Submitted:
16 July 2025
Posted:
17 July 2025
You are already at the latest version
Abstract
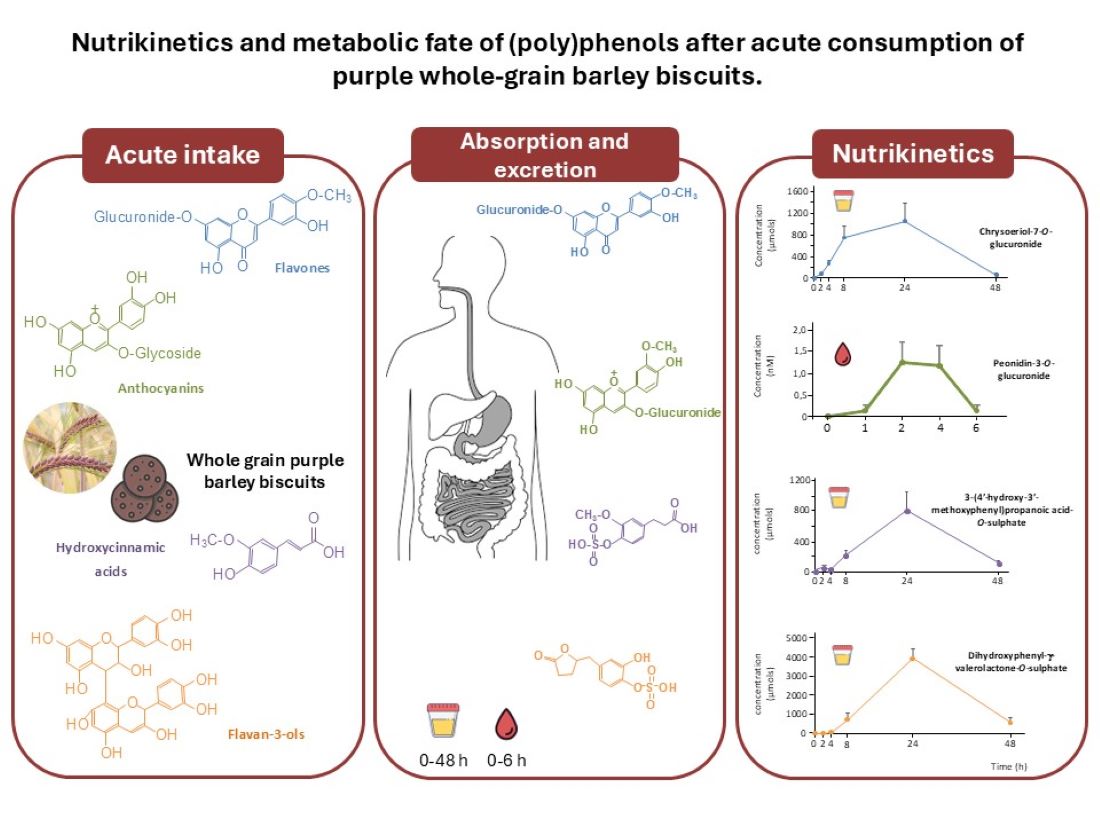
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Preparation of Whole-Grain Barley Biscuits
2.3. Subjects and Study Design
2.4. Biological Samples Pretreatment
2.4.1. Plasma Analysis
2.4.2. Urine Analysis
2.5. Analysis of (poly)phenolic Metabolites by UPLC-MS/MS
2.6. Statistical Analysis
3. Results and Discussion
3.1. Characterization of (poly)phenolic Compounds in Barley Biscuits
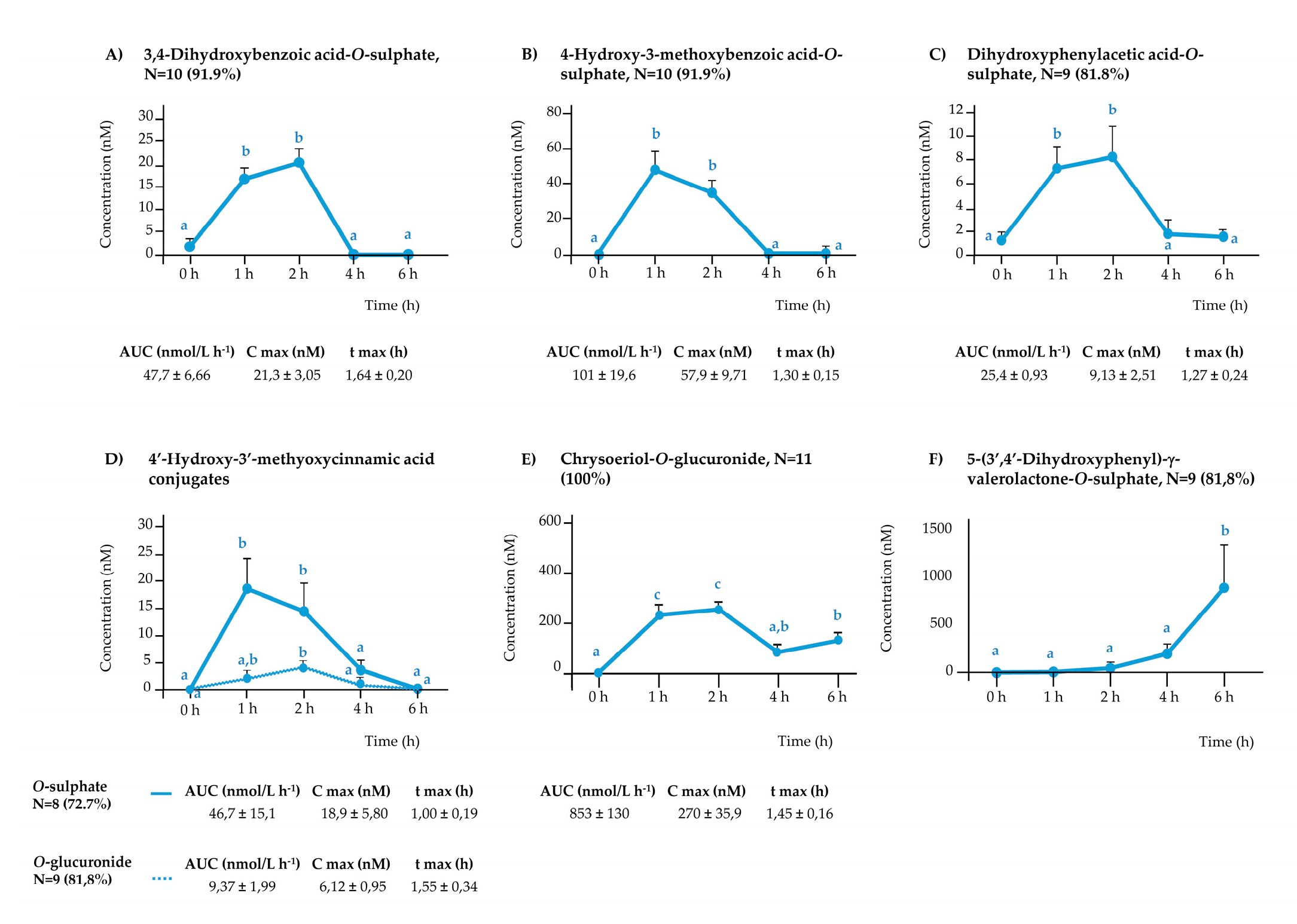
3.2. Plasma Appearance of Phenolic Metabolites
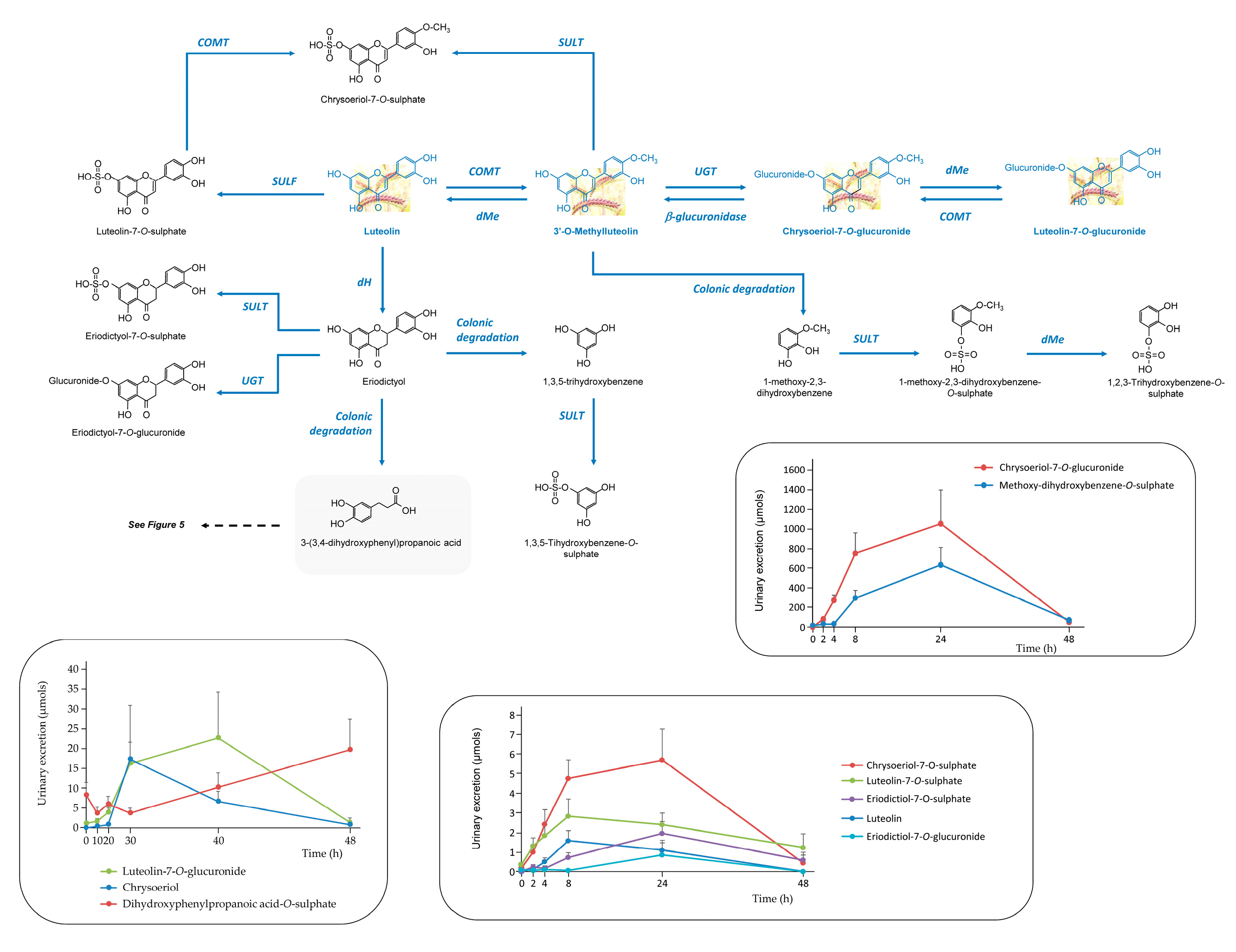
3.2.1. Anthocyanins
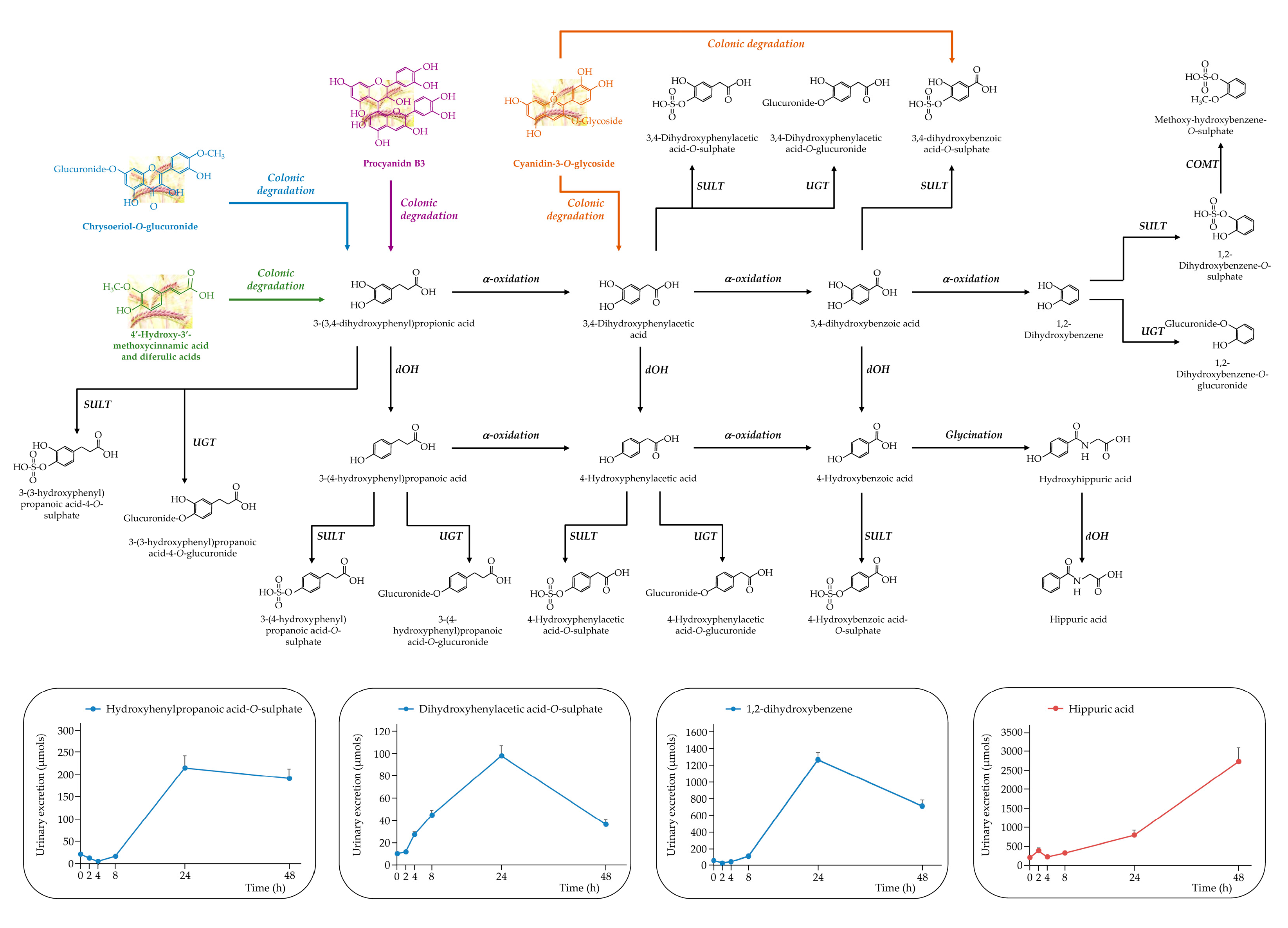
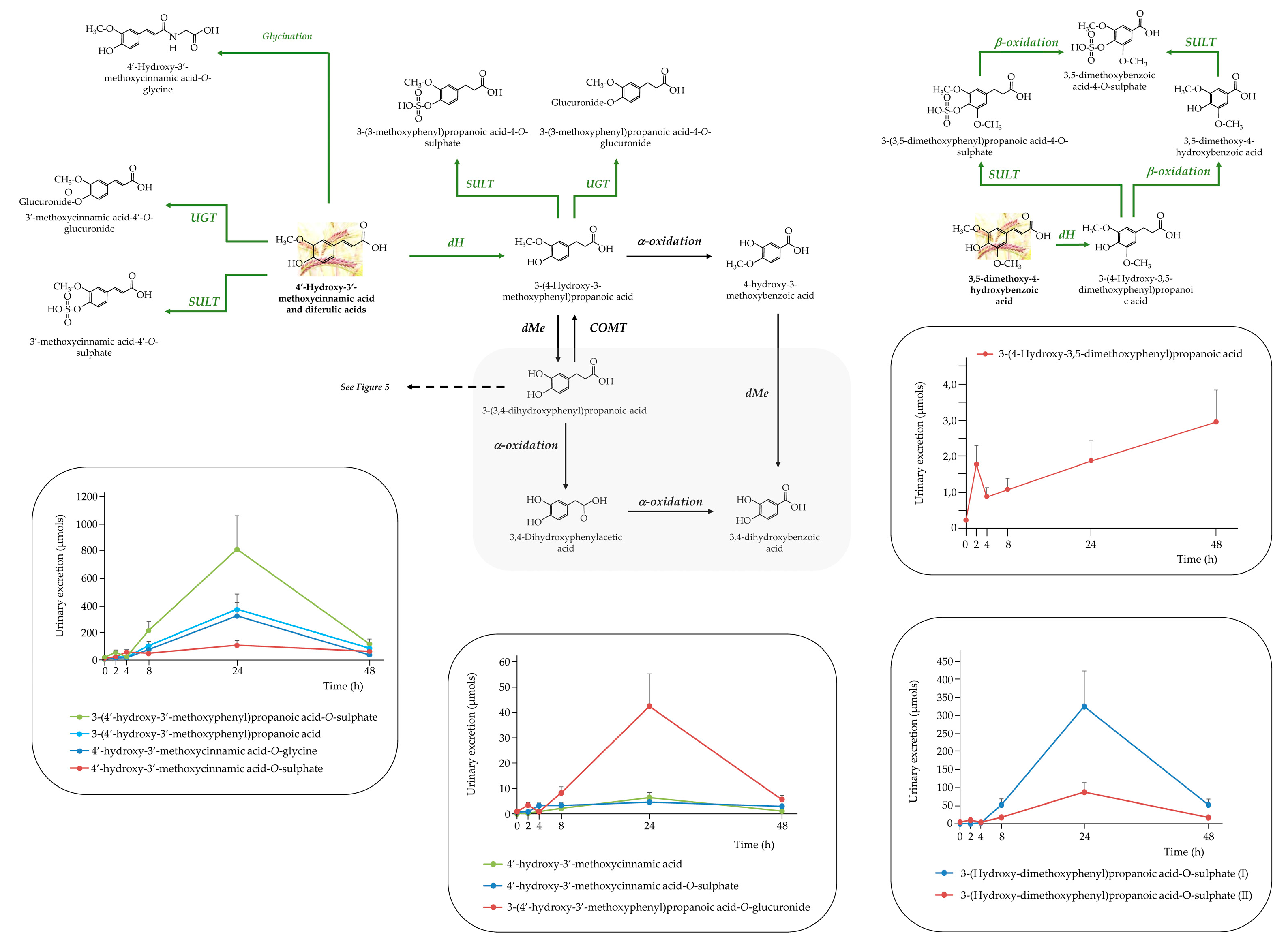
3.2.2. Rest of the (poly)phenolic Compounds
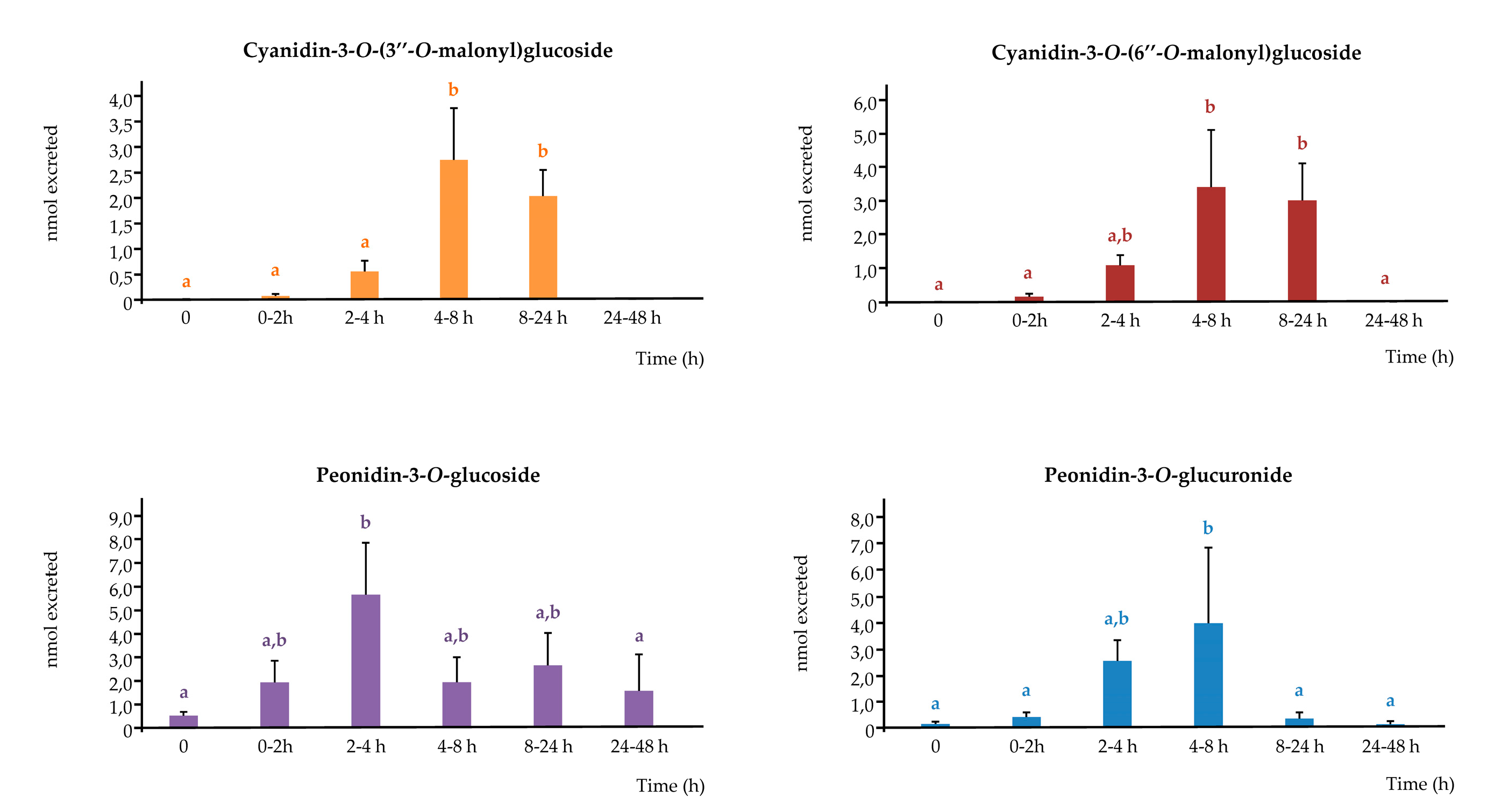
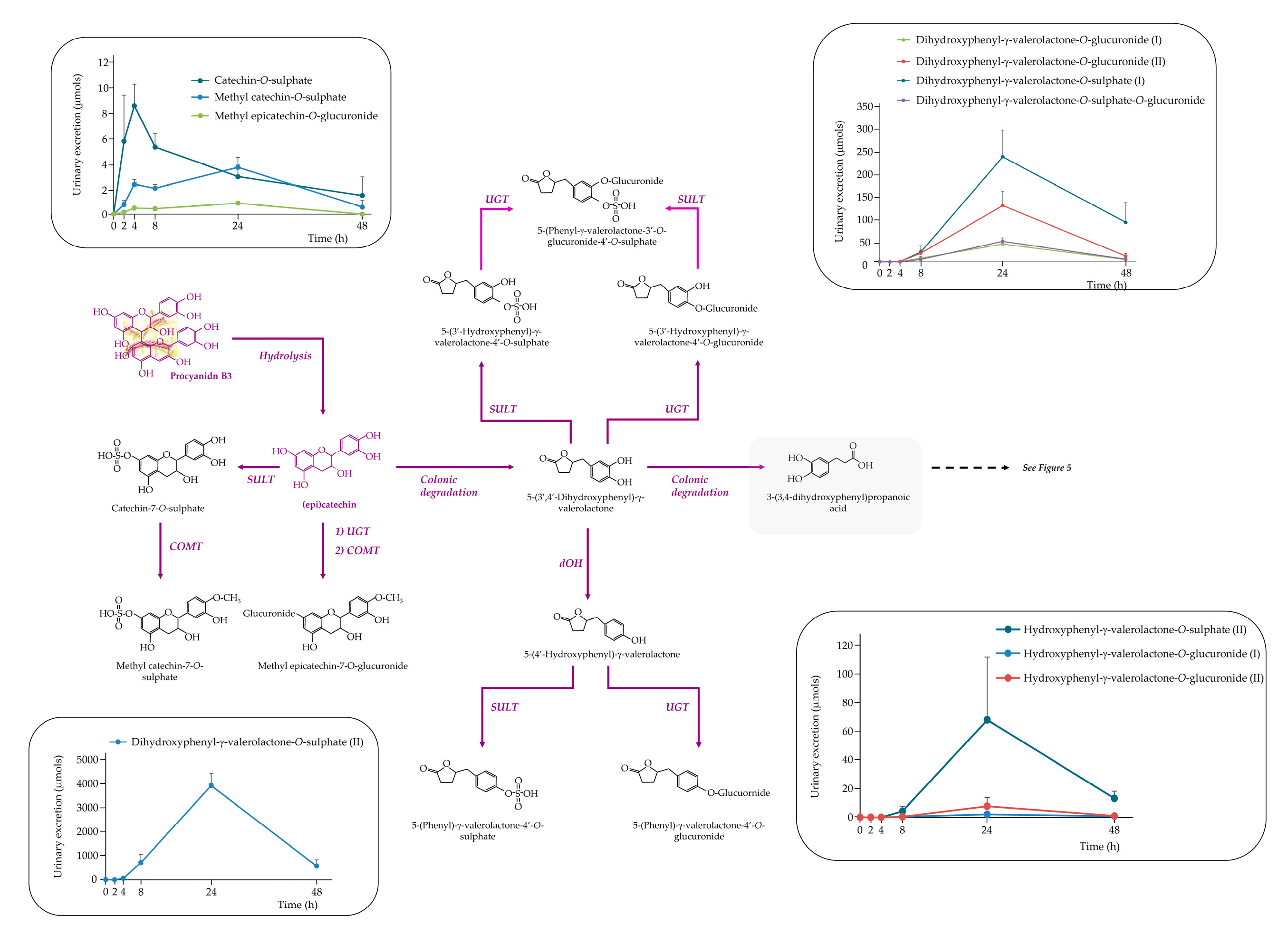
3.3. Urinary excretion of phenolic metabolites
3.3.1. Anthocyanins
3.3.2. The Rest of (poly) Phenolic Compounds
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgements
Conflicts of Interest
References
- D Aune, N Keum, E Giovannucci, LT Fadnes, P Boffetta, DC Greenwood, S Tonstad, LJ Vatten, E Riboli, T Norat. Whole grain consumption and risk of cardiovascular disease, cancer, and all cause and cause specific mortality: systematic review and dose-response meta-analysis of prospective studies. BMJ 2016, 353, i2716. [Google Scholar] [CrossRef]
