1. Introduction
One in eight women of the world population aged 16-74 years had experienced infertility, defined by unsuccessfully attempting pregnancy for a year or longer, during their life [
1].
The treatment of female infertility has improved substantially over the last 80 years with the advent of assisted reproduction. The main milestones in this continuous process were marked by the discovery of ovarian stimulation [
2,
3], intrauterine insemination [
4], ultrasound imaging of ovarian follicles [
5], in vitro fertilization (IVF) [
6], and the development of micromanipulation-assisted fertilization techniques [
7,
8]. Progressive improvements were also achieved in each of the above fields across time. However, in spite of these achievements, the success rates of assisted reproduction still remain relatively low [
9].
The causes of female infertility may be associated with impaired function of the ovaries (ovarian factor), the uterus (uterine factor), aberrant reaction of the immune system to the embryo (immunological factor), and persistent chronic disease of the female genital tract, mainly endometriosis and adenomyosis. Melatonin was first used to alleviate female infertility in the first decade of this century, when it was reported to improve pregnancy rates when used as an adjuvant in IVF treatment [
10]. Subsequent review articles highlighted the possible role of melatonin in different issues of the female reproductive function and its potential use for human infertility treatment [
11,
12,
13,
14,
15,
16,
17,
18,
19,
20,
21]. The present review is focused on the latest data, both from experimental animal research and human clinical studies, to explain how melatonin could be used as a biomedicine to improve human female fertility, while still acknowledging the pioneering articles published previously.
2. Different Facets of Melatonin Action
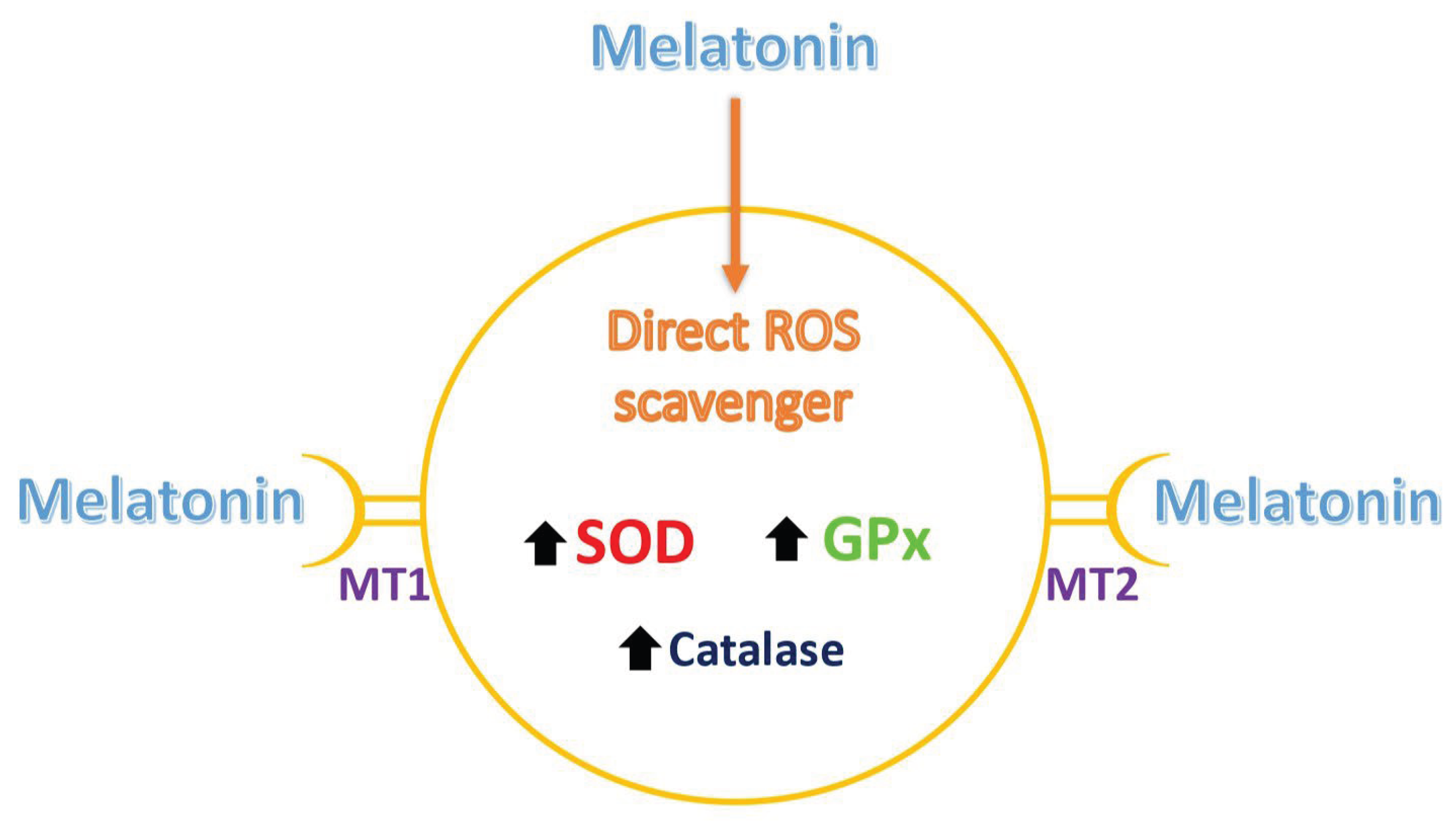
Melatonin is a multifaceted agent combining different molecular mechanisms of actions in cells and tissues. Actually, melatonin is a hormone acting at its complementary receptors in target cells, a direct antioxidant agent, and an immunomodulator influencing various aspects of the immune system
2.1. Melatonin as a Hormone
