M. Harth, and N. Wilde, “Silylated zeolites with enhanced hydrothermal stability for the aqueous-phase hydrogenation of levulinic acid to γ-valerolactone,” Front Chem, vol. 6, p. 143, 2018.
- C. Kim, H. S. Cho, S. Chang, S. J. Cho, and M. Choi, “An ethylenediamine-grafted y zeolite: A highly regenerable carbon dioxide adsorbent: Via temperature swing adsorption without urea formation,” Energy Environ Sci, vol. 9, pp. 1803–1811, May 2016. [CrossRef]
- Y. Guo, J. Qi, F. Wang, B. Fan, W. Hao, and R. Li, “Migration of Framework Aluminum and Tunable Acidity in Y Zeolite by Post-treatment,” Ind Eng Chem Res, vol. 62, no. 26, pp. 9951–9960, 2023.
- N. Khazipova, O. S. Travkina, M. R. Agliullin, I. N. Pavlova, B. I. Kutepov, and V. A. Dyakonov, “Modification of the Physicochemical Properties of High-Crystallinity Granular Y Zeolite by Steam Heating and Acid Treatment,” Petroleum Chemistry, vol. 61, pp. 284–291, 2021.
- M. Moosavifar and L. Fathyunes, “Influence of the post-synthesis method on the number and size of secondary mesoporous structure of NaY zeolite and its effect on catalyst loading. An efficient and eco-friendly catalyst for synthesis of xanthenes under conventional heating,” Journal of the Iranian Chemical Society, vol. 13, no. 11, pp. 2113–2120, 2016.
- S. Roy, A. Bhaskaran, and P. C. Meenu, “Microwave-assisted Synthesis of Porous Materials,” 2023.
- T. Le, T. Wang, A. V Ravindra, Y. Xuxiang, S. Ju, and L. Zhang, “Fast synthesis of submicron zeolite Y using microwave heating,” Kinetics and Catalysis, vol. 62, no. 3, pp. 436–444, 2021.
- T. Le, Q. Wang, B. Pan, A. V Ravindra, S. Ju, and J. Peng, “Process regulation of microwave intensified synthesis of Y-type zeolite,” Microporous and Mesoporous Materials, vol. 284, pp. 476–485, 2019.
- S. Abdulridha et al., “A comparative study on mesoporous Y zeolites prepared by hard-templating and post-synthetic treatment methods,” Appl Catal A Gen, vol. 612, p. 117986, 2021.
- M. Zhu, X. An, T. Gui, T. Wu, Y. Li, and X. Chen, “Effects of ion-exchange on the pervaporation performance and microstructure of NaY zeolite membrane,” Chin J Chem Eng, vol. 59, pp. 176–181, Jul. 2023. [CrossRef]
- H. Khan, A. S. Yerramilli, A. D’Oliveira, T. L. Alford, D. C. Boffito, and G. S. Patience, “Experimental methods in chemical engineering: X-ray diffraction spectroscopy—XRD,” Can J Chem Eng, vol. 98, no. 6, pp. 1255–1266, 2020.
- Salahudeen and A. S. Ahmed, “Synthesis of hexagonal zeolite Y from Kankara kaolin using a split technique,” J Incl Phenom Macrocycl Chem, vol. 87, pp. 149–156, 2017.
- N. Taufiqurrahmi, A. R. Mohamed, and S. Bhatia, “Nanocrystalline zeolite Y: synthesis and characterization,” in IOP Conference Series: Materials Science and Engineering, IOP Publishing, 2011, p. 012030.
- Ul-Hamid, A beginners’ guide to scanning electron microscopy, vol. 1. Springer, 2018.
- N. Kaur, “Transmission Electron Microscopy: A Powerful and Novel Scientific Technique with Nanoscale Resolution for Characterization of Materials,” in Microscopic Techniques for the Non-Expert, Springer, 2022, pp. 201–226.
- R. L. Volkov, V. N. Kukin, P. A. Kots, I. I. Ivanova, and N. I. Borgardt, “Complex pore structure of mesoporous zeolites: unambiguous TEM imaging using platinum tracking,” ChemPhysChem, vol. 21, no. 4, pp. 275–279, 2020.
- Huang et al., “Synthesis of the Y nanometer zeolites from fly ash and its adsorption models for aqueous Cs+ ions,” J Radioanal Nucl Chem, vol. 323, no. 1, pp. 65–72, 2020.
- F. S. Irwansyah et al., “How to read and determine the specific surface area of inorganic materials using the Brunauer-Emmett-Teller (BET) method,” ASEAN Journal of Science and Engineering, vol. 4, no. 1, pp. 61–70, 2024.
