Submitted:
21 September 2025
Posted:
23 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
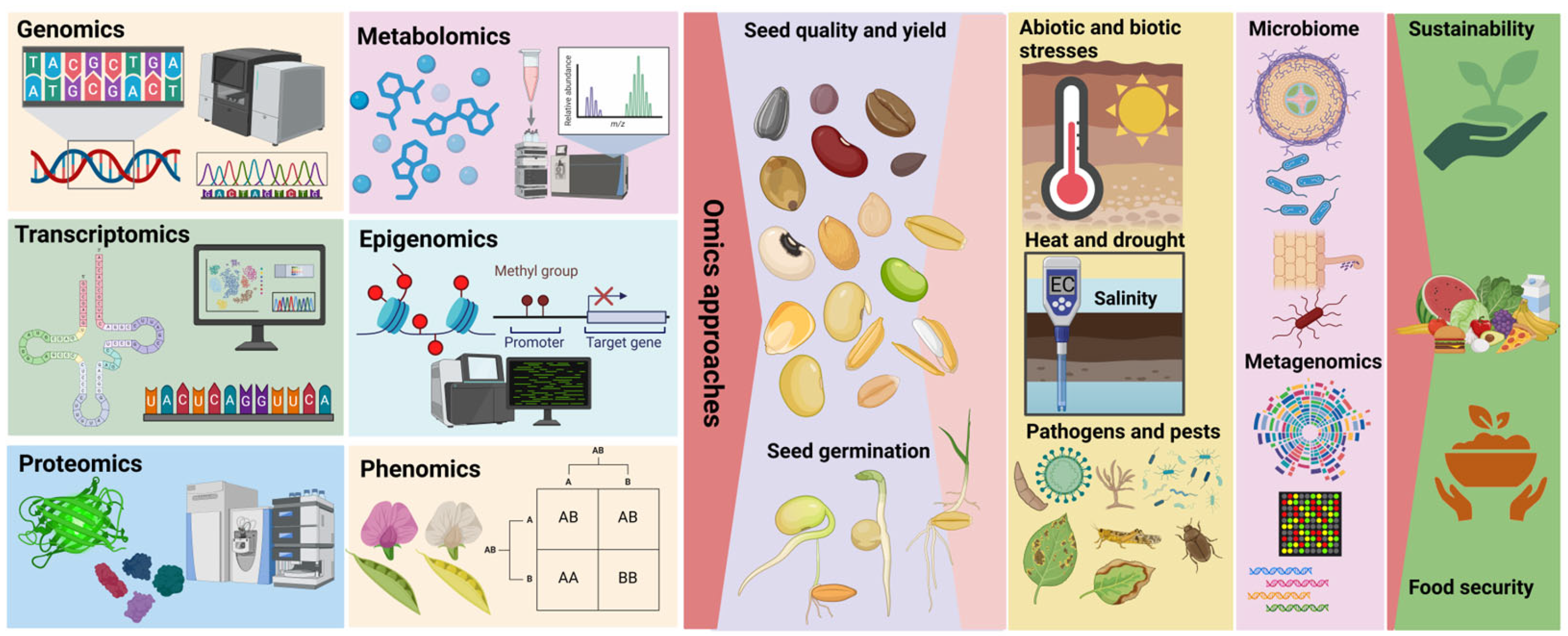
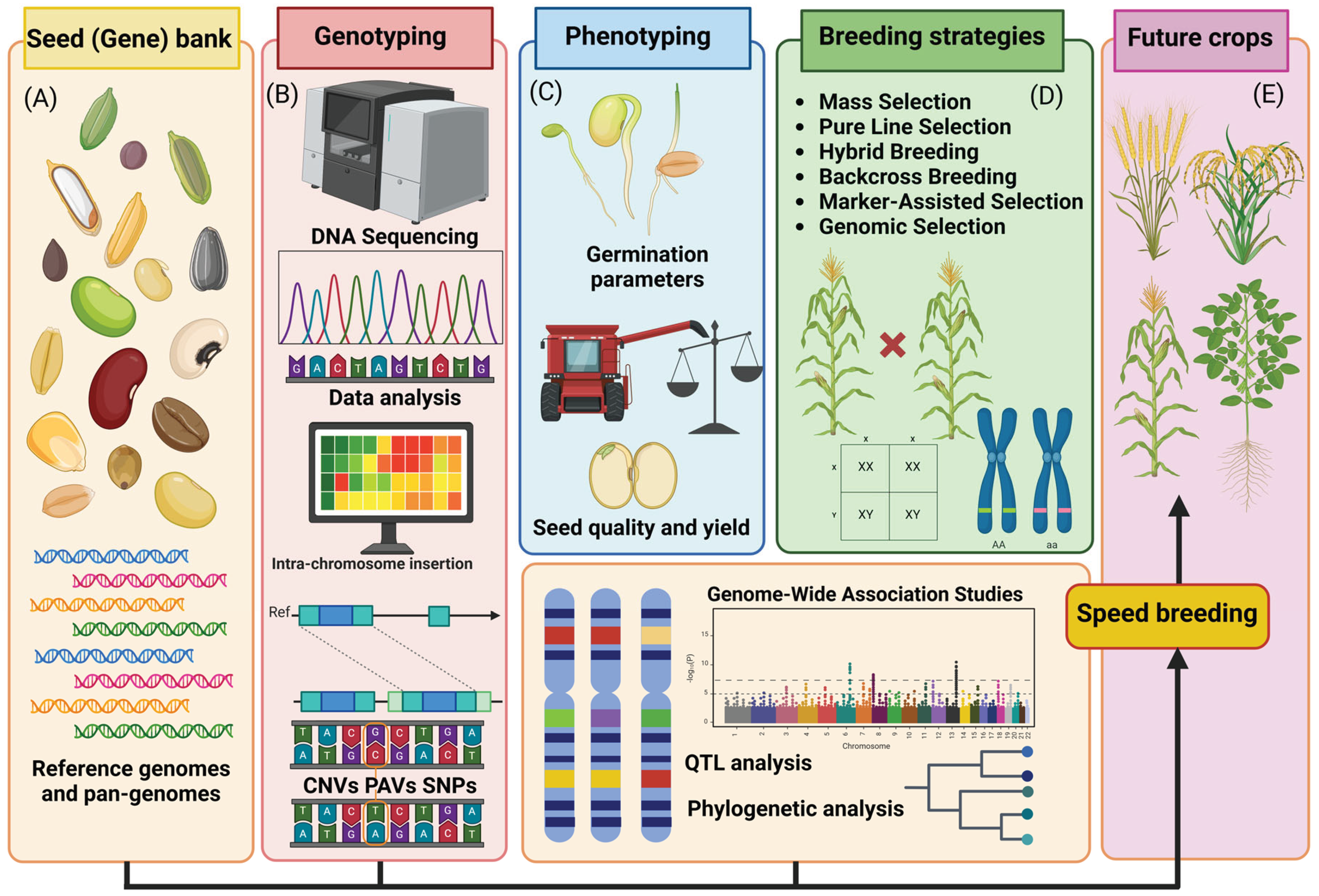
2. Genomics Approaches
3. Transcriptomics Insights
4. Proteomics Advances
5. Metabolomics
6. Epigenomics
7. Phenomics
8. Seed Dormancy and Germination
9. Abiotic and Biotic Stress-Resistant Seeds
10. The Role of Seed/Soil Microbiome in Quality and Yield
5. Conclusions and Future Directions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
- The following abbreviations are used in this manuscript: DNA – Deoxyribonucleic acid
- WGS – Whole genome sequencing
- SNP – Single nucleotide Polymorphism
- CNV – Copy number variant
- PAV – Presence/absence variant
- GWAS – Genome-wide association studies
- QTL – Quantitative trait loci
- QTL-seq – Quantitative trait loci sequencing
- MAS – Marker-assisted selection
- SSR – Simple sequence repeat
- SDN – Site directed nuclease
- CWR – Crop wild relatives
- RNA – Ribonucleic acid
- RT-PCR – Reverse transcription-polymerase chain reaction
- DEG – Differentially expressed gene
- LC-MS – Liquid chromatography mass spectrometry
- GC-MS – Gas chromatography mass spectrometry
- MS – Mass spectrometry
- DOG – Delay of Germination
- GA – Gibberellic acid
References
- Adeleke, B.S.; Babalola, O.O. The endosphere microbial communities, a great promise in agriculture. International Microbiology 2021, 24, 1–17. [Google Scholar] [CrossRef]
- Afzal, M.; Sielaff, M.; Distler, U.; Schuppan, D.; Tenzer, S.; Longin, C.F.H. Reference proteomes of five wheat species as starting point for future design of cultivars with lower allergenic potential. npj Science of Food 2023, 7, 9. [Google Scholar] [CrossRef]
- Agarwal, P.; Kapoor, S.; Tyagi, A.K. Transcription factors regulating the progression of monocot and dicot seed development. Bioessays: News and Reviews in Molecular, Cellular and Developmental Biology 2011, 33, 189–202. [Google Scholar] [CrossRef]
- Agnolucci, P.; Rapti, C.; Alexander, P.; De Lipsis, V.; Holland, R.A.; Eigenbrod, F.; Ekins, P. Impacts of rising temperatures and farm management practices on global yields of 18 crops. Nature Food 2020, 1, 562–571. [Google Scholar] [CrossRef]
- Ahmad, A.; Munawar, N.; Khan, Z.; Qusmani, A.T.; Khan, S.H.; Jamil, A.; Ashraf, S.; et al. An Outlook on Global Regulatory Landscape for Genome-Edited Crops. International Journal of Molecular Sciences 2021, 22. [Google Scholar] [CrossRef]
- Ahmad, P.; Abdel Latef, A.A.H.; Rasool, S.; Akram, N.A.; Ashraf, M.; Gucel, S. Role of proteomics in crop stress tolerance. Frontiers in Plant Science 2016, 7, 1336. [Google Scholar] [CrossRef]
- Akpertey, A.; Singh, R.J.; Diers, B.W.; Graef, G.L.; Mian, M.A.R.; Shannon, J.G.; Scaboo, A.M.; et al. Genetic Introgression from Glycine tomentella to Soybean to Increase Seed Yield. Crop science 2018, 58, 1277–1291. [Google Scholar] [CrossRef]
- Aleem, M.; Razzaq, M.K.; Aleem, M.; Yan, W.; Sharif, I.; Siddiqui, M.H.; Aleem, S.; et al. Genome-wide association study provides new insight into the underlying mechanism of drought tolerance during seed germination stage in soybean. Scientific Reports 2024, 14, 20765. [Google Scholar] [CrossRef] [PubMed]
- Amas, J.C.; Bayer, P.E.; Hong Tan, W.; Tirnaz, S.; Thomas, W.J.W.; Edwards, D.; Batley, J. Comparative pangenome analyses provide insights into the evolution of Brassica rapa resistance gene analogues (RGAs). Plant Biotechnology Journal 2023, 21, 2100–2112. [Google Scholar] [CrossRef] [PubMed]
- Angon, P.B.; Mondal, S.; Akter, S.; Sakil Md, A.; Jalil Md, A. Roles of CRISPR to mitigate drought and salinity stresses on plants. Plant Stress 2023, 8, 100169. [Google Scholar] [CrossRef]
- Bai, B.; van der Horst, S.; Cordewener, J.H.G.; America, T.A.H.P.; Hanson, J.; Bentsink, L. Seed-Stored mRNAs that Are Specifically Associated to Monosomes Are Translationally Regulated during Germination. Plant Physiology 2020, 182, 378–392. [Google Scholar] [CrossRef]
- Bailly, C.; Gomez Roldan, M.V. Impact of climate perturbations on seeds and seed quality for global agriculture. The Biochemical Journal 2023, 480, 177–196. [Google Scholar] [CrossRef]
