Introduction
Choledochal cyst (CC) is a congenital cystic dilatation of the biliary tree, representing one of the most common structural anomalies requiring surgical intervention in pediatric hepatobiliary practice[
1,
2]. Beyond the mechanical implications of biliary obstruction, patients with CC are plagued by recurrent cholangitis, pancreatitis, and a profoundly elevated lifetime risk of biliary malignancy. The precise pathogenesis of CC[
3,
4], however, remains enigmatic. While genetic predispositions and anomalous pancreaticobiliary ductal union are recognized etiological factors, they do not fully explain the chronic inflammation, recurrent infections, and heterogeneous clinical progression observed in patients[
5,
6].
The biliary tract is now understood to host a complex microbial community, the biliary microbiome, which is increasingly implicated in the pathophysiology of various hepatobiliary diseases[
3,
7]. This raises a compelling, yet under-explored, hypothesis: the microbial inhabitants of the choledochal cyst may act as dynamic contributors to local inflammation, tissue remodeling, and disease complications. Preliminary studies employing 16S rRNA gene sequencing have hinted at the existence of a dysbiotic bacterial community within CC[
8,
9]. However, the field is significantly constrained by critical limitations: small sample sizes that hinder robust statistical conclusions, a predominant focus on taxonomic cataloging with limited functional insight, and a notable lack of age-stratified analyses[
10,
11,
12]. The latter is particularly crucial, as pediatric CC spans a period of rapid host development-marked by immune system maturation, dietary transitions, and evolving environmental exposures-all of which are potent drivers of microbiome composition and function in other mucosal sites[
13,
14].
Thus, fundamental questions persist regarding whether the biliary microbiome in CC changes during child development[
13,
15] and, if so, whether such changes are confined to shifts in bacterial identity or extend to a deeper functional reprogramming of the microbial community in adaptation to the host's age-specific physiology[
16]. Addressing these questions necessitates moving beyond small-scale, descriptive studies to a large-scale, age-stratified, and functionally oriented investigation[
17].
To bridge this knowledge gap, we conducted a deep metagenomic sequencing[
4] study on a uniquely large cohort of 201 pediatric CC patients, stratified into infancy (under 1 year), early childhood (1-5 years), and later childhood (5-12 years). We hypothesized that while a core microbial structure might be conserved, the functional repertoire of the biliary microbiome undergoes significant, age-dependent remodeling. By integrating taxonomic profiling with comprehensive functional annotation, such as spanning core metabolism, carbohydrate utilization, antibiotic resistance, and virulence potential, this study aimed to establish a high-resolution functional map of the CC microbiome, assess age-related differences in community structure, and delineate the age-shared “core” versus age-unique “adaptive” features[
18,
19,
20]. Our findings reveal a paradigm of “taxonomic conservation coupled with profound functional remodeling,” identifying early childhood (1-5 years) as a critical window for microbial metabolic adaptation and highlighting distinct, age-specific profiles of antibiotic resistance and virulence factors. This work provides novel mechanistic insights into CC pathophysiology and underscores the importance of age as a key variable in understanding and potentially managing the disease.
Materials and Methods
Study Cohort and Sample Collection
This study was approved by the Institutional Review Board of Capital Institute of Pediatrics, Chinese Academy of Medical Sciences & Peking Union Medical College (Approval No. SHERLL2024053). All procedures were performed in accordance with the ethical standards of the responsible committee and with the Helsinki Declaration. Written informed consent was obtained from the parents or legal guardians of all participating children. A total of 201 pediatric patients diagnosed with choledochal cyst, who underwent radical cyst excision surgery between April 2024 and May 2025, were retrospectively enrolled. During surgery, fresh tissue samples from the excised cyst wall were aseptically collected, immediately snap-frozen in liquid nitrogen, and stored at -80°C until DNA extraction. Patients were stratified into three non-overlapping age groups for analysis: under 1 year old (Under1, n=86), from 1 to under 5 years old (Years1to5, n=82, including 1-year-olds but excluding 5-year-olds), and from 5 to 12 years old (Years5to12, n=33). Relevant clinical metadata were retrieved from the hospital’s electronic medical record system.
Taxonomic and Functional Annotation
For taxonomic assignment, the non-redundant gene catalog was aligned against the NCBI NR database using DIAMOND (v2.0.11) with a blastp e-value cutoff of 1e-5. Taxonomic lineage was assigned using the lowest common ancestor algorithm in MEGAN (v6.21.5). Functional annotation was performed against multiple databases: Clusters of Orthologous Groups (COG), Kyoto Encyclopedia of Genes and Genomes (KEGG), Carbohydrate-Active enZYmes (CAZy), Comprehensive Antibiotic Resistance Database (CARD), Virulence Factors Database (VFDB), and MetaCyc. Annotations were obtained using EggNOG-mapper (v2.1.6) for COG and KEGG, dbCAN3 for CAZy, DeepARG for CARD, and DIAMOND searches against the respective databases for VFDB and MetaCyc, all with standard recommended parameters.
