Submitted:
19 July 2025
Posted:
21 July 2025
You are already at the latest version
Abstract
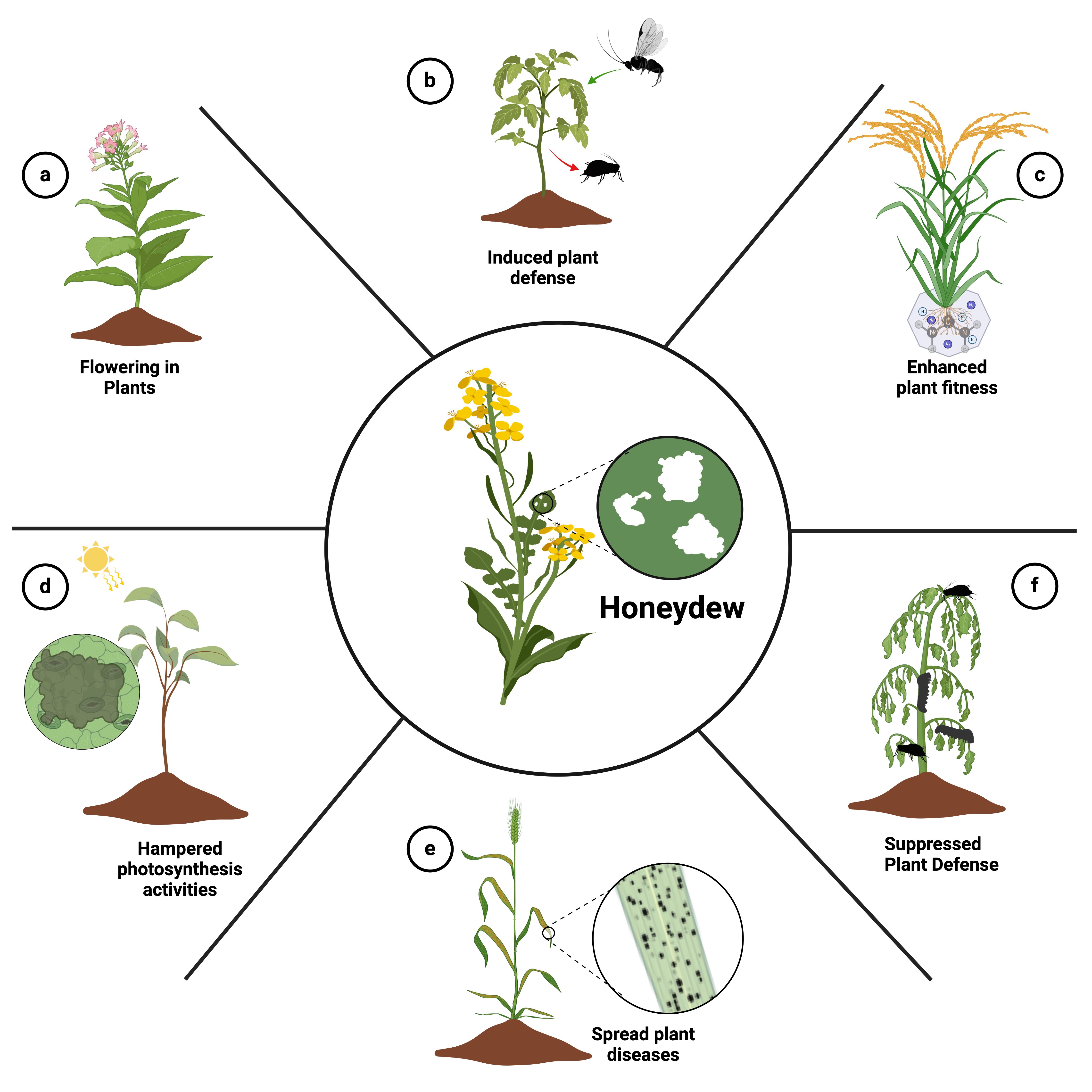
Keywords:
1. Introduction
2. Origin and Composition of Honeydew
3. Negative Effects of Honeydew on Plants
3.1. Impact on Photosynthetic Activity and Pollination
3.2. Making Plant Surface Prone for Pathogens
3.3. Suppression of Plant Defences
4. Positive Effects of Honeydew on Plants
4.1. Regulation of Flowering in Plants
4.2. Induction of Plant Defences
4.3. Indirect Positive Effects of Honeydew on Plants
4.4. Honeydew and Its Diverse Roles in Soil and Plant Ecosystems:
5. Consequences at the Agricultural Level
6. Conclusions and Future Prospects
Author Contributions
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alamgir, K. M. et al. (2016) ‘Systematic analysis of rice (Oryza sativa) metabolic responses to herbivory’, Plant, Cell & Environment, 39(2), pp. 453–466. [CrossRef]
- Ali, J. (2023) ‘The Peach Potato Aphid (Myzus persicae): Ecology and Management’, 1, p. 132. [CrossRef]
- Ali, J. et al. (2024) ‘Honeydew : A keystone in insect – plant interactions , current insights and future perspectives’, (January), pp. 1–7. [CrossRef]
- Álvarez-Pérez, S., Lievens, B. and de Vega, C. (2023) ‘Floral nectar and honeydew microbial diversity and their role in biocontrol of insect pests and pollination’, Current Opinion in Insect Science, p. 101138. [CrossRef]
- Arimura, G.-I., Ozawa, R. and Maffei, M. E. (2011) ‘Recent advances in plant early signaling in response to herbivory’, International Journal of Molecular Sciences, 12(6), pp. 3723–3739. [CrossRef]
- Auclair, J. L. (1963) ‘Aphid feeding and nutrition’, Annual review of entomology, 8(1), pp. 439–490.
- Bae, Hanhong et al. (2005) ‘Exogenous trehalose alters Arabidopsis transcripts involved in cell wall modification, abiotic stress, nitrogen metabolism, and plant defense’, Physiologia plantarum, 125(1), pp. 114–126. [CrossRef]
