Submitted:
05 September 2025
Posted:
10 September 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
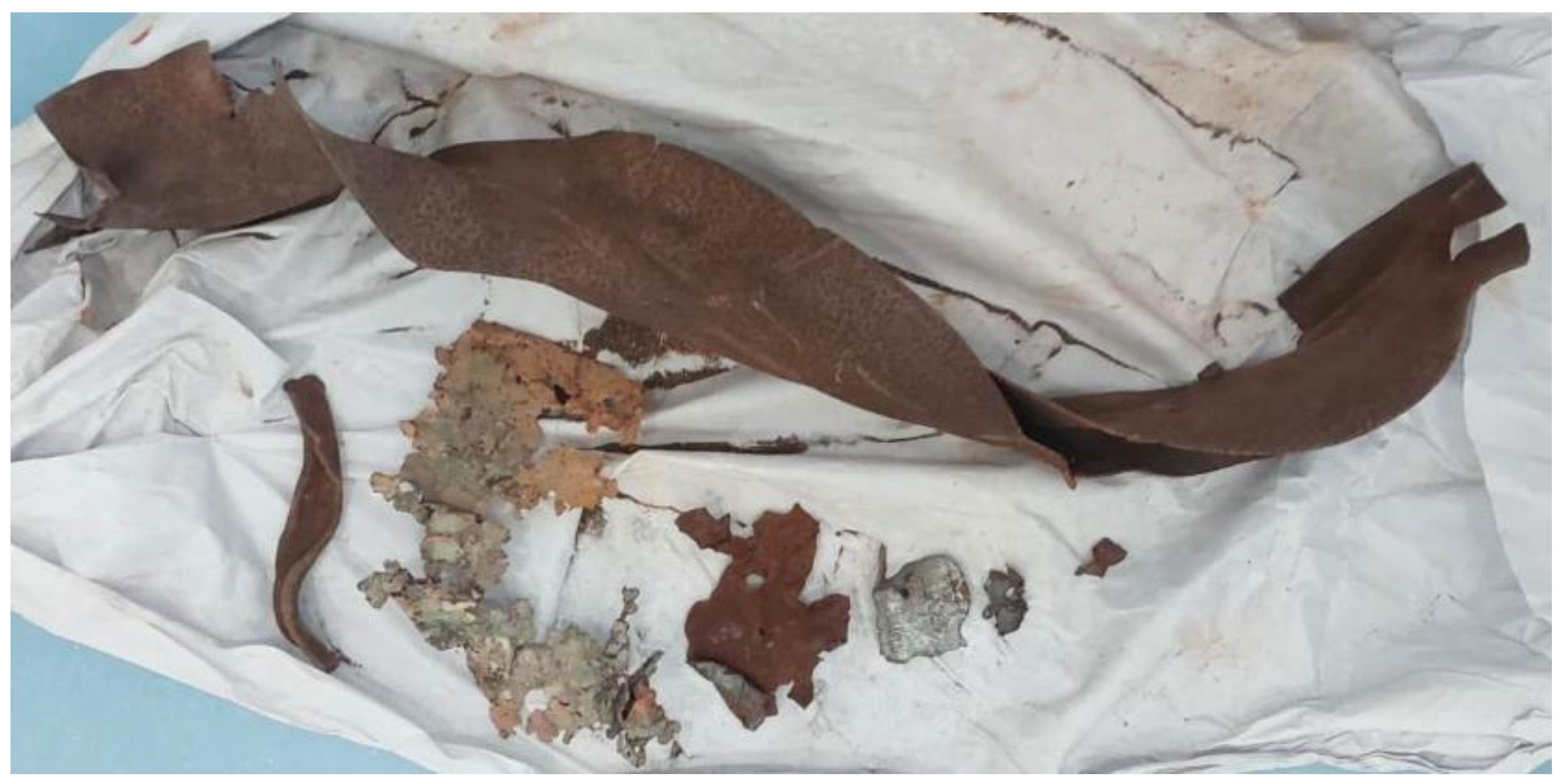
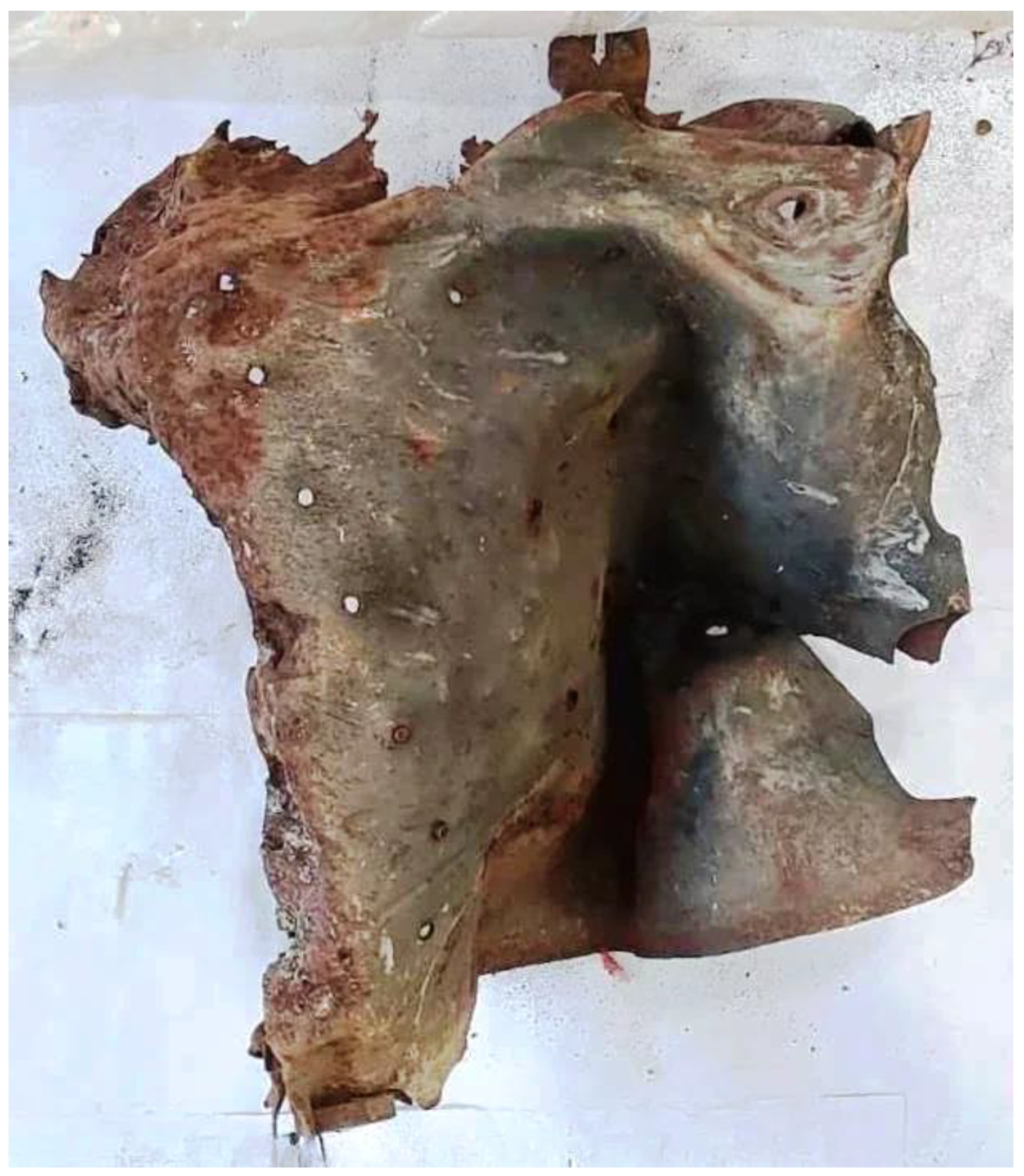
