Submitted:
22 December 2025
Posted:
24 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
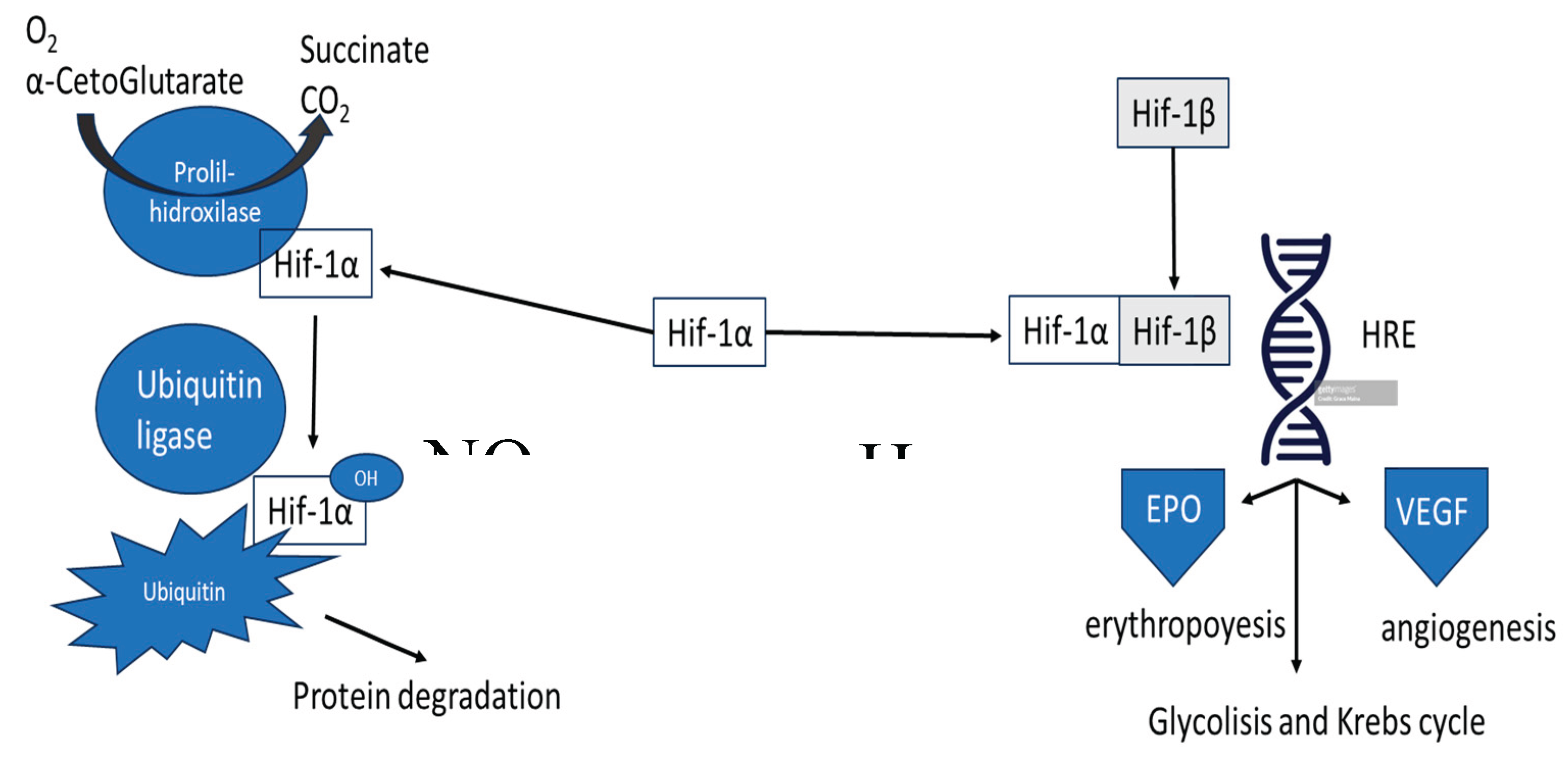
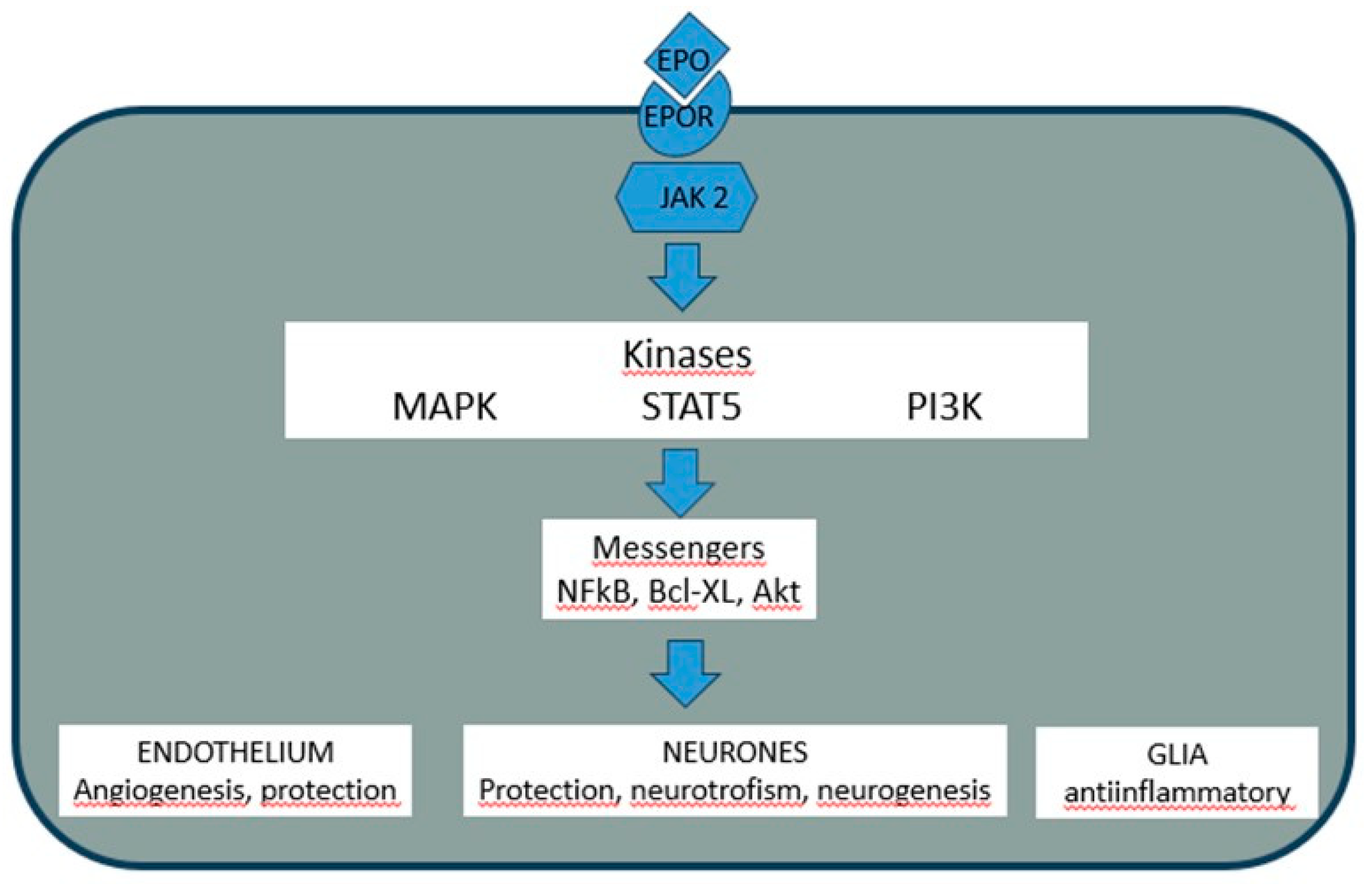
2. Erythropoietin: From Oxygen Sensing to Pleiotropic Signaling
3. Molecular Biology of EPO and Its Central Nervous System Expression
4. EPO and Neuroprotection: An Established yet Limited Frontier
5. EPO and Neuroplasticity: Introducing a Strategy for Tag-Guided Circuit Rewiring
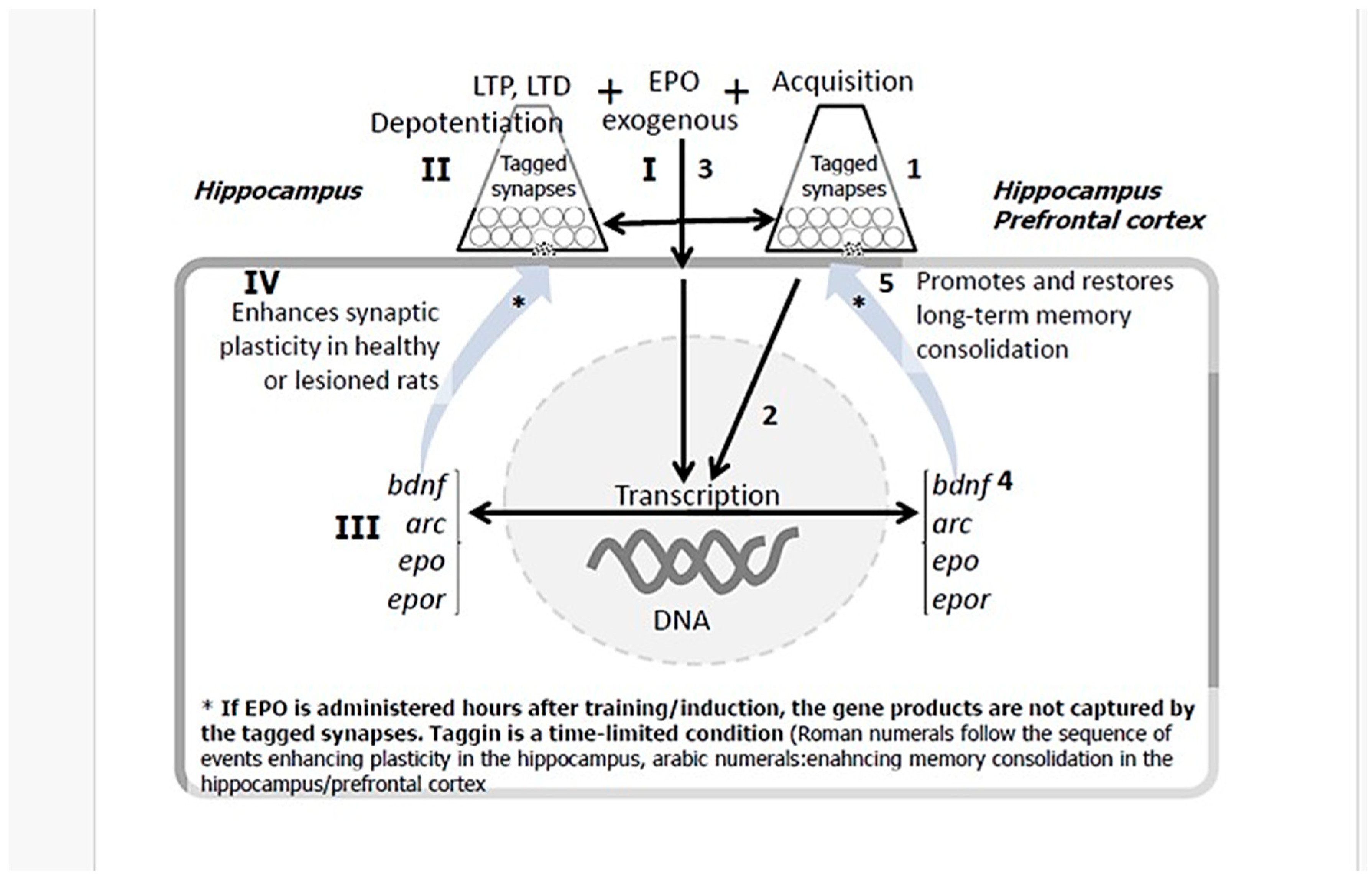
6. EPO Promotes Spatial Memory Recovery and Extends Memory Duration
7. EPO Expands the Boundaries of Synaptic Plasticity
8. EPO Induces Molecular Mediators of Plasticity in Memory Circuits
9. Endogenous EPO Signaling is an Activity-Dependent Component of Memory Formation
10. The Promise of EPO in Neuropsychiatry
11. Final Considerations and Conclusions
Author Contributions
Data Availability Statement
Conflicts of Interest
References
