Submitted:
05 December 2025
Posted:
08 December 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Cell Culture
Treatments
Measurement of Reactive Oxygen Species (ROS)
Measurement of Mitochondrial Membrane Potential (ΔΨm)
Assessment of Cell Viability
Assessment of Mitophagy via Mitochondrial-Lysosomal Colocalization
RNA Extraction and Quantitative Real-Time PCR
| gene | forward primer | reverse primer | product size |
| GAPDH | 5’-aggtgaaggtcggagtca | 5’-cgttctcagccttgacggt | 186bp |
| BAX | 5’-aggatcgagcagggcgaatg | 5’-ccaatgtccagcccatga | 330bp |
| BCL2 | 5’-tgtgtggagagcgtcaacc | 5’-tcaaacagaggccgcatg | 161bp |
| CASP3 | 5’-gcgaatcaatggactctgga | 5’-tgctccttttgctgtgatct | 267bp |
| NRF2 | 5’-gcgacggaaagagtatgagc | 5’-acgtagccgaagaaacctca | 296bp |
| DPR1 | 5’-agaaaatggggtggaagcaga | 5’-aggcaccttggtcattcctg | 220bp |
| FIS1 | 5’-tgtccaagagcacgcagt | 5’-gccttgtcaatgagccgct | 262bp |
| MFN1 | 5’-tgaagcactttgtgctggct | 5’-ccgagatagcacctcacca | 197bp |
| MFN2 | 5’-cgttgtaccagcttcctgg | 5’-gctcaaacttggtcttcactgc | 241bp |
| OPA1 | 5’-ggaaatgattgcccaagctc | 5’-atggtctcagggctaacggt | 231bp |
| PINK1 | 5’-tcaatcccttctacggccagg | 5’-atttgcggctactcgggca | 167bp |
| PRKN | 5’-ttccaaaccggatgagtggtg | 5’-cgatcattgagtcttgtcacacag | 262bp |