Melatonin (N-acetyl-5-methoxytryptamine), first isolated in 1958 as a neuro-hormone mainly synthesised and secreted from the pineal gland, is now known to be secreted by a number of other cell types, including those of gastrointestinal tract, brain, eye, lungs, skin, kidney, liver, thyroid, thymus, pancreas, immune system and reproductive organs (reviewed in Fernando and Rombauts [
10]) In mammals, melatonin acts at two types of receptors MT1 and MT2, located at the cell membrane and using a G protein pathway for further signal transduction [
22]. Since the melatonin receptors are present in brain neurons, and its production is linked to the light/dark cycle, its most notorious effect is associated with the regulation of circadian sleep-wake rhythm [
23]. However, a number of studies have shown that melatonin can act through its receptors in a variety of cell types, producing various kinds of effects, in addition to acting as a direct antioxidant [
24]. Hence, it is sometimes difficult to distinguish between the receptor-mediated and direct actions of melatonin in a particular condition (
Figure 1).
2.2. Melatonin as an Antioxidant
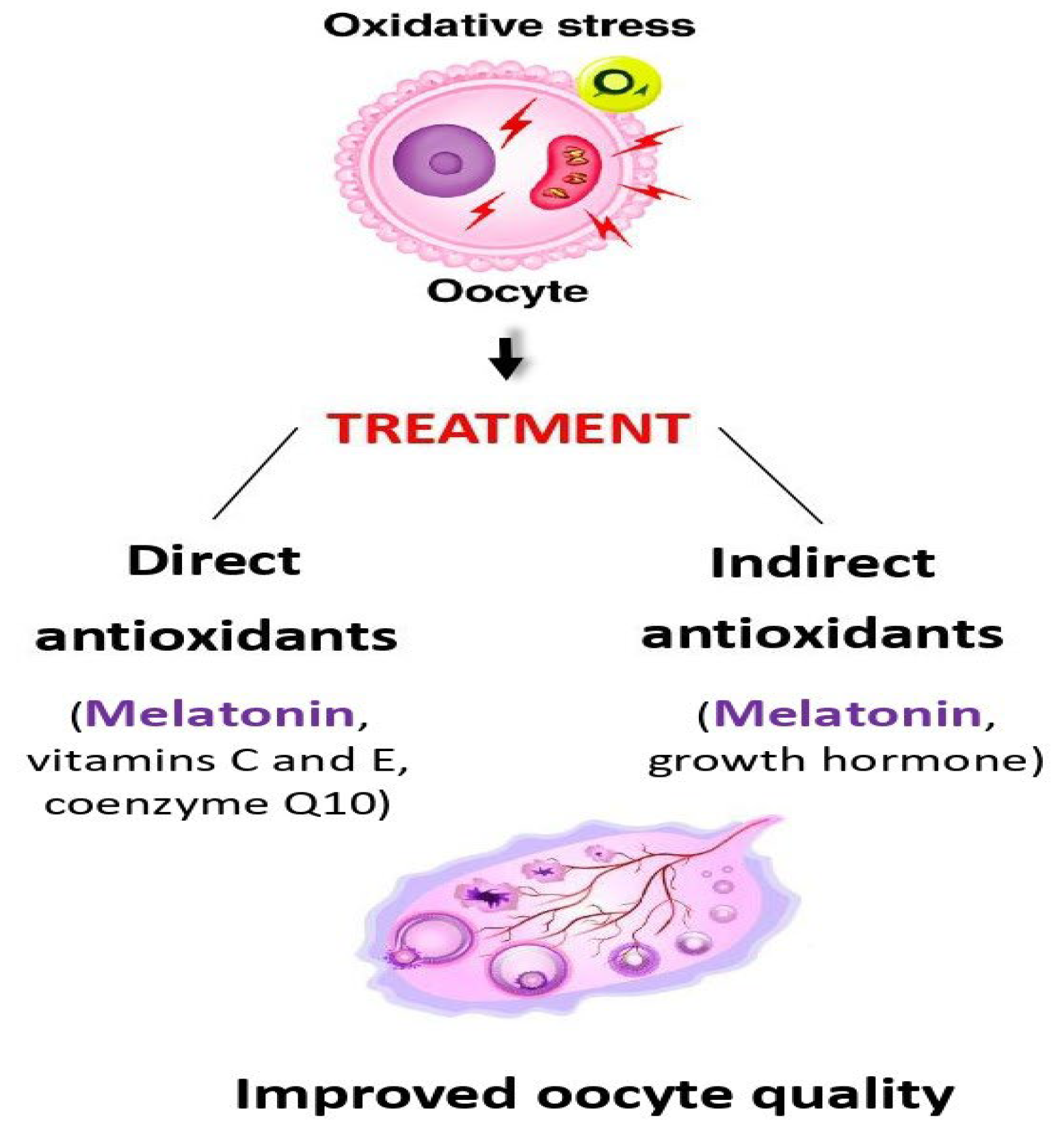
Owing to its amphiphilic nature, melatonin can easily penetrate into cells and exert effects independently of its receptor-mediated action. Antioxidant effect is one of its utmost receptor-independent actions. Unlike most of other antioxidant agents, melatonin does not switch between the oxidation-preventing and oxidation-supporting actions because, once the antioxidant effect executed, melatonin does not come back to its original condition in which it could be both the receptor (antioxidant) and donor (pro-oxidant) of free oxygen radicals [
25]. Consequently, melatonin does not switch between the antioxidant and pro-oxidant actions, and it can be employed as an antioxidant in combination with other molecules exerting the same effect (
Figure 2).
2.2. Melatonin as an Immunomodulator
In addition to its action as an antioxidant, melatonin also directly affects the immune system, making it more “intelligent” and less invasive towards healthy cells, first mentioned in 2022 [
26]. In fact, as exposed in the above article, melatonin attenuates the “primitive” innate immune response, destroying both affected cells and those unaffected in their neighbourhood, and favours a more “intelligent” way of action by reprogramming the activity of the natural killer (NK) cells. This action of melatonin is also important to mitigate symptoms of chronic diseases of the female genital tract (see below).
