Submitted:
23 December 2025
Posted:
24 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Feedstock Collection
2.3. Analytical Methods
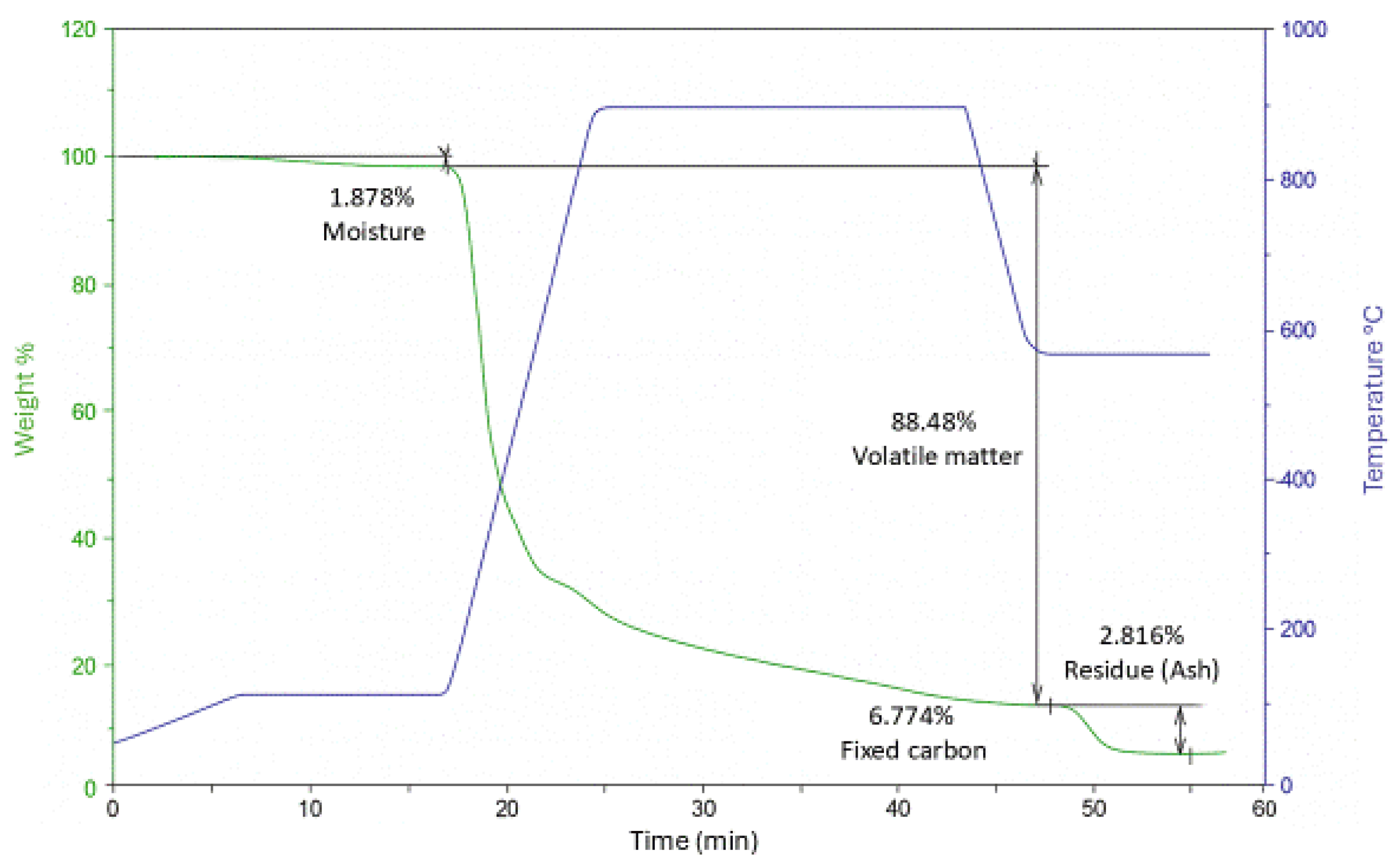
2.3.1. Dry Matter Determination and Thermogravimetric Analysis (TGA)
2.3.2. Elemental Analysis
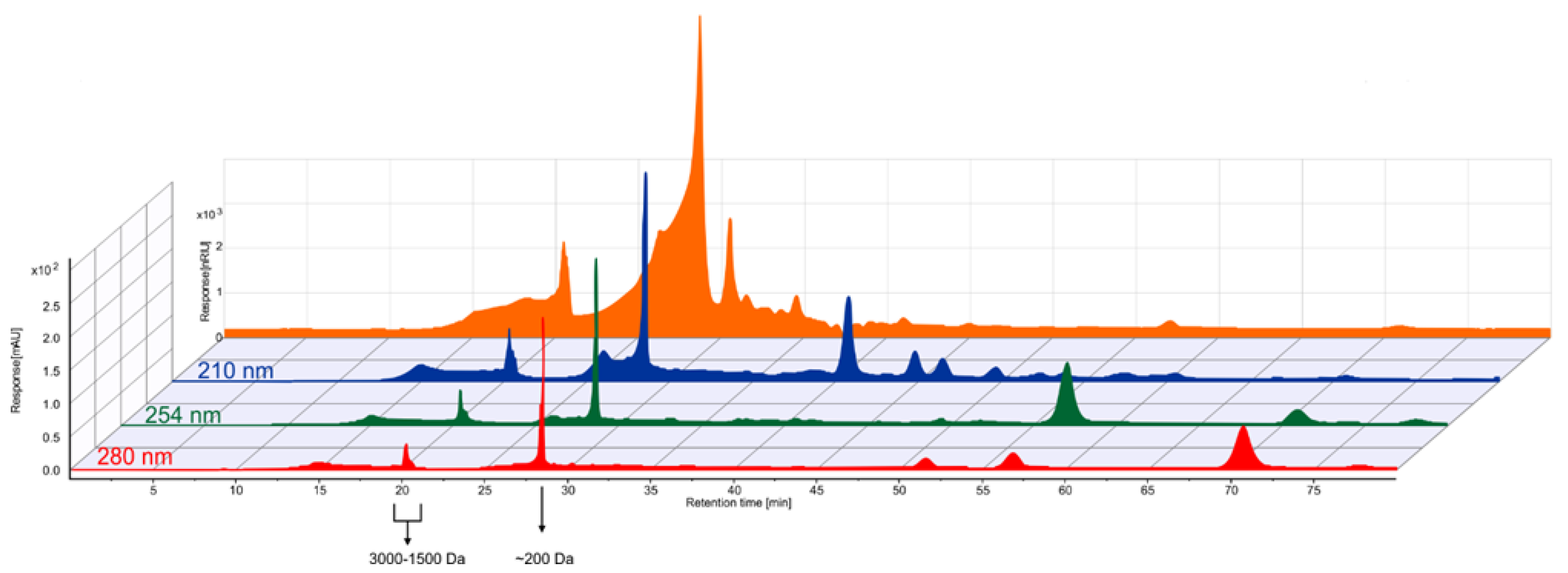
2.3.3. High-Performance Liquid Chromatography (HPLC)
2.3.4. Gas Chromatography-Mass Spectrometry (GC-MS)
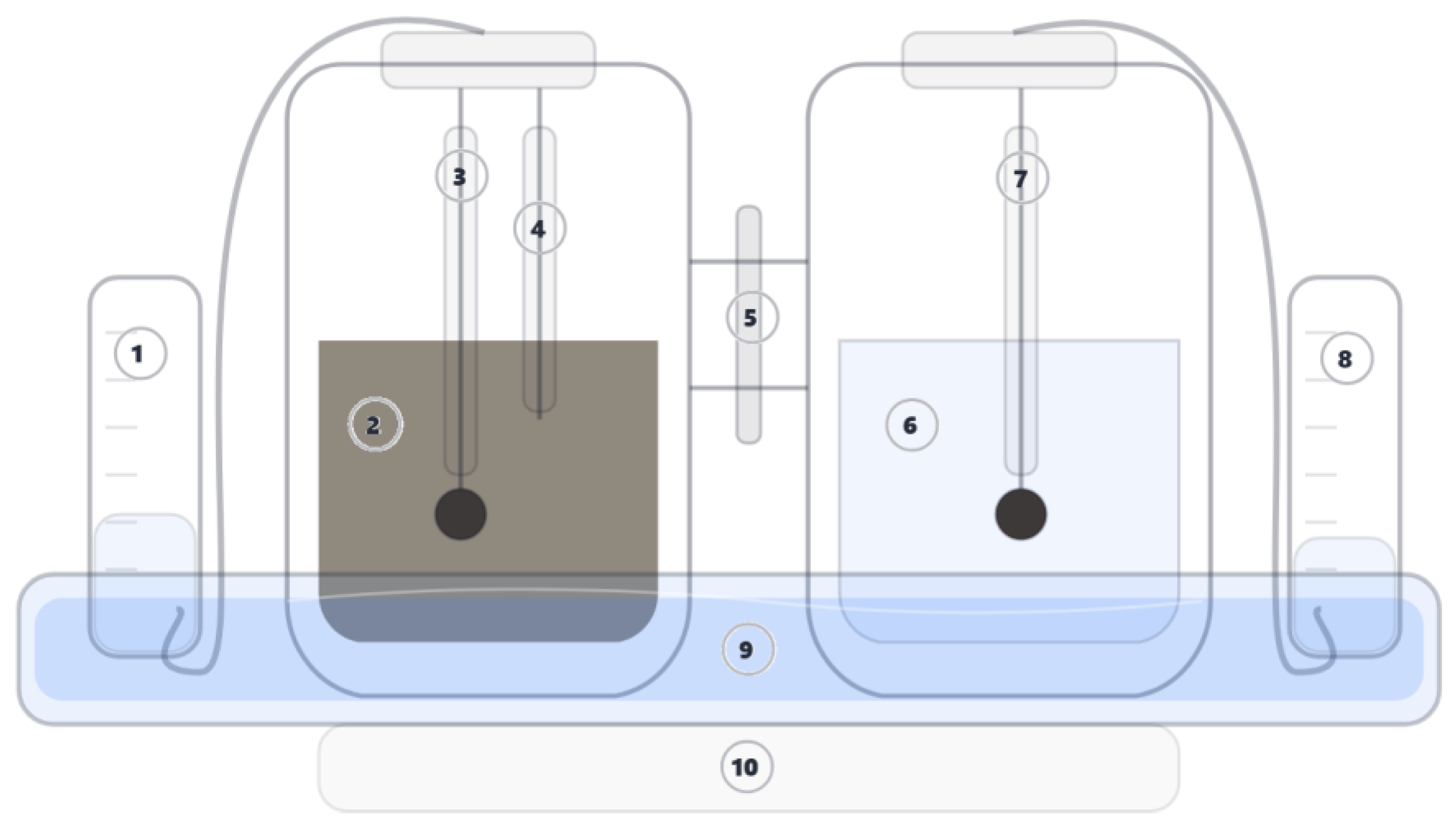
2.3.5. Electrochemical Measurements and Analysis (LSV, CA, CP, Bulk Electrolysis)
2.4. Nickel Oxide-Hydroxide on Nickel Foam Electrodeposition
2.5. Hydrogen Generation and Biomass Valorization
3. Results and Discussion
3.1. Biomass Byproduct Characterization
3.1.1. Dry Matter and Thermal Behavior
3.1.2. Elemental Composition
3.1.3. Analysis of Soluble Molecules and Molecular Weight Distribution
3.1.4. Volatile and Semi-Volatile Organic Compounds (GC-MS)
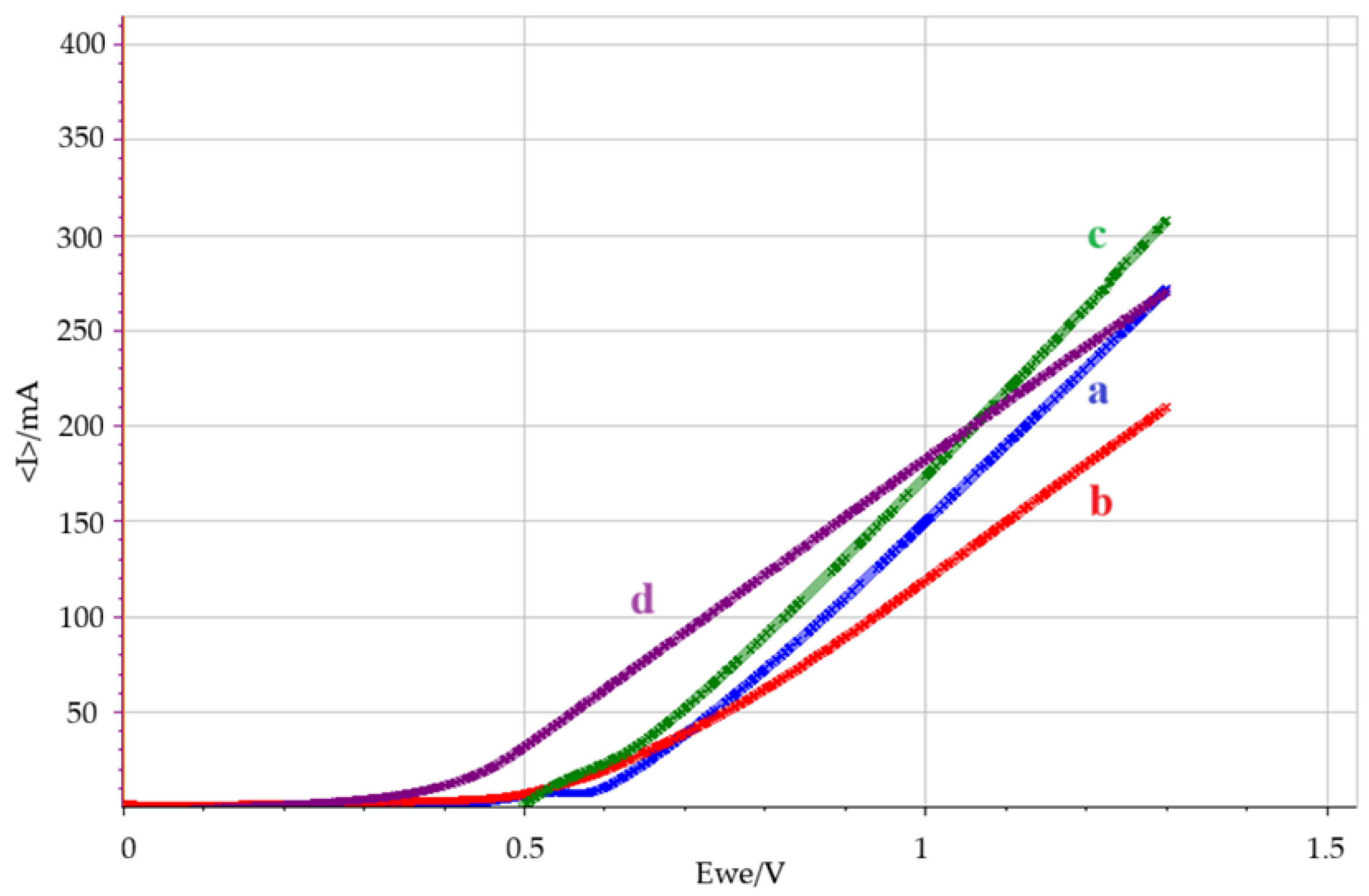
3.2. Polarization Curves
3.3. Tar Oxidation vs OER
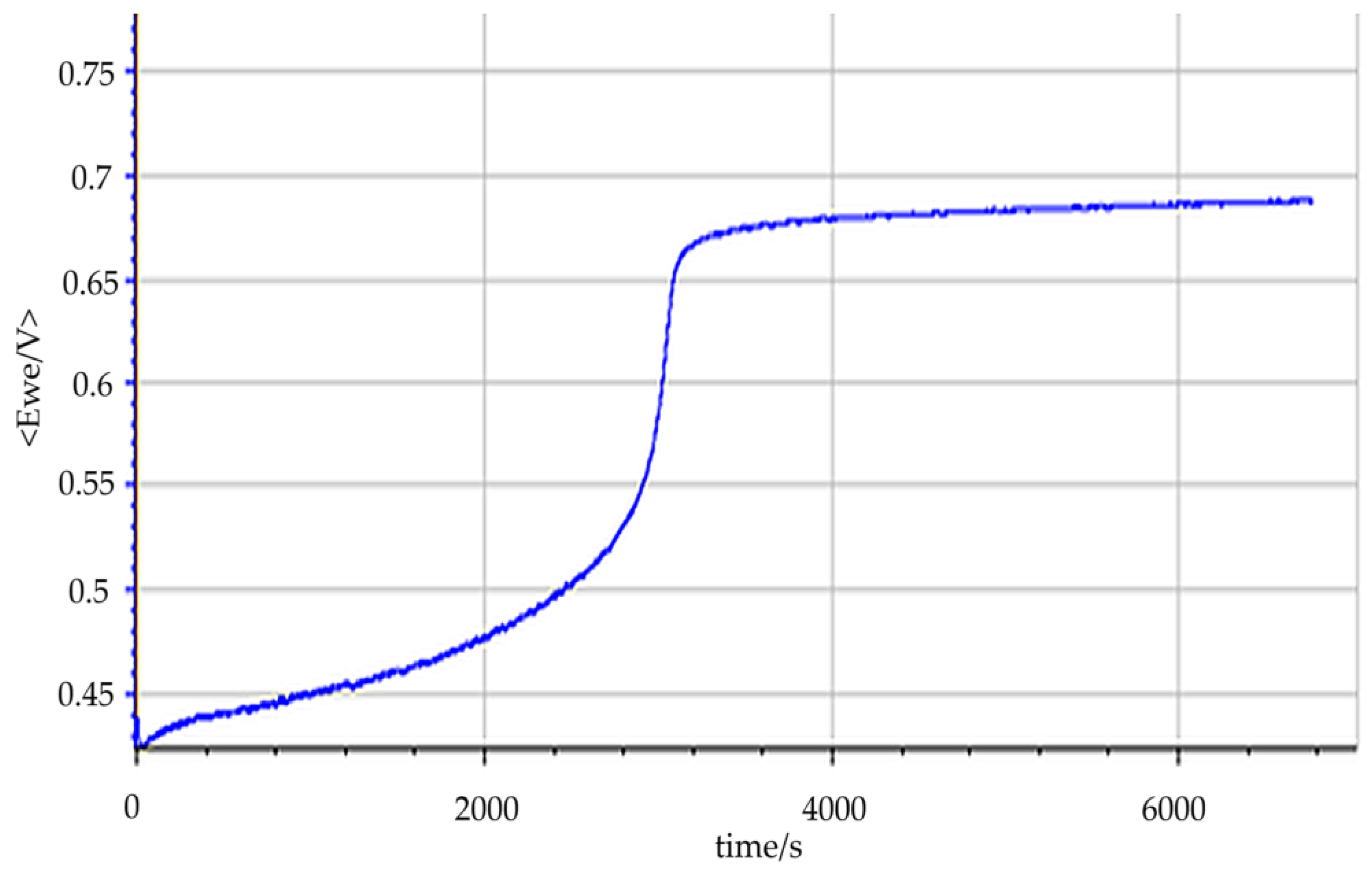
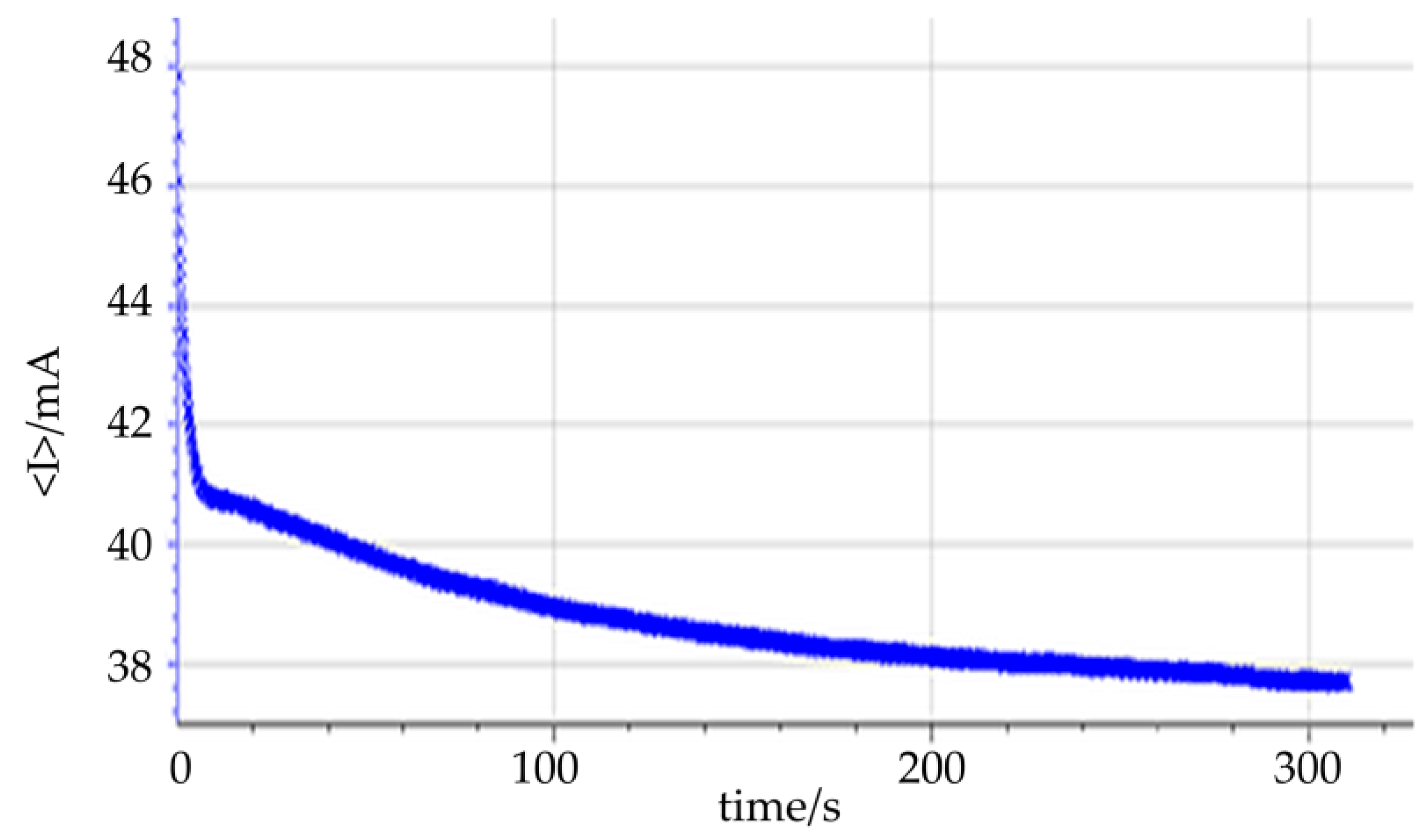
3.4. Hydrogen Production
3.5. Tar Electro-Reforming
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| AEM | Anion Exchange Membrane |
| CA | Chronoamperometry |
| CP | Chronopotentiometry |