- Y Zhang, J Sui, Y Xu, L Pan, H Xia, G Sun. Effect of whole grain and fiber consumption on chronic liver diseases: a systematic review and meta-analysis. Food Funct 2024, 15, 9707–9717. [Google Scholar] [CrossRef] [PubMed]
- P Vitaglione, A Napolitano, V Fogliano, Cereal dietary fibre: a natural functional ingredient to deliver phenolic compounds into the gut. Trends Food Sci Technol 2008, 19, 451–463. [CrossRef]
- D Junlin, Y Manyou, Y Yuehang, L Tinghui, X Zhuoya, C Jian, Y Kaijun, Z Ruiling, Z Boyu, Z Yongqing, Y Xing, W Yan, X Chen. Studies of phytochemical constituents by UPLC-QTOF-MS/MS of black hulless barley bran and its antioxidation and α-glucosidase inhibition effect. Arab J Chem 2024, 17, 105644. [Google Scholar] [CrossRef]
- B Ed Nignpense, N Francis, C Blanchard, A Santhakumar. The bioavailability of polyphenols following acute consumption of pigmented barley and wheat. Food Funct 2024, 15, 9330–9342. [Google Scholar] [CrossRef] [PubMed]
- D Junlin, X Zhuoya, L Changbin, Z Yongqing, Y Kaijun, L Tinghui, X Chen, C Jian, Z Wenhui, Z Yuhong, Z Boyu. Identification and quantification of free, esterified, and insoluble-bound phenolics in grains of hulless barley varieties and their antioxidant activities. LWT 2021, 151, 112001. [Google Scholar] [CrossRef]