- Blanchard, S. et al. (2022) ‘Combined Elevation of Temperature and CO2 Impacts the Production and Sugar Composition of Aphid Honeydew’, Journal of Chemical Ecology, 48(9–10), pp. 772–781.
- de Bobadilla, M. F. et al. (2024) ‘Honeydew management to promote biological control’, Current Opinion in Insect Science, 61, p. 101151. [CrossRef]
- Buckley, R. C. (1987) ‘Interactions involving plants, Homoptera, and ants’, Annual review of Ecology and Systematics, 18(1), pp. 111–135. [CrossRef]
- Buitenhuis, R. et al. (2004) ‘The role of honeydew in host searching of aphid hyperparasitoids’, Journal of chemical ecology, 30, pp. 273–285. [CrossRef]
- Byrne, D. N. and Bellows Jr, T. S. (1991) ‘Whitefly biology.’.
- Calvo-Agudo, M. et al. (2020) ‘IPM-recommended insecticides harm beneficial insects through contaminated honeydew’, Environmental Pollution, 267, p. 115581. [CrossRef]
- Capinera, J. (2020) Handbook of vegetable pests. Academic press.
- Carena, M. J. and Glogoza, P. (2004) ‘Resistance of maize to the corn leaf aphid: a review.’.
- Chomnunti, P. et al. (2014) ‘The sooty moulds’, Fungal diversity, 66, pp. 1–36.
- Cleland, C. F. (1974) ‘Isolation of flower-inducing and flower-inhibitory factors from aphid honeydew’, Plant Physiology, 54(6), pp. 899–903. [CrossRef]
- Cleland, C. F. and Ajami, A. (1974) ‘Identification of the flower-inducing factor isolated from aphid honeydew as being salicylic acid’, Plant Physiology, 54(6), pp. 904–906. [CrossRef]
- Colazza, S., Peri, E. and Cusumano, A. (2023) ‘Chemical ecology of floral resources in conservation biological control’, Annual Review of Entomology, 68, pp. 13–29. [CrossRef]
- Crawford, D. L. (1921) ‘Annual Address. Honey-Dew Smut and Photosynthesis’.