2.1. Evidence Collection from the Crime Scene
2.2. Exhibits Preparation
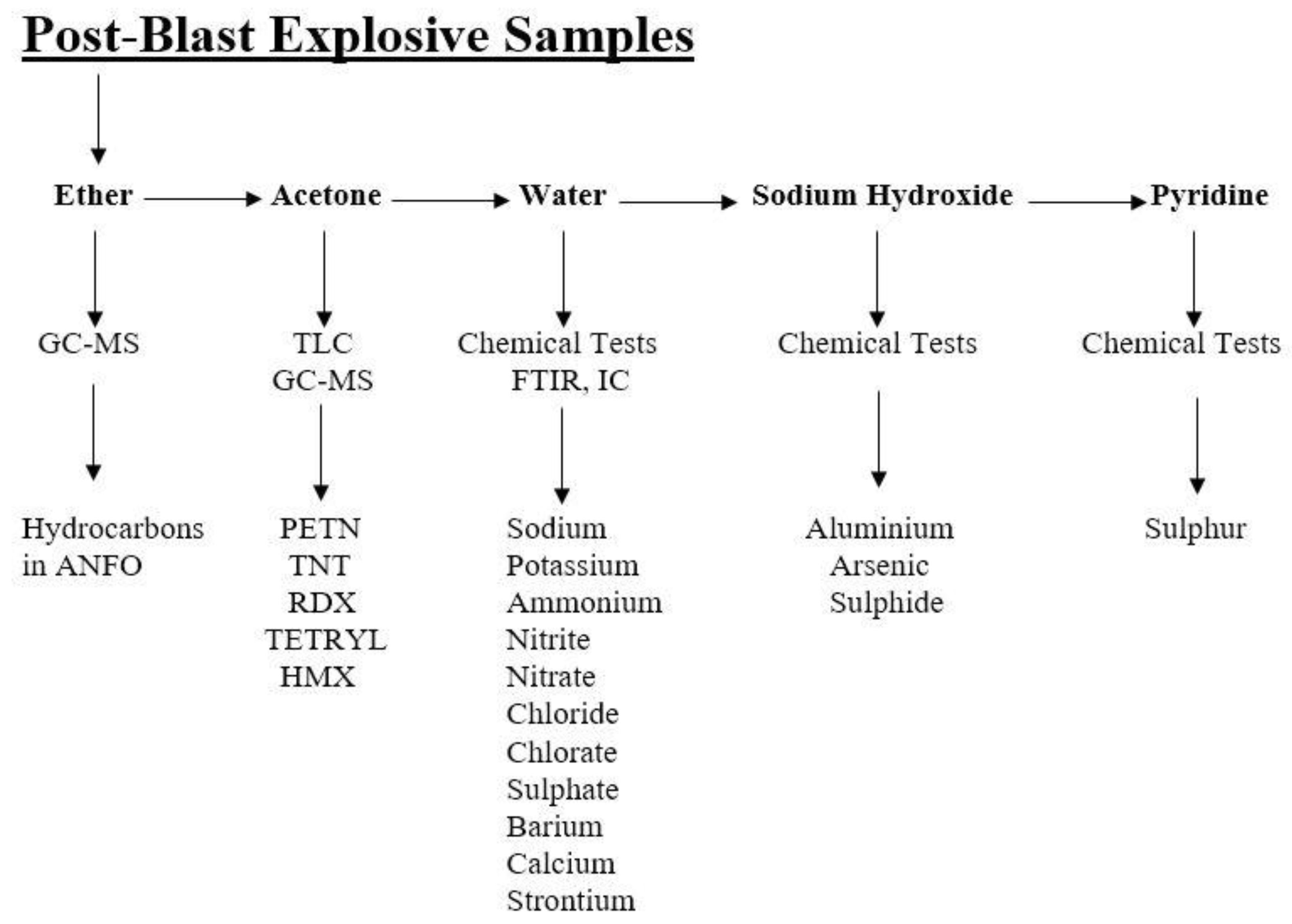
2.3. Extracts Collection and Analysis
2.4. Thin Layer Chromatography
