Submitted:
17 September 2025
Posted:
19 September 2025
You are already at the latest version
Abstract
Keywords:
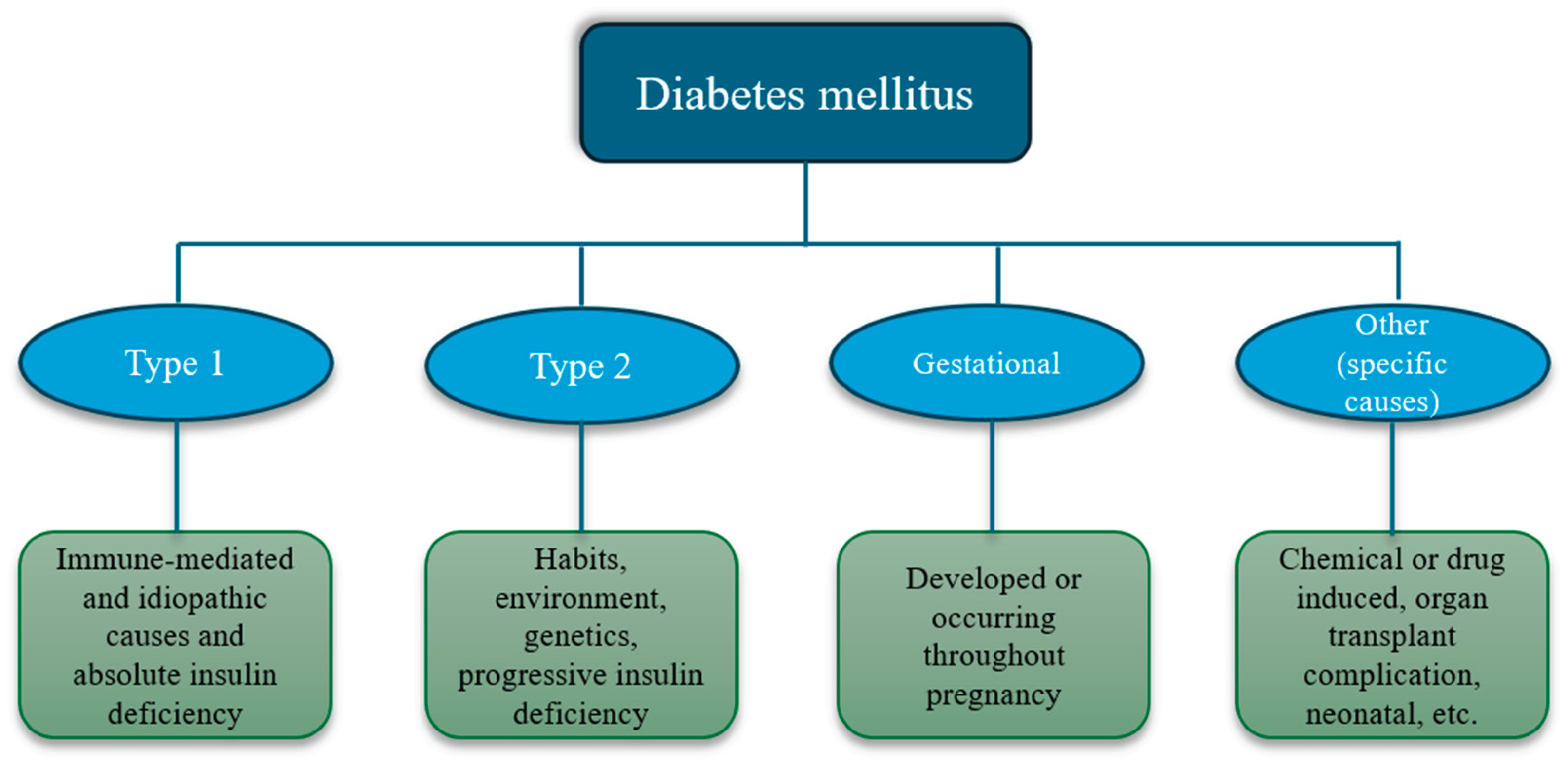
1. Introduction
2. Polyphenols
4. Microencapsulation
4.1. Physical Methods
4.1.1. Spray Drying
4.1.2. Freeze-Drying
4.1.3. Extrusion
4.2. Physico-Chemical Methods
4.2.1. Liposomes
4.2.2. Coacervation
4.2.3. Co-Crystallization
4.2.4. Ionic Gelation
5. Nanoencapsulation
6. Current Evidence Regarding the Efficacy of Encapsulated Polyphenols
6.1. In Vitro
6.2. In Vivo
7. ADMET Analysis of Polyphenols with Antidiabetic Properties
| Molecule | Class of compound | PubChem CID | Chemical Formula | Molecular Weight | H Bond donors | H Bond acceptor | Log p* | Lipinski Rule of 5 | |
| Cyanidin 3-glucoside | Anthocyanin | 197081 | C21H21ClO11 | 484.8 | 8 | 11 | -1.5 | No | |
| Curcumin | Curcuminoids | 969516 | C21H20O6 | 368.4 | 2 | 6 | 3.2 | Yes | |
| (+)-Catechin | Flavanol | 9064 | C15H14O6 | 290.27 | 5 | 6 | 1.4 | Yes | |
| (-)-Epicatechin | Flavanol | 72276 | C15H14O6 | 290.27 | 5 | 6 | 1.8 | Yes | |
| Liquiritin | Flavanone | 503737 | C21H22O9 | 418.4 | 5 | 9 | 0.4 | Yes | |
| Naringenin | Flavanone | 439246 | C15H12O5 | 272.25 | 3 | 5 | 2.2 | Yes | |
| Chrysin | Flavone | 5281607 | C15H10O4 | 254.24 | 2 | 4 | 2.5 | Yes | |
| Hesperidin | Flavone | 10621 | C28H34O15 | 610.6 | 8 | 15 | -1.1 | No | |
| Luteolin | Flavone | 5280445 | C15H10O6 | 286.24 | 4 | 6 | 2.0 | Yes | |
| Myricetin | Flavonol | 5281672 | C15H10O8 | 318.23 | 6 | 8 | 1.6 | No | |
| Quercetin | Flavonol | 5280343 | C15H10O7 | 302.23 | 5 | 7 | 1.5 | Yes | |
| Mangiferin | Glucosylxanthone | 5281647 | C19H18O11 | 422.3 | 8 | 11 | -0.4 | No | |
| Benzoic acid | Hydroxybenzoic acid | 243 | C7H6O2 | 122.12 | 1 | 2 | 1.87 | Yes | |
| Hydroxybenzoic acid | Hydroxybenzoic acid | 135 | C7H6O3 | 138.12 | 2 | 3 | 1.58 | yes | |
| Gallic acid | Hydroxybenzoic acid | 370 | C7H6O5 | 170.12 | 4 | 5 | 0.7 | Yes | |
| Ferulic acid | Hydroxycinnamic acid | 445858 | C10H10O4 | 194.18 | 2 | 4 | 1.5 | Yes | |
| Cinnamic acid | Hydroxycinnamic acid | 444539 | C9H8O2 | 148.16 | 1 | 2 | 2.1 | Yes | |
| Caffeic acid | Hydroxycinnamic acid | 689043 | C9H8O4 | 180.16 | 3 | 4 | 1.2 | Yes | |
| Coumaric acid | Hydroxycinnamic acid | 637542 | C9H8O3 | 164.16 | 2 | 3 | 1.5 | Yes | |
| Rosmarinic acid | Hydroxycinnamic acid | 5281792 | C18H16O8 | 360.3 | 5 | 8 | 2.4 | Yes | |
| Resveratrol | Stilbene | 445154 | C14H12O3 | 228.24 | 3 | 3 | 3.1 | Yes | |
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ADA | American Diabetes Association |
| ADME | Absorption, distribution, metabolism, and excretion |
| ALP | Alkaline phosphate |
| ALT | Alanine aminotransferase |
| AST | Aspartate aminotransferase |
| BUN | Blood urea nitrogen |
| BW | Body weight |
| DOAJ | Directory of open access journals |
| DM | Diabetes mellitus |
| FPG | Fasting plasma glucose |
| GLUT4 | Insulin-regulated glucose transporter |
| Hb1A1c | Glycosylated hemoglobin |
| HDL | High density lipoproteins |
| HOMA-B | Homeostasis model assessment of β-cell function |
| HOMA-IR | Homeostasis model assessment-insulin resistance |
| IC50 | Inhibitory Concentration 50 |
| LUV | Unilamellar vesicles |
| LD | Linear dichroism |
| LDL | Low density lipoproteins |
| LMPH | Longzhua mushroom polysaccharide hydrogel |
| MDPI | Multidisciplinary Digital Publishing Institute |
| MLV | Multilamellar vesicles |
| NLCs | Nanostructured Lipid Carriers |
| NSC | N-succinylated chitosan |
| OGTT | Oral glucose tolerance test |
| PGA | polyglycolides |
| PLA | Polylactides |
| PLA-PEG | poly(lactide)-poly(ethylene glycol) |
| PLGA | DL-polylactide/glycolide copolymer |
| PLGA-PEG | poly(lactide-co-glycolide)-poly(ethylene glycol) |
| SLNs | Solid Lipid Nanoparticles |
| STZ | streptozotocin |
| TC | Total cholesterol |
| TG | Triglycerides |
| T2DM | Type 2 Diabetes mellitus |
| 2-hPG | 2-h Plasma glucose |
References
- Committee, A.D.A.P.P. 2. Diagnosis and Classification of Diabetes: Standards of Care in Diabetes—2024. 2023, 47, S20–S42. [Google Scholar] [CrossRef]
- Popoviciu, M.S.; Paduraru, L.; Nutas, R.M.; Ujoc, A.M.; Yahya, G.; Metwally, K.; Cavalu, S. Diabetes Mellitus Secondary to Endocrine Diseases: An Update of Diagnostic and Treatment Particularities. 2023, 24, 12676.