Statistical Analysis
Alpha diversity indices (Observed species, Chao1, ACE, Shannon, Simpson) were calculated using the vegan package (v2.6-4) in R (v4.2.2) based on the gene abundance profile. Differences in alpha diversity indices among age groups were tested using the Kruskal-Wallis test. Venn diagrams were generated using the VennDiagram package to visualize shared and unique genes across age groups at both taxonomic (NR) and functional levels (COG, KEGG, etc.). All statistical tests were two-sided, and a p-value < 0.05 was considered significant.
Results
Study cohort and Demographic Characteristics
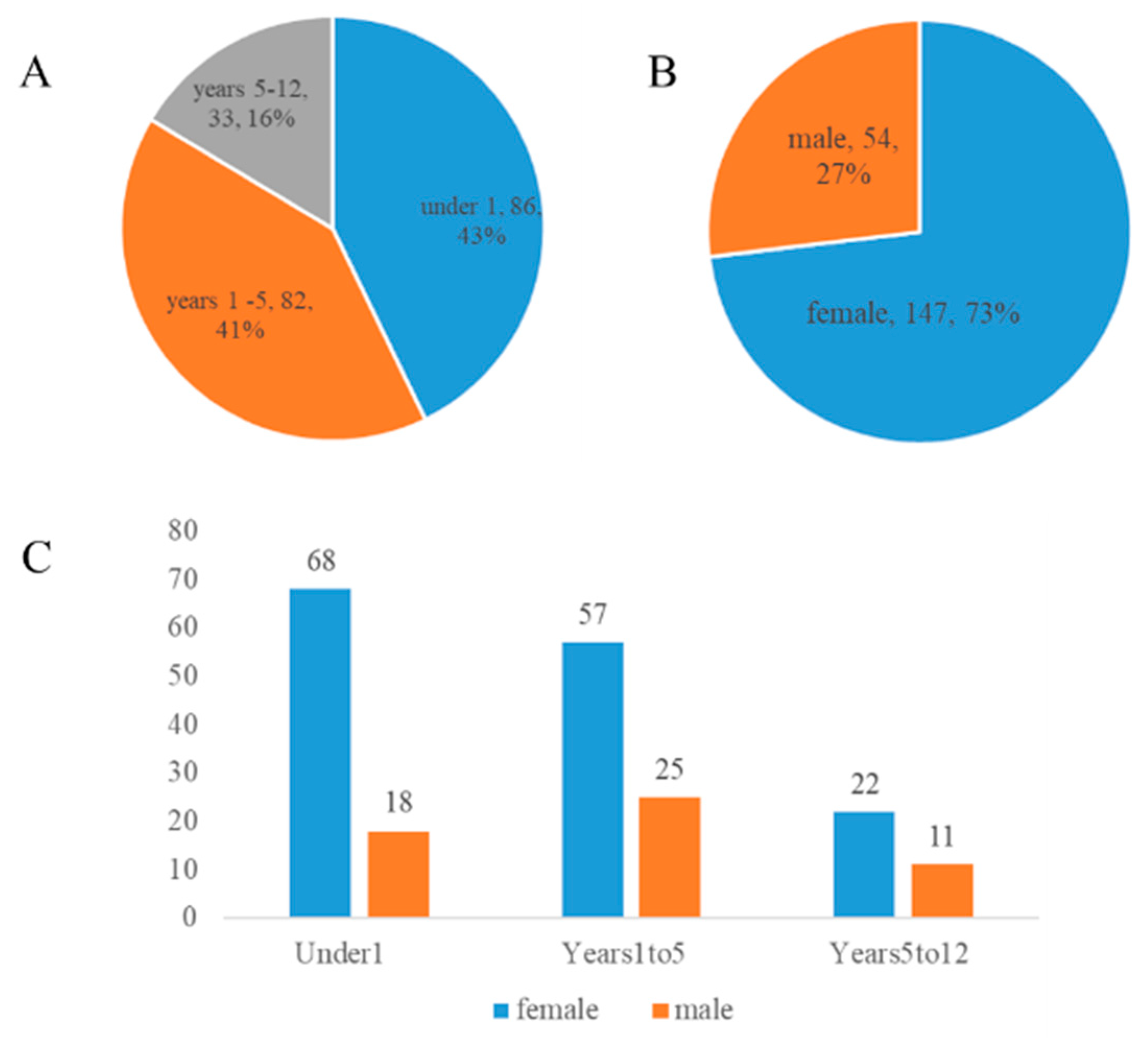
A total of 201 pediatric patients with choledochal cysts were included in this metagenomic study. The demographic composition of the cohort is detailed in
Figure 1.