2.5. Fourier Transform Infrared Spectroscopy (FTIR)
2.6. Challenges in the Analysis of Oversized Exhibits
3. Observations
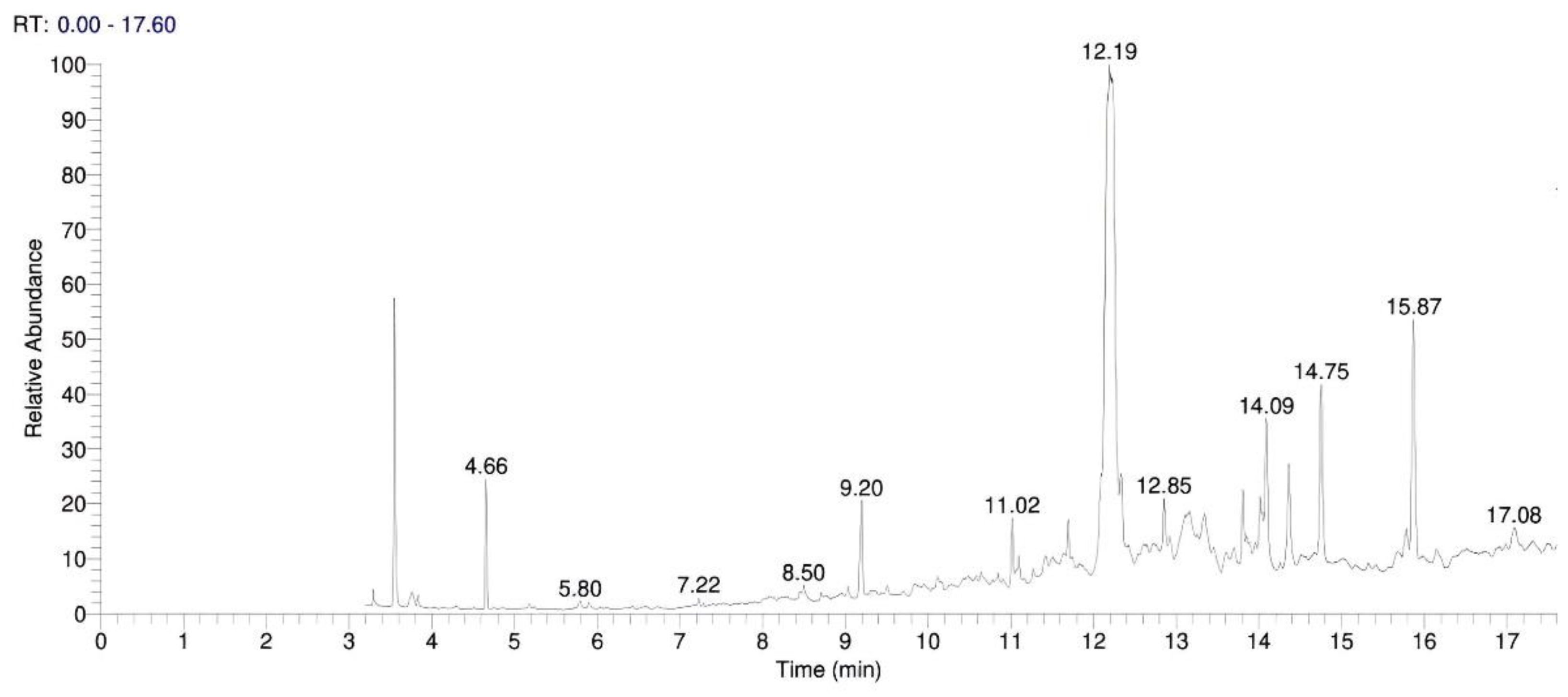
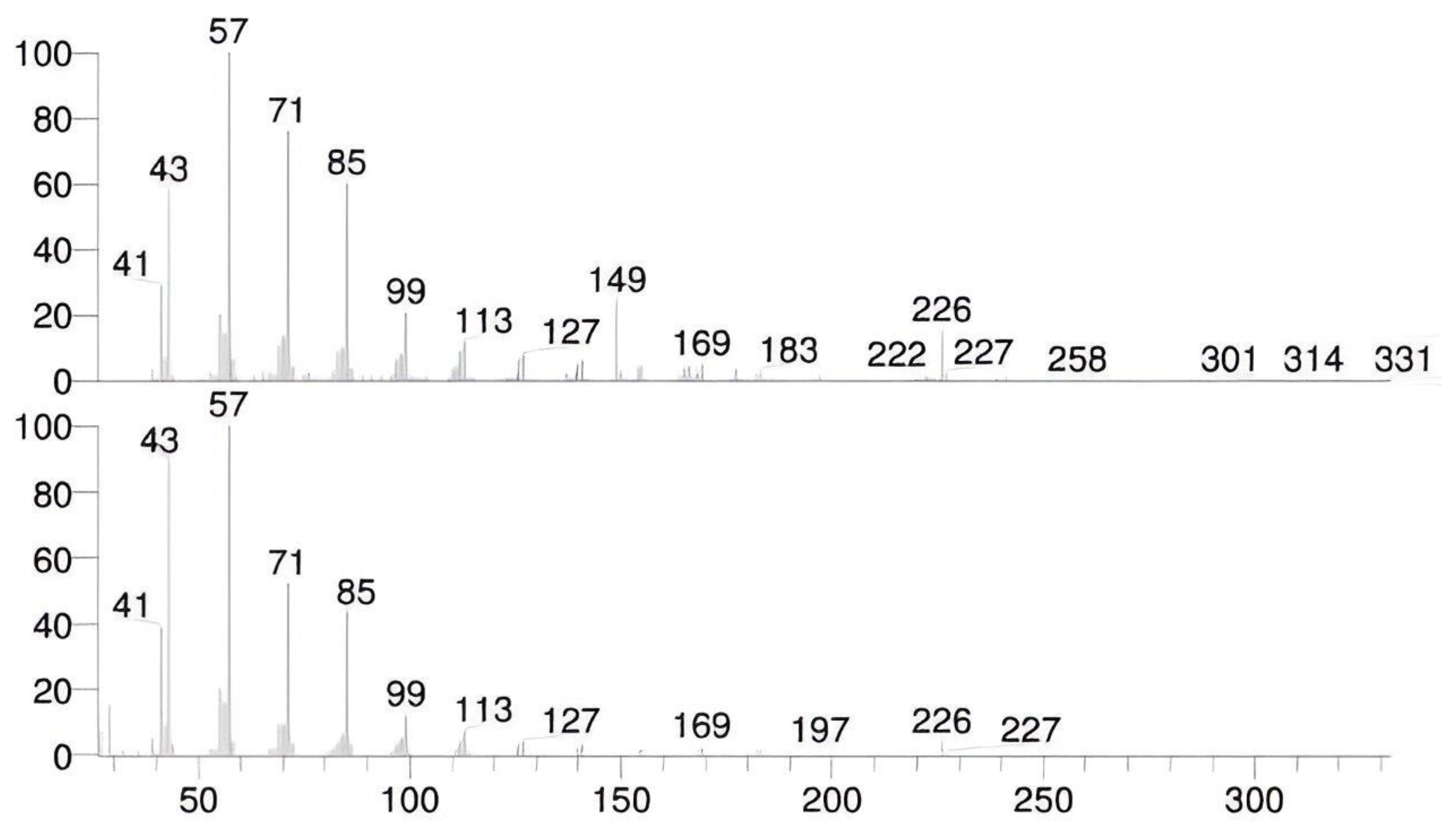