3. Melatonin Use in Different Types of Female Infertility
Melatonin began to be used to treat human female infertility in the late 2000s (reviewed in Tamura et al. [
27], mainly in order to improve oocyte quality through its antioxidant action. Since then, in addition to its usefulness in the treatment of the ovarian factor, melatonin has also proven efficient in cases of the uterine factor, endometriosis and adenomyosis, and repeated implantation failure and pregnancy loss due to incompatibility between the immune cells within the uterine cavity and the antigenic status of the embryo to implant, and its usefulness also goes beyond the early embryonic development, encompassing obstetrics issue and lactation [
21].
3.1. Ovarian Factor
The original use of melatonin to treat the ovarian factor was directed to women with a low ovarian reserve. In fact both the age-related and premature ovarian insufficiency are associated with an increased production and/or decreased inactivation of reactive oxygen species (ROS) within the cells of the ovarian follicles [
28]. Both types of these cells, granulosa cells [
29] and oocytes [
30], express the melatonin receptors (MT1 and MT2) on their surface and the binding of melatonin to these receptors promotes the production of intracellular antioxidant enzymes, such as glutathione peroxidase, superoxide dismutase and catalase [
31,
32] (
Figure 1).
In addition, because of its amphiphilic nature, melatonin penetrates easily into these cells where it acts as a direct ROS scavenger [
25]. Studies have shown that melatonin not only improves oocyte quantity and quality, but also is likely to slow down ovarian aging [
21]. This latter action can be explained by the fact that intrinsic melatonin production decreases with advancing age, and its external administration can restore normal melatonin levels in blood and follicular fluid (
Figure 2)
Interestingly, in addition to this action against ovarian aging, melatonin was also reported to improve oocyte quality in young women with polycystic ovarian syndrome (PCOS) [
33]. The mechanism of this latter effect is less clear, but it may be related to the fact that melatonin stimulates the production of growth hormone (GH) [
34], and the clinical utility of GH in women with PCOS has been clearly demonstrated [
35].