- M. Zheng, Y. Chu, Q. Wang, Y. Wang, J. Xu, and F. Deng, “Advanced solid-state NMR spectroscopy and its applications in zeolite chemistry,” Prog Nucl Magn Reson Spectrosc, vol. 140, pp. 1–41, 2024.
- S. Askarli et al., “Influence of Extra-Framework Aluminum Species on the Catalytic Properties of Acidic USY Zeolite in (Hydro) cracking Reactions,” ACS Catal, vol. 14, no. 18, pp. 13630–13639, 2024.
- M. Król, A. Koleżyński, and W. Mozgawa, “Vibrational spectra of zeolite Y as a function of ion exchange,” Molecules, vol. 26, no. 2, p. 342, 2021.
- Orlando et al., “A comprehensive review on Raman spectroscopy applications,” Chemosensors, vol. 9, no. 9, p. 262, 2021.
- H. T. Vu, M. Goepel, and R. Gläser, “Improving the hydrothermal stability of zeolite Y by La3+cation exchange as a catalyst for the aqueous-phase hydrogenation of levulinic acid,” RSC Adv, vol. 11, pp. 5568–5579, Jan. 2021. [CrossRef]
- M. Li et al., “Insight into the Key Role of Yttrium Modification for the Enhanced Hydrothermal Stability over Y-Cu-SSZ-39 Zeolite Catalysts,” Energy and Fuels, Dec. 2024. [CrossRef]
- L. Zhang, Y. Qin, X. Zhang, X. Gao, and L. Song, “Further Findings on the Stabilization Mechanism among Modified y Zeolite with Different Rare Earth Ions,” Ind Eng Chem Res, vol. 58, pp. 14016–14025, Aug. 2019. [CrossRef]
- C. Buttersack, A. König, and R. Gläser, “Stability of a highly dealuminated Y-zeolite in liquid aqueous media,” Microporous and Mesoporous Materials, vol. 281, pp. 148–160, Jun. 2019. [CrossRef]
- Z. Zhu et al., “Insight into tri-coordinated aluminum dependent catalytic properties of dealuminated Y zeolites in oxidative desulfurization,” Appl Catal B, vol. 288, p. 120022, 2021.
- L. Wang, Y. Jiang, J. Huang, and M. Hunger, “Relation between acidity and local structure of dealuminated zeolites y investigated by solid-state NMR spectroscopy,” in Chemeca 2011 (39th: 2011: Sydney, NSW), Engineers Australia Barton, ACT, 2011, pp. 550–558.
- S. Li et al., “Probing the spatial proximities among acid sites in dealuminated HY zeolite by solid-state NMR spectroscopy,” The Journal of Physical Chemistry C, vol. 112, no. 37, pp. 14486–14494, 2008.
- Z. Muhammad, A. Garba, Y. Ibrahim, and A. H. Birniwa, “Synthesis and characterization of zeolite sourced from rice husk lignocellulosic waste ash,” ChemSearch Journal, vol. 15, no. 1, pp. 80–85, 2024.
- F. H. Blindheim and J. Ruwoldt, “The effect of sample preparation techniques on lignin Fourier transform infrared spectroscopy,” Polymers (Basel), vol. 15, no. 13, p. 2901, 2023.
- K. Bahgaat, H. E.-S. Hassan, A. A. Melegy, and A. Abdel Karim, “Synthesis and characterization of zeolite-Y from natural clay of Wadi Hagul, Egypt,” Egypt J Chem, vol. 63, no. 10, pp. 3791–3800, 2020.
- H. Azzan, D. Danaci, C. Petit, and R. Pini, “Unary Adsorption Equilibria of Hydrogen, Nitrogen, and Carbon Dioxide on Y-Type Zeolites at Temperatures from 298 to 393 K and at Pressures up to 3 MPa,” J Chem Eng Data, vol. 68, no. 12, pp. 3512–3524, 2023.
- M. K. Singh and A. Singh, “Thermogravimetric analyzer,” in Characterization of Polymers and Fibres, Elsevier, 2022, pp. 223–240. [CrossRef]
- B. Bensafi, N. Chouat, A. Maziz, and F. Djafri, “Synthesis and Post-synthesis Modification of Zeolite Y for Improved Methanol Adsorption and Coke Formation Resistance,” Silicon, vol. 16, no. 15, pp. 5549–5561, 2024.
- M. Moreno-González, A. E. Palomares, M. Chiesa, M. Boronat, E. Giamello, and T. Blasco, “Evidence of a Cu2+–Alkane Interaction in Cu-Zeolite Catalysts Crucial for the Selective Catalytic Reduction of NO x with Hydrocarbons,” ACS Catal, vol. 7, no. 5, pp. 3501–3509, 2017.