As shown in
Figure 1A, the study population was stratified into three age groups for subsequent analysis. The under 1 year old group (Under1) constituted the largest proportion of the cohort (n=86, 43%), followed closely by the 1 to 5 years old group (Years1to5, n=82, 41%). The 5 to 12 years old group (Years5to12) accounted for the remaining 16% (n=33). The overall sex distribution of the cohort is presented in
Figure 1B, which reveals a notable female predominance. Female patients (n=147) comprised 73% of the cohort, while male patients (n=54) accounted for 27%, yielding an overall sex ratio (male: female) of approximately 1:2.7. Further stratification of the sex distribution within each age group is illustrated in
Figure 1C. This detailed view confirms that the female predominance was a consistent trend across all three age groups, with the Under1 group having the highest absolute number of both female and male patients.
Comprehensive Functional Profiling of the Biliary Microbiome
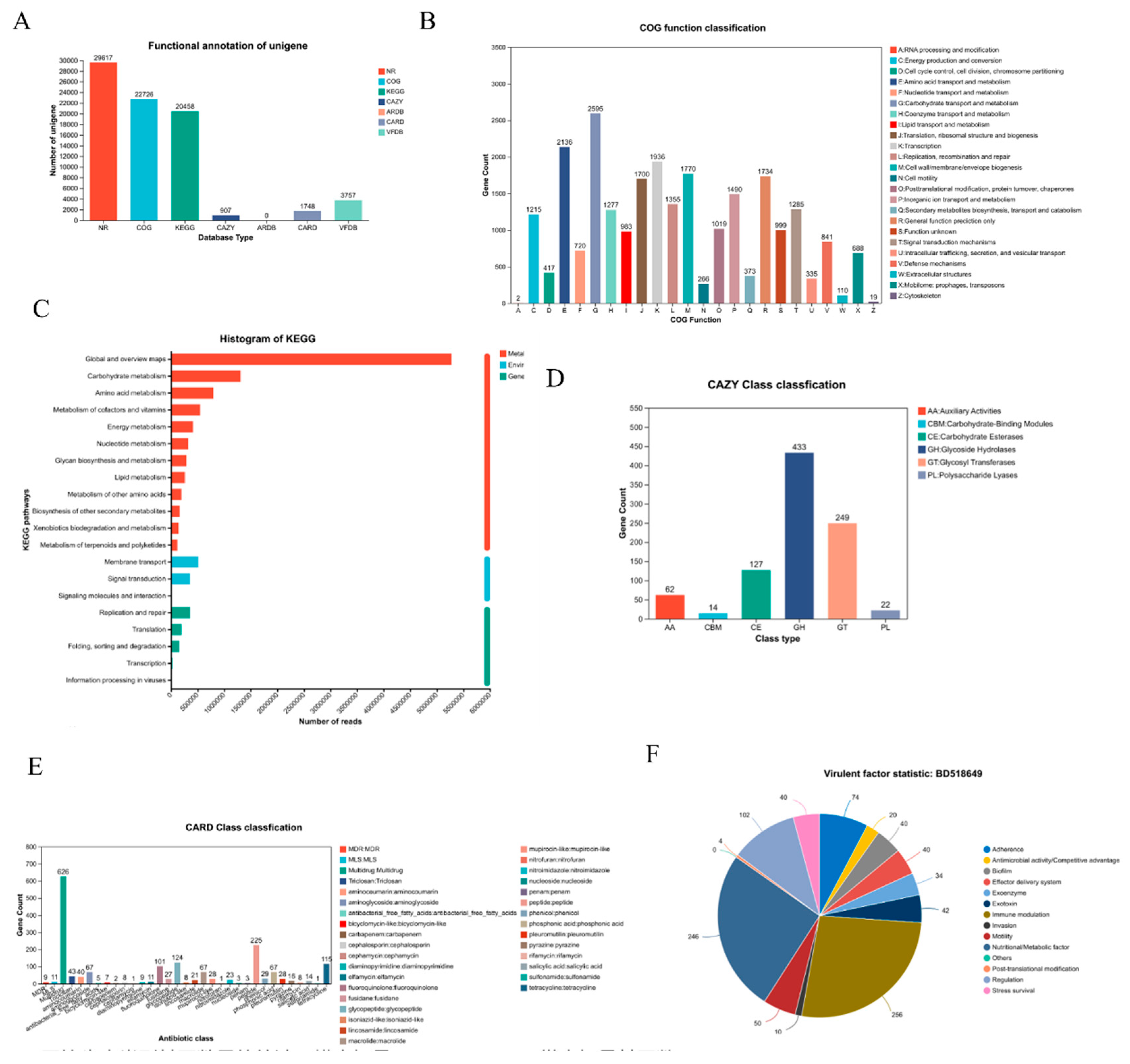
The functional potential of the biliary microbiome was extensively annotated and categorized, as summarized in
Figure 3.
