Submitted:
22 December 2025
Posted:
24 December 2025
You are already at the latest version
Abstract
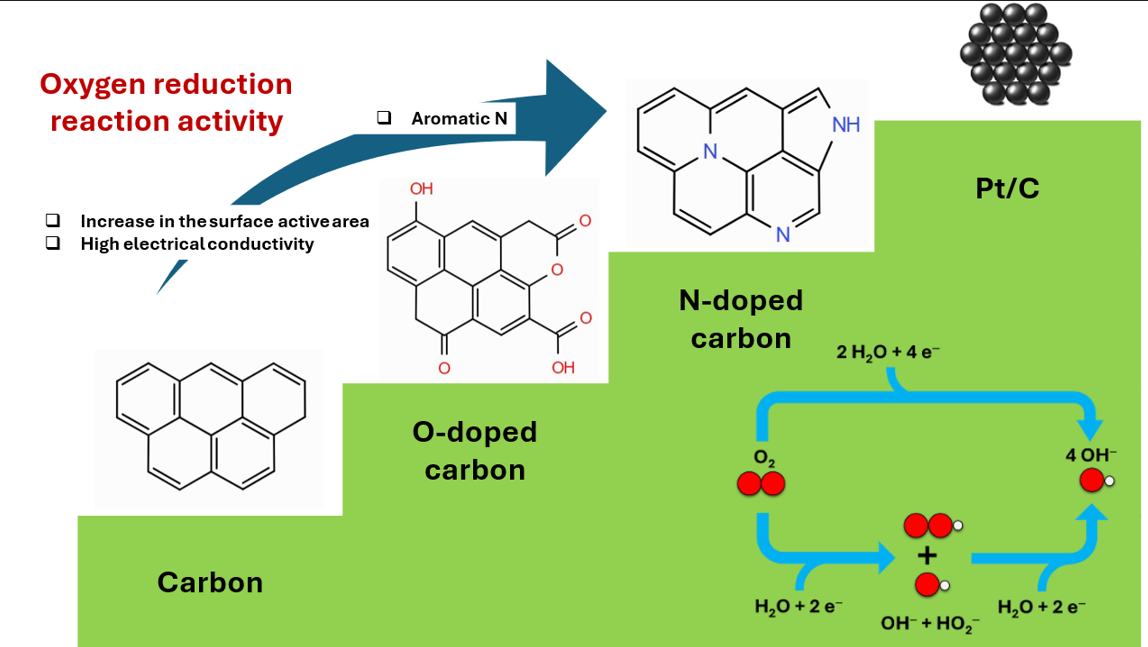
Keywords:
1. Introduction
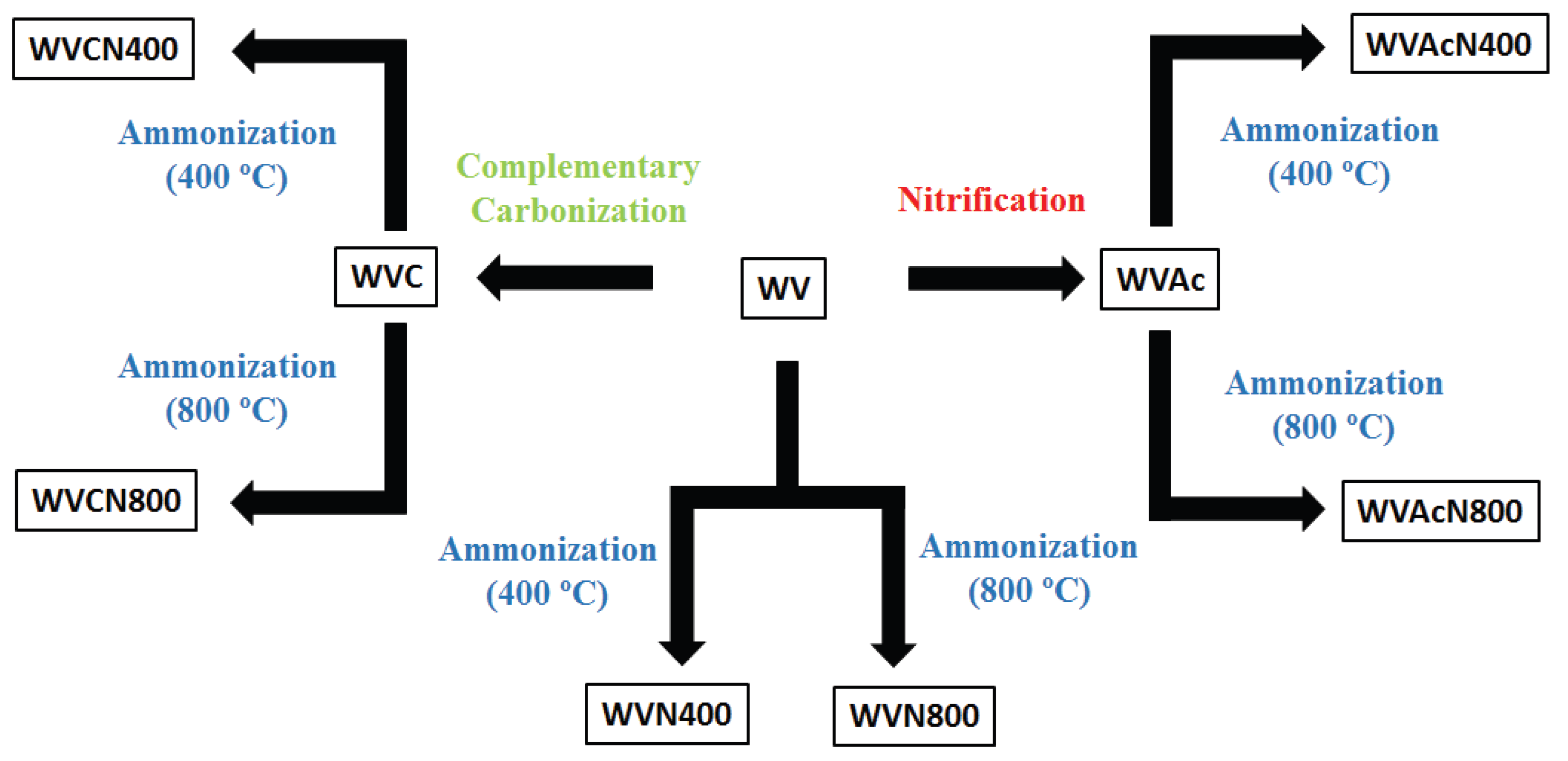
2. Materials and Methods
3. Results and Discussion
3.1. Activated Carbons
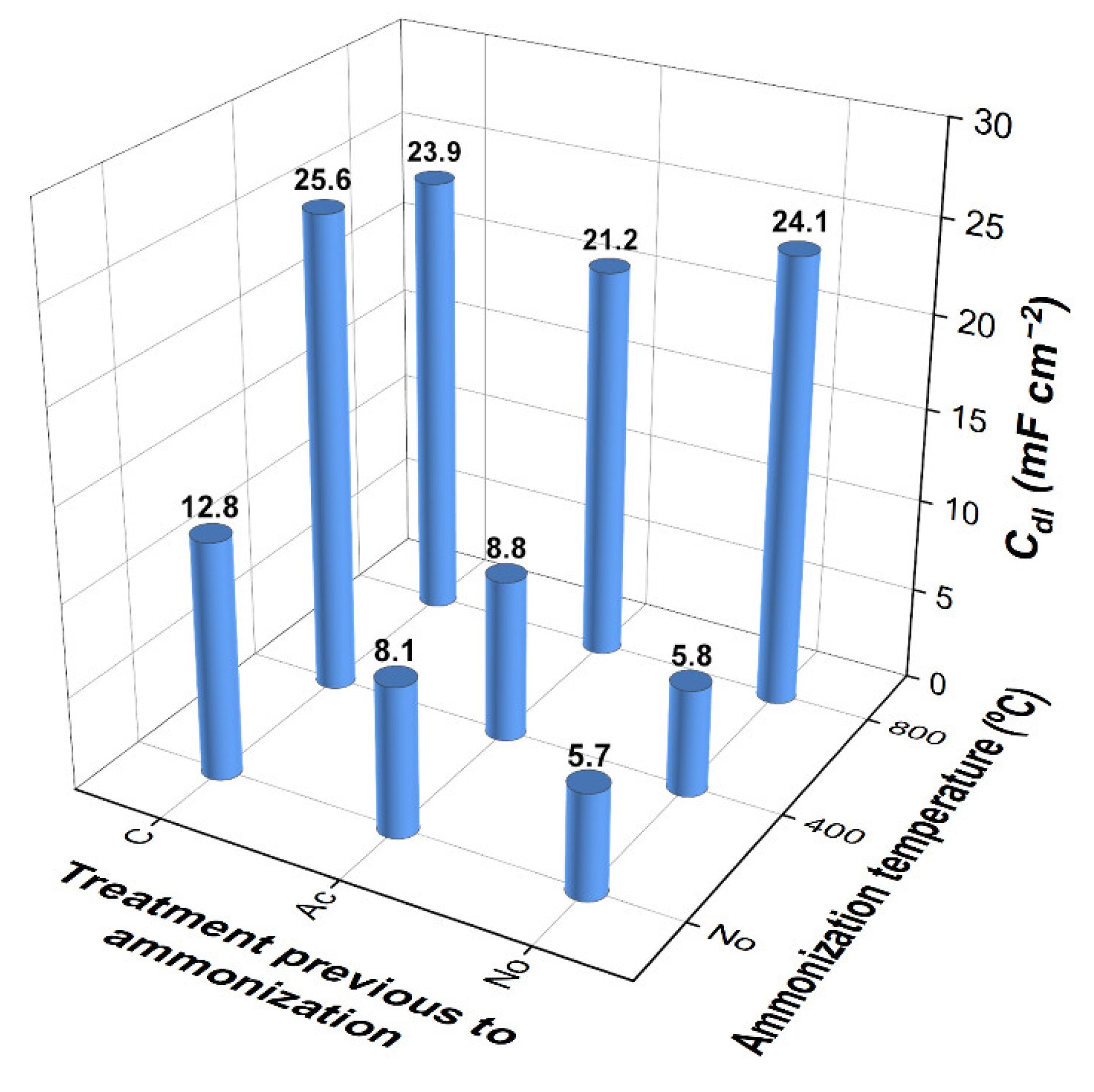
3.2. Capacitive Properties
3.3. ORR Activity
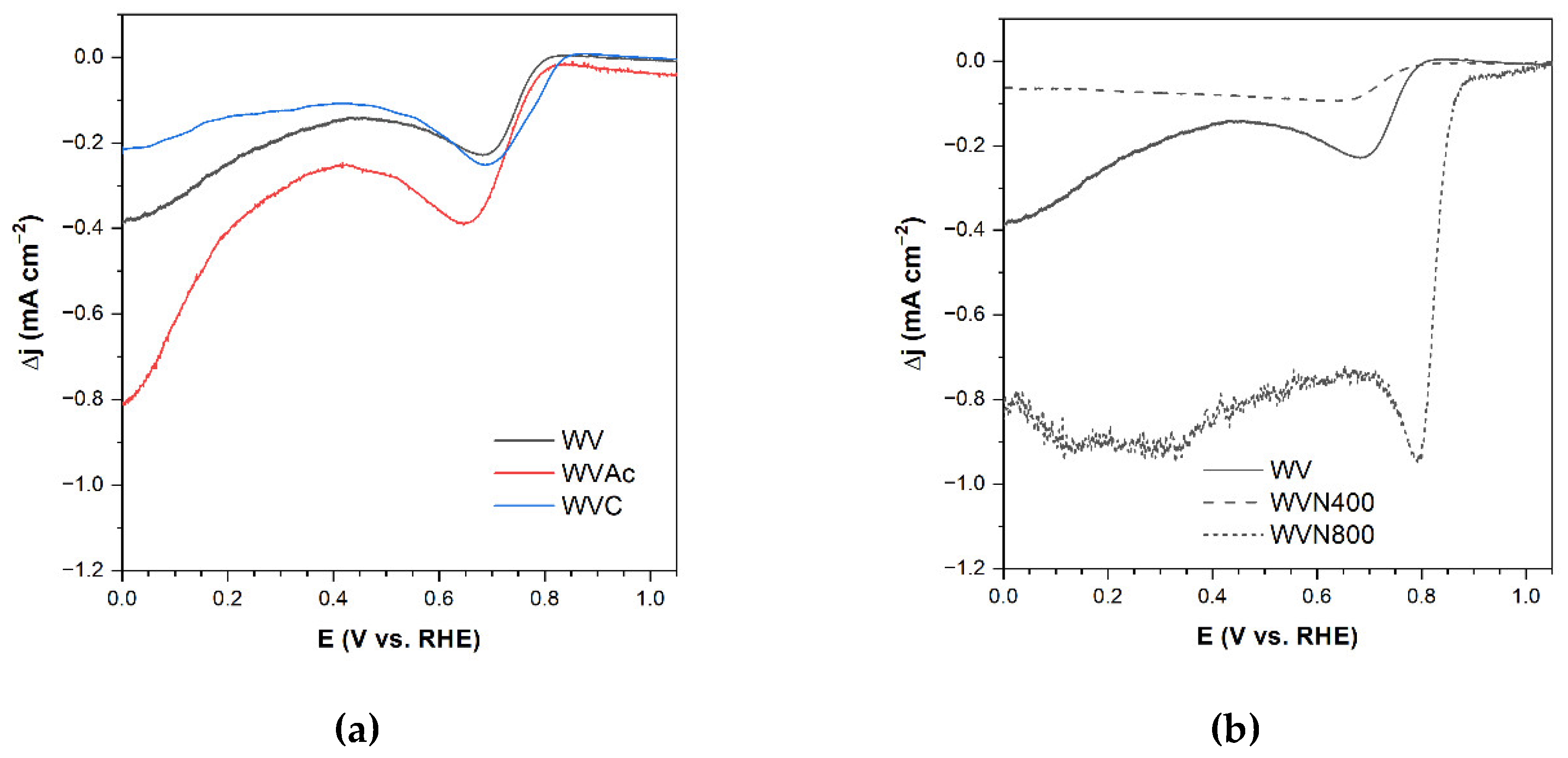
3.3.1. Unmodified AC WV
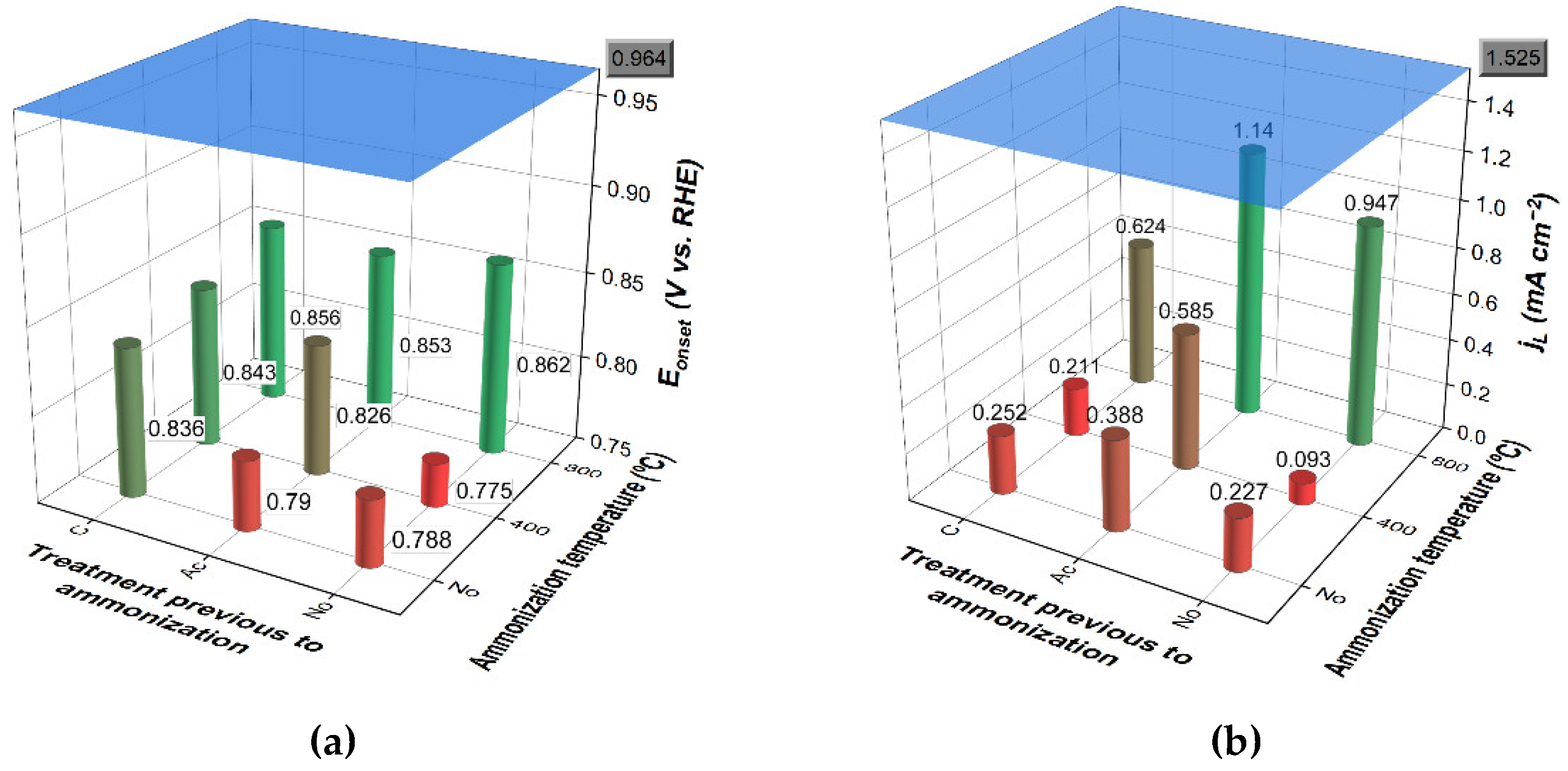
3.3.2. Effects of Nitrification
3.3.3. Effects of Complementary Carbonization
3.3.4. Effects of Ammonization
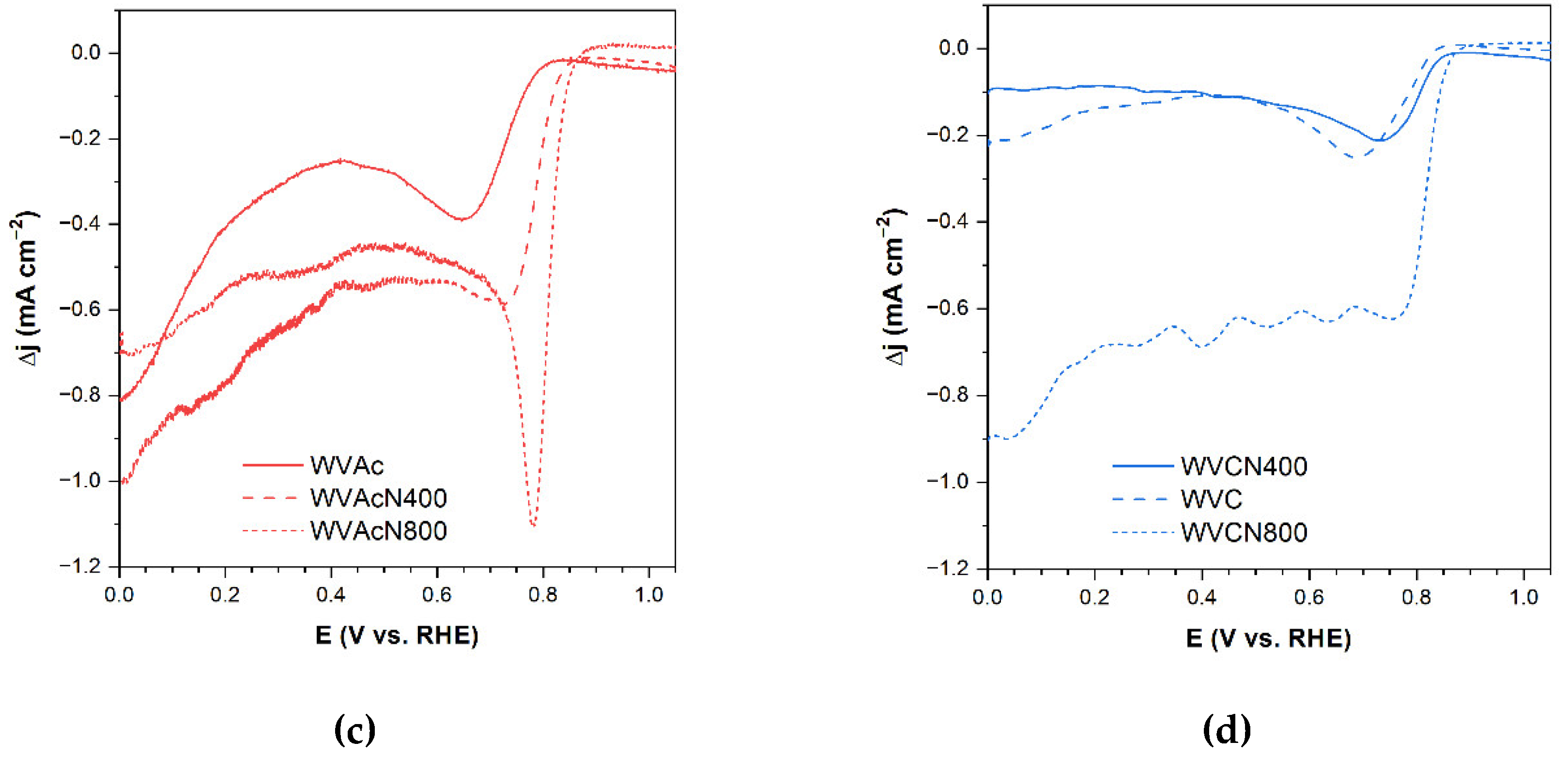
3.3.5. Final Considerations about ORR Performance
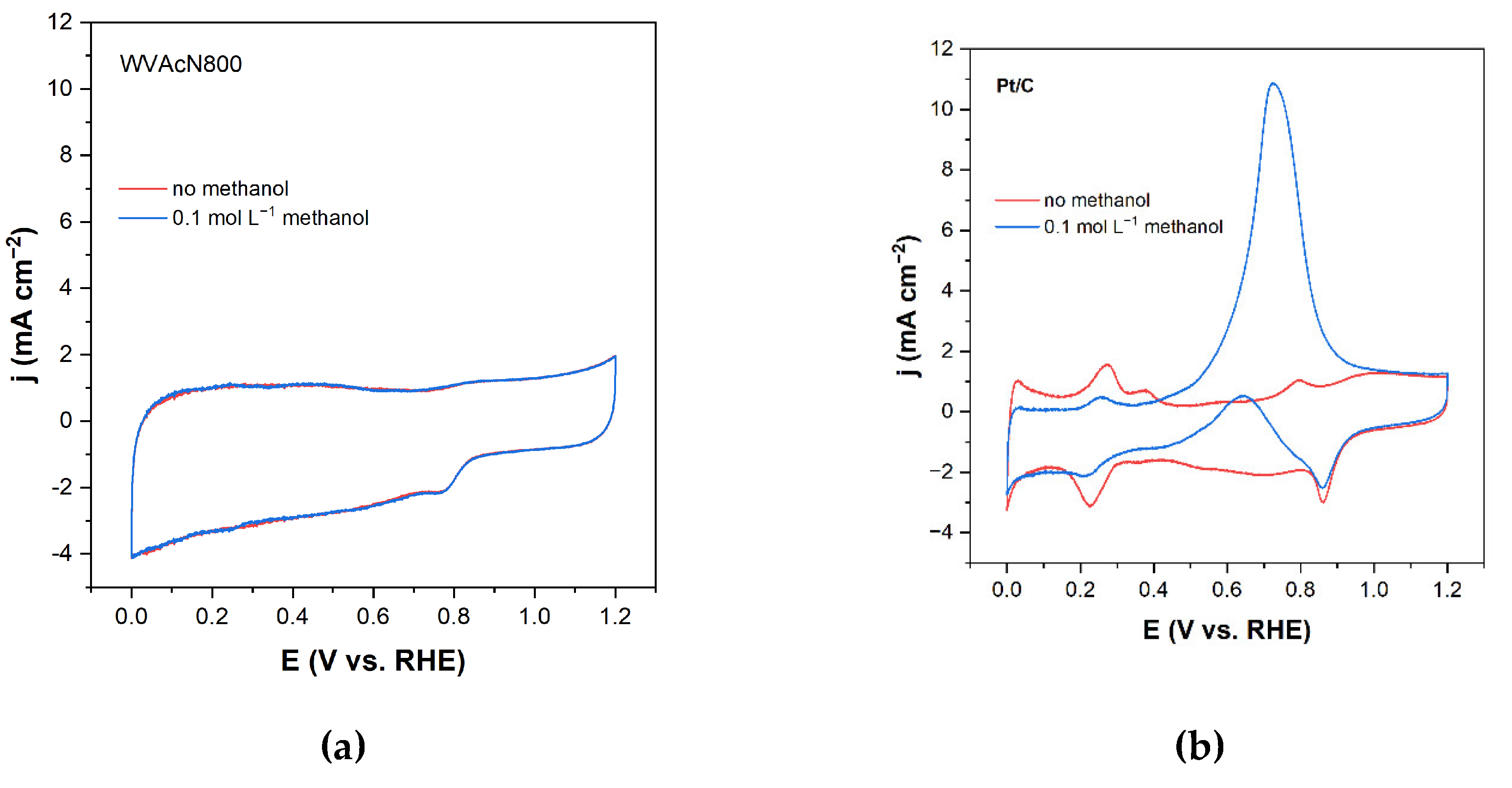
3.4. Methanol Tolerance
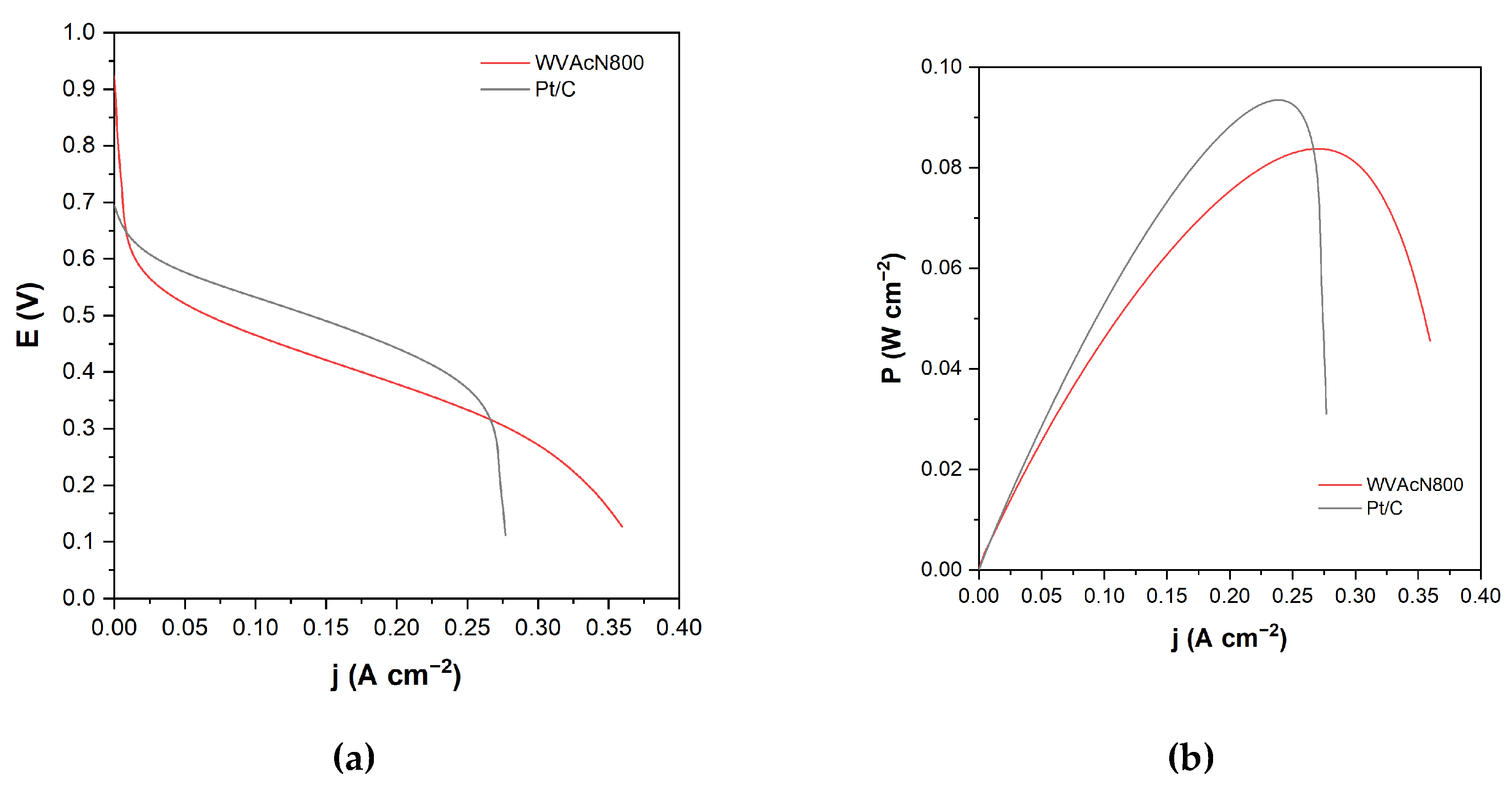
3.5. Single-Cell DMFC Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AC | Activated carbon |
| CV | Cyclic voltammetry |
| DMFC | Direct Methanol Fuel Cell |
| EA | Elemental analyses |
| Cdl | Geometric double-layer capacitance |
| LSV | Linear sweep voltammetry |
| OFG | Oxygenated functional group |
| Eonset | Onset potential |
| OCV | open circuit voltage |
| ORR | Oxygen reduction reaction |
| PZC | Point of zero charge |
| RHE | Reversible hydrogen electrode |
| SSA | Specific surface area |
| Vmic | Micropores volume |
| Vmes | Mesopores volume |
| XPS | X-ray photoelectron spectroscopy |
References
- Ge, X.; Sumboja, A.; Wuu, D.; An, T.; Li, B.; Goh, F.W.T.; Hor, T.S.A.; Zong, Y.; Liu, Z. Oxygen Reduction in Alkaline Media: From Mechanisms to Recent Advances of Catalysts. ACS Catal. 2015, 5, 4643–4667. [Google Scholar] [CrossRef]
- Li, S.; Shi, L.; Guo, Y.; Wang, J.; Liu, D.; Zhao, S. Selective Oxygen Reduction Reaction: Mechanism Understanding, Catalyst Design and Practical Application. Chem. Sci. 2024, 15, 11188–11228. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, A.; Siahrostami, S.; Patel, A.; Nørskov, J.K. Understanding Catalytic Activity Trends in the Oxygen Reduction Reaction. Chem. Rev. 2018, 118, 2302–2312. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, A.; Liu, Y.; Zhou, W.; Wen, H.; Zhang, H.; Sun, K.; Li, S.; Zhou, J.; Wang, Y.; et al. Catalytically Active Carbon for Oxygen Reduction Reaction in Energy Conversion: Recent Advances and Future Perspectives. Advanced Science 2024, 11, 2308040. [Google Scholar] [CrossRef]
