1. Introduction
The attached gingiva (AG) is the portion of the gingiva that is firm, dense, stippled, and tightly bound to the underlying periosteum, tooth, and bone. It is composed of keratinized epithelium (KT) supported by dense connective tissue largely filled with tightly packed collagen fibers bundles [
1].
In the apical direction, the gingiva is continuous with the non-keratinized alveolar mucosa that is mobile and loosely bound to the underlying tooth and bone. The demarcation line between the gingiva and the alveolar mucosa is named the mucogingival junction [
1].
Histologically, on its coronal aspect, the junctional epithelium (JE), which provides the first contact between the gingiva and the tooth, forms the epithelial attachment [
2]. The connective fibers that are embedded into root cementum just apical to the junctional epithelium, constitute the connective attachment to the tooth [
3].
In the permanent dentition, the width of the AG varies from 1 to 9mm [
4]. Clinically, the AG width is calculated by subtracting the sulcus or pocket depth from the distance between the gingival margin and the mucogingival junction. This measurement includes the JE as part of the AG, although their insertions differ in structure and functional role [
2]. Understanding these distinctions is essential when evaluating the functional significance of the AG in periodontal stability.
Carnio and co-workers proposed that the AG increases the resistance of the periodontium to external injury, contributes to the stability of the gingival margin position, and helps dissipate of physiological forces exerted by the muscular fibers of the alveolar mucosa onto the gingival tissues [
5].
From a clinical standpoint, several studies have suggested a minimal width of AG is required to maintain periodontal health [
6,
7,
8,
9,
10,
11,
12]. Lang & Loe (1972) and, more recently Scheyer et al. (2014) recommended a minimum of 2 mm of KT, with at least 1 mm of AG in patients with suboptimal plaque control [
13,
14]. However, subsequent investigations have challenged this requirement, demonstrating that periodontal health may be preserved even in areas with minimal or absent AG if inflammation and trauma are effectively controlled [
15,
16].
The average supracrestal tissue attachment (SCTA), previously referred as biologic width, consists of approximately 1 mm of epithelial attachment (EA) and 1 mm of connective tissue attachment. These dimensions have been confirmed histologically under healthy conditions and provide important reference parameters for periodontal and restorative treatment planning. (17–19)
