Submitted:
01 October 2025
Posted:
01 October 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Field and Reference Samples
2.2. Extraction Chemistries and Equipment
2.3. Polymerase Chain Reactions (PCR)
2.4. Statistical Analysis
3. Results
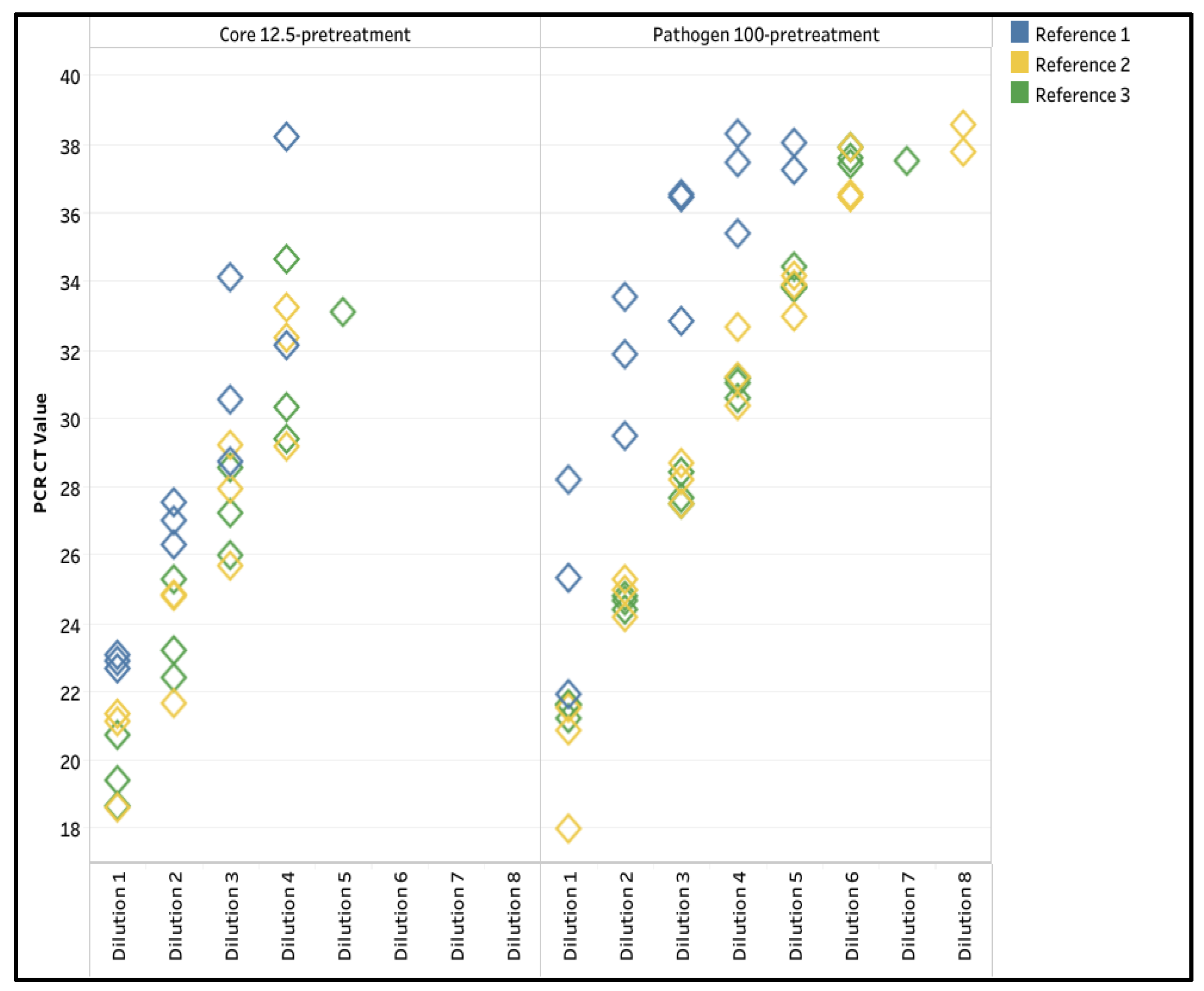
3.1. Extraction Method Optimization
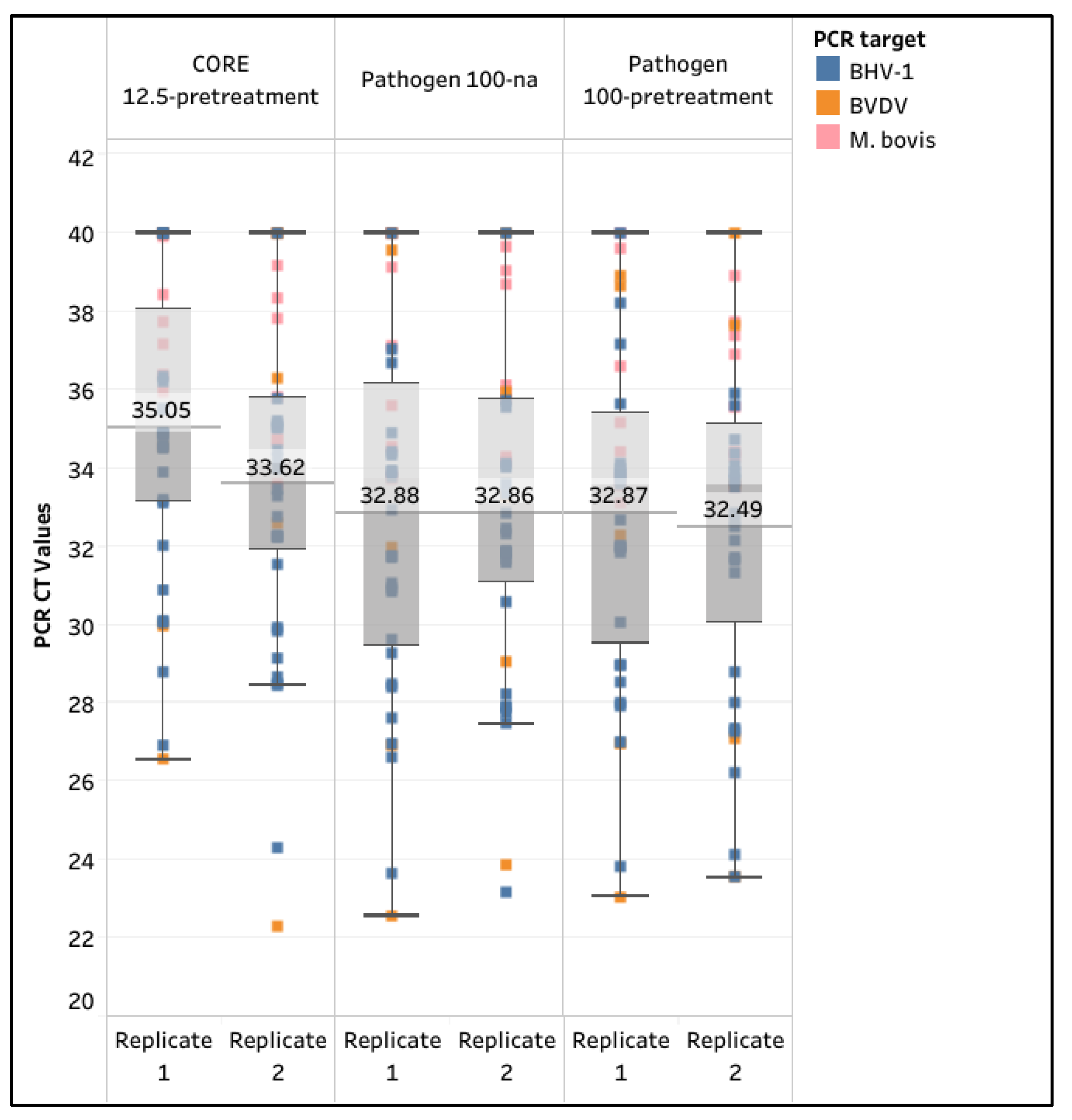
3.2. Evaluation of Diagnostic Sensitivity for the Selected Extraction Protocols
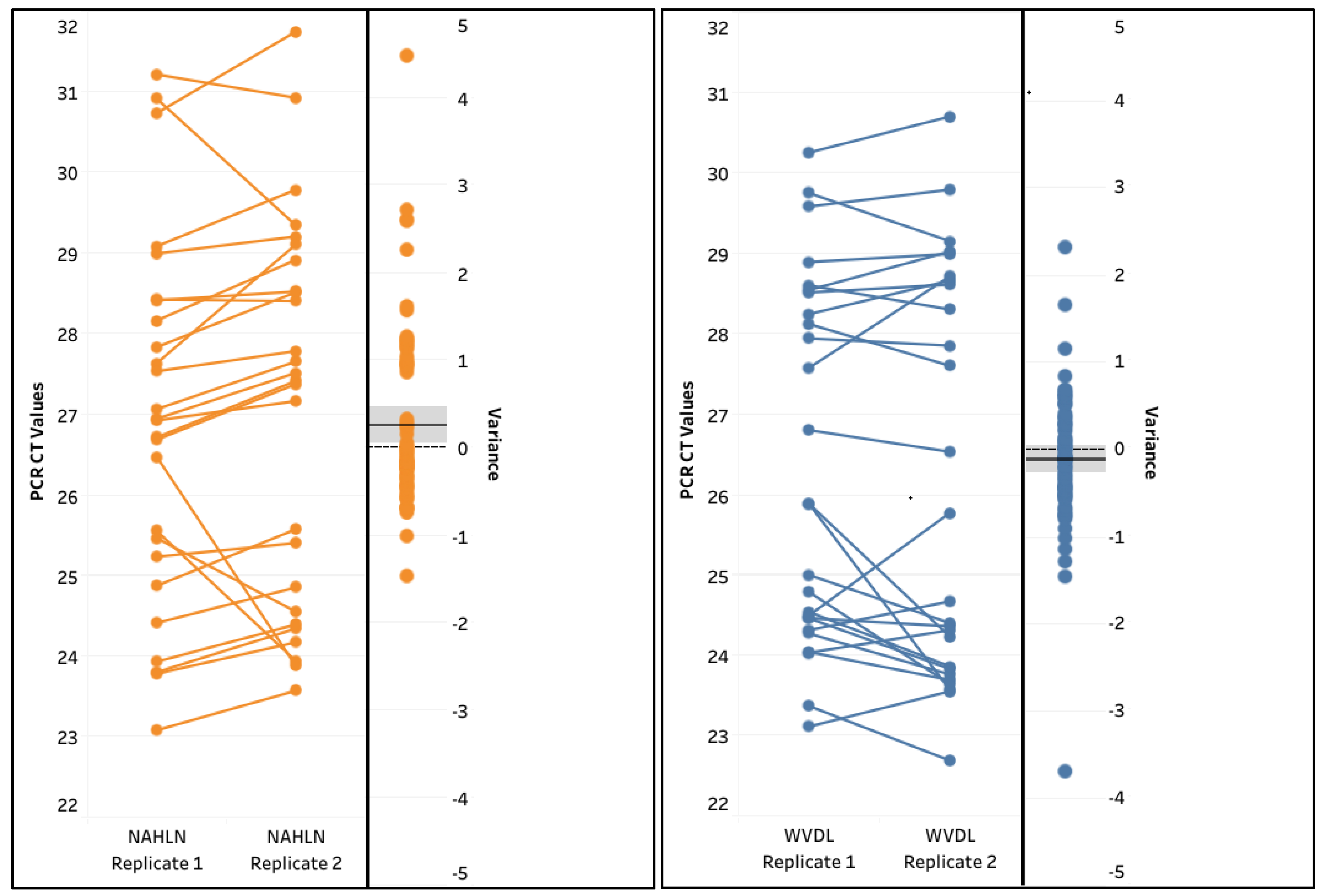
3.3. Evaluation of Performance for IAV Assays
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA BHV-1 BVDV CT |
Analysis of Variance Bovine herpesvirus-1 Bovine viral diarrhea virus Cycle threshold |
| FMDV HPAI |
Foot and mouth disease virus Highly Pathogenic Avian Influenza |
| IAV | Influenza A virus |
| LOD | Limit of detection |
| LPAI | Low Pathogenic Avian Influenza |
| MagMAX CORE | MagMAX CORE Nucleic Acid Purification Kit |
|
M. bovis NAHLN NVSL |