- Dai, L.; Xue, Y.; Qu, L.; Choi, H.-J.; Baek, J.-B. Metal-Free Catalysts for Oxygen Reduction Reaction. Chem. Rev. 2015, 115, 4823–4892. [Google Scholar] [CrossRef]
- Florent, M.; Wallace, R.; Bandosz, T.J. Oxygen Electroreduction on Nanoporous Carbons: Textural Features vs Nitrogen and Boron Catalytic Centers. ChemCatChem 2019, 11, 851–860. [Google Scholar] [CrossRef]
- Ma, R.; Lin, G.; Zhou, Y.; Liu, Q.; Zhang, T.; Shan, G.; Yang, M.; Wang, J. A Review of Oxygen Reduction Mechanisms for Metal-Free Carbon-Based Electrocatalysts. npj Comput Mater 2019, 5, 78. [Google Scholar] [CrossRef]
- Fang, W.; Wang, L.; Shen, D.; Meng, X.; Bai, H.; Li, C. Oxygen-Containing Functional Groups Cooperate to Boost Electrochemical ORR Selectivity to H2O2. Inorg. Chem. Front. 2025, 12, 7695–7705. [Google Scholar] [CrossRef]
- Zhao, G.; Chen, T.; Tang, A.; Yang, H. Roles of Oxygen-Containing Functional Groups in Carbon for Electrocatalytic Two-Electron Oxygen Reduction Reaction. Chemistry A European J 2024, 30, e202304065. [Google Scholar] [CrossRef]
- Wu, B.; Meng, H.; Morales, D.M.; Zeng, F.; Zhu, J.; Wang, B.; Risch, M.; Xu, Z.J.; Petit, T. Nitrogen-Rich Carbonaceous Materials for Advanced Oxygen Electrocatalysis: Synthesis, Characterization, and Activity of Nitrogen Sites. Adv Funct Materials 2022, 32, 2204137. [Google Scholar] [CrossRef]
- Al-Hajri, W.; De Luna, Y.; Bensalah, N. Review on Recent Applications of Nitrogen-Doped Carbon Materials in CO2 Capture and Energy Conversion and Storage. Energy Tech 2022, 10, 2200498. [Google Scholar] [CrossRef]
- Oliveira, S.D.C.; Dutra, R.C.; León, J.J.L.; Martins, G.A.V.; Silva, A.M.A.; Azevedo, D.C.S.D.; Santiago, R.G.; Ballesteros-Plata, D.; Rodríguez-Castellón, E.; Prauchner, M.J. Activated Carbon Ammonization: Effects of the Chemical Composition of the Starting Material and the Treatment Temperature. C 2025, 11, 15–40. [Google Scholar] [CrossRef]