When considering the collective evidence, it becomes clear that, in the presence of 2 mm of KT, the adherence corresponding to the AG would consist primarily of JE attachment [
13,
17,
18,
19,
20,
21,
22,
23,
24].
Schroeder and Listgarten noted that the JE serves as an anatomic feature that facilitates the migration of neutrophils and for the transient influx of mononuclear leucocytes, which serve as a major participant in host defenses against bacterial infection [
25]. However, its attachment, mediated through hemidesmosomes, is biologically fragile and insufficient to withstand mechanical forces exerted by alveolar mucosal onto the gingival margin [
20,
24]. The EA is important in the protection of the periodontium against biofilm accumulation, and such a role does not depend on the amount of KT present in the area [
13,
14,
15,
16,
17,
18,
19,
21,
22,
23].
Waerhaug demonstrated that inserting dental floss to the base of the sulcus and moving it back and forth three to five times against the tooth surface caused detachment of JE cells [
26]. Similarly, periodontal probing can disrupt the JE even in non-inflamed sites [
27]. The limited number of desmosomes connecting the junctional epithelial cells, and the variability the intercellular spaces dimensions explain why the EA is a weak barrier against trauma [
2,
18,
26].
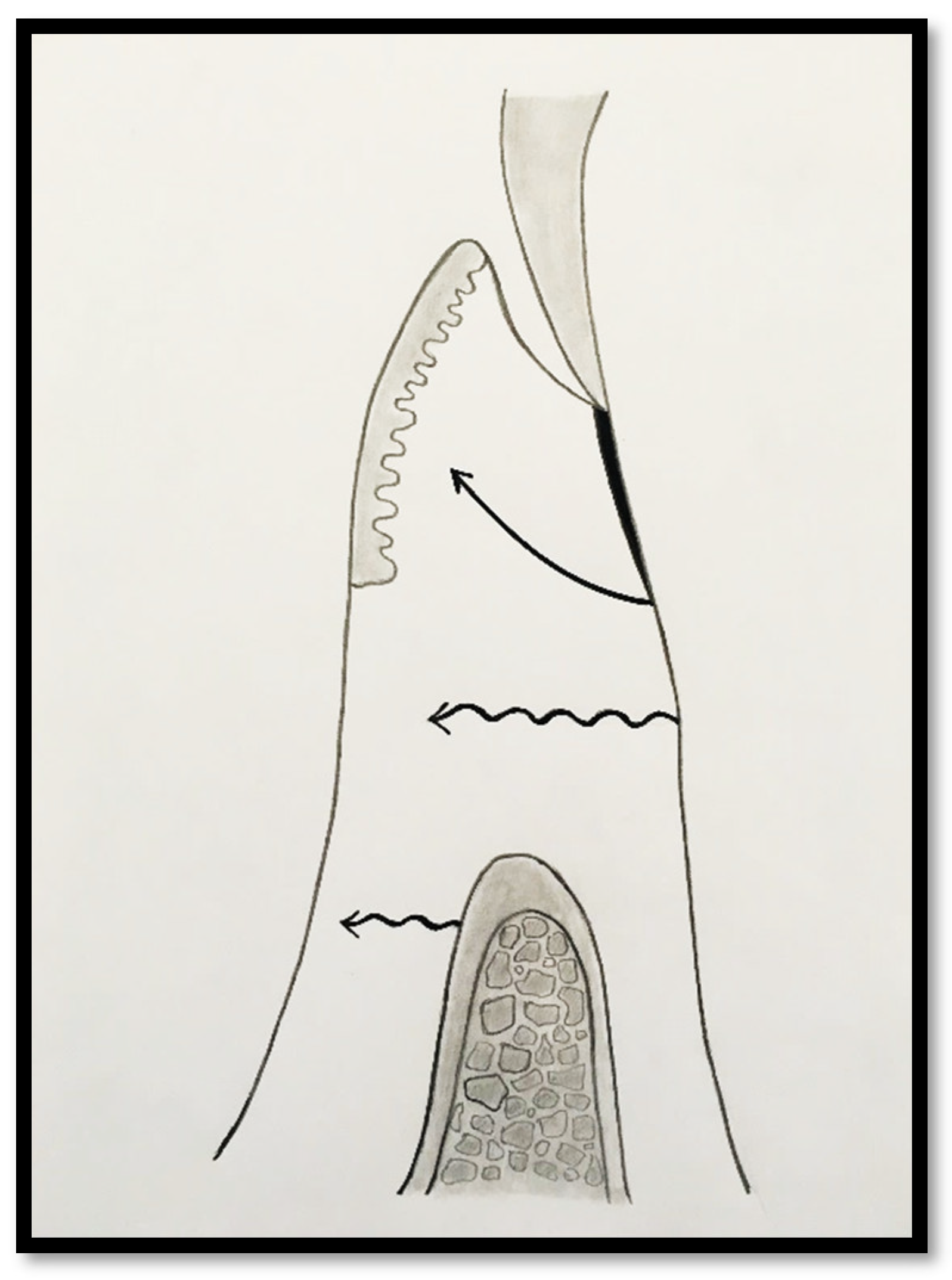
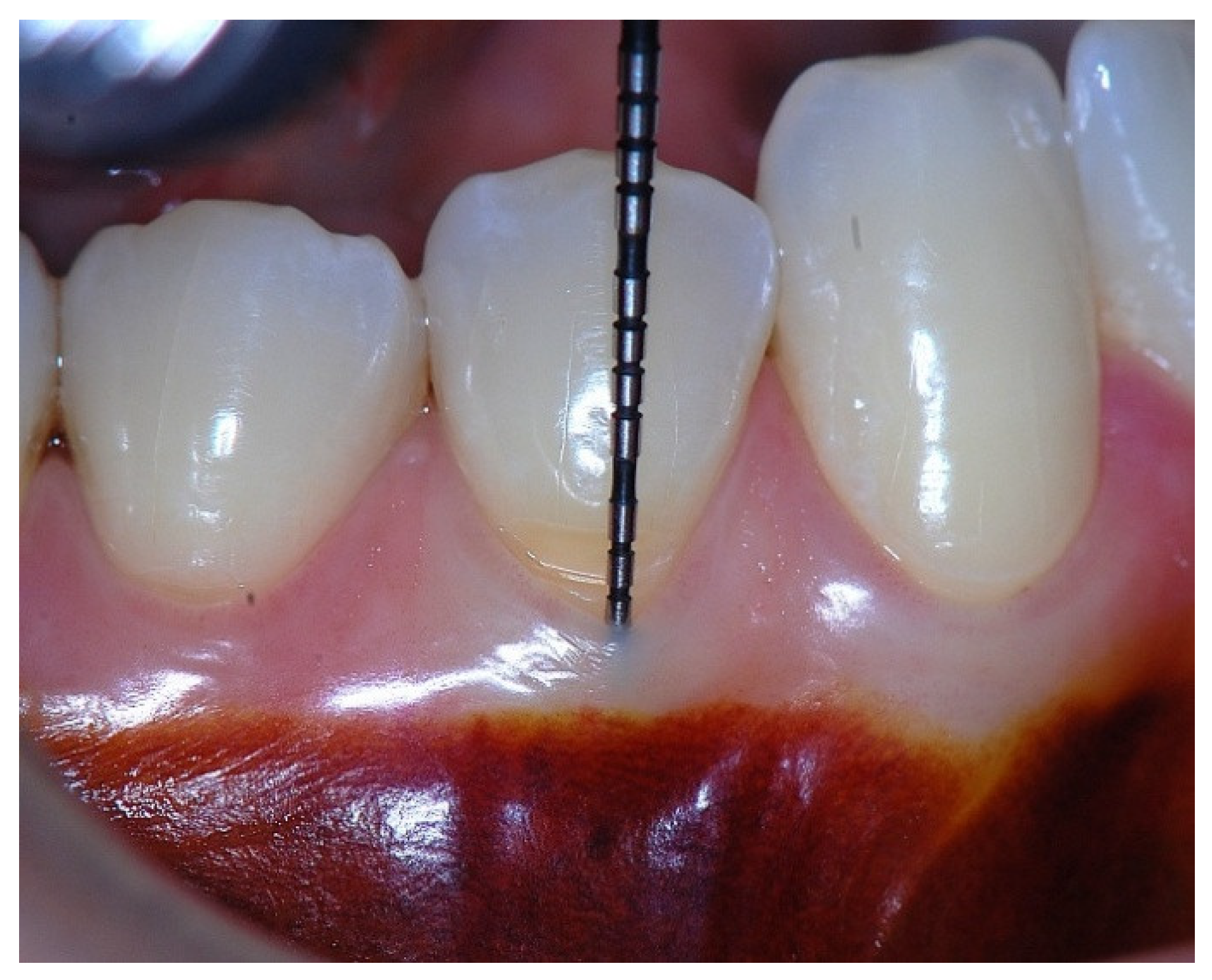
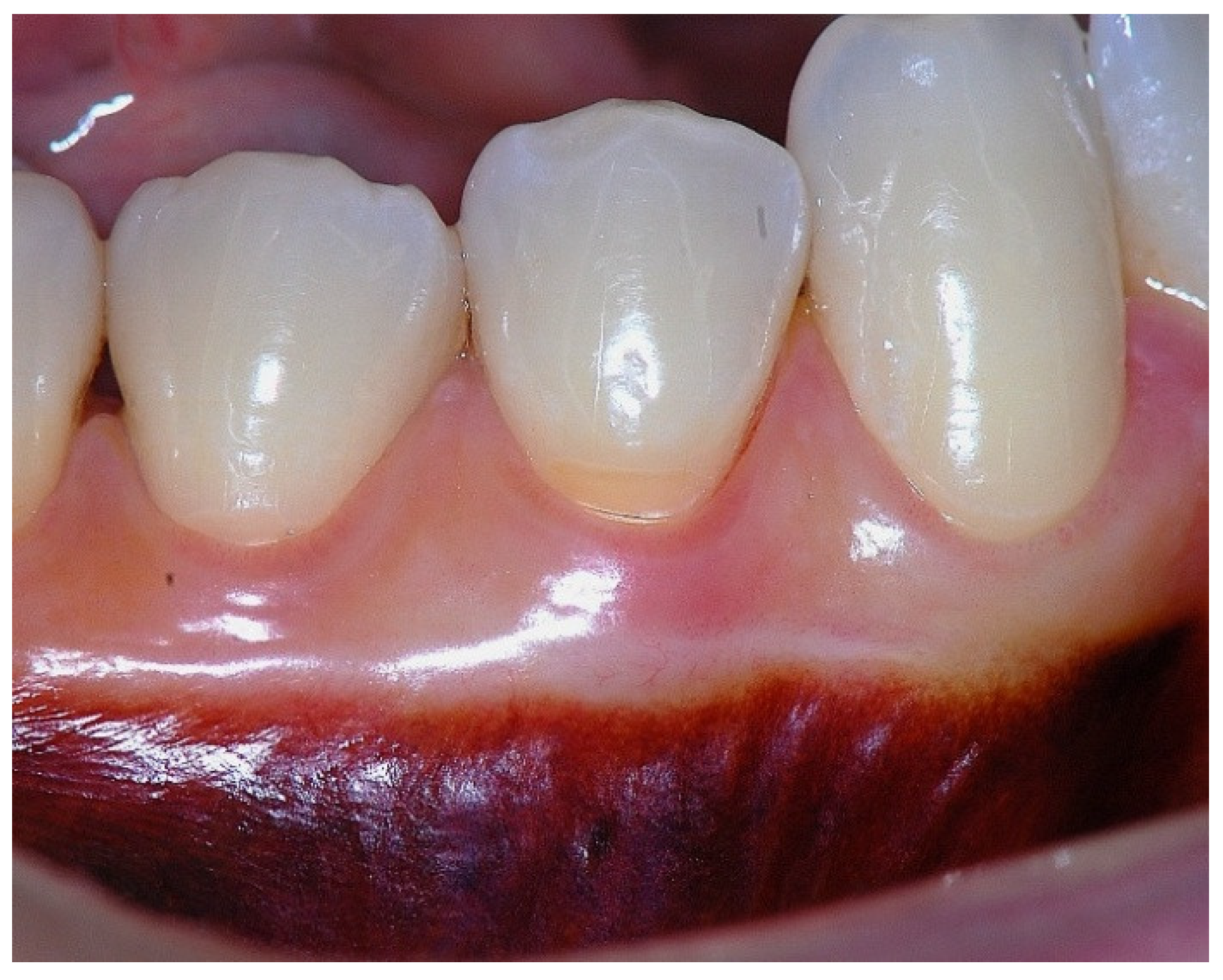
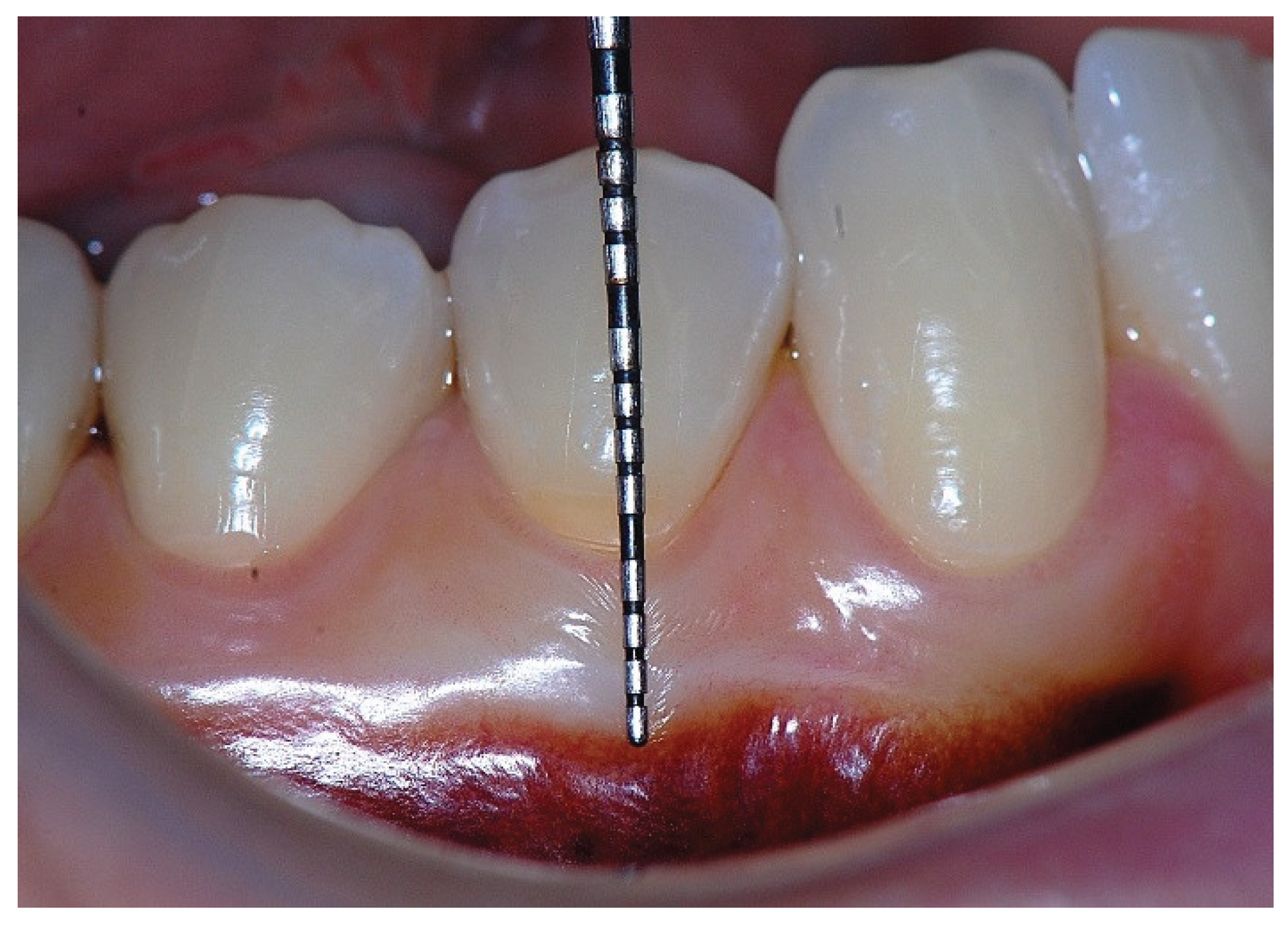
In a clinical scenario with 2 mm of keratinized tissue (KT) and a sulcus depth of 1 mm, a stable, firm, and resistant connective attachment is not present [
1]. This occurs because most of the connective tissue fibers inserting onto the root surface are associated with non-keratinized, mobile mucosal tissues (
Figure 1), which are elastic in nature and therefore unable to stabilize the gingival margin (
Figure 4 and
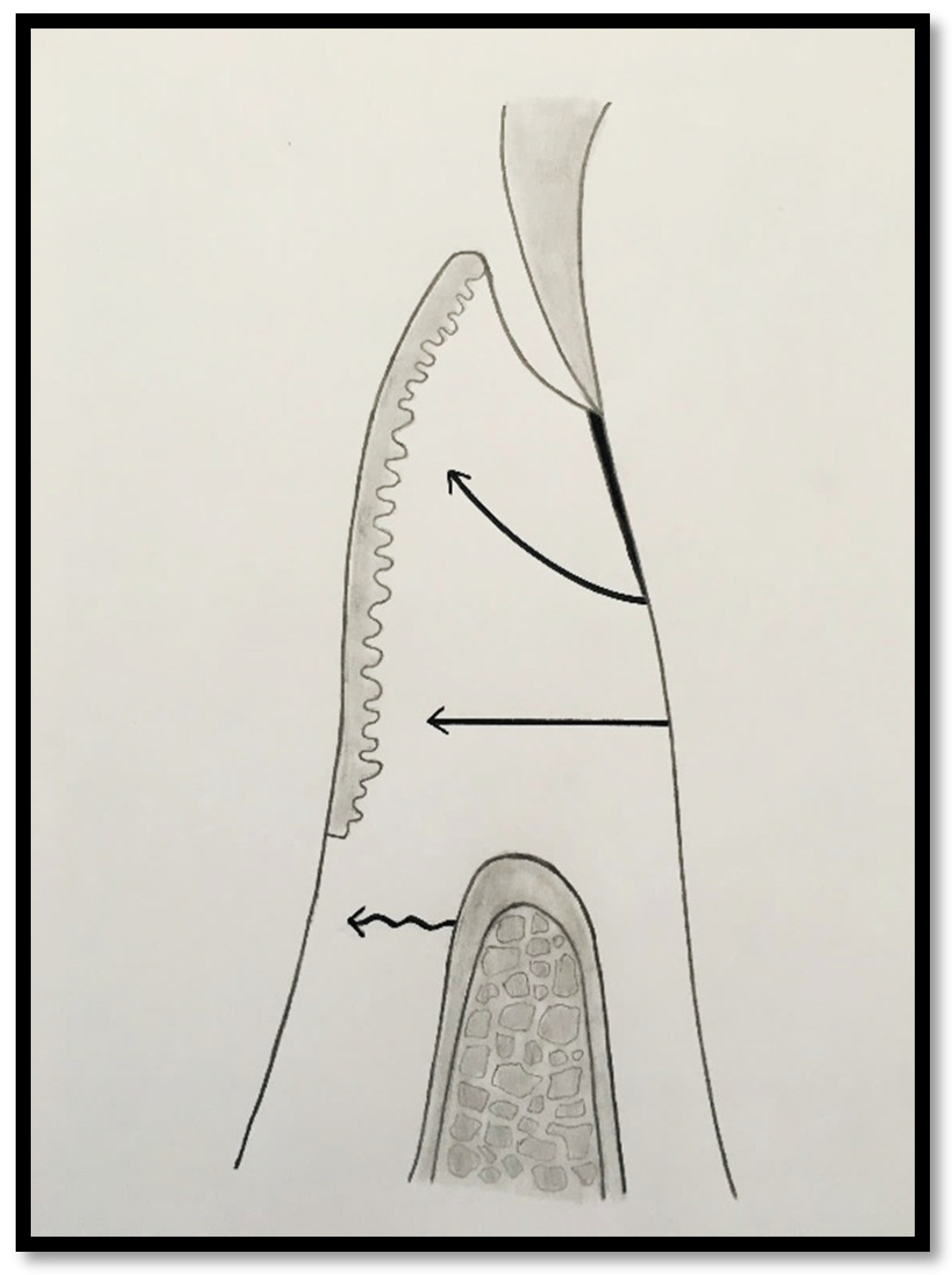
Figure 5). Conversely, when these fibers are embedded within keratinized tissue, they are firmly attached to the tooth and bone and are comparatively immobile, thereby contributing to gingival margin stability (
Figure 2,
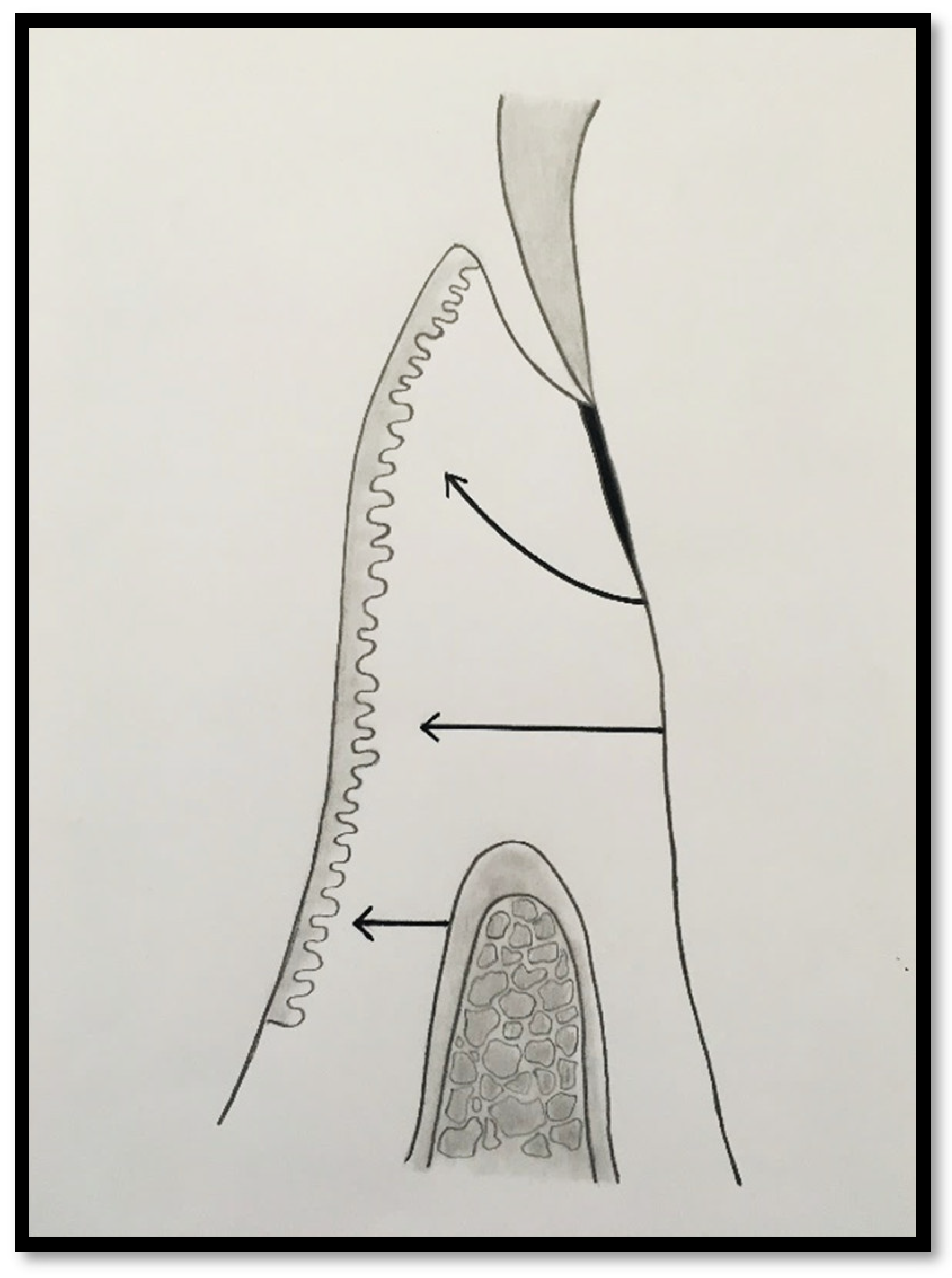
Figure 3, and
Figure 6).
Figure 1.