- CD Kay, MN Clifford, P Mena, GJ McDougall, C Andrés-Lacueva, A Cassidy, D Del Rio, N Kuhnert, C Manach, G Pereira-Caro, A Rodriguez-Mateos, A Scalbert, FA Tomás-Barberán, G Williamson, DS Wishart, A Crozier. Recommendations for standardizing nomenclature for dietary (poly)phenol catabolites. Am J Clin Nutr 2020, 112, 1051–1068. [Google Scholar] [CrossRef] [PubMed]
- C Curti, MN Clifford, CD Kay, P Mena, A Rodriguez-Mateos, D Del Rio, GJ McDougall, G Williamson, C Andrés-Lacueva, L Bresciani, B Burton Freeman, A Cassidy, Y Desjardin, CG Fraga, CCI Gill, PA Kroon, N Kuhnert, IA Ludwig, C Manach, D Milenkovic, C Nunes Dos Santos, PI Oteiza, G Pereira-Caro, FA Tomás Barberán, DS Wishart, A Crozier. Extended recommendations on the nomenclature for microbial catabolites of dietary (poly)phenols, with a focus on isomers. Food Funct 2025, 16, 3963–4000. [Google Scholar] [CrossRef] [PubMed]
- ME Cortijo-Alfonso, S Yuste, C Piñol-Felis, MP Romero, A Macià, L Rubió-Piqué. Finger-prick blood sampling using volumetric absorptive microsampling (VAMS) method for monitoring the main (poly)phenolic metabolites in human blood after barley biscuit intake. J Chromatogr B 2025, 1256, 124527. [Google Scholar] [CrossRef] [PubMed]
- S Yuste, A Macià, IA Ludwig, MP Romero, S Fernández-Castillejo, Ú Catalán, MJ Motilva, L Rubió. Validation of Dried Blood Spot Cards to Determine Apple Phenolic Metabolites in Human Blood and Plasma After an Acute Intake of Red-Fleshed Apple Snack. Mol Nutr Food Res 2018, 62, e1800623. [Google Scholar] [CrossRef] [PubMed]