- Dhami, M. K. et al. (2011) ‘Species-specific chemical signatures in scale insect honeydew’, Journal of chemical ecology, 37, pp. 1231–1241. [CrossRef]
- Dhami, M. K. et al. (2013) ‘Diverse honeydew-consuming fungal communities associated with scale insects’, PLoS One, 8(7), p. e70316. 0316. [CrossRef]
- Dietrich, C. H. (2009) ‘Auchenorrhyncha:(cicadas, spittlebugs, leafhoppers, treehoppers, and planthoppers)’, in Encyclopedia of insects. Elsevier, pp. 56–64.
- Dik, A. J. (1990) ‘Population dynamics of phyllosphere yeasts: influence of yeasts on aphid damage, diseases and fungicide activity in wheat’.
- Dik, A. J. and Van Pelt, J. A. (1992) ‘Interaction between phyllosphere yeasts, aphid honeydew and fungicide effectiveness in wheat under field conditions’, Plant pathology, 41(6), pp. 661–675. [CrossRef]
- Douglas, A. (2006) ‘Phloem-sap feeding by animals: problems and solutions’, Journal of experimental botany, 57(4), pp. 747–754. [CrossRef]
- Douglas, A. E. (1993) ‘The nutritional quality of phloem sap utilized by natural aphid populations’, Ecological Entomology, 18(1), pp. 31–38. [CrossRef]
- Douglas, A. E. (2009) ‘Honeydew’, in Encyclopedia of insects. Elsevier, pp. 461–463.
- Edde, P. A. (2022) ‘7-Arthropod pests of maize Zea mays (L.)’, Field crop arthropod pests of economic importance. Academic Press, Richmond, VA, pp. 410–465.
- Van Emden, H. F. and Harrington, R. (2017) Aphids as crop pests. Cabi.
- Erb, M. and Reymond, P. (2019) ‘Molecular interactions between plants and insect herbivores’, Annual review of plant biology, 70, pp. 527–557. [CrossRef]
- Fischer, M. K. et al. (2002) ‘Age-specific patterns in honeydew production and honeydew composition in the aphid Metopeurum fuscoviride: implications for ant-attendance’, Journal of Insect Physiology, 48(3), pp. 319–326. [CrossRef]
- Fischer, M. K. and Shingleton, A. W. (2001) ‘Host plant and ants influence the honeydew sugar composition of aphids’, Functional Ecology, 15(4), pp. 544–550. [CrossRef]
- Fischer, M. K., Völkl, W. and Hoffmann, K. H. (2005) ‘Honeydew production and honeydew sugar composition of polyphagous black bean aphid, Aphis fabae (Hemiptera: Aphididae) on various host plants and implications for ant-attendance’, European Journal of Entomology, 102(2), pp. 155–160.
- Fokkema, N. J. et al. (1983) ‘Aphid honeydew, a potential stimulant of Cochliobolus sativus and Septoria nodorum and the competitive role of saprophytic mycoflora’, Transactions of the British Mycological Society, 81(2), pp. 355–363. [CrossRef]
- Francis, F. et al. (2020) ‘From diverse origins to specific targets: role of microorganisms in indirect pest biological control’, Insects, 11(8), p. 533. [CrossRef]
- Fujita, D., Kohli, A. and Horgan, F. G. (2013) ‘Rice resistance to planthoppers and leafhoppers’, Critical Reviews in Plant Sciences, 32(3), pp. 162–191. [CrossRef]
- Garre, V., Tenberge, K. B. and Eising, R. (1998) ‘Secretion of a fungal extracellular catalase by Claviceps purpurea during infection of rye: putative role in pathogenicity and suppression of host defense’, Phytopathology, 88(8), pp. 744–753. [CrossRef]
- Grier, C. C. and Vogt, D. J. (1990) ‘Effects of aphid honeydew on soil nitrogen availability and net primary production in an Alnus rubra plantation in western Washington’, Oikos, pp. 114–118. [CrossRef]
- Hendrix, D. L., Wei, Y. and Leggett, J. E. (1992) ‘Homopteran honeydew sugar composition is determined by both the insect and plant species’, Comparative biochemistry and physiology part B: Comparative Biochemistry, 101(1–2), pp. 23–27. [CrossRef]
- Hequet, E. F., Henneberry, T. J. and Nichols, R. L. (2007) ‘Sticky Cotton: Causes, Effects, and Prevention’, United States Department of Agriculture, (1915), pp. 1–219.
