Submitted:
01 September 2025
Posted:
02 September 2025
You are already at the latest version
Abstract
Keywords:
1. The Clinical Burden of Fractures, Chronic Wounds, and Dysregulated Healing
2. TGF-β Signaling in Wound and Fracture Healing: Friend and Foe
3. Extracellular Matrix Protein 1 (ECM1)
3.1. EMC1: a Multifunctional ECM Protein in Skin and Wound Repair
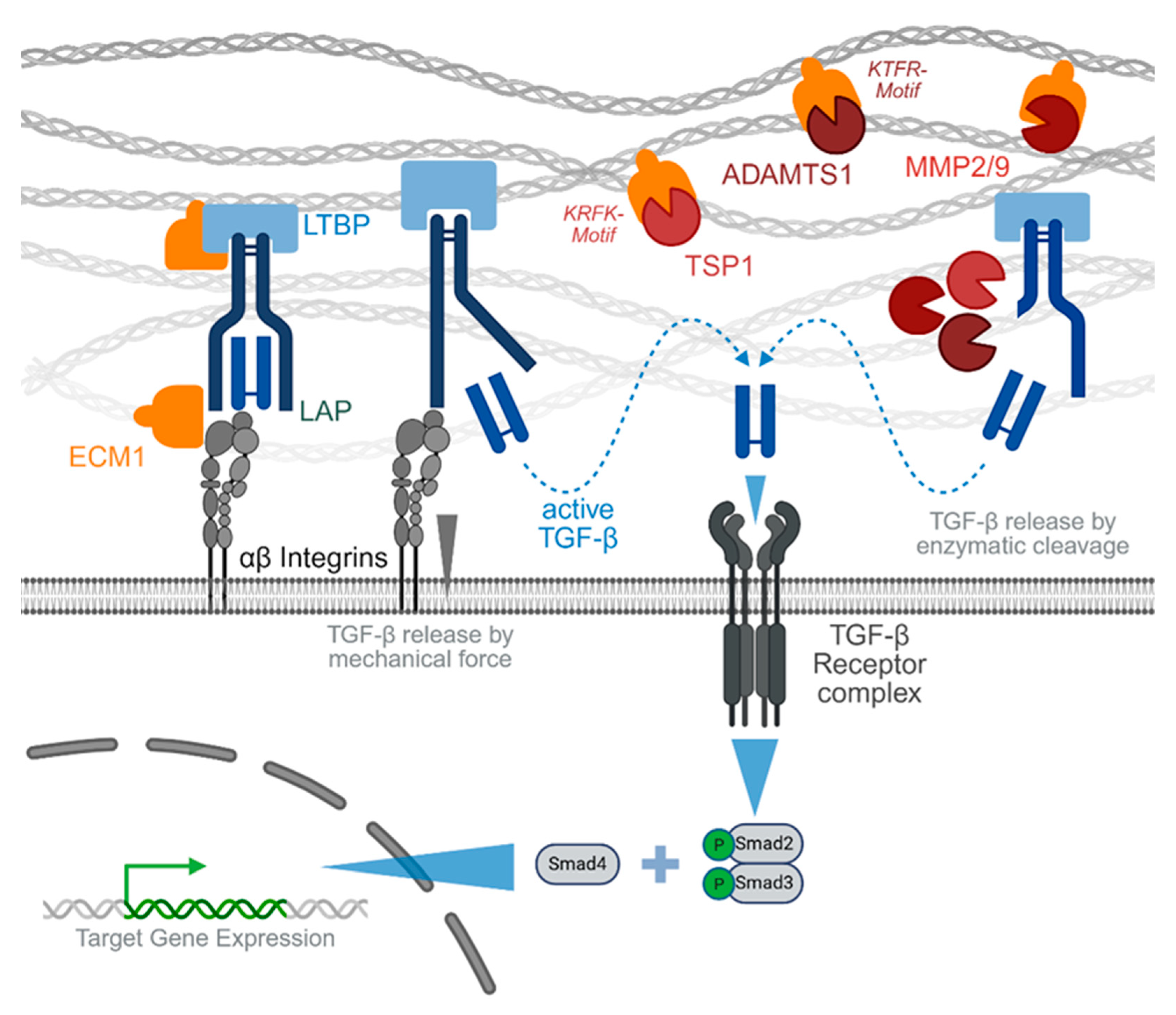
3.2. Interplay of ECM1 with TGF-β Pathway
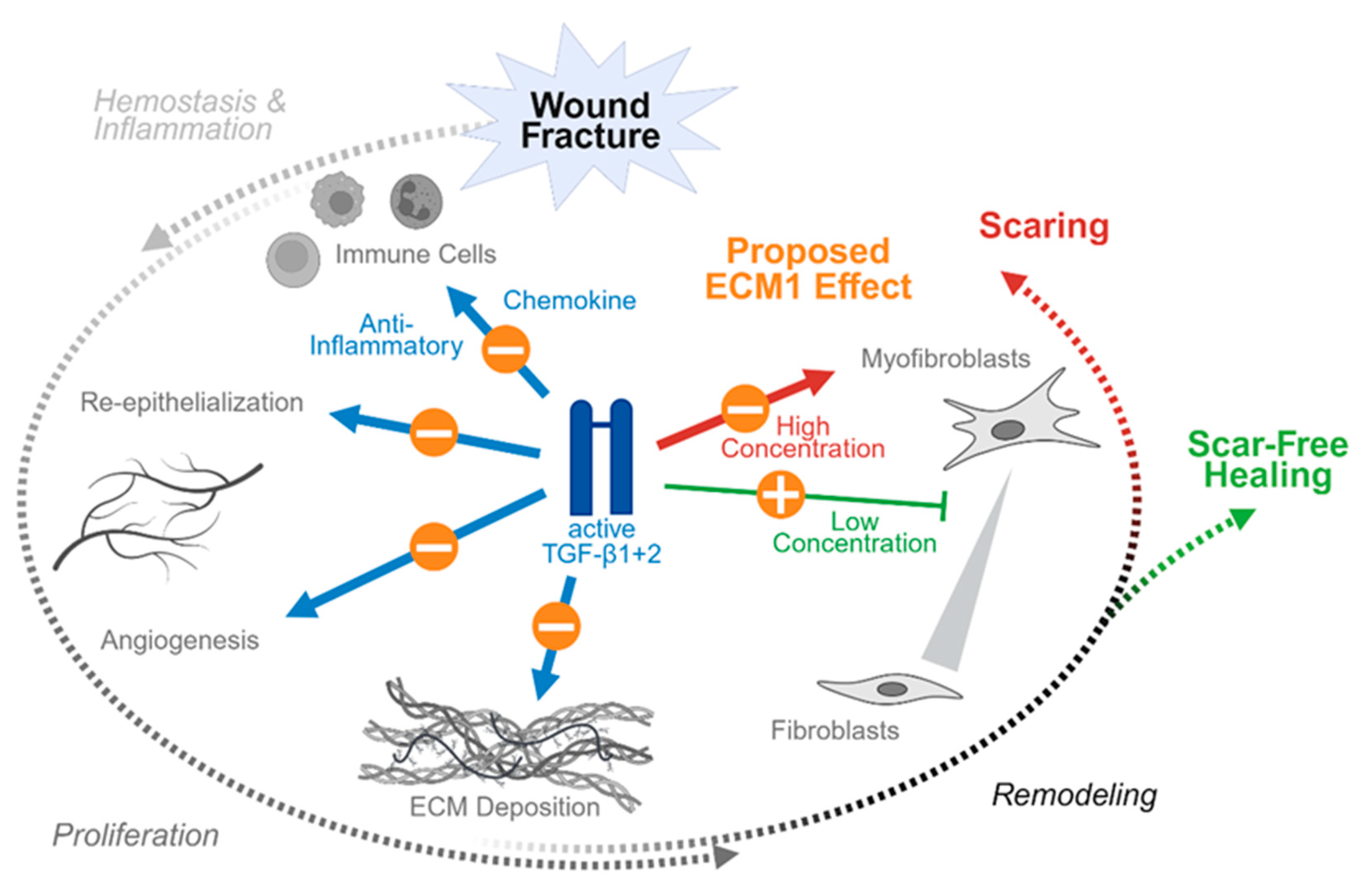
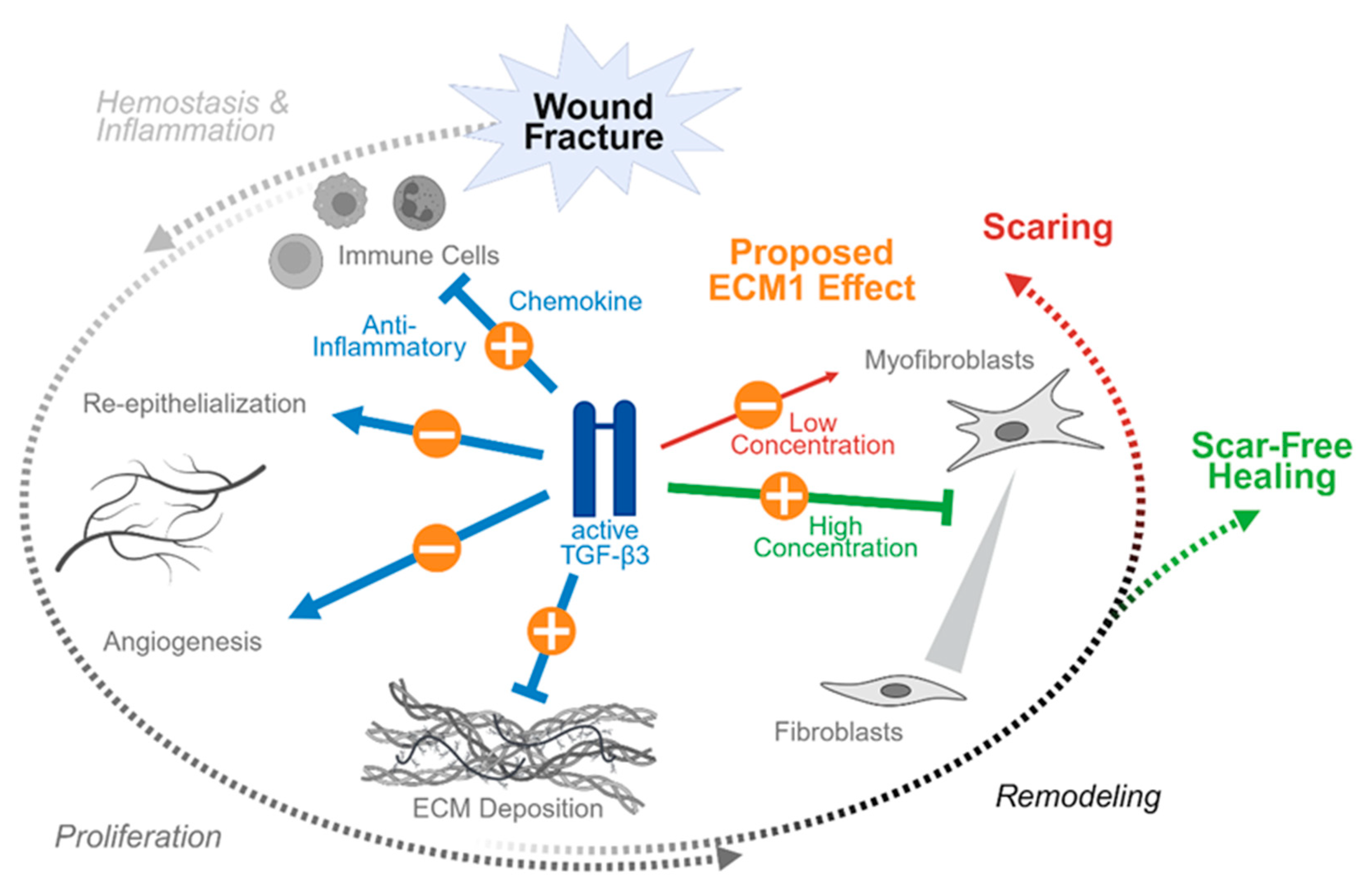
4. Proposed ECM1 Effects During Wound and Fracture Healing
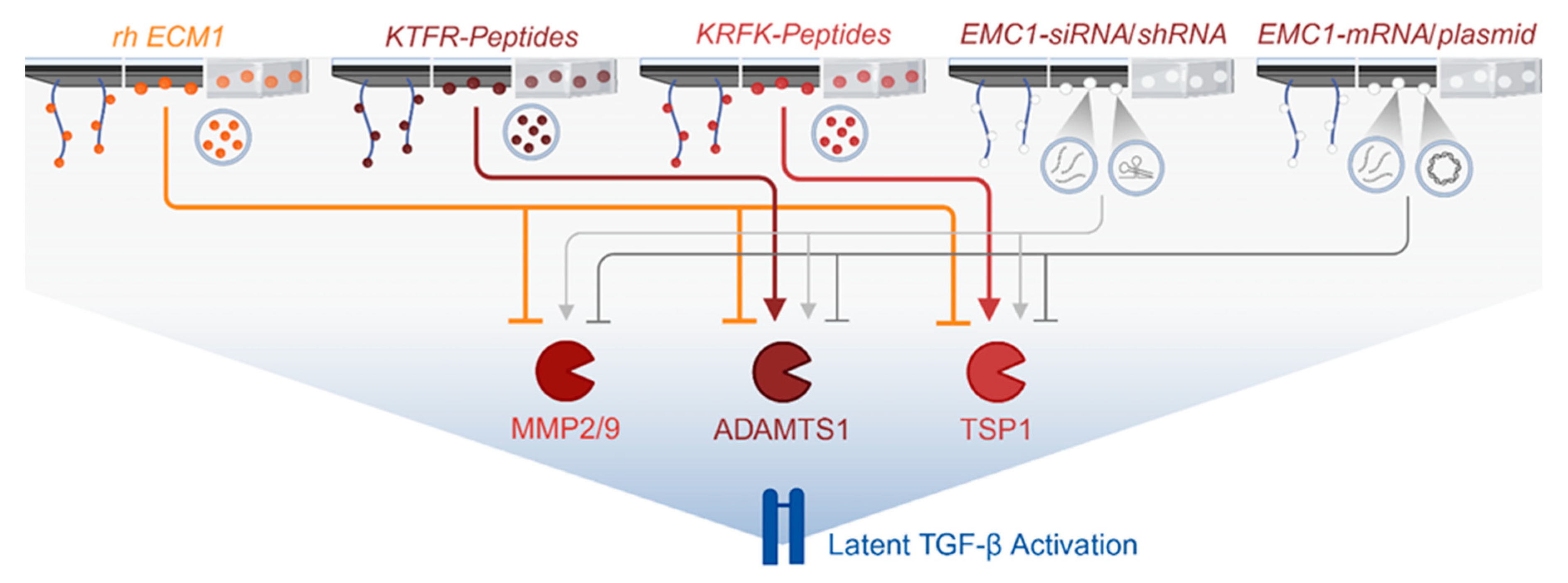
5. Biofunctionalization Strategies
5.1. Protein-, RNA- and DNA-Based Strategies
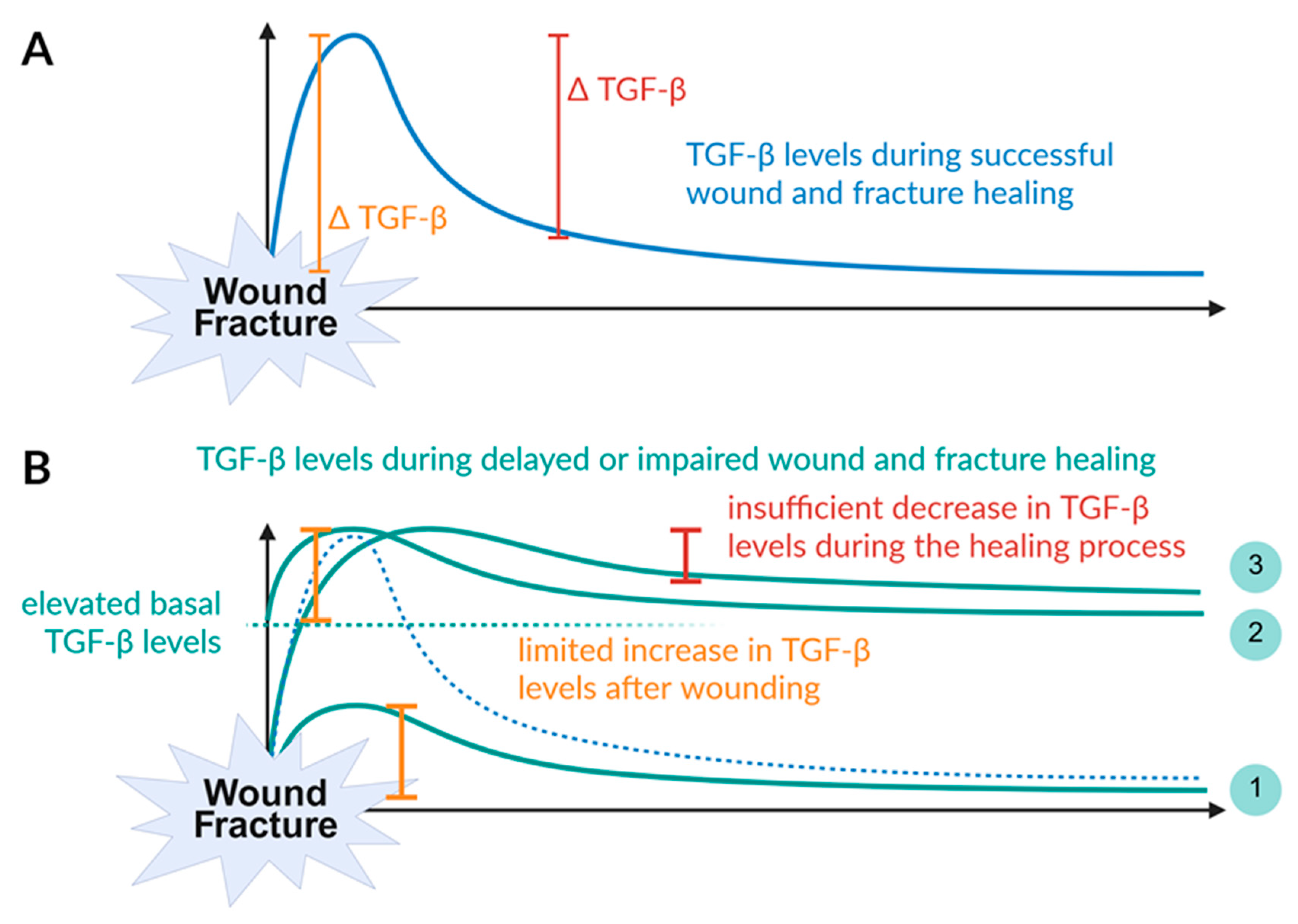
5.2. Alterations in TGF-β Levels Control Phyiological Wound and Fracture Healing ao
5.2.1. Proposed Use of Recombinant Human ECM1
5.2.1. Proposed Use of Tetrapeptide Sequences Targeting the Interaction of ECM1 with Proteases
5.2.1. Proposed Use of RNA and DNA-based Methods to Regulate ECM1 Expression
6. Conclusion
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ADAMTS1 | a disintegrin and metalloproteinase with thrombospondin motifs 1 |
| Alk | activin receptor-like kinase |
| CLD | chronic liver disease |
| DOAJ | Directory of open access journals |
