1. Introduction: What Causes Alzheimer’s Disease?
Identifying the cause of Alzheimer’s disease (AD) remains one of the most urgent challenges in modern medicine, yet its complexity continues to preclude a single parsimonious explanation of pathogenesis [
1]. Many risk factors have been identified, which combine and interact to confer relative risk or resilience [
2]. These include age, genetics, traumatic brain injury, comorbidities, physical inactivity, sleep disruption, viral or bacterial infection, among others [
1,
2]. The diversity of these ‘causes’ contrasts sharply with the relatively uniform sequence of pathological events that define AD. While individual progression patterns and clinical presentations can vary [
3,
4] (discussed in section 8), AD typically unfolds in the brain as follows: β-amyloid (Aβ) plaques emerge according to
Thal stages [
5], tau neurofibrillary tangles spread according to
Braak stages [
6], and the resulting tau-associated neurodegeneration drives canonical patterns of cognitive decline, ultimately leading to dementia [
7]. A system whereby varied causes lead to relatively uniform outcomes implies the existence of a single upstream vulnerability: a common breakpoint where diverse insults converge to initiate a pathological cascade (Postulate 1, Box 1). Furthermore, by Occam’s razor, a single breakpoint is much more likely an explanation of pathogenesis than multiple simultaneously dysfunctioning systems (Postulate 2, Box 1). This is especially true in a disorder as common as AD, which affects a third of people over the age of 85 [
8]. There is, therefore, a need for explanations of AD pathogenesis that unify the emergence of both Aβ and tau pathology under a single cause. Finally, given the high prevalence of AD, the single breakpoint is likely a feature of normal brain architecture rather than an obscure abnormality (Postulate 3, Box 1). Identification of this breakpoint is critical, as its protection would provide the most effective option for disease prevention [
9].
We argue that neuromodulatory subcortical projection neurons represent a prime candidate for this vulnerable breakpoint. These neurons are uniquely fragile: their large, sparsely myelinated, tau-rich axons have extraordinary energy demands, making them highly dependent on a steady supply of resources and fully functional repair mechanisms [
10,
11]. We propose that the heightened prevalence of AD among older adults therefore reflects a universal weakness within these projection systems, which is increasingly exposed in older age when cellular transport and maintenance mechanisms falter [
12,
13]. In a sentence: we believe that it is no coincidence that the hardest-to-maintain parts of the most fragile neurons are enriched for a pre-pathogenic component (tau) of the most common cause of dementia worldwide.
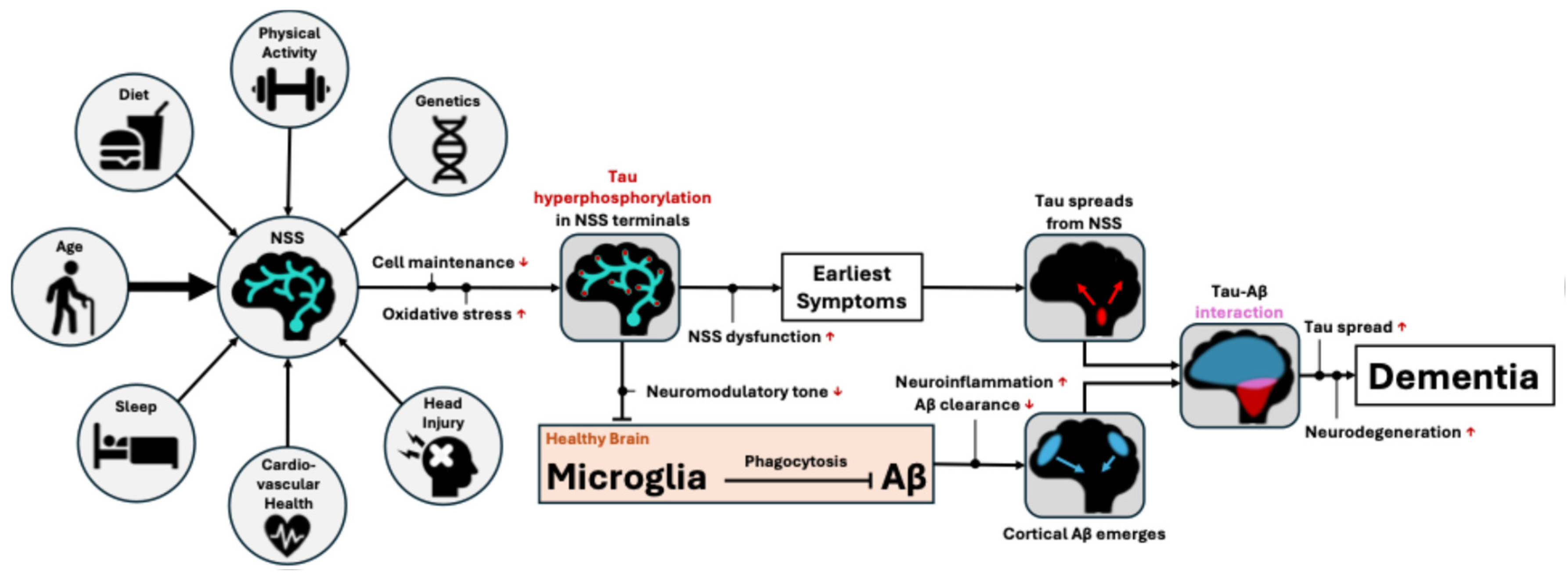
We propose the following mechanistic framework whereby AD, from its earliest pathogenesis to clinical dementia, can be understood as a downstream consequence of neuromodulatory fragility (
Figure 1). Declining metabolic efficiency and impaired cellular transport and clearance mechanisms in advancing age disproportionately affect these cells, particularly in tau-rich axons extremely distal from the cell nucleus. Accumulated oxidative stress drives tau hyperphosphorylation and misfolding, leading to axonal instability and reduced neuromodulatory tone at cortical endpoints. This loss of neuromodulatory input disrupts microglial homeostasis and impairs the clearance of early Aβ species, ultimately leading to plaque formation. Misfolded, hyperphosphorylated tau spreads slowly and transneuronally through highly active networks, while Aβ plaques continue to form across the cortex. Eventual local interactions of Aβ and tau pathology accelerate the activity-dependent transneuronal spread of tau due to Aβ-induced hyperexcitability, culminating in widespread neurodegeneration and dementia onset.
Box 1. Postulates of the neuromodulatory fragility framework of AD pathogenesis.
Postulate 1: Diverse causes leading to (relatively) uniform outcomes suggests a single upstream vulnerability.
Postulate 2: A single breakpoint is a simpler explanation of pathogenesis than multiple breakpoints, especially in an extremely common disorder (Occam’s Razor)
Postulate 3: In an extremely prevalent disorder (like AD), the breakpoint is more likely a feature of normal brain architecture than an obscure abnormality
Neuromodulatory subcortical projection neurons, specifically their tau-rich axonal terminals, are a prime candidate for this single vulnerable breakpoint. Multiple risk factors can converge on this system to confer risk of AD. Variation across individuals in neuromodulatory resilience is a likely predictor of relative risk for AD.