| DAD | Diode Array Detector |
| DM | Dry Matter |
| EI | Electron Impact |
| FE | Faradaic Efficiency |
| FID | Flame Ionization Detector |
| GC | Gas Chromatography |
| GC-MS | Gas Chromatography–Mass Spectrometry |
| GFC | Gel Filtration Chromatography |
| HPLC | High-Performance Liquid Chromatography |
| LSV | Linear Sweep Voltammetry |
| MW | Molecular Weight |
| Ni(O)OH | Nickel Oxide-Hydroxide |
| OER | Oxygen Evolution Reaction |
| PEG | Polyethylene Glycol |
| RID | Refractive Index Detector |
| SVOCs | Semi-Volatile Organic Compounds |
| TCD | Thermal Conductivity Detector |
| TGA | Thermogravimetric Analysis |
| VOCs | Volatile Organic Compounds |
References
- Giakoumakis, G.; Sidiras, D. Production and Storage of Hydrogen from Biomass and Other Sources: Technologies and Policies. Energies 2025, 18, 650. [Google Scholar] [CrossRef]
- Suleman, F.; Dincer, I.; Agelin-Chaab, M. Environmental Impact Assessment and Comparison of Some Hydrogen Production Options. Int J Hydrogen Energy 2015, 40, 6976–6987. [Google Scholar] [CrossRef]
- Skjærseth, J.B. Towards a European Green Deal: The Evolution of EU Climate and Energy Policy Mixes. Int Environ Agreem 2021, 21, 25–41. [Google Scholar] [CrossRef]
