1. Introduction
Pleural mesothelioma (PM) is an uncommon but highly aggressive malignancy associated with poor survival outcomes [
1]. A universally accepted treatment strategy has yet to be established, and prognosis remains dismal despite the use of multimodal approaches. Even with combinations of surgery, systemic chemotherapy, and radiotherapy, the 5-year overall survival rarely exceeds 10% [
1,
2]. Moreover, the role of surgery itself has become increasingly controversial. A recent phase III randomized controlled trial demonstrated that median overall survival was not improved with surgical intervention; instead, survival was slightly shorter than with chemotherapy alone, and serious adverse events occurred significantly more frequently in the surgical arm [
3]. These findings underscore the limitations and morbidity associated with aggressive surgical approaches in PM.
Systemic therapy has undergone substantial evolution in the past decade. Immune checkpoint inhibitors (ICIs), particularly nivolumab and ipilimumab, have extended median survival from approximately 12 months to nearly 18 months [
4]. While these results represent important progress, ICIs are also associated with a considerable rate of immune-related toxicities. More than one-fifth of patients discontinue treatment due to adverse events, highlighting the difficulties of sustaining long-term systemic therapy in this population [
4,
5,
6]. Radiation therapy has also advanced, particularly with the introduction of intensity-modulated radiation therapy (IMRT). Although small-cohort studies have suggested improved median survival times of up to 24 months [
7,
8], IMRT following radical surgery often results in substantial declines in pulmonary function, including approximately 30% reductions in forced vital capacity, forced expiratory volume in 1 second, and lung diffusing capacity [
9]. These functional declines have significant consequences for quality of life in a patient population already burdened by disease-related respiratory compromise.
Despite these therapeutic advances, recurrence continues to be a major therapeutic obstacle, with reported rates ranging from 26% to 63% even after conventional multimodal treatment [
10,
11,
12]. Recurrent PM is particularly difficult to manage because prior treatments often limit the feasibility of additional surgery or high-dose radiation, and systemic therapies may offer limited incremental benefit in the salvage setting. As a result, there is growing interest in identifying alternative, less invasive local treatments capable of providing meaningful tumor control while minimizing patient morbidity.
Radiofrequency ablation (RFA) is a minimally invasive technique that has demonstrated robust safety and efficacy across a wide range of tumors and anatomical sites [
13,
14,
15,
16]. Compared with surgery, RFA offers substantial advantages: it can be performed percutaneously, typically under conscious sedation and local anesthesia, and is associated with lower complication rates and shorter recovery periods. RFA is widely used worldwide, most notably for liver tumors—where local recurrence rates range from 2% to 41% [
17] —and for primary and metastatic lung tumors, with recurrence rates of 8%–21% [
18]. Mortality associated with RFA remains below 1%, and major complication rates are generally around 10% [
19,
20].
Despite its broad applications, the role of RFA in pleural mesothelioma has been minimally explored. To date, only a single case report has described its use in PM [
21], and no case-series studies have examined clinical outcomes systematically. Given the limited therapeutic options available for recurrent PM and the growing need for locally effective, minimally invasive treatments, RFA represents a potentially valuable strategy.
In the present study, we retrospectively analyzed outcomes of patients with recurrent PM treated with percutaneous RFA. Our objectives were to evaluate the safety profile of RFA, assess its efficacy in achieving local tumor control, and explore its potential contribution to prolonging survival in this challenging patient population.
2. Materials and Methods
2.1. Study Design
This retrospective single-center study was conducted with approval from the institutional review board of Hyogo Medical University (approval number: 202209-042), in accordance with the tenets of the Declaration of Helsinki. A waiver of patient consent for study enrollment was granted by the review board, but written informed consent was obtained from all patients for the RFA procedure.
2.2. Patient Selection
From July 2019 to June 2025, 14 consecutive patients underwent Computed Tomography (CT)-guided RFA for recurrent PM and were included in the analysis (
Table 1). The cohort consisted of 13 men (92.9%) and 1 woman (7.1%), with a median age of 69 (range, 54–77) years. Histological diagnoses were epithelioid in 11 patients (78.6%), biphasic in 2 (14.3%), and sarcomatoid in 1 (7.1%). All patients had previously received systemic therapy and 12 (85.7%) had also undergone surgical resection. Recurrence was established using Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography (FDG-PET/CT) or based on progressive growth on serial CT in 9 patients (64.3%) and using biopsy in 5 patient (35.7%).
Exclusion criteria included patients with poor performance status (ECOG ≥3), severe comorbidities that contraindicated RFA, uncontrolled coagulopathy, or tumors deemed unsuitable for percutaneous ablation (e.g., lesions adjacent to major vessels). Patients who declined to provide informed consent for the procedure were also excluded.
Seven patients (50%) presented with multiple lesions (2–6 tumors). In total, 25 tumors were treated: 12 in the chest wall, 11 in the lung, and 1 in the liver. Tumor size ranged from 0.5 to 7.0 (median, 1.8) cm.
2.3. RFA Procedure
A total of 23 RFA sessions were performed. One patient with a 7.0-cm tumor required three sessions; all others were treated in a single session. Procedures were performed on an inpatient basis under local anesthesia and moderate sedation. Fentanyl citrate was administered for analgesia and lidocaine was used for local anesthesia. Cefazolin was given prophylactically and continued for 2 days after treatment.
Real-time CT fluoroscopy (Aquilion One, Canon, Japan) guided the placement of 17- or 18-gauge internally cooled electrodes. Real-time CT fluoroscopy (Aquilion One, Canon, Otawara, Japan) guided the placement of 17- or 18-gauge internally cooled electrodes (Cool-Tip, Covidien, Boulder, CO, USA; or VIVA, STARmed, Gyeonggi, Republic of Korea).An 18-gauge electrode was preferentially used for lung lesions. The electrode number and exposure length were determined according to tumor size and morphology, with multiple electrodes used for tumors >3.0 cm. RF energy was delivered with an impedance-switching algorithm until three consecutive impedance spikes (≥30 W increase above baseline) occurred or a maximum of 12 min elapsed.