- Balouchi, H.; Soltani Khankahdani, V.; Moradi, A.; Gholamhoseini, M.; Piri, R.; Heydari, S.Z.; Dedicova, B. Seed Fatty Acid Changes Germination Response to Temperature and Water Potentials in Six Sesame (Sesamum indicum L.) Cultivars: Estimating the Cardinal Temperatures. Agriculture 2023, 13, 1936. [Google Scholar] [CrossRef]
- Barret, M.; Briand, M.; Bonneau, S.; Préveaux, A.; Valière, S.; Bouchez, O.; Hunault, G.; et al. Emergence shapes the structure of the seed microbiota. Applied and Environmental Microbiology 2015, 81, 1257–1266. [Google Scholar] [CrossRef] [PubMed]
- Begum, K.; Hasan, N.; Shammi, M. Selective biotic stressors’ action on seed germination: A review. Plant Science 2024, 346, 112156. [Google Scholar] [CrossRef]
- Begum, N.; Qin, C.; Ahanger, M.A.; Raza, S.; Khan, M.I.; Ashraf, M.; Ahmed, N.; et al. Role of arbuscular mycorrhizal fungi in plant growth regulation: implications in abiotic stress tolerance. Frontiers in Plant Science 2019, 10, 1068. [Google Scholar] [CrossRef]
- Bentsink, L.; Jowett, J.; Hanhart, C.J.; Koornneef, M. Cloning of DOG1, a quantitative trait locus controlling seed dormancy in Arabidopsis. Proceedings of the National Academy of Sciences of the United States of America 2006, 103, 17042–17047. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, A.; Devi, P.; Chaudhary, S.; Rani, A.; Jha, U.C.; Kumar, S.; Bindumadhava, H.; et al. “Omics” approaches in developing combined drought and heat tolerance in food crops. Plant Cell Reports 2022, 41, 699–739. [Google Scholar] [CrossRef]
- Bolingue, W.; Rosnoblet, C.; Leprince, O.; Vu, B.L.; Aubry, C.; Buitink, J. The MtSNF4b subunit of the sucrose non-fermenting-related kinase complex connects after-ripening and constitutive defense responses in seeds of Medicago truncatula. The Plant Journal 2010, 61, 792–803. [Google Scholar] [CrossRef]
- Bourgeois, M.; Jacquin, F.; Savois, V.; Sommerer, N.; Labas, V.; Henry, C.; Burstin, J. Dissecting the proteome of pea mature seeds reveals the phenotypic plasticity of seed protein composition. Proteomics 2009, 9, 254–271. [Google Scholar] [CrossRef]
- Brocklehurst, P.A.; Fraser, R.S.S. Ribosomal RNA integrity and rate of seed germination. Planta 1980, 148, 417–421. [Google Scholar] [CrossRef]
- Brozynska, M.; Furtado, A.; Henry, R.J. Genomics of crop wild relatives: expanding the gene pool for crop improvement. Plant Biotechnology Journal 2016, 14, 1070–1085. [Google Scholar] [CrossRef] [PubMed]
- Buchmann, J.P.; Holmes, E.C. Cell walls and the convergent evolution of the viral envelope. Microbiology and Molecular Biology Reviews 2015, 79, 403–418. [Google Scholar] [CrossRef]
- Campbell, M.T.; Hu, H.; Yeats, T.H.; Caffe-Treml, M.; Gutiérrez, L.; Smith, K.P.; Sorrells, M.E.; et al. Translating insights from the seed metabolome into improved prediction for lipid-composition traits in oat (Avena sativa L.). Genetics 2021, 217. [Google Scholar] [CrossRef]
- Chen, B.-X.; Fu, H.; Gao, J.-D.; Zhang, Y.-X.; Huang, W.-J.; Chen, Z.-J.; Qi-Zhang et, a.l. Identification of metabolomic biomarkers of seed vigor and aging in hybrid rice. Rice (New York, N.Y.) 2022, 15, 7. [Google Scholar] [CrossRef]
- Chen, K.; Arora, R.; Arora, U. Osmopriming of spinach (Spinacia oleracea L. cv. Bloomsdale) seeds and germination performance under temperature and water stress. Seed Science and Technology 2010, 38, 36–48. [Google Scholar] [CrossRef]
- Chu, W.; Liu, J.; Cheng, H.; Li, C.; Fu, L.; Wang, W.; Wang, H.; et al. A lignified-layer bridge controlled by a single recessive gene is associated with high pod-shatter resistance in Brassica napus L. The Crop journal 2021. [Google Scholar] [CrossRef]
- collection, handling, storage and pre-treatment of Prosopis seeds in Latin America. (n.d.). . Retrieved June 24, 2025, from https://www.fao.org/4/q2180e/Q2180E00.htm#TOC.
- Crop loss/waste on Australian horticulture farms, 2023–2024 - DAFF. (n.d.). . Retrieved September 20, 2025, from https://www.agriculture.gov.au/abares/research-topics/surveys/horticulture-crop-loss-23-24.
