Submitted:
12 December 2025
Posted:
16 December 2025
You are already at the latest version
Abstract
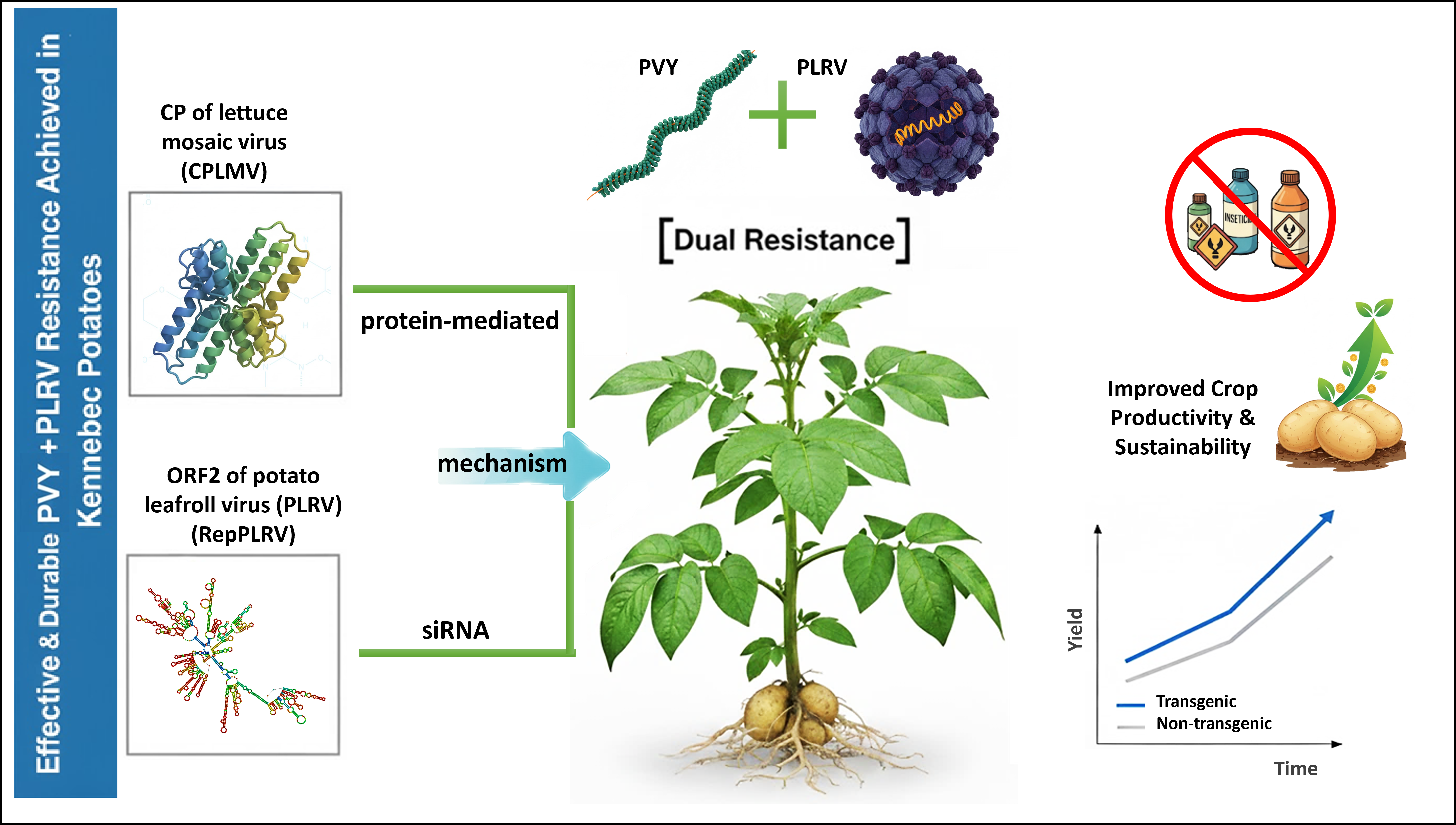
Keywords:
1. Introduction
2. Results
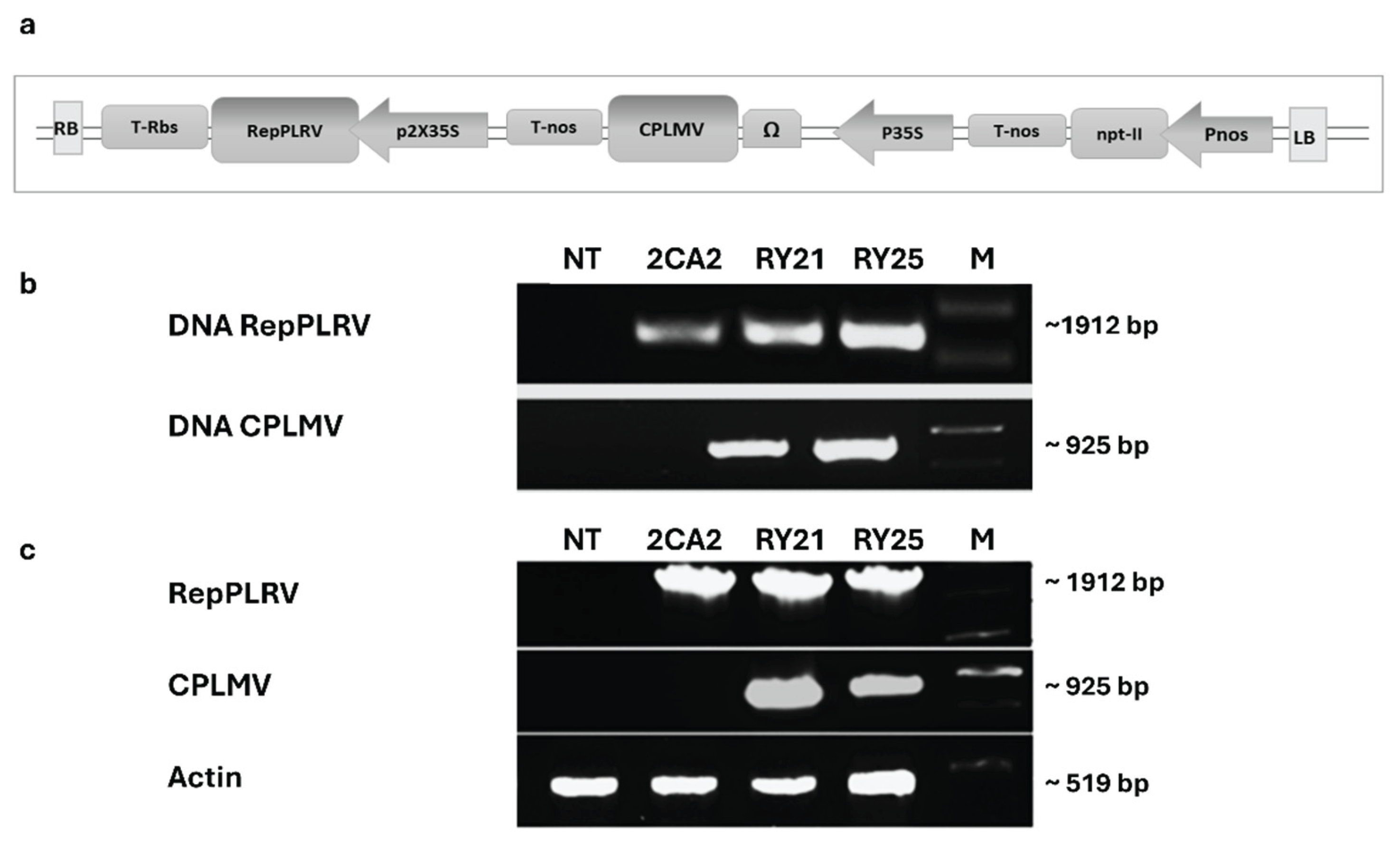
2.1. Generation of Transgenic Plants Engineered to Express the Coat Protein of Lettuce Mosaic Virus and the ORF2 Sequence of Potato Leafroll Virus
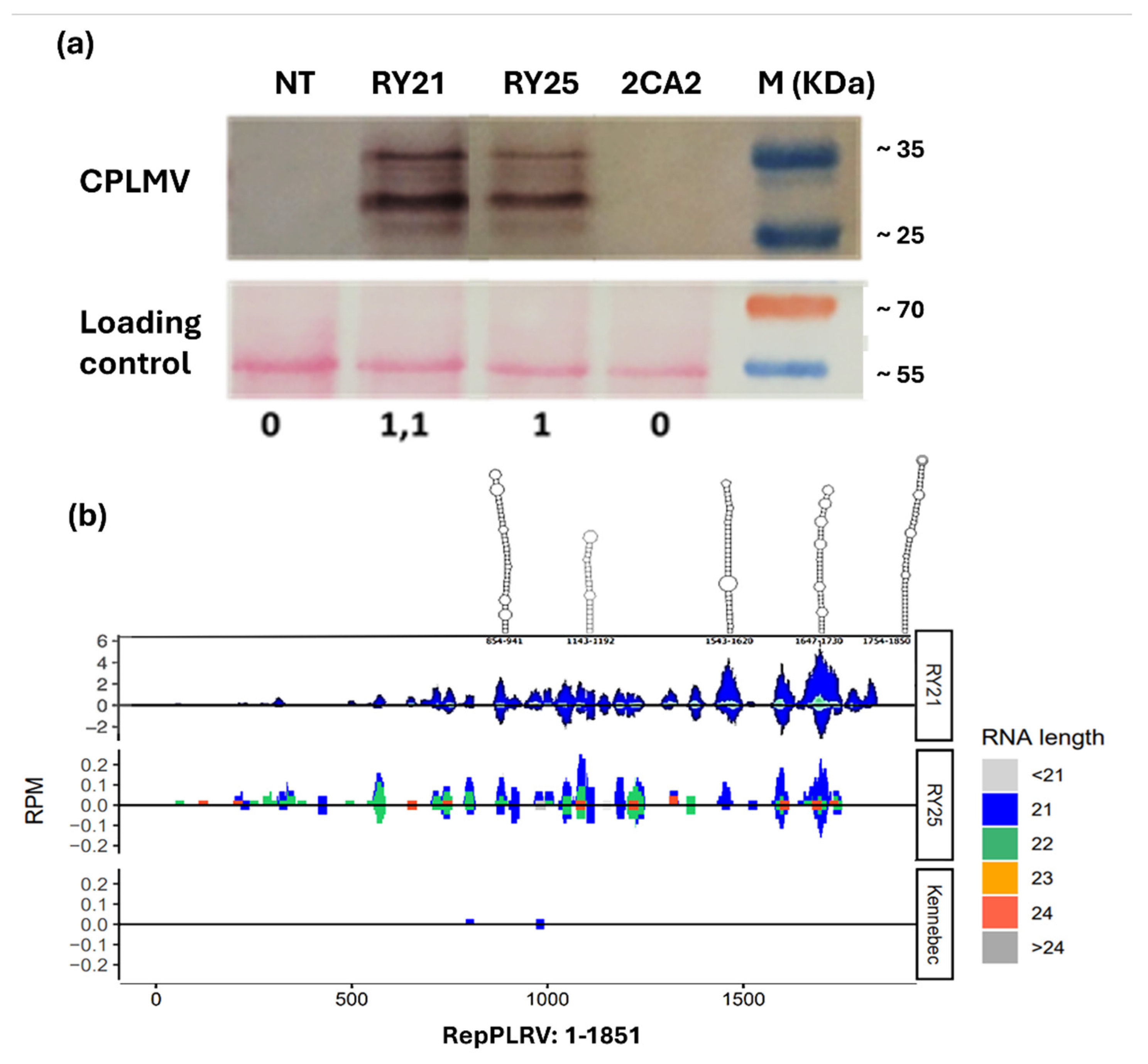