In this perspective paper we describe this
neuromodulatory fragility framework of AD pathogenesis in more detail, describing a broad view of the entire AD process from pathogenesis to dementia onset. The perspective this model provides shifts the focus from late-stage cortical hallmarks to vulnerable subcortical origins which become dysfunctional long before the clinical manifestation of the disease. The framework situates AD as a disorder of statistical vulnerability rather than a singular initiating insult
per se. In lieu of refuting the amyloid hypothesis, which has dominated thinking and research on AD for more than 30 years [
14], we suggest a reframing, whereby Aβ accumulation may be a downstream consequence, not the initiating trigger, of a more fundamental dysfunction of neuromodulatory subcortical projection neurons. By viewing AD through the lens of neuromodulatory fragility, we can integrate disparate findings on tau seeding, the neuroinflammatory response and nebulous prodromal behavioural changes into a unified theory of AD pathogenesis.
2. Neuromodulatory Projection Neurons Are a System-Wide Weak Point
Neuromodulatory subcortical systems can be analogised as the conductors to the orchestra of the brain. Where glutamatergic and GABA-ergic neurons facilitate the bulk of the brain’s activity (the orchestra), dramatically smaller populations of neuromodulatory neurons (the conductors) shape the dynamics and characteristics of their combined activity [
15,
16]. These systems release neurotransmitters such as noradrenaline, acetylcholine, dopamine, serotonin, and orexin. Given their pivotal role in modulating brain activity, these neuromodulators are implicated in many fundamental cognitive and behavioural processes including regulating sleep-wake cycles, mood stability, attention, appetite control, even interacting with the brain’s immune system [
17,
18,
19,
20,
21,
22].
A key distinction between neuromodulatory neurons and other projection neurons is their use of ‘volume transmission’: they can release neurotransmitters from varicosities along the axon into the extracellular space without a specific postsynaptic target, in addition to classical synaptic release [
23]. Several anatomical and physiological features simultaneously enable this mode of signalling while simultaneously heightening their fragility:
- 1)
Extreme axonal length and arborization: The projection neurons of neuromodulatory systems originate from small clusters of cell bodies (e.g. locus coeruleus, nucleus basalis of Meynert, dorsal raphe, ventral tegmental area, lateral hypothalamus), yet send vast, highly arborized projections across the entire neocortex. The extreme length and breadth of these combined arborizations allows each neuron to modulate large areas [
24,
25,
26]. However, maintaining such long, branched projections is metabolically expensive. These systems require continuous transport of proteins, mitochondria and other cellular components and rendering them chronically dependent on nutrients, growth factors and oxygen, as well as effective waste clearance and repair mechanisms [
27,
28].
- 2)
High tonic firing rates: To sustain basal neuromodulatory tone over large target areas, neuromodulatory neurons exhibit high tonic firing rates, with regular transient bursts during behaviourally relevant events [
29,
30,
31,
32]. This firing pattern is well-suited to slow, diffuse modulation via volume transmission, but further amplifies metabolic demand, increasing susceptibility to any disruption in oxidative balance, calcium homeostasis or energy production.
- 3)
Poor myelination. Neuromodulatory axons are typically thin and only sparsely myelinated. This facilitates ‘leakiness’ of neurotransmitter along the axon’s length – the definitive feature of ‘volume transmission’. However, the lack of myelination and thin calibre of these neurons offers little protection against oxidative or mechanical injury [
33,
34]. Cortical regions with higher myelination are relatively resistant to pathology and neurodegeneration, suggesting that myelin may represent a substrate of ‘brain reserve’ that these projection systems largely lack [
34,
35]. The constant axonal remodelling that may be required to maintain these delicate projections could further exacerbate the metabolic demands of these systems.
Taken together, the features that allow neuromodulatory systems to modulate large-scale brain states (extreme arborization, high tonic activity, poor myelination) also impose high energetic demands, low tolerance for metabolic and oxidative stress and a strong propensity to accumulate damage over the lifespan. The fact that these same features are essential for their function helps explain the apparent evolutionary maladaptiveness of these features: typical evolutionary routes to increase resilience, such as increasing myelination or pruning excess branches, would directly impair their modulatory capacity and are therefore unlikely to be favoured by natural selection.
Aging, the primary risk factor for sporadic AD, is poised to disproportionately affect these low-tolerance systems through changes such as mitochondrial dysfunction, oxidative stress and disrupted calcium homeostasis [
11,
12,
36]. Monoaminergic neurons are particularly at-risk for oxidative stress. By-products of noradrenaline and dopamine synthesis generate reactive oxygen species throughout life, particularly in axons and terminals where these neurotransmitters are produced [
37]. Neuromelanin, a compound found in monoaminergic neurons, normally sequesters these toxic metabolites, but when its capacity is reached in older age, intracellular toxicity rises sharply, possibly marking a transition from resilience to degeneration [
38]. In cholinergic neurons, age-related reductions in calcium-binding proteins increase their susceptibility to calcium-induced excitotoxicity and pathological processes [
39]. Aging therefore imposes a critical bottleneck on these inherently fragile neuronal systems.
Other risk factors for AD likely modulate the vulnerability of neuromodulatory projection neurons, either through specific direct mechanisms or through systemic changes to brain health. Take as an example the apolipoprotein E-ε4 (
APOε4) gene, the single largest genetic risk factor for sporadic AD. Carriers of one or two
APOε4 alleles face up to 3- or 15-fold greater risk, respectively, than
APOε3 homozygotes [
40].
APOε4 is thought to impair myelin regulation [
41]. It is feasible that myelin dysregulation would disproportionately increase the vulnerability of neuromodulatory projection neurons which already lack robust myelination and therefore have little to no redundancy on which to fall back. In effect, we propose that what little protection these neurons have under an
APOε3 genotype is weakened or lost with an
APOε4 genotype, further destabilizing an already fragile system. The inverse may well be true of the neuroprotective
APOε2 allele. This framework aligns with evidence of selective neuromodulatory system damage in demyelinating conditions such as multiple sclerosis and encephalomyelitis [
42].
We have so far explained how upstream risk factors converge onto neuromodulatory systems to cause disproportionate dysfunction. The next sections will explain how this dysfunction is sufficient to explain the downstream emergence of the Aβ and tau pathology which define AD.