- Das, S.S.; Karmakar, P.; Nandi, A.K.; Sanan-Mishra, N. Small RNA mediated regulation of seed germination. Frontiers in Plant Science 2015, 6, 828. [Google Scholar] [CrossRef] [PubMed]
- Dobiesz, M.; Piotrowicz-Cieślak, A.I. Proteins in Relation to Vigor and Viability of White Lupin (Lupinus albus L.) Seed Stored for 26 Years. Frontiers in Plant Science 2017, 8, 1392. [Google Scholar] [CrossRef] [PubMed]
- Dolatabadian, A.; Bayer, P.E.; Tirnaz, S.; Hurgobin, B.; Edwards, D.; Batley, J. Characterization of disease resistance genes in the Brassica napus pangenome reveals significant structural variation. Plant Biotechnology Journal 2020, 18, 969–982. [Google Scholar] [CrossRef]
- Domergue, J.-B.; Lalande, J.; Beucher, D.; Satour, P.; Abadie, C.; Limami, A.M.; Tcherkez, G. Experimental Evidence for Seed Metabolic Allometry in Barrel Medic (Medicago truncatula Gaertn.). International Journal of Molecular Sciences 2022, 23. [Google Scholar] [CrossRef]
- Dong, Y.; Yang, X.; Liu, J.; Wang, B.-H.; Liu, B.-L.; Wang, Y.-Z. Pod shattering resistance associated with domestication is mediated by a NAC gene in soybean. Nature Communications 2014, 5, 3352. [Google Scholar] [CrossRef]
- Dwivedi, S.L.; Spillane, C.; Lopez, F.; Ayele, B.T.; Ortiz, R. First the seed: Genomic advances in seed science for improved crop productivity and food security. Crop science 2021, 61, 1501–1526. [Google Scholar] [CrossRef]
- El-Maarouf-Bouteau, H. The seed and the metabolism regulation. Biology 2022, 11. [Google Scholar] [CrossRef]
- Estimating crop yield | Agriculture and Food. (n.d.). . Retrieved December 17, 2024, from https://www.agric.wa.gov.au/mycrop/estimating-crop-yield.
- Fadiji, A.E.; Babalola, O.O. Metagenomics methods for the study of plant-associated microbial communities: A review. Journal of Microbiological Methods 2020, 170, 105860. [Google Scholar] [CrossRef]
- Fait, A.; Angelovici, R.; Less, H.; Ohad, I.; Urbanczyk-Wochniak, E.; Fernie, A.R.; Galili, G. Arabidopsis seed development and germination is associated with temporally distinct metabolic switches. Plant Physiology 2006, 142, 839–854. [Google Scholar] [CrossRef] [PubMed]
- Faryad, A.; Aziz, F.; Tahir, J.; Kousar, M.; Qasim, M.; Shamim, A. Integration of OMICS technologies for crop improvement. Protein and Peptide Letters 2021, 28, 896–908. [Google Scholar] [CrossRef] [PubMed]
- Fernie, A.R.; Yan, J. De Novo Domestication: An Alternative Route toward New Crops for the Future. Molecular Plant 2019, 12, 615–631. [Google Scholar] [CrossRef] [PubMed]
- Fierer, N.; Leff, J.W.; Adams, B.J.; Nielsen, U.N.; Bates, S.T.; Lauber, C.L.; Owens, S.; et al. Cross-biome metagenomic analyses of soil microbial communities and their functional attributes. Proceedings of the National Academy of Sciences of the United States of America 2012, 109, 21390–21395. [Google Scholar] [CrossRef]
- Finch-Savage, W.E.; Bassel, G.W. Seed vigour and crop establishment: extending performance beyond adaptation. Journal of Experimental Botany 2016, 67, 567–591. [Google Scholar] [CrossRef]
- Flint-Garcia, S.A.; Bodnar, A.L.; Scott, M.P. Wide variability in kernel composition, seed characteristics, and zein profiles among diverse maize inbreds, landraces, and teosinte. TAG. Theoretical and Applied Genetics. Theoretische und Angewandte Genetik 2009, 119, 1129–1142. [Google Scholar] [CrossRef]
- Fujimoto, R.; Taylor, J.M.; Shirasawa, S.; Peacock, W.J.; Dennis, E.S. Heterosis of Arabidopsis hybrids between C24 and Col is associated with increased photosynthesis capacity. Proceedings of the National Academy of Sciences of the United States of America 2012, 109, 7109–7114. [Google Scholar] [CrossRef] [PubMed]
- Fujita, D.; Trijatmiko, K.R.; Tagle, A.G.; Sapasap, M.V.; Koide, Y.; Sasaki, K.; Tsakirpaloglou, N.; et al. NAL1 allele from a rice landrace greatly increases yield in modern indica cultivars. Proceedings of the National Academy of Sciences of the United States of America 2013, 110, 20431–20436. [Google Scholar] [CrossRef] [PubMed]
- Funatsuki, H.; Suzuki, M.; Hirose, A.; Inaba, H.; Yamada, T.; Hajika, M.; Komatsu, K.; et al. Molecular basis of a shattering resistance boosting global dissemination of soybean. Proceedings of the National Academy of Sciences of the United States of America 2014, 111, 17797–17802. [Google Scholar] [CrossRef] [PubMed]
- Fustier, M.A.; Brandenburg, J.T.; Boitard, S.; Lapeyronnie, J.; Eguiarte, L.E.; Vigouroux, Y.; Manicacci, D.; et al. Signatures of local adaptation in lowland and highland teosintes from whole-genome sequencing of pooled samples. Molecular Ecology 2017, 26, 2738–2756. [Google Scholar] [CrossRef]
- Gabur, I.; Chawla, H.S.; Lopisso, D.T.; von Tiedemann, A.; Snowdon, R.J.; Obermeier, C. Gene presence-absence variation associates with quantitative Verticillium longisporum disease resistance in Brassica napus. Scientific Reports 2020, 10, 4131. [Google Scholar] [CrossRef]
- Gali, K.K.; Sackville, A.; Tafesse, E.G.; Lachagari, V.B.R.; McPhee, K.; Hybl, M.; Mikić, A.; et al. Genome-Wide Association Mapping for Agronomic and Seed Quality Traits of Field Pea (Pisum sativum L.). Frontiers in Plant Science 2019, 10, 1538. [Google Scholar] [CrossRef]
- Galindo-Luján, R.; Pont, L.; Minic, Z.; Berezovski, M.V.; Sanz-Nebot, V.; Benavente, F. Characterization and differentiation of quinoa seed proteomes by label-free mass spectrometry-based shotgun proteomics. Food chemistry 2021, 363, 130250. [Google Scholar] [CrossRef]
- Galland, M.; Huguet, R.; Arc, E.; Cueff, G.; Job, D.; Rajjou, L. Dynamic proteomics emphasizes the importance of selective mRNA translation and protein turnover during Arabidopsis seed germination. Molecular & Cellular Proteomics 2014, 13, 252–268. [Google Scholar] [CrossRef]
- Gallardo, K.; Job, C.; Groot, S.P.; Puype, M.; Demol, H.; Vandekerckhove, J.; Job, D. Proteomic analysis of arabidopsis seed germination and priming. Plant Physiology 2001, 126, 835–848. [Google Scholar] [CrossRef]
- Gallego-Bartolomé, J. DNA methylation in plants: mechanisms and tools for targeted manipulation. The New Phytologist 2020, 227, 38–44. [Google Scholar] [CrossRef]
- Gallusci, P.; Dai, Z.; Génard, M.; Gauffretau, A.; Leblanc-Fournier, N.; Richard-Molard, C.; Vile, D.; et al. Epigenetics for plant improvement: current knowledge and modeling avenues. Trends in Plant Science 2017, 22, 610–623. [Google Scholar] [CrossRef] [PubMed]
- Gangurde, S.S.; Khan, A.W.; Janila, P.; Variath, M.T.; Manohar, S.S.; Singam, P.; Chitikineni, A.; et al. Whole-genome sequencing based discovery of candidate genes and diagnostic markers for seed weight in groundnut. The plant genome 2023, 16, e20265. [Google Scholar] [CrossRef]