2.4. Follow-Up
The patients underwent clinical examination, laboratory testing, and contrast-enhanced chest CT every 3 months. Follow-up continued until patient death or July 2025, whichever occurred first. The median follow-up duration was 22 (range, 2–72) months.
2.5. Endpoints and Definitions
Technical success was defined as complete tumor coverage within the ablation zone or disappearance of enhancement on CT within 1 week [
22]. Adverse events (AEs) were classified according to the Society of Interventional Radiology guidelines [
23]. Local tumor progression was defined as regr owth at the margin of an ablated lesion, whereas new lesions outside the ablation zone were categorized as distant metastases.
2.6. Statistical Analysis
Local progression and survival outcomes were estimated using the Kaplan–Meier method. Statistical analyses were performed with EZR software, version 1.61 (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). A p-value of <0.05 was considered statistically significant [
24].
3. Results
3.1. Safety
Technical success was achieved in all 23 sessions (100%). No procedures were interrupted due to pain or intraprocedural complications. One patient developed a refractory skin ulcer and another experienced a small subcutaneous hematoma not requiring intervention, corresponding to major and minor complication rates of 4.3% each.
3.2. Tumor Control and Recurrence
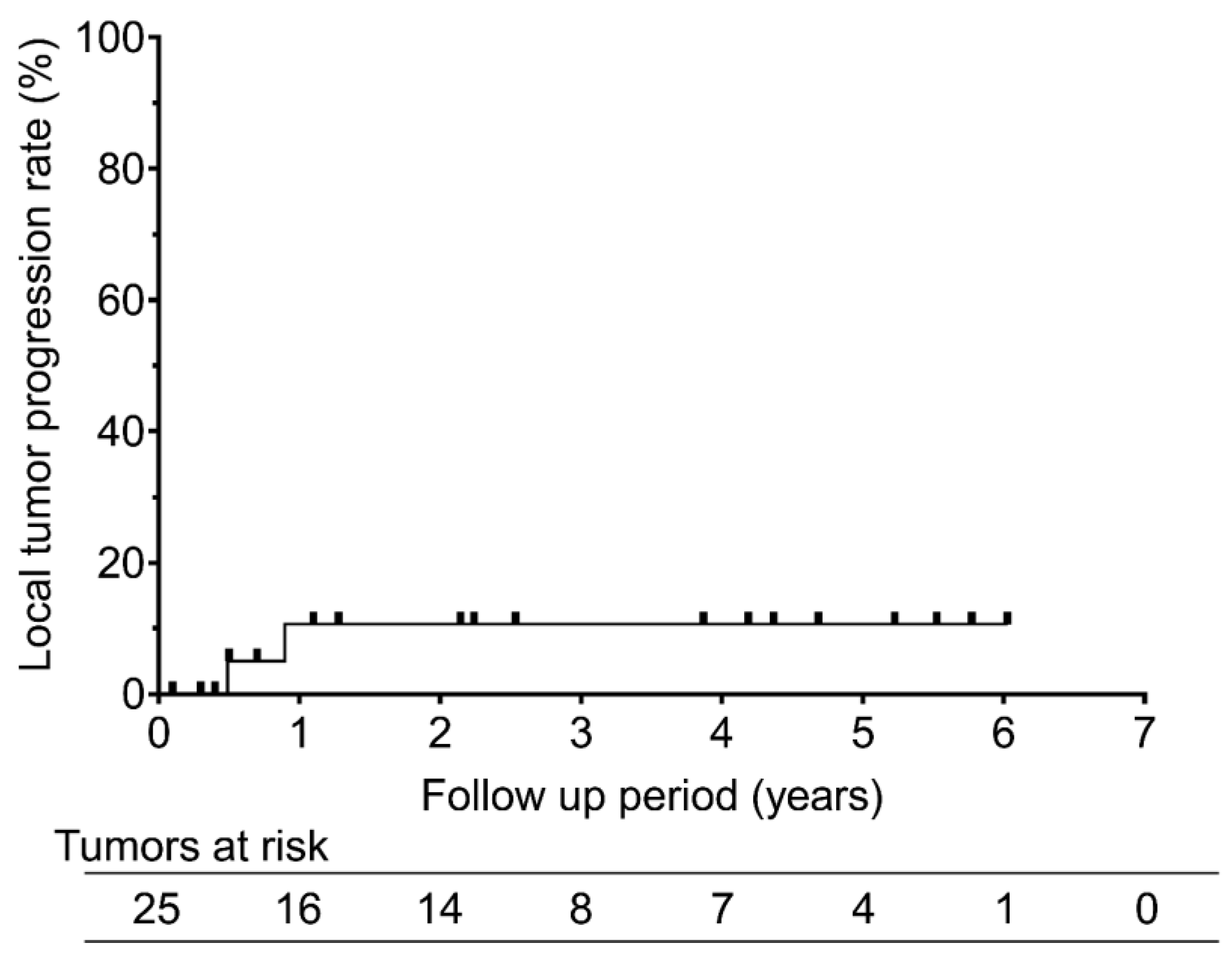
At a median follow-up of 22 months, 7 of the 14 patients (50%) experienced recurrence: two patients showed both local and distant progression and five showed distant metastases alone. The median time to progression was 11.7 (range, 5–20) months. Local progression occurred in 2 of the 25 treated tumors (8.0%), resulting in 1- and 2-year local progression rates of 10.6% (
Figure 1).
After recurrence, 5 patients (71.4%) received systemic therapy (chemotherapy or immune checkpoint inhibitors) and 2 patients received best supportive care.