| BNIP3 | 5’-taaacacccgaagcgcac | 5’-agctatgttgcaagctcaga | 244bp |
| NIX | 5’-aatgtcgtcccacctagtcg | 5’-gaggatggtacgtgttccagc | 189bp |
| FUNDC1 | 5’-cccaagactatgaaagtgatgacg | 5’-gaaagccaccacctactgca | 228bp |
| P62 | 5’-tgtggtaggaacccgctac | 5’-agtttcctggtggaccca | 215bp |
| OPTN | 5’-agacctgttgggcatcgtgt | 5’-ccagtggagactgttctcgtgg | 159bp |
Statistical Analysis
1. Results
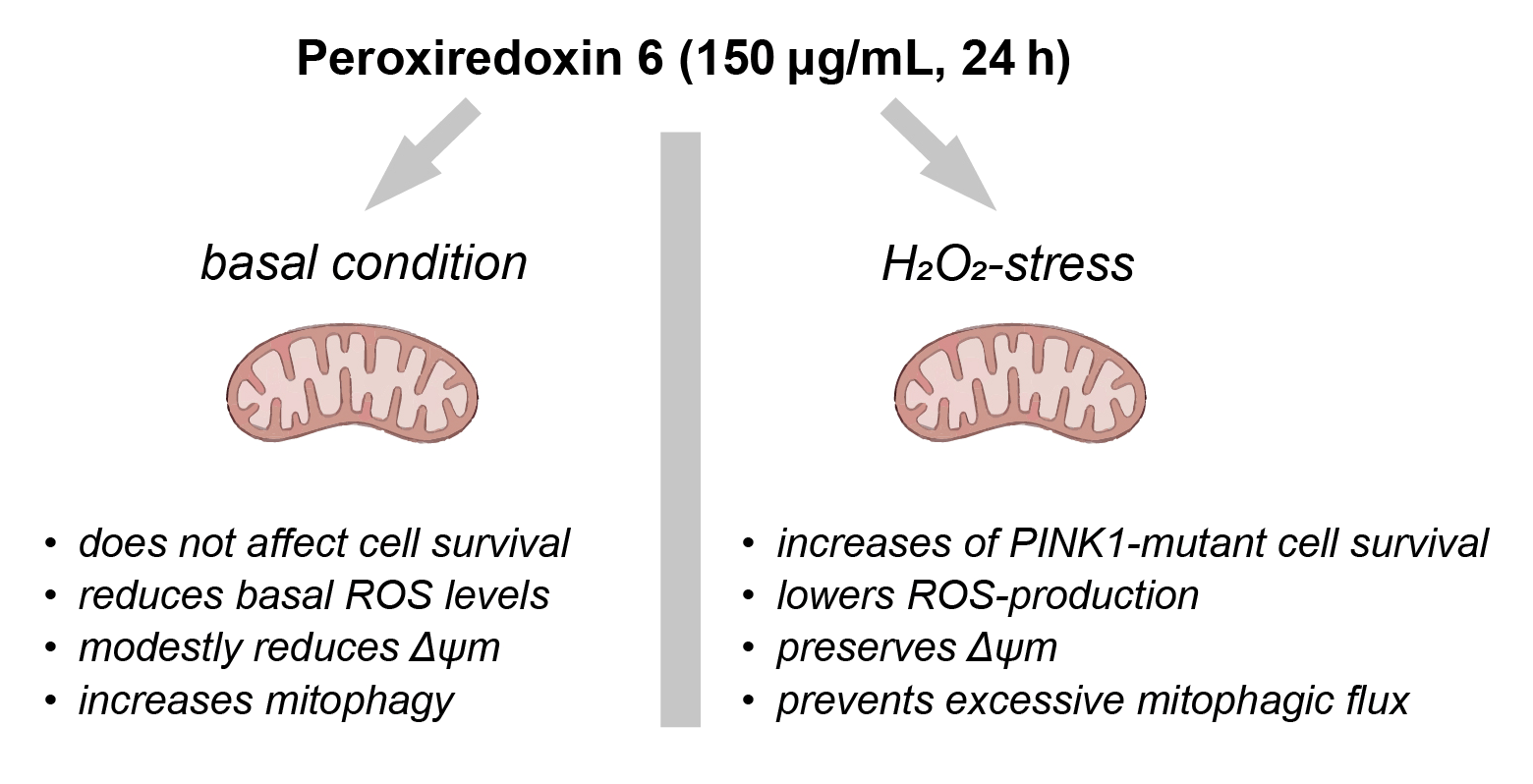
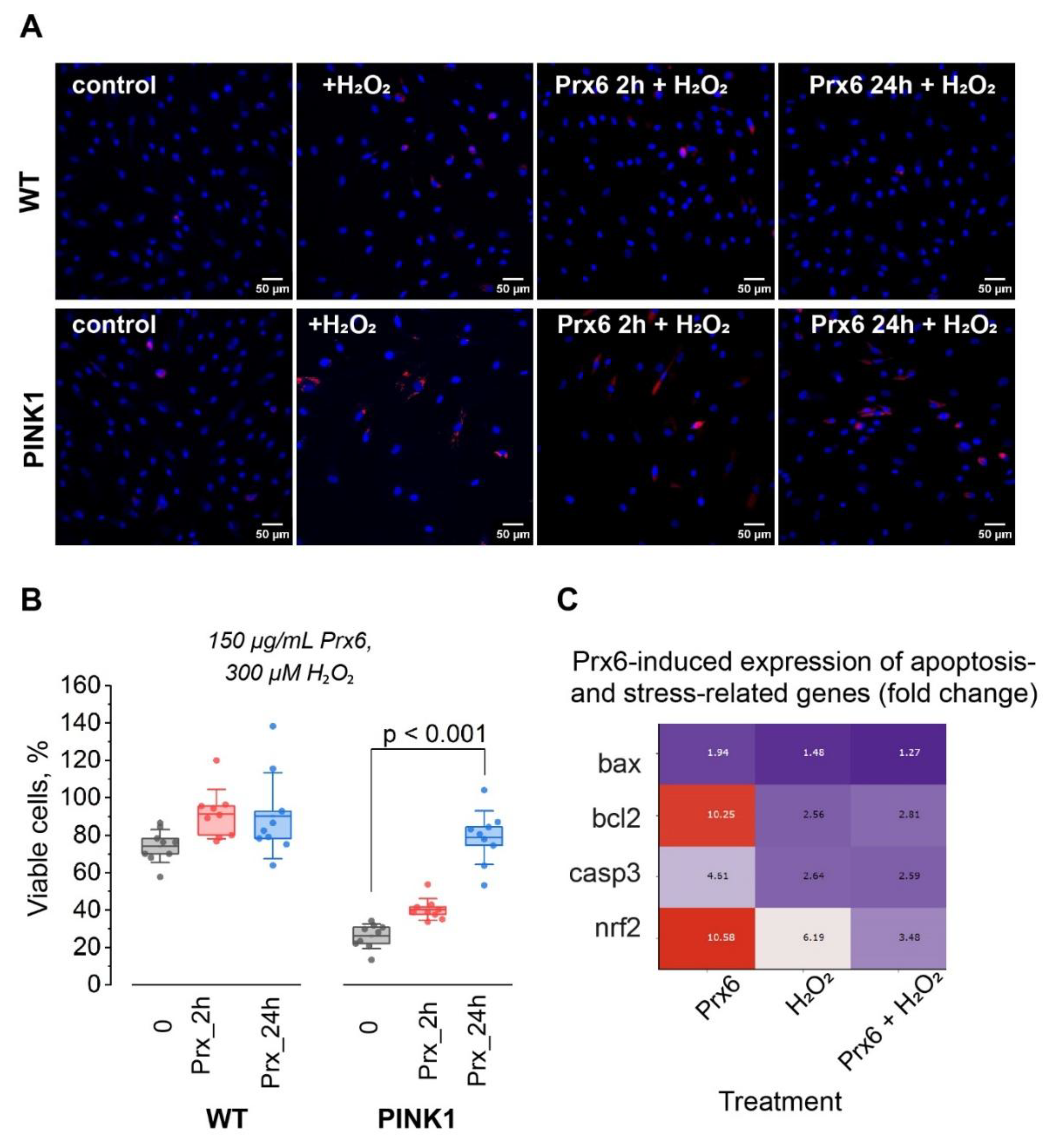
1.1. Prx6 Improves Survival of PINK1-Mutant Fibroblasts Under Oxidative Stress and Modulates Apoptosis-Related Gene Expression in Neuroblastoma Cells
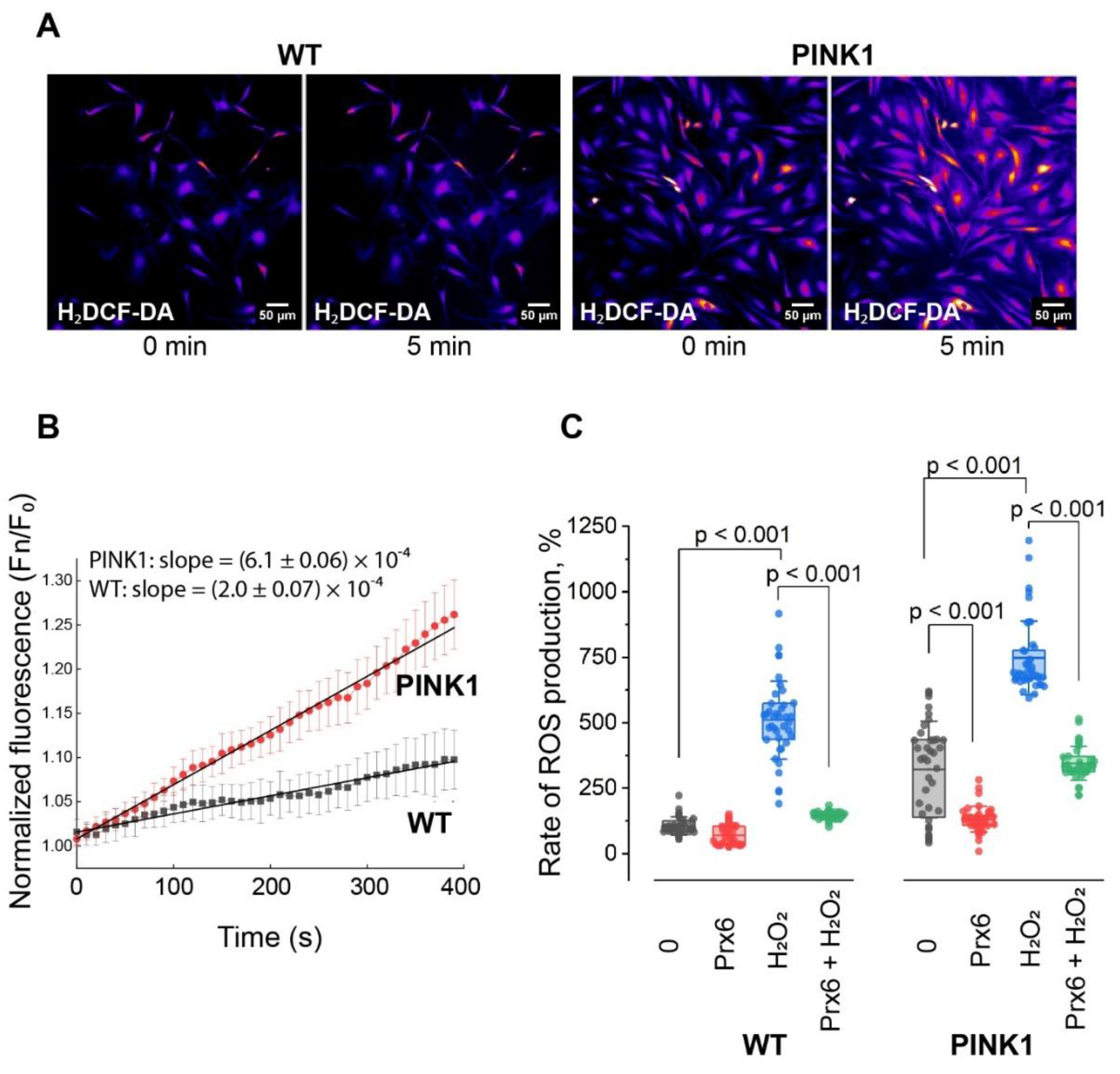
1.2. Prx6 Modulates ROS Production Rate in Wild-Type and PINK1-Mutant Fibroblasts
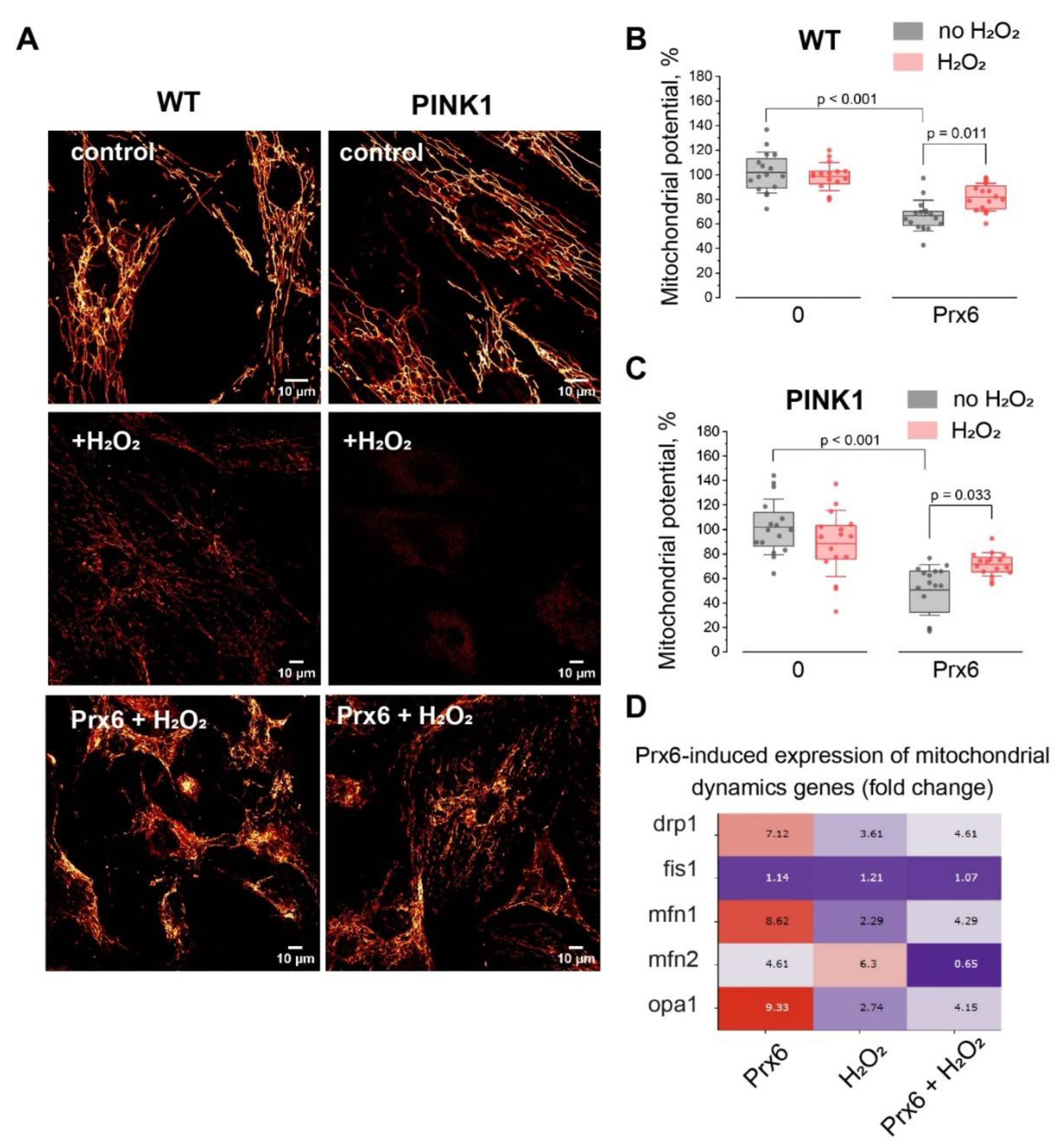
1.3. Prx6 Preserves Mitochondrial Membrane Potential in Fibroblasts Under Oxidative Stress and Modulates Mitochondrial Dynamics Gene Expression in Neuroblastoma Cells
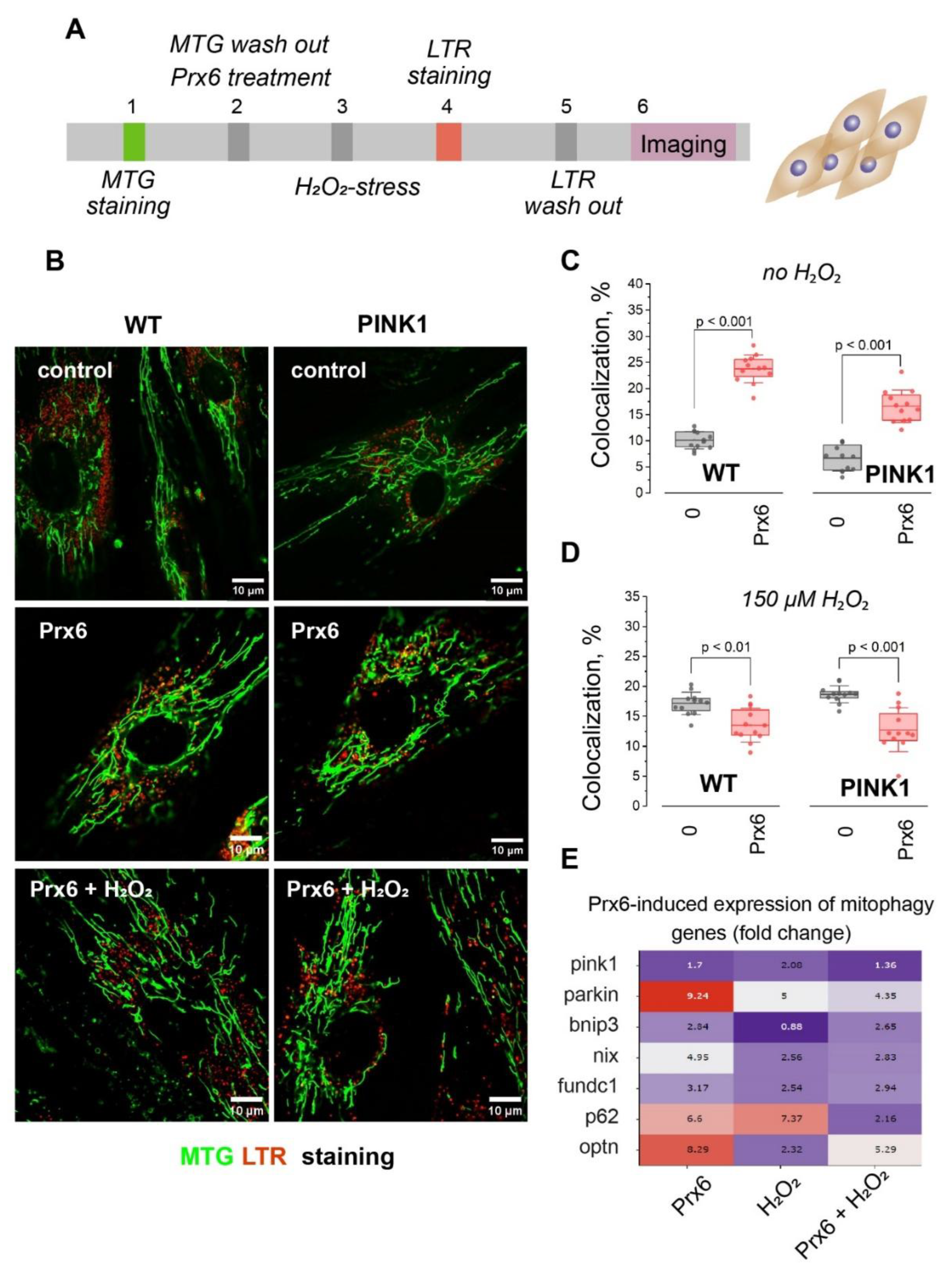
1.4. Prx6 Enhances Basal Mitophagy but Limits Stress-Induced Mitophagy in WT and PINK1-Deficient Fibroblasts
Discussion
Limited Effect After Short-Term Exposure; Cytoprotection Requires Prolonged Incubation
Prx6 Attenuates ROS Production Under Basal and Stress Conditions