- Sharma, P.; Hajam, Y.A.; Kumar, R.; Rai, S. Complementary and alternative medicine for the treatment of diabetes and associated complications: A review on therapeutic role of polyphenols. 2022; 2, 100188. [Google Scholar] [CrossRef]
- Vlacho, B.; Rossell-Rusiñol, J.; Granado-Casas, M.; Mauricio, D.; Julve, J. Overview on chronic complications of diabetes mellitus. In Chronic Complications of Diabetes Mellitus; Elsevier: 2024; pp. 1-10.
- International Diabetes Federation. Diabetes Atlas. Available online: https://diabetesatlas.org/atlas-reports (accessed on.
- World Health Organization. Diabetes. Available online: https://www.who.int/health-topics/diabetes#tab=tab_1 (accessed on 1 August 2024).
- Chandrasekaran, P.; Weiskirchen, R. The Role of Obesity in Type 2 Diabetes Mellitus—An Overview. 2024, 25, 1882.
- Rohm, T.V.; Meier, D.T.; Olefsky, J.M.; Donath, M.Y. Inflammation in obesity, diabetes, and related disorders. 2022, 55, 31-55.
- Li, Y.; Schoufour, J.; Wang, D.D.; Dhana, K.; Pan, A.; Liu, X.; Song, M.; Liu, G.; Shin, H.J.; Sun, Q. Healthy lifestyle and life expectancy free of cancer, cardiovascular disease, and type 2 diabetes: prospective cohort study. 2020, 368.
- Lingvay, I.; Sumithran, P.; Cohen, R.V.; le Roux, C.W. Obesity management as a primary treatment goal for type 2 diabetes: time to reframe the conversation. 2022, 399, 394-405.
- Magkos, F.; Hjorth, M.F.; Astrup, A. Diet and exercise in the prevention and treatment of type 2 diabetes mellitus. 2020, 16, 545-555.
- Akbari, B.; Baghaei-Yazdi, N.; Bahmaie, M.; Mahdavi Abhari, F. The role of plant-derived natural antioxidants in reduction of oxidative stress. 2022, 48, 611-633.
- Pasupuleti, V.R.; Arigela, C.S.; Gan, S.H.; Salam, S.K.N.; Krishnan, K.T.; Rahman, N.A.; Jeffree, M.S. A review on oxidative stress, diabetic complications, and the roles of honey polyphenols. 2020, 2020, 8878172.
- Pérez-Torres, I.; Castrejón-Téllez, V.; Soto, M.E.; Rubio-Ruiz, M.E.; Manzano-Pech, L.; Guarner-Lans, V. Oxidative stress, plant natural antioxidants, and obesity. 2021, 22, 1786.
- Unuofin, J.O.; Lebelo, S.L. Antioxidant effects and mechanisms of medicinal plants and their bioactive compounds for the prevention and treatment of type 2 diabetes: an updated review. 2020, 2020, 1356893.
- Arkoub-Djermoune, L.; Boulekbache-Makhlouf, L.; Zeghichi-Hamri, S.; Bellili, S.; Boukhalfa, F.; Madani, K. Influence of the Thermal Processing on the Physico-Chemical Properties and the Antioxidant Activity of A Solanaceae Vegetable: Eggplant. 2016; 39, 181–191. [Google Scholar] [CrossRef]
- Aryal, D.; Joshi, S.; Thapa, N.K.; Chaudhary, P.; Basaula, S.; Joshi, U.; Bhandari, D.; Rogers, H.M.; Bhattarai, S.; Sharma, K.R.; et al. Dietary phenolic compounds as promising therapeutic agents for diabetes and its complications: A comprehensive review. 2024; 12, 3025–3045. [Google Scholar] [CrossRef]
- Naz, R.; Saqib, F.; Awadallah, S.; Wahid, M.; Latif, M.F.; Iqbal, I.; Mubarak, M.S. Food Polyphenols and Type II Diabetes Mellitus: Pharmacology and Mechanisms. 2023, 28, 3996.
- Wang, Y.; Alkhalidy, H.; Liu, D. The Emerging Role of Polyphenols in the Management of Type 2 Diabetes. 2021, 26, 703.
- Durazzo, A.; Lucarini, M.; Souto, E.B.; Cicala, C.; Caiazzo, E.; Izzo, A.A.; Novellino, E.; Santini, A. Polyphenols: A concise overview on the chemistry, occurrence, and human health. 2019; 33, 2221–2243. [Google Scholar] [CrossRef]
- Sánchez Arribas, A.; Moreno, M.; Moreno, G.A.; Bermejo, E.; Zapardiel, A.; Chicharro, M. Characterization of White Wines by Electrochemical Indexes Obtained Using Carbon Nanotube-modified Electrodes. 2018; 30, 1461–1471. [Google Scholar] [CrossRef]
- Barbosa, S.; Pardo-Mates, N.; Hidalgo-Serrano, M.; Saurina, J.; Puignou, L.; Núñez, O. Detection and Quantitation of Frauds in the Authentication of Cranberry-Based Extracts by UHPLC-HRMS (Orbitrap) Polyphenolic Profiling and Multivariate Calibration Methods. 2018; 66, 9353–9365. [Google Scholar] [CrossRef]
- Brinsi, C.; Jedidi, S.; Sammari, H.; Selmi, H.; Sebai, H. Antidiarrheal, anti-inflammatory and antioxidant effects of Anethum graveolens L. fruit extract on castor oil-induced diarrhea in rats. 2024; e14892. [Google Scholar] [CrossRef]
- Arslaner, A.; Türkoğlu, Z. A potential antiviral and food-derived healty ingredient: Resveratrol. 2021; 7. [Google Scholar] [CrossRef]
- Santos Sánchez, N.; Salas-Coronado, R.; Villanueva, C.; Hernandez-Carlos, B. Antioxidant Compounds and Their Antioxidant Mechanism. 2019.
- Xuan Hoan, N.; Anh, L.; Quan, D.; Cuong, D.; Thai Ha, H.; Minh, N.; Hieu, D.; Thuat, N.; Pham, T.; Tuyen, D. Functional-Antioxidant Food. 2021.
