Submitted:
13 November 2025
Posted:
14 November 2025
You are already at the latest version
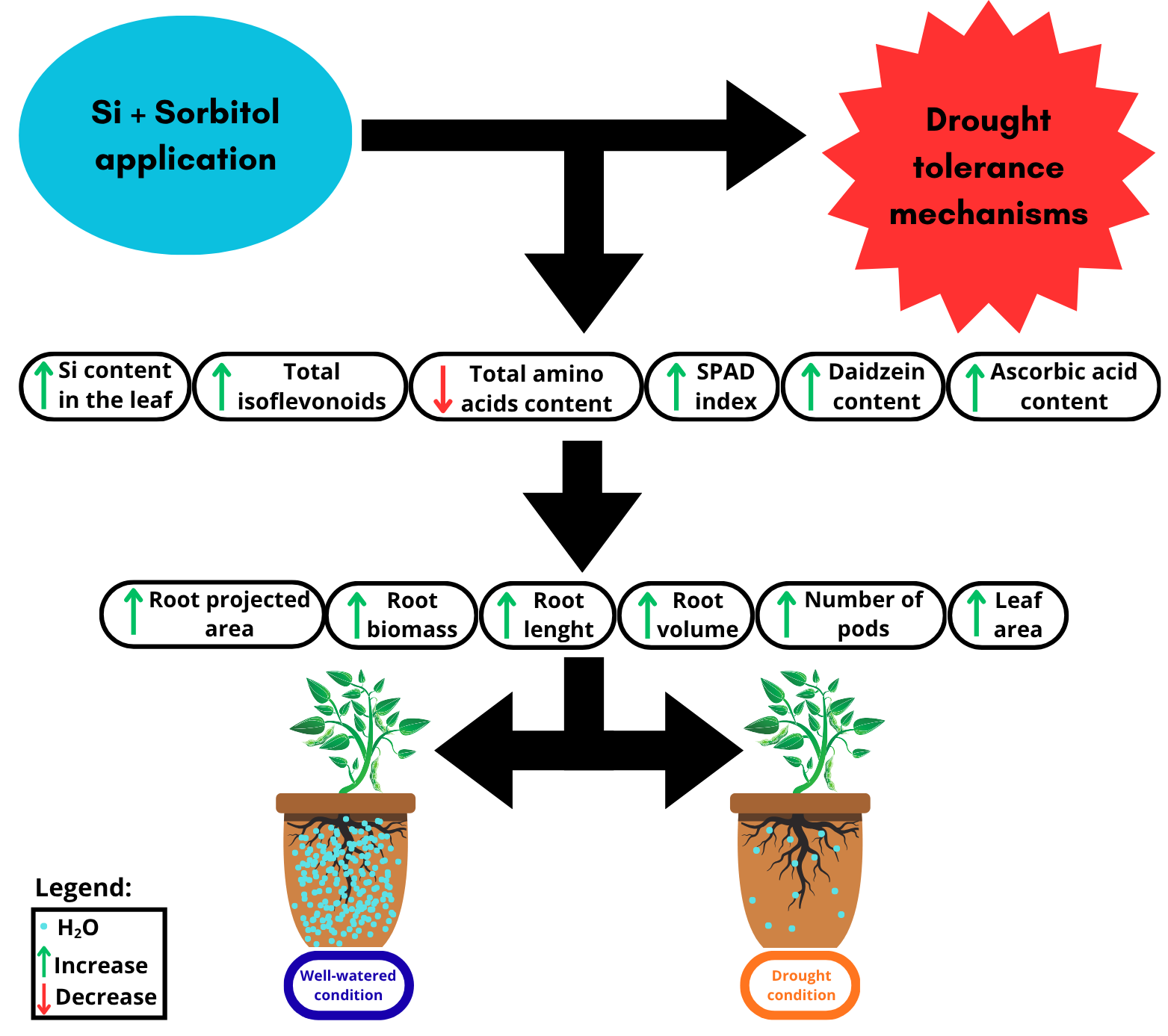
Abstract
Keywords:
1. Introduction
2. Results
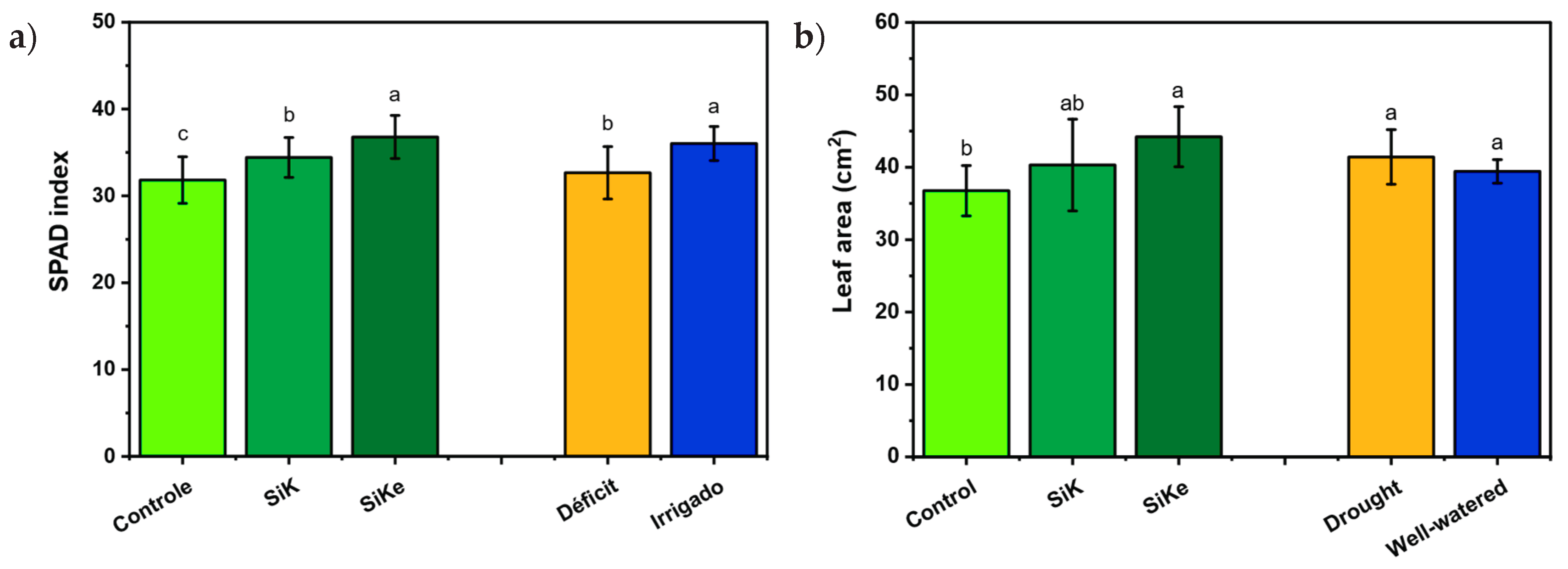
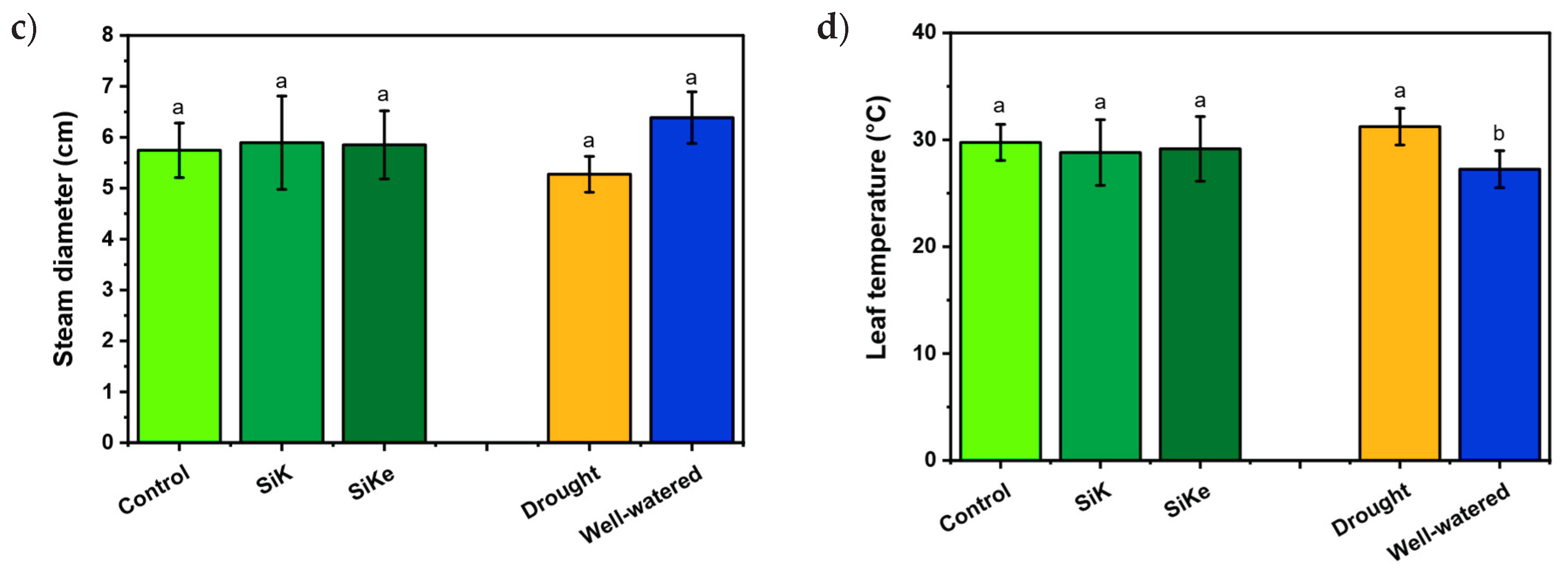
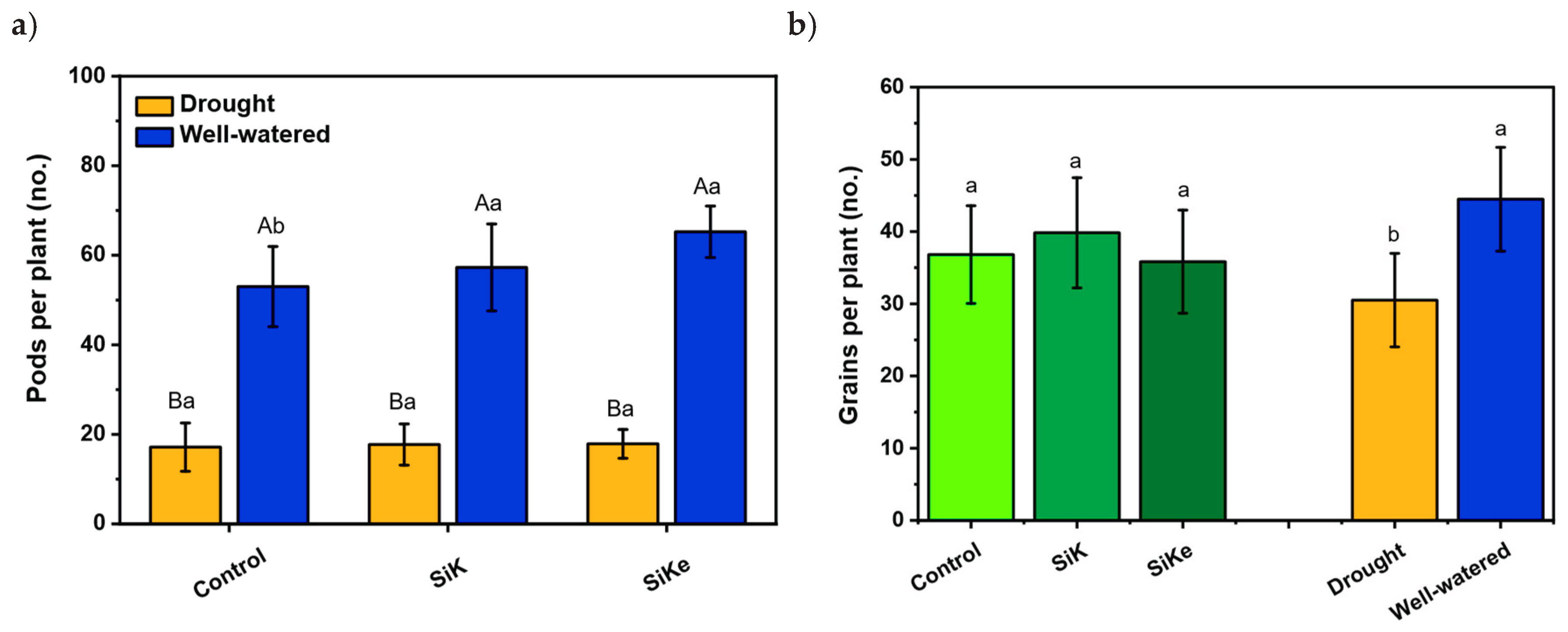
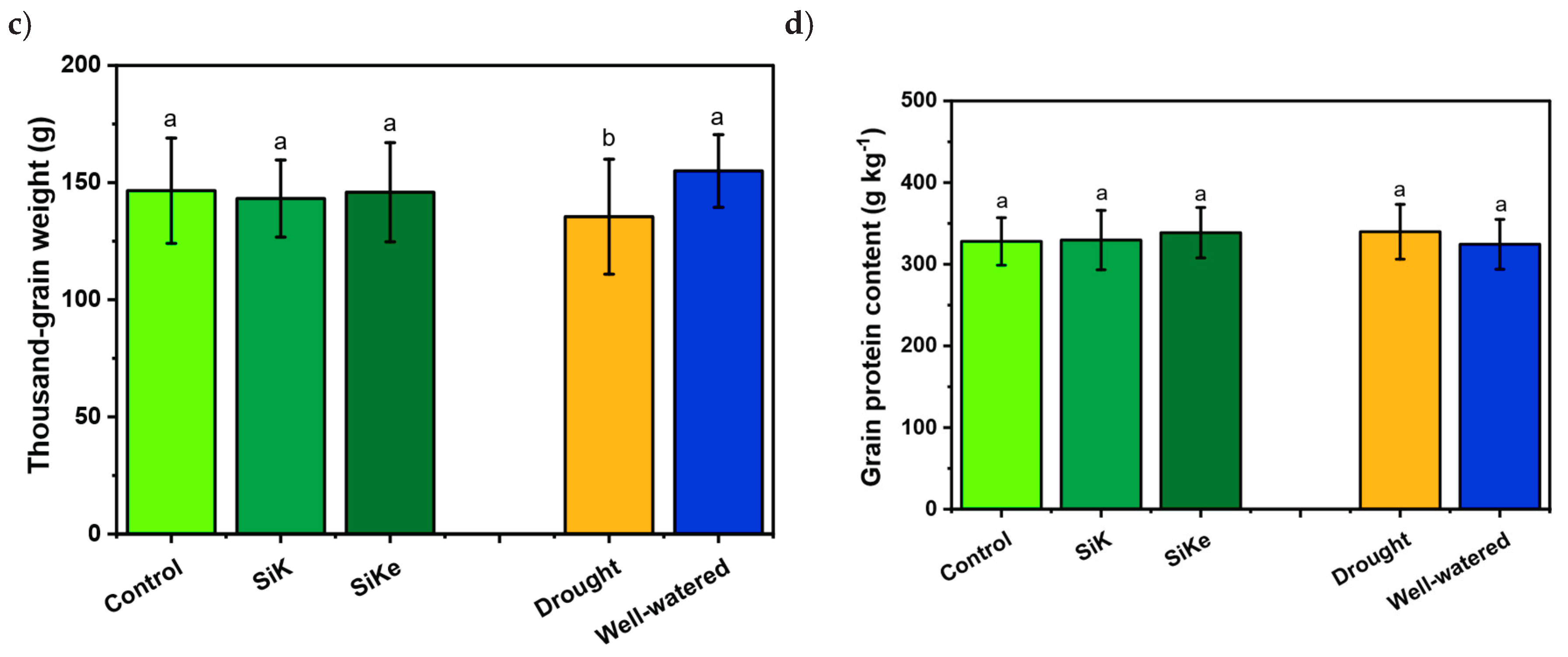