- Obiora, N.K.; Ujah, C.O.; Asadu, C.O.; Kolawole, F.O.; Ekwueme, B.N. Production of Hydrogen Energy from Biomass: Prospects and Challenges. Green Technologies and Sustainability 2024, 2, 100100. [Google Scholar] [CrossRef]
- Liu, W.; Liu, C.; Gogoi, P.; Deng, Y. Overview of Biomass Conversion to Electricity and Hydrogen and Recent Developments in Low-Temperature Electrochemical Approaches. Engineering 2020, 6, 1351–1363. [Google Scholar] [CrossRef]
- Valderrama Rios, M.L.; González, A.M.; Lora, E.E.S.; Almazán del Olmo, O.A. Reduction of Tar Generated during Biomass Gasification: A Review. Biomass Bioenergy 2018, 108, 345–370. [Google Scholar] [CrossRef]
- Cortazar, M.; Santamaria, L.; Lopez, G.; Alvarez, J.; Zhang, L.; Wang, R.; Bi, X.; Olazar, M. A Comprehensive Review of Primary Strategies for Tar Removal in Biomass Gasification. Energy Convers Manag 2023, 276, 116496. [Google Scholar] [CrossRef]
- Melis, N.; Mais, L.; Mascia, M.; Vacca, A. Assessing an Electrochemical Process for the Treatment of Tar-Containing Wastewater with Hydrogen Recovery. Chemical Engineering Journal 2024, 500, 156736. [Google Scholar] [CrossRef]
- Tian, Z.; Lu, Y.; Wang, J.; Shu, R.; Wang, C.; Chen, Y. Advances in Hydrogen Production by Aqueous Phase Reforming of Biomass Oxygenated Derivatives. Fuel 2024, 357, 129691. [Google Scholar] [CrossRef]