| ECM | extracellular matrix |
| BAMBI | BMP and Activin Membrane-Bound Inhibitor |
| ECM1 | Extracellular matrix protein-1 |
| HTRA1 | high temperature requirement factor A1 |
| KRFK | lysine-phenylalanine-arginine-lysine |
| KTRF | lysine-tryptophan-arginine-phenylalanine |
| LAP | latency-associated peptide |
| LD | Linear dichroism |
| LLC | large latent complex |
| LTBP | latent TGF-β binding protein |
| LTGF-β1 | latent TGF-β1 |
| MDPI | Multidisciplinary Digital Publishing Institute |
| MMP | matrix metalloproteinases |
| RGD | arginine-glycine-aspartic acid |
| shRNAs | small hairpin RNAs |
| siRNAs | small interfering RNAs |
| Smad | small mothers against decapentaplegic |
| TGF-β | transforming growth factor-beta |
| TLA | Three letter acronym |
| TSP1 | thrombospondin 1 |
| VEGF | vascular endothelial growth factor |
References
- C. M. Court-Brown and B. Caesar, “Epidemiology of adult fractures: A review,” Aug. 2006. [CrossRef]
- E. W. Unger et al., “Development of a dynamic fall risk profile in elderly nursing home residents: A free field gait analysis based study,” Arch Gerontol Geriatr, vol. 93, Mar. 2021. [CrossRef]
- O. Ström et al., “Osteoporosis: Burden, health care provision and opportunities in the EU,” Arch Osteoporos, vol. 6, no. 1–2, pp. 59–155, Dec. 2011. [CrossRef]