Figure 3A details the overall success of the functional annotation process for the non-redundant gene catalog. The highest annotation rate was achieved against the NR database, with 29,617 genes annotated, establishing a robust foundation for taxonomic assignment. Substantial annotations were also obtained for general and specialized functional databases: 22,726 genes in COG, 20,458 genes in KEGG, and 907 genes in CAZY. Critically, genes implicated in clinically relevant traits were identified, including 1,748 antibiotic resistance genes in the CARD database and 3,757 virulence factor genes in the VFDB.
Functional categorization using the COG database revealed the predominant metabolic nature of the microbial community (
Figure 3B). The most abundant categories were related to essential cellular processes, including amino acid transport and metabolism (Category E), transcription (K), replication and repair (L), and carbohydrate transport and metabolism (G). This profile indicates a community highly active in growth, maintenance, and energy acquisition.
At the pathway level, KEGG analysis further confirmed the dominance of core metabolic functions (
Figure 3C). The most represented pathways belonged to “Metabolism,” with “Global and overview maps,” “Carbohydrate metabolism,” and “Amino acid metabolism” being the top three categories. Pathways involved in “Environmental Information Processing,” such as “Membrane transport” and “Signal transduction,” were also notably abundant, suggesting adaptive mechanisms for the biliary niche.
A focused analysis on carbohydrate-active enzymes (CAZy) highlighted a rich repertoire for carbohydrate metabolism (
Figure 3D). Glycoside Hydrolases (GHs) and Glycosyl Transferases (GTs) were the two most abundant classes, underscoring a strong capacity for both the breakdown and synthesis of complex carbohydrates. A considerable number of genes encoding Carbohydrate-Binding Modules (CBMs) were also detected, which facilitate substrate recognition.