3.3. Survival Outcomes
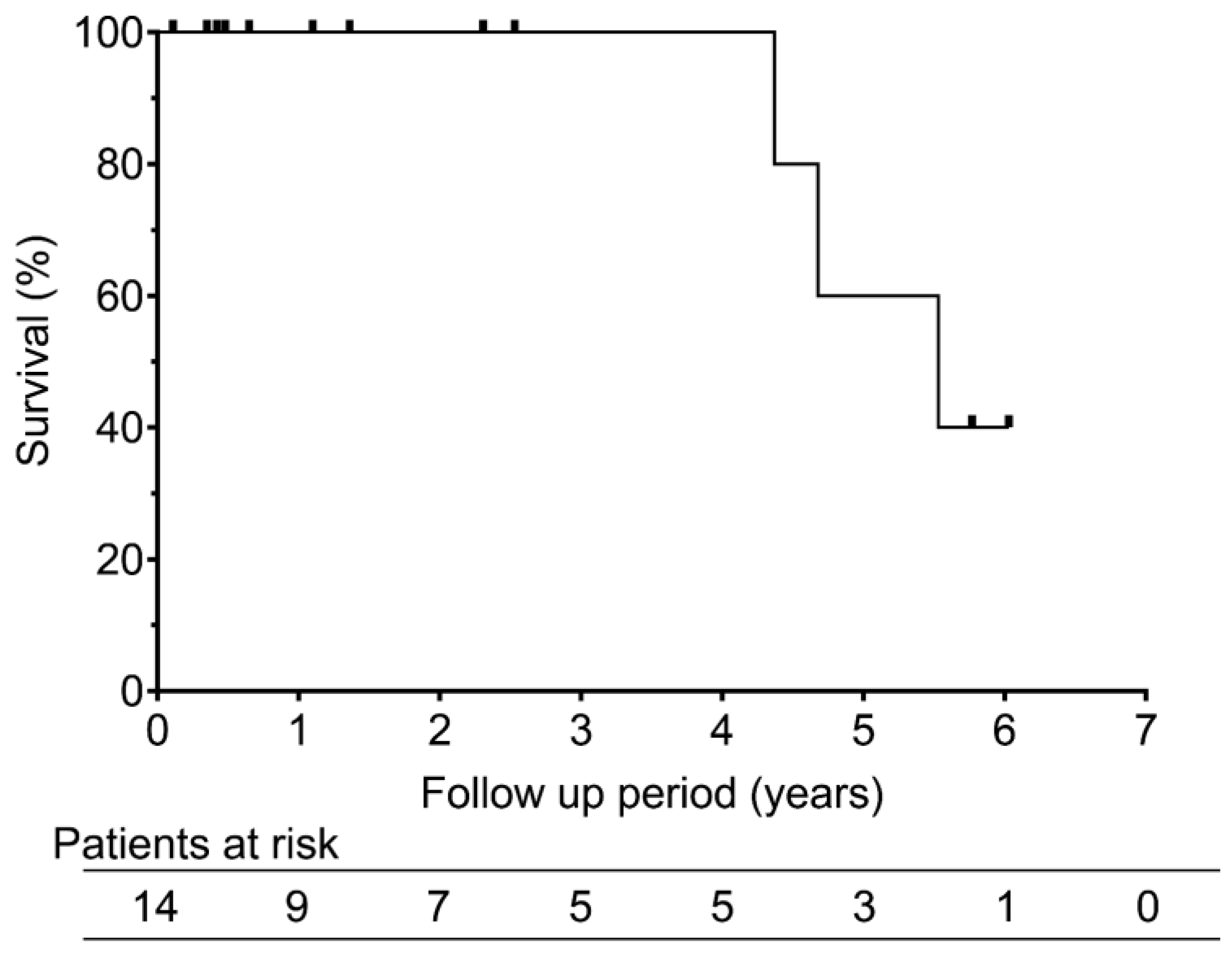
Three patients (21.4%) died: two from tumor progression and one from gastrointestinal perforation during chemotherapy for esophageal carcinoma. The overall survival rates were 100% at 1 and 3 years and 60% at 5 years (
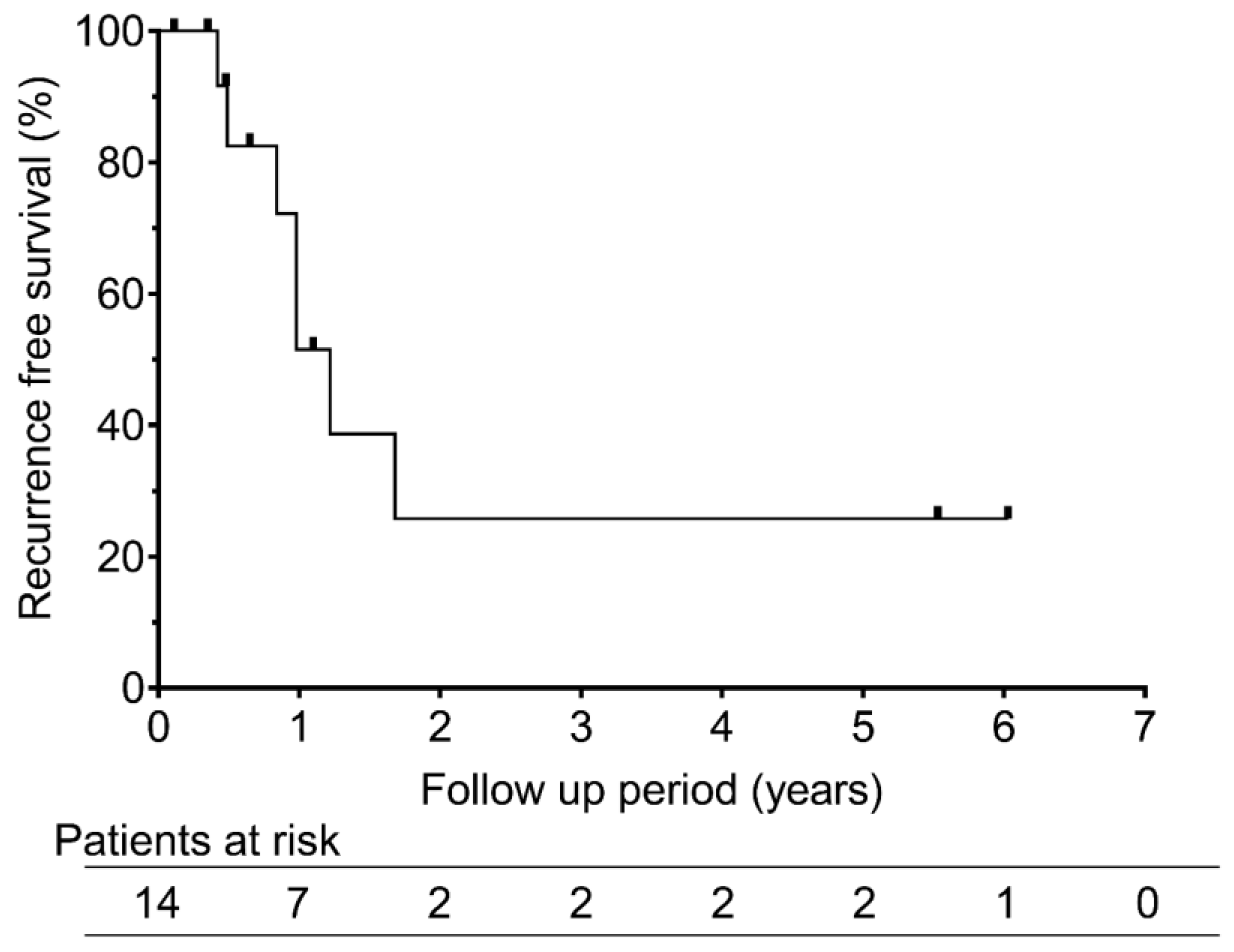
Figure 2). The median overall survival duration was 5.5 years. Recurrence-free survival was 51.6% at 1 year and 25.8% at both 3 and 5 years, with a median of 1.2 years (
Figure 3).