Probing depth of 1mm and 1mm of the epithelial attachment.
Figure 1.
Probing depth of 1mm and 1mm of the epithelial attachment.
Figure 2.
Probing depth of 1mm, 1mm of epithelial attachment, and 1 mm of connective tissue fiber attachment to the root cementum.
Figure 2.
Probing depth of 1mm, 1mm of epithelial attachment, and 1 mm of connective tissue fiber attachment to the root cementum.
Figure 3.
Probing depth of 1mm, 1mm of epithelial attachment, 1 mm of connective tissue fiber attachment to the root cementum, and connective tissue fibers inserted into alveolar bone.
Figure 3.
Probing depth of 1mm, 1mm of epithelial attachment, 1 mm of connective tissue fiber attachment to the root cementum, and connective tissue fibers inserted into alveolar bone.
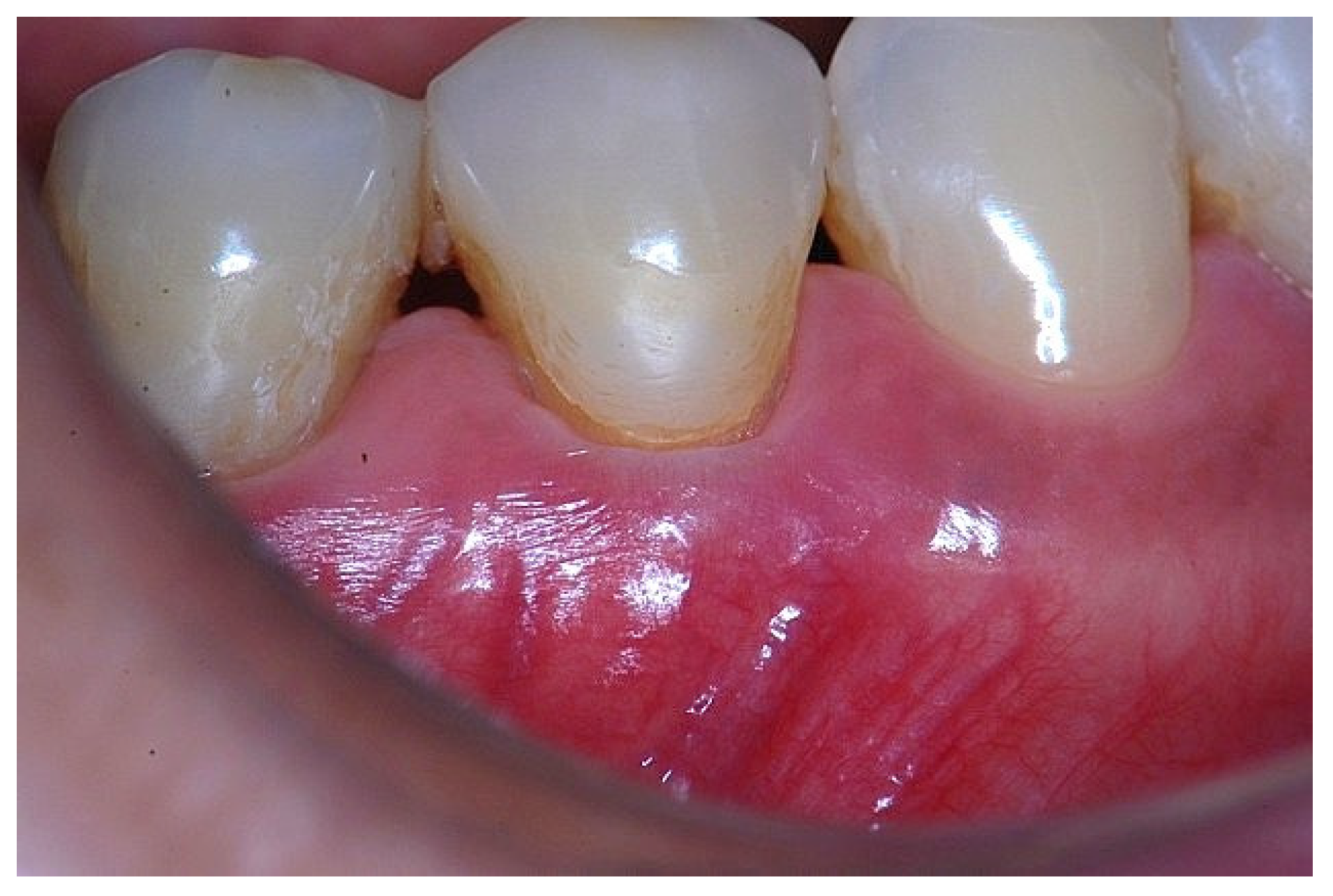
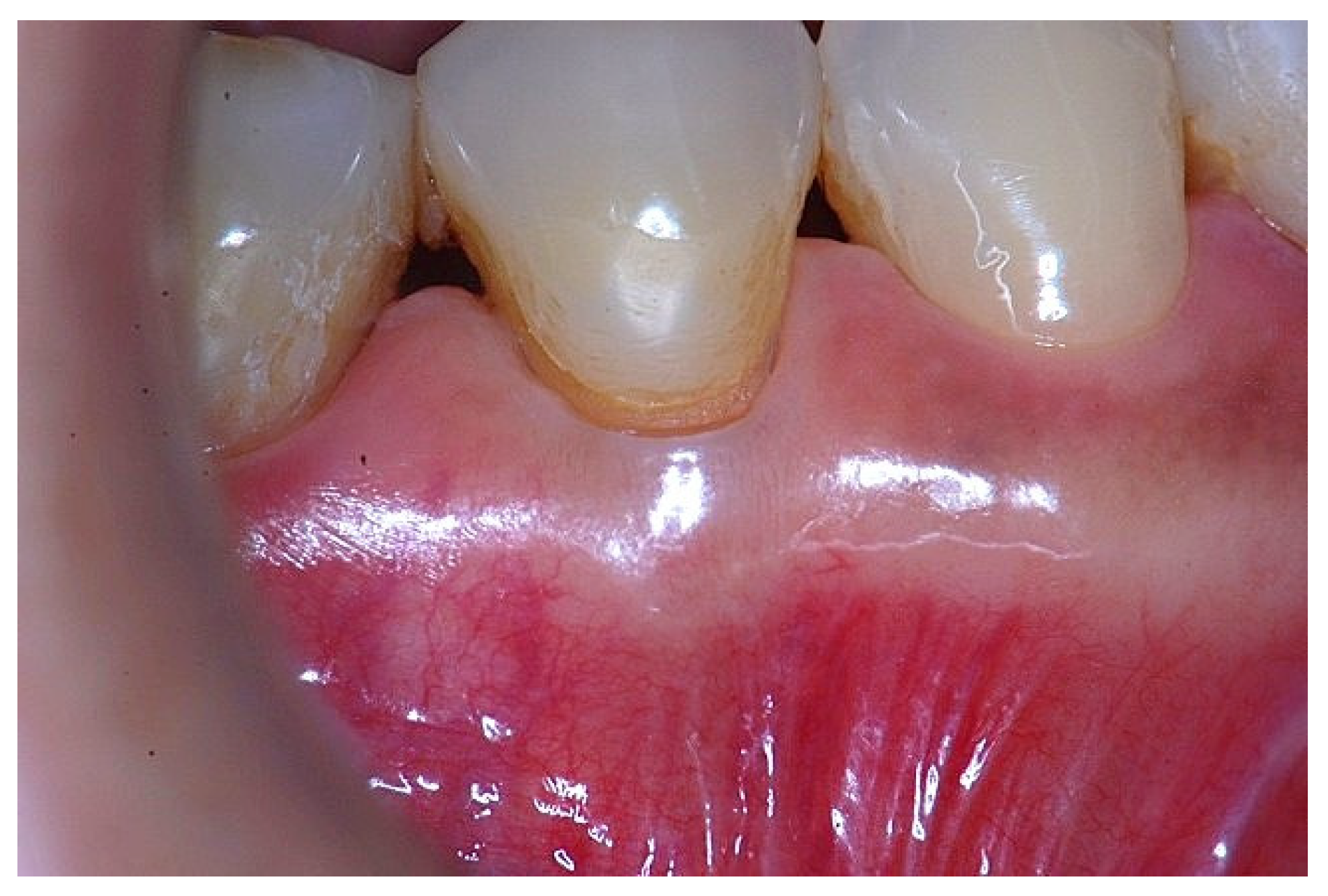
Figure 4.
Clinical image of tooth #29 showing an insufficient width of attached gingiva.
Figure 4.
Clinical image of tooth #29 showing an insufficient width of attached gingiva.
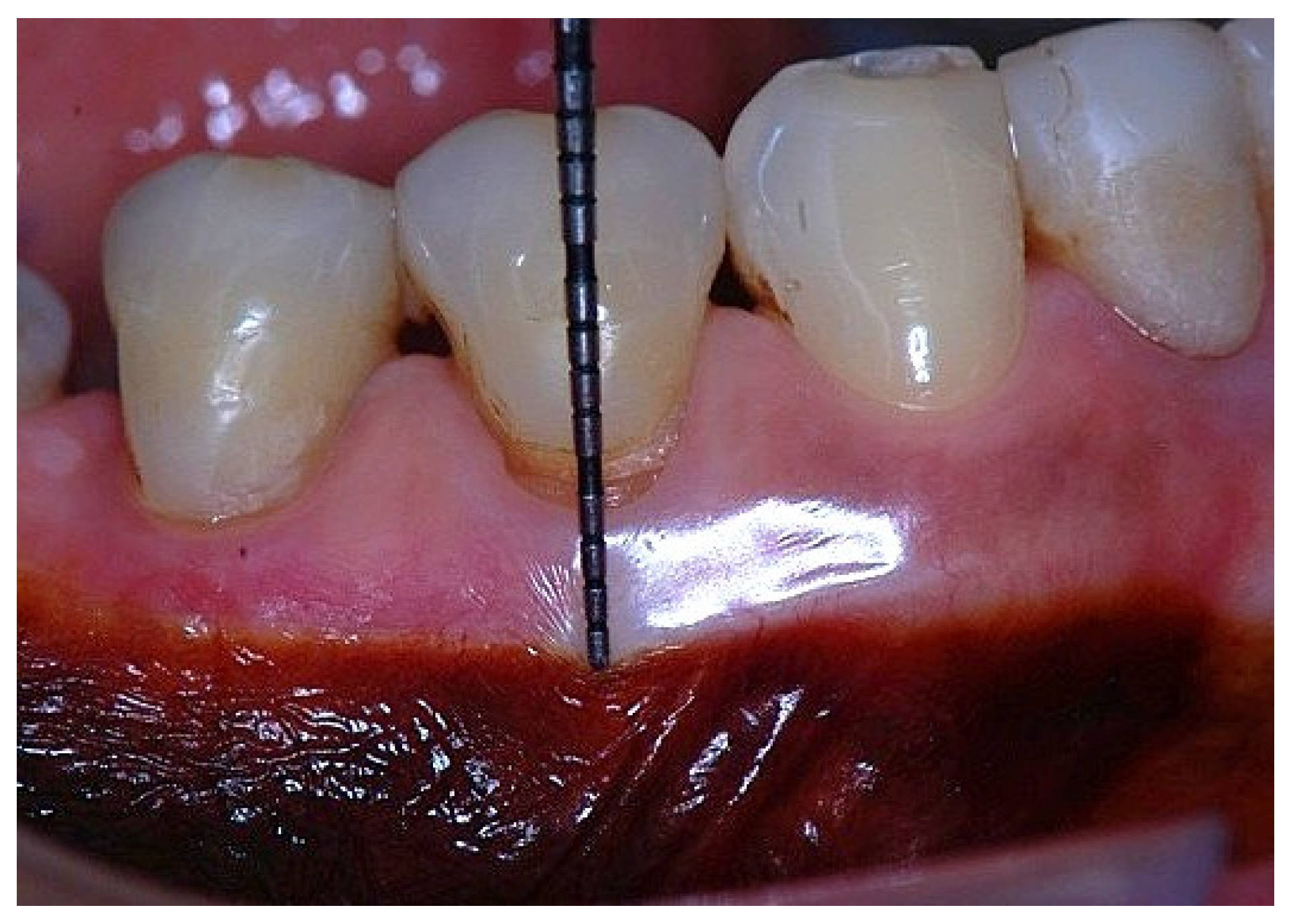
Figure 5.
Apical displacement of the gingival margin observed during deep lip retraction.
Figure 5.
Apical displacement of the gingival margin observed during deep lip retraction.