- De Falco, G.; Florent, M.; Jagiello, J.; Cheng, Y.; Daemen, L.L.; Ramirez-Cuesta, A.J.; Bandosz, T.J. Alternative View of Oxygen Reduction on Porous Carbon Electrocatalysts: The Substance of Complex Oxygen-Surface Interactions. iScience 2021, 24, 102216. [Google Scholar] [CrossRef] [PubMed]
- Brandão, R.D.; De Freitas Júnior, A.M.; Linares, J.J.; Suarez, P.A.Z.; Dutra, R.C.; Garnier, J.; Tonhá, M.S.; Ballesteros-Plata, D.; Rodríguez-Castellón, E.; Prauchner, M.J. Activated Carbon-Supported Pt Catalysts Intended for the Hydroprocessing of Lipid Feedstocks: Effects of Support Surface Composition and Impregnation Protocol. Molecules 2025, 30, 2862. [Google Scholar] [CrossRef] [PubMed]
- De Freitas Júnior, A.M.; Brandão, R.D.; Garnier, J.; Tonhá, M.S.; Mussel, W.D.N.; Ballesteros-Plata, D.; Rodríguez-Castellón, E.; Prauchner, M.J. Activated Carbons as Supports for Sulfided Mo-Based Catalysts Intended for the Hydroprocessing of Lipidic Feedstocks. Catalysts 2025, 15, 359. [Google Scholar] [CrossRef]
- Hou, C.; Zhang, X.; Yuan, W.; Zhang, Y.; Deng, H.; Liu, X. A Facile Synthesis for Nitrogen-doped Carbon Catalyst with High Activity of Oxygen Reduction Reaction in Acidic Media. Int J Energy Res 2021, 45, 19574–19585. [Google Scholar] [CrossRef]
- Farzaneh, A.; Goharshadi, E.K.; Gharibi, H.; Saghatoleslami, N.; Ahmadzadeh, H. Insights on the Superior Performance of Nanostructured Nitrogen-Doped Reduced Graphene Oxide in Comparison with Commercial Pt/C as Cathode Electrocatalyst Layer of Passive Direct Methanol Fuel Cell. Electrochimica Acta 2019, 306, 220–228. [Google Scholar] [CrossRef]
- Wan, K.; Long, G.-F.; Liu, M.-Y.; Du, L.; Liang, Z.-X.; Tsiakaras, P. Nitrogen-Doped Ordered Mesoporous Carbon: Synthesis and Active Sites for Electrocatalysis of Oxygen Reduction Reaction. Applied Catalysis B: Environmental 2015, 165, 566–571. [Google Scholar] [CrossRef]