- Miller, H.A.; Bouzek, K.; Hnat, J.; Loos, S.; Bernäcker, C.I.; Weißgärber, T.; Röntzsch, L.; Meier-Haack, J. Green Hydrogen from Anion Exchange Membrane Water Electrolysis: A Review of Recent Developments in Critical Materials and Operating Conditions. Sustain Energy Fuels 2020, 4, 2114–2133. [Google Scholar] [CrossRef]
- Dolle, C.; Neha, N.; Coutanceau, C. Electrochemical Hydrogen Production from Biomass. Curr Opin Electrochem 2022, 31, 100841. [Google Scholar] [CrossRef]
- Zhao, H.; Lu, D.; Wang, J.; Tu, W.; Wu, D.; Koh, S.W.; Gao, P.; Xu, Z.J.; Deng, S.; Zhou, Y.; et al. Raw Biomass Electroreforming Coupled to Green Hydrogen Generation. Nature Communications 2021, 1 2021(12), 2008. [Google Scholar] [CrossRef] [PubMed]
- Grasset, C.; Groeneveld, M.; Tranvik, L.J.; Robertson, L.P.; Hawkes, J.A. Hydrophilic Species Are the Most Biodegradable Components of Freshwater Dissolved Organic Matter. Environ Sci Technol 2023, 57, 13463–13472. [Google Scholar] [CrossRef]
- Shi, K.; Si, D.; Teng, X.; Chen, L.; Shi, J. Enhanced Electrocatalytic Glycerol Oxidation on CuCoN0.6/CP at Significantly Reduced Potentials. Chinese Journal of Catalysis 2023, 53, 143–152. [Google Scholar] [CrossRef]