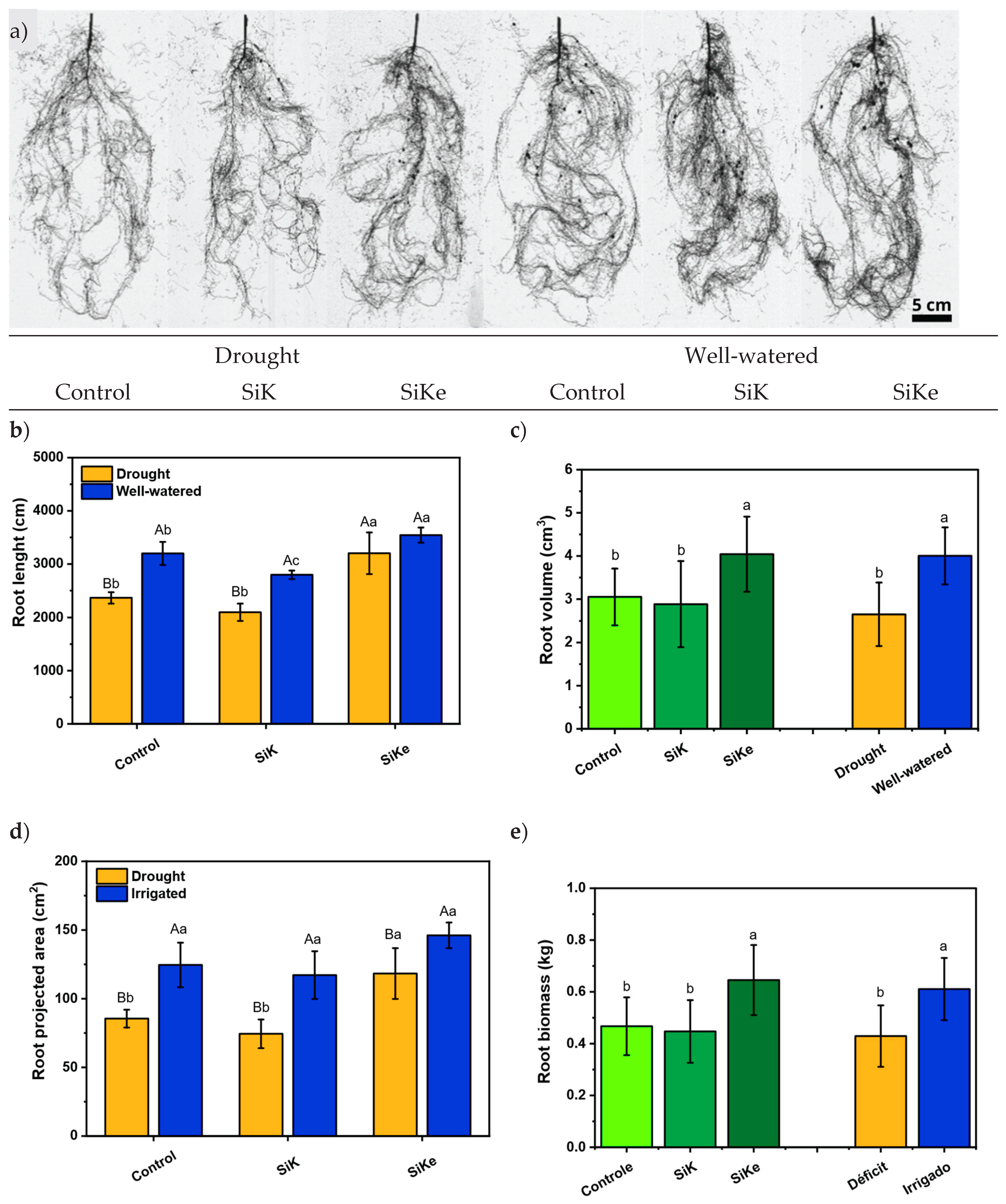
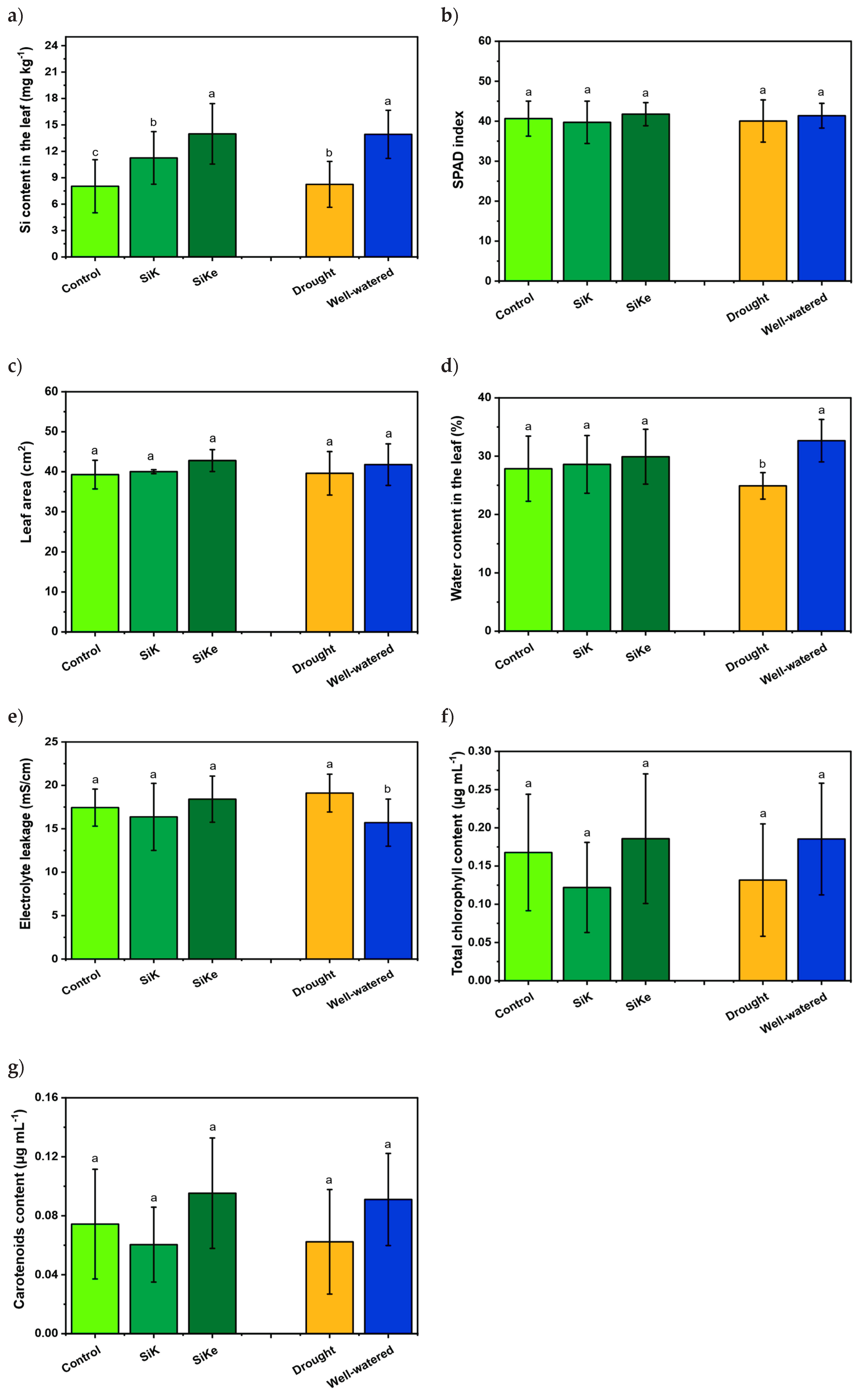
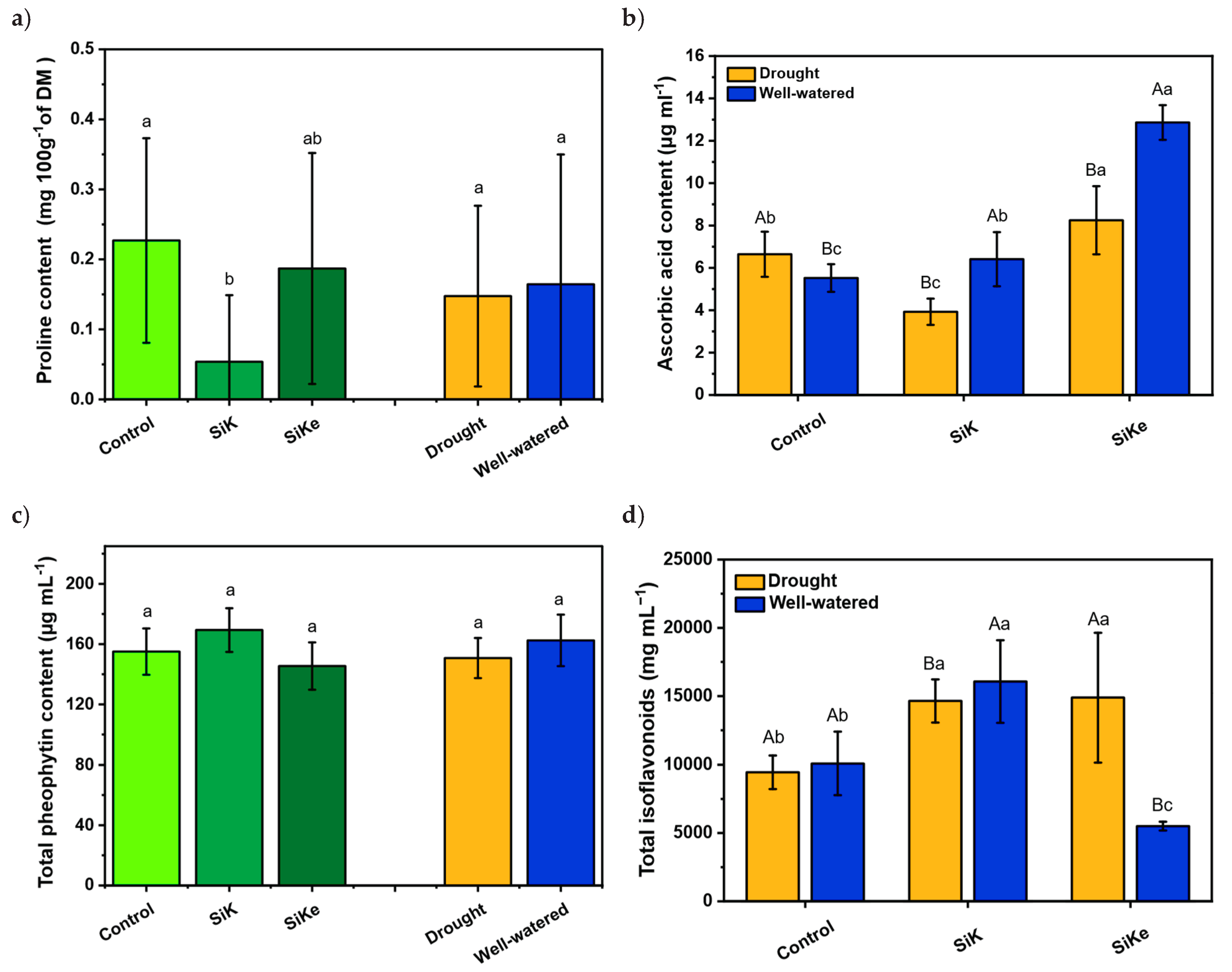
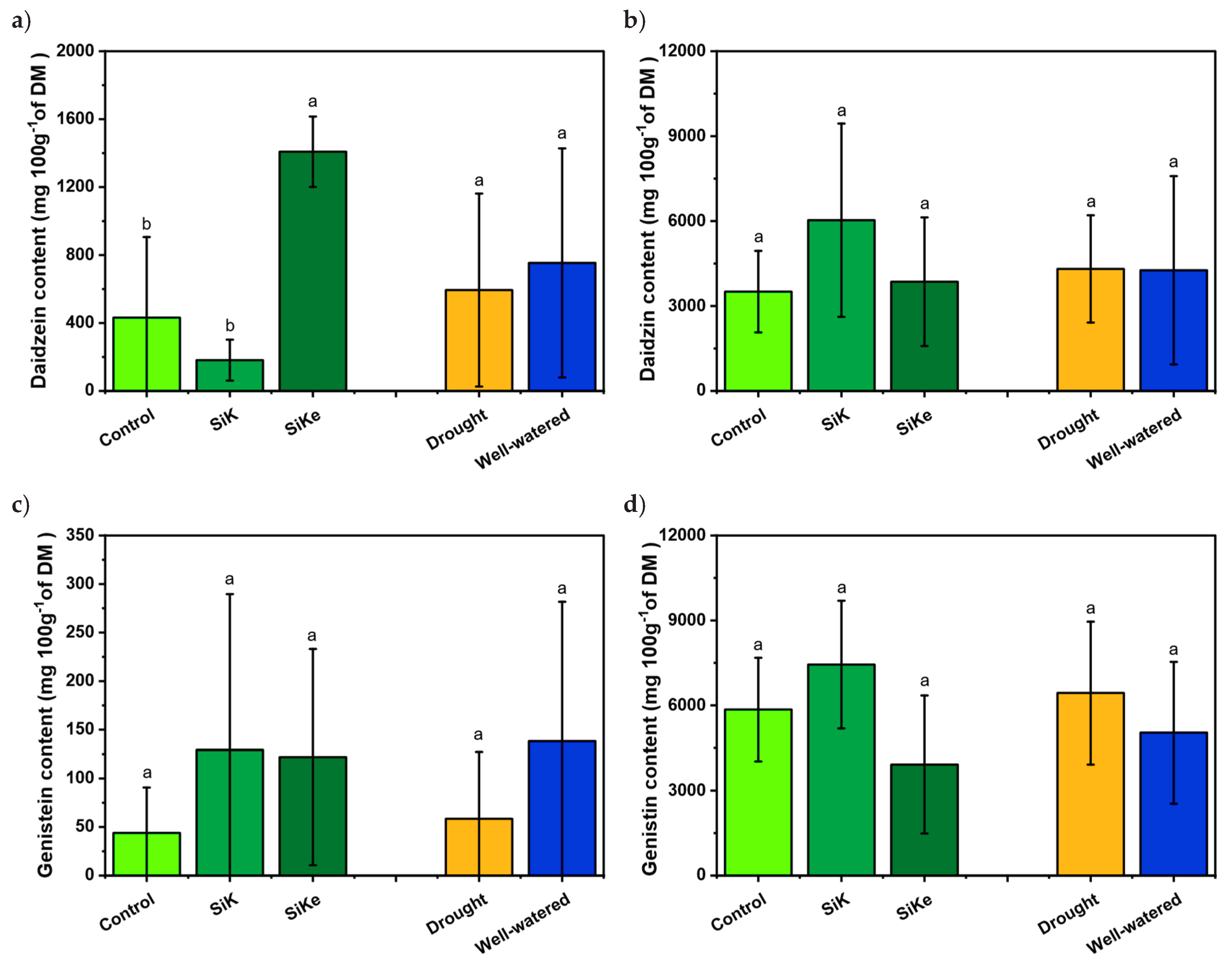
2.1. Experiment I
2.2. Experiment II
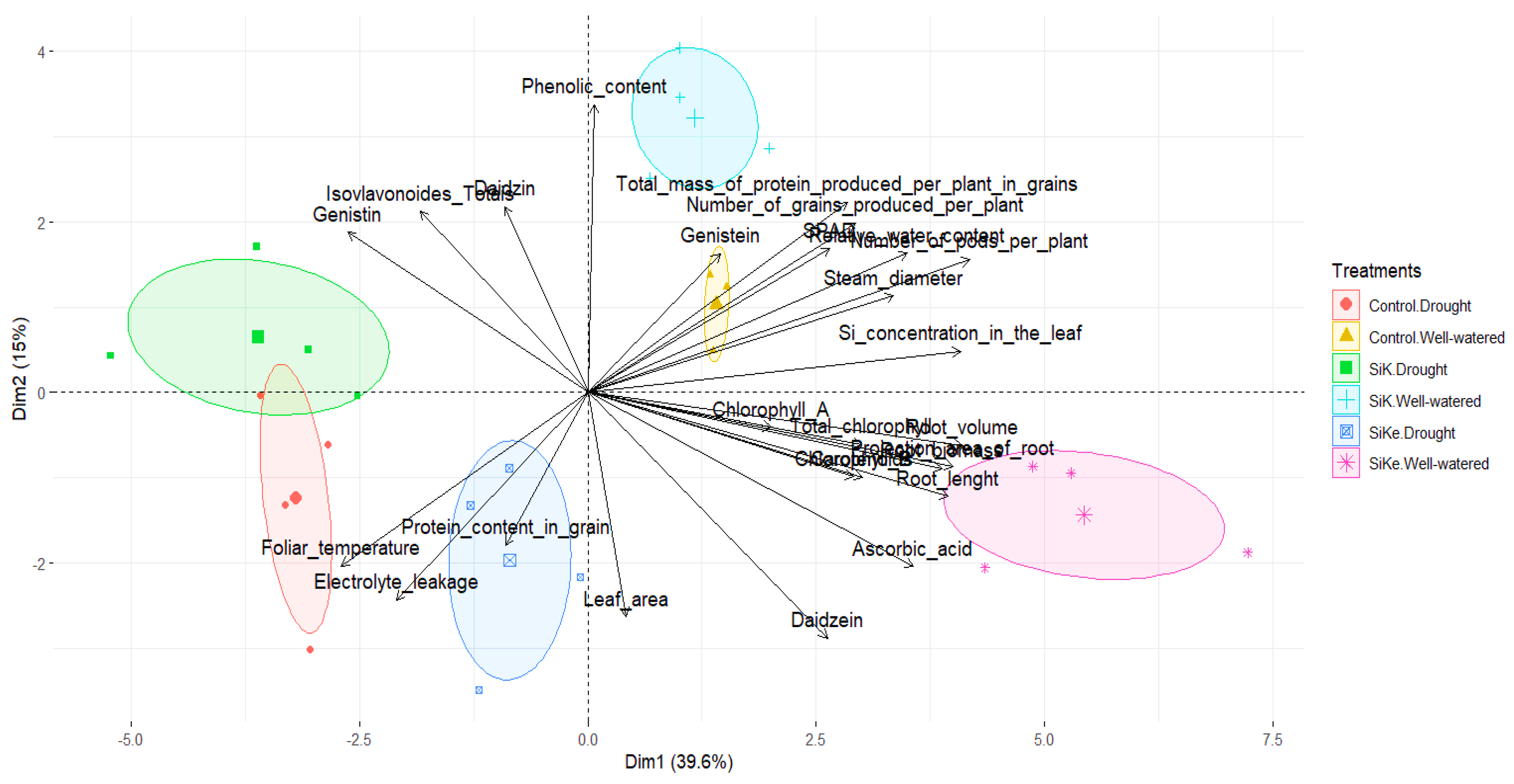
2.3. Principal Components Analysis (PCA)
2.4. Heatmap of Amino Acids Content in the Leaf