- A. M. Wu et al., “Global, regional, and national burden of bone fractures in 204 countries and territories, 1990–2019: a systematic analysis from the Global Burden of Disease Study 2019,” Lancet Healthy Longev, vol. 2, no. 9, pp. e580–e592, Sep. 2021. [CrossRef]
- T. A. Einhorn and L. C. Gerstenfeld, “Fracture healing: Mechanisms and interventions,” Jan. 01, 2015, Nature Publishing Group. [CrossRef]
- L. A. Mills, S. A. Aitken, and A. H. R. W. Simpson, “The risk of non-union per fracture: current myths and revised figures from a population of over 4 million adults,” Acta Orthop, vol. 88, no. 4, pp. 434–439, Jul. 2017. [CrossRef]
- B. J. Braun et al., “Increased therapy demand and impending loss of previous residence status after proximal femur fractures can be determined by continuous gait analysis – A clinical feasibility study,” Injury, vol. 50, no. 7, pp. 1329–1332, Jul. 2019. [CrossRef]
- M. Baumgarten et al., “Pressure ulcers in elderly patients with hip fracture across the continuum of care: Clinical investigations,” J Am Geriatr Soc, vol. 57, no. 5, pp. 863–870, May 2009. [CrossRef]
- S. Haleem, G. Heinert, and M. J. Parker, “Pressure sores and hip fractures,” Injury, vol. 39, no. 2, pp. 219–223, Feb. 2008. [CrossRef]