3.2. Uterine Factor
The term uterine factor infertility is used for a wide range of conditions in which women fail to get pregnant because of some issue(s) related to their uterus, ranging from its complete absence to more or less subtle abnormalities of its structure and function. Understandably, this section only deals with those abnormalitites that can be addressed by medical treatment, with a particular focus to fostering embryo implantation and postimplantation development. The term “uterine receptivity”, or “endometrial receptivity” is traditionally used to denote the intricate processes undertaken by the internal lining of the uterine cavity (endometrium) and the uterine muscle (myometrium) to make the uterus receptive for the upcoming embryo entry [
36].
Among a number of molecular pathways controlling uterine receptivity, involving hormones, adhesion molecules, cytokines, and growth factors, many are potential targets for melatonin action. Experimental animal studies have shown that melatonin treatment enhances the process of implantation, reflected by a higher number of implantation sites, implantation rate, pregnancy rate and litter size compared to untreated females, reviewed in Li et al. [
21]. As to data available for melatonin use in humans, the observation that melatonin supplementation improves uterine function has also been confirmed in women [
37].
3.3. In-Vitro Embryo Development Issues
During the time period between fertilization and implantation, newly formed embryos display an active metabolism generating a great amount of potentially harmful reactive oxygen species (ROS). Under in-vivo conditions, excess ROS are getting scavanged by intrinsic antioxidant enzymes present in the female genital tract. In contrast, when preimplantation embryos are cultured in vitro, these natural antioxidants are absent and have to be substituted by culture medium supplements [
20]. Owing to its dual mechanism of action (direct and receptor-mediated), melatonin appears to hold promise as compared to other ROS scavengers under embryo in-vitro culture conditions. Actually, independent in-vitro studies with human embryos demonstrated that melatonin improved the quality of human day-3 embryos in patients with repeated poor-quality embryos in previous attempts [
38], and increased fertilization, cleavage, high-quality blastocyst development, implantation, and clinical pregnancy rates [
39].
3.4. Gynecological Pathologies and Pregnancy Complications