Prx6 Preserves Δψm and Modulates Mitochondrial Remodeling Programs
Dual Effect of Prx6 on Mitophagy: Stimulation Under Basal Conditions, Suppression Under Stress
Prx6 Modulates Mitophagy-Related Gene Expression
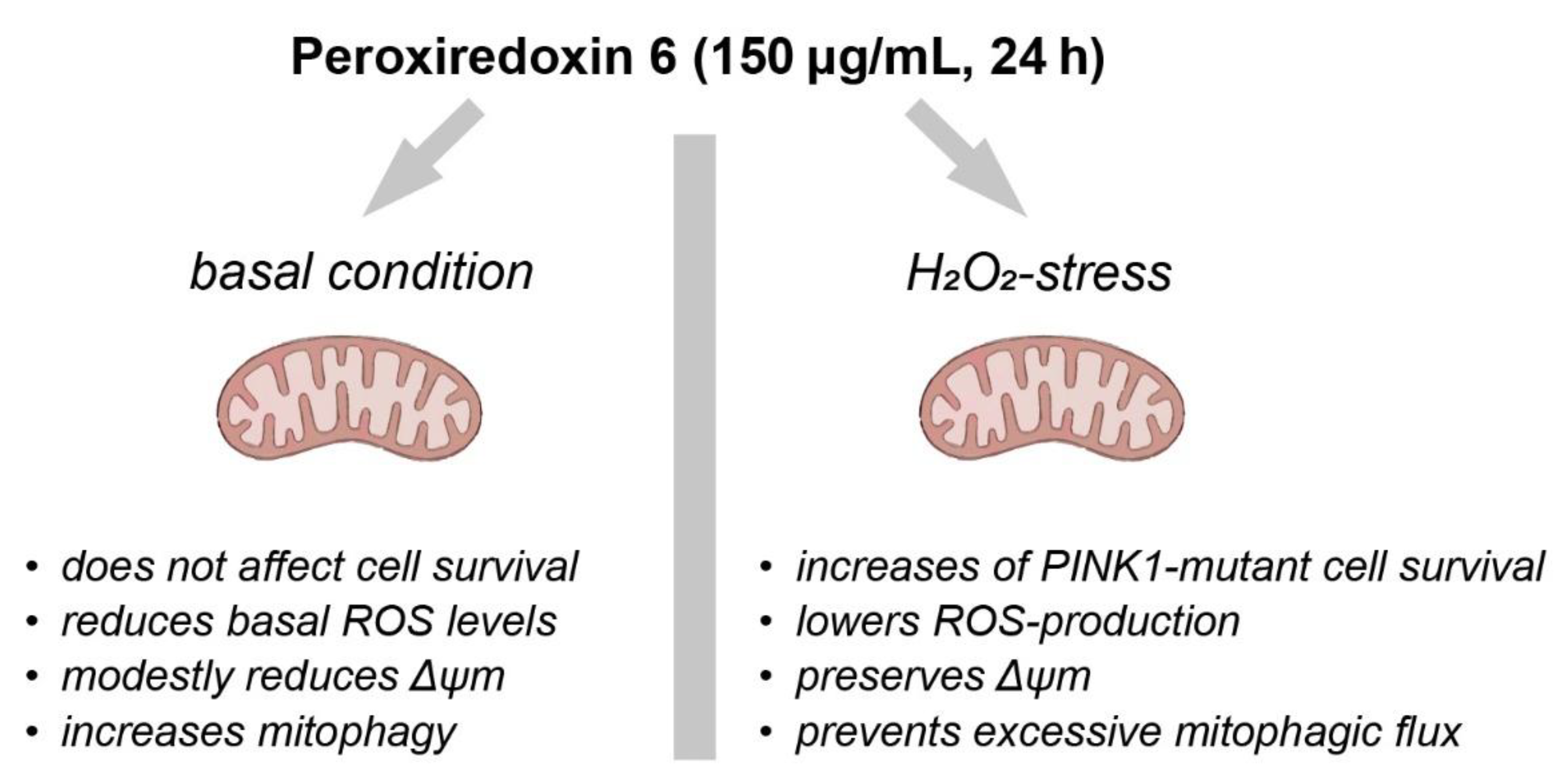
Integrated Model and Implications
Limitations and Future Directions
Conclusions
Fundings and acknowledgments
Ethics statement
Data statement
Declaration of competing interest
References
- Bertholet, A.; Delerue, T.; Millet, A.; Moulis, M.; David, C.; Daloyau, M.; Arnauné-Pelloquin, L.; Davezac, N.; Mils, V.; Miquel, M.; et al. Mitochondrial fusion/fission dynamics in neurodegeneration and neuronal plasticity. Neurobiol. Dis. 2016, 90, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zhao, H.; Li, Y. Mitochondrial dynamics in health and disease: mechanisms and potential targets. Signal Transduct. Target. Ther. 2023, 8, 333. [Google Scholar] [CrossRef] [PubMed]
- Fisher, A.B. Peroxiredoxin 6 in the repair of peroxidized cell membranes and cell signaling. Arch. Biochem. Biophys. 2017, 617, 68–83. [Google Scholar] [CrossRef]