- L. Martinengo et al., “Prevalence of chronic wounds in the general population: systematic review and meta-analysis of observational studies,” Jan. 01, 2019, Elsevier Inc. [CrossRef]
- B. Chan, S. Cadarette, W. Wodchis, J. Wong, N. Mittmann, and M. Krahn, “Cost-of-illness studies in chronic ulcers: a systematic review,” J Wound Care, vol. 26, no. sup4, pp. S4–S14, Apr. 2017. [CrossRef]
- C. K. Sen, “Human Wound and Its Burden: Updated 2020 Compendium of Estimates,” May 01, 2021, Mary Ann Liebert Inc. [CrossRef]
- G. Victoria, B. Petrisor, B. Drew, and D. Dick, “Bone stimulation for fracture healing: What′s all the fuss?,” in Indian Journal of Orthopaedics, Apr. 2009, pp. 117–120. [CrossRef]
- D. J. Hak et al., “Delayed union and nonunions: Epidemiology, clinical issues, and financial aspects,” Injury, vol. 45, no. SUPPL. 2, 2014. [CrossRef]
- K. S. Midwood, L. V. Williams, and J. E. Schwarzbauer, “Tissue repair and the dynamics of the extracellular matrix,” 2004, Elsevier Ltd. [CrossRef]
- R. O. Hynes, “The extracellular matrix: Not just pretty fibrils,” Nov. 27, 2009. [CrossRef]
- F. X. Maquart and J. C. Monboisse, “Extracellular matrix and wound healing,” Pathologie Biologie, vol. 62, no. 2, pp. 91–95, 2014. [CrossRef]