Endometriosis and adenomyosis are the main gynecological pathologies that can be treated with success by melatonin. In spite of the distinct clinical manifestations, both of these pathologies share a common denominator: a failure of the intrinsic mechanism of apoptosis, also called programmed cell death (PCD), to help the organism get rid of the endometrial cells released during menstruation. In the case of endometriosis, these cells undergo a retrograde migration across the oviduct and settle on the surface of various intra-abdominal organs, manly the ovaries, whereas in adenomyosis these cells migrate within the uterine wall towards the myometrium.
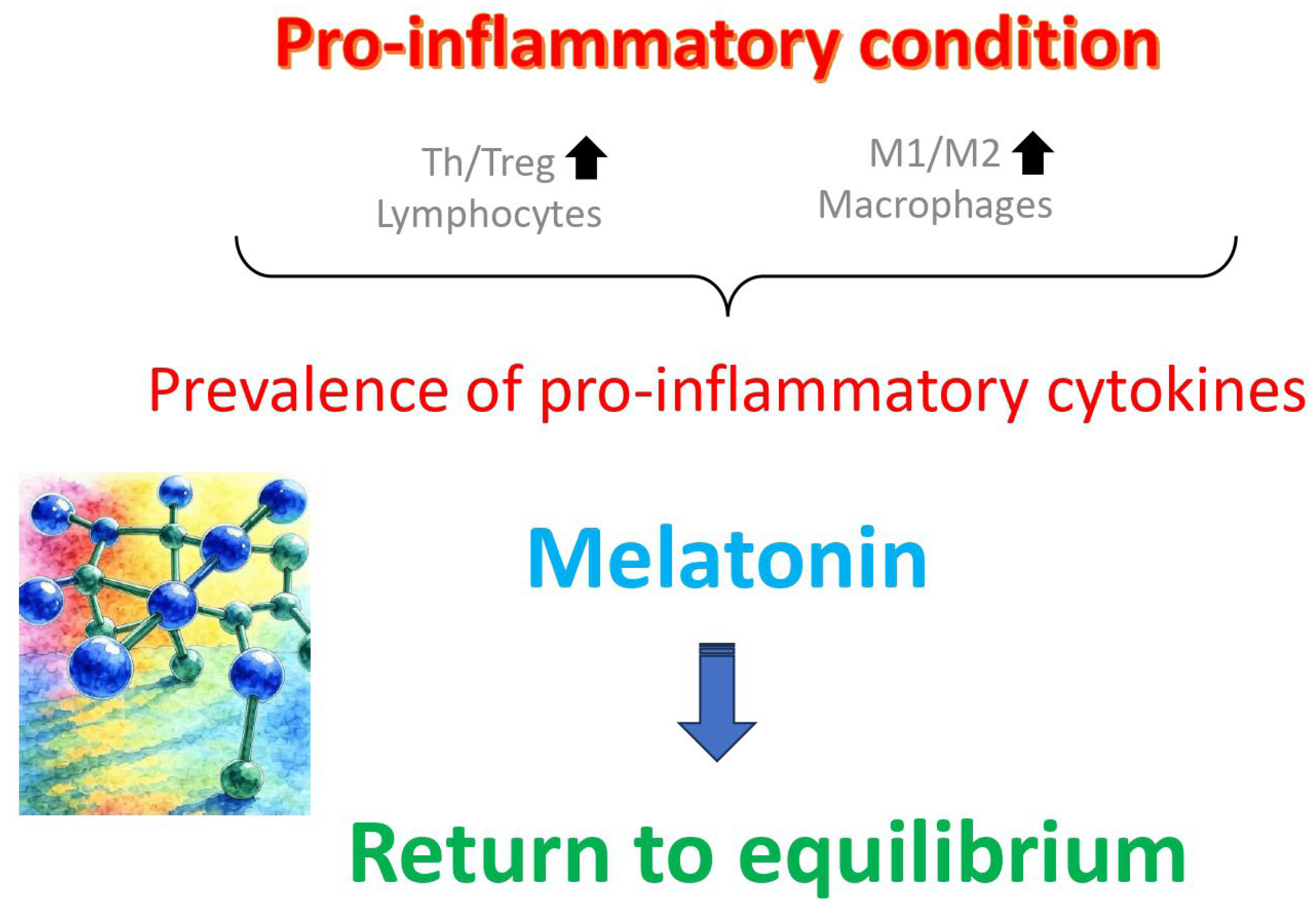
The immunological mechanisms underlying both of these pathologies are similar and involve an imbalance between different types of immune cells (mainly lymphocytes and macrophages) and their products (
Figure 3), especially an increased T helper cell/T regulatory cell and pro-inflammatory (M1)/anti-inflammatory (M2) ratios, along with a prevalence of pro-inflammatory cytokines over anti-inflammatory ones [
40]. In a controlled experimental study, melatonin was shown to revert this anomaly in adenomyosis mice, in addition to improve pregnancy outcomes [
41]. In addition to these animal experimental data, melatonin was shown to revert this kind of immunological imbalance in humans, too [
42,
43]. The well-demonstrated safety of melatonin medication in humans, even at much higher doses than those sufficient to control endometriosis and adenomyosis [
21,
44], further supports its use as the first noninvasive causal therapeutic agent for endometriosis and adenomyosis [
45], and in addition, this therapeutic action goes accompanied by other associated beneficial effects on female infertility [
21,
37] and preventive action against respiratory viral diseases [
46] and some types of cancer [
47].
4. Conclusions
This review provides convincing evidence in favor of the use of melatonin to treat human female infertility and to prevent a number of gynecological pathologies. By combining the actions of an indirect (receptor-mediated) antioxidant action with that of a direct reactive oxygen species scavenger, melatonin improves oocyte and preimplantation embryo quality as well as the uterine receptivity for the implanting embryo. In addition, it modulates the immune system (mainly T lymphocytes and macrophages) to make it more specific towards pathogenic agents and less destructive for uninfected cells. By the same mechanisms, it relieves consequences of some gynecological diseases, mainly endometriosis and adenomyosis. Taking into consideration its safety and zero toxicity, melatonin is highly recommended as a first-line treatment in all types of female infertility, and as an adjuvant treatment in cases in which the recourse to assisted reproduction techniques is required.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
No GenAI has been used. The authors take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
Explained in the text and Figure legends.