- Kanninen, P.; Borghei, M.; Sorsa, O.; Pohjalainen, E.; Kauppinen, E.I.; Ruiz, V.; Kallio, T. Highly Efficient Cathode Catalyst Layer Based on Nitrogen-Doped Carbon Nanotubes for the Alkaline Direct Methanol Fuel Cell. Applied Catalysis B: Environmental 2014, 156–157, 341–349. [Google Scholar] [CrossRef]
- Kruusenberg, I.; Ratso, S.; Vikkisk, M.; Kanninen, P.; Kallio, T.; Kannan, A.M.; Tammeveski, K. Highly Active Nitrogen-Doped Nanocarbon Electrocatalysts for Alkaline Direct Methanol Fuel Cell. Journal of Power Sources 2015, 281, 94–102. [Google Scholar] [CrossRef]
- Mirzaeian, M.; Abbas, Q.; Hunt, Michael.R.C.; Hall, P. Pseudocapacitive Effect of Carbons Doped with Different Functional Groups as Electrode Materials for Electrochemical Capacitors. Energies 2020, 13, 5577. [Google Scholar] [CrossRef]
- Morales, D.M.; Risch, M. Seven Steps to Reliable Cyclic Voltammetry Measurements for the Determination of Double Layer Capacitance. J. Phys. Energy 2021, 3, 034013. [Google Scholar] [CrossRef]
- Nascimento, A.P.; Linares, J.J. Performance of a Direct Glycerol Fuel Cell Using KOH Doped Polybenzimidazole as Electrolyte. Journal of the Brazilian Chemical Society 2014. [Google Scholar] [CrossRef]
- Lenne, Q.; Mattiuzzi, A.; Jabin, I.; Le Poul, N.; Leroux, Y.R.; Lagrost, C. Functionalizing Gold Nanoparticles with Calix [4]Arenes Monolayers for Enhancing Selectivity and Stability in ORR Electrocatalysis. Adv Materials Inter 2020, 7, 2001557. [Google Scholar] [CrossRef]
- Vaik, K. Electrochemical Reduction of Oxygen on Anodically Pre-Treated and Chemically Grafted Glassy Carbon Electrodes in Alkaline Solutions. Electrochemistry Communications 2004, 6, 1–5. [Google Scholar] [CrossRef]
- Gabe, A.; Ruiz-Rosas, R.; González-Gaitán, C.; Morallón, E.; Cazorla-Amorós, D. Modeling of Oxygen Reduction Reaction in Porous Carbon Materials in Alkaline Medium. Effect of Microporosity. Journal of Power Sources 2019, 412, 451–464. [Google Scholar] [CrossRef]
- Florent, M.; Hashmi, R.; Bandosz, T.J. The Extent of Carbon Surface Oxygen Affinity and Its Effects on the Activity of Metal-Free Carbon Catalysts in the Oxygen Reduction Reaction: The Interplay of Porosity and N-, O- and S-Enriched Surface Chemistry. Mater. Adv. 2022, 3, 8567–8578. [Google Scholar] [CrossRef]
