Submitted:
10 September 2025
Posted:
12 September 2025
You are already at the latest version
Abstract
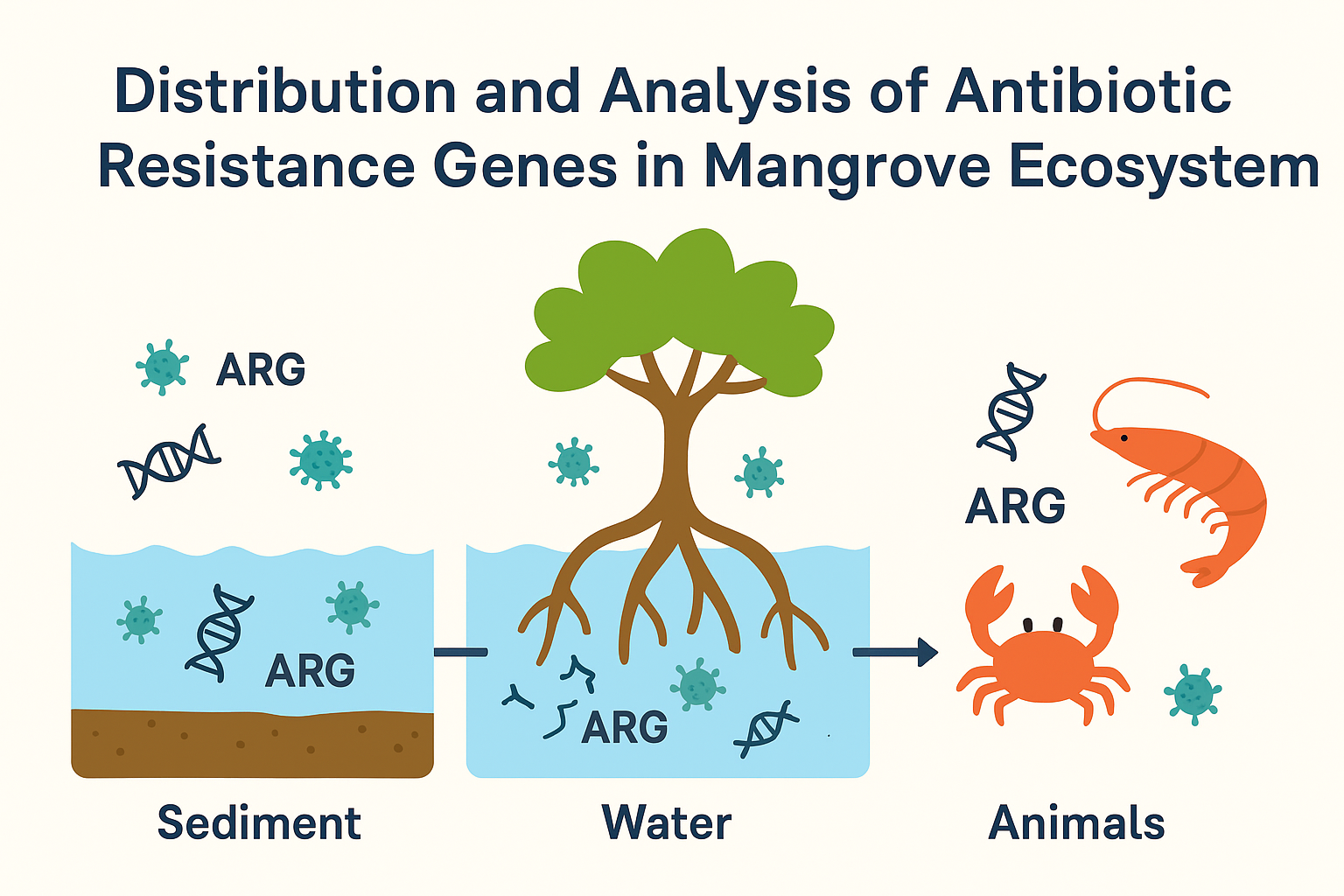
Keywords:
1. Introduction
1.1. Significance of Mangrove Ecosystems
1.2. Anthropogenic Impacts on Mangrove Ecosystems
1.3. Antibiotic Resistance in Coastal Ecosystems
1.4. Role of Mangroves in Antibiotic Resistance Dynamics
1.5. Objectives of the Study
- To assess the distribution and abundance of ARGs in mangrove sediments, water, and biota.
- To identify the primary sources and pathways of ARG contamination in mangrove environments.
- To evaluate the impact of anthropogenic activities on the prevalence and diversity of ARGs in mangrove ecosystems.
- To examine the potential for natural attenuation of ARGs within mangrove habitats.
- To provide recommendations for monitoring and managing antibiotic resistance in mangrove ecosystems.