The annotation against the CARD database uncovered a diverse and concerning antibiotic resistome (
Figure 3E). Genes conferring resistance to multiple drug classes (MDR) were highly abundant. More specifically, a high prevalence of genes encoding resistance to beta-lactam antibiotics-including penams, cephalosporins, and carbapenems-was identified, highlighting a significant challenge for clinical treatment. Finally, the virulence factor profile was delineated using the VFDB (
Figure 3F). Genes associated with “Biofilm” formation constituted the largest category, pointing to a key mechanism for persistence and antimicrobial tolerance. Substantial genetic potential was also found for “Exoenzyme” and “Exotoxin” production, “Immune modulation,” and “Adherence,” collectively outlining a multifaceted pathogenic strategy employed by the biliary microbiota.
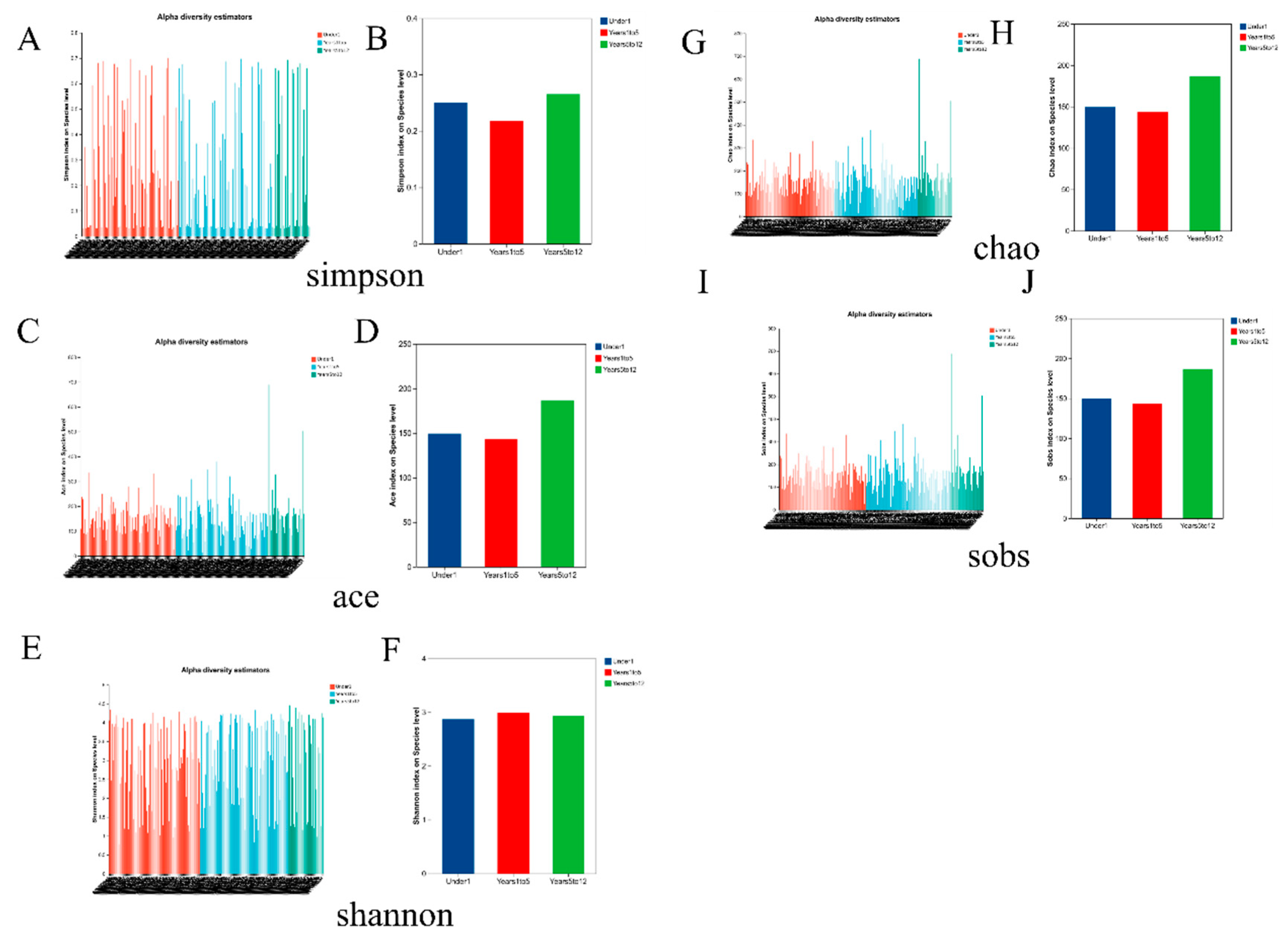
Alpha diversity of the biliary microbiota, assessed across all major indices, did not exhibit statistically significant differences among the three age groups. Specifically, comparisons of species richness (Observed species (SOBS), Chao, and ACE indices) and diversity metrics that incorporate both richness and evenness (Shannon and Simpson indices) all yielded p-values greater than 0.05 in the Kruskal-Wallis tests (
Figure 4). This indicates that the overall within-sample complexity, richness, and evenness of the microbial communities in the choledochal cyst were comparable in infants under 1 year, children aged 1-5 years, and those aged 5-12 years.
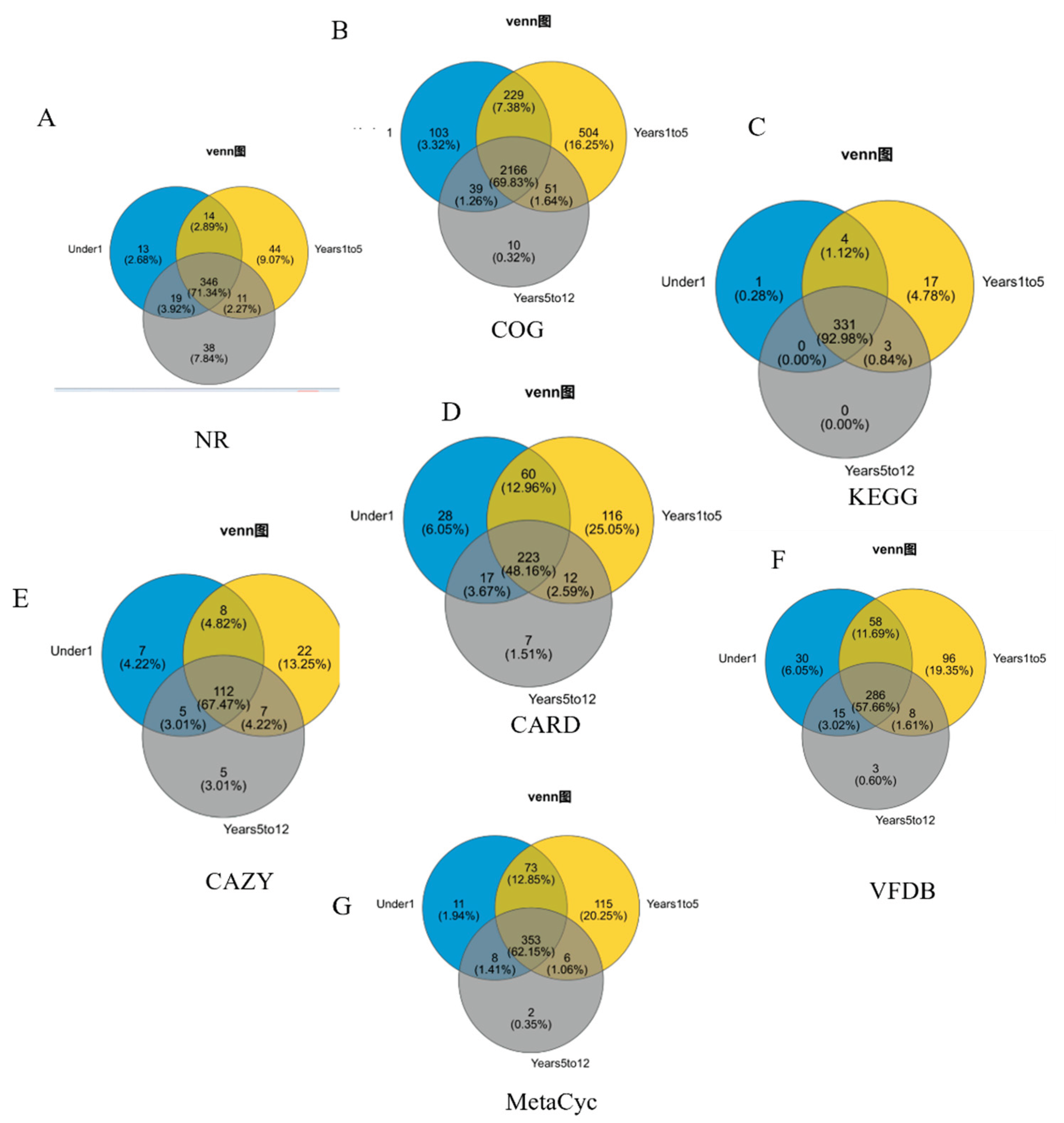
Age-Stratified Comparison of Taxonomic and Functional Gene Repertoires
To delineate the shared and age-specific components of the biliary microbiome, Venn diagram analyses were performed on the gene sets annotated from major taxonomic and functional databases (
Figure 5).