- Ramón y Cajal S. Degeneration and Regeneration of the Nervous System. New York: Harper Press; 1969.
- Bergado-Rosado JA, Almaguer-Melian W. [Cellular mechanisms of neuroplasticity]. Rev. Neurol. 2000; 31: 1074-95. PMID: 11190877.
- Rosenzweig MR, Bennett EL. Psychobiology of plasticity: Effects of training and experience on brain and behavior. Behav. Brain Res. 1996;78: 57-65. [CrossRef]
- Flexner JB, Flexner LB, Stellar E, De La HABA, Roberts RB. Inhibition of protein synthesis in brain and learning and memory following puromycin. J. Neurochem. 1962; 9:595-605. [CrossRef]
- Flexner LB, Flexner JB, Stellar E. Memory and cerebral protein synthesis in mice as affected by graded amounts of puromycin. Exp. Neurol. 1965; 13: 264-72. [CrossRef]
- Nottebohm F. Neuronal replacement in adulthood. Ann. N.Y. Acad. Sci. 1985; 457. [CrossRef]
- Nottebohm F. From bird song to neurogenesis. Sci. Am. 1989;260(2):74-9. [CrossRef]
- Bliss TV, Lomo T. Long-lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path. J. Physiol.(Lond). 1973; 232:331-56. [CrossRef]
- Bliss TV, Gardner-Medwin AR. Long-lasting potentiation of synaptic transmission in the dentate area of the unanaestetized rabbit following stimulation of the perforant path. J. Physiol. (Lond). 1973; 232:357-74. [CrossRef]
- Levy WB, Steward O. Synapses as associative memory elements in the hippocampal formation. Brain Res. 1979; 175:233-45. [CrossRef]
- Teyler TJ, DiScenna P. Long-term potentiation as a candidate mnemonic device. Brain Res. 1984; 319:15-28. [CrossRef]
- Krug M, Loessner B, Ott T. Anisomycin blocks the late phase of long-term potentiation in the dentate gyrus of freely moving rats. Brain Res.Bull. 1984; 13:39-42. [CrossRef]
- Loessner B, Schweigert C, Pchalek V, Krug M, Frey S, Matthies H. Training- and LTP-induced changes of protein synthesis in rat hippocampus. Neurosci. Suppl. 1987; 22:9512.