References
- Datta, J.; Palmer, M.J.; Tanton, C.; Gibson, L.J.; Jones, K.G.; Macdowall, W.; Glasier, A.; Sonnenberg, P.; Field, N.; Mercer, C.H.; Johnson, A.M.; Wellings, K. Prevalence of Infertility and Help Seeking Among 15 000 Women and Men. Hum. Reprod. 2016, 31, 2108–2118. [Google Scholar] [CrossRef] [PubMed]
- Gemzell, C.A.; Diczfalusy, E.; Tillinger, G. Clinical Effect of Human Pituitary Follicle-Stimulating Hormone (FSH). J. Clin. Endocrinol. Metab. 1958, 18, 1333–1348. [Google Scholar] [CrossRef]
- Buxton, C.L.; Herrmann, W. Induction of Ovulation in the Human with Human Gonadotropins. Yale J. Biol. Med. 1960, 33, 145–147. [Google Scholar] [PubMed]
- Kohlberg, K. Die Praxis der Samenübertragung beim Menschen [The Practice of Artificial Insemination in Humans]. Dtsch. Med. Wochenschr. 1953, 78, 835–839. [Google Scholar] [CrossRef]
- Hackelöer, B.J.; Robinson, H.P. Ultraschalldarstellung des wachsenden Follikels und Corpus luteum im normalen physiologischen Zyklus [Ultrasound Examination of the Growing Ovarian Follicle and of the Corpus Luteum During the Normal Physiologie Menstrual Cycle (author's transl)]. Geburtshilfe Frauenheilkd. 1978, 38, 163–168. [Google Scholar]
- Steptoe, P.C.; Edwards, R.G. Birth after Reimplantation of a Human Embryo. Lancet. 1978, 2, 366. [Google Scholar] [CrossRef]
- Palermo, G.; Joris, H.; Devroey, P.; Van Steirteghem, A.C. Pregnancies after Intracytoplasmic Injection of Single Spermatozoon into an Oocyte. Lancet. 1992, 340, 17–18. [Google Scholar] [CrossRef]
- Tesarik, J.; Mendoza, C.; Testart, J. Viable Embryos from Injection of Round Spermatids into Oocytes. N. Engl. J. Med. 1995, 333, 525. [Google Scholar] [CrossRef]
- Bashiri, A.; Halper, K.I.; Orvieto, R. Recurrent Implantation Failure-Update Overview on Etiology, Diagnosis, Treatment and Future Directions. Reprod. Biol. Endocrinol. 2018, 16, 121. [Google Scholar] [CrossRef]
- Fernando, S.; Rombauts, L. Melatonin: Shedding Light on Infertility?--A Review of the Recent Literature. J. Ovarian Res. 2014, 7, 98. [Google Scholar] [CrossRef]
- Espino, J.; Macedo, M.; Lozano, G.; Ortiz, Á.; Rodríguez, C.; Rodríguez, A.B.; Bejarano, I. Impact of Melatonin Supplementa-tion in Women with Unexplained Infertility Undergoing Fertility Treatment. Antioxidants (Basel). 2019, 8, 338. [Google Scholar] [CrossRef]
- Hu, K.L.; Ye, X.; Wang, S.; Zhang, D. Melatonin Application in Assisted Reproductive Technology: A Systematic Review and Meta-Analysis of Randomized Trials. Front. Endocrinol. (Lausanne). 2020, 11, 160. [Google Scholar] [CrossRef]
- OlceseJM Melatonin and Female Reproduction: An Expanding Universe. Front. Endocrinol. (Lausanne). 2020, 11, 85. [CrossRef]
- Yang, L.; Xu, H.; Chen, Y.; Miao, C.; Zhao, Y.; Xing, Y.; Zhang, Q. Melatonin: Multi-Target Mechanism Against Diminished Ovarian Reserve Based on Network Pharmacology. Front. Endocrinol. (Lausanne). 2021, 12, 630504. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Hung, S.W.; Zhang, R.; Man, G.C.; Zhang, T.; Chung, J.P.; Fang, L.; Wang, C.C. Melatonin in Endometriosis: Mecha-nistic Understanding and Clinical Insight. Nutrients. 2022, 14, 4087. [Google Scholar] [CrossRef]
- Tamura, I.; Tamura, H.; Kawamoto-Jozaki, M.; Shirafuta, Y.; Fujimura, T.; Doi-Tanaka, Y.; Mihara, Y.; Taketani, T.; Sugino, N. Effects of Melatonin on the Transcriptome of Human Granulosa Cells, Fertilization and Blastocyst Formation. Int. J. Mol. Sci. 2022, 23, 6731. [Google Scholar] [CrossRef]
- Patel, A.; Dewani, D.; Jaiswal, A.; Yadav, P. ; ReddyLS Exploring Melatonin's Multifaceted Role in Polycystic Ovary Syn-drome Management: A Comprehensive Review. Cureus. 2023, 15, e48929. [Google Scholar] [CrossRef]