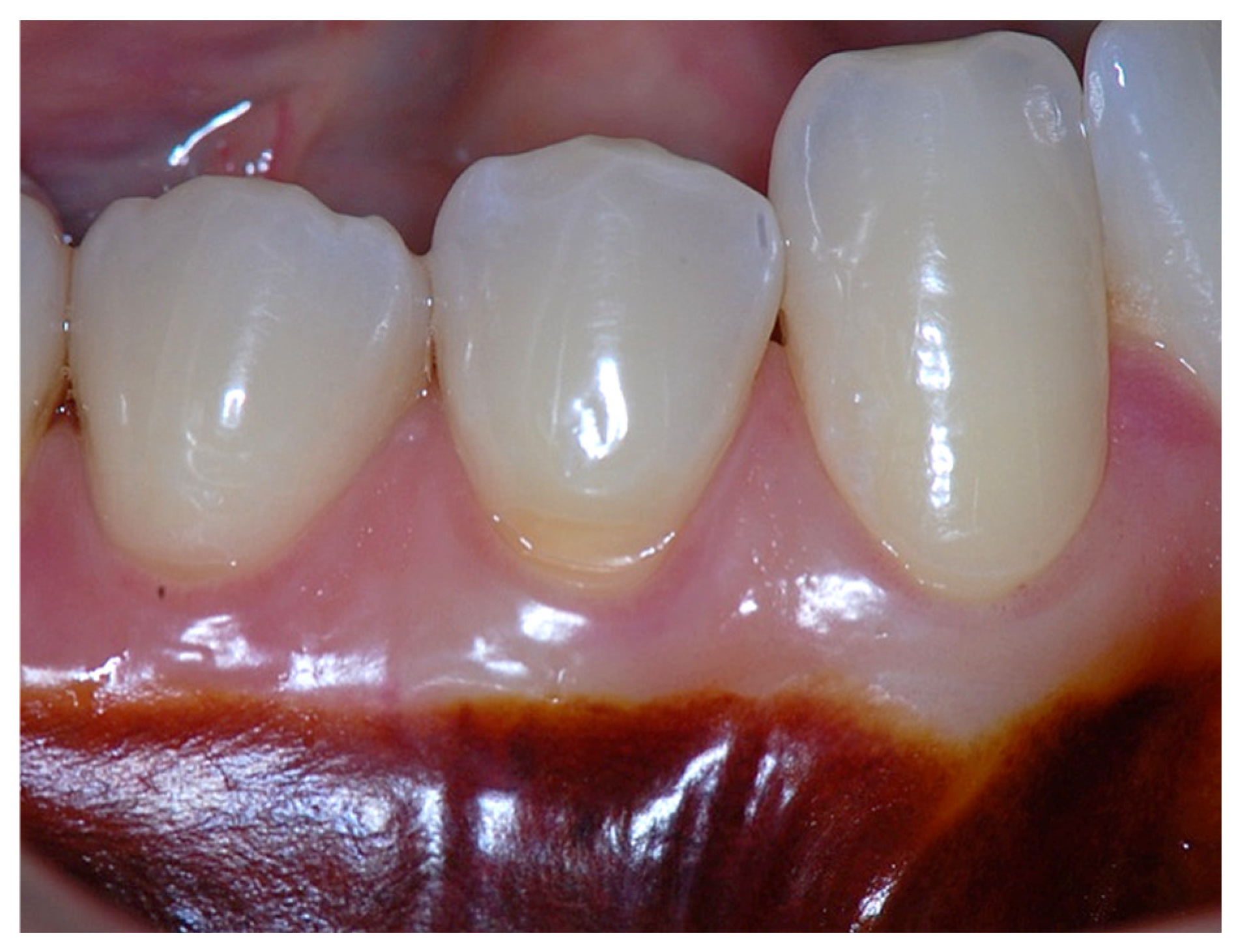
Figure 6.
Surgical establishment of a new 4 mm zone of attached gingiva.
Figure 6.
Surgical establishment of a new 4 mm zone of attached gingiva.
Therefore, while 2 mm of KT may be sufficient for the maintenance of periodontal health in patients with optimal oral hygiene, this width may not be sufficient to stabilize the gingival margin against external mechanical trauma [
13,
14,
28].
To achieve predictable resistance to mechanical trauma, at least 3 mm of KT is recommended, assuming a sulcus depth of 1 mm, allowing 1 mm of epithelial attachment and 1 mm of dense connective tissue attachment to root [
18].
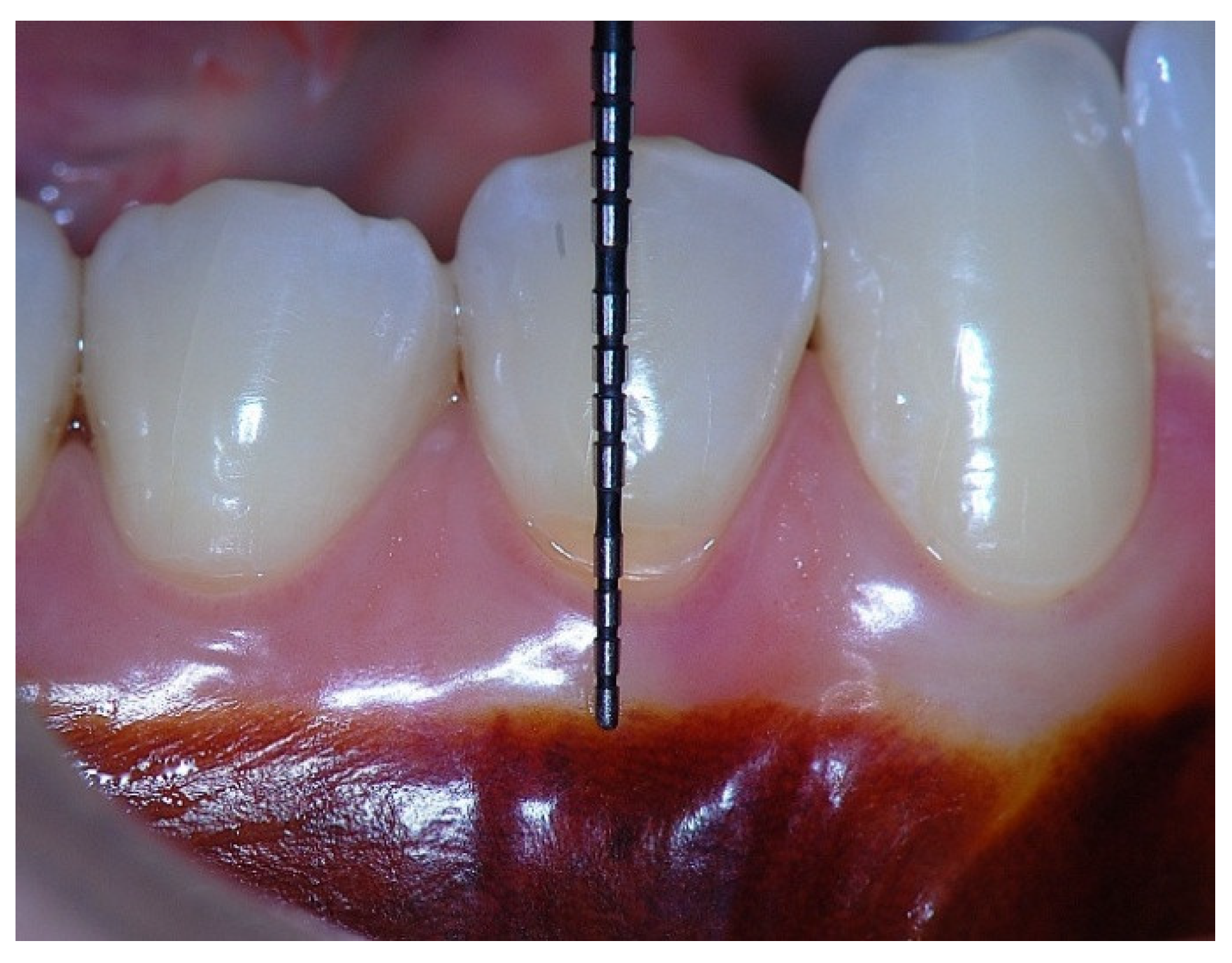
However, the ideal clinical scenario involves ≥3 mm of KT, as this supports connective tissue fiber insertion into both the root surface and the periosteum of the alveolar bone. (
Figure 7,
Figure 8,
Figure 9,
Figure 10 and
Figure 11) Notably, a KT width ≥3 mm is the only condition that fully aligns with all descriptions of AG in the periodontal literature and with the AAP criteria, in which the AG must be firmly attached to the underlying alveolar bone and root cementum by dense connective tissue fibers [
1,
29,
30,
31]
2. Material and Methods
A narrative literature review was conducted to summarize and critically analyze the existing knowledge regarding the morphology, biological concepts, and clinical relevance of the amount of attached gingiva (AG) in natural dentition A comprehensive bibliographic search was performed in the PubMed/MEDLINE, Scopus, and SciELO databases. The search included all articles published from 1961 to the present. Original research papers, clinical studies, and reviews addressing the morphology, histology, function, or clinical significance of the AG were eligible for inclusion. Classical studies that contributed to the establishment of the AG concept were intentionally prioritized due to their foundational relevance.