- Hettenhausen, C., Schuman, M. C. and Wu, J. (2015) ‘MAPK signaling: a key element in plant defense response to insects’, Insect science, 22(2), pp. 157–164. [CrossRef]
- Hijaz, F., Lu, Z. and Killiny, N. (2016) ‘Effect of host-plant and infection with “C andidatus Liberibacter asiaticus” on honeydew chemical composition of the Asian citrus psyllid, D iaphorina citri’, Entomologia Experimentalis et Applicata, 158(1), pp. 34–43.
- Hodkinson, I. D. (2009) ‘Life cycle variation and adaptation in jumping plant lice (Insecta: Hemiptera: Psylloidea): a global synthesis’, Journal of natural History, 43(1–2), pp. 65–179. [CrossRef]
- Hogervorst, P. A. M., Wäckers, F. L. and Romeis, J. (2007) ‘Effects of honeydew sugar composition on the longevity of Aphidius ervi’, Entomologia Experimentalis et Applicata, 122(3), pp. 223–232. [CrossRef]
- Hu, C. et al. (2024) ‘Molecular interaction network of plant-herbivorous insects’, Advanced Agrochem, 3(1), pp. 74–82. doi: 10.1016/j.aac.2023.08.008. [CrossRef]
- Leroy, P. D. et al. (2011) ‘Microorganisms from aphid honeydew attract and enhance the efficacy of natural enemies’, Nature communications, 2(1), p. 348. [CrossRef]
- Leroy, P. D. et al. (2014) ‘Aphid honeydew: An arrestant and a contact kairomone for Episyrphus balteatus (Diptera: Syrphidae) larvae and adults’, European Journal of Entomology, 111(2), pp. 237–242. [CrossRef]
- Li, X. et al. (2025) ‘Bacterial volatiles from aphid honeydew mediate ladybird beetles oviposition site choice’, Pest Management Science. [CrossRef]
- Liu, J. et al. (2024) ‘Chemical cues from honeydew-associated bacteria to enhance parasitism efficacy: from laboratory to field assay’, Journal of Pest Science, 97(2), pp. 873–884. [CrossRef]
- Llewellyn, M. (1972) ‘The effects of the lime aphid, Eucallipterus tiliae L.(Aphididae) on the growth of the lime Tilia x Vulgaris Hayne. I. Energy requirements of the aphid population’, Journal of Applied Ecology, pp. 261–282. [CrossRef]
- Llewellyn, M., Rashid, R. and Leckstein, P. (1974) ‘The ecological energetics of the willow aphid Tuberolachnus salignus (Gmelin); honeydew production’, The Journal of Animal Ecology, pp. 19–29. [CrossRef]
- LT, L. and RODRIGUEZ, J. G. (1985) ‘The leafhoppers and planthoppers’.
- Michalzik, B. (2011) ‘Insects, infestations, and nutrient fluxes’, in Forest hydrology and biogeochemistry: synthesis of past research and future directions. Springer, pp. 557–580.
- Michalzik, B. and Stadler, B. (2005) ‘Importance of canopy herbivores to dissolved and particulate organic matter fluxes to the forest floor’, Geoderma, 127(3–4), pp. 227–236. [CrossRef]
- Mihaela Fericean, L. (2012) ‘the Behaviour, Life Cycle and Biometrical Measurements of Aphis Fabae’, Research Journal of Agricultural Science, 44(4), pp. 31–37.
- Mittler, T. E. (1958) ‘Studies on the feeding and nutrition of Tuberolachnus salignus (Gmelin)(Homoptera, Aphididae) II. The nitrogen and sugar composition of ingested phloem sap and excreted honeydew’, Journal of Experimental Biology, 35(1), pp. 74–84. [CrossRef]
- Molyneux, R. J., Campbell, B. C. and Dreyer, D. L. (1990) ‘Honeydew analysis for detecting phloem transport of plant natural products: implications for host-plant resistance to sap-sucking insects’, Journal of Chemical Ecology, 16, pp. 1899–1909.