- Pinna, N.; Ben Abbou, S.; Ianni, F.; Angeles Flores, G.; Pietercelie, A.; Perretti, G.; Blasi, F.; Angelini, P.; Cossignani, L. Phenolic compounds from pumpkin pulp: Extraction optimization and biological properties. 2024; 23, 101628. [Google Scholar] [CrossRef]
- Upadhyaya, B.; Moreau, R.; Majumder, K. Antioxidant and Anti-Inflammatory Capacities of Three Dry Bean Varieties after Cooking and In Vitro Gastrointestinal Digestion. 2024. [Google Scholar] [CrossRef]
- Marzagalli, M.; Battaglia, S.; Raimondi, M.; Fontana, F.; Cozzi, M.; Ranieri, F.R.; Sacchi, R.; Curti, V.; Limonta, P. Anti-Inflammatory and Antioxidant Properties of a New Mixture of Vitamin C, Collagen Peptides, Resveratrol, and Astaxanthin in Tenocytes: Molecular Basis for Future Applications in Tendinopathies. 2024; 2024, 5273198. [Google Scholar] [CrossRef]
- Çetin, R.; Bahadir, S.; Basar, İ.; Aslanoglu, B.; Atlas, B.; Kaya, S.; Güzel, B.C.; Turan, Y. Neuroprotective effects of the combined treatment of resveratrol and urapidil in experimental cerebral ischemia-reperfusion injury in rats. 2024; 39, e395329. [Google Scholar] [CrossRef]
- Özturk, N.; Ceylan, H.; Demir, Y. The hepatoprotective potential of tannic acid against doxorubicin-induced hepatotoxicity: Insights into its antioxidative, anti-inflammatory, and antiapoptotic mechanisms. 2024; 38, e23798. [Google Scholar] [CrossRef]
- Wu, Z.; Shen, J.; Xu, Q.; Xiang, Q.; Chen, Y.; Lv, L.; Zheng, B.; Wang, Q.; Wang, S.; Li, L. Epigallocatechin-3-Gallate Improves Intestinal Gut Microbiota Homeostasis and Ameliorates Clostridioides difficile Infection. 2022; 14. [Google Scholar] [CrossRef]
- Zhao, X.; Guo, J.; Wang, Y.; Yi, X. High-tannin food enhances spatial memory and scatter-hoarding in rodents via the microbiota-gut-brain axis. 2024; 12, 140. [Google Scholar] [CrossRef]
- Tresserra-Rimbau, A.; Castro-Barquero, S.; Vitelli-Storelli, F.; Becerra-Tomas, N.; Vázquez-Ruiz, Z.; Díaz-López, A.; Corella, D.; Castañer, O.; Romaguera, D.; Vioque, J.; et al. Associations between Dietary Polyphenols and Type 2 Diabetes in a Cross-Sectional Analysis of the PREDIMED-Plus Trial: Role of Body Mass Index and Sex. 2019; 8. [Google Scholar] [CrossRef]
- Liao, M.; Wang, X. Ameliorating effect of Chinese jujube polyphenol on blood glucose oxidative stress in type 2 diabetic rats. 2024; 38, 108804. [Google Scholar] [CrossRef]
- Choockong, C.; Itharat, A.; Pipatrattanaseree, W.; Ninlaor, T.; Piwngam, K.; Intharit, N.; Sukkhum, S.; Davies, N.M. The most commonly used spices in Thai traditional medicine: in vitro evaluation of anti-hyperglycemic, antioxidant, polyphenol content, and nitric oxide production inhibitory activities. 2024; 19, 13–28. [Google Scholar] [CrossRef]
- Lee, S.M.; Park, S.Y.; Kim, J.Y. Comparative evaluation of the antihyperglycemic effects of three extracts of sea mustard (Undaria pinnatifida): In vitro and in vivo studies. 2024; 190, 114623. [Google Scholar] [CrossRef]
- Liu, C.; Zeng, H.; Cui, W.; Ouyang, J.; Zhou, F.; Wen, S.; Fang, W.; Zhang, S.; Huang, J.; Liu, Z. Theaflavins mitigate diabetic symptoms in GK rats by modulating the INSR/PI3K-Akt/GSK-3 pathway and intestinal microbiota. [CrossRef]
- Quetglas-Llabrés, M.M.; Monserrat-Mesquida, M.; Bouzas, C.; García, S.; Mateos, D.; Ugarriza, L.; Gómez, C.; Sureda, A.; Tur, J.A. Long-Term Impact of Nutritional Intervention with Increased Polyphenol Intake and Physical Activity Promotion on Oxidative and Inflammatory Profiles in Patients with Metabolic Syndrome. [CrossRef]
- Bohn, T. Dietary factors affecting polyphenol bioavailability. 2014, 72, 429-452.
- Castro-López, C.; Espinoza-González, C.; Ramos-González, R.; Boone-Villa, V.D.; Aguilar-González, M.A.; Martínez-Ávila, G.C.G.; Aguilar, C.N.; Ventura-Sobrevilla, J.M. Spray-drying encapsulation of microwave-assisted extracted polyphenols from Moringa oleifera: Influence of tragacanth, locust bean, and carboxymethyl-cellulose formulations. [CrossRef]
- Costa, M.; Sezgin-Bayindir, Z.; Losada-Barreiro, S.; Paiva-Martins, F.; Saso, L.; Bravo-Díaz, C. Polyphenols as Antioxidants for Extending Food Shelf-Life and in the Prevention of Health Diseases: Encapsulation and Interfacial Phenomena. [CrossRef]
- Gerloff, T. Impact of genetic polymorphisms in transmembrane carrier-systems on drug and xenobiotic distribution. 2004, 369, 69-77.
- Lampe, J.W. Interindividual differences in response to plant-based diets: implications for cancer risk. 2009, 89, 1553S-1557S.
- Hidalgo-Liberona, N.; González-Domínguez, R.; Vegas, E.; Riso, P.; Del Bo’, C.; Bernardi, S.; Peron, G.; Guglielmetti, S.; Gargari, G.; Kroon, P.A. Increased intestinal permeability in older subjects impacts the beneficial effects of dietary polyphenols by modulating their bioavailability. 2020, 68, 12476-12484.
- Margalef, M.; Pons, Z.; Iglesias-Carres, L.; Bravo, F.I.; Muguerza, B.; Arola-Arnal, A. Flavanol plasma bioavailability is affected by metabolic syndrome in rats. 2017, 231, 287-294.
- Zhang, H.; Yu, D.; Sun, J.; Liu, X.; Jiang, L.; Guo, H.; Ren, F. Interaction of plant phenols with food macronutrients: characterisation and nutritional–physiological consequences. 2014, 27, 1-15.
- Iqbal, Y.; Cottrell, J.J.; Suleria, H.A.; Dunshea, F.R. Gut microbiota-polyphenol interactions in chicken: A review. 2020, 10, 1391.
- Li, D.; Yang, Y.; Yang, X.; Wang, Z.; Yao, X.; Guo, Y. Enhanced bioavailability and anti-hyperglycemic activity of young apple polyphenols by complexation with whey protein isolates. 2022, 87, 1257-1267.
- Pasinetti, G.M.; Singh, R.; Westfall, S.; Herman, F.; Faith, J.; Ho, L. The role of the gut microbiota in the metabolism of polyphenols as characterized by gnotobiotic mice. 2018, 63, 409-421.
- Wang, Q.; Xu, K.; Cai, X.; Wang, C.; Cao, Y.; Xiao, J. Rosmarinic acid restores colonic mucus secretion in colitis mice by regulating gut microbiota-derived metabolites and the activation of inflammasomes. 2023, 71, 4571-4585.
- Zhou, N.; Gu, X.; Zhuang, T.; Xu, Y.; Yang, L.; Zhou, M. Gut microbiota: A pivotal hub for polyphenols as antidepressants. 2020, 68, 6007-6020.
- Corrêa, T.A.F.; Rogero, M.M.; Hassimotto, N.M.A.; Lajolo, F.M. The Two-Way Polyphenols-Microbiota Interactions and Their Effects on Obesity and Related Metabolic Diseases. [CrossRef]
- Shivashankara, K.; Acharya, S. Bioavailability of dietary polyphenols and the cardiovascular diseases. 2010, 3.
- Li, M.; Zheng, Y.; Zhao, J.; Liu, M.; Shu, X.; Li, Q.; Wang, Y.; Zhou, Y. Polyphenol mechanisms against gastric cancer and their interactions with gut microbiota: a review. 2022, 29, 5247-5261.
- Lila, M.A.; Hoskin, R.T.; Grace, M.H.; Xiong, J.; Strauch, R.; Ferruzzi, M.; Iorizzo, M.; Kay, C. Boosting the bioaccessibility of dietary bioactives by delivery as protein–polyphenol aggregate particles. 2022, 70, 13017-13026.
- Chen, L.; Gnanaraj, C.; Arulselvan, P.; El-Seedi, H.; Teng, H. A review on advanced microencapsulation technology to enhance bioavailability of phenolic compounds: Based on its activity in the treatment of Type 2 Diabetes. [CrossRef]
- Frakolaki, G.; Giannou, V.; Kekos, D.; Tzia, C. A review of the microencapsulation techniques for the incorporation of probiotic bacteria in functional foods. [CrossRef]
- Arenas-Jal, M.; Suñé-Negre, J.M.; García-Montoya, E. An overview of microencapsulation in the food industry: opportunities, challenges, and innovations. [CrossRef]
- Mohammadalinejhad, S.; Kurek, M.A. Microencapsulation of anthocyanins-critical review of techniques and wall materials. [CrossRef]
- Peanparkdee, M.; Iwamoto, S.; Yamauchi, R. Microencapsulation: a review of applications in the food and pharmaceutical industries. 2016, 4, 56-65.