3. Tau Hyperphosphorylation Is a Direct Outcome of Neuromodulatory System Disruption
Tau is a protein that binds and stabilizes microtubules, supporting axonal structure [
43]. In the highly arborized projection neurons of neuromodulatory systems, axons are extremely distal from the cell nucleus [
24,
25,
26]. This makes their axonal machinery, of which physiological tau is a key component, particularly susceptible to disruptions in transport, repair and clearance processes [
44,
45,
46]. In other words, these axons, especially distal, tau-rich regions, are the most vulnerable part of an already fragile system (
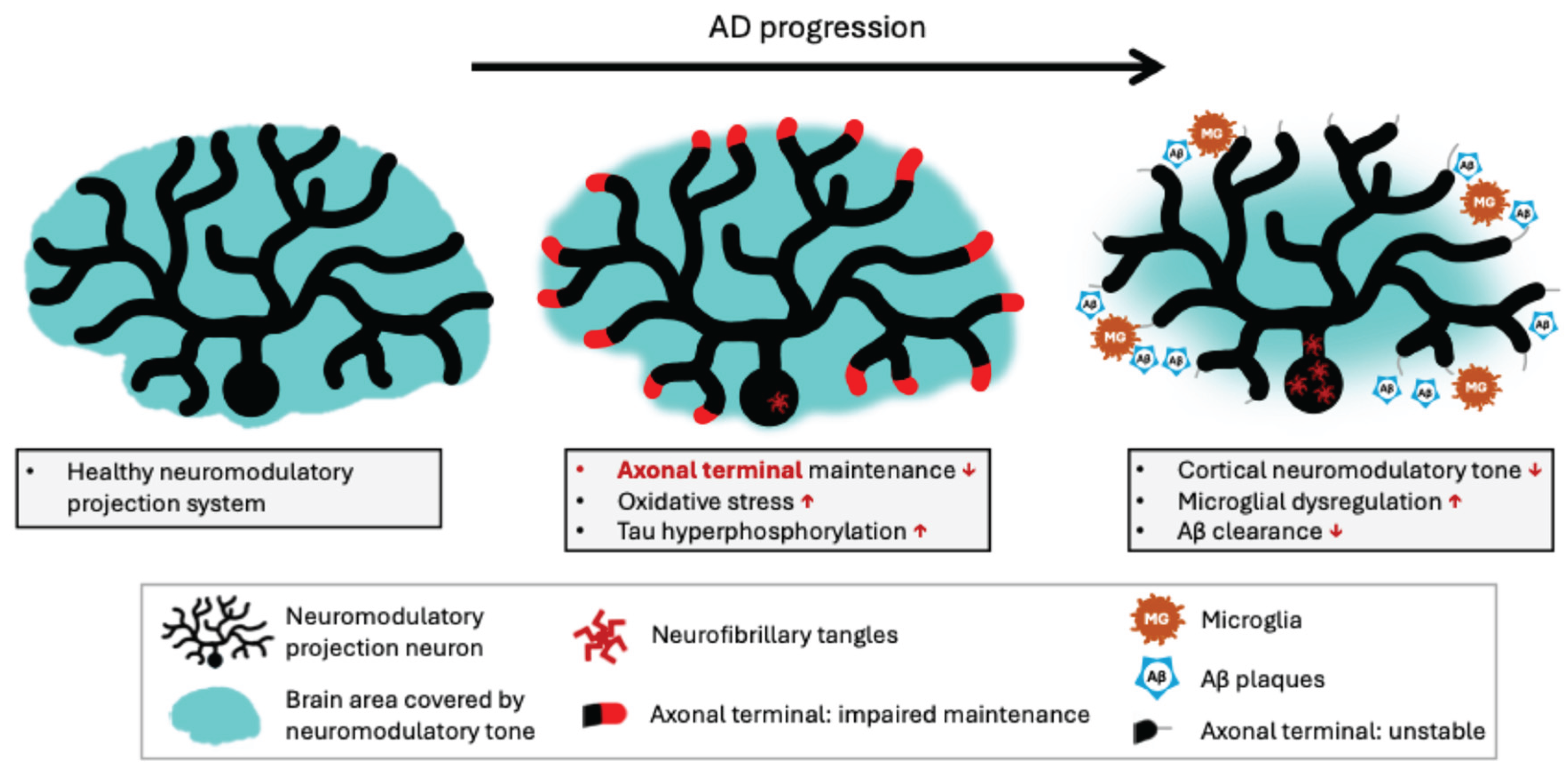
Figure 2).
Reliable clearance of reactive oxygen species from these distal structures is particularly critical. When reactive oxygen species accumulate (e.g. as by-products of the monoaminergic synthesis pathway [
47]), oxidative stress triggers tau hyperphosphorylation, producing misfolded forms of tau that can no longer bind microtubules [
48]. The result is axonal instability, disrupted intracellular transport and impaired neurotransmission [
44,
45].
Misfolded oligomers of hyperphosphorylated tau cluster together to form neurofibrillary tangles, which are first detectable in neuromodulatory nuclei in presymptomatic AD [
10,
49]. Tau tangles are reported as early as the second decade of life in the locus coeruleus [
18,
50], and the third decade of life in the nucleus basalis of Meynert [
51]. Tangles have also been found in orexinergic neurons of the lateral hypothalamus [
52] and serotonergic dorsal raphe neurons [
53,
54] in presymptomatic AD. In the locus coeruleus and basal forebrain, hyperphosphorylated tau species have been shown to lead to decreased fibre density and reduced neuromodulatory tone [
48,
55].
A growing body of evidence supports the idea that tau pathology spreads transneuronally in an activity-dependent manner, progressing most rapidly along densely connected pathways [
56,
57]. Early propagation likely occurs between neuromodulatory nuclei themselves, given their extensive inter-connectivity, which complicates efforts to identify the precise initial locus of pathology in population-based studies. Strong connections between these nuclei and cortical sites of early tau pathology in AD, together with evidence that tau pathology and atrophy in neuromodulatory nuclei precede corresponding changes in medial temporal regions [
58,
59,
60,
61,
62], have led to the proposal that tau advances from subcortical hubs to the medial temporal lobe and beyond, following canonical
Braak staging [
6]. Under the
neuromodulatory fragility framework, neuromodulatory nuclei act as seed points, propagating tau pathology to densely connected efferents to initiate cortical pathology. While tau accumulation in these early stages causes local neurodegeneration, the extent of spread and, by extension, overt clinical symptoms remain limited until the process is amplified by Aβ pathology (described in section 5) [
63].
4. Neuromodulatory System Degradation Disrupts Amyloid Clearance Through Neuroinflammatory Pathways
Interactions between neuromodulatory systems and Aβ are less well characterised than those with tau. The link is perhaps less obvious as Aβ plaques rarely accumulate around neuromodulatory cell bodies until relatively late stages of AD, typically
Thal stage five [
5]. Nevertheless, several mechanisms suggest that neuromodulatory system dysfunction could underlie the emergence of Aβ pathology.
As tau hyperphosphorylation destabilises the axons of neuromodulatory projections, distal axons degenerate, and cortical neuromodulatory tone declines [
64] (
Figure 2). Reduced neuromodulatory tone leads to the release of pro-inflammatory cytokines and microglial dysregulation, which, in turn, impairs Aβ clearance [
65,
66,
67,
68]. Experimentally induced degeneration of cholinergic and noradrenergic subcortical systems in animal models leads to Aβ plaque accumulation [
65,
69]. Conversely, replacement or receptor-agonist treatment can reverse these downstream effects, supporting a causal role for neuromodulatory dysfunction in promoting Aβ deposition [
70,
71]. While most direct evidence for the neuroinflammation-mediated dysregulation of Aβ clearance concerns the noradrenergic and cholinergic systems, similar anti-inflammatory and neuroprotective properties have been observed for orexin [
72].