- S Yuste, IA Ludwig, L Rubió, MP Romero, A Pedret, RM Valls, R Solà, MJ Motilva, A Macià. In vivo biotransformation of (poly)phenols and anthocyanins of red-fleshed apple and identification of intake biomarkers. J Funct Foods 2019, 55, 146–155. [Google Scholar] [CrossRef]
- ME Cortijo-Alfonso, S Yuste, C Piñol-Felis, MP Romero, A Macià, L Rubió-Piqué. Finger-prick blood sampling using volumetric absorptive microsampling (VAMS) method for monitoring the main (poly)phenolic metabolites in human blood after barley biscuit intake. J Chromatogr B 2025, 1256, 124527. [Google Scholar] [CrossRef] [PubMed]
- ME Cortijo-Alfonso, S Yuste, I Friero, M Martínez-Subirà, M Moralejo, C Piñol-Felis, L Rubió-Piqué, A Macià. Metabolic profiling of (poly)phenolic compounds in mouse urine following consumption of hull-less and purple-grain barley. Food Funct 2024, 15, 8300–8309. [Google Scholar] [CrossRef] [PubMed]
- I Friero, M Martínez-Subirà, A Macià, MP Romero, M Moralejo, Exploring the nutritional and techno-functional benefits of purple hull-less barley in extruded ready-to-eat cereals. LWT 2024, 212, 117018. [CrossRef]
- AD Assefa, OS Hur, BS Hahn, B Kim, NY Ro, JH Rhee. Nutritional Metabolites of Red Pigmented Lettuce (Lactuca sativa) Germplasm and Correlations with Selected Phenotypic Characters. Foods 2021, 10, 2504. [Google Scholar] [CrossRef] [PubMed]
- D Hernanz, V Nuñez, AI Sancho, CB Faulds, G Williamson, B Bartolomé, C Gómez-Cordovés. Hydroxycinnamic acids and ferulic acid dehydrodimers in barley and processed barley. J Agric Food Chem 2001, 49, 4884–4888. [Google Scholar] [CrossRef] [PubMed]