- A.B. Roberts, B. K. McCune, and M. B. Sporn, “TGF-β: Regulation of extracellular matrix,” in Kidney International, 1992, pp. 557–559. [CrossRef]
- S. Liarte, Á. Bernabé-García, and F. J. Nicolás, “Role of TGF-β in Skin Chronic Wounds: A Keratinocyte Perspective,” Jan. 28, 2020, NLM (Medline). [CrossRef]
- M. Pakyari, A. Farrokhi, M. K. Maharlooei, and A. Ghahary, “Critical Role of Transforming Growth Factor Beta in Different Phases of Wound Healing,” Adv Wound Care (New Rochelle), vol. 2, no. 5, pp. 215–224, Jun. 2013. [CrossRef]
- R. W. D. Gilbert, M. K. Vickaryous, and A. M. Viloria-Petit, “Signalling by transforming growth factor beta isoforms in wound healing and tissue regeneration,” Jun. 01, 2016, MDPI Multidisciplinary Digital Publishing Institute. [CrossRef]
- R. F. Diegelmann and M. C. Evans, “WOUND HEALING: AN OVERVIEW OF ACUTE, FIBROTIC AND DELAYED HEALING,” 2004.
- S. Guo and L. A. DiPietro, “Critical review in oral biology & medicine: Factors affecting wound healing,” J Dent Res, vol. 89, no. 3, pp. 219–229, Mar. 2010. [CrossRef]
- T. Velnar, T. Bailey, and V. Smrkolj, “The Wound Healing Process: an Overview of the Cellular and Molecular Mechanisms,” 2009.
- S. M. Wahl et al., “Transforming growth factor type 13 induces monocyte chemotaxis and growth factor production,” 1987. [Online]. Available: https://www.pnas.org.