- Mostafa, S. et al. (2022) ‘Plant responses to herbivory, wounding, and infection’, International journal of molecular sciences, 23(13), p. 7031.
- van Neerbos, F. A. C. et al. (2020) ‘Honeydew composition and its effect on life-history parameters of hyperparasitoids’, Ecological Entomology, 45(2), pp. 278–289. [CrossRef]
- Nelson, A. S. and Mooney, K. A. (2022) ‘The evolution and ecology of interactions between ants and honeydew-producing hemipteran insects’, Annual Review of Ecology, Evolution, and Systematics, 53(1), pp. 379–402. [CrossRef]
- Nelson, S. (2008) ‘Sooty mold’.
- Owen, D. F. and Wiegert, R. G. (1976) ‘Do consumers maximize plant fitness?’, Oikos, pp. 488–492. [CrossRef]
- Pellizzari, G. et al. (2012) ‘Phenology, ethology and distribution of Pseudococcus comstocki, an invasive pest in northeastern Italy’, Bulletin of Insectology, 65(2), pp. 209–215.
- Quesada, C. R., Scharf, M. E. and Sadof, C. S. (2020) ‘Excretion of non-metabolized insecticides in honeydew of striped pine scale’, Chemosphere, 249, p. 126167. [CrossRef]
- Rabbinge, R. et al. (1981) ‘Damage effects of cereal aphids in wheat’, Netherlands Journal of Plant Pathology, 87, pp. 217–232.
- Rabbinge, R. et al. (1984) ‘Effects of the saprophytic leaf mycoflora on growth and productivity of winter wheat’, Netherlands Journal of Plant Pathology, 90, pp. 181–197. [CrossRef]
- Rondoni, G. et al. (2018) ‘Vicia faba plants respond to oviposition by invasive Halyomorpha halys activating direct defences against offspring’, Journal of Pest Science, 91, pp. 671–679. [CrossRef]
- Sabri, A. et al. (2013) ‘Proteomic investigation of aphid honeydew reveals an unexpected diversity of proteins’, PloS one, 8(9), p. e74656. [CrossRef]
- Schillewaert, S. et al. (2017) ‘The effect of host plants on genotype variability in fitness and honeydew composition of Aphis fabae’, Insect science, 24(5), pp. 781–788. [CrossRef]
- Schoonhoven, L. M. et al. (2005) Insect-plant biology. Oxford University Press on Demand.
- Schwartzberg, E. G. and Tumlinson, J. H. (2014) ‘Aphid honeydew alters plant defence responses’, Functional Ecology, 28(2), pp. 386–394. [CrossRef]
- Seeger, J. and Filser, J. (2008) ‘Bottom-up down from the top: Honeydew as a carbon source for soil organisms’, European Journal of Soil Biology, 44(5–6), pp. 483–490. [CrossRef]
- Shaaban, B. et al. (2020) ‘Sugar, amino acid and inorganic ion profiling of the honeydew from different hemipteran species feeding on Abies alba and Picea abies’, PLoS One, 15(1), p. e0228171. [CrossRef]
- Shinya, T. et al. (2016) ‘Modulation of plant defense responses to herbivores by simultaneous recognition of different herbivore-associated elicitors in rice’, Scientific reports, 6(1), p. 32537. [CrossRef]
- Stadler, B. and Michalzik, B. (1998) ‘Linking aphid honeydew, throughfall, and forest floor solution chemistry of Norway spruce’, Ecology Letters, 1(1), pp. 13–16. [CrossRef]
- Starr, C. K. (2021) Encyclopedia of social insects. Springer.