1.6. Scope and Limitations
2. Methods
2.1. Literature Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction
2.4. Quality Assessment
2.5. Data Synthesis
2.6. Ethical Considerations
3. Results
3.1. Study Selection and Characteristics
3.2. Prevalence and Diversity of ARGs in Mangrove Ecosystems
3.3. Environmental Drivers and Sources of ARGs
3.4. Patterns in Microbial Community Composition
3.5. Temporal and Spatial Trends
3.6. Summary of Key Findings
- Mangrove sediments and water contain a broad range of ARGs, including tetracyclines, sulfonamides, β-lactams, and multidrug resistance genes.
- ARG abundance is highest near aquaculture and urban-influenced areas.
- Mobile genetic elements play a critical role in ARG dissemination.
- Microbial community composition and environmental factors (organic carbon, salinity, sediment type) influence ARG prevalence.
- Mangroves serve as both reservoirs and natural buffers, but anthropogenic pressure can overwhelm their natural mitigation capacity.
4. Discussion
4.1. Overview and Interpretation of Findings
4.2. Anthropogenic Influence on ARG Dissemination
4.3. Environmental Drivers and Natural Attenuation
4.4. Microbial Community Composition and ARG Dynamics
4.5. Implications for Environmental and Public Health
4.6. Management and Policy Recommendations
4.7. Limitations and Future Research Directions
5. Conclusion and Recommendations
5.1. Conclusion
5.2. Recommendations
- Reduction of Antibiotic Use in Aquaculture: Implementing stricter regulations on antibiotic usage in coastal aquaculture and promoting alternative disease management strategies (e.g., probiotics, vaccination, and biosecurity measures) can reduce ARG input into mangrove ecosystems (Cabello et al., 2013).
- Environmental Monitoring and Surveillance: Regular monitoring of ARGs and associated microbial communities in mangrove ecosystems should be conducted using standardized molecular techniques, including qPCR and metagenomic sequencing, to detect resistance hotspots and track temporal trends (Hendriksen et al., 2019).
- Mangrove Conservation and Restoration: Conservation of existing mangrove forests and restoration of degraded areas enhance natural filtration and microbial attenuation processes, contributing to the reduction of ARG dissemination in coastal environments (Palacios et al., 2021).
- Interdisciplinary Collaboration: Collaboration among environmental scientists, microbiologists, public health professionals, policymakers, and local communities is essential to develop sustainable management strategies that balance ecosystem protection with aquaculture productivity.
- Future Research Directions: Further studies should aim to (i) standardize ARG detection and quantification methods, (ii) expand geographic coverage to underrepresented mangrove regions, (iii) examine functional consequences of ARGs on microbial communities, and (iv) investigate the efficacy of mangrove restoration in mitigating ARG prevalence. Integrating metagenomics with functional and ecological assessments will provide a comprehensive understanding of resistance dynamics and environmental risk.
5.3. Final Remarks
Supplementary Material
Author Contributions
Funding
Ethics Statement
Data Availability Statement
Acknowledgements
Conflict of Interest
References
- Cabello, F.C.; Godfrey, H.P.; Tomova, A.; et al. Antimicrobial use in aquaculture re-examined: Its relevance to antimicrobial resistance and human health. Environ. Microbiol. 2013, 15, 1917–1945. [Google Scholar] [CrossRef] [PubMed]
- Hendriksen, R.S.; Munk, P.; Njage, P.; et al. Global monitoring of antimicrobial resistance based on metagenomics analyses of urban sewage. Nat. Microbiol. 2019, 4, 1977–1985. [Google Scholar] [CrossRef] [PubMed]
- Imchen, M.; Vennapu, R.K.; Ghosh, P.; Kumavath, R. Insights into antagonistic interactions of multidrug-resistant bacteria in mangrove sediments from the South Indian coast. Mar. Pollut. Bull. 2020, 161, 111789. [Google Scholar]
- Sresung, M.; Srathongneam, T.; Paisantham, P.; Sukchawalit, R.; Whangsuk, W.; Honda, R.; Satayavivad, J.; Mongkolsuk, S.; Sirikanchana, K. Quantitative distribution of antibiotic resistance genes and crAssphage in a tropical urbanized watershed. Sci. Total Environ. 2024, 954, 176569. [Google Scholar] [CrossRef] [PubMed]
- Yaikhan, T.; Suwannasin, S.; Singkhamanan, K.; Chusri, S.; Pomwised, R.; Wonglapsuwan, M.; Surachat, K. Genomic Characterization of Multidrug-Resistant Enterobacteriaceae Clinical Isolates from Southern Thailand Hospitals: Unraveling Antimicrobial Resistance and Virulence Mechanisms. Antibiotics 2024, 13, 531. [Google Scholar] [CrossRef] [PubMed]