- Bolarinwa, M.O.; Zakari, R.S.B.; Talib, S.H.; Stephen, S.; Polychronopoulou, K.; Elkadi, M.; Anjum, D.H. Electrochemical Oxidation of Glycerol to Glyceric Acid Using Iridium-Vanadium (IrV) Dual Atom Catalysts on Graphene Variants: Experimental and Computational Approach. Appl Surf Sci 2025, 688, 162457. [Google Scholar] [CrossRef]
- Velázquez-Hernández, I.; Álvarez-López, A.; Álvarez-Contreras, L.; Guerra-Balcázar, M.; Arjona, N. Electrocatalytic Oxidation of Crude Glycerol from the Biodiesel Production on Pd-M (M = Ir, Ru or Pt) Sub-10 Nm Nanomaterials. Appl Surf Sci 2021, 545. [Google Scholar] [CrossRef]
- White, J.; Peters, L.; Martín-Yerga, D.; Terekhina, I.; Anil, A.; Lundberg, H.; Johnsson, M.; Salazar-Alvarez, G.; Henriksson, G.; Cornell, A. Glycerol Electrooxidation at Industrially Relevant Current Densities Using Electrodeposited PdNi/Nifoam Catalysts in Aerated Alkaline Media. J Electrochem Soc 2023, 170, 086504. [Google Scholar] [CrossRef]
- Wulf, C.; Linssen, J.; Zapp, P. Power-to-Gas-Concepts, Demonstration, and Prospects. In Hydrogen Supply Chain: Design, Deployment and Operation; 2018; pp. 309–345. [Google Scholar] [CrossRef]
- Tichler, R.; Bauer, S.; Böhm, H. Power-to-Gas. In Storing Energy: with Special Reference to Renewable Energy Sources; 2022; pp. 595–612. [Google Scholar] [CrossRef]