- Taylor, S. H., Parker, W. E. and Douglas, A. E. (2012) ‘Patterns in aphid honeydew production parallel diurnal shifts in phloem sap composition’, Entomologia experimentalis et applicata, 142(2), pp. 121–129. [CrossRef]
- Tena, A. et al. (2013) ‘Energy reserves of parasitoids depend on honeydew from non-hosts’, Ecological Entomology, 38(3), pp. 278–289. [CrossRef]
- Tena, A. et al. (2016) ‘Parasitoid nutritional ecology in a community context: the importance of honeydew and implications for biological control’, Current opinion in insect science, 14, pp. 100–104. [CrossRef]
- Tosh, C. R. and Brogan, B. (2015) ‘Control of tomato whiteflies using the confusion effect of plant odours’, Agronomy for Sustainable Development, 35(1), pp. 183–193. [CrossRef]
- VanDoorn, A. et al. (2015) ‘Whiteflies glycosylate salicylic acid and secrete the conjugate via their honeydew’, Journal of Chemical Ecology, 41, pp. 52–58. [CrossRef]
- Vereijken, P. H. (1979) Feeding and multiplication of three cereal aphid species and their effect on yield of winter wheat. Wageningen University and Research.
- Völkl, W. et al. (1999) ‘Ant-aphid mutualisms: the impact of honeydew production and honeydew sugar composition on ant preferences’, Oecologia, 118, pp. 483–491. [CrossRef]
- Wäckers, F. L. (2000) ‘Do oligosaccharides reduce the suitability of honeydew for predators and parasitoids? A further facet to the function of insect-synthesized honeydew sugars’, Oikos, 90(1), pp. 197–201. [CrossRef]
- Wäckers, F. L., Van Rijn, P. C. J. and Heimpel, G. E. (2008) ‘Honeydew as a food source for natural enemies: making the best of a bad meal?’, Biological Control, 45(2), pp. 176–184. [CrossRef]
- Wardle, D. A. (2013) Communities and ecosystems: linking the aboveground and belowground components (MPB-34). Princeton University Press.
- Wari, D. et al. (2019) ‘Honeydew-associated microbes elicit defense responses against brown planthopper in rice’, Journal of experimental botany, 70(5), pp. 1683–1696. [CrossRef]
- Wasternack, C. and Song, S. (2017) ‘Jasmonates: biosynthesis, metabolism, and signaling by proteins activating and repressing transcription’, Journal of Experimental Botany, 68(6), pp. 1303–1321.
- Weintraub, P. G. (2009) ‘Physical control: an important tool in pest management programs’, Biorational Control of Arthropod Pests: Application and Resistance Management, pp. 317–324.
- Whitaker, M. R. L., Katayama, N. and Ohgushi, T. (2014) ‘Plant–rhizobia interactions alter aphid honeydew composition’, Arthropod-Plant Interactions, 8, pp. 213–220.
- Whitfield, A. E., Falk, B. W. and Rotenberg, D. (2015) ‘Insect vector-mediated transmission of plant viruses’, Virology, 479, pp. 278–289. [CrossRef]
- Woldemariam, M. G., Baldwin, I. T. and Galis, I. (2011) ‘Transcriptional regulation of plant inducible defenses against herbivores: a mini-review’, Journal of Plant Interactions, 6(2–3), pp. 113–119. [CrossRef]
- Wool, D., Hendrix, D. L. and Shukry, O. (2006) ‘Seasonal variation in honeydew sugar content of galling aphids (Aphidoidea: Pemphigidae: Fordinae) feeding on Pistacia: host ecology and aphid physiology’, Basic and Applied Ecology, 7(2), pp. 141–151. [CrossRef]
- Zebelo, S. A. and Maffei, M. E. (2015) ‘Role of early signalling events in plant–insect interactions’, Journal of Experimental Botany, 66(2), pp. 435–448. [CrossRef]
- Zhu, J. et al. (2020) ‘Proteomics of the honeydew from the brown planthopper and green rice leafhopper reveal they are rich in proteins from insects, rice plant and bacteria’, Insects, 11(9), p. 582. [CrossRef]
- Zipfel, C. et al. (2004) ‘Bacterial disease resistance in Arabidopsis through flagellin perception’, Nature, 428(6984), pp. 764–767. [CrossRef]

| Factors | Plant species | Insect species | Effects on honeydew | References |
|---|---|---|---|---|
| Species (Insect and Host Plant) | Populus tremula, P. alba | Chaitophorus populialbae, C. populeti | Contain high proportions of the trisaccharide melezitose | (Fischer and Shingleton, 2001) |