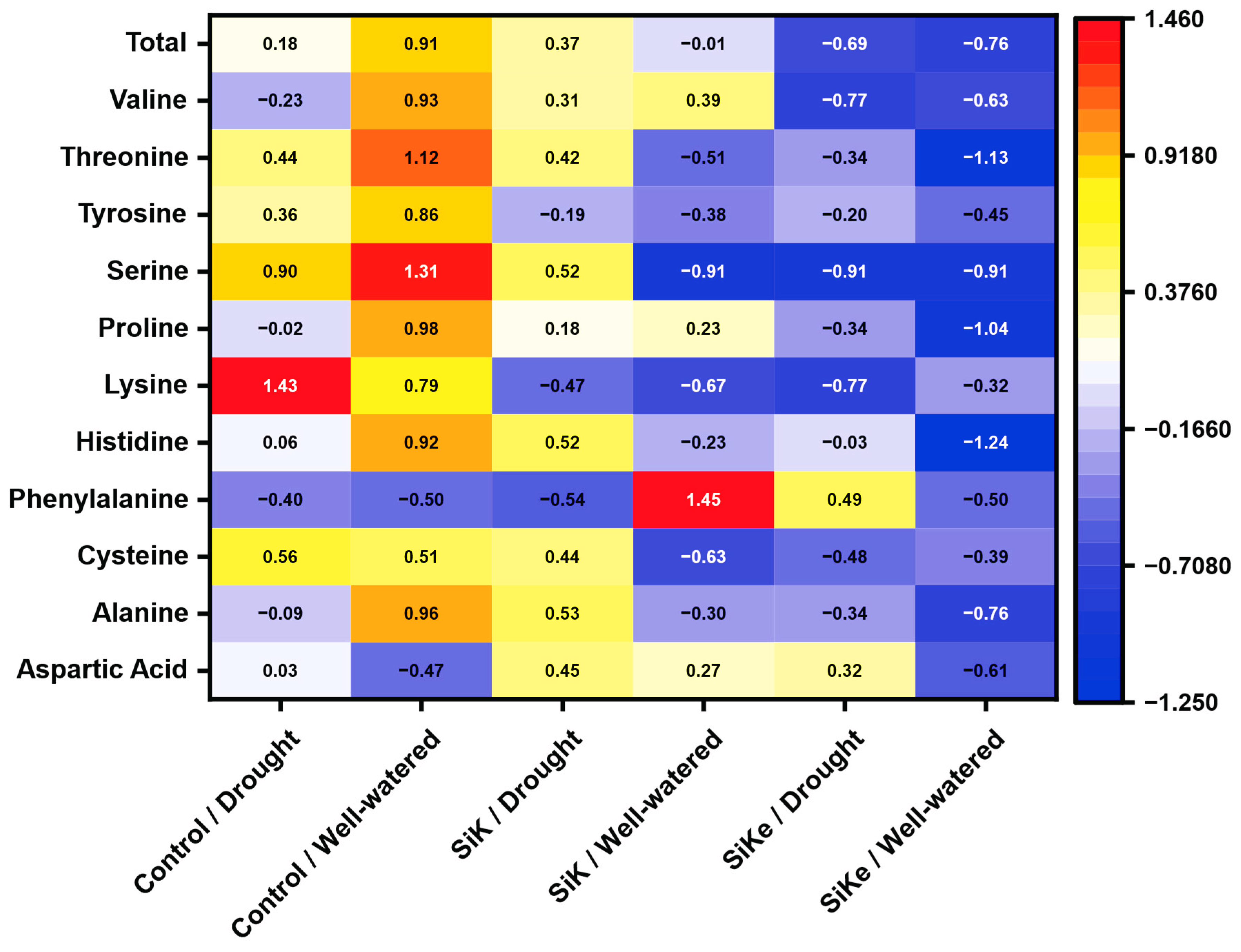
3. Discussion
4. Materials and Methods
4.1. Pot Experiment and Location
4.2. Experiment Design
4.3. Experiment I Measurements
4.4. Experiment II Measurements
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| SiK | Potassium silicate (K₂SiO₃) |
| SiKe | Potassium silicate (K₂SiO₃) + Sorbitol (C6H14O6) |
| ROS | Reactive oxygen species |
| RSN | Reactive nitrogen species |
| ¹O₂ | Singleton Oxygen |
| O₂⁻ | Superoxide |
| Vn | Soybean’s vegetative stages |
| Rn | Soybean’s reproductive stages |
| SPAD | Soil-Plant Analysis Development |
| EL | Electrolyte leakage |
| Ci | Initial conductivity |
| Cf | Final conductivity |
| DM | Dry matter |
| UV-B | Ultraviolet-B radiation |
| PCA | Principal component analysis |
References
- Marengo J.A., Schaeffer R., Zee D., Pinto H.S. (2009). Mudanças climáticas e eventos extremos no Brasil. Rio de Janeiro: FBDS, 2009. 76 p.
- IPCC (2021). Summary for Policymakers: Climate change 2021 the physical science basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change.
- Companhia Nacional de Abastecimento. (2025). Acompanhamento da safra brasileira de grãos: Safra 2024/2025 – 2º levantamento, outubro 2025. Brasília, DF: Conab. Recuperado de https://www.conab.gov.br/info-agro/safras/graos/boletim-da-safra-de-graos.