3.1. TLC and GC-MS Examinations
3.2. Chemical Examinations
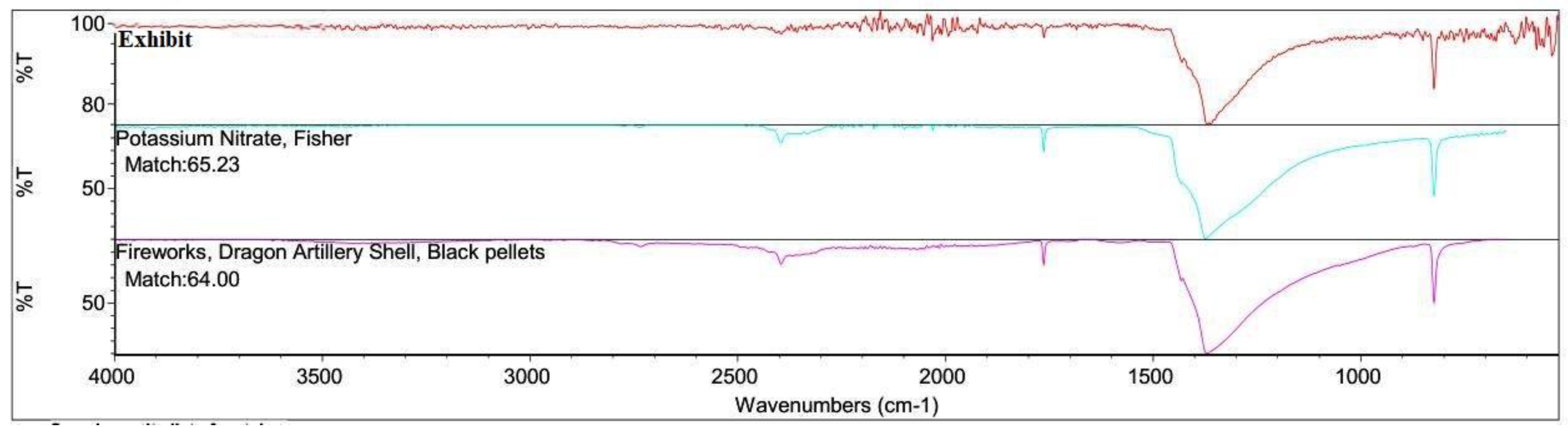
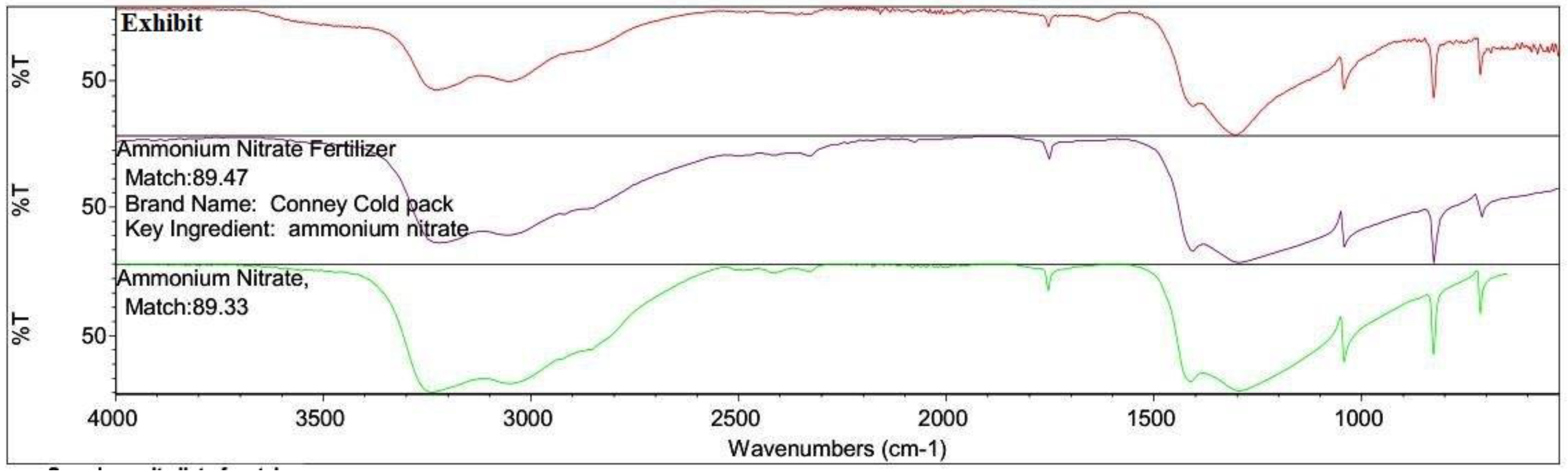
3.3. FTIR Analysis
4. Results and Discussions
4.1. Organic Residue Detection (GC–MS and TLC)
4.2. Inorganic Residue Detection (Chemical Tests and FTIR)
4.3. Workflow Efficacy in Oversized Exhibits
4.4. Forensic Interpretation
5. Conclusions
- The residue recovery efficiency was maximized by adapting the workflows to oversized exhibits, with syringe filtration proving particularly valuable.