2.2. Assessment of Key Molecules Involved in the Resistance Mechanism of RY Transgenic Lines
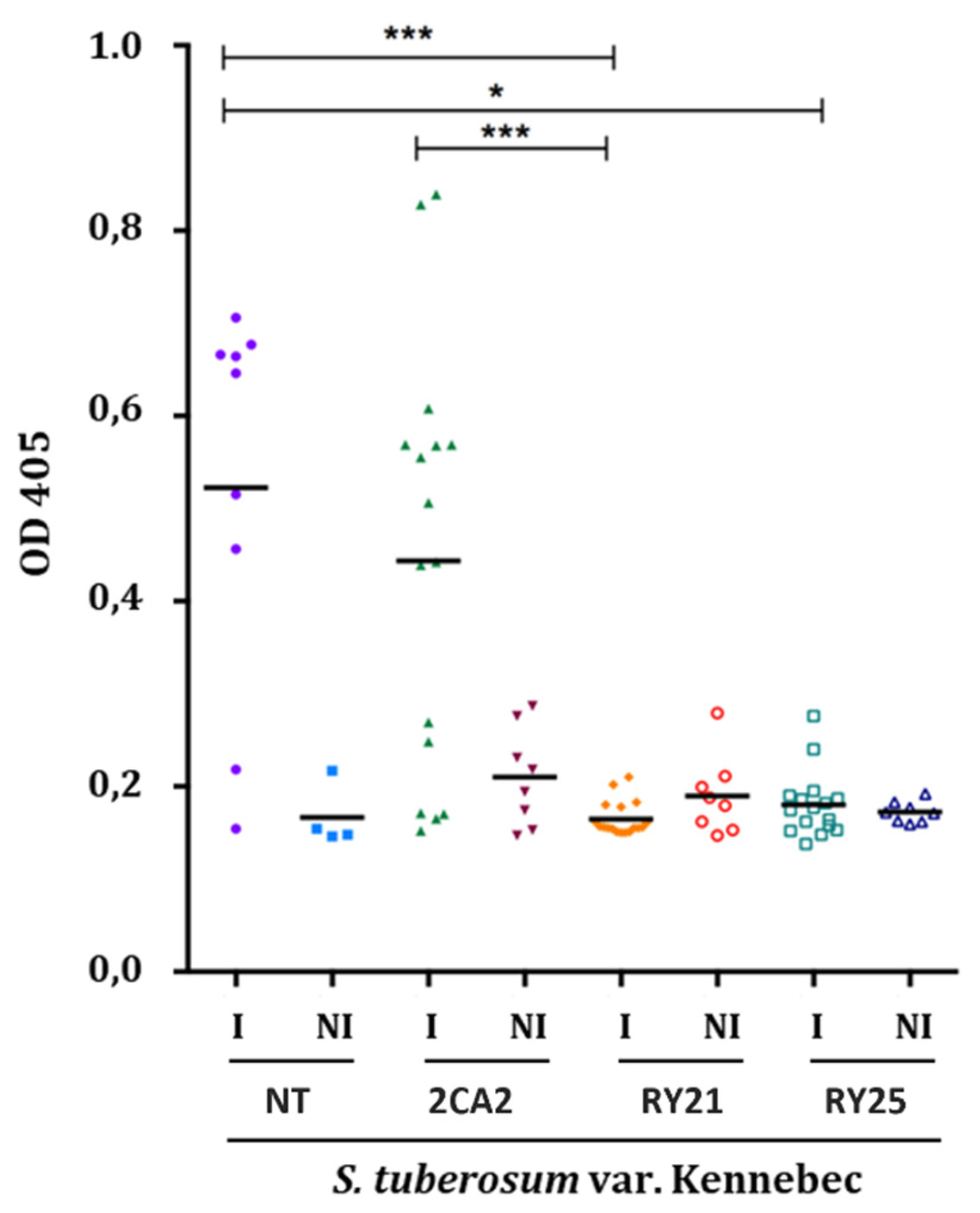
2.3. Assessment of PVY Viral Resistance of Transgenic Potato Lines Under Greenhouse Conditions
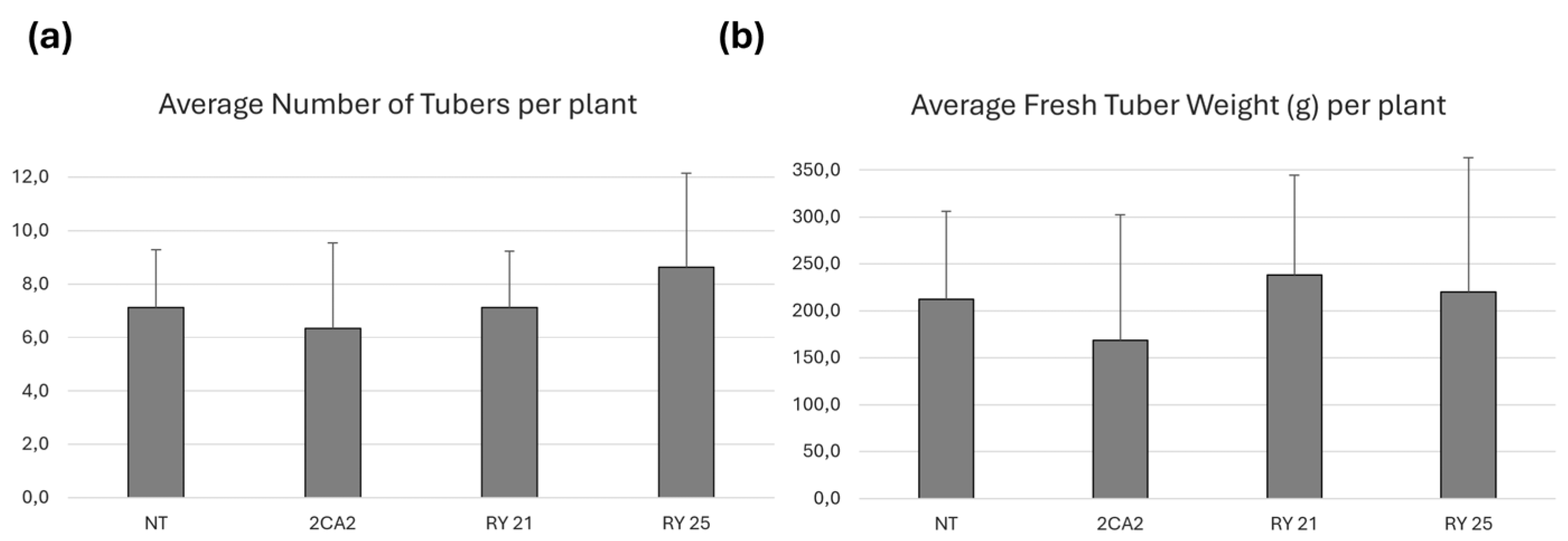
2.4. Evaluation of the Growth Parameters of Transgenic Potato Lines Under Greenhouse Conditions
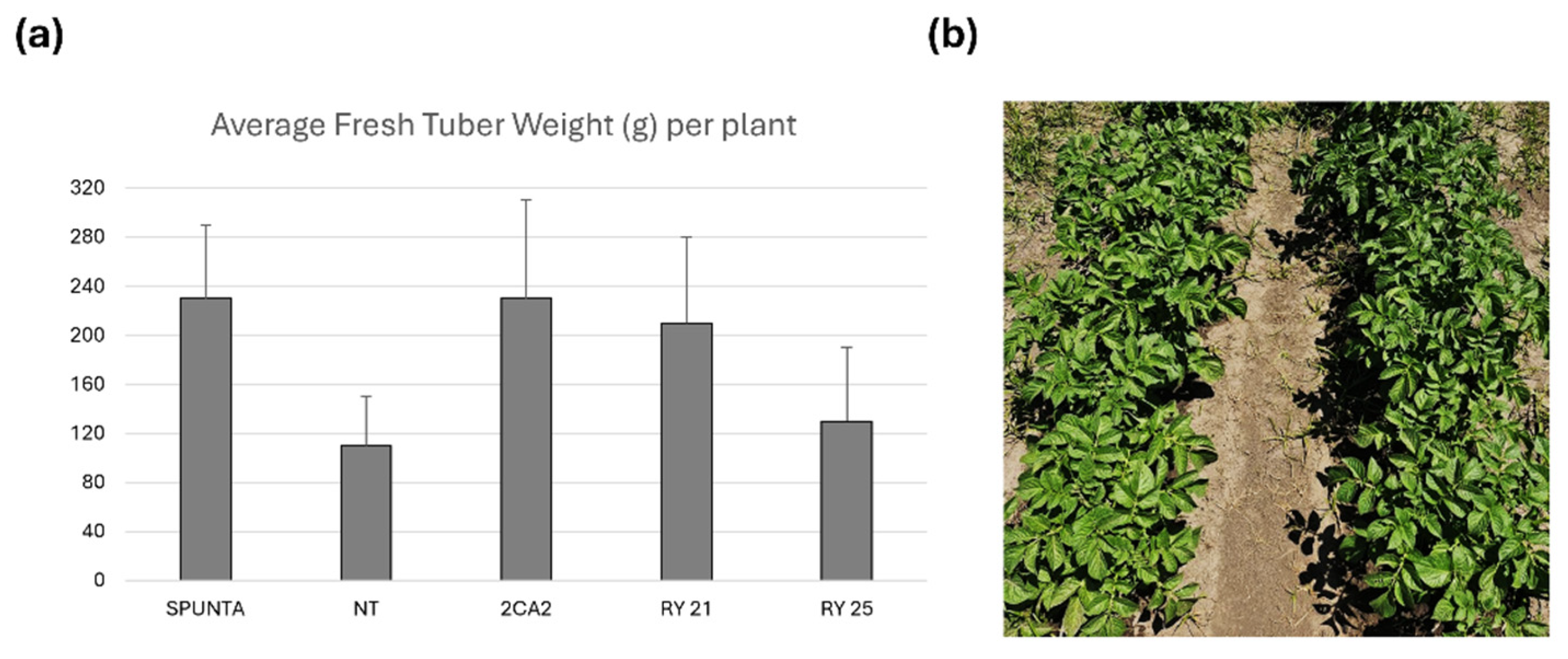
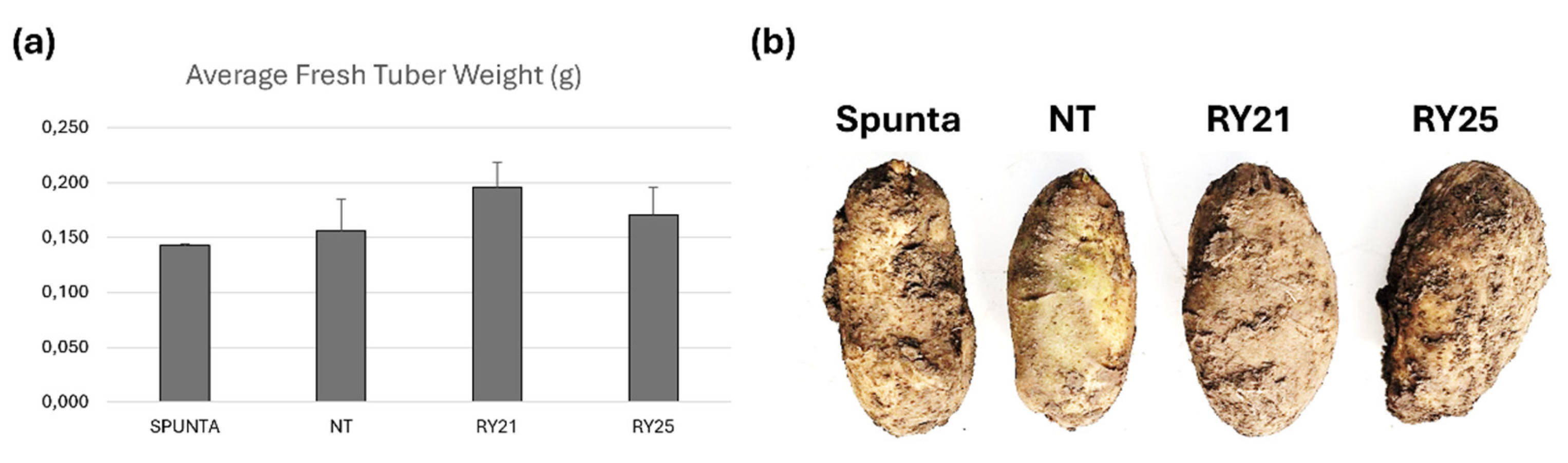