- Frey U, Krug M, Reymann KG, Matthies H. Anisomycin, an inhibitor of protein synthesis, blocks late phases of LTP phenomena in the hippocampal CA1 region in vitro. Brain Res. 1988; 452:57-65. [CrossRef]
- Krug M, Ruethrich H, Bergado J. The nootropic substance methylglucamine orotate prolongs both, postconditioning potentiation and posttetanic LTP in the dentate gyrus of freely moving rats. Act.Nerv.Sup.(Prague). 1988;30(3):232-3.
- Dąbrowski J, Czajka A, Zielińska-Turek J, Jaroszyński J, Furtak-Niczyporuk M, Mela A, et al. Brain Functional Reserve in the Context of Neuroplasticity after Stroke. Neural Plast. 2019; 2019:9708905. [CrossRef]
- Wadan AS, Raza ML, Moradikor N. Synaptic modulation by coffee compounds: Insights into neural plasticity. Prog Brain Res. 2024; 289:181-91. [CrossRef]
- Colucci-D’Amato L, Speranza L, Volpicelli F. Neurotrophic Factor BDNF, Physiological Functions and Therapeutic Potential in Depression, Neurodegeneration and Brain Cancer. Int. J. Mol. Sci. 2020;21(20). [CrossRef]
- Haase VH. Regulation of erythropoiesis by hypoxia-inducible factors. Blood Rev. 2013; 27:41-53. [CrossRef]
- Noguchi CT, Asavaritikrai P, Teng R, Jia Y. Role of erythropoietin in the brain. Crit. Rev. Oncol. Hematol. 2007; 64:159-71. [CrossRef]
- Frey U, Morris RGM. Synaptic tagging: implications for late maintenance of hippocampal long-term potentiation. Trends Neurosci. 1998; 21:181-8. [CrossRef]
- Frey U, Morris RGM. Synaptic tagging and long-term potentiation. Nature. 1997;385(6616):533-6. [CrossRef]
- Seidenbecher T, Reymann KG, Balschun D. A post-tetanic time window for the reinforcement of long-term potentiation by appetitive and aversive stimuli. Proc. Natl. Acad. Sci. USA. 1997; 94: 1494-9. [CrossRef]
- Masuda S, Nagao M, Takahata K, Konishi Y, Gallyas F, Tabira T, et al. Functional Erythropoietin Receptor of the Cells with Neural Characteristics. Comparison with receptor properties of erythroid cells. J. Biol. Chem. 1993;268(15):11208-16.
- Morishita E, Masuda S, Nagao M, Yasuda Y, Sasaki R. Erythropoietin receptor is expressed in rat hippocampal and cerebral cortical neurons, and erythropoietin prevents in vitro glutamate-induced neuronal death. Neuroscience. 1997;76: 105-16. [CrossRef]
- Sargin D, El-Kordi A, Agarwal A, Muller M, Wojcik SM, Hassouna I, et al. Expression of constitutively active erythropoietin receptor in pyramidal neurons of cortex and hippocampus boosts higher cognitive functions in mice. BMCBiol. 2011; 9: 27. [CrossRef]
- Miyake T, Kung CK, Goldwasser E. Purification of human erythropoietin. J. Biol. Chem. 1977; 252: 5558-64.