Mycoplasma bovis National Animal Health Laboratory Network National Veterinary Service Laboratories |
| PBS PCR |
Phosphate-buffered saline Polymerase chain reactions |
| R2 | Coefficient of correlation of the standard curve |
| Xeno RNA WVDL |
VetMAX Xeno Internal Positive Control RNA Wisconsin Veterinary Diagnostic Laboratory |
| WVDL IC | WVDL internal control |
Appendix A
| IAV | Subtype | Strain ID |
| IAV reference 1 | H9N2 | Influenza A Virus A/Turkey/CA/6889/1980 |
| IAV reference 2 | H5N9 | Influenza A Virus A/Turkey/Wisconsin/1968 |
| IAV reference 3 | H7N3 | Influenza A Virus A/Turkey/Oregon/1977 |
| LPAI 1 | H3N8 | Influenza A Virus A/Equine/Miami/1/63 |
| LPAI 2 | H1N7 | Influenza A Virus A/NJ/8/76/EQ-1 |
| LPAI 3 | HON3 | Influenza A Virus A/NWS-NOV2 |
| LPAI 4 | H10N7 | Influenza A Virus A/CK/GERM/49 |
| LPAI 5 | H2N3 | Influenza A Virus A/Mallard/A16/77 |
| LPAI 6 | H4N8 | Influenza A Virus A/MYNAH/Mass/71 |
| LPAI 7 | H7N3 | Influenza A Virus A/TY/ORE |
| LPAI 8 | H4N8 | Influenza A Virus A/DK/England/62 |
| LPAI 9 | H3N8 | Influenza A Virus A/DK/Ukraine/1/63 |
References
- Animal and Plant Health Inspection Service, U.S. DEPARTMENT OF AGRICULTURE. H5N1 Influenza. H5N1 Influenza. Available online: https://www.aphis.usda.gov/livestock-poultry-disease/avian/avian-influenza/hpai-detections?utm (accessed 2025-09-27).
- American Veterinary Medical Association. Avian Influenza Virus Type A (H5N1) in U.S. Dairy Cattle. Available online: https://www.avma.org/resources-tools/animal-health-and-welfare/animal-health/avianinfluenza/avian-influenza-virus-type-h5n1-us-dairy-cattle (accessed 2025-09-27).
- Fourment, M.; Darling, A. E.; Holmes, E. C. The Impact of Migratory Flyways on the Spread of Avian Influenza Virus in North America. BMC Evol. Biol. 2017, 17 (1), 118.
- Congressional Research Service; Biondo. The Highly Pathogenic Avian Influenza (HPAI) Outbreak in Poultry, 2022-Present; R48518; 2025. Available online: https://www.congress.gov/crs-product/R48518 (accessed 2025-09-27).