4. Discussion
This study demonstrated that percutaneous RFA for recurrent PM is technically feasible and associated with a low incidence of serious complications. The major AE rate of 4.3% was lower than that typically reported for lung RFA (10%–30%) [
25,
26] and comparable to that for small chest wall series (<10%) [
27,
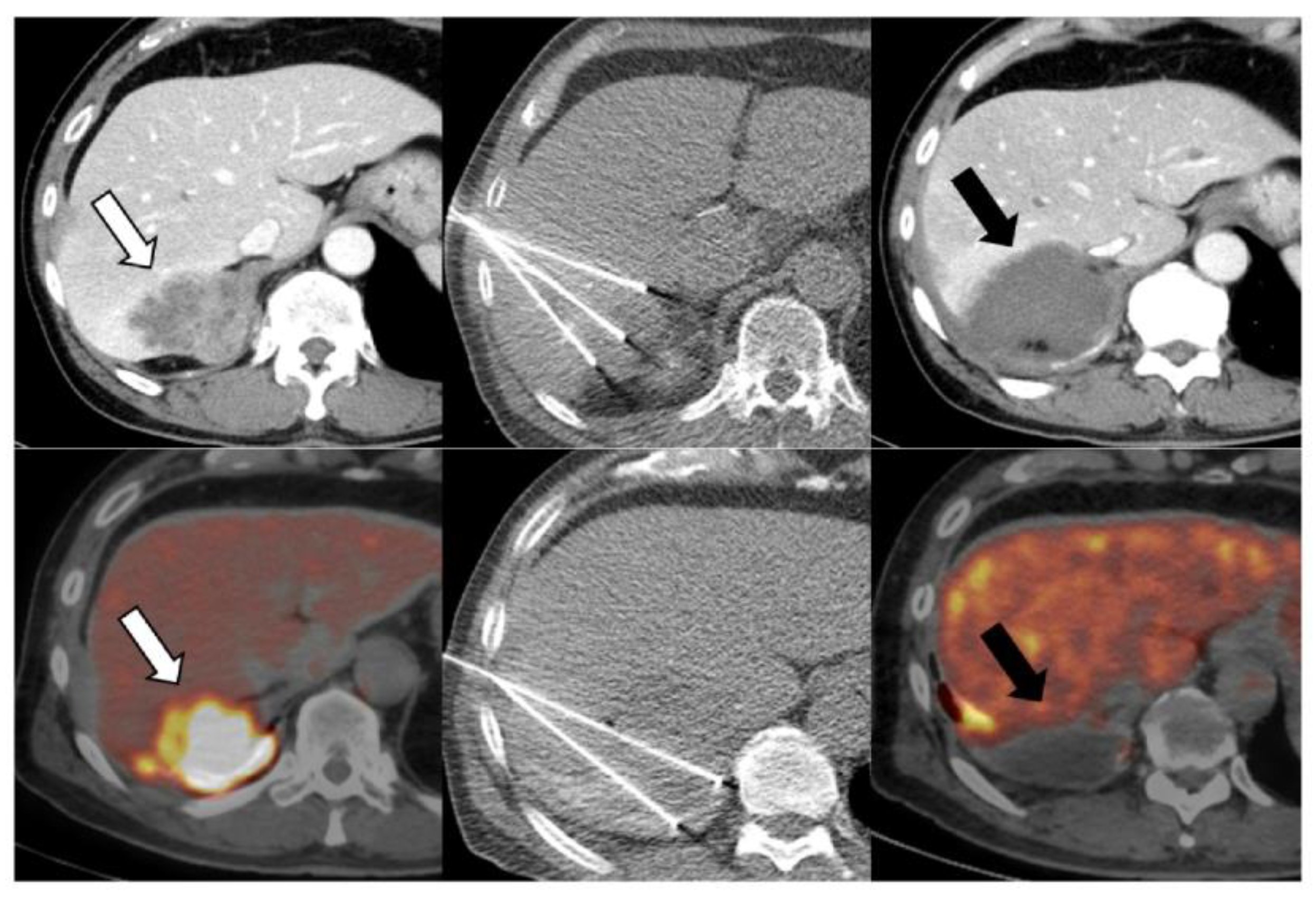
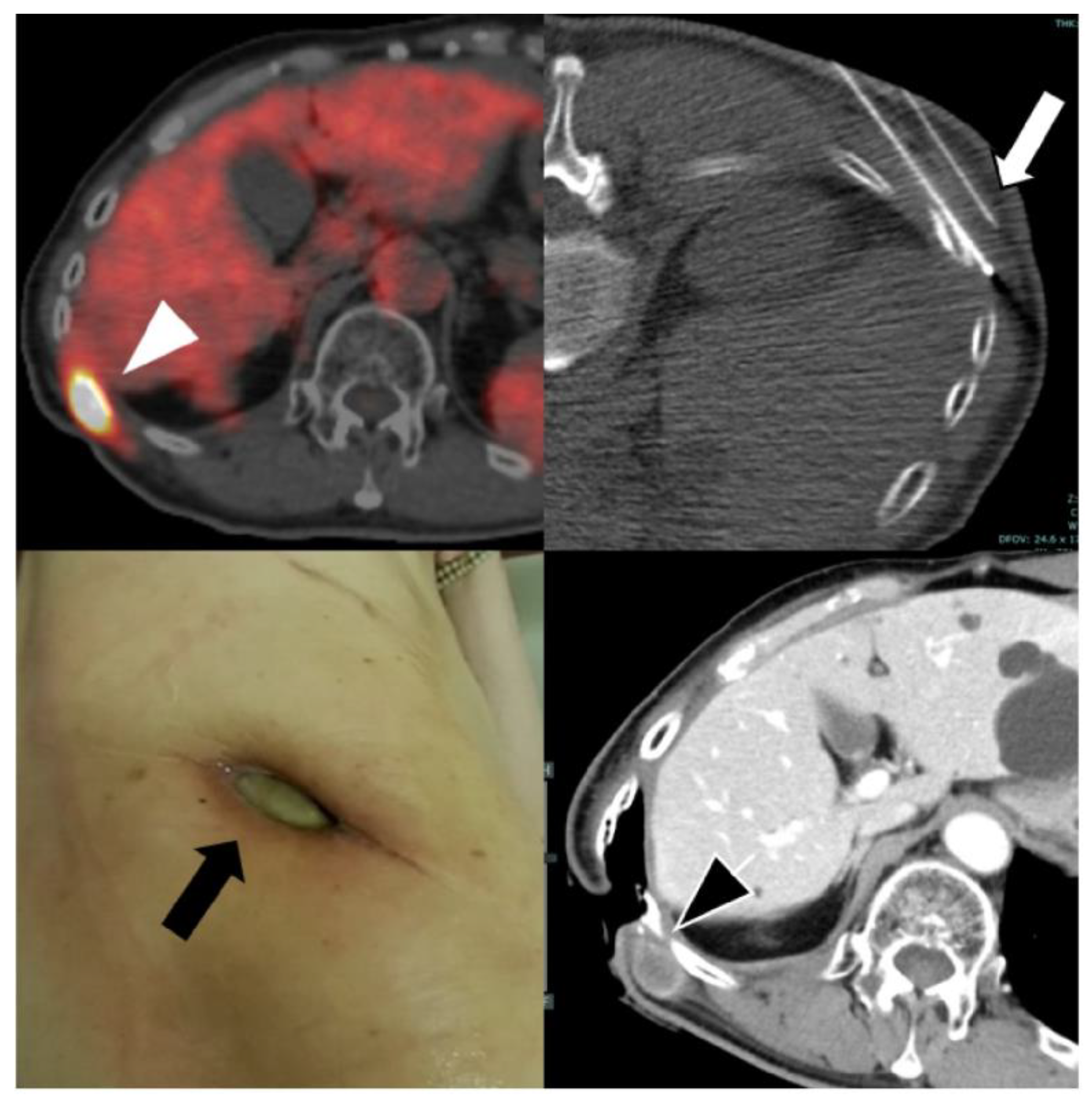
28]. Representative imaging findings are shown in
Figure 4 and
Figure 5. Nonetheless, the occurrence of a severe skin ulcer highlights the need for careful procedural planning and preventive measures when treating chest wall lesions.
Local tumor control was encouraging, with only 8% of treated tumors progressing, consistent with reported outcomes for lung RFA (10–20%) [
26] and musculoskeletal ablation [
29]. However, distant recurrence was frequent, occurring in half of the patients—a finding that aligns with published recurrence rates after surgery or systemic therapy (41–63%) [
10,
11]. These results reinforce the systemic nature of PM and the need to integrate local ablation with effective systemic therapy.
Radiation therapy remains a central component of multimodal PM management. IMRT, frequently employed after surgery, has demonstrated meaningful clinical benefit. Gomez et al. reported a median OS of 14.7 months following extrapleural pneumonectomy and IMRT [
30], while Rimner et al. demonstrated median PFS and OS of 12.4 and 23.7 months, respectively, after chemotherapy and pleurectomy/decortication followed by IMRT [
31]. Despite these advances, recurrence remains a major clinical challenge; a Japanese nationwide survey reported a 2-year local control rate of approximately 60% [
16], emphasizing the unmet need for additional local modalities with durable disease control.
In this context, RFA may serve as a complementary or alternative option to radiotherapy. RFA is minimally invasive, repeatable, and preserves pulmonary function—an important advantage in previously irradiated or surgically treated patients. Tochio et al. reported a case in which 50 lung tumors were ablated across 20 RFA sessions in a single patient [
32], illustrating the feasibility of repeated treatment. The ability to deliver multiple ablations without cumulative pulmonary toxicity contrasts sharply with IMRT, for which repeated administration is often limited by lung function decline and radiation-induced fibrosis.
Evidence from other organs supports the applicability of RFA in PM. Local recurrence rates after RFA range from 2–41% in hepatocellular carcinoma [
33]and 8–21% in lung tumors [
34], with mortality rates generally <1% [
35]. The 92% tumor control rate observed in the present study is comparable to, or slightly more favorable than, outcomes in these organs, despite the additional technical challenges posed by pleural anatomy.