- D Horvat, G Šimić, G Drezner, A Lalić, T Ledenčan, M Tucak, H Plavšić, L Andrić, Z Zdunić. Phenolic Acid Profiles and Antioxidant Activity of Major Cereal Crops. Antioxidants 2020, 9, 527–538. [Google Scholar] [CrossRef] [PubMed]
- I Friero, A Macià, MP Romero, I Romagosa, M Martínez-Subirà, M. Moralejo. Unlocking Phenolic Potential: Determining the Optimal Grain Development Stage in Hull-Less Barley Genotypes with Varying Grain Color. Foods 2024, 13, 1841–1860. [Google Scholar] [CrossRef] [PubMed]
- A Crozier, D Del Rio, MN. Clifford. Bioavailability of dietary flavonoids and phenolic compounds. Mol Aspects Med 2010, 31, 446–467. [Google Scholar] [CrossRef] [PubMed]
- H Gui, L Sun, R Liu, X Si, D Li, Y Wang, C Shu, X Sun, Q Jiang, Y Qiao, B Li, J Tian. Current knowledge of anthocyanin metabolism in the digestive tract: absorption, distribution, degradation, and interconversion. Crit Rev Food Sci Nutr 2023, 63, 5953–5966. [Google Scholar] [CrossRef] [PubMed]
- A Serra, A Macià, MP Romero, J Reguant, N Ortega, MJ. Motilva, Metabolic pathways of the colonic metabolism of flavonoids (flavonols, flavones and flavanones) and phenolic acids. Food Chem 2012, 130, 383–393. [Google Scholar] [CrossRef]