2.5. Assessment of Tuber Yield Under Field Conditions
2.6. Assessment of Viral Resistance of Transgenic Potato Lines in a Virus-Endemic Zone
3. Discussion
4. Materials and Methods
4.1. Construction of LMV-REP Plasmid
4.2. Potato cv. Kennebec Transformation and Regeneration
4.3. Plant DNA and RNA Extraction, cDNA Synthesis and RT-PCR
4.4. Plant Total Protein Extraction and Western Blot
4.5. Detection of Viral RNA
4.6. Small RNA Sequencing
4.7. Plant Inoculation and Assessment of Virus Resistance in Greenhouse
4.8. Field Trials Evaluations
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| PVY | potato virus Y |
| PVY | potato virus Y |
| PLRV | potato leafroll virus |
| CPLMV | lettuce mosaic virus capsid protein |
| TMV | Tobacco mosaic virus |
| PVX | Potato virus X |
| CMV | Cucumber mosaic virus |
| TRV | Tobacco rattle virus |
| NT | non-transgenic control plants |
| RT-PCR | reverse transcriptase Polymerase Chain Reaction |
| siRNA | small interfering RNA |
| NGS | next-generation sequencing |
| OD | Optical density |
References
- Noureen, A.; Khan, M.Z.; Amin, I.; Zainab, T.; Mansoor, S. CRISPR/Cas9-Mediated Targeting of Susceptibility Factor EIF4E-Enhanced Resistance Against Potato Virus Y. Front Genet 2022, 13. [Google Scholar] [CrossRef]
- Dominguez, J.P.J. Revista Mercados Agropecuarios 2021.