Emerging data also suggest that RFA may potentiate systemic immunotherapy. Preclinical studies indicate that thermal ablation induces immunogenic cell death, releases tumor antigens, and enhances dendritic cell activation, thereby priming the immune system and potentially augmenting the efficacy of immune checkpoint inhibitors (ICIs) [
36]. As ICIs have become standard first-line therapy for unresectable PM, combining RFA with ICI therapy—particularly for oligoprogressive lesions—may provide additional therapeutic benefit. Although clinical data remain limited, studies in other malignancies (HCC, lung cancer) offer a compelling rationale for exploring this strategy in PM.
Similarly, multimodal local therapy integrating RFA with radiotherapy (IMRT or SBRT) warrants investigation. For tumors >3 cm or those close to critical structures, RFA alone may be insufficient; consolidative SBRT following incomplete ablation, or RFA applied after partial radiotherapy response, may enhance local control while minimizing toxicity. Because thermal ablation produces a sharply demarcated necrotic zone, combining RFA with IMRT could also reduce high-dose radiation volumes, offering a lung-sparing alternative for previously irradiated patients.
Future directions may include biomarker-guided selection between RFA and radiotherapy. Imaging biomarkers such as ADC values, contrast enhancement patterns, or FDG-PET metabolic activity could potentially predict thermal versus radiation sensitivity. Furthermore, genomic features associated with PM aggressiveness—such as BAP1 loss or CDKN2A deletion—may correlate with responsiveness to local ablative therapies. Prospective studies integrating imaging and genomic profiling could refine patient selection and optimize treatment planning.
The rarity of PM must be considered when interpreting the clinical impact of our findings. Although the cohort is small, this study represents the largest case series to date evaluating RFA specifically for recurrent PM. For rare malignancies, even limited case series can provide valuable evidence and shape future research directions. The high local control rate, favorable survival, and low toxicity profile observed in this study support the potential incorporation of RFA into the multidisciplinary management algorithm for recurrent PM.
Nevertheless, several limitations must be acknowledged. The retrospective, single-institution design and small sample size may introduce selection bias. Follow-up duration varied, possibly underestimating late recurrences. In addition, treatment efficacy may be reduced for larger tumors (>3 cm), suggesting a role for combination strategies incorporating radiotherapy, ICIs, or surgery. Future multicenter prospective studies are needed to validate our findings, standardize procedural techniques, evaluate combination regimens such as RFA–ICI or RFA–IMRT, and identify biomarkers predictive of optimal local therapy.
In summary, RFA appears to be a feasible, safe, and effective minimally invasive treatment for recurrent PM, offering durable local tumor control and complementing established treatments such as radiotherapy and systemic immunotherapy. These findings warrant further investigation and contribute to expanding therapeutic possibilities in this challenging disease.
5. Conclusions
In conclusion, our study demonstrates that percutaneous RFA is a safe, feasible, and effective local treatment option for patients with recurrent pleural mesothelioma, achieving high rates of technical success and local tumor control with minimal morbidity. In an era in which immune checkpoint inhibitors have begun to reshape the therapeutic landscape of mesothelioma, the addition of RFA as a minimally invasive, repeatable modality may further expand treatment possibilities for patients with limited options. The combination or sequencing of RFA with systemic immunotherapy or advanced radiotherapy techniques holds particular promise, raising the possibility that the historically pessimistic outlook associated with this disease may gradually improve. Although our sample size is inherently limited due to the rarity of this malignancy, the present study provides important preliminary evidence supporting the incorporation of RFA into multidisciplinary care. Larger, prospective, and multi-institutional studies are warranted to clarify its optimal role and to define how this modality can best be integrated with emerging systemic therapies.
Author Contributions
Conceptualization, H.K. and K.Y.; Methodology, H.K.; Investigation, K.M., T.S., R.W. and A.O.; Writing – Original Draft Preparation, H.K.; Writing – Review & Editing, K.K., H.T, M.H, D.F., T.M., S.F. and T.K.; Supervision, K.K., T.K. and K.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of Hyogo Medical University (protocol code 202209-042; date of approval: 2 May 2023).
Informed Consent Statement
A waiver of patient consent for study enrollment was granted by the Institutional Review Board of Hyogo Medical University because this was a retrospective study using anonymized data, and the requirement for written informed consent was deemed unnecessary. All patients, however, provided informed consent for the RFA procedure itself.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy and ethical restrictions.
Acknowledgments
The authors would like to express their sincere gratitude to the clinical departments and the Interventional Radiology team of Hyogo Medical University for their technical assistance and patient care during the conduct of this study.