- Reiter, R.J.; Sharma, R.; Romero, A.; Manucha, W.; Tan, D.X.; Zuccari, D.A.P.C.; Chuffa, L.G.A. Aging-Related Ovarian Failure and Infertility: Melatonin to the Rescue. Antioxidants (Basel). 2023, 12, 695. [Google Scholar] [CrossRef]
- Veiga, E.C.A.; Samama, M.; Ikeda, F.; Cavalcanti, G.S.; Sartor, A.; Parames, S.F.; Baracat, E.C.; Ueno, J.; Junior, J.M.S. Melato-nin Improves Fertilization Rate in Assisted Reproduction: Systematic Review and Meta-Analysis. Clinics (Sao Paulo). 2024, 79, 100397. [Google Scholar] [CrossRef]
- Choi, J.W.; Kim, S.W.; Kim, H.S.; Kang, M.J.; Kim, S.A.; Han, J.Y.; Kim, H. ; KuSY Effects of Melatonin, GM-CSF, IGF-1, and LIF in Culture Media on Embryonic Development: Potential Benefits of Individualization. Int. J. Mol. Sci. 2024, 25, 751. [Google Scholar] [CrossRef]
- Li, Q.; Zheng, T.; Chen, J.; Li, B.; Zhang, Q.; Yang, S.; Shao, J.; Guan, W.; Zhang, S. Exploring Melatonin's Multifaceted Role in Female Reproductive Health: From Follicular Development to Lactation and its Therapeutic Potential in Obstetric Syndromes. J. Adv. Res. 2025, 70, 223–242. [Google Scholar] [CrossRef]
- Okamoto, H.H.; Cecon, E.; Nureki, O.; Rivara, S.; Jockers, R. Melatonin Receptor Structure and Signaling. J. Pineal Res. 2024, 76, e12952. [Google Scholar] [CrossRef]
- Cipolla-Neto, J.; do, A.m.a.r.a.l.F.G. Melatonin as a Hormone: New Physiological and Clinical Insights, Endocr. Rev. 2018, 39, 39–990. [Google Scholar] [CrossRef] [PubMed]
- Cecon, E.; Oishi, A.; Jockers, R. Melatonin Receptors: Molecular harmacology and Signalling in the Context of System Bias. Br. J. Pharmacol. 2018, 175, 3263–3280. [Google Scholar] [CrossRef] [PubMed]
- Bouayed, J. ; Bohn, T: Exogenous Antioxidants - Double-Eged Swords in Cellular Redox State: Health Beneficial Effects at Physiologic Doses Versus Deleterious Effects at High Doses. Oxid. Med. Cell Longev. 2010, 3, 228–237. [Google Scholar] [CrossRef]
- Reiter, R.J.; Sharma, R.; Simko, F.; Dominguez-Rodriguez, A.; Tesarik, J.; Neel, R.L.; Slominski, A.T.; Kleszczynski, K.; Mar- tin-Gimenez, V.M.; Manucha, W. ; CardinaliDP Melatonin: Highlighting its Use as a Potential Treatment for SARS-CoV-2 Infection. Cell. Mol. Life Sci. 2022, 79, 143. [Google Scholar] [CrossRef] [PubMed]
- Tamura, H.; Nakamura, Y.; Korkmaz, A.; Manchester, L.C.; Tan, D.X.; Sugino, N. ; ReiterRJ Melatonin and the Ovary: Physiological and Pathophysiological Implications. Fertil. Steril. 2009, 92, 328–343. [Google Scholar] [CrossRef]
- Tesarik, J. Endocrinology of Primary Ovarian Insufficiency: Diagnostic and Therapeutic Clues. Endocrines. 2025, 6, 18. [Google Scholar] [CrossRef]
- Yie SM, Niles, L. P.; YounglaiEV Melatonin Receptors on Human Granulosa Cell Membranes. J. Clin. Endocrinol. Metab. 1995, 80, 1747–1749. [Google Scholar] [CrossRef]
- Tamura, H.; Jozaki, M.; Tanabe, M.; Shirafuta, Y.; Mihara, Y.; Shinagawa, M.; Tamura, I.; Maekawa, R.; Sato, S.; Taketani, T.; Takasaki, A.; Reiter, R.J.; Sugino, N. Importance of Melatonin in Assisted Reproductive Technology and Ovarian Aging. Int. J. Mol. Sci. 2020, 21, 1135. [Google Scholar] [CrossRef]
- Mayo, J.C.; Sainz, R.M.; Antoli, I.; Herrera, F.; Martin, V.; Rodriguez, C. Melatonin Regulation of Antioxidant Enzyme Gene Expression. Cell. Mol. Life Sci. 2002, 59, 1706–1713. [Google Scholar] [CrossRef]
- Mayo, J.C.; Tan, D.X.; Sainz, R.M.; Lopez-Burillo, S. ; ReiterRJ Oxidative Damage to Catalase Induced by Peroxyl Radicals: Functional Protection by Melatonin and Other Antioxidants. Free Radic. Res. 2003, 37, 543–553. [Google Scholar] [CrossRef]