- JI Mosele, B Viadel, S Yuste, L Tomás-Cobos, S García-Benlloch, MT Escribano Bailón, I García Estévez, P Moretón Fraile, F Rodríguez de Rivera, S de Domingo Casado, MJ Motilva. Application of a dynamic colonic gastrointestinal digestion model to red wines: a study of flavanol metabolism by the gut microbiota and the cardioprotective activity of microbial metabolites. Food Funct 2025, 16, 885–899. [Google Scholar] [CrossRef] [PubMed]
- Z Zhao, Y Egashira, H Sanada. Ferulic Acid Sugar Esters Are Recovered in Rat Plasma and Urine Mainly as the Sulfoglucuronide of Ferulic Acid. J Nutr 2003, 133, 1355–1361. [Google Scholar] [CrossRef] [PubMed]
- X Feng, Y Li, M Brobbey Oppong, F Qiu. Insights into the intestinal bacterial metabolism of flavonoids and the bioactivities of their microbe-derived ring cleavage metabolites. Drug Metab Rev 2018, 50, 343–356. [Google Scholar] [CrossRef] [PubMed]
- HG Hur, RD Beger, TM Heinze, JO Lay, JP Freeman, J Dore, F Rafii. Isolation of an anaerobic intestinal bacterium capable of cleaving the C-ring of the isoflavonoid daidzein. Arch Microbiol 2002, 178, 75–75. [Google Scholar] [CrossRef]
- J Winter, MR Popoff, P Grimont, VD Bokkenheuser. Clostridium orbiscindens sp. nov., a Human Intestinal Bacterium Capable of Cleaving the Flavonoid C-Ring. Int J Syst Bacteriol 1991, 41, 355–357. [Google Scholar] [CrossRef] [PubMed]
- G Williamson, MN Clifford. A critical examination of human data for the biological activity of phenolic acids and their phase-2 conjugates derived from dietary (poly)phenols, phenylalanine, tyrosine and catecholamines. Crit Rev Food Sci Nutr 2024, 9, 1–60. [Google Scholar] [CrossRef]