- Cortiello, M.; Milc, J.; Sanfelici, A.; Martini, S.; Tagliazucchi, D.; Caccialupi, G.; Ben Hassine, M.; Giovanardi, D.; Francia, E.; Caradonia, F. Genotype and Plant Biostimulant Treatments Influence Tuber Size and Quality of Potato Grown in the Pedoclimatic Conditions in Northern Apennines in Italy. Int J Plant Prod 2024, 18, 579–599. [Google Scholar] [CrossRef]
- Kreuze, J.F.; Souza-Dias, J.A.C.; Jeevalatha, A.; Figueira, A.R.; Valkonen, J.P.T.; Jones, R.A.C. Viral Diseases in Potato. In The Potato Crop: Its Agricultural, Nutritional and Social Contribution to Humankind; Springer International Publishing, 2019; pp. 389–430. ISBN 9783030286835. [Google Scholar]
- Loebenstein, G. Virus and Virus-like Diseases of Potatoes and Production of Seed-Potatoes; Loebenstein, G., Berger, P.H., Brunt, A.A., Lawson, R.H., Eds.; Springer Netherlands: Dordrecht, 2001; ISBN 978-94-010-3736-5. [Google Scholar]
- Valkonen, J.P.T. Elucidation of Virus-Host Interactions to Enhance Resistance Breeding for Control of Virus Diseases in Potato. Breed Sci 2015, 65, 69–76. [Google Scholar] [CrossRef]
- Wang, B.; Ma, Y.; Zhang, Z.; Wu, Z.; Wu, Y.; Wang, Q.; Li, M. Potato Viruses in China. Crop Protection 2011, 30, 1117–1123. [Google Scholar] [CrossRef]
- Hamm, P.B.; Hane, D.C. Effects of Seedborne Potato Leafroll Virus on Russet Norkotah Potato. Plant Dis 1999, 83, 1122–1124. [Google Scholar] [CrossRef] [PubMed]
- Chatzivassiliou, E.K.; Moschos, E.; Gazi, S.; Koutretsis, P.; Tsoukaki, M. INFECTION OF POTATO CROPS AND SEEDS WITH POTATO VIRUS Y AND POTATO LEAFROLL VIRUS IN GREECE. Journal of Plant Pathology 2008, 90, 253–261. [Google Scholar]
- Byarugaba, A.A.; Mukasa, S.B.; Barekye, A.; Rubaihayo, P.R. Interactive Effects of Potato Virus Y and Potato Leafroll Virus Infection on Potato Yields in Uganda. Open Agric 2020, 5, 726–739. [Google Scholar] [CrossRef]
- Barker, H.; Dale, M.F.B. Resistance to Viruses in Potato. In Natural Resistance Mechanisms of Plants to Viruses; Springer Netherlands: Dordrecht; pp. 341–366.
- Davidson, T.M.W. Assessing Resistance to Leafroll in Potato Seedlings. Potato Res 1973, 16, 99–108. [Google Scholar] [CrossRef]
- Thomas, P.E.; Kaniewski, W.K.; Lawson, E.C. Reduced Field Spread of Potato Leafroll Virus in Potatoes Transformed with the Potato Leafroll Virus Coat Protein Gene 1997.