- Suresh S, Rajvanshi PK, Noguchi CT. The Many Facets of Erythropoietin Physiologic and Metabolic Response. Sci. Translat. Med. 2019; 10:1534. [CrossRef]
- Tan CC, Eckardt K, Firth JD, Ratcliffe PJ. Feedback modulation of renal and hepatic erythropoietin mRNA in response to graded anemia and hypoxia. Am. J. Physiol. 1992;263: F474-81. [CrossRef]
- Konishi Y, Chui DH, Hirose H, Kunishita H, Tabira T. Trophic effect of erythropoietin and other hematopoietic factors on central cholinergic neurons in vivo and in vivo. Brain Res. 1993; 609:29-35. [CrossRef]
- Shingo T, Sorokan ST, Shimazaki T, Weiss S. Erythropoietin regulates the in vitro and in vivo production of neuronal progenitors by mammalian forebrain neural stem cells. J. Neurosci. 2001; 21: 9733-43. [CrossRef]
- Sirén AL, Knerlich F, Poser W, Gleiter CH, Brück W, Ehrenreich H. Erythropoietin and erythropoietin receptor in human ischemic/hypoxic brain. Acta Neuropathol. 2001; 101: 271-6. [CrossRef]
- Kumral A, Uysal N, Tugyan K, Sonmez A, Yilmaz O, Gokmen N, et al. Erythropoietin improves long-term spatial memory deficits and brain injury following neonatal hypoxia-ischemia in rats. Behav. Brain Res. 2004;153(1):77-86. [CrossRef]
- Brines M, Cerami A. Emerging biological roles for erythropoietin in the nervous system. Nature. 2005; 6:484-94. [CrossRef]
- Chen J, Chopp M. Neurorestorative treatment of stroke: cell and pharmacological approaches. NeuroRx. 2006;3: 466-73. [CrossRef]
- Adamcio B, Sargin D, Stradomska A, Medrihan L, Gertler C, Theis F, et al. Erythropoietin enhances hippocampal long-term potentiation and memory. BMC Biol. 2008; 6:37. [CrossRef]
- Bartels C, Spate K, Krampe H, Ehrenreich H. Recombinant Human Erythropoietin: Novel Strategies for Neuroprotective/Neuro-regenerative Treatment of Multiple Sclerosis. Ther. Adv. Neurol. Disord. 2008; 1:193-206. [CrossRef]
- Ehrenreich H, Bartels C, Sargin D, Stawicki S, Krampe H. Recombinant human erythropoietin in the treatment of human brain disease: focus on cognition. J. Ren. Nutr. 2008; 18:146-53. [CrossRef]
- Jing M, Shingo T, Yasuhara T, Kondo A, Morimoto T, Wang F, et al. The combined therapy of intrahippocampal transplantation of adult neural stem cells and intraventricular erythropoietin-infusion ameliorates spontaneous recurrent seizures by suppression of abnormal mossy fiber sprouting. Brain Res. 2009; 1295:203-17. [CrossRef]
- Kamal A, Al ST, Ramakers G. Erythropoietin decreases the excitatory neurotransmitter release probability and enhances synaptic plasticity in mice hippocampal slices. Brain Res. 2011; 1410:33-7. [CrossRef]
- Sedriep S, Xia X, Marotta F, Zhou L, Yadav H, Yang H, et al. Beneficial nutraceutical modulation of cerebral erythropoietin expression and oxidative stress: an experimental study. J. Biol. Regul. Homeost. Agents. 2011;25(2):187-94. PMID: 21880207.
- Arabpoor Z, Hamidi G, Rashidi B, Shabrang M, Alaei H, Sharifi MR, et al. Erythropoietin improves neuronal proliferation in dentate gyrus of hippocampal formation in an animal model of Alzheimer’s disease. Adv. Biomed. Res. 2012; 1:50. [CrossRef]
- Mengozzi M, Cervellini I, Villa P, Erbayraktar Z, Gokmen N, Yilmaz O, et al. Erythropoietin-induced changes in brain gene expression reveal induction of synaptic plasticity genes in experimental stroke. Proc. Natl. Acad. Sci. USA. 2012; 109:9617-22. [CrossRef]
- Wobrock T, Hasan A, Falkai P. Innovative treatment approaches in schizophrenia enhancing neuroplasticity: aerobic exercise, erythropoetin and repetitive transcranial magnetic stimulation. Curr. Pharm. Biotechnol. 2012; 13:1595-605. [CrossRef]
- Esmaeili TP, Moosavi SM, Shabani M, Haghani M. Erythropoietin improves synaptic plasticity and memory deficits by decrease of the neurotransmitter release probability in the rat model of Alzheimer’s disease. Pharmacol. Biochem. Behav. 2015; 130:15-21. [CrossRef]
- Miskowiak KW, Petersen JZ, Macoveanu J, Ysbæk-Nielsen AT, Lindegaard IA, Cramer K, et al. Effect of erythropoietin on cognitive side-effects of electroconvulsive therapy in depression: A randomized, double-blind, placebo-controlled trial. Europ. Neuropsychopharmacol. 2024; 79:38-48. [CrossRef]