| FREE (poly)phenolics | BOUND (poly)phenolics | |
|---|---|---|
| Cyanidin-3-O-glucoside | 6.20 ± 0.24 | 0.06 ± 0.01 |
| Cyanidin-3-O-(3’’-malonyl)glucoside | 4.90 ± 0.20 | n.d. |
| Cyanidin-3-O-(6’’-malonyl)glucoside | 12.5 ± 0.50 | n.d. |
| Cyanidin-3-O-(3’’,6’’-dimalonyl)glucoside | 16.9 ± 0.64 | n.d. |
| Peonidin-3-O-glucoside | 0.38 ± 0.07 | 0.02 ± 0.00 |
| Peonidin-3-O-(3’’-O-malonyl)glucoside | 0.31 ± 0.01 | n.d. |
| Peonidin-3-O-(6’’-O-malonyl)glucoside | 1.12 ± 0.07 | n.d. |
| Peonidin-3-O-(3’’,6’’-dimalonyl)glucoside | 0.46 ± 0.01 | n.d. |
| Pelargonidin-3-O-glucoside | 0.27 ± 0.02 | 0.02 ± 0.00 |
| Pelargonidin-3-O-malonylglucoside | 0.22 ± 0.02 | n.d. |
| Pelargonidin-3-O-(6’’-malonyl)glucoside | 1.24 ± 0.08 | n.d. |
| Delphinidin-3-O-glucoside | 0.20 ± 0.06 | n.d. |
| Total anthocyanins | 44.7 ± 1.80 | 0.10 ± 0.01 |
| 4-Hydroxybenzoic acid | 0.22 ± 0.02 | 0.87 ± 0.03 |
| Hydroxybenzoic acid | n.d. | 0.18 ± 0.01 |
| 3,4-Dihydroxybenzoic acid (PCA) | 0.16 ± 0.06 | 0.03 ± 0.01 |
| 4-Hydroxy-3-methoxybenzoic acid (VA) | 0.16 ± 0.03 | 0.44 ± 0.01 |
| 4-Hydroxy-3,5-dimethoxybenzoic acid (Syr) | n.d. | 0.16 ± 0.02 |
| Cinnamic acid | n.d. | 0.11 ± 0.01 |
| 4’-Hydroxycinnamic acid | 0.10 ± 0.02 | 1.18 ± 0.04 |
| 3,4-Dihydroxycinnamic acid (CA) | n.d. | 0.02 ± 0.00 |
| 4’-Hydroxy-3’-methoxycinnamic acid (FA) | 1.22 ± 0.14 | 40.8 ± 0.80 |