- Taliansky, M.; Mayo, M.A.; Barker, H. Potato Leafroll Virus: A Classic Pathogen Shows Some New Tricks. Mol Plant Pathol 2003, 4, 81–89. [Google Scholar] [CrossRef]
- Palukaitis, P. Resistance to Viruses of Potato and Their Vectors. Plant Pathol J (Faisalabad) 2012, 28, 248–258. [Google Scholar] [CrossRef]
- Velásquez, A.C.; Mihovilovich, E.; Bonierbale, M. Genetic Characterization and Mapping of Major Gene Resistance to Potato Leafroll Virus in Solanum Tuberosum Ssp. Andigena. Theoretical and Applied Genetics 2007, 114, 1051–1058. [Google Scholar] [CrossRef]
- Mihovilovich, E.; Alarcón, L.; Pérez, A.L.; Alvarado, J.; Arellano, C.; Bonierbale, M. High Levels of Heritable Resistance to Potato Leafroll Virus (PLRV) in Solanum Tuberosum Subsp. Andigena. Crop Sci 2007, 47, 1091–1103. [Google Scholar] [CrossRef]
- Orbegozo, J.; Solorzano, D.; Cuellar, W.J.; Bartolini, I.; Roman, M.L.; Ghislain, M.; Kreuze, J. Marker-Free PLRV Resistant Potato Mediated by Cre-LoxP Excision and RNAi. Transgenic Res 2016, 25, 813–828. [Google Scholar] [CrossRef]
- Valkonen, J.P.T.; Gebhardt, C.; Zimnoch-Guzowska, E.; Watanabe, K.N. Resistance to Potato Virus Y in Potato. In Potato virus Y: biodiversity, pathogenicity, epidemiology and management; Springer International Publishing: Cham, 2017; pp. 207–241. [Google Scholar]
- Eck, J. Van Genome Editing and Plant Transformation of Solanaceous Food Crops; 2018. [Google Scholar]
- Radcliffe; Ragsdale. Aphid-Transmitted Potato Viruses The Importance of Understanding Vector Biology; 2002. [Google Scholar]
- Mowry, T.M. Insecticidal Reduction of Potato Leafroll Virus Transmission by Myzus Persicae. Annals of Applied Biology 2005, 146, 81–88. [Google Scholar] [CrossRef]
- Dupuis, B.; Cadby, J.; Goy, G.; Tallant, M.; Derron, J.; Schwaerzel, R.; Steinger, T. Control of Potato Virus Y (PVY) in Seed Potatoes by Oil Spraying, Straw Mulching and Intercropping. Plant Pathol 2017, 66, 960–969. [Google Scholar] [CrossRef]
- Watanabe, K. Potato Genetics, Genomics, and Applications. Breed Sci 2015, 65, 53–68. [Google Scholar] [CrossRef] [PubMed]
- Beachy, R.N.; Tumer, N.E. COAT PROTEIN-MEDIATED RESISTANCE AGAINST VIRUS INFECTION 1990, Vol. 28.
- Majumdar, A.; Sharma, A.; Belludi, R. Natural and Engineered Resistance Mechanisms in Plants against Phytoviruses. Pathogens 2023, 12. [Google Scholar] [CrossRef]
- Dinant S, B.F.K.C.A.-M.S.A.J. Heterologous Resistance to Potato Virus Y in Transgenic Tobacco Plants Expressing the Coat Protein Gene of Lettuce Mosaic Potyvirus. Phytopathology 1993, 818–824. [Google Scholar] [CrossRef]
- Dinant, S.; Maisonneuve, B.; Albouy, J.; Chupeau, Y.; Chupeau, M.-C.; Bellec, Y.; Gaudefroy, F.; Kusiak, C.; Souche, S.; Robaglia, C.; et al. Coat Protein Gene-Mediated Protection in Lactuca Sativa against Lettuce Mosaic Potyvirus Strains; Kluwer Academic Publishers, 1997; Vol. 3. [Google Scholar]
- Hassairi, A.; Masmoudi, K.; Albouy, J.; Robaglia, C.; Jullien, M.; Ellouz, R. Transformation of Two Potato Cultivars ‘Spunta’ and ‘Claustar’ (Solanum Tuberosum) with Lettuce Mosaic Virus Coat Protein Gene and Heterologous Immunity to Potato Virus Y. Plant Science 1998, 136, 31–42. [Google Scholar] [CrossRef]
- Rovere, C.V.; Asurmendi, S.; Hopp, H.E. Transgenic Resistance in Potato Plants Expressing Potato Leaf Roll Virus (PLRV) Replicase Gene Sequences Is RNA-Mediated and Suggests the Involvement of Post-Transcriptional Gene Silencing * 2001, Vol. 146.
- Savenkov, E.I.; Valkonen, J.P.T. Coat Protein Gene-Mediated Resistance to Potato Virus A in Transgenic Plants Is Suppressed Following Infection with Another Potyvirus. Journal of General Virology 2001, 82, 2275–2278. [Google Scholar] [CrossRef] [PubMed]
- Cecilia Vazquez Rovere*, M. del V. and H.E.H. RNA-Mediated Virus Resistance; 2002.