- Sharapov, M.G.; Goncharov, R.G.; Parfenyuk, S.B.; Glushkova, O.V.; Novoselov, V.I. The Role of Phospholipase Activity of Peroxiredoxin 6 in Its Transmembrane Transport and Protective Properties. Int. J. Mol. Sci. 2022, 23, 15265. [Google Scholar] [CrossRef]
- Jia, W.; Dong, C.; Li, B. Anti-Oxidant and Pro-Oxidant Effects of Peroxiredoxin 6: A Potential Target in Respiratory Diseases. Cells 2023, 12, 181. [Google Scholar] [CrossRef]
- Pacifici, F.; Della-Morte, D.; Capuani, B.; Coppola, A.; Scioli, M.G.; Donadel, G.; Andreadi, A.; Ciccosanti, F.; Fimia, G.M.; Bellia, A.; et al. Peroxiredoxin 6 Modulates Insulin Secretion and Beta Cell Death via a Mitochondrial Dynamic Network. Front. Endocrinol. 2022, 13, 842575. [Google Scholar] [CrossRef]
- Sharapov, M.G.; Gudkov, S.V.; Lankin, V.Z.; Novoselov, V.I. Role of Glutathione Peroxidases and Peroxiredoxins in Free Radical-Induced Pathologies. Biochem. (Moscow) 2021, 86, 1418–1433. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Zhang, X.; Zheng, L.; Li, Z.; Zhao, X.; Lai, W.; Shen, H.; Lv, J.; Yang, G.; Wang, Q.; et al. Peroxiredoxin 6 Is a Crucial Factor in the Initial Step of Mitochondrial Clearance and Is Upstream of the PINK1-Parkin Pathway. Antioxidants Redox Signal. 2015, 24, 486–501. [Google Scholar] [CrossRef]