- Burrough, E. R.; Magstadt, D. R.; Petersen, B.; Timmermans, S. J.; Gauger, P. C.; Zhang, J.; Siepker, C.; Mainenti, M.; Li, G.; Thompson, A. C.; Gorden, P. J.; Plummer, P. J.; Main, R. Highly Pathogenic Avian Influenza A(H5N1) Clade 2.3.4.4b Virus Infection in Domestic Dairy Cattle and Cats, United States, 2024. Emerg. Infect. Dis. 2024, 30 (7), 1335–1343. https://doi.org/10.3201/eid3007.240508. [CrossRef]
- Caserta, L. C.; Frye, E. A.; Butt, S. L.; Laverack, M.; Nooruzzaman, M.; Covaleda, L. M.; Thompson, A. C.; Koscielny, M. P.; Cronk, B.; Johnson, A.; Kleinhenz, K.; Edwards, E. E.; Gomez, G.; Hitchener, G.; Martins, M.; Kapczynski, D. R.; Suarez, D. L.; Alexander Morris, E. R.; Hensley, T.; Beeby, J. S.; Lejeune, M.; Swinford, A. K.; Elvinger, F.; Dimitrov, K. M.; Diel, D. G. Spillover of Highly Pathogenic Avian Influenza H5N1 Virus to Dairy Cattle. Nature 2024, 634 (8034), 669–676. https://doi.org/10.1038/s41586-024-07849-4. [CrossRef]
- Guan, L.; Eisfeld, A. J.; Pattinson, D.; Gu, C.; Biswas, A.; Maemura, T.; Trifkovic, S.; Babujee, L.; Presler, R.; Dahn, R.; Halfmann, P. J.; Barnhardt, T.; Neumann, G.; Thompson, A.; Swinford, A. K.; Dimitrov, K. M.; Poulsen, K.; Kawaoka, Y. Cow’s Milk Containing Avian Influenza A(H5N1) Virus — Heat Inactivation and Infectivity in Mice. N. Engl. J. Med. 2024, 391 (1), 87–90. https://doi.org/10.1056/NEJMc2405495. [CrossRef]
- Frye, E. A.; Nooruzzaman, M.; Cronk, B.; Laverack, M.; Oliveira, P. S. B. de; Caserta, L. C.; Lejeune, M.; Diel, D. G. Isolation of Highly Pathogenic Avian Influenza A(H5N1) Virus from Cat Urine after Raw Milk Ingestion, United States - Volume 31, Number 8—August 2025 - Emerging Infectious Diseases Journal - CDC. https://doi.org/10.3201/eid3108.250309. [CrossRef]
- Givens, M. D. Review: Risks of Disease Transmission through Semen in Cattle. Animal 2018, 12, s165–s171. https://doi.org/10.1017/S1751731118000708. [CrossRef]
- Samanta, L.; Parida, R.; Dias, T. R.; Agarwal, A. The Enigmatic Seminal Plasma: A Proteomics Insight from Ejaculation to Fertilization. Reprod. Biol. Endocrinol. 2018, 16 (1), 41. https://doi.org/10.1186/s12958-018-0358-6. [CrossRef]
- Gautier, C.; Aurich, C. “Fine Feathers Make Fine Birds” – The Mammalian Sperm Plasma Membrane Lipid Composition and Effects on Assisted Reproduction. Anim. Reprod. Sci. 2022, 246, 106884. https://doi.org/10.1016/j.anireprosci.2021.106884. [CrossRef]
- Raheja, N.; Choudhary, S.; Grewal, S.; Sharma, N.; Kumar., N. A Review on Semen Extenders and Additives Used in Cattle and Buffalo Bull Semen Preservation. J. Entomol. Zool. Stud. 6 (3), 293–245.
- Bodu, M.; Hitit, M.; Greenwood, O. C.; Murray, R. D.; Memili, E. Extender Development for Optimal Cryopreservation of Buck Sperm to Increase Reproductive Efficiency of Goats. Front. Vet. Sci. 2025, 12. https://doi.org/10.3389/fvets.2025.1554771. [CrossRef]
- National Animal Health Laboratory Network. Standard operating procedure for Real-time RT-PCR Detection of Influenza A and Avian Paramyxovirus Type-1. (NVSL-SOP-0068). Available online: https://www.aphis.usda.gov/animal_health/lab_info_services/downloads/ ApprovedSOPList.pdf (accessed 2025-09-26).
- National Animal Health Laboratory Network. Standard operating procedure for Nucleic Acid Extraction Using the MagMAX Core Nucleic Acid Purification Kit on a Magnetic Particle Processor. (NVSL-SOP-0643). Available online: https://www.aphis.usda.gov/animal_health/lab_info_services/downloads/ApprovedSOPList.pdf (accessed 2025-09-26).
- National Animal Health Laboratory Network. Standard operating procedure for Nucleic Acid Extraction Using the INDICAL BIOSCIENCE IndiMag Pathogen Kit on a Magnetic Particle Processor. (NVSL-SOP-0645). Available online: https://www.aphis.usda.gov/animal_health/lab_info_services/downloads/ApprovedSOPList.pdf (accessed 2025-09-26).