- S. Ellis, E. J. Lin, and D. Tartar, “Immunology of Wound Healing,” Dec. 01, 2018, Current Medicine Group LLC 1. [CrossRef]
- B. Hinz, “Formation and function of the myofibroblast during tissue repair,” 2007, Nature Publishing Group. [CrossRef]
- S. Fang, N. Pentinmikko, M. Ilmonen, and P. Salven, “Dual action of TGF-β induces vascular growth in vivo through recruitment of angiogenic VEGF-producing hematopoietic effector cells,” Angiogenesis, vol. 15, no. 3, pp. 511–519, Sep. 2012. [CrossRef]
- M. G. Tonnesen, 2 Xiaodong Feng, and R. A. F. Clark, “Angiogenesis in Wound Healing,” 2000.
- C. J. Morrison, G. S. Butler, D. Rodríguez, and C. M. Overall, “Matrix metalloproteinase proteomics: substrates, targets, and therapy,” Oct. 2009. [CrossRef]
- T. Zhang et al., “Current potential therapeutic strategies targeting the TGF-β/Smad signaling pathway to attenuate keloid and hypertrophic scar formation,” Sep. 01, 2020, Elsevier Masson s.r.l. [CrossRef]
- S. Ehnert et al., “Transforming growth factor β1inhibits bone morphogenic protein (BMP)-2 and BMP-7 signaling via upregulation of Ski-related novel protein N (SnoN): Possible mechanism for the failure of BMP therapy?,” BMC Med, vol. 10, Sep. 2012. [CrossRef]
- Y. Guang CHEN and A. Ming MENG, “Negative regulation of TGF-β signaling in development,” 2004. [Online]. Available: http://www.cell-research.com.
- D. Onichtchouk et al., “Silencing of TGF-β signalling by the pseudoreceptor BAMBI,” Nature, vol. 401, no. 6752, pp. 480–485, Sep. 1999. [CrossRef]
- Y. Fan et al., “BAMBI elimination enhances alternative TGF-b signaling and glomerular dysfunction in diabetic mice,” Diabetes, vol. 64, no. 6, pp. 2220–2233, Jun. 2015. [CrossRef]
- M. K. Lichtman, M. Otero-Vinas, and V. Falanga, “Transforming growth factor beta (TGF-β) isoforms in wound healing and fibrosis,” Mar. 01, 2016, Blackwell Publishing Inc. [CrossRef]
- S. Sercu et al., “Interaction of extracellular matrix protein 1 with extracellular matrix components: ECM1 is a basement membrane protein of the skin,” Journal of Investigative Dermatology, vol. 128, no. 6, pp. 1397–1408, 2008. [CrossRef]
- S. Sercu et al., “ECM1 interacts with fibulin-3 and the beta 3 chain of laminin 332 through its serum albumin subdomain-like 2 domain,” Matrix Biology, vol. 28, no. 3, pp. 160–169, Apr. 2009. [CrossRef]
- Y. L. Liu, Z. Y. O. Zhang, and X. M. Chen, “A Sporadic Family of Lipoid Proteinosis with Novel ECM1 Gene Mutations,” Clin Cosmet Investig Dermatol, vol. 17, pp. 885–889, 2024. [CrossRef]
- F. Link et al., “ECM1 Attenuates Hepatic Fibrosis by Interfering with Mediators of Latent TGF-β1 Activation,” Dec. 12, 2023. [CrossRef]
- L. E. Tracy, R. A. Minasian, and E. J. Caterson, “Extracellular Matrix and Dermal Fibroblast Function in the Healing Wound,” Mar. 01, 2016, Mary Ann Liebert Inc. [CrossRef]
- P. Rousselle, M. Montmasson, and C. Garnier, “Extracellular matrix contribution to skin wound re-epithelialization,” Jan. 01, 2019, Elsevier B.V. [CrossRef]
- C. Frantz, K. M. Stewart, and V. M. Weaver, “The extracellular matrix at a glance,” Dec. 15, 2010. [CrossRef]
- N. Beaufort et al., “Cerebral small vessel disease-related protease HtrA1 processes latent TGF-β binding protein 1 and facilitates TGF-β signaling,” Proc Natl Acad Sci U S A, vol. 111, no. 46, pp. 16496–16501, Nov. 2014. [CrossRef]
- G. A. Secker, A. J. Shortt, E. Sampson, Q. P. Schwarz, G. S. Schultz, and J. T. Daniels, “TGFβ stimulated re-epithelialisation is regulated by CTGF and Ras/MEK/ERK signalling,” Exp Cell Res, vol. 314, no. 1, pp. 131–142, Jan. 2008. [CrossRef]
- L. F. Bonewald and S. L. Dallas, “Role of active and latent transforming growth factor β in bone formation,” J Cell Biochem, vol. 55, no. 3, pp. 350–357, 1994. [CrossRef]
- Y. Shi and J. Massagué, “Review Mechanisms of TGF-Signaling from Cell Membrane to the Nucleus have been observed in both TGF-family receptors and the Smad proteins. The TGF-type II receptor is inacti-vated by mutation in most human gastrointestinal can,” 2003.