Neuromodulatory neurons may also contribute directly to early Aβ seeding. In both noradrenergic and cholinergic subcortical systems, these neurons have been shown to accumulate intracellular Aβ oligomer species in neurites and axon terminals [
73,
74]. These locally generated Aβ oligomers can be removed quickly when clearance mechanisms are performing well, with little chance to cause damage or cluster into plaques. However, they could act as an initial seed that facilitates Aβ plaque formation once microglial dysfunction impairs Aβ clearance.
In contrast to the transneuronal spread of tau, Aβ plaque formation appears wherever distal neuromodulatory tone is depleted. As with leaves falling from a tree in autumn, the exact branch to lose foliage first may differ between individuals of even similar trees, but loss is inevitable once environmental conditions shift, and the tree restricts nutrients to more proximal regions. This metaphor aligns with the observed interindividual variability in Aβ plaque localisation in early
Thal stages [
5], despite the high prevalence of Aβ among even cognitively unimpaired older adult populations [
75].
Neuroinflammation is a critical but poorly understood element of AD, often debated as either a cause, effect or epiphenomenon of the disease process. The neuromodulatory fragility framework highlights a specific role for neuroinflammation in triggering and amplifying spread of Aβ pathology. The positive feedback loops by which neuroinflammation shifts from neuroprotection to neurotoxicity amplify the difficulty of studying the exact causative role of neuroinflammation in AD. Our model clarifies the sequence of events, which will aid generation of specific testable hypotheses to better understand this complex relationship.
5. Tau Misfolding Precedes Plaque Accumulation But Is Slow in the Absence of Aβ
Neurodegeneration and symptom onset correlate more strongly with tau neurofibrillary tangles than with Aβ pathology [
76]. However, in the absence of Aβ, tau-associated toxicity, behavioural impairments and the spatial extent of tau are greatly attenuated in AD [
77]. This latter detail likely reflects the interaction of two key mechanisms driven by the AD hallmarks: Aβ causes neuronal hyperexcitability, and tau spreads transneuronally in an activity-dependent manner [
63,
78].
In our
neuromodulatory fragility framework of AD pathogenesis, tau misfolding originates in neuromodulatory projection neurons and spreads slowly along dense, highly active pathways, most notably projecting from neuromodulatory nuclei to the medial temporal lobe. In the absence of Aβ, as in primary age-related tauopathy, tau remains spatially restricted but still colocalizes with neurodegeneration [
79]. In AD, as both tau and Aβ pathologies spread, they eventually converge. At this convergence point, significant Aβ-driven neuronal hyperexcitability amplifies transneuronal tau propagation, driving rapid global distribution of tau. Graph theoretical and network modelling studies support this model, identifying the inferior temporal lobe as the most common site of Aβ-tau colocalization [
80,
81]. This colocalization and subsequent increasing rate of progression is likely the tipping point between presymptomatic and symptomatic disease.
Because tau spread and symptom severity depend on Aβ pathology, Aβ is sometimes regarded as an ‘upstream’ disease feature relative to altered tau proteostasis [
77]. However, our model positions tau dysregulation as the earliest molecular consequence of neuromodulatory system failure, preceding Aβ pathology. These seemingly contradictory views are reconcilable when considered across scales. With a traditional systems-level view of disease, focusing on clinical symptom onset, Aβ moderates the spread and severity of tau pathology, and hence is considered ‘upstream’ [
77,
78]. However, as we have described in the previous sections, at the cellular level, resource disruption in fragile neuromodulatory systems provides the initiating conditions that trigger tau misprocessing, which is sufficient to explain Aβ accumulation through microglial dysregulation [
45,
48,
64,
65]. It is clear that these views are not in fact contradictory, rather, they prioritise different scales at which the term ‘upstream’ is defined. The idea that tau misfolding precedes that of Aβ is not new, but the two processes are typically regarded as independent inciting events [
82,
83]. In contrast, our framework positions neuromodulatory fragility as a single upstream cause for both.
In humans, Aβ and tau interact continuously throughout AD. Pathogenic tau species can promote Aβ deposition (as discussed above), while Aβ may directly augment tau hyperphosphorylation e.g. through GSK-3β signalling [
37]. Direct injection of Aβ can initiate tau pathology in rodent models expressing mutant human tau [
84], but we argue that this injection ‘skips ahead’ in the natural pathological progression, bypassing the neuromodulatory dysfunction-associated altered tau proteostasis and triggering the positive feedback loop from an alternate entry point. In other words, this rodent model lacks true construct validity and, while useful for studying downstream stages of the disease, does not recapitulate natural AD pathogenesis. In our
neuromodulatory fragility framework, we highlight how a single point of fragility can trigger a self-perpetuating state of pathology. These closely interacting factors rapidly obscure the direction of these cause-effect relationships in experimental settings. Mechanistic explanations such as the
neuromodulatory fragility framework presented here are therefore crucial for forming effective research questions and hypotheses aimed at disentangling these relationships.
6. Early Behavioural Symptoms Are Consistent with Early Neuromodulatory Dysfunction
We acknowledge that compensatory mechanisms (potentially within these neuromodulatory systems) maintain near-normal cognitive functioning for many years, despite concurrent pathophysiological processes [
85]. Thus, clinically detectable symptoms most likely emerge as these mechanisms reach their limits and begin to fail. In AD, an episodic memory deficit is the most definitive and clinically recognised early symptom, however patients often report symptoms months or years before measurable memory impairment [
86]. These subtle perturbations in cognition and behaviour align closely with a model of early neuromodulatory system dysfunction, including: dysregulation of sleep-wake cycles (orexin, noradrenaline), attention deficits (acetylcholine, noradrenaline) and mood instability (serotonin) [
10,
86]. The emergence of neuropsychiatric symptoms in later disease stages likely reflects a more substantial breakdown of global neuromodulatory control [
87].
7. Predictions of the Neuromodulatory Fragility Framework
The proposed framework makes several testable predictions about AD that could guide future research. A key prediction of this framework is that enhancing the resilience of neuromodulatory systems should significantly delay or prevent the onset of AD. Strengthening these systems, through interventions that reduce oxidative stress or support intracellular transport and repair, could slow or halt early pathogenesis if applied early enough. Rather than proposing a single ‘cure-all’ mechanism, this framework aligns with existing evidence on the importance of modifiable risk factors such as cardiovascular health, sleep regulation, and protection from head injury as effective preventive strategies in midlife [
2]. That said, a theoretical pharmacological augmentation of neuromodulatory resilience would provide effective targeted AD protection [
9].
The
neuromodulatory fragility framework also predicts that accurate tracking of neuromodulatory system health could improve early detection and monitoring of disease progression. Recent advances in neuroimaging, including quantitative and neuromelanin-sensitive MRI, ultra-high-field diffusion imaging and positron emission tomography, have improved
in vivo assessment of these systems [
88,
89,
90]. As these tools mature and become more widely available, clinical monitoring of neuromodulatory integrity may become feasible for early diagnosis and therapeutic evaluation.