- Vandenburg-Carroll, A.; Marthaler, D. G.; Lim, A. Enhancing Diagnostic Resilience: Evaluation of Extraction Platforms and IndiMag Pathogen Kits for Rapid Animal Disease Detection. Microbiol. Res. 2025, 16 (4), 80. https://doi.org/10.3390/microbiolres16040080. [CrossRef]
- Yan, L.; Toohey-Kurth, K. L.; Crossley, B. M.; Bai, J.; Glaser, A. L.; Tallmadge, R. L.; Goodman, L. B. Inhibition Monitoring in Veterinary Molecular Testing. J. Vet. Diagn. Investig. Off. Publ. Am. Assoc. Vet. Lab. Diagn. Inc 2020, 32 (6), 758–766. https://doi.org/10.1177/1040638719889315. [CrossRef]
- Lee, S. M.; Balakrishnan, H. K.; Doeven, E. H.; Yuan, D.; Guijt, R. M. Chemical Trends in Sample Preparation for Nucleic Acid Amplification Testing (NAAT): A Review. Biosensors 2023, 13 (11), 980. https://doi.org/10.3390/bios13110980. [CrossRef]
- Schellhammer, S. K.; Hudson, B. C.; Cox, J. O.; Dawson Green, T. Alternative Direct-to-amplification Sperm Cell Lysis Techniques for Sexual Assault Sample Processing. J. Forensic Sci. 2022, 67 (4), 1668–1678. https://doi.org/10.1111/1556-4029.15027. [CrossRef]
- Hennekens, C. M.; Cooper, E. S.; Cotton, R. W.; Grgicak, C. M. The Effects of Differential Extraction Conditions on the Premature Lysis of Spermatozoa. J. Forensic Sci. 2013, 58 (3), 744–752. https://doi.org/10.1111/1556-4029.12098. [CrossRef]