- Hajdukiewicz, P.; Svab, Z.; Maliga, P. The Small, VersatilepPZP Family OfAgrobacterium Binary Vectors for Plant Transformation. Plant Mol Biol 1994, 25, 989–994. [Google Scholar] [CrossRef]
- María Pilar Barrios Barón Caracterización de Un Aislamiento Local Del Potato Leafroll Virus (PLRV) y Evaluación de Eventos Transgénicos de Papa Para Resistencia a PLRV y PVY. Tesis presentada para optar al título de;Doctor de la Universidad de Buenos Aires en el área CIENCIAS BIOLOGICAS, 2019.
- Kenzhebekova, R.; Pozharskiy, A.; Adilbayeva, K.; Gritsenko, D. Molecular Mechanisms of Potato Plant–Virus–Vector Interactions. Plants 2025, 14, 2282. [Google Scholar] [CrossRef] [PubMed]
- Loebenstein, G.; Gaba, V. Viruses of Potato. In; 2012; pp. 209–246.
- Gottula, J.; Fuchs, M. Toward a Quarter Century of Pathogen-Derived Resistance and Practical Approaches to Plant Virus Disease Control. In; 2009; pp. 161–183.
- Chung, B.N.; Yoon, J.-Y.; Palukaitis, P. Engineered Resistance in Potato against Potato Leafroll Virus, Potato Virus A and Potato Virus Y. Virus Genes 2013, 47, 86–92. [Google Scholar] [CrossRef]
- Mondal, S.; Gray, S.M. Sequential Acquisition of Potato Virus Y Strains by Myzus Persicae Favors the Transmission of the Emerging Recombinant Strains. Virus Res 2017, 241, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Missiou, A.; Kalantidis, K.; Boutla, A.; Tzortzakaki, S.; Tabler, M.; Tsagris, M. Generation of Transgenic Potato Plants Highly Resistant to Potato Virus Y (PVY) through RNA Silencing. Molecular Breeding 2004, 14, 185–197. [Google Scholar] [CrossRef]
- Bukovinszki, Á.; Götz, R.; Johansen, E.; Maiss, E.; Balázs, E. The Role of the Coat Protein Region in Symptom Formation on Physalis Floridana Varies between PVY Strains. Virus Res 2007, 127, 122–125. [Google Scholar] [CrossRef]
- Bravo-Almonacid, F.; Rudoy, V.; Welin, B.; Segretin, M.E.; Bedogni, M.C.; Stolowicz, F.; Criscuolo, M.; Foti, M.; Gomez, M.; López, M.; et al. Field Testing, Gene Flow Assessment and Pre-Commercial Studies on Transgenic Solanum Tuberosum Spp. Tuberosum (Cv. Spunta) Selected for PVY Resistance in Argentina. Transgenic Res 2012, 21, 967–982. [Google Scholar] [CrossRef] [PubMed]
- Kaniewski, W.K.; Thomas, P.E. A Two-Step ELISA for Rapid, Reliable Detection of Potato Viruses. Am Potato J 1988, 65, 561–571. [Google Scholar] [CrossRef]
- Kaniewski and Thomas Kaniewski and Thomas. 2004.
- Arif, M.; Azhar, U.; Arshad, M.; Zafar, Y.; Mansoor, S.; Asad, S. Engineering Broad-Spectrum Resistance against RNA Viruses in Potato. Transgenic Res 2012, 21, 303–311. [Google Scholar] [CrossRef]
- Wen-jun, S.; Forde, B.G. Efficient Transformation of Agrobacterium Spp. by High Voltage Electroporation. Nucleic Acids Res 1989, 17, 8385–8385. [Google Scholar] [CrossRef]
- Dellaporta, S.L.; Wood, J.; Hicks, J.B. A Plant DNA Minipreparation: Version II. Vol. 1. [CrossRef]
- Isogai, A.; Kato, Y. Preparation of Polyuronic Acid from Cellulose by TEMPO-Mediated Oxidation. Cellulose 1998, 5, 153–164. [Google Scholar] [CrossRef]
| Line | NT | 2CA2 | RY21 | RY25 |
|---|---|---|---|---|
| Plants (Tubers) | 21 | 18 | 31 | 22 |
| PLRV-positive Tubers | 5 | 4 | 0 | 0 |
| PVY-positive Tubers | 5 | 4 | 0 | 0 |
| PLRV and PVY-incidence | 23.81 % | 22.22 % | 0% | 0% |
| Fisher’s Exact Test (vs. NT) | — | 1 | 0.0078 * | 0,0211* |
| Line | NT | 2CA2 | RY21 | RY25 |
|---|---|---|---|---|
| Plants (Tubers) | 89 (275) | 73 (176) | 49 (148) | 72 (249) |
| PLRV-positive Tubers | 0 / 275 | 0 / 176 | 0 / 148 | 0 / 249 |
| PVY-positive Tubers | 36 / 275 | 26 / 176 | 0 / 148 | 0 / 249 |
| PVY-incidence | 22.5 % | 31.5 % | 0% | 0% |
| Fisher’s Exact Test (vs. NT) | — | 0.2143 | 0.0001 * | < 0.00001 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