An additional implication of the model concerns the limited efficacy of anti-Aβ treatments, especially if applied late in the disease process [
82]. Once cortical Aβ burden is high, positive feedback loops characteristic of AD are already established, and the spreading of tau pathology is already well underway. Removing Aβ at this stage may modestly slow disease progression but is unlikely to halt it. While even incremental slowing of disease remains valuable (if side-effects can be minimized) we argue that therapies targeting patients who are already Aβ-positive may miss the critical early window of intervention. Within the
neuromodulatory fragility framework, Aβ is positioned as an important but downstream element of AD pathology, while preventing or mitigating neuromodulatory dysfunction offers a more promising means of arresting AD progression.
8. Unanswered Questions
While the neuromodulatory fragility framework ties together many known features of the AD trajectory, several questions remain open and warrant further investigation.
One such question concerns atypical subtypes of AD. It is becoming increasingly apparent that although progression patterns of tau are often categorised into canonical Braak stages, there is evidence for multiple subtypes of tau progression pattern, each with subtly different characteristics in terms of severity, topography, speed of progression and behavioural presentation [
3,
4]. It remains unclear whether all AD subtypes share the same underlying aetiology. Tau transmission along neuromodulatory projection pathways may follow probabilistic routes that vary across individuals leading to different clinical and anatomical presentations. Alternatively, different subtypes might arise from dysfunction in distinct neuromodulatory systems or through entirely separate mechanisms.
A related question is whether all neuromodulatory systems are equally vulnerable or whether specific nuclei are consistently affected first. If the latter is true, targeted strategies to augment resilience of a given, particularly vulnerable, neuromodulatory system could be highly effective. Alternatively, neuromodulatory systems may be strongly interdependent, and dysfunction in any one of a few candidate systems may be swiftly transmitted to others, necessitating a broader therapeutic strategy.
Additionally, while we have proposed that environmental and demographic factors confer risk of AD through their impact on neuromodulatory systems, the specific underlying mechanisms remain poorly understood. For example, AD is more prevalent in women, possibly reflecting menopause-related neuroendocrine changes that increase neuromodulatory fragility [
91]. Likewise, differences in disease incidence and clinical presentation across ethnic groups highlight the need to clarify how genetic and sociocultural factors interact with brain resilience and neuromodulatory system health [
92].
Finally, this proposed
neuromodulatory fragility framework must address the question of why AD appears so human-specific, despite neuromodulatory systems being relatively well conserved across species. Non-human primates do develop amyloid pathology, but tau burden diminishes rapidly with increasing phylogenetic distance from humans [
93]. Viewed through the lens of the
neuromodulatory fragility framework, this pattern suggests we should look closely at species differences in these systems. Many mammals express neuromelanin in monoaminergic nuclei (e.g. locus coeruleus, substantia nigra) but typically at much lower levels than humans [
94]. Even across primates, neuromelanin content is greater in species more closely-related to us [
94]. An striking exception, in terms of both the expression of AD-like pathology and neuromelanin can be seen in certain toothed whales (odontocetes) [
95,
96]. In some odontocetes, investigators have reported human-like co-occurrence of amyloid plaques and tau pathology [
95] as well as neuromelanin in the locus coeruleus with ultrastructural features remarkably similar to humans [
96]. Although direct quantitative comparisons across primates and odontocetes are still lacking, this apparent qualitative correlation between neuromelanin and AD-like pathology raises the hypothesis that neuromelanin burden may serve as a cross-species marker of cumulative neuromodulatory strain, which facilitates the emergence of AD.
The origin of this putative species-specific neuromodulatory strain remains unclear. One possibility is that a dramatically expanded neocortex provides unusually large target fields that must be modulated by relatively small populations of neuromodulatory cells. Indeed, human locus coeruleus and nucleus basalis of Meynert contain far fewer neurons relative to neocortical volume compared with non-human primates [
97,
98], a pattern that may hypothetically extend to odontocetes which have among the largest brain-to-body size ratios of any mammals [
99]. A second plausible contributor is our unusually long post-reproductive lifespan, a feature shared almost exclusively by humans and toothed whales [
100]. Consistent with this, odontocetes are also among the few non-human animals known to undergo menopause [
101]. This convergence raises the possibility that menopause-related hormonal shifts may interact with neuromodulatory vulnerability, potentially helping to explain the higher incidence of AD in women [
102] and fitting established links between oestrogen and cholinergic function [
103].
Together, these observations hint that AD may emerge where evolutionarily conserved neuromodulatory systems are pushed beyond originally selected limits to modulate much broader regions across significantly longer lifetimes. Comparative studies of neuromodulatory systems across humans, non-human primates and odontocetes could provide powerful opportunities to test the proposed framework.
9. Conclusions
We have proposed a neuromodulatory fragility framework for AD pathogenesis. This model offers a parsimonious account of how a single point of vulnerability (neuromodulatory projection neurons) can give rise to downstream hallmarks of AD, including the emergence of neurofibrillary tangles, Aβ plaques, neuroinflammation, cognitive-behavioural symptoms and dementia onset. The key insight of this model is that neuromodulatory system dysfunction is inherently tied to, and sufficient to explain, both tau hyperphosphorylation and increases in neocortical Aβ, the two hallmarks of AD. Given the inherent fragility of these volume transmission projection neurons in humans, it is no surprise through the lens of this framework that AD has such high global prevalence.
In this framework, genetic and environmental risk factors modulate AD risk through their cumulative effects on neuromodulatory resilience. The model predicts that strengthening these systems therefore represents the most effective strategy for preventing AD. Conversely, treatments that target downstream pathology are expected to offer only modest benefit once neuromodulatory dysfunction is established.
Our ideas and conclusions complement and expand upon recently proposed priorities for AD research from the Neuromodulatory Subcortical Systems Professional Interest Area of The Alzheimer's Association International Society to Advance Alzheimer's Research and Treatment [
17]. By describing this framework, we hope to further raise awareness of the importance of neuromodulatory subcortical systems in early AD, while providing a testable mechanistic explanation of pathogenesis that can help guide future research questions and the development of a new generation of effective disease-modifying therapies.
Funding
This work was funded by a NIH: National Institute of Aging R01 (RNS: AG068563), the Alzheimer’s Association (RNS: AARG-22-927100), Fonds de Recherche du Québec - Santé (AW # 317644, RNS), a Jeanne Timmins Costello Postdoctoral Fellowship, McGill University (AW). Funding sources had no say in the writing of this manuscript, nor decision to submit this manuscript for publication.
Acknowledgments
We would like to thank the Laboratory of Brain and Cognition at the Montreal Neurological Institute for productive discussions and helpful feedback.
Conflicts of Interest
Authors have no competing interests to declare.