- Liu, Y.; Li, K.; Ge, B.; Pu, L.; Liu, Z. Influence of Micropore and Mesoporous in Activated Carbon Air-Cathode Catalysts on Oxygen Reduction Reaction in Microbial Fuel Cells. Electrochimica Acta 2016, 214, 110–118. [Google Scholar] [CrossRef]
- Seredych, M.; Szczurek, A.; Fierro, V.; Celzard, A.; Bandosz, T.J. Electrochemical Reduction of Oxygen on Hydrophobic Ultramicroporous PolyHIPE Carbon. ACS Catal. 2016, 6, 5618–5628. [Google Scholar] [CrossRef]
- Encalada, J.; Savaram, K.; Travlou, N.A.; Li, W.; Li, Q.; Delgado-Sánchez, C.; Fierro, V.; Celzard, A.; He, H.; Bandosz, T.J. Combined Effect of Porosity and Surface Chemistry on the Electrochemical Reduction of Oxygen on Cellular Vitreous Carbon Foam Catalyst. ACS Catal. 2017, 7, 7466–7478. [Google Scholar] [CrossRef]
- Barrera, D.; Florent, M.; Kulko, M.; Bandosz, T.J. Ultramicropore-Influenced Mechanism of Oxygen Electroreduction on Metal-Free Carbon Catalysts. J. Mater. Chem. A 2019, 7, 27110–27123. [Google Scholar] [CrossRef]
- Barrera, D.; Florent, M.; Sapag, K.; Bandosz, T.J. Insight into the Mechanism of Oxygen Reduction Reaction on Micro/Mesoporous Carbons: Ultramicropores versus Nitrogen-Containing Catalytic Centers in Ordered Pore Structure. ACS Appl. Energy Mater. 2019, 2, 7412–7424. [Google Scholar] [CrossRef]
- Guo, D.; Shibuya, R.; Akiba, C.; Saji, S.; Kondo, T.; Nakamura, J. Active Sites of Nitrogen-Doped Carbon Materials for Oxygen Reduction Reaction Clarified Using Model Catalysts. Science 2016, 351, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Hu, J.; Wei, J.; Liu, X.; Hou, H. Insights into Nitrogen-doped Carbon for Oxygen Reduction: The Role of Graphitic and Pyridinic Nitrogen Species. ChemPhysChem 2023, 24, e202200734. [Google Scholar] [CrossRef]
- Mazzucato, M.; Durante, C. Insights on Oxygen Reduction Reaction to H2O2: The Role of Functional Groups and Textural Properties on the Activity and Selectivity of Doped Carbon Electrocatalysts. Current Opinion in Electrochemistry 2022, 35, 101051. [Google Scholar] [CrossRef]