- Gautam, T.; Amardeep Saripalli, G.; Rakhi Kumar, A.; Gahlaut, V.; Gadekar, D.A.; et al. Introgression of a drought insensitive grain yield QTL for improvement of four Indian bread wheat cultivars using marker assisted breeding without background selection. Journal of plant biochemistry and biotechnology 2021, 30, 172–183. [Google Scholar] [CrossRef]
- Ghamkhar, K.; Hay, F.R.; Engbers, M.; Dempewolf, H.; Schurr, U. Realizing the potential of plant genetic resources: the use of phenomics for genebanks. Plants, People, Planet 2025, 7, 23–32. [Google Scholar] [CrossRef]
- Ghazy, M.I.; El-Naem, S.A.; Hefeina, A.G.; Sallam, A.; Eltaher, S. Genome-Wide Association Study of Rice Diversity Panel Reveals New QTLs for Tolerance to Water Deficit Under the Egyptian Conditions. Rice (New York, N.Y.) 2024, 17, 29. [Google Scholar] [CrossRef]
- Gibney, E.R.; Nolan, C.M. Epigenetics and gene expression. Heredity 2010, 105, 4–13. [Google Scholar] [CrossRef]
- Gijzen, M.; Kuflu, K.; Qutob, D.; Chernys, J.T. A class I chitinase from soybean seed coat. Journal of Experimental Botany 2001, 52, 2283–2289. [Google Scholar] [CrossRef] [PubMed]
- Gogolev, Y.V.; Ahmar, S.; Akpinar, B.A.; Budak, H.; Kiryushkin, A.S.; Gorshkov, V.Y.; Hensel, G.; et al. Omics, epigenetics, and genome editing techniques for food and nutritional security. Plants 2021, 10. [Google Scholar] [CrossRef]
- Graeber, K.; Linkies, A.; Steinbrecher, T.; Mummenhoff, K.; Tarkowská, D.; Turečková, V.; Ignatz, M.; et al. DELAY OF GERMINATION 1 mediates a conserved coat-dormancy mechanism for the temperature- and gibberellin-dependent control of seed germination. Proceedings of the National Academy of Sciences of the United States of America 2014, 111, E3571–80. [Google Scholar] [CrossRef]
- Gu, J.; Chen, J.; Zhao, C.; Hong, D. Mutating BnEOD1s via CRISPR-Cas9 increases the seed size and weight in Brassica napus. Molecular breeding : new strategies in plant improvement 2023, 43, 79. [Google Scholar] [CrossRef]
- Guo, J.; Qu, L.; Hu, Y.; Lu, W.; Lu, D. Proteomics reveals the effects of drought stress on the kernel development and starch formation of waxy maize. BMC Plant Biology 2021, 21, 434. [Google Scholar] [CrossRef] [PubMed]
- Gupta, C.; Salgotra, R.K. Epigenetics and its role in effecting agronomical traits. Frontiers in Plant Science 2022, 13, 925688. [Google Scholar] [CrossRef] [PubMed]
- Hacisalihoglu, G.; Armstrong, P. Crop Seed Phenomics: Focus on Non-Destructive Functional Trait Phenotyping Methods and Applications. Plants 2023, 12. [Google Scholar] [CrossRef] [PubMed]
- Hamdan, M.F.; Tan, B.C. Genetic modification techniques in plant breeding: A comparative review of CRISPR/Cas and GM technologies. Horticultural Plant Journal 2024. [Google Scholar] [CrossRef]
- Han, B.; Wu, D.; Zhang, Y.; Li, D.-Z.; Xu, W.; Liu, A. Epigenetic regulation of seed-specific gene expression by DNA methylation valleys in castor bean. BMC Biology 2022, 20, 57. [Google Scholar] [CrossRef]
- Han, Y.; Gao, S.; Muegge, K.; Zhang, W.; Zhou, B. Advanced applications of RNA sequencing and challenges. Bioinformatics and biology insights 2015, 9 (Suppl 1), 29–46. [Google Scholar] [CrossRef]
- Hatfield, J.L.; Prueger, J.H. Temperature extremes: Effect on plant growth and development. Weather and Climate Extremes 2015, 10, 4–10. [Google Scholar] [CrossRef]
- Hayden, H.L.; Savin, K.W.; Wadeson, J.; Gupta, V.V.S.R.; Mele, P.M. Comparative metatranscriptomics of wheat rhizosphere microbiomes in disease suppressive and non-suppressive soils for Rhizoctonia solani AG8. Frontiers in Microbiology 2018, 9, 859. [Google Scholar] [CrossRef]
- Hu, G.; Wei, Y.; Yuan, Y.; Jin, H.; Gu, X.; Xiang, D.; Wang, J.; et al. Transcriptome analysis reveals the nutrient metabolism processes during the germination of Longzi black barley. Agricultural Products Processing and Storage 2025, 1, 19. [Google Scholar] [CrossRef]
- Hu, H.; Wang, J.; Nie, S.; Zhao, J.; Batley, J.; Edwards, D. Plant pangenomics, current practice and future direction. Agriculture Communications 2024, 2, 100039. [Google Scholar] [CrossRef]
- Hu, Y.; Liu, Y.; Lu, L.; Tao, J.-J.; Cheng, T.; Jin, M.; Wang, Z.-Y.; et al. Global analysis of seed transcriptomes reveals a novel PLATZ regulator for seed size and weight control in soybean. The New Phytologist 2023, 240, 2436–2454. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Hua, W.; Huang, S.; Yang, H.; Zhan, G.; Wang, X.; Liu, G.; et al. Discovery of pod shatter-resistant associated SNPs by deep sequencing of a representative library followed by bulk segregant analysis in rapeseed. Plos One 2012, 7, e34253. [Google Scholar] [CrossRef]
- Huang, D.; Koh, C.; Feurtado, J.A.; Tsang, E.W.T.; Cutler, A.J. MicroRNAs and their putative targets in Brassica napus seed maturation. BMC Genomics 2013, 14, 140. [Google Scholar] [CrossRef]
- Huang, F.; Li, X.; Du, X.; Li, S.; Li, N.; Lü, Y.; Zou, S.; et al. SNP-based identification of QTLs for thousand-grain weight and related traits in wheat 8762/Keyi 5214 DH lines. Journal of integrative agriculture 2023, 22, 2949–2960. [Google Scholar] [CrossRef]
- Huang, X.; Wei, X.; Sang, T.; Zhao, Q.; Feng, Q.; Zhao, Y.; Li, C.; et al. Genome-wide association studies of 14 agronomic traits in rice landraces. Nature Genetics 2010, 42, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Hubert, B.; Leprince, O.; Buitink, J. Sleeping but not defenceless: seed dormancy and protection. Journal of Experimental Botany 2024, 75, 6110–6124. [Google Scholar] [CrossRef] [PubMed]
- Huo, H.; Wei, S.; Bradford, K.J. DELAY OF GERMINATION1 (DOG1) regulates both seed dormancy and flowering time through microRNA pathways. Proceedings of the National Academy of Sciences of the United States of America 2016, 113, E2199–206. [Google Scholar] [CrossRef]
- Hussain, S.; Rao, M.J.; Anjum, M.A.; Ejaz, S.; Zakir, I.; Ali, M.A.; Ahmad, N.; et al. (2019). Oxidative stress and antioxidant defense in plants under drought conditions. In M. Hasanuzzaman, K.R. Hakeem, K. Nahar, & H. F. Alharby (Eds.), Plant abiotic stress tolerance: agronomic, molecular and biotechnological approaches (pp. 207–219). Cham: Springer International Publishing.
- Hyten, D.L.; Song, Q.; Zhu, Y.; Choi, I.-Y.; Nelson, R.L.; Costa, J.M.; Specht, J.E.; et al. Impacts of genetic bottlenecks on soybean genome diversity. Proceedings of the National Academy of Sciences of the United States of America 2006, 103, 16666–16671. [Google Scholar] [CrossRef]
- International Rules for Seed Testing - International Seed Testing Association. (n.d.). . Retrieved December 12, 2024, from https://www.seedtest.org/en/publications/international-rules-seed-testing.html.