- Yoshida, K.; Sekiguchi, K.; Mizuno, N.; Kasai, K.; Sakai, I.; Sato, H.; Seta, S. The Modified Method of Two-Step Differential Extraction of Sperm and Vaginal Epithelial Cell DNA from Vaginal Fluid Mixed with Semen. Forensic Sci. Int. 1995, 72 (1), 25–33. https://doi.org/10.1016/0379-0738(94)01668-U. [CrossRef]
- Hsiao, C.-C.; Lin, C.-C.; Chen, Y.-M.; Cheng, M.-C.; Davison, S.; Ma, J.; Dai, H.-L. Quantitative Real-Time PCR Detection of Inactivated H5 Avian Influenza Virus in Raw Milk Samples by Miniaturized Instruments Designed for On-Site Testing. bioRxiv 2025, 2025.06.02.657307. https://doi.org/10.1101/2025.06.02.657307. [CrossRef]
- Miller, M. R.; Braun, E.; Ip, H. S.; Tyson, G. H. Domestic and Wild Animal Samples and Diagnostic Testing for SARS-CoV-2. Vet. Q. 43 (1), 1–11. https://doi.org/10.1080/01652176.2023.2263864. [CrossRef]
- Sharma, G.; Subramaniam, S.; De, A.; Das, B.; Dash, B.; Sanyal, A.; Misra, A.; Pattnaik, B. Detection of Foot-and-Mouth Disease Virus in Semen of Infected Cattle Bulls. Indian J. Anim. Sci. 2012, 82, 1472–1476.
- Alexandersen, S.; Zhang, Z.; Donaldson, A. I.; Garland, A. J. M. The Pathogenesis and Diagnosis of Foot-and-Mouth Disease. J. Comp. Pathol. 2003, 129 (1), 1–36. https://doi.org/10.1016/S0021-9975(03)00041-0. [CrossRef]