- Mehta, N.; Kumar, P.; Verma, A.K.; Umaraw, P.; Kumar, Y.; Malav, O.P.; Sazili, A.Q.; Domínguez, R.; Lorenzo, J.M. Microencapsulation as a Noble Technique for the Application of Bioactive Compounds in the Food Industry: A Comprehensive Review. [CrossRef]
- Leyva-Jiménez, F.J.; Lozano-Sánchez, J.; de la Luz Cádiz-Gurrea, M.; Fernández-Ochoa, Á.; Arráez-Román, D.; Segura-Carretero, A. Spray-drying microencapsulation of bioactive compounds from lemon verbena green extract. [CrossRef]
- Santos, S.S.d.; Rodrigues, L.M.; da Costa, S.C.; Bergamasco, R.d.C.; Madrona, G.S. Microencapsulation of bioactive compounds from blackberry pomace (Rubus fruticosus) by spray drying technique. 2017, 13, 20170047.
- Navarro-Flores, M.J.; Ventura-Canseco, L.M.C.; Meza-Gordillo, R.; Ayora-Talavera, T.R.; Abud-Archila, M. Spray drying encapsulation of a native plant extract rich in phenolic compounds with combinations of maltodextrin and non-conventional wall materials. [CrossRef]
- Ribeiro, A.M.; Estevinho, B.N.; Rocha, F. Spray Drying Encapsulation of Elderberry Extract and Evaluating the Release and Stability of Phenolic Compounds in Encapsulated Powders. [CrossRef]
- Ferreira, L.M.D.M.C.; Pereira, R.R.; Carvalho-Guimarães, F.B.D.; Remígio, M.S.D.N.; Barbosa, W.L.R.; Ribeiro-Costa, R.M.; Silva-Júnior, J.O.C. Microencapsulation by Spray Drying and Antioxidant Activity of Phenolic Compounds from Tucuma Coproduct (Astrocaryum vulgare Mart.) Almonds. 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Da Rosa, C.G.; Borges, C.D.; Zambiazi, R.C.; Rutz, J.K.; da Luz, S.R.; Krumreich, F.D.; Benvenutti, E.V.; Nunes, M.R. Encapsulation of the phenolic compounds of the blackberry (Rubus fruticosus). [CrossRef]
- Wilkowska, A.; Ambroziak, W.; Czyzowska, A.; Adamiec, J. Effect of Microencapsulation by Spray-Drying and Freeze-Drying Technique on the Antioxidant Properties of Blueberry (Vaccinium myrtillus) Juice Polyphenolic Compounds. [CrossRef]
- Stach, M.; Kolniak-Ostek, J. The Influence of the Use of Different Polysaccharide Coatings on the Stability of Phenolic Compounds and Antioxidant Capacity of Chokeberry Hydrogel Microcapsules Obtained by Indirect Extrusion. [CrossRef]
- Bakry, A.M.; Abbas, S.; Ali, B.; Majeed, H.; Abouelwafa, M.Y.; Mousa, A.; Liang, L. Microencapsulation of Oils: A Comprehensive Review of Benefits, Techniques, and Applications. [CrossRef]
- Gibis, M.; Ruedt, C.; Weiss, J. In vitro release of grape-seed polyphenols encapsulated from uncoated and chitosan-coated liposomes. [CrossRef]
- Has, C.; Sunthar, P. A comprehensive review on recent preparation techniques of liposomes. [CrossRef]
- Bozzuto, G.; Molinari, A. Liposomes as nanomedical devices. [CrossRef]
- Choudhury, N.; Meghwal, M.; Das, K. Microencapsulation: An overview on concepts, methods, properties and applications in foods. [CrossRef]
- Ozkan, G.; Franco, P.; De Marco, I.; Xiao, J.; Capanoglu, E. A review of microencapsulation methods for food antioxidants: Principles, advantages, drawbacks and applications. [CrossRef]
- de Souza, V.B.; Thomazini, M.; Echalar Barrientos, M.A.; Nalin, C.M.; Ferro-Furtado, R.; Genovese, M.I.; Favaro-Trindade, C.S. Functional properties and encapsulation of a proanthocyanidin-rich cinnamon extract (Cinnamomum zeylanicum) by complex coacervation using gelatin and different polysaccharides. [CrossRef]
- Soliman, T.N.; Mohammed, D.M.; El-Messery, T.M.; Elaaser, M.; Zaky, A.A.; Eun, J.B.; Shim, J.H.; El-Said, M.M. Microencapsulation of Plant Phenolic Extracts Using Complex Coacervation Incorporated in Ultrafiltered Cheese Against AlCl3-Induced Neuroinflammation in Rats. [CrossRef]
- Sarabandi, K.; Mahoonak, A.S.; Akbari, M. Physicochemical properties and antioxidant stability of microencapsulated marjoram extract prepared by co-crystallization method. [CrossRef]
- Ali, N.A.; Dash, K.K.; Pandey, V.K.; Tripathi, A.; Mukarram, S.A.; Harsányi, E.; Kovács, B. Extraction and Encapsulation of Phytocompounds of Poniol Fruit via Co-Crystallization: Physicochemical Properties and Characterization. [CrossRef]
- Cutrim, C.S.; Alvim, I.D.; Cortez, M.A.S. Microencapsulation of green tea polyphenols by ionic gelation and spray chilling methods. [CrossRef]
- Kurozawa, L.E.; Hubinger, M.D. Hydrophilic food compounds encapsulation by ionic gelation. [CrossRef]
- de Moura, S.C.S.R.; Berling, C.L.; Germer, S.P.M.; Alvim, I.D.; Hubinger, M.D. Encapsulating anthocyanins from Hibiscus sabdariffa L. calyces by ionic gelation: Pigment stability during storage of microparticles. [CrossRef]
- da Silva Júnior, M.E.; Araújo, M.V.R.L.; Martins, A.C.S.; dos Santos Lima, M.; da Silva, F.L.H.; Converti, A.; Maciel, M.I.S. Microencapsulation by spray-drying and freeze-drying of extract of phenolic compounds obtained from ciriguela peel. [CrossRef]
- Thakur, N.S.; Thakur, A. Microencapsulation of wild pomegranate flavedo phenolics by lyophilization: Effect of maltodextrin concentration, structural morphology, functional properties, elemental composition and ingredient for development of functional beverage. [CrossRef]
- Vallejo-Castillo, V.; Rodríguez-Stouvenel, A.; Martínez, R.; Bernal, C. Development of alginate-pectin microcapsules by the extrusion for encapsulation and controlled release of polyphenols from papaya (Carica papaya L.). 2020, 44, 44. [Google Scholar] [CrossRef] [PubMed]
- Bannikova, A.; Zyainitdinov, D.; Evteev, A.; Drevko, Y.; Evdokimov, I. Microencapsulation of polyphenols and xylooligosaccharides from oat bran in whey protein-maltodextrin complex coacervates: In-vitro evaluation and controlled release. [CrossRef]
- Istenič, K.; Cerc Korošec, R.; Poklar Ulrih, N. Encapsulation of (-)-epigallocatechin gallate into liposomes and into alginate or chitosan microparticles reinforced with liposomes. [CrossRef]
- Nik, A.B.; Vazifedoost, M.; Didar, Z.; Hajirostamloo, B. The antioxidant and physicochemical properties of microencapsulated bioactive compounds in Securigera securidaca (L.) seed extract by co-crystallization. [CrossRef]
- Chezanoglou, E.; Goula, A.M. Properties and Stability of Encapsulated Pomegranate Peel Extract Prepared by Co-Crystallization. [CrossRef]
- Anandharamakrishnan, C. Techniques for nanoencapsulation of food ingredients; Springer: 2014; Volume 8.
- Contreras-Angulo, L.A.; Gutiérrez-Grijalva, E.P.; Cabanillas-Bojórquez, L.A.; Jiménez-Ortega, L.A.; Heredia, J.B. Chapter 7 - Nanoformulations applied to the delivery of alkaloids. In Phytochemical Nanodelivery Systems as Potential Biopharmaceuticals, Heredia, J.B., Gutiérrez-Grijalva, E.P., Licea-Claverie, A., Gutierrez-Uribe, J.A., Patra, J.K., Eds.; Elsevier: 2023; pp. 257-281.
- Garcia-Carrasco, M.; Parra-Aguilar, I.F.; Gutiérrez-Grijalva, E.P.; Licea-Claverie, A.; Basilio Heredia, J. Chapter 18 - Nano-formulations in drug delivery. In Food, Medical, and Environmental Applications of Nanomaterials, Pal, K., Sarkar, A., Sarkar, P., Bandara, N., Jegatheesan, V., Eds.; Elsevier: 2022; pp. 473-491.