- Analytical corroboration across GC–MS, TLC, chemical tests, and FTIR reinforced evidentiary strength and reduced uncertainty.
- Forensic reconstruction confirmed the exclusive use of ANFO, providing insights into the nature of the explosive device and its deployment strategy.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Braga, J. W. B.; Logrado, L. P. L. Evaluation of interferents in sampling materials for analysis of post-explosion residues (explosive emulsion/ANFO) using gas chromatography–mass spectrometry (GC–MS). J. Forensic Sci. 2024, 70(1), 1–9. [Google Scholar]
- De Vooght-Johnson, R. Contamination concerns for GC–MS analysis of explosive residues. Anal. Sci. 2024. Wiley Analytical Science. [Google Scholar]
- Smith, J.; Brown, K. Post-blast explosive residue: A review of formation and dispersion theories and experimental research. RSC Adv. 2014, 4(41), 12345–12360. [Google Scholar] [CrossRef]
- Doe, A.; Lee, P. Sampling of explosive residues: The use of a gelatine-based medium for the recovery of ammonium nitrate. Forensic Sci. Int. 2020, 310, 110234. [Google Scholar] [CrossRef]
- Green, D.; White, S. Recent advances in ambient mass spectrometry of trace explosives. J. Mass Spectrom. 2018, 53(9), 873–889. [Google Scholar] [CrossRef]
- Johnson, T. The application of mass spectrometry to explosive casework: Opportunities and challenges. In Applications of Mass Spectrometry for the Provision of Forensic Intelligence; Royal Society of Chemistry: London, UK, 2017; pp. 201–223. [Google Scholar]
- Yinon, J. Detection of explosives by Fourier transform infrared spectrometry. J. Forensic Sci. 1995, 40(5), 865–870. [Google Scholar] [CrossRef]
- Kumar, D. Explosive device reconstruction through chemical and trace evidence analysis: A homicide case investigation. Preprint (Version 1), Research Square, 17 August 2025. [CrossRef]
- Central Forensic Science Laboratory Pune. Working Procedure Manual; Doc. No.: CFSL/PUNE/WPM/EXPL/11, Issue No. 01; Directorate of Forensic Science Services, Ministry of Home Affairs: Pune, India, 2022.
- Kumar, D.; Prajakta, U. K. Optimizing forensic detection of explosive substances: Extended column analysis of TNT. Int. J. Res. Appl. Sci. Eng. Technol. 2025. [Google Scholar] [CrossRef]
- Kumar, D.; Prajakta, U. K. Thermal decomposition approach for PETN detection in improvised explosive devices. Int. J. Innov. Res. Technol. 2025, 11(12). Braga, J.W.B.; Logrado, L.P.L. Evaluation of interferents in sampling materials for analysis of post-explosion residues (explosive emulsion/ANFO) using gas chromatography–mass spectrometry (GC–MS). J. Forensic Sci. 2024, 70, 1–9. [CrossRef]





| Sl.No | Chemical Test | Target Ion/Analyte | Observation |
| 1 | Silver Nitrate | Chloride | Present |
| 2 | Griess Test | Nitrite | Present |
| 3 | Griess reagent + Zn dust | Nitrate | Present |
| 4 | Aniline sulphate | Chlorate | Absent |
| 5 | Methylene blue indicator | Perchlorate | Absent |
| 6 | Barium chloride | Sulphate | Present |
| 7 | Zinc Uranyl Acetate | Sodium | Absent |
| 8 | Sodium Cobaltinitrate | Potassium | Present |
| 9 | Nessler’s reagent | Ammonium | Present |
| 10 | Magneson-I | Magnesium | Absent |
| 11 | Sodium Rhodizonate | Barium, Calcium, Strontium | Absent |
| 12 | Sodium Nitroprusside | Sulphide (NaOH Extract) | Absent |
| 13 | Alizarine-S | Metallic Aluminium (NaOH Extract) | Absent |
| 14 | Gutzeit’s Test | Arsenic (NaOH Extract) | Absent |
| 15 | Pyridine + NaOH | Elemental Sulphur | Absent |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