- World Organisation for Animal Health. Foot and Mouth Disease; OIE Terrestrial Manual 2009. Available online: https://www.woah.org/fileadmin/Home/eng/Animal_Health_in_the_World/docs/pdf/2.01.05_FMD.pdf (accessed 2025-09-26).
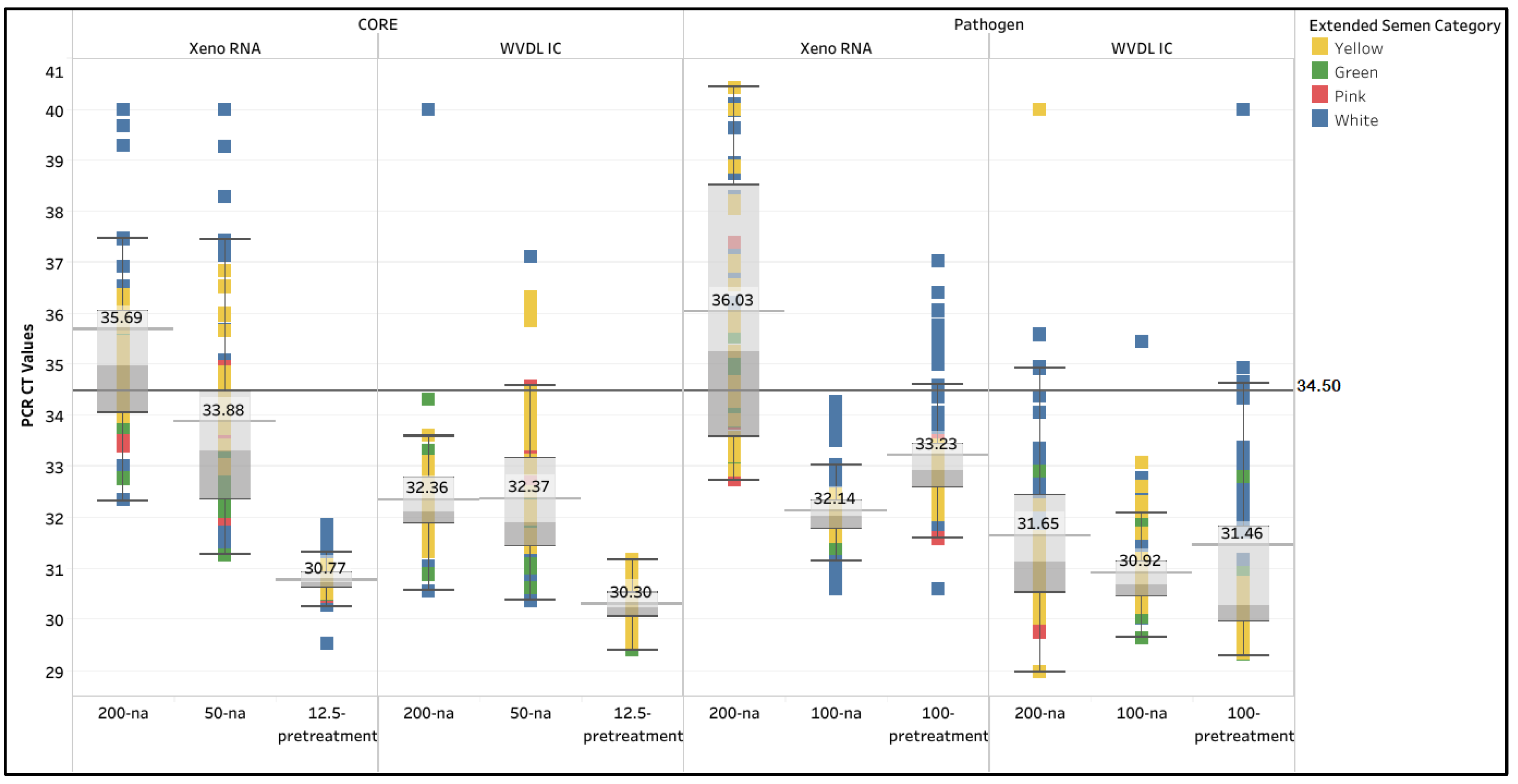
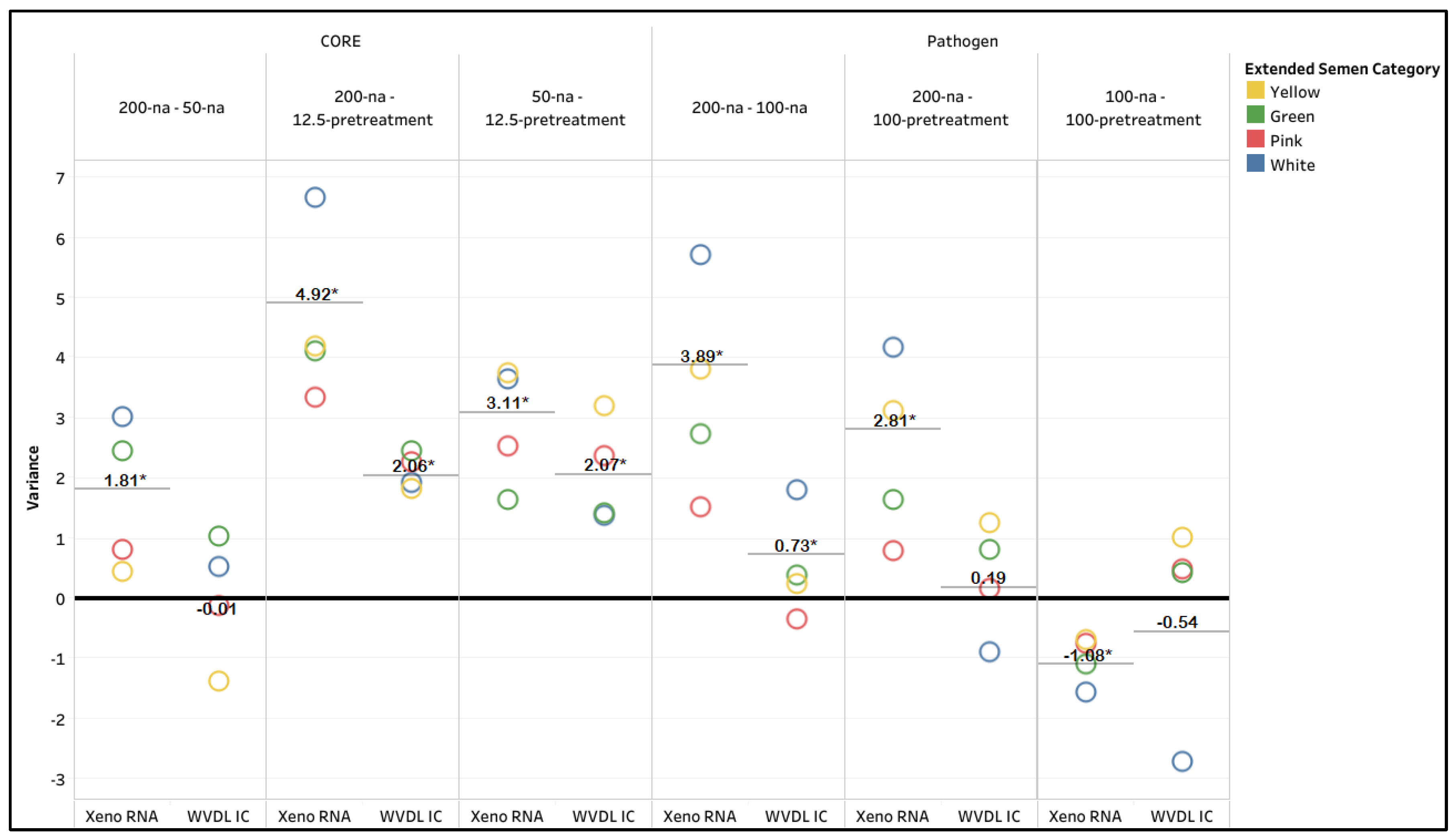
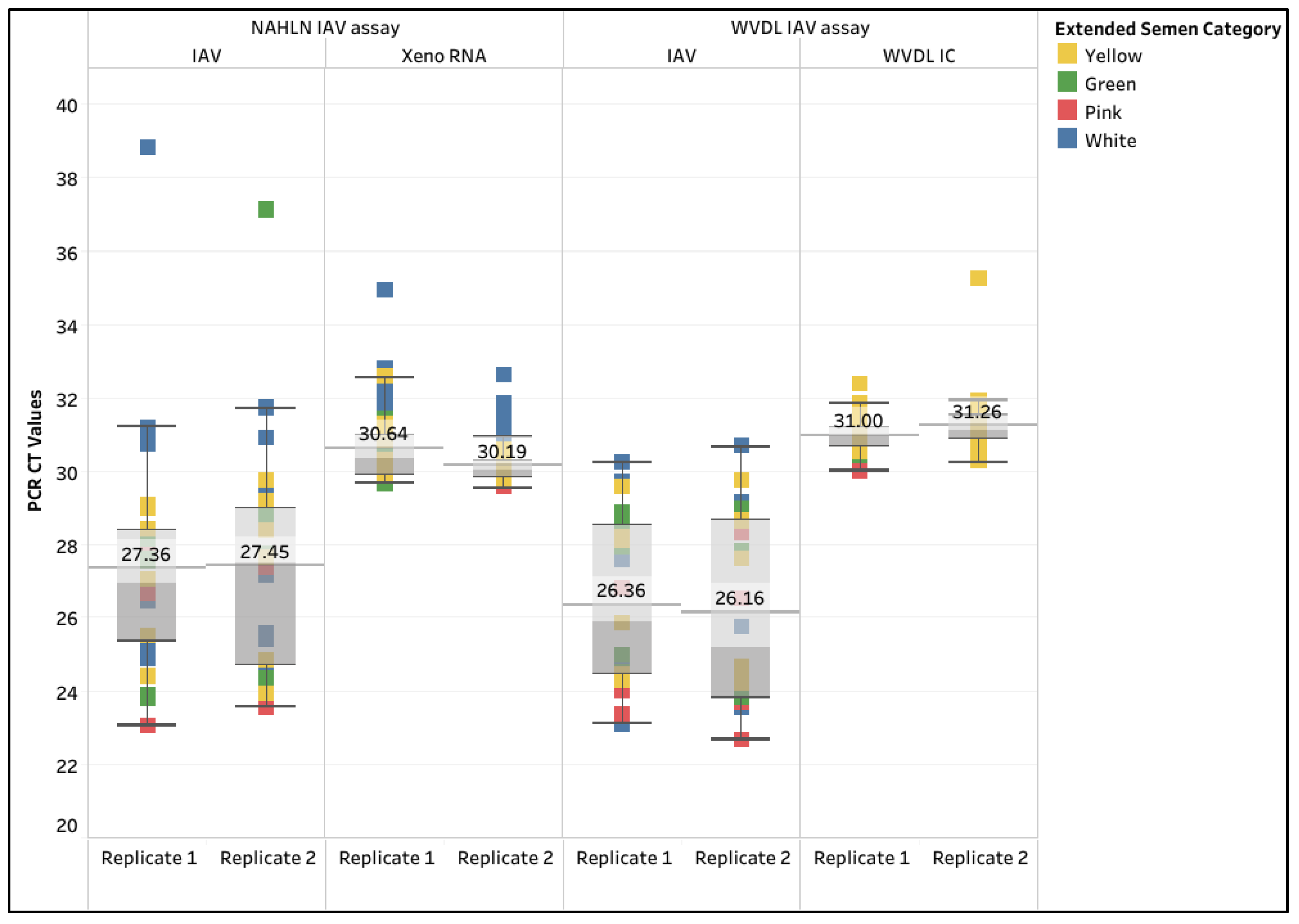
| PCR Methodology | CORE | Pathogen | ||||||||||
| Extraction Kit | Xeno RNA | WVDL IC | Xeno RNA | WVDL IC | ||||||||
| Semen input & modification | 200-na | 50-na | 12.5-pretreat-ment | 200-na | 50-na | 12.5-pretreat-ment | 200-na | 100-na | 100-pretreat-ment | 200-na | 100-na | 100-pretreat-ment |
| Yellow (n=24) | 25.0 | 62.5 | 100.0 | 100.0 | 83.3 | 100.0 | 41.7 | 100.0 | 100.0 | 95.8 | 100.0 | 100.0 |
| Green (n=16) | 12.5 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 50.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Pink (n=16) | 81.3 | 87.5 | 100.0 | 100.0 | 93.8 | 100.0 | 87.5 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| White (n=32) | 21.9 | 62.5 | 100.0 | 93.8 | 96.9 | 100.0 | 3.1 | 100.0 | 59.4 | 83.3 | 100.0 | 68.8 |