- Cigolotti, V.; Genovese, M.; Piraino, F.; Fragiacomo, P. Applications | Overview of Energy Storage Systems. Encyclopedia of Electrochemical Power Sources: Volume 1-7, Second Edition 2024, 7, V7:47–V7:59. [Google Scholar] [CrossRef]
- Cerone, N.; Zimbardi, F. Gasification of Agroresidues for Syngas Production. Energies 2018, 11, 1280. [Google Scholar] [CrossRef]
- Srinivasan, V.; Weidner, J.W. An Electrochemical Route for Making Porous Nickel Oxide Electrochemical Capacitors. J Electrochem Soc 1997, 144, L210–L213. [Google Scholar] [CrossRef]
- Wu, M.S.; Huang, Y.A.; Yang, C.H.; Jow, J.J. Electrodeposition of Nanoporous Nickel Oxide Film for Electrochemical Capacitors. Int J Hydrogen Energy 2007, 32, 4153–4159. [Google Scholar] [CrossRef]
- Abawalo, M.; Pikoń, K.; Landrat, M.; Ścierski, W. Hydrogen Production from Biowaste: A Systematic Review of Conversion Technologies, Environmental Impacts, and Future Perspectives. Energies (Basel) 2025, 18. [Google Scholar] [CrossRef]
- Mustapha, S.I.; Anekwe, I.M.S.; Akpasi, S.O.; Muritala, K.B.; Tetteh, E.K.; Joel, A.S.; Isa, Y.M. Biomass Conversion for Sustainable Hydrogen Generation: A Comprehensive Review. Fuel Processing Technology 2025, 272, 108210. [Google Scholar] [CrossRef]
- Sun, Y.; Miao, J.; Fan, X.; Zhang, K.; Zhang, T. Recent Progress in Electrochemical Conversion from Biomass Derivatives into High-Value-Added Chemicals. Small Struct 2024, 5, 2300576. [Google Scholar] [CrossRef]