- Pedrozo, R.C.; Antônio, E.; Khalil, N.M.; Mainardes, R.M. Bovine serum albumin-based nanoparticles containing the flavonoid rutin produced by nano spray drying. [CrossRef]
- Rambaran, T.F. Nanopolyphenols: a review of their encapsulation and anti-diabetic effects. [CrossRef]
- Rehman, A.; Ahmad, T.; Aadil, R.M.; Spotti, M.J.; Bakry, A.M.; Khan, I.M.; Zhao, L.; Riaz, T.; Tong, Q. Pectin polymers as wall materials for the nano-encapsulation of bioactive compounds. [CrossRef]
- Ribeiro, E.F.; de Barros-Alexandrino, T.T.; Assis, O.B.G.; Junior, A.C.; Quiles, A.; Hernando, I.; Nicoletti, V.R. Chitosan and crosslinked chitosan nanoparticles: Synthesis, characterization and their role as Pickering emulsifiers. [CrossRef]
- Jiang, T.; Jin, K.; Liu, X.; Pang, Z. 8 - Nanoparticles for tumor targeting. In Biopolymer-Based Composites, Jana, S., Maiti, S., Jana, S., Eds.; Woodhead Publishing: 2017; pp. 221-267.
- Costa-Almeida, R.; Soares, R.; Costa, R. Polyphenol-Based Nanoparticles as Multifaceted Diabetes Modulators. In Functional Bionanomaterials: From Biomolecules to Nanoparticles, Thangadurai, D., Sangeetha, J., Prasad, R., Eds.; Springer International Publishing: Cham, 2020; pp. 251–270. [Google Scholar]
- Rai, V.K.; Gupta, G.D.; Pottoo, F.H.; Barkat, M.A. Potential of Nano-Structured Drug Delivery System for Phytomedicine Delivery. In Nanophytomedicine: Concept to Clinic, Beg, S., Barkat, M.A., Ahmad, F.J., Eds.; Springer Singapore: Singapore, 2020; pp. 89–111. [Google Scholar]
- Wu, M.; Jin, J.; Jin, P.; Xu, Y.; Yin, J.; Qin, D.; Wang, K.; Du, Q. Epigallocatechin gallate-β-lactoglobulin nanoparticles improve the antitumor activity of EGCG for inducing cancer cell apoptosis. [CrossRef]
- Palacio, J.; Monsalve, Y.; Ramírez-Rodríguez, F.; López, B. Study of encapsulation of polyphenols on succinyl-chitosan nanoparticles. [CrossRef]
- Gadkari, P.V.; Balaraman, M. Extraction of catechins from decaffeinated green tea for development of nanoemulsion using palm oil and sunflower oil based lipid carrier systems. [CrossRef]
- Harwansh, R.K.; Mukherjee, P.K.; Kar, A.; Bahadur, S.; Al-Dhabi, N.A.; Duraipandiyan, V. Enhancement of photoprotection potential of catechin loaded nanoemulsion gel against UVA induced oxidative stress. [CrossRef]
- Antônio, E.; Khalil, N.M.; Mainardes, R.M. Bovine Serum Albumin Nanoparticles Containing Quercetin: Characterization and Antioxidant Activity. Journal of nanoscience and nanotechnology 2016, 16, 1346–1353. [Google Scholar] [CrossRef] [PubMed]
- Javani, R.; Hashemi, F.S.; Ghanbarzadeh, B.; Hamishehkar, H. Quercetin-loaded niosomal nanoparticles prepared by the thin-layer hydration method: Formulation development, colloidal stability, and structural properties. [CrossRef]
- Romeo, A.; Musumeci, T.; Carbone, C.; Bonaccorso, A.; Corvo, S.; Lupo, G.; Anfuso, C.D.; Puglisi, G.; Pignatello, R. Ferulic Acid-Loaded Polymeric Nanoparticles for Potential Ocular Delivery. Pharmaceutics 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Nallamuthu, I.; Devi, A.; Khanum, F. Chlorogenic acid loaded chitosan nanoparticles with sustained release property, retained antioxidant activity and enhanced bioavailability. [CrossRef]
- Neelakandan, M.; Manoharan, S.; Muralinaidu, R.; Thara, J.M. Tumor preventive and antioxidant efficacy of chlorogenic acid–loaded chitosan nanoparticles in experimental skin carcinogenesis. [CrossRef]
- Mariadoss, A.V.A.; Vinayagam, R.; Senthilkumar, V.; Paulpandi, M.; Murugan, K.; Xu, B.; K.M, G.; Kotakadi, V.S.; David, E. Phloretin loaded chitosan nanoparticles augments the pH-dependent mitochondrial-mediated intrinsic apoptosis in human oral cancer cells. 2019, 130, 997–1008. [CrossRef]
- Kuai, L.; Liu, F.; Ma, Y.; Goff, H.D.; Zhong, F. Regulation of nano-encapsulated tea polyphenol release from gelatin films with different Bloom values. [CrossRef]
- Gaber Ahmed, G.H.; Fernández-González, A.; Díaz García, M.E. Nano-encapsulation of grape and apple pomace phenolic extract in chitosan and soy protein via nanoemulsification. [CrossRef]
- Soleimanifar, M.; Jafari, S.M.; Assadpour, E. Encapsulation of olive leaf phenolics within electrosprayed whey protein nanoparticles; production and characterization. [CrossRef]
- Oskoueian, E.; Karimi, E.; Noura, R.; Ebrahimi, M.; Shafaei, N.; Karimi, E. Nanoliposomes encapsulation of enriched phenolic fraction from pistachio hulls and its antioxidant, anti-inflammatory, and anti-melanogenic activities. [CrossRef]
- Gharehbeglou, P.; Jafari, S.M.; Homayouni, A.; Hamishekar, H.; Mirzaei, H. Fabrication of double W1/O/W2 nano-emulsions loaded with oleuropein in the internal phase (W1) and evaluation of their release rate. [CrossRef]
- Grama, C.N.; Suryanarayana, P.; Patil, M.A.; Raghu, G.; Balakrishna, N.; Kumar, M.N.V.R.; Reddy, G.B. Efficacy of Biodegradable Curcumin Nanoparticles in Delaying Cataract in Diabetic Rat Model. [CrossRef]
- Zhang, J.; Li, S.; An, F.-F.; Liu, J.; Jin, S.; Zhang, J.-C.; Wang, P.C.; Zhang, X.; Lee, C.-S.; Liang, X.-J. Self-carried curcumin nanoparticles for in vitro and in vivo cancer therapy with real-time monitoring of drug release. [CrossRef]
- Chockalingam, S.; Thada, R.; Dhandapani, R.K.; Panchamoorthy, R. Biogenesis, characterization, and the effect of vicenin-gold nanoparticles on glucose utilization in 3T3-L1 adipocytes: A bioinformatic approach to illuminate its interaction with PTP 1B and AMPK. [CrossRef]
- Das, S.; Roy, P.; Pal, R.; Auddy, R.G.; Chakraborti, A.S.; Mukherjee, A. Engineered silybin nanoparticles educe efficient control in experimental diabetes. [CrossRef]
- Salah, M.; Mansour, M.; Zogona, D.; Xu, X. Nanoencapsulation of anthocyanins-loaded β-lactoglobulin nanoparticles: Characterization, stability, and bioavailability in vitro. [CrossRef]
- Sun, J.; Chen, J.; Mei, Z.; Luo, Z.; Ding, L.; Jiang, X.; Bai, W. Synthesis, structural characterization, and evaluation of cyanidin-3-O-glucoside-loaded chitosan nanoparticles. [CrossRef]
- Liang, T.; Guan, R.; Shen, H.; Xia, Q.; Liu, M. Optimization of Conditions for Cyanidin-3-OGlucoside (C3G) Nanoliposome Production by Response Surface Methodology and Cellular Uptake Studies in Caco-2 Cells. [CrossRef]
- Verônica Cardoso de Souza, B.; de Morais Sousa, M.; Augusto Gasparotto Sattler, J.; Cristina Sousa Gramoza Vilarinho Santana, A.; Bruno Fonseca de Carvalho, R.; de Sousa Lima Neto, J.; de Matos Borges, F.; Angelica Neri Numa, I.; Braga Ribeiro, A.; César Cunha Nunes, L. Nanoencapsulation and bioaccessibility of polyphenols of aqueous extracts from Bauhinia forficata link. [CrossRef]
- Kerbab, K.; Sansone, F.; Zaiter, L.; Esposito, T.; Celano, R.; Franceschelli, S.; Pecoraro, M.; Benayache, F.; Rastrelli, L.; Picerno, P.; et al. Halimium halimifolium: From the Chemical and Functional Characterization to a Nutraceutical Ingredient Design. [CrossRef]
- Zorzenon, M.R.T.; Formigoni, M.; da Silva, S.B.; Hodas, F.; Piovan, S.; Ciotta, S.R.; Jansen, C.A.; Dacome, A.S.; Pilau, E.J.; Mareze-Costa, C.E.; et al. Spray drying encapsulation of stevia extract with maltodextrin and evaluation of the physicochemical and functional properties of produced powders. [CrossRef]
- De Silva, N.D.; Attanayake, A.P.; Karunaratne, D.N.; Arawwawala, L.; Pamunuwa, G.K. Bael (Aegle marmelos L. Correa) fruit extracts encapsulated alginate nanoparticles as a potential dietary supplement with improved bioactivities. [CrossRef]
- Chauhan, P.; Tamrakar, A.K.; Mahajan, S.; Prasad, G. Chitosan encapsulated nanocurcumin induces GLUT-4 translocation and exhibits enhanced anti-hyperglycemic function. [CrossRef]
- Pandey, S.; Dvorakova, C.M. Future Perspective of Diabetic Animal Models. [CrossRef]
- Goyal, S.N.; Reddy, N.M.; Patil, K.R.; Nakhate, K.T.; Ojha, S.; Patil, C.R.; Agrawal, Y.O. Challenges and issues with streptozotocin-induced diabetes – A clinically relevant animal model to understand the diabetes pathogenesis and evaluate therapeutics. [CrossRef]
- Panwar, R.; Raghuwanshi, N.; Srivastava, A.K.; Sharma, A.K.; Pruthi, V. In-vivo sustained release of nanoencapsulated ferulic acid and its impact in induced diabetes. [CrossRef]
- Yao, Y.; Luong, T.N.; Lepik, M.; Aftab, N.; Fong, V.H.; Vieira, A. Synergism of antioxidant phytochemicals: comparisons among purified polyphenols and dietary-plant extracts. 2012; pp. 121-127.