- M. Shi et al., “Latent TGF-β structure and activation,” Nature, vol. 474, no. 7351, pp. 343–351, Jun. 2011. [CrossRef]
- R. W. D. Gilbert, M. K. Vickaryous, and A. M. Viloria-Petit, “Signalling by transforming growth factor beta isoforms in wound healing and tissue regeneration,” Jun. 01, 2016, MDPI Multidisciplinary Digital Publishing Institute. [CrossRef]
- A. Meyer et al., “Platelet TGF-β1 contributions to plasma TGF-β1, cardiac fibrosis, and systolic dysfunction in a mouse model of pressure overload,” Blood, vol. 119, no. 4, pp. 1064–1074, Jan. 2012. [CrossRef]
- D. J. Grainger, D. E. Mosedale, and J. C. Metcalfe, “TGF-b in blood: a complex problem.” [Online]. Available: www.elsevier.com/locate/cytogfr.
- T. A. Mustoe, G. F. Pierce, A. Thomason, P. Gramates, M. B. Sporn, and T. F. Deuel, “Accelerated Healing of Incisional Wounds in Rats Induced by Transforming Growth Factor-β,” Science (1979), vol. 237, no. 4820, pp. 1333–1336, Sep. 1987. [CrossRef]
- M. Le et al., “Transforming Growth Factor Beta 3 Is Required for Excisional Wound Repair In Vivo,” PLoS One, vol. 7, no. 10, Oct. 2012. [CrossRef]
- M. E. Mercado-Pimentel and R. B. Runyan, “Multiple transforming growth factor-β isoforms and receptors function during epithelial-mesenchymal cell transformation in the embryonic heart,” in Cells Tissues Organs, Jun. 2007, pp. 146–156. [CrossRef]
- K. M. Welch-Reardon et al., “Angiogenic sprouting is regulated by endothelial cell expression of Slug,” J Cell Sci, vol. 127, no. 9, pp. 2017–2028, 2014. [CrossRef]
- S. Piera-Velazquez, F. A. Mendoza, and S. A. Jimenez, “Endothelial to Mesenchymal Transition (EndoMT) in the pathogenesis of human fibrotic diseases,” Apr. 11, 2016, MDPI. [CrossRef]
- G. S. Chin et al., “Differential Expression of Transforming Growth Factor-β Receptors I and II and Activation of Smad 3 in Keloid Fibroblasts,” Plast Reconstr Surg, vol. 108, no. 2, pp. 423–429, Aug. 2001. [CrossRef]
- M. Shah, D. M. Foreman, and M. W. J. Ferguson, “Neutralisation of TGF-β1 and TGF-β2 or exogenous addition of TGF-β3 to cutaneous rat wounds reduces scarring,” J Cell Sci, vol. 108, no. 3, pp. 985–1002, Mar. 1995. [CrossRef]
- G. Serini and G. Gabbiani, “Modulation of α-smooth muscle actin expression in fibroblasts by transforming growth factor-β isoforms: An in vivo and in vitro study,” Wound Repair and Regeneration, vol. 4, no. 2, pp. 278–287, 1996. [CrossRef]
- O. Khorkova, J. Stahl, A. Joji, C. H. Volmar, and C. Wahlestedt, “Amplifying gene expression with RNA-targeted therapeutics,” Jul. 01, 2023, Nature Research. [CrossRef]
- H. C. Tsai et al., “Current strategies employed in the manipulation of gene expression for clinical purposes,” Dec. 01, 2022, BioMed Central Ltd. [CrossRef]
- S. Mali, “Delivery systems for gene therapy,” Indian J Hum Genet, vol. 19, no. 1, p. 3, 2013. [CrossRef]
- G. Zimmermann et al., “TGF-β1 as a marker of delayed fracture healing,” Bone, vol. 36, no. 5, pp. 779–785, 2005. [CrossRef]
- M. Pujani, V. Chauhan, K. Singh, S. Rastogi, C. Agarwal, and K. Gera, “The effect and correlation of smoking with platelet indices, neutrophil lymphocyte ratio and platelet lymphocyte ratio,” Hematol Transfus Cell Ther, vol. 43, no. 4, pp. 424–429, Oct. 2021. [CrossRef]
- S. Kanzler et al., “Prediction of progressive liver fibrosis in hepatitis C infection by serum and tissue levels of transforming growth factor-β,” J Viral Hepat, vol. 8, no. 6, pp. 430–437, 2001. [CrossRef]
- A. M. Gressner, “Roles of TGF-beta in hepatic fibrosis,” Frontiers in Bioscience, vol. 7, no. 4, p. A812, 2002. [CrossRef]
- Y. C. Qiao et al., “Changes of Regulatory T Cells and of Proinflammatory and Immunosuppressive Cytokines in Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis,” 2016, Hindawi Limited. [CrossRef]
- J. L. Chen et al., “Specific targeting of TGF-β family ligands demonstrates distinct roles in the regulation of muscle mass in health and disease,” Proc Natl Acad Sci U S A, vol. 114, no. 26, pp. E5266–E5275, Jun. 2017. [CrossRef]