- Wang, Z.; Li, B.; Yang, T.; Wang, J.; Li, R. Valorization to Hydrogen of Aqueous Phase from Biomass Hydrothermal Liquefaction Via Aqueous Phase Reforming Over Ni/Ac Catalyst. 2024. [Google Scholar] [CrossRef]

| Unit | Value | ± (sd) | ||
|---|---|---|---|---|
| DM (80 °C) | % | 13.6 | 0.1 | |
| Carbon | % | 53.39 | 0.04 | |
| Hydrogen | % | 5.4 | 0.1 | |
| Nitrogen | % | 0.91 | 0.02 | |
| Sulfur | % | < 0.02 | ||
| Unit | Value | ± (sd) | ||
|---|---|---|---|---|
| DM (80 °C) | % | 13.6 | 0.1 | |
| Acetic acid | g/kg | 114 | 3 | |
| Formic acid | g/kg | <0.003 | ||
| 2-furaldehyde | g/kg | 0.95 | 0.04 | |
| 5-hydroxymethylfurfural | g/kg | 0.36 | 0.02 | |
| Compound | Initial concentration | Final concentration | Conversion |
|---|---|---|---|
| g/kg | g/kg | % | |
| 1-Hydroxy-2-butanone | 0.58 | 0 | 100 |
| 3-Furaldehyde | 0.69 | 0 | 100 |
| 2-Propanone, 1-(acetyloxy)- | 0.16 | 0 | 100 |
| 1H-Imidazole-4-carboxaldehyde | 0.09 | 0 | 100 |
| 1H-1,2,4-Triazole, 1-vinyl- | 0.09 | 0 | 100 |
| 2(5H)-Furanone | 0.35 | 0 | 100 |
| Butyrolactone | 0.38 | 0 | 100 |
| 2-Cyclopenten-1-one, 2-hydroxy- | 0.1 | 0 | 100 |
| 2-Cyclopenten-1-one, 3-methyl- | 0.18 | 0 | 100 |
| Phenol | 0.13 | 0 | 100 |