| 3’-Hydroxy-4’-methoxycinnamic acid (IsoFA) | 0.20 ± 0.07 | 8.64 ± 0.17 |
| 4’-Hydroxy-3,5-dimethoxycinnamic acid | n.d. | 3.07 ± 0.16 |
| 4’-Hydroxy-3,5-dimethoxycinnamic acid-O-glucoside | n.d. | 0.95 ± 0.25 |
| Diferulic acid | n.d. | 12.4 ± 0.65 |
| Diferulic acid DC | n.d. | 1.20 ± 0.24 |
| Triferulic acid | n.d. | 2.06 ± 0.39 |
| Total phenolic acids | 2.06 ± 0.16 | 72.1 ± 1.24 |
| Catechin | 2.62 ± 0.56 | n.d. |
| Catechin glucoside | 2.09 ± 0.20 | n.d. |
| Procyanidin B3 | 8.40 ± 1.49 | n.d. |
| Gallocatechin-catechin or prodelphinidin B4 | 3.67 ± 0.39 | n.d. |
| Total flavan-3-ols | 16.8 ± 1.40 | n.d. |
| Apigenin-7-O-glucoside | n.d. | 0.04 ± 0.01 |
| Apigenin-6-C-arabinoside-8-C-glucoside | 0.46 ± 0.09 | 0.05 ± 0.01 |
| Isovitexin-C-glucoside | 0.56 ± 0.05 | 0.09 ± 0.01 |
| Isovitexin-C-rutinoside | 0.25 ± 0.03 | n.d. |
| Isoscoparin-C-glucoside | 0.89 ± 0.12 | 0.09 ± 0.02 |
| Isoscoparin-C-rutinoside | 0.26 ± 0.04 | n.d. |
| Luteolin | 5.92 ± 0.18 | 0.03 ± 0.00 |
| Methyl luteolin (Chrysoeriol) | 16.2 ± 2.63 | 0.07 ± 0.02 |
| Luteolin-O-glucoside | 0.13 ± 0.02 | n.d. |
| Luteolin-7-O-glucuronide | 32.9 ± 5.91 | 0.46 ± 0.03 |
| Methyl Luteolin-O-glucoside | 0.22 ± 0.03 | n.d. |
| Methyl Luteolin-O-glucuronide | 160 ± 24.6 | 4.64 ± 0.49 |
| Total flavones | 217 ± 33.0 | 5.53 ± 0.50 |
| Total (poly)phenolics | 281 ± 32.4 | 77.7 ± 0.73 |
| TOTAL (POLY)PHENOLS (Free and Bound) | 359 ± 32.2 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