| Overall Passing rate (n=88) | 31.8 | 73.9 | 100.0 | 97.7 | 93.2 | 100.0 | 37.5 | 100.0 | 85.2 | 94.3 | 100.0 | 88.6 |
| Extraction Kit | Core | Pathogen | ||||
| Semen input & Modification | 12.5-pretreatment | 100-na | 100-pretreatment | |||
| Extraction Rep | 1 | 2 | 1 | 2 | 1 | 2 |
| M. bovis (n=8) | 75.0% | 87.5% | 75.0% | 100.0% | 87.5% | 100.0% |
| BVDV (n=5) | 60.0% | 60.0% | 80.0% | 80.0% | 100.0% | 80.0% |
| BHV-1 (n=23) | 87.0% | 95.7% | 95.7% | 95.7% | 95.7% | 100.0% |
| Sensitivity (n=36) | 80.6% | 88.9% | 88.9% | 94.4% | 94.4% | 97.2% |
| Reference Strain |
NAHLN IAV assay | WVDL IAV assay | |||||
|---|---|---|---|---|---|---|---|
| Replicate 1 | Replicate 2 |
Replicate 3 | Replicate 1 | Replicate 2 |
Replicate 3 | ||
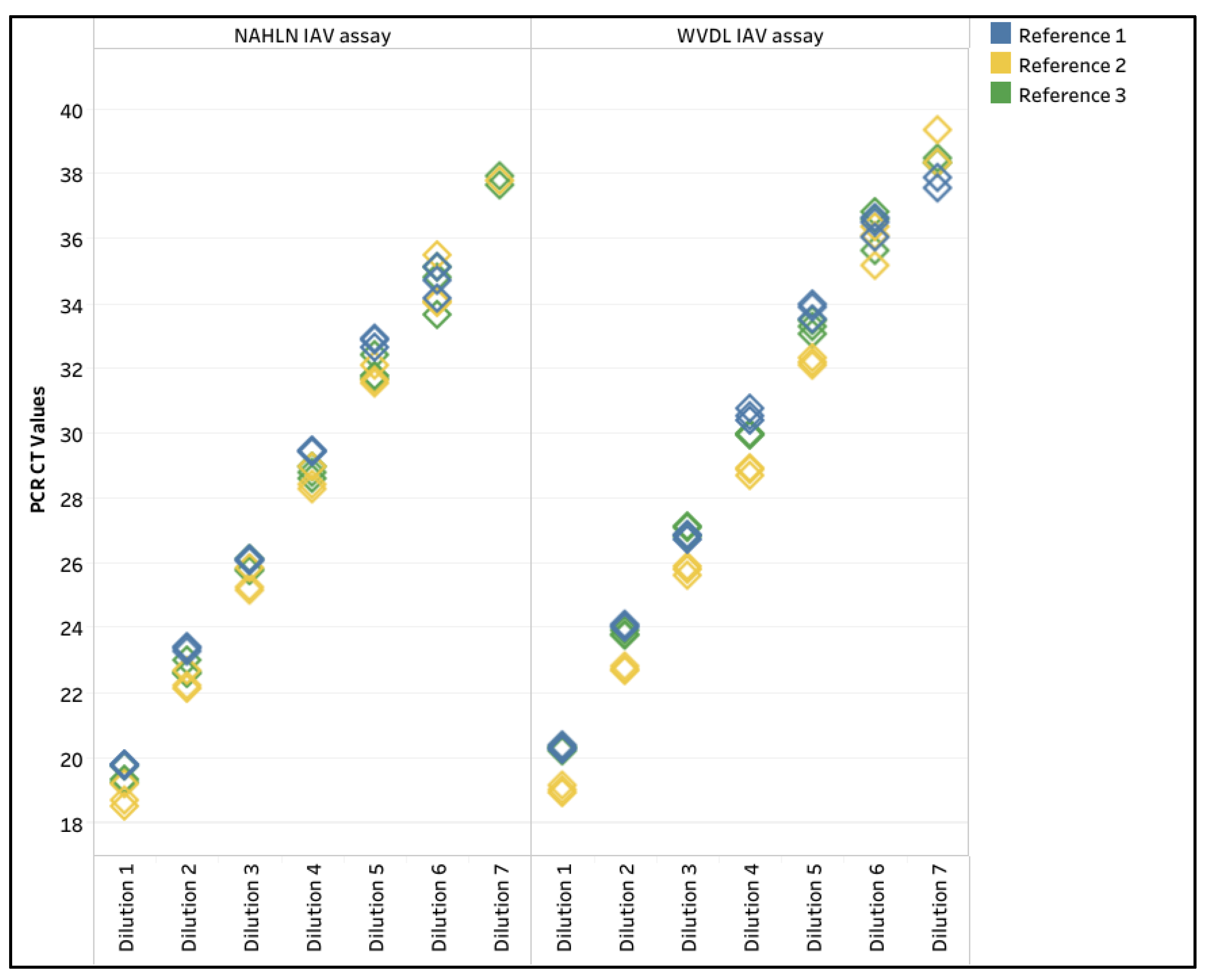
| Limit of Detection (LOD) | 1 | 6 | 6 | 6 | 6 | 6 | 6 |
| 2 | 7 | 6 | 6 | 6 | 7 | 7 | |
| 3 | 7 | 6 | 7 | 6 | 7 | 7 | |
| R2 value | 1 | 0.997 | 0.99 | 0.994 | 0.997 | 0.995 | 0.999 |
| 2 | 0.999 | 0.998 | 0.998 | 0.999 | 0.999 | 0.996 | |
| 3 | 0.997 | 0.995 | 0.999 | 0.997 | 0.995 | 0.996 | |
| PCR Efficiency (%) | 1 | 110.6 | 118.0 | 113.3 | 102.1 | 104.8 | 103.8 |
| 2 | 98.5 | 109.0 | 101.3 | 106.2 | 97.8 | 101.5 | |
| 3 | 100.6 | 120.7 | 110.9 | 116.4 | 112.2 | 109.9 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