| 2-Cyclopenten-1-one, 2-hydroxy-3-methyl- | 0.90 | 0.47 | 48 |
| 1-Propanone, 1-cyclopropyl- | 0.07 | 0 | 100 |
| Propanoic acid, 2-methyl-, anhydride | 0.2 | 0 | 100 |
| Phenol, 2-methoxy- | 0.52 | 0.07 | 86 |
| 2-Cyclopenten-1-one, 3-ethyl-2-hydroxy- | 0.12 | 0 | 100 |
| Catechol | 2.09 | 0.78 | 73 |
| Creosol | 0.55 | 0 | 100 |
| Naphthalene | 0.22 | 0 | 100 |
| 1,2-Benzenediol, 3-methyl- | 0.29 | 0 | 100 |
| 1,2-Benzenediol, 3-methoxy- | 0.13 | 0 | 100 |
| 1,2,4,5-Tetrazine | 0.17 | 0 | 100 |
| 1,2-Benzenediol, 4-methyl- | 0.59 | 0 | 100 |
| 2,3-Dimethoxyphenol | 0.18 | 0 | 100 |
| Vanillin | 0.37 | 0 | 100 |
| Ethanone, 1-(3-hydroxy-4-methoxyphenyl)- | 0.26 | 0 | 100 |
| b-D-Glucopyranose, 1,6-anhydro- | 0.24 | 0 | 100 |
| 2-Propanone, 1-(4-hydroxy-3-methoxyphenyl)- | 0.44 | 0.24 | 45 |
| Benzenepropanol, 4-hydroxy-3-methoxy- | 0.62 | 0.42 | 32 |
| 2-Methoxy-5-methylphenol | 0.29 | 0 | 100 |
| 1,4:3,6-Dianhydro-α-d-glucopyranose | 0.18 | 0.13 | 29 |
| Benzene, 1,3-bis(1,1-dimethylethyl)- | 0.08 | 0.07 | 6 |
| Hydroquinone | 0.3 | 0.19 | 36 |
| Phenol, 2,6-dimethoxy- | 0.21 | 0.11 | 49 |
| Apocynin | 0.47 | 0.27 | 42 |
| 2H-1-Benzopyran-2-one, 3,4-dihydro-6-hydroxy- | 0.31 | 0 | 100 |
| TOTAL | 12.7 | 2.75 | 78 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