- Fortunato, G.V.; Kronka, M.S.; Cardoso, E.S.F.; Dos Santos, A.J.; Roveda, A.C.; Lima, F.H.B.; Ledendecker, M.; Maia, G.; Lanza, M.R.V. A Comprehensive Comparison of Oxygen and Nitrogen Functionalities in Carbon and Their Implications for the Oxygen Reduction Reaction. Journal of Catalysis 2022, 413, 1034–1047. [Google Scholar] [CrossRef]
- Jiang, Y.; Yang, L.; Sun, T.; Zhao, J.; Lyu, Z.; Zhuo, O.; Wang, X.; Wu, Q.; Ma, J.; Hu, Z. Significant Contribution of Intrinsic Carbon Defects to Oxygen Reduction Activity. ACS Catal. 2015, 5, 6707–6712. [Google Scholar] [CrossRef]
- Yan, X.; Jia, Y.; Yao, X. Defects on Carbons for Electrocatalytic Oxygen Reduction. Chem. Soc. Rev. 2018, 47, 7628–7658. [Google Scholar] [CrossRef]
- Deng, H.; Li, Q.; Liu, J.; Wang, F. Active Sites for Oxygen Reduction Reaction on Nitrogen-Doped Carbon Nanotubes Derived from Polyaniline. Carbon 2017, 112, 219–229. [Google Scholar] [CrossRef]
- Peng, W.; Liu, J.; Liu, X.; Wang, L.; Yin, L.; Tan, H.; Hou, F.; Liang, J. Facilitating Two-Electron Oxygen Reduction with Pyrrolic Nitrogen Sites for Electrochemical Hydrogen Peroxide Production. Nat Commun 2023, 14, 4430. [Google Scholar] [CrossRef]
- Li, L.; Tang, C.; Zheng, Y.; Xia, B.; Zhou, X.; Xu, H.; Qiao, S. Tailoring Selectivity of Electrochemical Hydrogen Peroxide Generation by Tunable Pyrrolic-Nitrogen-Carbon. Advanced Energy Materials 2020, 10, 2000789. [Google Scholar] [CrossRef]
- Shi, Y.; Jiao, M.; Wang, C.; Zhang, L.-L.; Sun, S. Ultrahigh Pyrrolic Nitrogen Triggers High-Activity Sites in Carbon Catalysts for Selective Oxygen Reduction to Efficient H2O2 Production. Carbon 2025, 242, 120450. [Google Scholar] [CrossRef]
- Zhang, Y.; Pang, Y.; Xia, D.; Chai, G. Regulable Pyrrolic-N-Doped Carbon Materials as an Efficient Electrocatalyst for Selective O2 Reduction to H2 O2. New J. Chem. 2022, 46, 14510–14516. [Google Scholar] [CrossRef]
- Prauchner, M.J.; Oliveira, S.D.C.; Rodríguez-Reinoso, F. Tailoring Low-Cost Granular Activated Carbons Intended for CO2 Adsorption. Front. Chem. 2020, 8, 581133. [Google Scholar] [CrossRef]