- Basso, M. F., Neves, M. F., & Grossi-de-Sa, M. F. (2024). Agriculture evolution, sustainability and trends, focusing on Brazilian agribusiness: A review. Frontiers in Sustainable Food Systems, 7, 1296337. [CrossRef]
- Ferreira, R. C., Sibaldelli, R. N. R., Crusiol, L. G. T., Neumaier, N., & Farias, J. R. B. (2024). Soybean Yield Losses Related to Drought Events in Brazil: Spatial–Temporal Trends over Five Decades and Management Strategies. Agriculture, 14(12), 2144. [CrossRef]
- Khan I., Awan S.A., Ikram R., Rizwan M., Akhtar N., Yasmin H., Sayyed R.Z., Ali S., Ilyas N. (2021). Effects of epibrassinolide on plant growth, antioxidants defense system, and endogenous hormones in two wheat varieties under drought stress. Physiologia Plantarum 172:696–7062. [CrossRef]
- Khan A., Bilal S., Khan A.L., Imran M., Al-Harrasi A., Al-Rawahi A., Lee I-J (2020). Silicon mediated alleviation of combined salinity and cadmium stress in date palm (Phoenix dactylifera L.) by regulating physio-hormonal alteration. Ecotoxicology and Environmental Safety 188:109885. [CrossRef]
- Bhardwaj S., Sharma D., Singh S., Ramamurthy PC, Verma T, Pujari M, Singh J, Kapoor D, Prasad R (2022). Physiological and molecular insights into the role of silicon in improving plant. Plant and Soil 486:25-43. [CrossRef]
- Sonobe K., Hattori T., An P., Tsuji W., Eneji A.E., Kobayashi S., Kawamura Y., Tanaka K., Inanaga S. (2010).tripa Effect of silicon application on sorghum root responses to water stress. Journal of Plant Nutrition 341:7182. [CrossRef]
- Wang M., Wang R., Mur L.A.J., Ruan J., Shen Q., Guo S. (2021). Functions of silicon in plant drought stress responses. Horticulture Research. [CrossRef]
- Sharma A., Kumar V., Shahzad B., Ramakrishnan M., Singh Sidhu G.P., Bali A.S., Handa N., Kapoor D., Yadav P., Khanna K. (2020). Photosynthetic response of plants under different abiotic stresses: A review. Journal of Plant Growth Regulation 392:509–531. [CrossRef]
- Hussain S., Shuxian Li., Mumtaz M., Shafiq I., Iqbal N., Brestic M., Shoaib M., Sisi Q., Li W., Mei X., Bing C., Zivcak M., Rastogi A., Skalicky M., Hejnak V., Weiguo L., Wenyu Y. (2021) Foliar application of silicon improves stem strength under low light stress by regulating lignin biosynthesis genes in soybean (Glycine max (L.) Merr.). Journal of Hazardous Materials, 401:123-256. [CrossRef]
- Shi Y., Zhang Y., Han W., Feng R., Hu Y., Guo J., Gong H. (2016). Silicon enhances water stress tolerance by improving root hydraulic conductance in Solanum lycopersicum L. Frontiers in Plant Science 7:196. [CrossRef]
- Shen X, Zhou Y, Duan L, Li Z, Egrinya A, Li J (2010) Silicon effects on photosynthesis and antioxidant parameters of soybean seedlings under drought and ultraviolet-B radiation. Journal of Plant Physiology 167:1248-1252. [CrossRef]
- Greger M., Landberg T., Vaculík M. (2018). Silicon influences soil availability and accumulation of mineral nutrients in various plant species. Plants 7:41. [CrossRef]
- Knight C.T., Kinrade S.D. (2001). A primer on the aqueous chemistry of silicon. Studies in plant science. Elsevier 8:57–84. [CrossRef]
- Kapoor D., Bhardwaj S., Landi M., Sharma A., Ramakrishnan M., Sharma A. (2020). The impact of drought in plant metabolism: How to exploit tolerance mechanisms to increase crop production. Applied Science 1016:5692. [CrossRef]
- Lambers H., Chapin F., Pons T. (2008). Plant physiological ecology. (Springer: New York). [CrossRef]
- Laxa M., Liebthal M., Telman W., Chibani K., Dietz K-J (2019). The role of the plant antioxidant system in drought tolerance. Antioxidants 84:94. [CrossRef]
- Cerny, M., Habanová, H., Berka, M., Luklová, M., & Brzobohatý, B. (2018). Hydrogen Peroxide: Its Role in Plant Biology and Crosstalk with Signalling Networks. International Journal of Molecular Sciences, 19(9), 2812. [CrossRef]
- Baxter A, Mittler R., Suzuki N. (2014) ROS as key players in plant stress signaling. Journal of Experimental Botany. [CrossRef]
- Chen D., Wang S., Yin L., Deng X. (2018) How does silicon mediate plant water uptake and loss under water deficiency? Frontiers in Plant Science 9:281. [CrossRef]
- Andrade, A.F. de, Bueno, A.M., de Carvalho, A.d.S. Innovative Soluble Silicon Leaf Source Increase Gas Exchange, Grain Yield and Economic Viability in Common Bean. Silicon 14, 3739 3747 (2022). [CrossRef]
- Souza Júnior, J. P., Prado, R. de M., Diniz, J. F., Guedes, V. H. de F., Silva, J. L. F. da, Roque, C. G., & Alvarez, R. de C. F. (2022). Foliar application of innovative sources of silicon in soybean, cotton, and maize. Journal of Soil Science and Plant Nutrition, 22(6), 5186–5199. [CrossRef]
- Oliveira S., Lemes E.S., Meneghello G.E., Tavares L.C., Barros A.C.A. (2015). Aplicação de silício via solo no rendimento e na qualidade fisiológica de sementes de soja. Semina: Ciências Agrárias 36:3029–3042. [CrossRef]
- Moreira A.R., Fagan E.B., Martins K.V., Souza C.H.E. (2010) Resposta da cultura de soja a aplicação de silício foliar. Bioscience Journal 26:413-423.