- Sakanaka M, Wen TC, Matsuda S, Masuda S, Morishita E, Nagao M, et al. In vivo evidence that erythropoietin protects neurons from ischemic damage. Proc. Natl. Acad. Sci. USA. 1998; 95:4635-40. [CrossRef]
- Li SJ, Cui KF, Fu JJ, Fu XJ, Gao YF, Zhang D, et al. EPO promotes axonal sprouting via upregulating GDF10. Genet. Mol. Biol. 2019; 711:134412. [CrossRef]
- Ben Attia M, N’Gouemo P, Belaidi M, Rondouin G, Chicheportiche R. Kindling and electrode effects on the benzodiazepine receptors density of olfactory bulb and hippocampus after olfactory bulb kindling. Neurosci. Lett. 1992; 143:74-8. [CrossRef]
- Marti HH, Gassmann M, Wenger RH, Kvietikova I, Morganti-Kossmann MC, Kossmann T, et al. Detection of erythropoietin in human liquor: intrinsic erythropoietin production in the brain. Kidney internat. 1997;51:416-8. [CrossRef]
- Peng W, Xing Z, Yang J, Wang Y, Wang W, Huang W. The efficacy of erythropoietin in treating experimental traumatic brain injury: a systematic review of controlled trials in animal models. J. Neurosurg. 2014; 121: 653-64. [CrossRef]
- Blixt J, Song Y, Wanecek M, Gunnarson E. EPO has multiple positive effects on astrocytes in an experimental model of ischemia. Brain Res. 2023; 1802:148207. [CrossRef]
- Xiong Y, Zhang Y, Mahmood A, Meng Y, Qu C, Chopp M. Erythropoietin mediates neurobehavioral recovery and neurovascular remodeling following traumatic brain injury in rats by increasing expression of vascular endothelial growth factor. Translat. Stroke Res. 2011; 2:619-32. [CrossRef]
- Ureña-Guerrero ME, Castañeda-Cabral JL, Rivera-Cervantes MC, Macias-Velez RJ, Jarero-Basulto JJ, Gudiño-Cabrera G, et al. Neuroprotective and Neurorestorative Effects of Epo and VEGF: Perspectives for New Therapeutic Approaches to Neurological Diseases. Curr. Pharmaceut. Design. 2020;2 6:1263-76. [CrossRef]
- Demers EJ, McPherson RJ, Juul SE. Erythropoietin protects dopaminergic neurons and improves neurobehavioral outcomes in juvenile rats after neonatal hypoxia-ischemia. Pediat. Res. 2005; 58:297-301. [CrossRef]
- Xu Y, Huang L, Han J, Zhou Y. Effects of EPO combined with mild hypothermia on oxidative stress and neuroprotection in neonates with hypoxic-ischemic encephalopathy. Cell. Mol. Biol. (Noisy-le-Grand). 2022; 68:36-45. [CrossRef]
- Lai YF, Lin TY. Erythropoietin in Optic Neuropathies: Current Future Strategies for Optic Nerve Protection and Repair. 2022; 23(13). [CrossRef]
- Boesch S, Indelicato E. Erythropoietin and Friedreich Ataxia: Time for a Reappraisal? Front. Neurosci. 2019; 13:386. [CrossRef]
- Bartels C, Späte K, Krampe H, Ehrenreich H. Recombinant Human Erythropoietin: Novel Strategies for Neuroprotective/Neuro-regenerative Treatment of Multiple Sclerosis. Ther. Adv. Neurol. Dis. 2008; 1:193-206. [CrossRef]
- Dayyat EA, Zhang SX, Wang Y, Cheng ZJ, Gozal D. Exogenous erythropoietin administration attenuates intermittent hypoxia-induced cognitive deficits in a murine model of sleep apnea. BMC Neurosci. 2012; 13:77. [CrossRef]
- Maurice T, Mustafa MH, Desrumaux C, Keller E, Naert G, de la CG-BM, et al. Intranasal formulation of erythropoietin (EPO) showed potent protective activity against amyloid toxicity in the Aβ25₋35 non-transgenic mouse model of Alzheimer’s disease. J. Psychopharmacol. 2013; 27:1044-57. [CrossRef]
- Auzmendi J, Puchulu MB, Rodríguez JCG, Balaszczuk AM, Lazarowski A, Merelli A. EPO and EPO-Receptor System as Potential Actionable Mechanism for the Protection of Brain and Heart in Refractory Epilepsy and SUDEP. Neurochem. Res. 2020;26(12):1356-64. [CrossRef]
- Miskowiak KW, Macoveanu J, Vinberg M, Assentoft E, Randers L, Harmer CJ, et al. Effects of erythropoietin on memory-relevant neurocircuitry activity and recall in mood disorders. Acta Psychiat. Scand. 2016; 134:249-59. [CrossRef]
- Vinberg M, Weikop P, Olsen NV, Kessing LV, Miskowiak K. Effect of recombinant erythropoietin on inflammatory markers in patients with affective disorders: A randomised controlled study. Brain Behav. Immun. 2016; 57:53-7. [CrossRef]