Conflicts of Interest
Daichi Fujimoto reports grants and personal fees from AstraZeneca KK and Boehringer Ingelheim Japan Inc., as well as personal fees from Ono Pharmaceutical Co. Ltd., Bristol-Myers Squibb Co. Ltd., Taiho Pharmaceutical Co. Ltd., Chugai Pharmaceutical Co. Ltd., MSD KK, Eli Lilly Japan KK, Kyowa Kirin, Daiichi Sankyo Co. Ltd., Nippon Kayaku Co. Ltd., Janssen Pharmaceutical KK, and Novartis Pharma KK, outside the submitted work. Koichiro Yamakado has served as a consultant for Boston Scientific Japan, Century Medical Inc., Covidien Japan Inc., and Canon Medical. The other authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AE |
Adverse event |
| CT |
Computed Tomography |
| FDG-PET/CT |
Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography |
| PM |
Pleural mesothelioma |
| RFA |
Radiofrequency ablation |
References
- Khan, A.M.H.; Anwer, S.H.; Sayed, S.; Mansha, M.A.; Kamran, Y.B.; Khursheed, A.; Haroon, F.; Soomro, N.H.; Idrees, R.; Abbasi, A.N. Comprehensive clinical overview of malignant pleural mesothelioma. Respir. Med. 2024, 222, 107511. [Google Scholar] [CrossRef]
- Bertin, B.; Zugman, M.; Schvartsman, G. The current treatment landscape of malignant pleural mesothelioma and future directions. Cancers 2023, 15, 5808. [Google Scholar] [CrossRef]
- Rintoul, R.C.; Arnold, D.T.; Hughes, V.; et al. Surgery in Malignant Pleural Mesothelioma (MARS-2): A Randomized Controlled Trial. N. Engl. J. Med. 2023, 389, 1955–1965. [Google Scholar]
- Baas, P.; Scherpereel, A.; Nowak, A.K.; Fujimoto, N.; Peters, S.; Tsao, A.S.; Mansfield, A.S.; Popat, S.; Jahan, T.; Antonia, S.; Oulkhouir, Y.; Bautista, Y.; Cornelissen, R.; Greillier, L.; Grossi, F.; Kowalski, D.; Rodríguez-Cid, J.; Aanur, P.; Oukessou, A.; Baudelet, C.; Zalcman, G. First-line nivolumab plus ipilimumab in unresectable malignant pleural mesothelioma (CheckMate 743): A multicentre, randomised, open-label, phase 3 trial. Lancet 2021, 397, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Janes, S.M.; Alrifai, D.; Fennell, D.A. Perspectives on the treatment of malignant pleural mesothelioma. N. Engl. J. Med. 2021, 385, 1207–1218. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Li, M.; Wang, Z.; An, D.; Li, B. Adverse events of immunotherapy in non-small cell lung cancer: A systematic review and network meta-analysis. Int. Immunopharmacol. 2022, 102, 108353. [Google Scholar] [CrossRef] [PubMed]
- Rimner, A.; Zauderer, M.G.; Gomez, D.R.; et al. Phase II Study of Hemithoracic Intensity-Modulated Radiation Therapy After Pleurectomy/Decortication and Chemotherapy for Malignant Pleural Mesothelioma. J. Clin. Oncol. 2016, 34, 2761–2768. [Google Scholar] [CrossRef]
- Gomez, D.R.; Hong, D.S.; Allen, P.K.; et al. Patterns of Failure, Toxicity, and Survival After Hemithoracic Intensity-Modulated Radiation Therapy for Malignant Pleural Mesothelioma. J. Thorac. Oncol. 2013, 8, 238–245. [Google Scholar] [CrossRef]
- Allen, A.M.; Czerminska, M.; Janne, P.A.; et al. Fatal Toxicity Associated with High-Dose Thoracic Radiotherapy for Lung Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2015, 93, 613–622. [Google Scholar] [CrossRef]
- Nakamura, A.; Takuwa, T.; Hashimoto, M.; Kuroda, A.; Nakamichi, T.; Matsumoto, S.; Kondo, N.; Kijima, T.; Yamakado, K.; Hasegawa, S. clinical outcomes with recurrence after pleurectomy/decortication for malignant pleural mesothelioma. Ann. Thorac. Surg. 2020, 109, 1537–1543. [Google Scholar] [CrossRef]
- Nowak, A.K.; Jackson, A.; Sidhu, C. Management of advanced pleural mesothelioma—At the crossroads. JCO Oncol. Pract. 2022, 18, 116–124. [Google Scholar] [CrossRef]
- Nakanishi-Imai, M.; Murai, T.; Onishi, M.; Mouri, A.; Komiyama, T.; Omura, M.; Kudo, S.; Miyamoto, A.; Hoshino, M.; Ogawa, S.; Ohashi, S.; Koizumi, M.; Omagari, J.; Mayahara, H.; Karasawa, K.; Okumura, T.; Shibamoto, Y. Survey of malignant pleural mesothelioma treatment in Japan: Patterns of practice and clinical outcomes in tomotherapy facilities. J. Radiat. Res. (Tokyo) 2022, 63, 281–289. [Google Scholar] [CrossRef]
- Hasegawa, T.; Takaki, H.; Kodama, H.; Yamanaka, T.; Nakatsuka, A.; Sato, Y.; Takao, M.; Katayama, Y.; Fukai, I.; Kato, T.; Tokui, T.; Tempaku, H.; Adachi, K.; Matsushima, Y.; Inaba, Y.; Yamakado, K. Three-year survival rate after radiofrequency ablation for surgically resectable colorectal lung metastases: A prospective multicenter study. Radiology 2020, 294, 686–695. [Google Scholar] [CrossRef]
- Kinota, N.; Takaki, H.; Kobayashi, K.; Kako, Y.; Kodama, H.; Ogasawara, A.; Maruyama, M.; Takahagi, M.; Taniguchi, J.; Moriyama, T.; Yokoyama, H.; Komoto, H.; Kunimoto, R.; Yoshimura, N.; Yamakado, K. Clinical experience of percutaneous radiofrequency ablation using an Arfa RF ABLATION SYSTEMⓇ in various organs. Interv. Radiol. Higashimatsuyama-Shi Jpn. 2022, 7. [Google Scholar] [CrossRef]
- Fujimori, M.; Takaki, H.; Nakatsuka, A.; Uraki, J.; Yamanaka, T.; Hasegawa, T.; Shiraki, K.; Takei, Y.; Sakuma, H.; Yamakado, K. Survival with up to 10-year follow-up after combination therapy of chemoembolization and radiofrequency ablation for the treatment of hepatocellular carcinoma: Single-center experience. J. Vasc. Interv. Radiol. 2013, 24, 655–666. [Google Scholar] [CrossRef] [PubMed]