- L. B. Shi et al., “Transforming growth factor beta1 from endometriomas promotes fibrosis in surrounding ovarian tissues via Smad2/3 signaling†,” Biol Reprod, vol. 97, no. 6, pp. 873–882, Dec. 2017. [CrossRef]
- S. Ehnert et al., “Factors circulating in the blood of type 2 diabetes mellitus patients affect osteoblast maturation - Description of a novel in vitro model,” Exp Cell Res, vol. 332, no. 2, pp. 247–258, Mar. 2015. [CrossRef]
- T. Freude et al., “Hyperinsulinemia reduces osteoblast activity in vitro via upregulation of TGF-β,” J Mol Med, vol. 90, no. 11, pp. 1257–1266, Nov. 2012. [CrossRef]
- F. H. Epstein, W. A. Border, and N. A. Noble, “Transforming Growth Factor β in Tissue Fibrosis,” New England Journal of Medicine, vol. 331, no. 19, pp. 1286–1292, Nov. 1994. [CrossRef]
- A. V. Fedulov et al., “Serum TGF-Beta 1 and TNF-Alpha Levels and Cardiac Fibrosis in Experimental Chronic Renal Failure,” Immunol Invest, vol. 34, no. 2, pp. 143–152, Jan. 2005. [CrossRef]
- A. Leask, “TGFβ, cardiac fibroblasts, and the fibrotic response,” May 01, 2007. [CrossRef]
- S. Ehnert et al., “TGF-β1 impairs mechanosensation of human osteoblasts via HDAC6-mediated shortening and distortion of primary cilia,” J Mol Med, vol. 95, no. 6, pp. 653–663, Jun. 2017. [CrossRef]
- L. Lu et al., “The Temporal Effects of Anti-TGF-β1, 2, and 3 Monoclonal Antibody on Wound Healing and Hypertrophic Scar Formation,” J Am Coll Surg, vol. 201, no. 3, pp. 391–397, Sep. 2005. [CrossRef]
- E. Traversa et al., “Tuning hierarchical architecture of 3D polymeric scaffolds for cardiac tissue engineering,” in Journal of Experimental Nanoscience, Jun. 2008, pp. 97–110. [CrossRef]
- C. Chocarro-Wrona, E. López-Ruiz, M. Perán, P. Gálvez-Martín, and J. A. Marchal, “Therapeutic strategies for skin regeneration based on biomedical substitutes,” Mar. 01, 2019, Blackwell Publishing Ltd. [CrossRef]
- J. S. Lee, D. Suarez-Gonzalez, and W. L. Murphy, “Mineral coatings for temporally controlled delivery of multiple proteins,” Advanced Materials, vol. 23, no. 37, pp. 4279–4284, Oct. 2011. [CrossRef]
- S. Talebian et al., “Biopolymeric Coatings for Local Release of Therapeutics from Biomedical Implants,” Apr. 26, 2023, John Wiley and Sons Inc. [CrossRef]
- S. Saeed et al., “Flexible Topical Hydrogel Patch Loaded with Antimicrobial Drug for Accelerated Wound Healing,” Gels, vol. 9, no. 7, Jul. 2023. [CrossRef]
- P. Thongpon, M. Tang, and Z. Cong, “Peptide-Based Nanoparticle for Tumor Therapy,” Jun. 01, 2025, Multidisciplinary Digital Publishing Institute (MDPI). [CrossRef]
- J. H. Yen, C. C. Chang, T. Y. Wu, C. H. Yang, H. J. Hsu, and J. W. Liou, “Therapeutic peptides and their delivery using lipid-based nanoparticles,” Jul. 01, 2025, Wolters Kluwer Medknow Publications. [CrossRef]
- W. Fan et al., “ECM1 Prevents Activation of Transforming Growth Factor β, Hepatic Stellate Cells, and Fibrogenesis in Mice,” Gastroenterology, vol. 157, no. 5, pp. 1352-1367.e13, Nov. 2019. [CrossRef]
- D. Zhang et al., “ECM1 protects against liver steatosis through PCBP1-mediated iron homeostasis,” Hepatology, May 2025. [CrossRef]
- Q. Liu et al., “ECM1 modified HF-MSCs targeting HSC attenuate liver cirrhosis by inhibiting the TGF-β/Smad signaling pathway,” Cell Death Discov, vol. 8, no. 1, Dec. 2022. [CrossRef]
- M. A. Cichon and D. C. Radisky, “Extracellular matrix as a contextual determinant of transforming growth factor-b signaling in epithelial-mesenchymal transition and in cancer http://www.tandfonline.com/doi/pdf/10.4161/19336918.2014.972788,” Nov. 01, 2014, Landes Bioscience. [CrossRef]
- J. H. Yoon, J. Kim, and M. Gorospe, “Long noncoding RNA turnover,” Oct. 15, 2015, Elsevier. [CrossRef]
- K. D. Kovacevic, J. C. Gilbert, and B. Jilma, “Pharmacokinetics, pharmacodynamics and safety of aptamers,” Sep. 01, 2018, Elsevier B.V. [CrossRef]
- G. Hutvagner and M. J. Simard, “Argonaute proteins: Key players in RNA silencing,” Jan. 2008. [CrossRef]
- S. Subramanya, S. S. Kim, N. Manjunath, and P. Shankar, “RNA interference-based therapeutics for human immunodeficiency virus HIV-1 treatment: Synthetic siRNA or vector-based shRNA?,” Feb. 2010. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