- Sudatip, D.; Tiengrim, S.; Chasiri, K.; Kritiyakan, A.; Phanprasit, W.; Morand, S.; Thamlikitkul, V. One Health Surveillance of Antimicrobial Resistance Phenotypes in Selected Communities in Thailand. Antibiotics 2022, 11, 556. [Google Scholar] [CrossRef] [PubMed]
- Hinthong, W.; Thaotumpitak, V.; Sripradite, J.; Indrawattana, N.; Srisook, T.; Kongngoen, T.; Atwill, E. R.; Jeamsripong, S. Antimicrobial resistance, virulence profile, and genetic analysis of ESBL-producing Escherichia coli isolated from Nile tilapia in fresh markets and supermarkets in Thailand. PLoS ONE 2024, 19, e0296857. [Google Scholar] [CrossRef] [PubMed]
- Intahphuak, S.; Apidechkul, T.; Kuipiaphum, P. Antibiotic resistance among the Lahu hill tribe people, northern Thailand: a cross-sectional study. BMC Infect. Dis. 2021, 21, 385. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhang, Y.; Li, Y. Diverse and abundant antibiotic resistance genes in mangrove area and their relationship with bacterial communities—A study in Hainan Island, China. Sci. Total Environ. 2021, 755, 142544. [Google Scholar]
- Liu, Z.; Wan, X.; Zhang, C.; Cai, M.; Pan, Y.; Li, M. Deep sequencing reveals comprehensive insight into the prevalence, mobility, and hosts of antibiotic resistance genes in mangrove ecosystems. J. Environ. Manag. 2023, 335, 117580. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Munn, Z.; Peters, M.D.; Stern, C.; et al. Systematic review or scoping review? Guidance for authors when choosing between a systematic or scoping review approach. BMC Med. Res. Methodol. 2015, 15, 143. [Google Scholar] [CrossRef] [PubMed]
- Palacios, O.A.; Adame-Gallegos, J.R.; Rivera-Chavira, B.E.; Nevarez-Moorillon, G.V. Antibiotics, multidrug-resistant bacteria, and antibiotic resistance genes: Indicators of contamination in mangroves? Antibiotics (Basel) 2021, 10, 1103. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, X.X.; Zhang, T.; Fang, H.H.P. Antibiotic resistance genes in water environment. Environ. Sci. Pollut. Res. 2020, 27, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Yan, B.; Mo, X.; Li, P.; Li, B.; Li, Q.; Li, N.; Mo, S.; Ou, Q.; Shen, P.; Wu, B.; Jiang, C. Prevalence and proliferation of antibiotic resistance genes in the subtropical mangrove wetland ecosystem of South China Sea. MicrobiologyOpen 2019, 8, e871. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhang, J.; Chen, X. Environmental drivers of antibiotic resistance gene distribution in mangrove sediments. Sci. Total Environ. 2022, 810, 152268. [Google Scholar]
- Zhou, L.; Qiao, M.; Zhu, L.; et al. Antibiotic resistance genes in coastal sediments: Abundance, distribution, and environmental factors. Sci. Total Environ. 2021, 755, 142544. [Google Scholar]
| Study | Location | Sample Type | ARGs Detected | Detection Method | Notes |
| Liu et al., 2021 | Hainan, China | Sediment | tetA, tetM, sul1 | qPCR | Aquaculture-influenced |
| Palacios et al., 2021 | Mexico | Water | mdt, acr | Metagenomics | Urban runoff |
| Zhao et al., 2022 | South China | Sediment | sul2, bla_CTX-M | Metagenomics | Fine sediment, high organic content |
| Imchen et al., 2020 | India | Sediment | tetM, bla_TEM | 16S + qPCR | Human-impacted mangrove |
| Zhang et al., 2020 | China | Water & Sediment | tetA, sul1, bla_CTX-M, mdt | Metagenomics | High MGE content |
| Hendriksen et al., 2019 | Global | Sediment | Various ARGs | Metagenomics | Global sewage comparison |
| ARG Class | Representative Genes | Reported Occurrence (%) | Notes / Sample Type | References |
| Tetracycline | tetA, tetM | 40–55 | Water, sediment, mangrove sediments | Liu et al., 2021; Imchen et al., 2020; Palacios et al., 2021 |
| Sulfonamide | sul1, sul2 | 25–35 | Urban watershed, community water | Sresung et al., 2024; Hendriksen et al., 2019 |
| β-lactamase | blaTEM, blaSHV, blaCTX-M | 20–30 | Water, sediment, Enterobacteriaceae | Yaikhan et al., 2024; Hinthong et al., 2024 |
| Multidrug | mdtK, acrB | 15–25 | Mangrove sediments, aquaculture | Cabello et al., 2013; Palacios et al., 2021 |
| Aminoglycoside | aac(3)-II, aph(3')-III | 10–20 | Mangrove sediments | Liu et al., 2021; Zhao et al., 2019 |
| Macrolide | ermB, mefA | 5–15 | Sediment, human-associated samples | Intahphuak et al., 2021; Zhao et al., 2022 |
| Quinolone | qnrS, qnrB | 5–10 | Coastal sediments | Zhou et al., 2021; Liu et al., 2023 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