- Qi C, Xu M, Gan J, Yang X, Wu N, Song L, et al. Erythropoietin improves neurobehavior by reducing dopaminergic neuron loss in a 6-hydroxydopamine-induced rat model. Internat. J. Mol. Med. 2014; 34: 440-50. [CrossRef]
- Rey F, Ottolenghi S. Mitochondrial Metabolism as Target of the Neuroprotective Role of Erythropoietin in Parkinson’s Disease. Antioxidants (Basel) 2021;10(1). [CrossRef]
- Pedroso I, Bringas ML, Aguiar A, Morales L, Alvarez M, Valdés PA, et al. Use of Cuban recombinant human erythropoietin in Parkinson’s disease treatment. MEDICC Review. 2012; 14:11-7. [CrossRef]
- Fischer HS, Reibel NJ, Bührer C, Dame C. Prophylactic Erythropoietin for Neuroprotection in Very Preterm Infants: A Meta-Analysis Update. Front. Pediat. 2021; 9:657228. [CrossRef]
- Ma Y, Zhou Z, Yang GY, Ding J, Wang X. The Effect of Erythropoietin and Its Derivatives on Ischemic Stroke Therapy: A Comprehensive Review. Front. Pharmacol. 2022; 13:743926. [CrossRef]
- Robertson C, Sadrameli S. Erythropoietin in the neurology ICU. Curr. Treat. Options Neurol. 2013; 15:104-12. [CrossRef]
- Hemani S, Lane O. Systematic Review of Erythropoietin (EPO) for Neuroprotection in Human Studies. Neurochem. Res. 2021; 46:732-9. [CrossRef]
- Cassel J-C, Duconseille E, Jeltsch H, Will B. The fimbria-fornix/cingular bundle pathways: a review of neurochemical and behavioural approaches using lesions and transplantation techniques. Prog. Neurobiol. 1997; 51:663-716. [CrossRef]
- Vizi ES, Kiss JP. Neurochemistry and pharmacology of the major hippocampal transmitter systems: synaptic and non-synaptic interaction. Hippocampus. 1998; 8:566-607. [CrossRef]
- Liu J, Zhang Z, Li JT, Zhu YH, Zhou HL, Liu S, et al. Effects of NT-4 gene modified fibroblasts transplanted into AD rats. Neurosci. Lett. 2009; 466:1-5. [CrossRef]
- Xuan AG, Luo M, Ji WD, Long DH. Effects of engrafted neural stem cells in Alzheimer’s disease rats. Neurosci. Lett. 2009; 450:167-71. [CrossRef]
- Xuan AG, Long DH, Gu HG, Yang DD, Hong LP, Leng SL. BDNF improves the effects of neural stem cells on the rat model of Alzheimer’s disease with unilateral lesion of fimbria-fornix. Neurosci. Lett. 2008; 440:331-5. [CrossRef]
- Almaguer-Melian W, Merceron-Martinez D, Pav¢n-Fuentes N, Alberti Amador E, Leon-Martinez R, Ledon N, et al. Erythropoietin promotes neural plasticity and spatial memory recovery in fimbria-fornix-lesioned ats. Neurorehabil. Neural Repair. 2015; 29: 979-88. [CrossRef]
- Almaguer-Melian W, Mercerón-Martinez D, Delgado-Ocaña S, Alberti-Amador E, Gonzalez-Gómez R, Bergado JA. Erythropoietin improves object placement recognition memory in a time dependent manner in both, uninjured animals and fimbria-fornix-lesioned male rats. Horm. Behav. 2018; 100:6. [CrossRef]
- Almaguer-Melian W, Mercerón-Martinez D, Alberti-Amador E, Alacán-Ricardo L, de Bardet JC, Orama-Rojo N, et al. Learning induces EPO/EPOr expression in memory relevant brain areas, whereas exogenously applied EPO promotes remote memory consolidation. Synapse. 2024;78:e22282. [CrossRef]
- Tse D, Langston RF, Kakeyana M, Bethus I, Spooner PA, Wood ER, et al. Schemas and Memory Consolidation. Science. 2007; 316:76-82. [CrossRef]
- Dudai Y. Molecular basis of long-term memories: a question of persistence. Curr. Op. Neurobiol. 2002; 12:211-6. [CrossRef]
- Malenka RC, Bear MF. LTP and LTD; An Embarrassment of Riches. Neuron. 2004; 44:5-21. [CrossRef]
- Almaguer-Melian W, Mercerón-Martínez D, Delgado-Ocaña S, Pavón-Fuentes N, Ledón N, Bergado JA. EPO Induces Changes in Synaptic Transmission and Plasticity in the Dentate Gyrus of Rats. Synapse. 2016; 70: 240-52. [CrossRef]
- Bramham CR, Messaoudi E. BDNF function in adult synaptic plasticity: The synaptic consolidation hypothesis. Prog. Neurobiol. 2005; 76: 99-125. [CrossRef]
- Soule J, Messaoudi E, Bramham CR. Brain-derived neurotrophic factor and control of synaptic consolidation in the adult brain. Biochem. Soc. Trans. 2006; 34 (Pt 4): 600-4. [CrossRef]