- Kritskaya, K.A.; Fedotova, E.I.; Berezhnov, A.V. Impaired Mitochondrial Network Morphology and Reactive Oxygen Species Production in Fibroblasts from Parkinson’s Disease Patients. Biomedicines 2024, 12, 282. [Google Scholar] [CrossRef]
- Sharapov, M.G.; Novoselov, V.I.; Ravin, V.K. Cloning, expression and comparative analysis of peroxiredoxin 6 from different species. Mol. Biol. 2009, 43, 465–471. [Google Scholar] [CrossRef]
- Sharapov, M.G.; Novoselov, V.I.; Fesenko, E.E.; Bruskov, V.I.; Gudkov, S.V. The role of peroxiredoxin 6 in neutralization of X-ray mediated oxidative stress: effects on gene expression, preservation of radiosensitive tissues and postradiation survival of animals. Free. Radic. Res. 2017, 51, 148–166. [Google Scholar] [CrossRef]
- Berezhnov, A.V.; Soutar, M.P.; Fedotova, E.I.; Frolova, M.S.; Plun-Favreau, H.; Zinchenko, V.P.; Abramov, A.Y. Intracellular pH modulates autophagy and mitophagy. J. Biol. Chem. 2016, 291, 8701–8708. [Google Scholar] [CrossRef]
- Fedotova, E.I.; Dolgacheva, L.P.; Abramov, A.Y.; Berezhnov, A.V. Lactate and Pyruvate Activate Autophagy and Mitophagy that Protect Cells in Toxic Model of Parkinson’s Disease. Mol. Neurobiol. 2022, 59, 177–190. [Google Scholar] [CrossRef]
- Kozera, B.; Rapacz, M. Reference genes in real-time PCR. J. Appl. Genet. 2013, 54, 391–406. [Google Scholar] [CrossRef]