- Mostazo-López, M.J.; Salinas-Torres, D.; Ruiz-Rosas, R.; Morallón, E.; Cazorla-Amorós, D. Nitrogen-Doped Superporous Activated Carbons as Electrocatalysts for the Oxygen Reduction Reaction. Materials 2019, 12, 1346. [Google Scholar] [CrossRef]
- Quílez-Bermejo, J.; Pérez-Rodríguez, S.; Torres, D.; Canevesi, R.; Morallón, E.; Cazorla-Amorós, D.; Celzard, A.; Fierro, V. Nitrogen Sites Prevail over Textural Properties in N-Doped Carbons for the Oxygen Reduction Reaction. Journal of Colloid and Interface Science 2024, 654, 446–453. [Google Scholar] [CrossRef]
- Lima, R.A.C.; Pinto Júnior, A.J.C.; Pocrifka, L.A.; Passos, R.R. Investigation of Nitrogen-Doping Influence on the Electrocatalytic Activity of Graphene in Alkaline Oxygen Reduction Reaction. Mat. Res. 2021, 24, e20200375. [Google Scholar] [CrossRef]
- Ruiz-Marizcal, J.M.; Paez-Ornelas, J.I.; Fernández-Escamilla, H.N.; Murillo-Bracamontes, E.A.; Alonso-Núñez, G.; Perez-Tijerina, E.G.; Takeuchi, N.; Romo-Herrera, J.M. From Graphene Oxide to N-Doped Graphene: Understanding the Doping Process. Adv Energy and Sustain Res 2025, 6, 2400310. [Google Scholar] [CrossRef]
- Béjar, J.; Espinosa‒Magaña, F.; Avelar, J.; Aguilar‒Elguezabal, A.; Guerra‒Balcázar, M.; Arjona, N.; Álvarez‒Contreras, L. Rational Design of Nitrogen‒doped Carbon Nanotubes by Defect Engineering for Zn‒air Batteries with High Performance. Carbon 2023, 204, 411–426. [Google Scholar] [CrossRef]
- Nabais, J.M.V.; Mouquinho, A.; Galacho, C.; Carrott, P.J.M.; Ribeiro Carrott, M.M.L. In Vitro Adsorption Study of Fluoxetine in Activated Carbons and Activated Carbon Fibres. Fuel Processing Technology 2008, 89, 549–555. [Google Scholar] [CrossRef]
- Tamaki, T.; Yamauchi, A.; Ito, T.; Ohashi, H.; Yamaguchi, T. The Effect of Methanol Crossover on the Cathode Overpotential of DMFCs. Fuel Cells 2011, 11, 394–403. [Google Scholar] [CrossRef]
- Li, X.; Miao, Z.; Marten, L.; Blankenau, I. Experimental Measurements of Fuel and Water Crossover in an Active DMFC. International Journal of Hydrogen Energy 2021, 46, 4437–4446. [Google Scholar] [CrossRef]