At the species level (
Figure 5A, NR database), a substantial core microbiome of 346 species (71.34%) was common to all three age groups, while each group possessed unique species (Under1: 13; Years1to5: 38; Years5to12: 11). Analysis of general protein functions (COG database) revealed a highly conserved core functional repertoire of 2,166 genes (69.83%) (
Figure 5B). Strikingly, the Years1to5 group harbored the largest number of unique functional genes (229 genes, 7.38%), far exceeding those in the Under1 (103 genes) and Years5to12 (39 genes) groups. This pattern of high conservation coupled with age-specific features was consistent at the pathway level. KEGG pathway analysis showed an exceptionally high proportion of core pathways (331 pathways, 92.98%) shared across all ages (
Figure 5C). Again, the Years1to5 group was distinguished by the greatest number of unique pathways (17 pathways, 4.78%). A similar trend was observed in the MetaCyc database of metabolic pathways, which identified 353 core pathways (62.15%) and highlighted the Years1to5 group as possessing the most unique metabolic pathways (115 pathways, 20.25%) (
Figure 5G).
Focusing on specialized metabolic capabilities, the CAZy database analysis identified a core set of 112 carbohydrate-active enzyme families (67.47%) (
Figure 5E). Consistent with other functional analyses, the Years1to5 group contained the highest number of unique CAZy families (22 families, 13.25%). Clinically relevant gene pools also exhibited distinct patterns. The antibiotic resistome (CARD database) comprised a large shared core of 223 resistance genes (48.16%) (
Figure 5D). The Under1 group featured the most unique resistance genes (60 genes, 12.96%). In contrast, the virulome (VFDB database) analysis showed a core of 286 virulence factor genes (57.66%) common to all groups (
Figure 5F). Notably, the Under1 group possessed the largest unique set of virulence factors (58 genes, 11.69%).
Discussion
This study, through deep metagenomic sequencing of biliary tissue from 201 pediatric patients with choledochal cysts (CC), provides the first systematic characterization of age-dependent dynamics within the CC-associated biliary microbiome. Our central finding reveals a paradigm of “taxonomic conservation coupled with profound functional remodeling”. While the taxonomic composition and alpha diversity of the microbial community remained stable across age groups, its functional potential underwent significant and age-specific restructuring[
21]. This pattern offers a novel microbial ecological perspective for understanding the chronic inflammatory and infectious processes in CC.
Firstly, the lack of significant differences in species composition and alpha diversity across the three age groups contrasts with the conventional view that host development markedly alters the microbiota at mucosal sites. This may suggest that the pathological structure of the CC provides a relatively isolated and stable physical and biochemical microenvironment, buffering the microbial community from the direct selective pressures associated with systemic host maturation, such as immune development and dietary shifts. However, in-depth functional analyses revealed a distinctly different dynamic landscape. The early childhood period (1-5 years) exhibited the greatest enrichment of unique functional genes, metabolic pathways, and carbohydrate-active enzymes (CAZys), indicating that this stage is a critical window for metabolic network reorganization and functional adaptation of the microbial community. This likely correlates with the transition from a milk-based diet to complex solid foods during this developmental period, prompting a functional "upgrade" of the microbiota to utilize new nutritional substrates.