Articles were screened based on relevance to the following domains:
(1) evolution of the biological and clinical concept of AG;
(2) methods for clinical and histological assessment;
(3) functional and protective roles of AG in periodontal health; and
(4) implications for periodontal stability, susceptibility to mechanical trauma, and treatment outcomes.
The selected articles were analyzed descriptively, focusing on the evolution of the concept, the clinical assessment methods, the functional role of AG, and its implication in periodontal health, protection to periodontal structures and treatment outcomes.
3. Clinical Presentation
Based on current evidence and the biological characteristics of the SCTA entities that comprise the dento-gingival unit the attached gingiva (AG) can be clinically differentiated into the following categories (
Figure 1,
Figure 2 and
Figure 3): [
17].
1 – Attached Gingiva Associated Primarily with the Epithelial Attachment.
In this situation, most of the AG is composed of junctional epithelium (JE), with only a limited number of connective tissue fibers extending into the coronal aspect of the keratinized papilla. The majority t of the connective tissue and supracrestal bone fibers extend to insert into the alveolar mucosa.
2 – Attached Gingiva Composed of Epithelial Attachment and Connective Tissue Fibers Inserted into Root Cementum
In this condition, the connective tissue fibers extend into the keratinized tissue (KT). The attached gingiva (AG) comprises both the epithelial attachment and the insertion of connective tissue fibers into the root surface. However, the supracrestal bone fibers continue to insert into the alveolar mucosa.
3 – Attached Gingiva Corresponding to Epithelial Attachment and Connective Tissue Fibers Inserted into Both Root Cementum and Alveolar Bone
This category represents the ideal anatomical and functional scenario, in which connective tissue fibers and supracrestal bone fibers extend into the KT. This configuration provides maximal resistance to mechanical forces and optimal stabilization of the gingival margin.
4. Discussion
The anatomy-related clinical situations of the attached gingiva (AG) presented in this review are based on the dimensional characteristics of the supracrestal tissue attachment (SCTA), which comprises both the junctional epithelium (JE) and the connective tissue attachment. These anatomical parameters have long served as the primary reference for perio-restorative decision-making [
13,
17,
18,
32,
33,
34].
Additionally, the direction and insertion pattern of the supracrestal connective tissue fibers relative to the keratinized tissue (KT) play a critical role in determining gingival margin stability, provided that the anatomical landmarks remain within normal limits (i.e., 1 mm of epithelial attachment and 1 mm of probing depth). (
Figure 8,
Figure 9,
Figure 10,
Figure 11 and Figure 12)
It is well established that the supracrestal connective tissue fibers of the dento-gingival unit must extend into the dense connective tissue covered by keratinized epithelium on the outer surface of the gingiva. This extension is essential for achieving the protective and resistant functions required to stabilize the gingival margin [
35].
However, a lack of agreement has persisted among clinicians regarding the appropriate apico-coronal dimension of the AG [
22,
23]. One contributing factor is the long-standing misinterpretation of the classic findings by Lang and Löe, whose data were taken to suggest that a minimum of 2 mm of keratinized tissue was required to maintain periodontal health in individuals with suboptimal plaque control [
13]. In their study, 80% of sites with ≥2 mm of KT remained healthy, whereas sites with <2 mm exhibited clinical signs of inflammation, leading to the conclusion that 2 mm of KT is necessary for periodontal stability.
Conversely, Maynard and Wilson proposed that when restorative margins are placed within the gingival sulcus, a minimum of 5 mm of KT, of which at least 3 mm should be attached is required [
10]. Their rationale was that the AG provides a protective barrier not only against bacterial inflammation but also against traumatic injuries associated with restorative procedures [
11].
These different opinions reflect the coexistence of two distinct biological concepts: (1) the role of keratinized tissue in modulating susceptibility to bacterial-induced inflammation, and (2) its capacity to enhance the mechanical resistance of soft tissues to trauma resulting from excessive or aggressive toothbrushing or dental interventions (trauma-induced inflammation). While 2 mm of KT may be sufficient to maintain periodontal health, such dimension may be inadequate when mechanical trauma is taken into consideration [
10,
12,
13,
14,
21,
36].
This interpretation is closely related to the biological nature of the soft-tissue attachment. A width of 2 mm of KT typically corresponds to a predominance of epithelial attachment, which, although functional, may not provide sufficient protection to the gingival margin when subjected to traumatic forces. In contrast, when 3 mm of KT is present, connective tissue fibers attached to the root cementum offer the first effective protective barrier for maintaining the gingival margin stability. The additional millimeter of KT is also likely to engage the alveolar crest incorporating both the connective tissue attachment and the underlying bone within the protective complex [
1,
19]. This situation is considered the most desirable protection for the gingival margin and offers superior resistance against the development or progression of gingival recession.
Schroeder described the gingiva as a “collar of masticatory mucosa” forming an attachment to the tooth, alveolar crest, interdental septa, and coronal alveolar process [
24]. However, such protective function, may not be consistently effective when only 2 mm of KT is present.
Despite ongoing debate regarding the minimal width of AG required for periodontal stability, most clinicians generally agree that a wider band facilitates oral hygiene, preserves clinical attachment, and reduces the risk of gingival recession. Evidence also suggests that in patients with inadequate plaque control, wider KT/AG reduces the likelihood of inflammation and less attachment loss [
9]. As a result, surgical augmentation of AG remains a widely used and predictable procedure [
37].
Finally, an insufficient width of AG, together with the histological characteristics of the epithelial and connective tissue attachments, may constitute a contributing risk factor for the initiation and progression of gingival recession, particularly in the presence of mechanical trauma.
5. Conclusions
This review provides a rational and structured description of the various possible relationships between the attached gingiva (AG) and the underlying tissues, based on the most relevant studies that established the widely accepted concepts in dentistry and those consistently observed in clinical practice. It also highlights that one of the main reasons for the existing controversies regarding the amount and necessity of AG lies in the type of clinical attachment being evaluated. A 1-mm width of AG may, in fact, represent only the attachment provided by the junctional epithelium. Proper and atraumatic toothbrushing may help maintain a stable gingival margin under such conditions. Although this stability may be preserved for some time, the evidence consistently supports that the presence of AG is preferable to its absence.
The information summarized in this review offers a practical and reliable framework for clinicians to evaluate the quality and dimensions of the AG and to determine, when indicated, whether surgical augmentation procedures are warranted to ensure long-term periodontal health and stability.
6. Clinical Relevance
Understanding the anatomical variations and biological characteristics of the attached gingiva is essential for accurate clinical evaluation and treatment planning. Recognizing the limits of functional stability in areas with reduced AG allow clinicians to make informed decisions regarding the need for surgical augmentation, thereby contributing to the long-term preservation of periodontal health, stability, and esthetics.