- Jain, R.; Dhaka, N.; Yadav, P.; Sharma, M.K.; Danish, M.; Sharma, S.; Kumari, S.; et al. Integrated analysis of transcriptomic and small RNA sequencing data provides miRNA candidates for engineering agronomically important seed traits in Brassica juncea. Current Plant Biology 1003, 36. [Google Scholar]
- Jha, U.C.; Nayyar, H.; Palakurthi, R.; Jha, R.; Valluri, V.; Bajaj, P.; Chitikineni, A.; et al. Major QTLs and Potential Candidate Genes for Heat Stress Tolerance Identified in Chickpea (Cicer arietinum L.). Frontiers in Plant Science 2021, 12, 655103. [Google Scholar] [CrossRef]
- Jia, J.; Wang, H.; Cai, Z.; Wei, R.; Huang, J.; Xia, Q.; Xiao, X.; et al. Identification and validation of stable and novel quantitative trait loci for pod shattering in soybean [Glycinemax (L.) Merr.]. Journal of integrative agriculture 2022, 21, 3169–3184. [Google Scholar] [CrossRef]
- Joseph Fernando, E.A.; Selvaraj, M.; Ghamkhar, K. The power of phenomics: Improving genebank value and utility. Molecular Plant 2023, 16, 1099–1101. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.J.; Kang, H. Expression and functional analyses of microRNA417 in Arabidopsis thaliana under stress conditions. Plant Physiology and Biochemistry 2007, 45, 805–811. [Google Scholar] [CrossRef]
- Kachhap, B.; Pandey, S.; Banerjee, M.; Pandey, A.K.; Kumari, N. Comprehensive Review of Seed-Borne Pathogens: Challenges and Control in Crop Production. Current Agriculture Research Journal 2025, 13, 427–442. [Google Scholar] [CrossRef]
- Kakoulidou, I.; Avramidou, E.V.; Baránek, M.; Brunel-Muguet, S.; Farrona, S.; Johannes, F.; Kaiserli, E.; et al. Epigenetics for crop improvement in times of global change. Biology 2021, 10. [Google Scholar] [CrossRef]
- Kalemba, E.M.; Dufour, S.; Gevaert, K.; Impens, F.; Meimoun, P. Proteomics- and metabolomics-based analysis of the regulation of germination in Norway maple and sycamore embryonic axes. Tree Physiology 2025, 45. [Google Scholar] [CrossRef]
- Kameswara Rao, N.; Dulloo, M.E.; Engels, J.M.M. A review of factors that influence the production of quality seed for long-term conservation in genebanks. Genetic resources and crop evolution 2017, 64, 1061–1074. [Google Scholar] [CrossRef]
- Kausar, R.; Wang, X.; Komatsu, S. Crop Proteomics under Abiotic Stress: From Data to Insights. Plants 2022, 11. [Google Scholar] [CrossRef]
- Kawakatsu, T.; Nery, J.R.; Castanon, R.; Ecker, J.R. Dynamic DNA methylation reconfiguration during seed development and germination. Genome Biology 2017, 18, 171. [Google Scholar] [CrossRef] [PubMed]
- Ke, J.; Wang, B.; Yoshikuni, Y. Microbiome engineering: synthetic biology of plant-associated microbiomes in sustainable agriculture. Trends in Biotechnology 2021, 39, 244–261. [Google Scholar] [CrossRef]
- Kedzierski, W.; PaweŁkiewicz, J. Effect of seed germination on levels of tRNA aminoacylation. Phytochemistry 1977, 16, 503–504. [Google Scholar] [CrossRef]
- Khajeh-Hosseini, M.; Powell, A.A.; Bingham, I.J. The interaction between salinity stress and seed vigour during germination of soyabean seeds. Seed Science and Technology 2003, 31, 715–725. [Google Scholar] [CrossRef]
- Khan, M.H.U.; Hu, L.; Zhu, M.; Zhai, Y.; Khan, S.U.; Ahmar, S.; Amoo, O.; et al. Targeted mutagenesis of EOD3 gene in Brassica napus L. regulates seed production. Journal of Cellular Physiology 2021, 236, 1996–2007. [Google Scholar] [CrossRef]
- Khoury, C.K.; Brush, S.; Costich, D.E.; Curry, H.A.; de Haan, S.; Engels, J.M.M.; Guarino, L.; et al. Crop genetic erosion: understanding and responding to loss of crop diversity. The New Phytologist 2022, 233, 84–118. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kwak, K.J.; Jung, H.J.; Lee, H.J.; Kang, H. MicroRNA402 affects seed germination of Arabidopsis thaliana under stress conditions via targeting DEMETER-LIKE Protein3 mRNA. Plant & Cell Physiology 2010, 51, 1079–1083. [Google Scholar]
- Kim, J.Y.; Lee, H.J.; Jung, H.J.; Maruyama, K.; Suzuki, N.; Kang, H. Overexpression of microRNA395c or 395e affects differently the seed germination of Arabidopsis thaliana under stress conditions. Planta 2010, 232, 1447–1454. [Google Scholar] [CrossRef] [PubMed]
- Krzyszton, M.; Yatusevich, R.; Wrona, M.; Sacharowski, S.P.; Adamska, D.; Swiezewski, S. Single seeds exhibit transcriptional heterogeneity during secondary dormancy induction. Plant Physiology 2022, 190, 211–225. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Das, S.P.; Choudhury, B.U.; Kumar, A.; Prakash, N.R.; Verma, R.; Chakraborti, M.; et al. Advances in genomic tools for plant breeding: harnessing DNA molecular markers, genomic selection, and genome editing. Biological research 2024, 57, 80. [Google Scholar] [CrossRef] [PubMed]
- Lahijanian, S.; Schmidt, J.; Feuerstein, U.; Polle, A. Effects of cover crops and microbial inoculants in different farming systems on soil microbial communities and yield of maize. Biology and Fertility of Soils 2025, 61, 1165–1182. [Google Scholar] [CrossRef]
- Laurençon, M.; Legrix, J.; Wagner, M.-H.; Demilly, D.; Baron, C.; Rolland, S.; Ducournau, S.; et al. Genomic and phenomic predictions help capture low-effect alleles promoting seed germination in oilseed rape in addition to QTL analyses. TAG. Theoretical and Applied Genetics. Theoretische und Angewandte Genetik 2024, 137, 156. [Google Scholar] [CrossRef]
- Li, D.; Wang, L.; Liu, X.; Cui, D.; Chen, T.; Zhang, H.; Jiang, C.; et al. Deep sequencing of maize small RNAs reveals a diverse set of microRNA in dry and imbibed seeds. Plos One 2013, 8, e55107. [Google Scholar] [CrossRef]
- Li Jianying Yuan, D.; Wang, P.; Wang, Q.; Sun, M.; Liu, Z.; Si, H.; et al. Cotton pan-genome retrieves the lost sequences and genes during domestication and selection. Genome Biology 2021, 22, 119. [Google Scholar] [CrossRef] [PubMed]
- Li Jing Cao, L.; Xie, Q.; Chen, G.; Hu, Z. Transcriptome sequencing and analysis during seed growth and development in tomato. Scientia horticulturae 2023, 310, 111763. [Google Scholar]
- Li, W.; Liu, J.; Zhang, H.; Liu, Z.; Wang, Y.; Xing, L.; He, Q.; et al. Plant pan-genomics: recent advances, new challenges, and roads ahead. Journal of Genetics and Genomics = Yi Chuan Xue Bao 2022, 49, 833–846. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Sheng, W.; Dong, Q.; Huang, R.; Dong, R.; Liu, G.; Ding, X.; et al. Analysis of seed production and seed shattering in a new artificial grassland forage: pigeon pea. Frontiers in Plant Science 2023, 14, 1146398. [Google Scholar] [CrossRef]
- Links, M.G.; Demeke, T.; Gräfenhan, T.; Hill, J.E.; Hemmingsen, S.M.; Dumonceaux, T.J. Simultaneous profiling of seed-associated bacteria and fungi reveals antagonistic interactions between microorganisms within a shared epiphytic microbiome on Triticum and Brassica seeds. The New Phytologist 2014, 202, 542–553. [Google Scholar] [CrossRef]
- Lipka, A.E.; Tian, F.; Wang, Q.; Peiffer, J.; Li, M.; Bradbury, P.J.; Gore, M.A.; et al. GAPIT: genome association and prediction integrated tool. Bioinformatics 2012, 28, 2397–2399. [Google Scholar] [CrossRef]
- Liu, M.; Li, Y.; Ma, Y.; Zhao, Q.; Stiller, J.; Feng, Q.; Tian, Q.; et al. The draft genome of a wild barley genotype reveals its enrichment in genes related to biotic and abiotic stresses compared to cultivated barley. Plant Biotechnology Journal 2020, 18, 443–456. [Google Scholar] [CrossRef]
- Liu, P.-P.; Montgomery, T.A.; Fahlgren, N.; Kasschau, K.D.; Nonogaki, H.; Carrington, J.C. Repression of AUXIN RESPONSE FACTOR10 by microRNA160 is critical for seed germination and post-germination stages. The Plant Journal 2007, 52, 133–146. [Google Scholar] [CrossRef]
- Liu, Y.; Du, H.; Li, P.; Shen, Y.; Peng, H.; Liu, S.; Zhou, G.-A.; et al. Pan-Genome of Wild and Cultivated Soybeans. Cell 2020, 182, 162–176.e13. [Google Scholar] [CrossRef]
- Long, Y.; Xia, W.; Li, R.; Wang, J.; Shao, M.; Feng, J.; King, G.J.; et al. Epigenetic QTL mapping in Brassica napus. Genetics 2011, 189, 1093–1102. [Google Scholar] [CrossRef] [PubMed]
- MacNish, T.R.; Al-Mamun, H.A.; Bayer, P.E.; McPhan, C.; Fernandez, C.G.T.; Upadhyaya, S.R.; Liu, S.; et al. Brassica Panache: A multi-species graph pangenome representing presence absence variation across forty-one Brassica genomes. The plant genome 2025, 18, e20535. [Google Scholar] [CrossRef]
- Martin, R.C.; Liu, P.-P.; Goloviznina, N.A.; Nonogaki, H. microRNA, seeds, and Darwin?: diverse function of miRNA in seed biology and plant responses to stress. Journal of Experimental Botany 2010, 61, 2229–2234. [Google Scholar] [CrossRef] [PubMed]
- Mathur, S.; Paritosh, K.; Tandon, R.; Pental, D.; Pradhan, A.K. Comparative Analysis of Seed Transcriptome and Coexpression Analysis Reveal Candidate Genes for Enhancing Seed Size/Weight in Brassica juncea. Frontiers in genetics 2022, 13, 814486. [Google Scholar] [CrossRef]
- Mercader, J. Mozambican grass seed consumption during the Middle Stone Age. Science 2009, 326, 1680–1683. [Google Scholar] [CrossRef]
- Mertz, E.T.; Bates, L.S.; Nelson, O.E. Mutant gene that changes protein composition and increases lysine content of maize endosperm. Science 1964, 145, 279–280. [Google Scholar] [CrossRef]
- Meyer, P. Epigenetic variation and environmental change. Journal of Experimental Botany 2015, 66, 3541–3548. [Google Scholar] [CrossRef]
- Miernyk, J.A.; Johnston, M.L. Proteomic analysis of the testa from developing soybean seeds. Journal of Proteomics 2013, 89, 265–272. [Google Scholar] [CrossRef]
- Mira, S.; Pirredda, M.; Martín-Sánchez, M.; Marchessi, J.E.; Martín, C. DNA methylation and integrity in aged seeds and regenerated plants. Seed science research 2020, 1–9. [Google Scholar] [CrossRef]
- Miyaji, N.; Fujimoto, R. (2018). Hybrid Vigor. Plant epigenetics coming of age for breeding applications, Advances in botanical research (Vol. 88, pp. 247–275). Elsevier.