- Brückner, “In situ electron paramagnetic resonance: A unique tool for analyzing structure–reactivity relationships in heterogeneous catalysis,” Chem Soc Rev, vol. 39, pp. 4673–4684, Nov. 2010. [CrossRef]
- V. A. Reyes Villegas et al., “Sonochemical post-synthesis modification of Y zeolite with iron species,” Mater Chem Phys, vol. 331, Feb. 2025. [CrossRef]
- C. Douvris, T. Vaughan, D. Bussan, G. Bartzas, and R. Thomas, “How ICP-OES changed the face of trace element analysis: Review of the global application landscape,” Dec. 2023, Elsevier B.V. [CrossRef]
- V. A. Reyes Villegas, J. I. De León Ramirez, L. Pérez-Cabrera, S. Pérez-Sicairos, J. R. Chávez-Méndez, and V. Petranovskii, “Analysis of catalytic sites in FeY zeolite prepared by sono-assisted exchange of iron (II) ions,” Microporous and Mesoporous Materials, vol. 380, Dec. 2024. [CrossRef]
- K. Argaw Shiferaw, J. M. Mathews, E. Yu, E. Y. Choi, and N. H. Tarte, “Sodium Methoxide/Zeolite-Supported Catalyst for Transesterification of Soybean Waste Cooking Oil for Biodiesel Production,” Inorganics (Basel), vol. 11, Apr. 2023. [CrossRef]
| Characterization Technique | Purpose | Typical Conditions | Example in Literature | Ref. |
| XRD | Determine crystalline structure and phase purity. | Ambient; powdered samples; X-rays | XRD was used to confirm the crystalline structure of synthesized hexagonal zeolite Y from Kankara kaolin. | [133,134] |
| SEM | Study surface morphology and particle size. | Vacuum; conductive coating required | SEM was used to analyze the microstructure of nanocrystalline zeolite Y. | [135,136] |
| TEM | Provide detailed internal structure and morphology at an atomic scale. | High vacuum; ultrathin samples | The mesopores of the zeolite Y (CBV 720) containing platinum tracking were visualized by TEM, including inside crystals and those that emerge at its surface. | [137,138] |
| BET | Determine surface area, pore volume, and pore size distribution. | Liquid nitrogen; under vacuum, gas adsorption |
The specific surface area of nano zeolite Y (NFA-Y) that was synthesized from fly ash was obtained by BET | [139,140] |
| NMR | Analyze local chemical environment | Ambient; solid-state or powdered samples; magnetic field | 27Al NMR showed steam calcination reduces framework aluminum, creating extra-framework aluminum that alters the acidity and catalytic behavior of USY. | [141,142] |
| Raman | Provide information on vibrational modes of the framework. | Ambient; laser excitation | Raman was utilized to analyze the vibrational spectra of zeolite Y after ion exchange with different cations, comparing computed spectra of optimized structures with experimental data. | [143,144] |
| Characterization Technique | Purpose | Typical Conditions | Example in Literature | Ref. |
| FTIR | Identify functional groups and framework vibrations. | Ambient; powdered or pellet samples | FTIR confirmed the successful synthesis of zeolite NaY from Rice husk ash by identifying the distinct functional groups, such as Si-O and Al-O stretches. | [152,153] |
| XRF | Determine elemental composition. | Ambient; solid or powdered samples | XRF measured the elemental content in zeolite Y synthesized from natural clay. | [154] |
| TGA | Study thermal stability and decomposition behavior. | commonly up to 1000°C, controlled atmosphere (air or inert) | The thermal stability of NaTMA-Y zeolite was quantified before and after activation utilizing TGA, which revealed a mass loss between (625-1050K) due to the decomposition of the TMA+ cations. | [155,156] |
| NH₃-TPD | Determines the acidity strength and distribution of acidic sites. | Variable temperatures; controlled ammonia gas flow | NH3-TPD analysis showed that modified zeolite Y via dealumination increased the concentration of strong acid sites. | [157] |
| EPR | Study paramagnetic species and defects. | Ambient or low temperatures; under magnetic field | EPR identified the formation of Cu2+ NO₃- species at room temperature in Cu-Y zeolites when used for the catalytic reduction of NOx with hydrocarbons. | [158,159] |
| ICP-OES | determining the elemental composition and metal loading | Ambient; sample digested in solution | The Si/Al ratio of modified zeolite Y with iron species was measured using ICP-OES. | [160,161] |
| DR UV-Vis | Analyze electronic transitions in metal-substituted zeolites. | Ambient; solid samples in diffuse reflectance mode | DR UV-Vis is used to study the electronic transitions in the FeY zeolite, helping to identify the presence and nature of iron species within the zeolite structure. | [162] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.