- Ying SW, Futter M, Rosenblum K, Webber MJ, Hunt SP, Bliss TV, et al. Brain-derived neurotrophic factor induces long-term potentiation in intact adult hippocampus: requirement for ERK activation coupled to CREB and upregulation of Arc synthesis. J. Neurosci. 2002; 22:1532-40. [CrossRef]
- Buzsaki G, Gage FH. Absence of long-term potentiation in the subcortically deafferented dentate gyrus. Brain Res. 1989; 484:94-101. [CrossRef]
- Almaguer-Melian W, Mercerón-Martínez D, Bergado-Rosado J. A unique erythropoietin dosage induces the recovery of long-term synaptic potentiation in fimbria-fornix lesioned rats. Brain Res. 2023; 1799:148178. [CrossRef]
- Bear MF, Malenka RC. Synaptic plasticity: LTP and LTD. Curr. Op. Neurobiol. 1994; 4:389-99. [CrossRef]
- Malenka RC. Synaptic Plasticity in the Hippocampus: LTP and LTD. Cell. 1994; 78:535-8. [CrossRef]
- Bliss TVP, Collingridge GL. A synaptic model of memory: Long-term potentiation in the hippocampus. Nature. 1993; 361:31-9. [CrossRef]
- Matthies H. Neurobiological aspects of learning and memory. Ann. Rev. Psychol. 1989; 40:381-404. [CrossRef]
- McCormick DA, Nestvogel DB, He BJ. Neuromodulation of Brain State and Behavior. Ann. Rev. Neurosci. 2020; 43:391-415. [CrossRef]
- Rolls ET. The hippocampus, ventromedial prefrontal cortex, and episodic and semantic memory. Prog. Neurobiol. 2022; 217:102334. [CrossRef]
- Frey U, Morris RGM. Synaptic tagging and long-term potentiation. Nature. 1997; 385:533-6. [CrossRef]
- Frey U, Morris RGM. Synaptic tagging: implications for late maintenance of hippocampal long-term potentiation. Trends Neurosci. 1997; 21:181-8. [CrossRef]
- McGaugh JL. Memory-a century of consolidation. Science. 2000; 287:248-51. [CrossRef]
- Ballarini F, Moncada D, Mart¡nez MC, Alen N, Viola H. Behavioral tagging is a general mechanism of long-term memory formation. Proc. Nat. Acad. Sci. USA. 2009; 106:14599-604. [CrossRef]
- Moncada D, Ballarini F, Viola H. Behavioral Tagging: A Translation of the Synaptic Tagging and Capture Hypothesis. Neural Plast. 2015; 2015: 650780. [CrossRef]
- Bramham CR, Worley PF, Moore MJ, Guzowski JF. The immediate early gene arc/arg3.1: regulation, mechanisms, and function. J. Neurosci. 2008;28: 11760-7. [CrossRef]
- Bramham CR. Opioid receptor dependent long-term potentiation: Peptidergic regulation of synaptic plasticity in the hippocampus. Neurochemistry International. 1992; 20: 441-55. [CrossRef]
- Tyler WJ, Alonso M, Bramham CR, Pozzo-Miller LD. From acquisition to consolidation: on the role of brain-derived neurotrophic factor signaling in hippocampal-dependent learning. Learn. Mem. 2002; 9: 224-37. [CrossRef]
- Wakhloo D, Scharkowski F. Functional hypoxia drives neuroplasticity and neurogenesis via brain erythropoietin. Nat. Commun. 2020;11: 1313. [CrossRef]
- Miskowiak KW, Vinberg M, Macoveanu J, Ehrenreich H, Køster N, Inkster B, et al. Effects of Erythropoietin on Hippocampal Volume and Memory in Mood Disorders. Biol Psychiatry. 2015;78: 270-7. [CrossRef]
- Miskowiak KW, Vinberg M, Glerup L, Paulson OB, Knudsen GM, Ehrenreich H, et al. Neural correlates of improved executive function following erythropoietin treatment in mood disorders. Psychol Med. 2016;46: 1679-91. [CrossRef]
- Miskowiak KW, Rush AJ, Jr., Gerds TA, Vinberg M, Kessing LV. Targeting Treatments to Improve Cognitive Function in Mood Disorder: Suggestions From Trials Using Erythropoietin. The Journal of clinical psychiatry. 2016;77: e1639-46. [CrossRef]
- Miskowiak KW, Petersen NA, Harmer CJ, Ehrenreich E, Kessing LV. Neural correlates of improved recognition of happy faces after erythropoietin treatment in bipolar disorder. 2018;138: 336-47. [CrossRef]
- Yaeger JDW, John MM, Ledesma LJ, Krupp KT, Booth CD, Jones NT, et al. Acute carbamoylated erythropoietin reduces social stress-induced anxiety and depression-related behaviors. Neuropharmacology. 2025:110558. [CrossRef]
- Martin L, Garcia Rodriguez JC, Audran M, Ericsson M, Maurice T. Detection of a nonerythropoietic erythropoietin, Neuro-EPO, in blood after intranasal administration in rat. Drug Test Anal. 2020;12: 1605-13. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