- Revathi, G.; Elavarasi, S.; Saravanan, K.; Ashokkumar, M.; Egbuna, C. Greater efficiency of polyherbal drug encapsulated biosynthesized chitosan nano-biopolymer on diabetes and its complications. [CrossRef]
- El-Hussien, D.; El-Zaafarany, G.M.; Nasr, M.; Sammour, O. Chrysin nanocapsules with dual anti-glycemic and anti-hyperlipidemic effects: Chemometric optimization, physicochemical characterization and pharmacodynamic assessment. [CrossRef]
- Sudirman, S.; Lai, C.-S.; Yan, Y.-L.; Yeh, H.-I.; Kong, Z.-L. Histological evidence of chitosan-encapsulated curcumin suppresses heart and kidney damages on streptozotocin-induced type-1 diabetes in mice model. [CrossRef]
- El-Shahawy, A.A.G.; Abdel-Moneim, A.; Ebeid, A.S.M.; Eldin, Z.E.; Zanaty, M.I. A novel layered double hydroxide-hesperidin nanoparticles exert antidiabetic, antioxidant and anti-inflammatory effects in rats with diabetes. [CrossRef]
- Wang, Q.; Wei, C.; Weng, W.; Bao, R.; Adu-Frimpong, M.; Toreniyazov, E.; Ji, H.; Xu, X.-M.; Yu, J. Enhancement of oral bioavailability and hypoglycemic activity of liquiritin-loaded precursor liposome. [CrossRef]
- Foudah, A.I.; Ayman Salkini, M.; Alqarni, M.H.; Alam, A. Preparation and evaluation of antidiabetic activity of mangiferin-loaded solid lipid nanoparticles. [CrossRef]
- Wang, Y.; Karmakar, T.; Ghosh, N.; Basak, S.; Gopal Sahoo, N. Targeting mangiferin loaded N-succinyl chitosan-alginate grafted nanoparticles against atherosclerosis – A case study against diabetes mediated hyperlipidemia in rat. [CrossRef]
- Upadhyay, M.; Hosur, R.V.; Jha, A.; Bharti, K.; Mali, P.S.; Jha, A.K.; Mishra, B.; Kumar, A. Myricetin encapsulated chitosan nanoformulation for management of type 2 diabetes: preparation, optimization, characterization and in vivo activity. [CrossRef]
- Joshi, R.; Laddha, A.P.; Kulkarni, Y.A.; Wairkar, S. Improved performance of naringenin herbosomes over naringenin in streptozotocin-induced diabetic rats: In vitro and in vivo evaluation. [CrossRef]
- Maity, S.; Chakraborti, A.S. Formulation, physico-chemical characterization and antidiabetic potential of naringenin-loaded poly D, L lactide-co-glycolide (N-PLGA) nanoparticles. [CrossRef]
- Ahmad, M.Z.; Pathak, K.; Das, R.J.; Saikia, R.; Sarma, H.; Gogoi, N.; Gogoi, U.; Das, A.; Alasiri, A.S.; Abdel-Wahab, B.A.; et al. Design and optimization of quercetin-loaded polymeric Eudragit L-100 nanoparticles for anti-diabetic activity with improved oral delivery: in-vitro and in-vivo evaluation. J Inorg Organomet Polym Mater 2023, 33, 2411–2428. [Google Scholar] [CrossRef]
- Chitkara, D.; Nikalaje, S.K.; Mittal, A.; Chand, M.; Kumar, N. Development of quercetin nanoformulation and in vivo evaluation using streptozotocin induced diabetic rat model. [CrossRef]
- Wang, S.; Du, S.; Wang, W.; Zhang, F. Therapeutic investigation of quercetin nanomedicine in a zebrafish model of diabetic retinopathy. [CrossRef]
- Du, S.; Lv, Y.; Li, N.; Huang, X.; Liu, X.; Li, H.; Wang, C.; Jia, Y.-F. Biological investigations on therapeutic effect of chitosan encapsulated nano resveratrol against gestational diabetes mellitus rats induced by streptozotocin. [CrossRef]
- Benyelles, M.; Merzouk, H.; Merzouk, A.Z.; Imessaoudene, A.; Medjdoub, A.; Mebarki, A. Valorization of Encapsulated Coffee Parchment Extracts as Metabolic Control for High Fructose Diet-Induced Obesity, Using Wistar Rat as Animal Model. [CrossRef]
- Colorado, D.; Fernandez, M.; Orozco, J.; Lopera, Y.; Muñoz, D.L.; Acín, S.; Balcazar, N. Metabolic activity of anthocyanin extracts loaded into non-ionic niosomes in diet-induced obese mice. [CrossRef]
- de Paulo Farias, D.; de Araújo, F.F.; Neri-Numa, I.A.; Pastore, G.M. Antidiabetic potential of dietary polyphenols: A mechanistic review. [CrossRef]
- Huang, Y.-C.; Chen, B.-H. A comparative study on improving streptozotocin-induced type 2 diabetes in rats by hydrosol, extract and nanoemulsion prepared from cinnamon leaves. Antioxidants 2023, 12, 29. [Google Scholar] [CrossRef] [PubMed]
- Wickramasinghe, A.S.D.; Attanayake, A.P.; Kalansuriya, P. Gelatine nanoparticles encapsulating three edible plant extracts as potential nanonutraceutical agents against type 2 diabetes mellitus. [CrossRef]
- Ammar, N.M.; Hassan, H.A.; Mohammed, M.A.; Serag, A.; Abd El-Alim, S.H.; Elmotasem, H.; El Raey, M.; El Gendy, A.N.; Sobeh, M.; Abdel-Hamid, A.-H.Z. Metabolomic profiling to reveal the therapeutic potency of Posidonia oceanica nanoparticles in diabetic rats. [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. 1997; 23, 3–25. [Google Scholar] [CrossRef]
| Source | Encapsulation method | Wall material | Conditions | Results | Reference |
| Tucuma Coproduct (Astrocaryum vulgare Mart.) Almonds |
Spray Drying | Maltodextrin (5%) | Temperature: 100 °C; flow rate: 7.5 mL/min, and pressure: 6 bar. | The microparticles showed spherical and heterogeneous structures and good encapsulation efficiency. | [67] |
| Blackberry Pomace (Rubus fruticosus) | Spray Drying | Maltodextrin DE 10, in a 1:1 (w/w) ratio | Inlet drying air temperature: 170 °C; atomization pressure: 4 bar; drying air flow: 3.5 m3/h, and flow rate: 0.5 L/h. | Microparticles have a rounded outer structure and are agglomerated into different sizes. | [64] |
| Chipilin (Crotalaria longirostrata) methanolic extracts | Spray Drying | Maltodextrin, Arabic gum, Cajanus gum, cocoa shell pectin, Cajanus protein, and soy protein. | Inlet air temperature: 120 °C; feed flow: 3 mL min-1; drop pressure: 1.35 bar | Microcapsules with mostly irregular amorphous structures, smooth surfaces, and depressions. Size between 3 and 8 μm | [65] |
| Sambucus Nigra L. (elderberry) | Spray Drying | Modified chitosan, sodium alginate, and Arabic gum. | Flow rate: 4 mL/min (15%); inlet temperature: 115 °C; air pressure: 5–6 bar, and aspiration rate: 100% (36 m3/h) | Very small particles (between 5 and 19 μm). | [66] |
| Extract from Lippia citriodora leaves | Spray Drying | Maltodextrin and inulin | Inlet air temperature 135–195 °C; airflow: 0.30 m3/min; feeding flow: 2 mL/min, atomization air flow: 13 L/min | Inulin increased powder and polar compounds recovery, whereas maltodextrin achieved a higher encapsulation efficiency. | [63] |
| Ciriguela (Spondias purpurea L.) | Freeze-drying | Maltodextrin 10 DE and arabic gum | 48 h in a freeze dryer at −80 °C and 0.28 mbar chamber pressure. | Microcapsules with irregular shape, extensive wrinkles, and a serrated surface. | [84] |
| Blackberry (Rubus fruticosus) |