- Mou, C.; Chen, Y.; Zhang, P.; Tong, Q.; Zhu, Z.; Ma, T.; Wang, P.; et al. Prolongation of seed viability and grain quality in rice by editing OsLOX1 using CRISPR/Cas9. Molecular breeding : new strategies in plant improvement 2024, 44, 72. [Google Scholar] [CrossRef] [PubMed]
- Nakabayashi, K.; Bartsch, M.; Xiang, Y.; Miatton, E.; Pellengahr, S.; Yano, R.; Seo, M.; et al. The time required for dormancy release in Arabidopsis is determined by DELAY OF GERMINATION1 protein levels in freshly harvested seeds. The Plant Cell 2012, 24, 2826–2838. [Google Scholar] [CrossRef]
- Nasar, S.; Muhammad, M.; Siddiqi, E.H.; Majeed, M.; Ameen, N.; Ullah, S.; Kousar, S.; et al. (2024). omics-based knowledge for achieving food and nutritional security. In S. Fiaz & C. S. Prakash (Eds.), Omics-based techniques for global food security (pp. 67–90). Wiley.
- Nelson, E.B. The seed microbiome: Origins, interactions, and impacts. Plant and soil 2018, 422, 7–34. [Google Scholar] [CrossRef]
- Nietzel, T.; Dudkina, N.V.; Haase, C.; Denolf, P.; Semchonok, D.A.; Boekema, E.J.; Braun, H.-P.; et al. The native structure and composition of the cruciferin complex in Brassica napus. The Journal of Biological Chemistry 2013, 288, 2238–2245. [Google Scholar] [CrossRef] [PubMed]
- Nonogaki, H. Seed dormancy and germination-emerging mechanisms and new hypotheses. Frontiers in Plant Science 2014, 5, 233. [Google Scholar] [CrossRef]
- Nouri, M.-Z.; Komatsu, S. Comparative analysis of soybean plasma membrane proteins under osmotic stress using gel-based and LC MS/MS-based proteomics approaches. Proteomics 2010, 10, 1930–1945. [Google Scholar] [CrossRef]
- Omar, G.; Saqer, M.M.; Adwan, G. Phylogenetic Relationship among Some species of the Genera Lens, Vicia, Lathyrus and Pisum ( Leguminosae ) in Palestine. Biology, Environmental Science 2019. [Google Scholar]
- Pagán, I. Transmission through seeds: The unknown life of plant viruses. PLoS Pathogens 2022, 18, e1010707. [Google Scholar] [CrossRef]
- Paulis, J.W.; Wall, J.S. Comparison of the protein compositions of selected corns and their wild relatives, teosinte and Tripsacum. Journal of Agricultural and Food Chemistry 1977, 25, 265–270. [Google Scholar] [CrossRef]
- Peixoto Araujo, N.M.; Arruda, H.S.; Dos Santos, F.N.; de Morais, D.R.; Pereira, G.A.; Pastore, G.M. LC-MS/MS screening and identification of bioactive compounds in leaves, pulp and seed from Eugenia calycina Cambess. Food research international (Ottawa, Ont.) 2020, 137, 109556. [Google Scholar] [CrossRef] [PubMed]
- Petereit, J.; Bayer, P.E.; Tay Fernandez, C.G.; Batley, J.; Edwards, D. Changes of gene content in four crop species during domestication and breeding. Agriculture Communications 2025, 3, 100077. [Google Scholar] [CrossRef]
- Pikaard, C.S.; Mittelsten Scheid, O. Epigenetic regulation in plants. Cold Spring Harbor Perspectives in Biology 2014, 6, a019315. [Google Scholar] [CrossRef]
- Podevin, N.; Davies, H.V.; Hartung, F.; Nogué, F.; Casacuberta, J.M. Site-directed nucleases: a paradigm shift in predictable, knowledge-based plant breeding. Trends in Biotechnology 2013, 31, 375–383. [Google Scholar] [CrossRef]
- Pourkheirandish, M.; Golicz, A.A.; Bhalla, P.L.; Singh, M.B. Global role of crop genomics in the face of climate change. Frontiers in Plant Science 2020, 11, 922. [Google Scholar] [CrossRef]
- Pourkheirandish, M.; Kanamori, H.; Wu, J.; Sakuma, S.; Blattner, F.R.; Komatsuda, T. Elucidation of the origin of “agriocrithon” based on domestication genes questions the hypothesis that Tibet is one of the centers of barley domestication. The Plant Journal 2018, 94, 525–534. [Google Scholar] [CrossRef]
- Preiner, J.; Steccari, I.; Oburger, E.; Wienkoop, S. Rhizobium symbiosis improves amino acid and secondary metabolite biosynthesis of tungsten-stressed soybean (Glycine max). Frontiers in Plant Science 2024, 15, 1355136. [Google Scholar] [CrossRef]
- Punia, A.; Kumari, M.; Chouhan, M.; Saini, V.; Joshi, R.; Kumar, A.; Kumar, R. Proteomic and metabolomic insights into seed germination of Ferula assa-foetida. Journal of Proteomics 2024, 300, 105176. [Google Scholar] [CrossRef]
- Qi, Q.; Hu, B.; Jiang, W.; Wang, Y.; Yan, J.; Ma, F.; Guan, Q.; et al. Advances in plant epigenome editing research and its application in plants. International Journal of Molecular Sciences 2023, 24. [Google Scholar] [CrossRef]
- Qi Xiantao Zhang, C.; Zhu, J.; Liu, C.; Huang, C.; Li, X.; Xie, C. Genome Editing Enables Next-Generation Hybrid Seed Production Technology. Molecular Plant 2020, 13, 1262–1269. [Google Scholar] [CrossRef]
- Qi Xinpeng Li, M.-W.; Xie, M.; Liu, X.; Ni, M.; Shao, G.; Song, C.; et al. Identification of a novel salt tolerance gene in wild soybean by whole-genome sequencing. Nature Communications 2014, 5, 4340. [Google Scholar] [CrossRef]
- Quezada-Martinez, D.; Addo Nyarko, C.P.; Schiessl, S.V.; Mason, A.S. Using wild relatives and related species to build climate resilience in Brassica crops. TAG. Theoretical and Applied Genetics. Theoretische und Angewandte Genetik 2021, 134, 1711–1728. [Google Scholar] [CrossRef]
- Rahaman, M.M.; Chen, D.; Gillani, Z.; Klukas, C.; Chen, M. Advanced phenotyping and phenotype data analysis for the study of plant growth and development. Frontiers in Plant Science 2015, 6, 619. [Google Scholar] [CrossRef] [PubMed]
- Rajpal, V.R.; Singh, A.; Kathpalia, R.; Thakur, R.K.; Khan, M.K.; Pandey, A.; Hamurcu, M.; et al. The Prospects of gene introgression from crop wild relatives into cultivated lentil for climate change mitigation. Frontiers in Plant Science 2023, 14, 1127239. [Google Scholar] [CrossRef] [PubMed]
- Raman, H.; Raman, R.; Kilian, A.; Detering, F.; Carling, J.; Coombes, N.; Diffey, S.; et al. Genome-wide delineation of natural variation for pod shatter resistance in Brassica napus. Plos One 2014, 9, e101673. [Google Scholar] [CrossRef]
- Raman, H.; Raman, R.; Sharma, N.; Cui, X.; McVittie, B.; Qiu, Y.; Zhang, Y.; et al. Novel quantitative trait loci from an interspecific Brassica rapa derivative improve pod shatter resistance in Brassica napus. Frontiers in Plant Science 2023, 14, 1233996. [Google Scholar] [CrossRef]
- Ran, Z.; Li, Z.; Xiao, X.; Yan, C.; An, M.; Chen, J.; Tang, M. Extensive targeted metabolomics analysis reveals the identification of major metabolites, antioxidants, and disease-resistant active pharmaceutical components in Camellia tuberculata (Camellia L.) seeds. Scientific Reports 2024, 14, 8709. [Google Scholar] [CrossRef]