| Bemisia tabaci, Trialeurodes abutilonea and T. vaporariorum | Gossypium hirsutum, (Euphorbia pulcherrima, and Lycopersicon esculentum | Honeydew from the whitefly Bemisia tabaci and the whitefly genus Trialeurodes contains a greater proportion of oligomers larger than disaccharides, in addition to a higher concentration of turanose, at a ratio of over 20%. | (Hendrix, Wei and Leggett, 1992) | |
| Tanacetum vulgare, | Aphis fabae, Vicia faba or Chenopodium album, Cirsium arvense | The composition is predominantly characterised by the presence of the higher level of trisaccharide melezitose. |
(Fischer, Völkl and Hoffmann, 2005) |
|
| Solanum tuberosum L., Triticum aestivumL., cv. | Aphidius ervi | The composition of honeydew sugar (comprising sucrose, glucose, and fructose) differed between the various aphid species on the various plants. | (Hogervorst, Wäckers and Romeis, 2007) | |
| Large variation in phloem composition of host plants, herbivore–plant interactions | Vicia faba | Aphis fabae | The study revealed alterations in melezitose and other carbohydrates, which were dependent on the host plant. | (Schillewaert et al., 2017) |
| Host aphid and its microbiota, including endosymbiotic bacteria and gut flora | Acyrthosiphon pisum | Vicia faba L. | It is widely accepted that aphid honeydew comprises not only carbohydrates but also proteins. The total protein concentration was notably high, comprising not only insect proteins but also bacterial and aphid proteins, including peroxidease, inositol monophosphates (IMPase) and dihydrofolate reductase (DHFR), as well as GroEL, DnaK, and Hsp70 chaperones, and flagellin. | (Sabri et al., 2013) |
| Species of scale insect, local environments, geography | Nothofagus solandri, N. trucnata, N. fusca | Ultracoelostoma spp., Coelostomidia wairoensis, Coelostomidia Zealandica | A comparative analysis of scale insect species has revealed a significant impact on the chemical composition of their respective honeydew, which may elucidate the distinctive consumer communities associated with them. | (Dhami et al., 2011) |
| Climate change, elevated temperature, and/or CO2 conditions | Vicia faba | Aphis fabae | An increase in temperature and carbon dioxide concentration resulted in a significant elevation in the concentration of fructose, a principal honeydew sugar. Furthermore, there were insignificant increases in the volume of honeydew produced and the melezitose content. | (Blanchard et al., 2022) |
| Plant species, weather, and position on leaves | Acer pseudoplanatus, Prunus domestica, Euonymus europaeus, Vicia faba | Drepanosiphum platanoides, Hyalopterus pruni, Aphis fabae | Honeydew was found to be dominated by non-essential amino acids, including glutamic acid, glutamine, asparagine and serine. | (Douglas, 1993) |
| Seasonal variation in host-tree sap quality, | Pistacia palaestina | Aphidoidea, Pemphigidae, Fordinae | A modification in the concentration of sugars was observed. The concentration of glucose was found to be higher than that of fructose. | (Wool, Hendrix and Shukry, 2006) |
| L. niger. L. nigerworkers, higher total sugar concentration of host plants | Tanacetum vulgare | Metopeurum fuscoviride and Brachycaudus cardui, | The honeydew of M. fuscoviride and B. cardui was found to contain notable quantities of melezitose and raffinose. | (Völkl et al., 1999) |
| Age-related | T. vulgare | M. fuscoviride | In addition to melezitose, the age-related effects were not observed for other sugar compositions. Furthermore, the concentration of asparagine and glutamine amino acids was observed to be elevated. | (Fischer et al., 2002) |
| Plant age, circadian clocks | Solanum tuberosum L | Macrosiphum euphorbiae (Thomas) and Myzus persicae | The mean values for honeydew production during daylight hours exhibited a 1.9-fold increase for Ma. euphorbiae and a 2.6-fold increase for My. persicae in comparison to night-time honeydew production. A significant alteration was observed in both the ratio of sucrose to amino acids and the composition of amino acids. | (Taylor, Parker and Douglas, 2012) |
| Ant presence | Ant-tended aphids produce melezitose-rich honeydew, while others have higher glucose. | (Fischer and Shingleton, 2001; Mihaela Fericean, 2012) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