- Mimura, H.; Arai, Y.; Yamakado, K.; Sone, M.; Takeuchi, Y.; Miki, T.; Gobara, H.; Sakuhara, Y.; Yamamoto, T.; Sato, Y.; Kanazawa, S. Phase I/II study of radiofrequency ablation for malignant renal tumors: Japan Interventional Radiology in Oncology Study Group 0701. Cardiovasc. Intervent. Radiol. 2016, 39, 717–723. [Google Scholar] [CrossRef]
- Livraghi, T.; Meloni, F.; Solbiati, L.; et al. Complications of Microwave Ablation for Liver Tumors: Results of a Multicenter Study. Radiology 2012, 265, 409–417. [Google Scholar] [CrossRef]
- Abtin, F.G.; Suh, R.; Kwan, S.W.; et al. Image-Guided Thermal Ablation of Lung Malignancies: A Clinical Review. J. Thorac. Imaging 2019, 34, 130–142. [Google Scholar] [CrossRef]
- Charboneau, J.W.; Callstrom, M.R.; Reading, C.C.; et al. CT- and US-Guided Percutaneous Radiofrequency Ablation of Renal Cell Carcinoma. Radiographics 2013, 33, 197–209. [Google Scholar]
- Healey, T.T.; Sofocleous, C.T.; Maybody, M.; et al. Radiofrequency Ablation of Lung Tumors: Review of the Literature and Update on Current Practice. Semin. Intervent. Radiol. 2011, 28, 307–317. [Google Scholar] [CrossRef]
- Tsuchiya, N.; Yoshizako, T.; Yoshida, R.; et al. Radiofrequency Ablation for Malignant Pleural Mesothelioma: A Case Report. Clin. J. Gastroenterol. 2018, 11, 150–154. [Google Scholar]
- Ahmed, M.; Solbiati, L.; Brace, C.L.; Breen, D.J.; Callstrom, M.R.; Charboneau, J.W.; Chen, M.-H.; Choi, B.I.; de Baère, T.; Dodd, G.D.; Dupuy, D.E.; Gervais, D.A.; Gianfelice, D.; Gillams, A.R.; Lee, F.T.; Leen, E.; Lencioni, R.; Littrup, P.J.; Livraghi, T.; Lu, D.S.; McGahan, J.P.; Meloni, M.F.; Nikolic, B.; Pereira, P.L.; Liang, P.; Rhim, H.; Rose, S.C.; Salem, R.; Sofocleous, C.T.; Solomon, S.B.; Soulen, M.C.; Tanaka, M.; Vogl, T.J.; Wood, B.J.; Goldberg, S.N.; International Working Group on Image-Guided Tumor Ablation; Interventional Oncology Sans Frontières Expert Panel; Technology Assessment Committee of the Society of Interventional Radiology; Standard of Practice Committee of the Cardiovascular and Interventional Radiological Society of Europe. Image-guided tumor ablation: standardization of terminology and reporting criteria—A 10-year update. Radiology 2014, 273, 241–260. [Google Scholar] [CrossRef] [PubMed]
- Baerlocher, M.O.; Nikolic, B.; Sze, D.Y. Adverse event classification: clarification and validation of the society of interventional radiology specialty–specific system. J. Vasc. Interv. Radiol. 2023, 34, 1–3. [Google Scholar] [CrossRef]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013, 48, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Kashima, M.; Yamakado, K.; Takaki, H.; Kodama, H.; Yamada, T.; Uraki, J.; Nakatsuka, A. Complications after 1000 lung radiofrequency ablation sessions in 420 patients: A single center’s experiences. AJR Am. J. Roentgenol. 2011, 197, W576–580. [Google Scholar] [CrossRef]
- Kodama, H.; Yamakado, K.; Taniguchi, J.; Ogasawara, A.; Kako, Y.; Takaki, H.; Kobayashi, K. Advances in percutaneous lung tumor therapy: 2019 update. Chin. Clin. Oncol. 2019, 8, 64. [Google Scholar] [CrossRef]
- M, H.; X, Z.; J, Z. Radiofrequency ablation (RFA) for palliative treatment of painful non-small cell lung cancer (NSCLC) rib metastasis: experience in 12 patients. Thorac. Cancer 2015, 6. [Google Scholar] [CrossRef]
- Yamakado, K.; Matsumine, A.; Nakamura, T.; Nakatsuka, A.; Takaki, H.; Matsubara, T.; Asanuma, K.; Sudo, A.; Sugimura, Y.; Sakuma, H. Radiofrequency ablation for the treatment of recurrent bone and soft-tissue sarcomas in non-surgical candidates. Int. J. Clin. Oncol. 2014, 19. [Google Scholar] [CrossRef]
- Efthymiou, E.; Charalampopoulos, G.; Velonakis, G.; Grigoriadis, S.; Kelekis, A.; Kelekis, N.; Filippiadis, D. Ablative techniques for sarcoma metastatic disease: Current role and clinical applications. Medicina 2023, 59, 485. [Google Scholar] [CrossRef]
- Gomez, D.R.; Hong, D.S.; Allen, P.K.; et al. Intensity-Modulated Radiotherapy After Extrapleural Pneumonectomy: Clinical Outcomes and Toxicities. J. Thorac. Oncol. 2013, 8(1), 112–118. [Google Scholar]
- Rimner, A.; Zauderer, M.G.; Gomez, D.R.; et al. Phase II Study of Hemithoracic Intensity-Modulated Pleural Radiation Therapy After Chemotherapy for Malignant Pleural Mesothelioma. J. Clin. Oncol. 2016, 34(23), 2761–2768. [Google Scholar] [CrossRef] [PubMed]
- Tochio, N.; Tanabe, M.; Kawaguchi, T.; et al. Long-Term Outcome of Repeated Radiofrequency Ablation for Multiple Lung Metastases in One Patient. Intern. Med. 2010, 49(21), 2421–2425. [Google Scholar]
- Nodwell, R.; Lafreniere, R.; Ding, L.; et al. Radiofrequency Ablation of Liver Tumors: Long-Term Results and Factors Affecting Outcomes. HPB (Oxford) 2011, 13(10), 761–766. [Google Scholar] [CrossRef]
- Yamakado, K.; Takao, M.; Yamada, T.; et al. Radiofrequency Ablation for Pulmonary Metastases: Outcomes and Prognostic Factors. J. Vasc. Interv. Radiol. 2019, 30(3), 349–356. [Google Scholar] [CrossRef]
- de Baère, T.; Palussière, J.; Aupérin, A.; et al. Radiofrequency Ablation of Lung Tumors: Factors Influencing Local Tumor Progression. Radiology 2013, 268(1), 267–275. [Google Scholar]
- Kuribayashi, K.; Hirano, J. True Benefits of Immune Checkpoint Inhibitors in the Treatment of Malignant Pleural Mesothelioma in Japan. Anticancer Res. 2024, 44(10), 4135–4145. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).