- Rayner, T.; Saalbach, G.; Vickers, M.; Paajanen, P.; Martins, C.; Wouters, R.H.M.; Chinoy, C.; et al. Rebalancing the seed proteome following deletion of vicilin-related genes in pea (Pisum sativum L.). Journal of Experimental Botany 2024. [Google Scholar] [CrossRef]
- Reeve, M.A.; Pollard, K.M. MALDI-TOF MS-based analysis of dried seed proteins immobilized on filter paper. Biology methods and protocols 2019, 4, bpz007. [Google Scholar] [CrossRef]
- Repetto, O.; Rogniaux, H.; Firnhaber, C.; Zuber, H.; Küster, H.; Larré, C.; Thompson, R.; et al. Exploring the nuclear proteome of Medicago truncatula at the switch towards seed filling. The Plant Journal 2008, 56, 398–410. [Google Scholar] [CrossRef]
- Rey, O.; Danchin, E.; Mirouze, M.; Loot, C.; Blanchet, S. Adaptation to Global Change: A Transposable Element-Epigenetics Perspective. Trends in Ecology & Evolution 2016, 31, 514–526. [Google Scholar]
- Reyes, J.L.; Chua, N.H. ABA induction of miR159 controls transcript levels of two MYB factors during Arabidopsis seed germination. The Plant Journal 2007, 49, 592–606. [Google Scholar] [CrossRef]
- Ribeiro, D.G.; Carmo, L.S.T.; Santos, I.R.; Almeida, R.F.; Silva, L.P.; Oliveira-Neto, O.B.; Scherwinski-Pereira, J.E.; et al. MALDI TOF MS-profiling: Applications for bacterial and plant sample differentiation and biological variability assessment. Journal of Proteomics 2020, 213, 103619. [Google Scholar] [CrossRef]
- Rodriguez, D.F.C.; Urban, M.O.; Santaella, M.; Gereda, J.M.; Contreras, A.D.; Wenzl, P. Using phenomics to identify and integrate traits of interest for better-performing common beans: A validation study on an interspecific hybrid and its Acutifolii parents. Frontiers in Plant Science 2022, 13, 1008666. [Google Scholar] [CrossRef]
- Ruperao, P.; Thirunavukkarasu, N.; Gandham, P.; Selvanayagam, S.; Govindaraj, M.; Nebie, B.; Manyasa, E.; et al. Sorghum Pan-Genome Explores the Functional Utility for Genomic-Assisted Breeding to Accelerate the Genetic Gain. Frontiers in Plant Science 2021, 12, 666342. [Google Scholar] [CrossRef] [PubMed]
- Saikkonen, K.; Young, C.A.; Helander, M.; Schardl, C.L. Endophytic Epichloë species and their grass hosts: from evolution to applications. Plant Molecular Biology 2016, 90, 665–675. [Google Scholar] [CrossRef]
- Sato, H.; Yamane, H. Histone modifications affecting plant dormancy and dormancy release: common regulatory effects on hormone metabolism. Journal of Experimental Botany 2024, 75, 6142–6158. [Google Scholar] [CrossRef] [PubMed]
- Schopfer, P.; Bajracharya, D.; Plachy, C. Control of Seed Germination by Abscisic Acid: I. Time Course of Action in Sinapis alba L. Plant Physiology 1979, 64, 822–827. [Google Scholar] [CrossRef]
- Shen, W.; Yao, X.; Ye, T.; Ma, S.; Liu, X.; Yin, X.; Wu, Y. Arabidopsis aspartic protease ASPG1 affects seed dormancy, seed longevity and seed germination. Plant & Cell Physiology 2018, 59, 1415–1431. [Google Scholar]
- Shi, L.; Cui, X.; Shen, Y. The roles of histone methylation in the regulation of abiotic stress responses in plants. Plant Stress 2024, 11, 100303. [Google Scholar] [CrossRef]
- Shu, K.; Liu, X.-D.; Xie, Q.; He, Z.-H. Two faces of one seed: hormonal regulation of dormancy and germination. Molecular Plant 2016, 9, 34–45. [Google Scholar] [CrossRef]
- Simon, S.A.; Meyers, B.C. Small RNA-mediated epigenetic modifications in plants. Current Opinion in Plant Biology 2011, 14, 148–155. [Google Scholar] [CrossRef]
- Singh, A.; Mazahar, S.; Chapadgaonkar, S.S.; Giri, P.; Shourie, A. Phyto-microbiome to mitigate abiotic stress in crop plants. Frontiers in Microbiology 2023, 14, 1210890. [Google Scholar] [CrossRef]
- So, K.K.Y.; Duncan, R.W. Breeding Canola (Brassica napus L.) for Protein in Feed and Food. Plants 2021, 10. [Google Scholar] [CrossRef]
- Solanki, M.K.; Abdelfattah, A.; Sadhasivam, S.; Zakin, V.; Wisniewski, M.; Droby, S.; Sionov, E. Analysis of Stored Wheat Grain-Associated Microbiota Reveals Biocontrol Activity among Microorganisms against Mycotoxigenic Fungi. Journal of fungi (Basel, Switzerland) 2021, 7. [Google Scholar] [CrossRef]
- Song, J.-M.; Guan, Z.; Hu, J.; Guo, C.; Yang, Z.; Wang, S.; Liu, D.; et al. Eight high-quality genomes reveal pan-genome architecture and ecotype differentiation of Brassica napus. Nature Plants 2020, 6, 34–45. [Google Scholar] [CrossRef]
- Song, Y.; Zhu, J. The roles of metabolic pathways in maintaining primary dormancy of Pinus koraiensis seeds. BMC Plant Biology 2019, 19, 550. [Google Scholar] [CrossRef] [PubMed]
- de Souza Vidigal, D.; Willems, L.; van Arkel, J.; Dekkers, B.J.W.; Hilhorst, H.W.M.; Bentsink, L. Galactinol as marker for seed longevity. Plant Science 2016, 246, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Subedi, U.; Jayawardhane, K.N.; Pan, X.; Ozga, J.; Chen, G.; Foroud, N.A.; Singer, S.D. The potential of genome editing for improving seed oil content and fatty acid composition in oilseed crops. Lipids 2020, 55, 495–512. [Google Scholar] [CrossRef]
- Suman, J.; Rakshit, A.; Ogireddy, S.D.; Singh, S.; Gupta, C.; Chandrakala, J. Microbiome as a key player in sustainable agriculture and human health. Frontiers in Soil Science 2022, 2. [Google Scholar] [CrossRef]
- Sun, Z.; Adeleke, B.S.; Shi, Y.; Li, C. The seed microbiomes of staple food crops. Microbial biotechnology 2023, 16, 2236–2249. [Google Scholar] [CrossRef]
- Takehisa, H.; Sato, Y. Transcriptome monitoring visualizes growth stage-dependent nutrient status dynamics in rice under field conditions. The Plant Journal 2019, 97, 1048–1060. [Google Scholar] [CrossRef]
- Takehisa, H.; Sato, Y. Transcriptome-based approaches for clarification of nutritional responses and improvement of crop production. Breeding science 2021, 71, 76–88. [Google Scholar] [CrossRef]
- Tanaka, N.; Shenton, M.; Kawahara, Y.; Kumagai, M.; Sakai, H.; Kanamori, H.; Yonemaru, J.; et al. Whole-Genome Sequencing of the NARO World Rice Core Collection (WRC) as the Basis for Diversity and Association Studies. Plant & Cell Physiology 2020, 61, 922–932. [Google Scholar]
- Tang, K.; Lang, Z.; Zhang, H.; Zhu, J.-K. The DNA demethylase ROS1 targets genomic regions with distinct chromatin modifications. Nature Plants 2016, 2, 16169. [Google Scholar] [CrossRef]
- Tao, Y.; Zhao, X.; Mace, E.; Henry, R.; Jordan, D. Exploring and Exploiting Pan-genomics for Crop Improvement. Molecular Plant 2019, 12, 156–169. [Google Scholar] [CrossRef]
- Taylor, A.G. (2003). SEED DEVELOPMENT | seed quality. Encyclopedia of applied plant sciences (pp. 1285–1291). Elsevier.