- Pacchiarotti, A.; Carlomagno, G.; Antonini, G.; Pacchiarotti, A. Effect of Myo-Inositol and mMelatonin Versus Myo-Inositol, in a Randomized Controlled Trial, for Improving In Vitro Fertilization of Patients with Polycystic Ovarian Syndrome. Gynecol. Endocrinol. 2016, 32, 69–73. [Google Scholar] [CrossRef]
- Valcavi, R.; Zini, M.; Maestroni, G.J.; Conti, A.; Portioli, I. Melatonin Stimulates Growth Hormone Secretion Through Path-ways Other Than the Growth Hormone-Releasing hHormone. Clin. Endocrinol. (Oxf). 1993, 39, 193–199. [Google Scholar] [CrossRef]
- Vitale, S.G.; Palumbo, M.; Conde-López, C.; Mendoza, N.; Mendoza-Tesarik, R.; Tesarik, J. Effect of Growth Hormone Ad-ministration on ICSI Outcomes in Patients With Polycystic Ovary Syndrome and Recurrent Implantation Failure: A Retro-spective Cross-Over Study. Int. J. Gynaecol. Obstet. 2021, 153, 357–358. [Google Scholar] [CrossRef]
- Blanco-Breindel, M.F.; Singh, M.; Kahn, J. Endometrial Receptivity. 2023 Jun 7. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. [PubMed]
- Chuffa, L.G.A.; Lupi, L.A.; Cucielo, M.S.; Silveira, H.S.; Reiter, R.J.; Seiva, F.R.F. Melatonin Promotes Uterine and Placental Health: Potential Molecular Mechanisms. Int J Mol Sci. 2019, 21, 300. [Google Scholar] [CrossRef]
- Bao, Z.; Li, G.; Wang, R.; Xue, S.; Zeng, Y.; Deng, S. Melatonin Improves Quality of Repeated-Poor and Frozen-Thawed Em-bryos in Human, a Prospective Clinical Trial. Front. Endocrinol. (Lausanne). 2022, 13, 853999–10.3389. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Wang, K.; Zhang, C.; Chen, B.; Zou, H.; Zou, W.; Xue, R.; Ji, D.; Yu, Z.; Rao, B.; Huo, R.; Cao, Y.; Ding, D.; Zhang, Z. Effect of Melatonin on the Clinical Outcome of Patients with Repeated Cycles after Failed Cycles of in Vitro Fertilization and Intracytoplasmic Sperm Injection. Zygote. 2022, 30, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Yang, D.; Cai, S.; Yang, L.; Yu, S.; Geng, Q.; Mo, M.; Li, W.; Wei, Y.; Li, Y.; Yin, T.; Diao, L. Adenomyosis-Associated Infertility: An Update of the Immunological Perspective. Reprod. Biomed. Online. 2025, 50, 104703. [Google Scholar] [CrossRef]
- Guan, X.; Liu, D.; Zhou, H.; Dai, C.; Wang, T.; Fang, Y.; Jia, Y.; Li, K. Melatonin Improves Pregnancy Outcomes in Adenomyosis Mice by Restoring Endometrial Receptivity Via NF-κB/Apoptosis Signaling. Ann. Transl. Med. 2022, 10, 1317. [Google Scholar] [CrossRef]
- Qi, S.; Yan, L.; Liu, Z.; Mu, Y.L.; Li, M.; Zhao, X.; Chen, Z.J.; Zhang, H. Melatonin Inhibits 17β-Estradiol-Induced Migration, Invasion and Epithelial-Mesenchymal Transition in Normal and Endometriotic Endometrial Epithelial Cells. Reprod. Biol. Endocrinol. 2018, 16, 62. [Google Scholar] [CrossRef]
- Joseph, T.T.; Schuch, V.; Hossack, D.J.; Chakraborty, R. ; JohnsonEL Melatonin: The Placental Antioxidant and An-ti-Inflammatory. Front. Immunol. 2024, 15, 1339304. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.P.; Gögenur, I.; Rosenberg, J. ; ReiterRJ The Safety of Melatonin in Humans. Clin. Drug Investig. 2016, 36, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Tesarik, J.; Mendoza Tesarik, R. Melatonin: The First Noninvasive Causal Therapy for Both Endometriosis and Adenomyosis? J. Gynecol. Women’s Health. 2018, 12, 555829. [Google Scholar] [CrossRef]
- Alomari, T.; Al-Abdallat, H.; Hamamreh, R.; Alomari, O.; Hos, B.H. : Reiter, R.J., 2024. Assessing the Antiviral Potential of Melatonin: A Comprehensive Systematic Review. Rev. Med. Virol. 2024, 34, e2499. [Google Scholar] [CrossRef]
- Wang, L.; Wang, C. ; ChoiWS Use of Melatonin in Cancer Treatment: Where Are We? Int. J. Mol. Sci. 2022, 23, 3779. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).