Freeze-drying | Chitosan, xanthan, β-cyclodextrin, and hydrogel | Mixture: 0.003 mol of polymer and the same proportion of extract, diluted in 50 mL of water. The solution was frozen at -80 °C for 24 h, with subsequent lyophilization. | Only chitosan and xanthan showed the characteristic shape. | [68] |
| Blueberry (Vaccinium myrtillus) Juice | Freeze-drying | HP- β-cyclodextrin and β-cyclodextrin | β-CD in 15% (w/w) ratio to hot (75°C) blueberry juice. The precipitated product was freeze-dried at −50°C | Formation of amorphous material and a 78.1% product yield. | [69] |
| Pomegranate (Punica granatum L.) | Freeze-drying | Maltodextrin (20 DE) | The extract and maltodextrin mixture (1:2 (w/w)) was lyophilized at -30 ◦C and vacuum pressure: 0.04 mbar. | Homogeneous coating on particle surface. | [85] |
| Black chokeberry (Aronia melanocarpa) |
Indirect extrusion | Sodium alginate, low-molecular-weight chitosan, carrageenan, Low-methoxyl pectin | Alginate was mixed in equal proportions (1:1 g/g) with other encapsulants. Encapsulator; vibrating nozzle: 150 m; pressure: 200 mbar; frequency: 400 Hz; electrode: 1000 V; solidification temperature: 30 ◦C and complexation time: 10 min. | Hydrogel beads differ in shape and structure. The most regular capsules were obtained with the mixture of alginate + carrageenan. | [70] |
| Papaya fruit (Carica papaya L.) | Extrusion | Pectin-alginate | The papaya extract was encapsulated through the in situ and two-step methodologies. Alginate:pectin ratio was 55:45. | Bioactive compounds are dispersed in the encapsulation matrix, improving their thermal stability. | [86] |
| Proanthocyanidin cinnamon extract | Complex coacervation | Gelatin and five different polysaccharides (gum Arabic, pectin, cashew tree gum, carboxymethylcellulose, and κ-carrageenan | The proanthocyanidin-rich cinnamon extract was dispersed in distilled water. The gelatin dispersion was added, and then the polysaccharide solution. The decanted material was frozen at -20 °C and dried in freeze-dryer. | Particles presented resistance when submitted to different stress conditions, except pH lower than 2 and temperatures higher than 50 °C. | [77] |
| Polyphenols from oat bran | Complex coacervation | Whey protein concentrates 10% Maltodextrin 10% |
The wall materials were mixed in ratios 10:0, 8:2, 6:4, 4:6, and 2:8 by gentle magnetic stirring for 1 h. BAS extract was then added to the wall material at 10% (1:10 ratio) and the microcapsules solution was formed using a Magnetic Stirrer for 15 min. | The encapsulation efficiency was 95.28%. The release percentage of polyphenols coated in a capsule ranged between 70 and 83% after 2 h of digestion. | [87] |
| (−)-Epigallocatechin gallate (≥94%) | Liposomes | Phospholipon | Phospholipon and Epigallocatechin gallate were dissolved in ethanol. Citric acid (0.1%) was added while stirring, and the mixture was heated to 60 °C. The microparticles were prepared using an encapsulator. | Encapsulation efficiency (>97%) and sustained release; in 14 days, no more than 15% of EGCG was released. The sizes of the liposomes were estimated at 1–2 μm. | [88] |
| Grape-seed extract | Liposomes | Soy lecithin |
Grape-seed extract was incorporated into liposomes (1.1% w/w soy lecithin) using high-pressure homogenization (22,500 psi). | Entrapment efficiency for uncoated liposomes was 88.2 ± 4.7%. The release rate after 24 h from uncoated liposomes was 0.55*h. | [72] |
| Green tea extract (C. sinensis) | Ionic gelation | Amidated low methoxyl pectin, calcium chloride, hydrogenated palm oil |
Association of a double emulsion (water/oil/water) with ionic gelation. The final emulsion was sprayed through a double-fluid atomizer on a CaCl2 crosslinking solution acidified with citric acid (pH 3). | 72.6 ± 0.4% encapsulation efficiency for ionic gelation microparticles. | [81] |
| Anthocyanins from Hibiscus sabdariffa L. calyces | Ionic gelation | Rapeseed oil, pectin, calcium chloride | Ionic gelation using two techniques: drip-extrusion and atomization, both using a double emulsion (Hibiscus extract /rapeseed oil/pectin) and a cross-linked solution (CaCl2). | The median diameter (D50) of the particles ranged from 78 to 1100μm, and encapsulation efficiency ranged from 67.9 to 93.9%. | [83] |
| Securigera securidaca (L) seed extract | Co-crystallization | Saccharose | Sucrose and S. securidaca extract were mixed on a heater at 132° C. The co-crystallized product was dried in an oven at 40°C for 15 h, then ground and sieved. | The production efficiency and moisture content of the extract-containing co-crystallized powder were 84% and 0.14%, respectively. | [89] |
| Pomegranate Peel Extract |
Co-Crystallization | Food-grade crystal sucrose | Sucrose solution and extract were mixed at 700 rpm. The mixture is placed in a water bath and stirred until it reaches 45 °C. The powder is kept in a desiccator for 24 h. | The co-crystallized powder had low moisture content (0.59%), low hygroscopicity (0.011%), high apparent density (0.803 g/cm3) and solubility (61 s). | [90] |
| Polyphenols Loaded | Nanosystem | Encapsulating Material | Technique Nanoencapsulation | Ref. |
| Epigallocatechin-3-gallate | Nanoparticle | Bovine β-lactoglobulin | Co-assembled with preheated | [101] |
| Epigallocatechin-3-gallate | Nanoparticles | Succinyl-chitosan (modified chitosan), pentasodium tripolyphosphate | Ionic crosslinking | [102] |
| Propyl gallate | Nanoparticles | Succinyl-chitosan (modified chitosan), pentasodium tripolyphosphate | Ionic crosslinking | [102] |
| Gallic acid | Nanoparticles | Succinyl-chitosan (modified chitosan), pentasodium tripolyphosphate | Ionic crosslinking | [102] |
| Catechin | Nanoemulsion | Palm oil and sunflower oil | Nanoemulsification | [103] |
| Catechin | Nanoemulsion |
Ethyl oleate, the surfactant span 80, and the cosurfactant trancutol CG | Nanoemulsification | [104] |
| Rutin | Nanoparticle | Bovine serum albumins | Nanospray drying | [94] |
| Quercetin | Nanoparticle | Bovine serum albumins and glutaraldehyde as a crosslinking agent | Desolvation | [105] |
| Quercetin | Nanoniosome | Surfactants (span 60 and 80, tween 60 and 80), polymers (polyethylene glycol, propylene glycol, glycerol, and cholesterol. | Thin-layer hydration combined with sonication | [106] |
| Trans-Ferulic acid | Nanoparticle | Nanoparticle A: poly (lactic acid) Nanoparticle B: poly (lactic acid)/poly (lactic-co-glycolic acid) |
Nanoprecipitation | [107] |
| Chlorogenic acid | Nanoparticle | Chitosan, pentasodium tripolyphosphate | Ionic gelation | [108,109] |
| Phloretin | Nanoparticle | Chitosan, sodium tripolyphosphate | Ionotropic gelation | [110] |