References
- Duchesne, S. et al. A Scoping Review of Alzheimers Disease Hypotheses: An Array of Uni- and Multi-Factorial Theories. J Alzheimers Dis 99, 843–856 (2024). [CrossRef]
- Livingston, G. et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. The Lancet 396, 413–446 (2020). [CrossRef]
- Jellinger, K. A. Pathobiological Subtypes of Alzheimer Disease. Dement Geriatr Cogn Disord 49, 321–333 (2020). [CrossRef]
- Vogel, J. W. et al. Four distinct trajectories of tau deposition identified in Alzheimer’s disease. Nature Medicine 1–11 (2021). [CrossRef]
- Thal, D. R., Rüb, U., Orantes, M. & Braak, H. Phases of A-Beta-deposition in the human brain and its relevance for the development of AD. Neurology 58, 1791–1800 (2002). [CrossRef]
- Braak, H. & Braak, E. Staging of alzheimer’s disease-related neurofibrillary changes. Neurobiology of Aging 16, 271278 (1995). [CrossRef]
- Jack Jr., C. R. et al. Revised criteria for diagnosis and staging of Alzheimer’s disease: Alzheimer’s Association Workgroup. Alzheimer’s & Dementia 20, 5143–5169 (2024).
- Alzheimer’s Association. 2024 Alzheimer’s disease facts and figures. Alzheimers Dement 20, 3708–3821 (2024).
- Shanks, H. R. C. et al. p75 neurotrophin receptor modulation in mild to moderate Alzheimer disease: a randomized, placebo-controlled phase 2a trial. Nat Med 30, 1761–1770 (2024). [CrossRef]
- Theofilas, P., Dunlop, S., Heinsen, H. & Grinberg, L. T. Turning on the light within: subcortical nuclei of the isodentritic core and their role in Alzheimer’s disease pathogenesis. J Alzheimers Dis 46, 17–34 (2015). [CrossRef]
- Kampmann, M. Molecular and cellular mechanisms of selective vulnerability in neurodegenerative diseases. Nat. Rev. Neurosci. 1–21 (2024). [CrossRef]
- Mattson, M. P. & Arumugam, T. V. Hallmarks of Brain Aging: Adaptive and Pathological Modification by Metabolic States. Cell Metabolism 27, 1176–1199 (2018). [CrossRef]
- Saxena, S. & Caroni, P. Selective Neuronal Vulnerability in Neurodegenerative Diseases: from Stressor Thresholds to Degeneration. Neuron 71, 35–48 (2011). [CrossRef]
- Selkoe, D. J. & Hardy, J. The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Molecular Medicine 8, 595–608 (2016). [CrossRef]
- Shine, J. M. et al. Human cognition involves the dynamic integration of neural activity and neuromodulatory systems. Nat Neurosci 22, 289–296 (2019). [CrossRef]
- Shine, J. M. Neuromodulatory Influences on Integration and Segregation in the Brain. Trends in Cognitive Sciences 23, 572–583 (2019). [CrossRef]
- Ehrenberg, A. J. et al. Priorities for research on neuromodulatory subcortical systems in Alzheimer’s disease: Position paper from the NSS PIA of ISTAART. Alzheimer’s & Dementia. [CrossRef]
- Mather, M. & Harley, C. W. The Locus Coeruleus: Essential for Maintaining Cognitive Function and the Aging Brain. Trends in Cognitive Sciences 20, 214–226 (2016). [CrossRef]
- Picciotto, M. R., Higley, M. J. & Mineur, Y. S. Acetylcholine as a Neuromodulator: Cholinergic Signaling Shapes Nervous System Function and Behavior. Neuron 76, 116–129 (2012). [CrossRef]
- Pignatelli, M. & Bonci, A. Role of Dopamine Neurons in Reward and Aversion: A Synaptic Plasticity Perspective. Neuron 86, 1145–1157 (2015). [CrossRef]
- Sakurai, T. The neural circuit of orexin (hypocretin): maintaining sleep and wakefulness. Nat Rev Neurosci 8, 171–181 (2007). [CrossRef]
- Hornung, J.-P. The human raphe nuclei and the serotonergic system. Journal of Chemical Neuroanatomy 26, 331–343 (2003). [CrossRef]
- Özçete, Ö. D., Banerjee, A. & Kaeser, P. S. Mechanisms of neuromodulatory volume transmission. Mol Psychiatry 29, 3680–3693 (2024). [CrossRef]
- Wu, H., Williams, J. & Nathans, J. Complete morphologies of basal forebrain cholinergic neurons in the mouse. eLife 3, e02444 (2014). [CrossRef]
- Aransay, A., Rodríguez-López, C., García-Amado, M., Clascá, F. & Prensa, L. Long-range projection neurons of the mouse ventral tegmental area: a single-cell axon tracing analysis. Front. Neuroanat. 9, (2015). [CrossRef]
- Wu, Y.-M., Hung, W.-C., Chang, Y., Min, M.-Y. & Yang, H.-W. Axon collateral pattern of a sparse locus coeruleus norepinephrine neuron in mouse cerebral cortex. 2025.02.04.636453. [CrossRef]
- Trillo, L. et al. Ascending monoaminergic systems alterations in Alzheimer’s disease. Translating basic science into clinical care. Neuroscience & Biobehavioral Reviews 37, 1363–1379 (2013). [CrossRef]
- Giguère, N. et al. Increased vulnerability of nigral dopamine neurons after expansion of their axonal arborization size through D2 dopamine receptor conditional knockout. PLOS Genetics 15, e1008352 (2019). [CrossRef]
- Aston-Jones, G. & Cohen, J. D. AN INTEGRATIVE THEORY OF LOCUS COERULEUS-NOREPINEPHRINE FUNCTION: Adaptive Gain and Optimal Performance. Annu. Rev. Neurosci. 28, 403–450 (2005). [CrossRef]
- Grace, A. A. & Bunney, B. S. The control of firing pattern in nigral dopamine neurons: single spike firing. The Journal of neuroscience: the official journal of the Society for Neuroscience 4, 2866–2876 (1984). [CrossRef]
- Allers, K. A. & Sharp, T. Neurochemical and anatomical identification of fast- and slow-firing neurones in the rat dorsal raphe nucleus using juxtacellular labelling methods in vivo. Neuroscience 122, 193–204 (2003). [CrossRef]
- Ananth, M. R., Rajebhosale, P., Kim, R., Talmage, D. A. & Role, L. W. Basal forebrain cholinergic signalling: development, connectivity and roles in cognition. Nat Rev Neurosci 24, 233–251 (2023). [CrossRef]
- Braak, H., Del Tredici, K., Schultz, C. & Braak, E. Vulnerability of Select Neuronal Types to Alzheimer’s Disease. Annals of the New York Academy of Sciences 924, 53–61 (2000). [CrossRef]
- Braak, H. & Del Tredici, K. Poor and protracted myelination as a contributory factor to neurodegenerative disorders. Neurobiol Aging 25, 19–23 (2004). [CrossRef]
- Braak, H. & Braak, E. Development of Alzheimer-related neurofibrillary changes in the neocortex inversely recapitulates cortical myelogenesis. Acta Neuropathol 92, 197–201 (1996). [CrossRef]
- Pontzer, H. et al. Daily energy expenditure through the human life course. Science 373, 808–812 (2021). [CrossRef]
- Matchett, B. J., Grinberg, L. T., Theofilas, P. & Murray, M. E. The mechanistic link between selective vulnerability of the locus coeruleus and neurodegeneration in Alzheimer’s disease. Acta Neuropathol 141, 631–650 (2021). [CrossRef]
- Iannitelli, A. F. & Weinshenker, D. Riddles in the dark: Decoding the relationship between neuromelanin and neurodegeneration in locus coeruleus neurons. Neurosci Biobehav Rev 152, 105287 (2023). [CrossRef]
- Geula, C. et al. Loss of calbindin-D28k from aging human cholinergic basal forebrain: relation to neuronal loss. J Comp Neurol 455, 249–259 (2003). [CrossRef]
- Troutwine, B. R., Hamid, L., Lysaker, C. R., Strope, T. A. & Wilkins, H. M. Apolipoprotein E and Alzheimer’s disease. Acta Pharmaceutica Sinica B 12, 496–510 (2022).