- Farhangi-Abriz S., Torabian S. (2018). Nano-silicon alters antioxidant activities of soybean seedlings under salt toxicity. Protoplasma 255:953–962. [CrossRef]
- Sah, S. K., Reddy, K. R., & Li, J. (2022). Silicon Enhances Plant Vegetative Growth and Soil Water Retention of Soybean (Glycine max) Plants under Water-Limiting Conditions. Plants (Basel, Switzerland), 11(13), 1687. [CrossRef]
- Abdullah, M. M., Waraich, E. A., Ahmad, M., Hussain, S., Asghar, H. N., Haider, A., Zulfiqar, U., Ahmad, Z., Soufan, W., Prasad, P. V., & Djalovic, I. (2025). Improving soybean drought tolerance via silicon-induced changes in growth, physiological, biochemical, and root characteristics. Plant signaling & behavior, 20(1), 2465232. [CrossRef]
- Wei, J., Liu, L., Wei, Z., Qin, Q., Bai, Q., Zhao, C., Zhang, S., & Wang, H. (2025). Silicon Nano-Fertilizer-Enhanced Soybean Resilience and Yield Under Drought Stress. Plants, 14(5), 751. [CrossRef]
- Tripathi, P., Tayade, R., Mun, B.-G., Yun, B.-W., & Kim, Y. (2022). Silicon application differentially modulates root morphology and expression of PIN and YUCCA family genes in soybean (Glycine max L.). Frontiers in Plant Science, 13, 842832. [CrossRef]
- La, H.; et al. (2023). Drought stress-responsive abscisic acid and salicylic acid crosstalk with the phenylpropanoid pathway in soybean seeds. Frontiers in Plant Science. [CrossRef]
- Biju, S., Fuentes, S., & Gupta, D. (2023). Novel insights into the mechanism(s) of silicon-induced drought stress tolerance in lentil plants revealed by RNA sequencing analysis. BMC Plant Biology, 23, 498. [CrossRef]
- Trush, K., & Pal'ove-Balang, P. (2023). Biosynthesis and role of isoflavonoids in legumes under different environmental conditions. Plant Stress, 8, 100153. [CrossRef]
- Santos, H. G. dos, Jacomine, P. K. T., Anjos, L. H. C. dos, Oliveira, V. A. de, Lumbreras, J. F., Coelho, M. R., Almeida, J. A. de, Araujo Filho, J. C., Oliveira, J. B. de, & Cunha, T. J. F. (Eds.). (2018). Brazilian Soil Classification System (5th ed., rev. & exp.; Translated by C. E. A. Reichart, L. H. C. dos Anjos, M. C. Alves, H. G. dos Santos, & J. F. Lumbreras). Brasília, DF: Embrapa. ISBN 978-85-7035-821-9.
- Raij, B. van; Andrade, J. C.; Cantarella, H.; Quaggio, J. A. (2001). Chemical Analysis for Soil Fertility Evaluation in Tropical Soils. Campinas: Instituto Agronômico de Campinas. 285 p.
- Datnoff, L. E., Snyder, G. H., & Korndörfer, G. H. (Eds.). (2001). Silicon in agriculture. Studies in Plant Science, 8. Elsevier Science B.V., Amsterdam, The Netherlands. 403 p.
- Fehr, W. R. & Caviness, C. E. Stages of soybean development. (1977).
- Sinclair, T. R. & Ludlow, M. M. (1986). Influence of soil water supply on the plant water balance of four tropical grain legumes. Australian Journal of Plant Physiology, 13(3), 329–341. [CrossRef]
- Gong, H., Zhu, X., Chen, K., Wang, S., & Zhang, C. (2005). Silicon alleviates oxidative damage of wheat plants in pots under drought. Plant Science, 169(2), 313–321. [CrossRef]
- Birchall, J. D. The essentiality of silicon in biology. Chem. Soc. Rev. 24, 351–357 (1995). [CrossRef]
- Adami, M., Hastenreiter, F.A., Flumignan, D.L., Faria, R.T. (2008). Estimativa de área de folíolos de soja usando imagens digitais e dimensões foliares. Bragantia, 67, 1053-1058. [CrossRef]
- AOAC International. (2005). Official methods of analysis of AOAC International (18th ed., Method 988.05 – Protein (Crude)). AOAC International.
- Barrs, H.D. and Weatherley, P.E. (1962) A Re-Examination of the Relative Turgidity Techniques for estimating water deficits in leaves. Australian Journal of Biological Sciences, 15, 413-428. [CrossRef]
- Tarhanen, S. Metsärinne, S., Holopainen, T., Oksanen, J. (1999) Membrane permeability response of lichen Bryoria fuscescens to wet deposited heavy metals and acid rain. Environmental Pollution. 104, 121-129. [CrossRef]
- Lichtenthaler, H. (1987). Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods in Enzymology, 148C, 350-382. [CrossRef]
- Stroehecker R., Henning H.M. (1967). Análises de vitaminas: Métodos comprobados. Madrid: Paz Montalvo.
- Singleton V.L., Rossi J.R. (1965). Colorimetry of total phenolics with phosphomolybdic- acid reagents. American Journal of Enology and Viticulture 16:144-158. [CrossRef]
- Carrão-Panizzi, M.C., Favoni, S.P.G., Kikuchu, A. (2002). Extraction time for isoflavone determination. Brasilian Archives of Biology and Technology, v. 45, p. 515-518. [CrossRef]
- Oliveira, I.C., Santana, D.C., de Oliveira, J.L.G. et al. Flavonoids and their relationship with the physiological quality of seeds from different soybean genotypes. Sci Rep 14, 17008 (2024). [CrossRef]
- Bates, L., Waldren, R. P., Teare, I. D. (1973). Rapid determination of free proline for water-stress studies. Plant and Soil 39:205–207. [CrossRef]
- Seron, A.C.d.S.C.; Santana, D.C.; Oliveira, I.A.; Campos, C.N.S.; Teodoro, L.P.R.; Silva, E.V.M.; Ratke, R.F.; Baio, F.H.R.; da Silva Junior, C.A.; Teodoro, P.E. Relationship Between Hyperspectral Data and Amino Acid Composition in Soybean Genotypes. AgriEngineering 2025, 7, 265. [CrossRef]
- Silva, F. C. da (2009). Manual de análises químicas de solos, plantas e fertilizantes. Embrapa Informação Tecnológica.
- Elliott C.L., Snyder G.H. (1991). Autoclave-induced digestion for the colorimetric determination of silicon in rice straw. Journal of Agricultural Food Chemistry 39:1118-1119. [CrossRef]
- Bouma, T. J., Nielsen, K. L., & Koutstaal, B. (2000). Sample preparation and scanning protocol for computerised analysis of root length and diameter. Plant and Soil, 218(1), 185–196. [CrossRef]
| pH CaCl2 |
O.M. | P(resin) | S | Ca | Mg | K | Al | H+Al | BS | CEC | BS | m |
| g dm⁻³ | --- mg dm⁻³ --- | --------------------------------- mmolc dm⁻³ ------------- | % | % | ||||||||
| 5.71 | 22.9 | 17.6 | 10.2 | 37.6 | 12 | 3.24 | <0.1 | 14.1 | 52.8 | 66.9 | 79 | 0 |
| B | Cu | Fe | Mn | Zn | Na | Si | Sand total | Silt | Clay | Texture class | ||
| --------------------- mg dm⁻³ -------------------------------- | ------------g kg⁻¹--------------- | |||||||||||
| 0.4 | 1.1 | 25.3 | 38.2 | 2.9 | 9 | <9.7 | 751 | 25 | 224 | Medium | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