7. Future Perspectives
Future investigations employing standardized clinical methodologies and well-defined histological criteria are needed to further elucidate the minimal width and biological requirements of the AG required to sustain periodontal health. Such studies may provide deeper insights into the interactions among the junctional epithelium, connective tissue attachment, and keratinized mucosa. A more comprehensive understanding of these relationships will contribute refine clinical parameters for diagnosing mucogingival deficiencies and will support more evidence-based decision-making regarding the need for and indications of surgical augmentation procedures.
Author Contributions Statement
Joao Carnio: Conceptualization. Investigation. Visualization; Joao Kreling Carnio: Writing – Original Draft Preparation; Paulo M Camargo: Writing – Review & Editing.
References
- Glossary of Periodontal Terms 2001, 4th edition, American Academy of Periodontology.
- Cho, MI. Garant PR. Development and general structure of the periodontium. Periodontology 2000;24:9-27.
- Newman MG, Takei HH, Klokkevold PR, Carranza FA. Carranza’s Clinical Periodontology, 11th Edition, Elsevier Saunders;2012:9-57.
- Bowers, G.M. A Study of the Width of Attached Gingiva. J. Periodontol. 1963, 34, 201–209. [Google Scholar] [CrossRef]
- Carnio, J.; Camargo, P.M.; Passanezi, E. Increasing the Apico-Coronal Dimension of Attached Gingiva Using the Modified Apically Repositioned Flap Technique: A Case Series With a 6-Month Follow-Up. J. Periodontol. 2007, 78, 1825–1830. [Google Scholar] [CrossRef]
- Wennstrom J, Lindhe J, Nyman S. Role of keratinized gingiva for gingival health. Clinical and histologic study of normal and regenerated gingival tissue in dogs. J Clin Periodontol 1981;8:311-328.
- Ericsson I, Lindhe J. Recession in sites with inadequate width of keratinized gingiva. J Clin Periodontol 1984;11:95-103.
- Wennström, J.L. Mucogingival Therapy. Ann. Periodontol. 1996, 1, 671–701. [Google Scholar] [CrossRef]
- Pini Prato, GP. Mucogingival deformities. Ann Periodontol 1999;4;98-101.
- Maynard JG, Jr. , Wilson RDK. Physiologic dimension of the periodontium significant to the restorative dentist. J Periodontol 1979;50:170-174.
- Stetler, K.J.; Bissada, N.F. Significance of the Width of Keratinized Gingiva on the Periodontal Status of Teeth with Submarginal Restorations. J. Periodontol. 1987, 58, 696–700. [Google Scholar] [CrossRef]
- Nevins, M. Attached gingiva--mucogingival therapy and restorative dentistry. . 1986, 6, 9–27. [Google Scholar]
- Lang, N.P.; Löe, H. The Relationship Between the Width of Keratinized Gingiva and Gingival Health. J. Periodontol. 1972, 43, 623–627. [Google Scholar] [CrossRef]
- Scheyer, E.T.; Sanz, M.; Dibart, S.; Greenwell, H.; John, V.; Kim, D.M.; Langer, L.; Neiva, R.; Rasperini, G. Periodontal Soft Tissue Non–Root Coverage Procedures: A Consensus Report From the AAP Regeneration Workshop. J. Periodontol. 2015, 86, S73–S76. [Google Scholar] [CrossRef] [PubMed]
- Wennström, JL. Lack of association between width of attached gingiva and development of soft tissue recession: A 5-year longitudinal study. J Clin Periodontol 1987;14:181-184.
- Freedman, AL. The clinical significance of the width of keratinized gingiva. J Periodontol. 1977;48:595–596.
- Schmidt, J.C.; Sahrmann, P.; Weiger, R.; Schmidlin, P.R.; Walter, C. Biologic width dimensions – a systematic review. J. Clin. Periodontol. 2013, 40, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Gargiulo, A.W.; Wentz, F.M.; Orban, B. Dimensions and Relations of the Dentogingival Junction in Humans. J. Periodontol. 1961, 32, 261–267. [Google Scholar] [CrossRef]
- Passanezi E, Sant’Ana ACP, Rezende MLR, Greghi SLA, Janson WA. Distancias biologicas periodontais: principios para a reconstrucao periodontal, estetica e protetica. 1o Edicao, Artes Medicas, 2011.
- Magnusson, I.; Runstad, L.; Nyman, S.; Lindhe, J. A long junctional epithelium - A locus minoris resistentiae in plaque infection? J. Clin. Periodontol. 1983, 10, 333–340. [Google Scholar] [CrossRef]
- Wennström, J.; Lindhe, J. Plaque-induced gingival inflammation in the absence of attached gingiva in dogs. J. Clin. Periodontol. 1983, 10, 266–276. [Google Scholar] [CrossRef]
- Miyasato, M.; Crigger, M.; Egelberg, J. Gingival condition in areas of minimal and appreciable width of keratinized gingival. J. Clin. Periodontol. 1977, 4, 200–209. [Google Scholar] [CrossRef]
- Kennedy, J.E.; Bird, W.C.; Palcanis, K.G.; Dorfman, H.S. A longitudinal evaluation of varying widths of attached gingiva. J. Clin. Periodontol. 1985, 12, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, H.E.; Listgarten, M.A. The gingival tissues: the architecture of periodontal protection. Periodontology 2000 1997, 13, 91–120. [Google Scholar] [CrossRef] [PubMed]
- E Schroeder, H.; A Listgarten, M. Fine structure of the developing epithelial attachment of human teeth. . 1971, 2, 1–134. [Google Scholar]
- Waerhaug, J. Healing of the dento-epithelial junction following the use of dental floss. J. Clin. Periodontol. 1981, 8, 144–150. [Google Scholar] [CrossRef]
- Armitage, G.C. Manual periodontal probing in supportive periodontal treatment. Periodontology 2000 1996, 12, 33–39. [Google Scholar] [CrossRef]
- Phatale, S.; Marawar, P.; Byakod, G.; Lagdive, S.B.; Kalburge, J.V. Effect of retraction materials on gingival health: A histopathological study. J. Indian Soc. Periodontol. 2010, 14, 35–9. [Google Scholar] [CrossRef]
- Schroeder, HE. The periodontium. Handbook of microscopic anatomy. Vol V/5. Berlin:Springer, 1986.
- Lindhe J, Lang NP, Karring T. Clinical Periodontology and Implant Dentistry. 6th Edition, Oxford, Wiley Blackwell, 2015.
- Newman MG, Takei HH, Klokkevold PR, Carranza FA. Carranza’s Clinical Periodontology. 12th Edition, St.Louis, Elsevier Saunders, 2015.
- Jepsen, S.; Caton, J.G.; Albandar, J.M.; Bissada, N.F.; Bouchard, P.; Cortellini, P.; Demirel, K.; de Sanctis, M.; Ercoli, C.; Fan, J.; et al. Periodontal manifestations of systemic diseases and developmental and acquired conditions: Consensus report of workgroup 3 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Periodontol. 2018, 89, S237–S248. [Google Scholar] [CrossRef]
- Hempton TJ, Dominici JT. Contemporary crown-lengthening therapy: a review. J Am Dent Assoc 2010;141:647-655.
- Marzadori, M.; Stefanini, M.; Sangiorgi, M.; Mounssif, I.; Monaco, C.; Zucchelli, G. Crown lengthening and restorative procedures in the esthetic zone. Periodontology 2000 2018, 77, 84–92. [Google Scholar] [CrossRef]
- Hassel, TM. Tissues and cells of the periodontium. Periodontol 2000 1993;3:9-28.
- Proceedings of the 1996 Word Workshop in Periodontics. Ann Periodontol 1996;1:1-947.
- Agudio, G.; Nieri, M.; Rotundo, R.; Cortellini, P.; Prato, G.P. Free Gingival Grafts to Increase Keratinized Tissue: A Retrospective Long-Term Evaluation (10 to 25 years) of Outcomes. J. Periodontol. 2008, 79, 587–594. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).