- Blanchard, J. W. et al. APOE4 impairs myelination via cholesterol dysregulation in oligodendrocytes. Nature 611, 769–779 (2022). [CrossRef]
- Carandini, T., Cercignani, M., Galimberti, D., Scarpini, E. & Bozzali, M. The distinct roles of monoamines in multiple sclerosis: A bridge between the immune and nervous systems? Brain, Behavior, and Immunity 94, 381–391 (2021). [CrossRef]
- Barbier, P. et al. Role of Tau as a Microtubule-Associated Protein: Structural and Functional Aspects. Front. Aging Neurosci. 11, (2019). [CrossRef]
- de Vos, K. J., Grierson, A. J., Ackerley, S. & Miller, C. C. J. Role of Axonal Transport in Neurodegenerative Diseases*. Annual Review of Neuroscience 31, 151–173 (2008). [CrossRef]
- Wang, Z.-X., Tan, L. & Yu, J.-T. Axonal transport defects in Alzheimer’s disease. Mol Neurobiol 51, 1309–1321 (2015). [CrossRef]
- Scholz, T. & Mandelkow, E. Transport and diffusion of Tau protein in neurons. Cell. Mol. Life Sci. 71, 3139–3150 (2014). [CrossRef]
- Hussain, L. S., Reddy, V. & Maani, C. V. Physiology, Noradrenergic Synapse. in StatPearls (StatPearls Publishing, Treasure Island (FL), 2025).
- Chalermpalanupap, T., Weinshenker, D. & Rorabaugh, J. M. Down but Not Out: The Consequences of Pretangle Tau in the Locus Coeruleus. Neural Plast 2017, 7829507 (2017). [CrossRef]
- Braak, H. & Del Tredici, K. The preclinical phase of the pathological process underlying sporadic Alzheimer’s disease. Brain 138, 2814–2833 (2015). [CrossRef]
- Braak, H., Thal, D. R., Ghebremedhin, E. & Del Tredici, K. Stages of the Pathologic Process in Alzheimer Disease: Age Categories From 1 to 100 Years. Journal of Neuropathology & Experimental Neurology 70, 960–969 (2011). [CrossRef]
- Geula, C., Nagykery, N., Nicholas, A. & Wu, C.-K. Cholinergic neuronal and axonal abnormalities are present early in aging and in Alzheimer disease. J Neuropathol Exp Neurol 67, 309–318 (2008). [CrossRef]
- Satpati, A. et al. The wake- and sleep-modulating neurons of the lateral hypothalamic area demonstrate a differential pattern of degeneration in Alzheimer’s disease. 2024.03.06.583765. [CrossRef]
- Grinberg, L. T. et al. The dorsal raphe nucleus shows phospho-tau neurofibrillary changes before the transentorhinal region in Alzheimer’s disease. A precocious onset? Neuropathology and Applied Neurobiology 35, 406–416 (2009). 2009. [CrossRef]
- Pierson, S. R. et al. Tau pathology in the dorsal raphe may be a prodromal indicator of Alzheimer’s disease. Mol Psychiatry 30, 532–546 (2025). [CrossRef]
- Cranston, A. L. et al. Cholinergic and inflammatory phenotypes in transgenic tau mouse models of Alzheimer’s disease and frontotemporal lobar degeneration. Brain Commun 2, fcaa033 (2020). [CrossRef]
- Wu, J. W. et al. Neuronal activity enhances tau propagation and tau pathology in vivo. Nat Neurosci 19, 1085–1092 (2016). [CrossRef]
- Therriault, J. et al. Intrinsic connectivity of the human brain provides scaffold for tau aggregation in clinical variants of Alzheimer’s disease. Science Translational Medicine 14, eabc8693 (2022). [CrossRef]
- Bueichekú, E. et al. Spatiotemporal patterns of locus coeruleus integrity predict cortical tau and cognition. Nat Aging 4, 625–637 (2024). [CrossRef]
- Jacobs, H. I. L. et al. In vivo and neuropathology data support locus coeruleus integrity as indicator of Alzheimer’s disease pathology and cognitive decline. Sci. Transl. Med. 13, eabj2511 (2021). [CrossRef]
- Schmitz, T. W. & Spreng, R. N. Basal forebrain degeneration precedes and predicts the cortical spread of Alzheimer’s pathology. Nature Communications 7, 13249 (2016). [CrossRef]
- Fernández-Cabello, S. et al. Basal forebrain volume reliably predicts the cortical spread of Alzheimer’s degeneration. Brain 143, 993–1009 (2020). [CrossRef]
- Yi, D. et al. Locus coeruleus tau is linked to successive cortical tau accumulation. Alzheimer’s & Dementia 21, e14426 (2025). [CrossRef]
- Roemer-Cassiano, S. N. et al. Amyloid-associated hyperconnectivity drives tau spread across connected brain regions in Alzheimer’s disease. Sci Transl Med 17, eadp2564 (2025). [CrossRef]
- Salvadores, N., Gerónimo-Olvera, C. & Court, F. A. Axonal Degeneration in AD: The Contribution of Aβ and Tau. Front Aging Neurosci 12, 581767 (2020). [CrossRef]
- Heneka, M. T. et al. Locus ceruleus controls Alzheimer’s disease pathology by modulating microglial functions through norepinephrine. Proceedings of the National Academy of Sciences 107, 6058–6063 (2010). [CrossRef]
- Gamage, R. et al. Cholinergic Modulation of Glial Function During Aging and Chronic Neuroinflammation. Front. Cell. Neurosci. 14, (2020). [CrossRef]
- Schmitz, T. W., Soreq, H., Poirier, J. & Spreng, R. N. Longitudinal Basal Forebrain Degeneration Interacts with TREM2/C3 Biomarkers of Inflammation in Presymptomatic Alzheimer’s Disease. J. Neurosci. 40, 1931–1942 (2020). [CrossRef]
- Wang, H. et al. Nicotinic acetylcholine receptor α7 subunit is an essential regulator of inflammation. Nature 421, 384–388 (2003).