| Tea Polyphenol | Nanoparticle | Chitosan, sulfobutylether-β-cyclodextrin | Inclusion complexes | [111] |
| Phenolics of grape pomace | Nanocapsules | Chitosan, soy protein | Nanoemulsification | [112] |
| Phenolics of apple pomace | Nanocapsules | Chitosan, soy protein | Nanoemulsification | [112] |
| Olive leaf phenolics | Nanoparticle | Whey protein concentrate and tween 20 as surfactant | Electrospray | [113] |
| Phenolics of pistachio hulls | Nanoliposome | Lecithin | Sonication | [114] |
| Oleuropin | Nanoemulsion | Soybean oil, span 80 (surfactant), whey protein concentrate, and pectin | Double emulsification | [115] |
| Curcumin | Nanoparticle | Polyvinyl alcohol, Poly(lactide-co-glycolic) acid |
Modified emulsion- diffusion-evaporation method | [116] |
| Curcumin | Nanoparticle | Poly(maleic anhydride-alt-1-octadecene), poly(ethylene glycol)-amine and 1-Ethyl-3-(3-dimethylaminopropyl)-carbodiimide |
Sonication | [117] |
| Vicenin-2 | Nanoparticle | Chloroauric acid | Ultrasonication | [118] |
| Sylbin | Nanoparticle | Chitosan, poly(lactide-co-glycolic) acid, pluronic F-127 | Solvent diffusion and polyelectrolyte deposition | [119] |
| Anthocyanin from raspberry | Nanoparticle | β-Lactoglobulin, N-(3-Dimethylaminopropyl)-N-ethyl carbodiimide hydrochloride (cross-linking) | Desolvatation | [120] |
| Cyanidin 3-O-Glucoside | Nanoparticle | Nanoparticle1: Chitosan, and PGA Nanoparticle 2: Chitosan oligosaccharide, and polyglutamic acid Nanoparticle 3: Carboxymethyl chitosan, CaCl2 |
Ionic crosslinking | [121] |
| Cyanidin 3-O-Glucoside | Nanoliposome | Phosphatidylcholine and cholesterol | Reverse-phase evaporation | [122] |
| Compound | Polymer/Particle size | Dosage | In vivomodel | Effect* | Ref. |
| Chrysin | PLGA/176.0±2.1 nm | One administration of 20 mg/kg | STZ-induced diabetes in male albino rats (180-200 g) | ↓Blood glucose ↓TG, LDL ↑HDL |
[133] |
| Curcumin | Chitosan/n.s | 150 mg/kg once a day, for 28 days | STZ-induced type 1 diabetes in C57Bl/6 mice | ↓Blood glucose ↑Insulin secretion ↓Fibrosis in the kidney |
[134] |
| Ferulic acid | Chitosan/211.3±5.1 nm | 10 mg/kg once a day, for 14 days | STZ-induced diabetes in Wistar albino rats (110-150 g) | ↓Blood glucose ↑Plasma insulin levels ↓TC, TG -Recovered islets of Langerhans in the pancreas |
[130] |
| Hesperidin | MgAl-double layered hydroxide/330-380 nm | 50 mg/kg once a day, for 30 days | Nicotinamide+STZ-induced diabetes in male albino rats (200-300 g) | ↓Plasma glucose, HbA1c ↑Insulin, HOMA-B -Restored the pancreatic Islets of Langerhans |
[135] |
| Liquiritin | Phospholipid complex/91.8±1.9 nm | 200 mg/kg once a day, for 28 days | STZ-diabetes induced in male ICR mice (18-22 g) | ↓Blood glucose - Improved the glomerular and renal cortical structure of the kidney |
[136] |
| Mangiferin | Labrafil M 2130 CS/138.4±3.4 nm | One administration of 40 mg/kg | High-fat diet + STZ-diabetes induced in male Wistar rats (250 g) | ↓Blood glucose ↓TC, TG ↑HDL ↓AST, ALT |
[137] |
| Mangiferin | NSC-alginate/124 nm | 10 mg/kg once a day, for 28 days | STZ-induced diabetes in Wistar rats (100-150 g) | ↓Blood glucose ↓TC, TG, LDL ↑HDL |
[138] |
| Myricetin | Chitosan/ 184.4±4.1 nm | 50 mg/kg once a day, for 28 days | STZ-induced diabetes in male Wistar rats (~250 g) | ↓Blood glucose ↓TG, TC ↑BW |
[139] |
| Naringenin | Phospholipid LECIVA-S70/564.4 nm | Single dose of 25 mg/kg or 50 mg/kg, for 28 days |
STZ-induced diabetes in male Sprague Dawley rats (180-220 g) | ↓Plasma glucose level ↓TC, TG, BUN ↓ALT, AST |
[140] |
| Naringenin | PLGA/129 nm | One dose of 10 mg/kg, and a second dose after 10 days, period of 7-49 days | STZ-induced diabetes in male Wistar rats | ↓Blood glucose ↑Insulin level ↓HbA1c -Restored pancreas and kidney cells |
[141] |
| Quercetin | Eudragit L-100/144.7±1.7 nm | 200 mg/kg once a day, during 21 days | STZ-induced diabetes in albino female Wistar rats (150-200 g) | ↓Blood glucose ↓TG, TC, LDL ↓ALP, ALT, AST ↓cellular damage in the pancreas |
[142] |
| Quercetin | PLGA/179.9±11.2 nm | 150 mg/kg every 5th day, during 15 days |
STZ-stimulated male Sprague-Dawley rats (~250 g) | ↓Blood glucose | [143] |
| Quercetin | Poloxamer-180-stearic acid/157.1 to 528.2 nm | 5 or 10 mg/kg, for 21 days | STZ-induced diabetic retinopathy in male adult zebra fish (< 8 months) | ↓Plasma glucose | [144] |
| Resveratrol | Chitosan/38.0 nm | 100 mg /kg, for 28 days | STZ-indued gestational diabetes mellitus in Wistar albino rats (180-200 g) | ↓Blood glucose ↑Insulin level ↓TC, TG, LDL ↑HDL |
[145] |
| Plant specie | Components of the extract | Encapsulating material/particle size | In vivomodel, dosage | Effect* | Ref |
| Cinnamomoum osmophloeum Kanehira | Cinnamaldehyde, benzoic acid, caffeic acid, caffeoylquinic acid, cinnamic acid, coumaric acid, rutin, kaempferol, eugenol, quercetin, and derivatives | Nanoemulsion (soybean oil, lecithin and Tween 80)/ 36.6 nm |
Nicotinamide + STZ-induced diabetes in male Wistar rats (7 weeks old), 60 mg/kg (cinammaldehyde equivalents) | ↓Blood glucose, HOMA-IR ↓TC, TG, AST, ALT, BUN |
[149] |
| Coccinia grandis | Phenolics and flavonoids | Gelatin/ 468±14 nm |
High-fat diet+STZ-induced diabetes in male Wistar rats (135-165 g), single dose of 330 mg/kg | ↓Plasma glucose | [150] |
| Coffea arabica | Caffeine, chlorogenic acid | Maltodextrin + whey protein/ 1-2 µm |
Fructose-induced obesity in male Wistar rats (85-120 g), 100 mg/kg per day (during 28 days) | ↓Glucose, HOMA-IR ↓TC, TG, AST, ALT ↓Liver-TG, liver-TC |
[146] |
| Murraya koenigii | Phenolics and flavonoids | Gelatin/ 520±33 nm |
High-fat diet + STZ-induced diabetes in male Wistar rats (135-165 g), single dose of 65 mg/kg |
↓Plasma glucose | [150] |
| Posidonia oceanica | Hydroxybenzoic acid, protocatechuic acid, ferulic acid, gallic acid, coumaric acid, sinapic acid, vanillic acid, catechin, epicatechin, luteolin, naringenin, apigenin, among others. | Bovine gelatine/ 274.7±30.5 |
STZ-induced diabetes in male Wistar albino rats (150-170 g), 100 mg/kg (for 28 days) | ↓Glucose, HOMA-IR ↑GLUT4 |
[151] |
| Senna auriculata | Phenolics and flavonoids | Gelatin/ 563±4 nm |
High-fat diet + STZ-induced diabetes in male Wistar rats (135-165 g), Single dose of 45 mg/kg |
↓Plasma glucose | [150] |
| Vaccinium meridionale | Delphinidin 3-hexoside, cyanidin-3-galactoside, cyanidin-3-glucoside, cyanidin 3-arabinoside | Pro-nanosome Nio-N/ 219.7±3.1 nm | High-fat diet-induced obesity in C57BL/6 mice, 160 µg/mL (during 28 days) | ↓Glucose ↓TC, leptin |
[147] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