Secondly, our analysis of the clinically relevant resistome and virulome revealed clear age stratification. Infants (<1 year) harbored the most unique antibiotic resistance genes (ARGs) and virulence factor genes (VFGs). This may reflect the selective pressure from early exposure to specific antibiotics in maternal or hospital environments, as well as the enhanced colonization and defense capabilities of initial colonizers establishing a niche in an immunologically immature host. In contrast, the resistome and virulome appeared to become more "streamlined" with age, with an increasing proportion of shared core genes. This may suggest that as the host immune system matures and the microbial community stabilizes, functional selection favors maintaining core ecological homeostasis over expressing redundant invasive traits. Notably, genes associated with biofilm formation were highly enriched across all age groups, strongly supporting biofilm formation as a core, conserved strategy for persistent colonization, resistance to bile flow, and antibiotic attack within the CC biliary environment.
Our findings elevate the role of microbes in CC from mere "infectious agents" to "dynamic modulators of disease progression." Functional remodeling, particularly adaptive changes in metabolism and virulence, may directly participate in regulating the intensity of local inflammation, tissue repair processes, and potentially influence the long-term risk of complications such as biliary malignancy. This provides a potential microbiological explanation for the heterogeneous clinical progression observed in CC patients.
This study has several limitations. The most significant is the lack of a true healthy pediatric biliary microbiome for comparison. Obtaining biliary samples from healthy children is nearly impossible due to profound ethical and practical constraints. Therefore, we cannot definitively conclude whether the microbial features observed here are unique to CC or partially reflect the developing biliary microbiome in normal physiology. Future studies could partially address this by comparing samples from patients undergoing surgery for other non-infectious, non-inflammatory biliary conditions or by employing advanced non-invasive sampling techniques for indirect inference[
22,
23]. Additionally, as a cross-sectional design, this study reveals age-associated differences but cannot establish causality. Subsequent research combining longitudinal follow-up data and functional validation in gnotobiotic animal models is needed to elucidate the specific mechanistic roles of particular microbial functions in CC pathogenesis.
In conclusion, through large-scale, age-stratified, and functionally oriented analysis, this study maps the developmental dynamics of the biliary microbiome in pediatric CC. It establishes the core paradigm of taxonomic conservation with functional remodeling and identifies early childhood as a critical period for functional adaptation. These findings not only deepen the understanding of the microbial pathophysiology of CC but also suggest that future adjunctive microbiome-modulating strategies should consider patient age to achieve more precise management.
Author Contributions
Conceptualization, T.L., X.W. and M.D.; Data curation, X.W., R.D., A.M and Y. Z.; Investigation, X.W., A.M. and Y. Z.; Methodology, T.L.; Supervision, T.L.; Writing—review & editing, T.L. and X.W.; All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare that they have no conflict of interest.
References
- Khan, M Y; Rehman, A; Chaudhary, M A. Double Common Bile Duct Associated With Choledochal Cyst: A Rare Developmental Anomaly[J]. Cureus 2025. [Google Scholar] [CrossRef]
- Kim, M; Lam, S; Thirunavaya Kalathil, M; Paterson, A; Bowden, D; Liau, S. A new variant of gallbladder duplication mimicking a choledochal cyst: stepwise management of an unexpected surgical finding[J]. Annals of the Royal College of Surgeons of England 2025, 107(3). [Google Scholar] [CrossRef]
- Kumar, Y; Ram, A K; Menon, P; Kanojia, R P; Bhatia, A; Minz, R W. Cytomegalovirus and Epstein-Barr virus infections in pediatric patients of choledochal cyst[J]. IP Indian Journal of Immunology and Respiratory Medicine 2024, 9(2), 58–61. [Google Scholar] [CrossRef]
- Banerjee, T; Goswami, A G; Basu, S. Biliary microbiome and gallstones: A silent friendship[J]. World Journal of Gastrointestinal Surgery 2024, 16(11), 3395–3399. [Google Scholar] [CrossRef]
- Wang, Z M; Su, S; Ling-Hu, E Q; Chai, N L. Type III choledochal cyst confirmed by aspiration and treated with endoscopic fenestration plus internal drainage: A case report[J]. World Journal of Gastrointestinal Surgery 2025, 17(4), 412–417. [Google Scholar] [CrossRef]