- Liu, M.; Zuo, S.; Guo, X.; Peng, J.; Xing, Y.; Guo, Y.; Li, C.; Xing, H. The Study of Overexpression of Peroxiredoxin-2 Reduces MPP+-Induced Toxicity in the Cell Model of Parkinson’s Disease. Neurochem. Res. 2023, 48, 2129–2137. [Google Scholar] [CrossRef]
- Dinkova-Kostova, A.T.; Abramov, A.Y. The emerging role of Nrf2 in mitochondrial function. Free. Radic. Biol. Med. 2015, 88, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Abramov, A.Y.; Gegg, M.; Grunewald, A.; Wood, N.W.; Klein, C.; Schapira, A.H.V. Bioenergetic consequences of PINK1 mutations in parkinson disease. PLOS ONE 2011, 6, e25622. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, S.; Wood-Kaczmar, A.; Yao, Z.; Plun-Favreau, H.; Deas, E.; Klupsch, K.; Downward, J.; Latchman, D.S.; Tabrizi, S.J.; Wood, N.W.; et al. PINK1-Associated Parkinson's Disease Is Caused by Neuronal Vulnerability to Calcium-Induced Cell Death. Mol. Cell 2009, 33, 627–638. [Google Scholar] [CrossRef]
- Zorov, D.B.; Andrianova, N.V.; Babenko, V.A.; Pevzner, I.B.; Popkov, V.A.; Zorov, S.D.; Zorova, L.D.; Plotnikov, E.Y.; Sukhikh, G.T.; Silachev, D.N. Neuroprotective Potential of Mild Uncoupling in Mitochondria. Pros and Cons. Brain Sci. 2021, 11, 1050. [Google Scholar] [CrossRef]