- Ramos-Rodriguez, J. J. et al. Rapid β-amyloid deposition and cognitive impairment after cholinergic denervation in APP/PS1 mice. J Neuropathol Exp Neurol 72, 272–285 (2013). [CrossRef]
- Chalermpalanupap, T. et al. Targeting norepinephrine in mild cognitive impairment and Alzheimer’s disease. Alzheimers Res Ther 5, 21 (2013). [CrossRef]
- Gil-Bea, F. J. et al. Cholinergic denervation exacerbates amyloid pathology and induces hippocampal atrophy in Tg2576 mice. Neurobiol Dis 48, 439–446 (2012). [CrossRef]
- Frick, M. A. et al. Orexin/Hypocretin Modulates Neuroinflammatory Response to LPS in a Sex and Brain-Region Specific Manner in Young Rats. J Neurochem 169, e70175 (2025). [CrossRef]
- Muresan, Z. & Muresan, V. Seeding Neuritic Plaques from the Distance: A Possible Role for Brainstem Neurons in the Development of Alzheimer’s Disease Pathology. Neurodegener Dis 5, 250–253 (2008). [CrossRef]
- Baker-Nigh, A. et al. Neuronal amyloid-β accumulation within cholinergic basal forebrain in ageing and Alzheimer’s disease. Brain 138, 1722–1737 (2015). [CrossRef]
- Ossenkoppele, R. et al. Amyloid and tau PET-positive cognitively unimpaired individuals are at high risk for future cognitive decline. Nature Medicine 28, 2381 (2022). [CrossRef]
- La Joie, R. et al. Prospective longitudinal atrophy in Alzheimer’s disease correlates with the intensity and topography of baseline tau-PET. Sci Transl Med 12, eaau5732 (2020).
- Bloom, G. S. Amyloid-β and Tau: The Trigger and Bullet in Alzheimer Disease Pathogenesis. JAMA Neurol 71, 505–508 (2014).
- Busche, M. A. & Hyman, B. T. Synergy between amyloid-β and tau in Alzheimer’s disease. Nat Neurosci 23, 1183–1193 (2020).
- Crary, J. F. et al. Primary age-related tauopathy (PART): a common pathology associated with human aging. Acta Neuropathol 128, 755–766 (2014). [CrossRef]
- Lee, W. J. et al. Regional Aβ-tau interactions promote onset and acceleration of Alzheimer’s disease tau spreading. Neuron 110, 1932-1943.e5 (2022). [CrossRef]
- Vogel, J. W. et al. Connectome-based modelling of neurodegenerative diseases: towards precision medicine and mechanistic insight. Nat. Rev. Neurosci. 24, 620–639 (2023). [CrossRef]
- Karran, E. & De Strooper, B. The amyloid hypothesis in Alzheimer disease: new insights from new therapeutics. Nat Rev Drug Discov 21, 306–318 (2022).
- Braak, H. & Del Tredici, K. Alzheimer’s disease: intraneuronal alterations precede insoluble amyloid-β formation. Neurobiology of Aging 25, 713–718 (2004). [CrossRef]
- Götz, J., Chen, F., van Dorpe, J. & Nitsch, R. M. Formation of Neurofibrillary Tangles in P301L Tau Transgenic Mice Induced by Aβ42 Fibrils. Science 293, 1491–1495 (2001). [CrossRef]
- Stern, Y., Barnes, C. A., Grady, C., Jones, R. N. & Raz, N. Brain Reserve, Cognitive Reserve, Compensation, and Maintenance: Operationalization, Validity, and Mechanisms of Cognitive Resilience. Neurobiol Aging 83, 124–129 (2019). [CrossRef]
- Stella, F. et al. Neuropsychiatric symptoms in the prodromal stages of dementia. Current Opinion in Psychiatry 27, 230 (2014). [CrossRef]
- Orlando, I. F., Shine, J. M., Robbins, T. W., Rowe, J. B. & O’Callaghan, C. Noradrenergic and cholinergic systems take centre stage in neuropsychiatric diseases of ageing. Neuroscience & Biobehavioral Reviews 149, 105167 (2023). [CrossRef]
- Engels-Domínguez, N. et al. State-of-the-art imaging of neuromodulatory subcortical systems in aging and Alzheimer’s disease: Challenges and opportunities. Neuroscience & Biobehavioral Reviews 144, 104998 (2023). [CrossRef]
- Wearn, A. et al. Neuromodulatory subcortical nucleus integrity is associated with white matter microstructure, tauopathy and APOE status. Nat Commun 15, 4706 (2024). [CrossRef]
- Chakraborty, S. et al. Multimodal gradients of basal forebrain connectivity across the neocortex. Nat Commun 15, 8990 (2024). [CrossRef]
- Wood Alexander, M. et al. The interplay between age at menopause and synaptic integrity on Alzheimer’s disease risk in women. Science Advances 11, eadt0757 (2025). [CrossRef]
- ang, M.-X. et al. Incidence of AD in African-Americans, Caribbean Hispanics, and Caucasians in northern Manhattan. Neurology 56, 49–56 (2001). [CrossRef]
- Toussaint, C., Bézard, E., Lemoine, M. & Planche, V. The biological scaling of Alzheimer’s disease neuropathological changes across primate species. Alzheimers Res Ther 17, 242 (2025). [CrossRef]
- Marsden, C. D. Pigmentation in the nucleus substantiae nigrae of mammals. J Anat 95, 256–261 (1961).
- Vacher, M. C. et al. Alzheimer’s disease-like neuropathology in three species of oceanic dolphin. Eur J Neurosci 57, 1161–1179 (2023). [CrossRef]
- Sacchini, S. et al. Toothed Whales Have Black Neurons in the Blue Spot. Vet Sci 9, 525 (2022). [CrossRef]
- Sharma, Y. et al. Comparative anatomy of the locus coeruleus in humans and nonhuman primates. J Comp Neurol 518, 963–971 (2010). [CrossRef]
- Raghanti, M. A. et al. Comparative analysis of the nucleus basalis of Meynert among primates. Neuroscience 184, 1–15 (2011). [CrossRef]
- Worthy, G. A. J. & Hickie, J. P. Relative Brain Size in Marine Mammals. The American Naturalist 128, 445–459 (1986). [CrossRef]
- Gunn-Moore, D., Kaidanovich-Beilin, O., Iradi, M. C. G., Gunn-Moore, F. & Lovestone, S. Alzheimer’s disease in humans and other animals: A consequence of postreproductive life span and longevity rather than aging. Alzheimer’s & Dementia 14, 195–204 (2018). [CrossRef]
- Ellis, S., Franks, D. W., Nielsen, M. L. K., Weiss, M. N. & Croft, D. P. The evolution of menopause in toothed whales. Nature 627, 579–585 (2024). [CrossRef]
- Chêne, G. et al. Gender and incidence of dementia in the Framingham Heart Study from mid-adult life. Alzheimer’s & Dementia 11, 310–320 (2015). [CrossRef]
- Newhouse, P. & Dumas, J. Estrogen-Cholinergic Interactions: Implications for Cognitive Aging. Horm Behav 74, 173–185 (2015). [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).