- Terskikh, V.; Kermode, A.R. In vivo nuclear magnetic resonance metabolite profiling in plant seeds. Methods in Molecular Biology 2011, 773, 307–318. [Google Scholar]
- Thenveettil, N.; Bheemanahalli, R.; Reddy, K.N.; Gao, W.; Reddy, K.R. Temperature and elevated CO2 alter soybean seed yield and quality, exhibiting transgenerational effects on seedling emergence and vigor. Frontiers in Plant Science 2024, 15, 1427086. [Google Scholar] [CrossRef]
- Thines, M. Phylogeny and evolution of plant pathogenic oomycetes—a global overview. European Journal of Plant Pathology 2014, 138, 431–447. [Google Scholar] [CrossRef]
- Tonosaki, K.; Fujimoto, R.; Dennis, E.S.; Raboy, V.; Osabe, K. Will epigenetics be a key player in crop breeding? Frontiers in Plant Science 2022, 13, 958350. [Google Scholar] [CrossRef]
- Tripathi, N.; Khare, D. Molecular approaches for genetic improvement of seed quality and characterization of genetic diversity in soybean: a critical review. Biotechnology Letters 2016, 38, 1645–1654. [Google Scholar] [CrossRef] [PubMed]
- Varunjikar, M.S.; Bøhn, T.; Sanden, M.; Belghit, I.; Pineda-Pampliega, J.; Palmblad, M.; Broll, H.; et al. Proteomics analyses of herbicide-tolerant genetically modified, conventionally, and organically farmed soybean seeds. Food control 2023, 151, 109795. [Google Scholar] [CrossRef]
- Vidigal, D.S.; Marques, A.C.S.S.; Willems, L.A.J.; Buijs, G.; Méndez-Vigo, B.; Hilhorst, H.W.M.; Bentsink, L.; et al. Altitudinal and climatic associations of seed dormancy and flowering traits evidence adaptation of annual life cycle timing in Arabidopsis thaliana. Plant, Cell & Environment 2016, 39, 1737–1748. [Google Scholar] [CrossRef]
- Vikram, P.; Kumar, A.; Singh, A.; Singh, N.K. (2012). Rice: genomics-assisted breeding for drought tolerance. In N. Tuteja, S.S. Gill, A.F. Tiburcio, & R. Tuteja (Eds.), Improving crop resistance to abiotic stress (pp. 715–731). Wiley.
- Vincent, H.; Wiersema, J.; Kell, S.; Fielder, H.; Dobbie, S.; Castañeda-Álvarez, N.P.; Guarino, L.; et al. A prioritized crop wild relative inventory to help underpin global food security. Biological Conservation 2013, 167, 265–275. [Google Scholar] [CrossRef]
- Wang, W.; Pan, Q.; Tian, B.; He, F.; Chen, Y.; Bai, G.; Akhunova, A.; et al. Gene editing of the wheat homologs of TONNEAU1-recruiting motif encoding gene affects grain shape and weight in wheat. The Plant Journal 2019, 100, 251–264. [Google Scholar] [CrossRef]
- Wang, W.; Simmonds, J.; Pan, Q.; Davidson, D.; He, F.; Battal, A.; Akhunova, A.; et al. Gene editing and mutagenesis reveal inter-cultivar differences and additivity in the contribution of TaGW2 homoeologues to grain size and weight in wheat. TAG. Theoretical and Applied Genetics. Theoretische und Angewandte Genetik 2018, 131, 2463–2475. [Google Scholar] [CrossRef]
- Wang Xingqiang Zhao, Z.; Li, H.; Wei, Y.; Hu, J.; Yang, H.; Zhou, Y.; et al. The growth-promoting and disease-suppressing mechanisms of Trichoderma inoculation on peanut seedlings. Frontiers in Plant Science 2024, 15, 1414193. [Google Scholar] [CrossRef] [PubMed]
- Wang Xuemeng Zhang, H.; Song, R.; Sun, M.; Liu, P.; Tian, P.; Mao, P.; et al. Multiple omics datasets reveal significant physical and physiological dormancy in alfalfa hard seeds identified by multispectral imaging analysis. The Crop journal 2023, 11, 1458–1468. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, Y. Research on multilateral collaboration strategies in agricultural seed quality assurance. Scientific Reports 2024, 14, 11310. [Google Scholar] [CrossRef]
- Welbaum, G.E.; Bradford, K.J. Water Relations of Seed Development and Germination in Muskmelon (Cucumis melo L.) : V. Water Relations of Imbibition and Germination. Plant Physiology 1990, 92, 1046–1052. [Google Scholar] [CrossRef]
- West, J.S.; Kharbanda, P.D.; Barbetti, M.J.; Fitt, B.D.L. Epidemiology and management ofLeptosphaeria maculans (phoma stem canker) on oilseed rape in Australia, Canada and Europe. Plant pathology 2001, 50, 10–27. [Google Scholar] [CrossRef]
- von Wettberg, E.J.B.; Chang, P.L.; Başdemir, F.; Carrasquila-Garcia, N.; Korbu, L.B.; Moenga, S.M.; Bedada, G.; et al. Ecology and genomics of an important crop wild relative as a prelude to agricultural innovation. Nature Communications 2018, 9, 649. [Google Scholar] [CrossRef] [PubMed]
- Willmann, M.R.; Mehalick, A.J.; Packer, R.L.; Jenik, P.D. MicroRNAs regulate the timing of embryo maturation in Arabidopsis. Plant Physiology 2011, 155, 1871–1884. [Google Scholar] [CrossRef]
- Wu, G.; Park, M.Y.; Conway, S.R.; Wang, J.-W.; Weigel, D.; Poethig, R.S. The sequential action of miR156 and miR172 regulates developmental timing in Arabidopsis. Cell 2009, 138, 750–759. [Google Scholar] [CrossRef]
- Xie, M.; Chung, C.Y.-L.; Li, M.-W.; Wong, F.-L.; Wang, X.; Liu, A.; Wang, Z.; et al. A reference-grade wild soybean genome. Nature Communications 2019, 10, 1216. [Google Scholar] [CrossRef] [PubMed]
- Yacoubi, R.; Job, C.; Belghazi, M.; Chaibi, W.; Job, D. Proteomic analysis of the enhancement of seed vigour in osmoprimed alfalfa seeds germinated under salinity stress. Seed science research 2013, 23, 99–110. [Google Scholar] [CrossRef]
- Yang, Q.-X.; Chen, D.; Zhao, Y.; Zhang, X.-Y.; Zhao, M.; Peng, R.; Sun, N.-X.; et al. RNA-seq analysis reveals key genes associated with seed germination of Fritillaria taipaiensis P.Y.Li by cold stratification. Frontiers in Plant Science 2022, 13, 1021572. [Google Scholar] [CrossRef]
- Yang, W.; Feng, H.; Zhang, X.; Zhang, J.; Doonan, J.H.; Batchelor, W.D.; Xiong, L.; et al. Crop Phenomics and High-Throughput Phenotyping: Past Decades, Current Challenges, and Future Perspectives. Molecular Plant 2020, 13, 187–214. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Gao, P.; Song, J.; Yang, H.; Qin, L.; Yu, X.; Song, H.; et al. Spatiotemporal transcriptomics and metabolic profiling provide insights into gene regulatory networks during lentil seed development. The Plant Journal 2023, 115, 253–274. [Google Scholar] [CrossRef]
- Zhai, Y.; Cai, S.; Hu, L.; Yang, Y.; Amoo, O.; Fan, C.; Zhou, Y. CRISPR/Cas9-mediated genome editing reveals differences in the contribution of INDEHISCENT homologues to pod shatter resistance in Brassica napus L. TAG. Theoretical and Applied Genetics. Theoretische und Angewandte Genetik 2019, 132, 2111–2123. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, L.; Li, H.; He, J.; Chao, H.; Yan, S.; Yin, Y.; et al. 3D genome structural variations play important roles in regulating seed oil content of Brassica napus. Plant communications 2024, 5, 100666. [Google Scholar] [CrossRef]
- Zhang, X.; Sun, J.; Cao, X.; Song, X. Epigenetic mutation of RAV6 affects leaf angle and seed size in rice. Plant Physiology 2015, 169, 2118–2128. [Google Scholar] [CrossRef]
- Zhang Yanjie Liang, W.; Shi, J.; Xu, J.; Zhang, D. MYB56 encoding a R2R3 MYB transcription factor regulates seed size in Arabidopsis thaliana. Journal of Integrative Plant Biology 2013, 55, 1166–1178. [Google Scholar] [CrossRef] [PubMed]
- Zhang Yuting Zhang, H.; Zhao, H.; Xia, Y.; Zheng, X.; Fan, R.; Tan, Z.; et al. Multi-omics analysis dissects the genetic architecture of seed coat content in Brassica napus. Genome Biology 2022, 23, 86. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Zhang, Y.; Du, J.; Guo, X.; Wen, W.; Gu, S.; Wang, J.; et al. Crop phenomics: current status and perspectives. Frontiers in Plant Science 2019, 10, 714. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Bayer, P.E.; Ruperao, P.; Saxena, R.K.; Khan, A.W.; Golicz, A.A.; Nguyen, H.T.; et al. Trait associations in the pangenome of pigeon pea (Cajanus cajan). Plant Biotechnology Journal 2020, 18, 1946–1954. [Google Scholar] [CrossRef]
- Zhou, B.-J.; Li, J.; Ma, C.-L.; Wang, Y.-J.; Zhang, J.-L.; Chen, H.-H.; Lao, Q.-X.; et al. Metabolomics analysis of the nutraceutical diversity and physiological quality of Torreya yunnanensis seeds during cold storage. Plant Physiology and Biochemistry 2024, 206, 108183. [Google Scholar] [CrossRef]
- Zsögön, A.; Čermák, T.; Naves, E.R.; Notini, M.M.; Edel, K.H.; Weinl, S.; Freschi, L.; et al. De novo domestication of wild tomato using genome editing. Nature Biotechnology 2018. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