- Frank, M.; Duvezin-Caubet, S.; Koob, S.; Occhipinti, A.; Jagasia, R.; Petcherski, A.; Ruonala, M.O.; Priault, M.; Salin, B.; Reichert, A.S. Mitophagy is triggered by mild oxidative stress in a mitochondrial fission dependent manner. Biochim. et Biophys. Acta (BBA) - Mol. Cell Res. 2012, 1823, 2297–2310. [Google Scholar] [CrossRef] [PubMed]
- Palikaras, K.; Lionaki, E.; Tavernarakis, N. Mechanisms of mitophagy in cellular homeostasis, physiology and pathology. Nat. Cell Biol. 2018, 20, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- Sharapov, M.G.; Glushkova, O.V.; Parfenyuk, S.B.; Gudkov, S.V.; Lunin, S.M.; Novoselova, E.G. The role of TLR4/NF-κB signaling in the radioprotective effects of exogenous Prdx6. Arch. Biochem. Biophys. 2021, 702, 108830. [Google Scholar] [CrossRef] [PubMed]
- Komilova, N.R.; Angelova, P.R.; Berezhnov, A.V.; Stelmashchuk, O.A.; Mirkhodjaev, U.Z.; Houlden, H.; Gourine, A.V.; Esteras, N.; Abramov, A.Y. Metabolically induced intracellular pH changes activate mitophagy, autophagy, and cell protection in familial forms of Parkinson's disease. FEBS J. 2021, 289, 699–711. [Google Scholar] [CrossRef]