- Bora, D J; Rajesh, P. Biliary anatomical variation in a patient with a large choledochal cyst malformation requiring complex biliary reconstruction surgery: a case report[J]. HPB:Hepato pancreato Biliary 2024, 26(Sup1), S465–S466. [Google Scholar] [CrossRef]
- Jung, E. The First Single-Port Robotic-Assisted Excision of Choledochal Cyst and Hepaticojejunostomy in Children[J]. International Journal of Medical Robotics & Computer Assisted Surgery 2025, 21(2). [Google Scholar]
- Murali, K; Dhua, A K; Jain, V; Yadav, D K; Goel, P; Agarwala, S; Kumar, R; Kandasamy, D; Potturu, M. Short-term Surgical Outcomes after Laparoscopic Excision of Choledochal Cyst and Roux-en-Y Hepaticojejunostomy in Children[J]. Journal of Indian Association of Pediatric Surgeons 2025, 30(3). [Google Scholar] [CrossRef] [PubMed]
- Chen, W; Li, L; Hao, X; Gao, R; Chi, Y; Zhao, J; Jin, S; Lu, Q. A comparative study of childhood versus adult-onset biliary complications following pediatric choledochal cyst surgery[J]. Pediatric Surgery International 2025, 41(1). [Google Scholar] [CrossRef]
- Chung, P; Wong, K; Yeung, F; Ma, M. Pre-operative external biliary drainage is associated with a higher chance of conversion in paediatric laparoscopic excision of choledochal cyst - a review of 17 years' experience [Oral presentation][J]. 2023. [Google Scholar]
- Oliveira-Filho, A G D; Farias, M G O; Márcia Alessandra Cavalaro Pereira da, S; Bento, A P N; Bustorff-Silva, J M. Long-term follow-up of laparoscopic treatment and hepaticojejunostomy without Roux-en-Y for choledochal cyst in children. Report of two cases and review of the literature[J]. International Journal of Surgery Case Reports 2025, 129. [Google Scholar] [CrossRef]
- Ai, C; Xie, X; Lv, Y; Zheng, Q; Yang, J; Xiang, B; Chen, J. LncRNA–mRNA coexpression analysis reveals distinct pathogenic mechanisms for subtypes of congenital biliary dilatation[J]. Journal of Hepato -- Biliary -- Pancreatic Sciences 2023, 30(11). [Google Scholar] [CrossRef]
- Liwinski, T; Heinemann, M; Schramm, C. The intestinal and biliary microbiome in autoimmune liver disease—current evidence and concepts[J]. Seminars in Immunopathology 2022, 44(4), 485–507. [Google Scholar] [CrossRef]
- Fuliang, L I; Qinshou, C; Junwei, H; Sibo, P; Longguang, H E. Application of modified cholangio-intestinal loop anastomosis for congenital cystic dilatation of the common bile duct and its influence on QOL score of patients[J]. China Modern Doctor 2019. [Google Scholar]
- Behrens, S; Sutton, T L; Patel, R; Schwantes, I R; O'Grady, J; Johnson, A; Grossberg, A; Lopez, C; Gilbert, E; Mayo, S. Neoadjuvant therapy does not impact the biliary microbiome in patients with pancreatic cancer[J]. Journal of Surgical Oncology 2023. [Google Scholar] [CrossRef] [PubMed]
- Plieskatt, J L; Deenonpoe, R; Mulvenna, J P; Krause, L; Sripa, B; Bethony, J M; Brindley, P J. Infection with the carcinogenic liver fluke Opisthorchis viverrini modifies intestinal and biliary microbiome[J]. Faseb Journal Official Publication of the Federation of American Societies for Experimental Biology 2013, 27(11), 4572–4584. [Google Scholar] [CrossRef]
- Jose, T. Isolated Cystic Duct Dilatation: Type VI Choledochal Cyst[J]. Kerala Surgical Journal 2024, 30(1), 30–31. [Google Scholar] [CrossRef]
- Kili, R M; Subai, I D; Kaya, A D. Correction to: Magnetic resonance urography in the assesment of congenital urinary system dilatation in pediatric patients[J]. Journal of Experimental & Clinical Medicine / Deneysel ve Klinik Tip Dergisi 2024, 41(2). [Google Scholar]
- Li, L; Guan, S; Xie, X; Tan, Y; Yu, J K; Zeng, J. Timing of minimally invasive surgery for neonates with congenital cystic dilatation[J]. 2019. [Google Scholar]
- Nakamata, A; Matsuki, M; Muramatsu, K. Special Feature: Imaging of infectious disease Imaging features of congenital cytomegalovirus infection[J]. Journal of Japanese Society of Pediatric Radiology 2024, 40(2). [Google Scholar]
- Elayyouti, M; Elshafey, A; Saied, A E; Albishbishy, M. Comparison of Efficacy and Safety of Hepatico-Duodenostomy versus Hepatico-Jejenostomy after Excision of Choledochal Cyst in Children[J]. Egyptian Journal of Hospital Medicine 2025, 98(1). [Google Scholar]
- Sánchez, M; Caballero, F; Massa, M; Janikow, C; Lada, P E. Congenital cystic dilatation of the intrahepatic bile ducts (Caroli's disease). Left hepatectomy[J]. Prensa Médica Argentina 2014, 100(7), 467–474. [Google Scholar]
- Senaha, Y; Kumori, K; Funabashi, N; Ishibashi, S; Manako, J; Ohkura, T; Nishi, T; Hayashi, H; Kawabata, Y; Tajima, Y. Preoperative Percutaneous Transhepatic Biliary Drainage for Congenital Biliary Dilatation in a Two-Year-Old Girl[J]. Journal of the Japanese Society of Pediatric Surgeons 2023, 59(1), 44–50. [Google Scholar]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).