- Hu, Y.; Jensen, J.O.; Bretzler, P.; Cleemann, L.N.; Yu, J.; Li, Q. Revealing the Genuine Stability of the Reference Pt/C Electrocatalyst toward the ORR. Electrochimica Acta 2021, 391, 138963. [Google Scholar] [CrossRef]
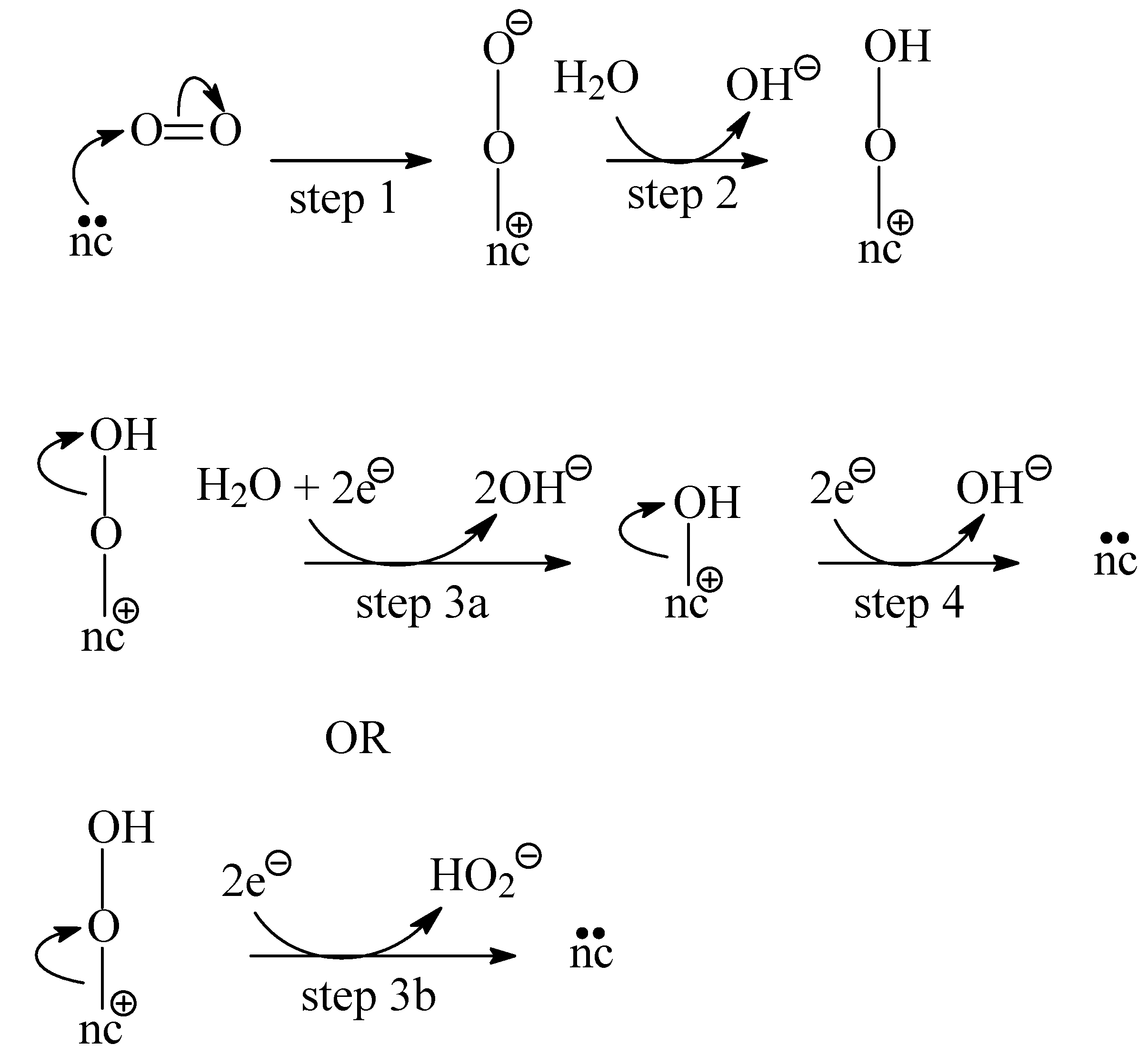
| Sample | Δmam (%)1 | PZC | H/C2 | XPS (wt%) | |||
|---|---|---|---|---|---|---|---|
| C | O | N | Inorganics3 | ||||
| WV | - | 4.7 | 0.41 | 83.1 | 13.7 | 0.3 | 2.9 |
| WVN400 | −4.2 | 6.8 | 0.39 | 83.2 | 10.7 | 3.1 | 3.0 |
| WVN800 | −10.8 | 6.2 | 0.29 | 84.9 | 7.2 | 5.1 | 2.8 |
| WVC | - | 6.2 | 0.16 | 86.2 | 8.4 | 0.8 | 4.6 |
| WVCN400 | +3.2 | 6.1 | 0.15 | 85.3 | 8.1 | 3.1 | 3.5 |
| WVCN800 | −1.5 | 6.3 | 0.19 | 85.3 | 7.6 | 3.9 | 3.3 |
| WVAc | - | 1.8 | 0.48 | 80.1 | 17.0 | 1.7 | 1.2 |
| WVAcN400 | −1.8 | 3.4 | 0.50 | 81.4 | 9.5 | 7.8 | 1.3 |
| WVAcN800 | −32.4 | 6.9 | 0.19 | 89.1 | 8.3 | 1.9 | 0.7 |
| Sample | SSA (m2 g−1)1 | Vmic (cm3 g−1)2 | V0.95 (cm3 g−1)3 | Vmes (cm3 g−1)4 |
|---|---|---|---|---|
| WV | 1506 | 0.51 | 0.92 | 0.41 |
| WVN400 | 1238 | 0.39 | 0.72 | 0.33 |
| WVN800 | 1247 | 0.42 | 0.74 | 0.32 |
| WVC | 1296 | 0.44 | 0.75 | 0.31 |
| WVCN400 | 1289 | 0.43 | 0.74 | 0.31 |
| WVCN800 | 1313 | 0.46 | 0.76 | 0.30 |
| WVAc | 1392 | 0.47 | 0.82 | 0.35 |
| WVAcN400 | 1226 | 0.41 | 0.72 | 0.31 |
| WVAcN800 | 1504 | 0.50 | 0.83 | 0.33 |
| Entry | Original material | N-doping methodology | Eonset (V) | Reference | |
|
Original material |
N-doped material |
||||
| 1 | 1 AC WVA-1050 | Ammonization | 0.788 | 0.853–0.862 | Present work |
| 2 | 2,3 AC | 4 Amidation | 0.82 | 0.84 | [45] |
| 3 | 2,3 AC | 5 Composite with polyaniline | 0.82 | 0.88 | [45] |
| 4 | 1,6 AC MSC30 | 7 Urea (800 °C) | 0.83 | 0.86 | [46] |
| 5 | 1 Carbon Black Pearls 2000 | 7 Urea (850 °C) | 0.737 | 0.777 | [27] |
| 6 | 1 Carbon Black Pearls 2000 | 8 HNO3 + urea (850 °C) | 0.737 | 0.817 | [27] |
| 7 | 2 graphene | 7 Urea (800 °C) | 0.73 | 0.78 | [47] |
| 8 | 2 graphene oxide | 7 Melamine (900 °C) | - | 0.82 | [48] |
| 9 | 2 carbon nanotubes | 7 Urea (800 °C) | 0.78 | 0.89 | [49] |
| 10 | 2 carbon nanotubes | 7 Melamine (800 °C) | 0.78 | 0.90 | [49] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