- Nadeev, A.D.; Kritskaya, K.A.; Fedotova, E.I.; Berezhnov, A.V. One Small Step for Mouse»: High CO2 Inhalation as a New Therapeutic Strategy for Parkinson’s Disease. Biomedicines 2022, 10, 2832. [Google Scholar] [CrossRef]
- Georgakopoulos, N.D.; Wells, G.; Campanella, M. The pharmacological regulation of cellular mitophagy. Nat. Chem. Biol. 2017, 13, 136–146. [Google Scholar] [CrossRef]
- Mantey, I.; Langerscheidt, F.; Durmaz, Ç.Ç.; Baba, N.; Burghardt, K.; Karakaya, M.; Zempel, H. The POLG Variant c.678G>C; p.(Gln226His) Is Associated with Mitochondrial Abnormalities in Fibroblasts Derived from a Patient Compared to a First-Degree Relative. Genes 2025, 16, 198. [Google Scholar] [CrossRef]
- Vinokurov, A.Y.; Palalov, A.A.; Kritskaya, K.A.; Demyanenko, S.V.; Garbuz, D.G.; Evgen’eV, M.B.; Esteras, N.; Abramov, A.Y. Cell-Permeable HSP70 Protects Neurons and Astrocytes Against Cell Death in the Rotenone-Induced and Familial Models of Parkinson’s Disease. Mol. Neurobiol. 2024, 61, 7785–7795. [Google Scholar] [CrossRef]
- Kritskaya, K.A.; Stelmashchuk, O.A.; Abramov, A.Y. Point of No Return—What Is the Threshold of Mitochondria With Permeability Transition in Cells to Trigger Cell Death. J. Cell. Physiol. 2025, 240, e31521. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



