Submitted:
19 June 2025
Posted:
23 June 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
1.1. The Current State of Research
1.2. Specific Aims of the Study
- To evaluate the effectiveness of compounded semaglutide for weight loss.
- To study the effects of compounded semaglutide in healthy individuals who are of normal weight or overweight (BMI < 29.9).
- To evaluate safety, efficacy, and the extent of weight loss with likely consequential health benefits.
- To propose a novel method for declaring the ideal or target weight that accounts for differences in body composition, bone structure, and sex. Achieving this target weight is also proposed as a metric for program success.
1.3. The Primary Research Question
2. Materials and Methods
2.1. Study Design
2.2. Intervention Protocol
2.3. Inclusion and Exclusion Criteria
2.4. Sample Size and Recruitment
2.5. Informed Consent
2.6. Data Collection and Follow-Up
2.7. Success Zone and Weight Goals
2.8. Monitoring and Safety Measures
2.9. Data Management and Confidentiality
2.10. Risk and Benefit Analysis
2.11. Patient-Reported Outcomes
2.12. Participant Demographics and Follow-Up
3. Results
3.1. Outcome
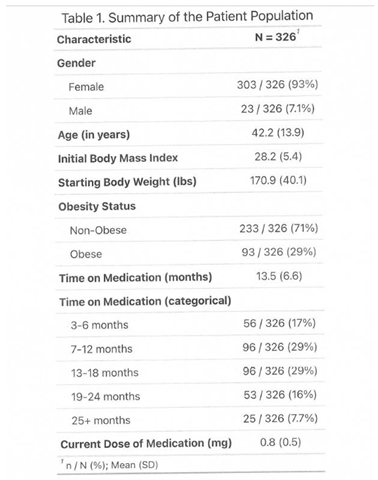 |
 |
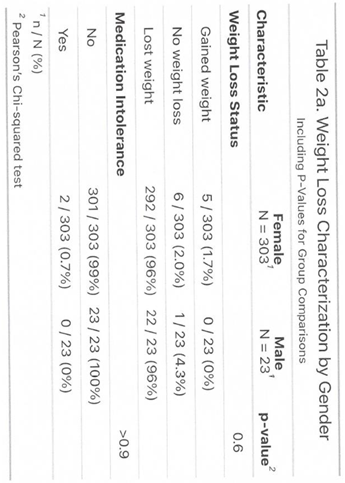 |
 |
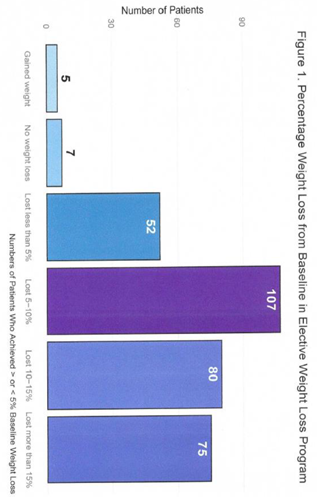

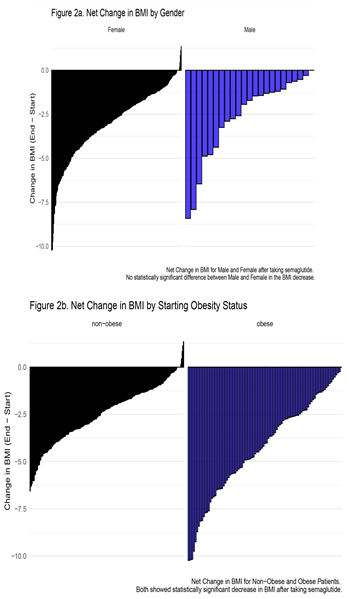
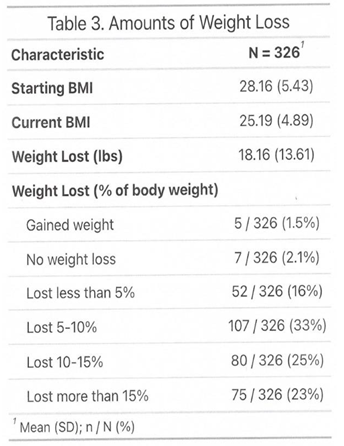
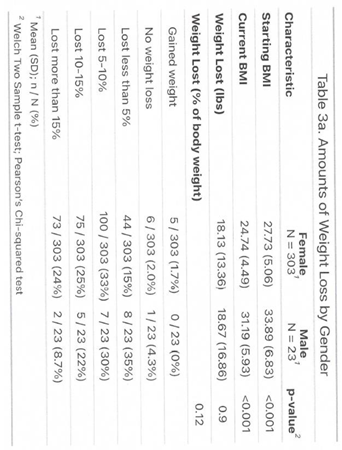
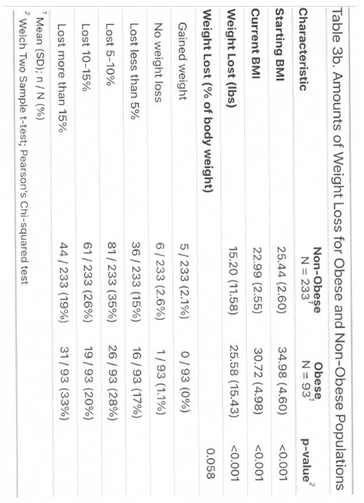






3.2. Side Effects
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ward, Z. J.; Bleich, S. N.; Cradock, A. L.; Barrett, J. L.; Giles, C. M.; Flax, C.; Gortmaker, S. L. Projected US state-level prevalence of adult obesity and severe obesity. N. Engl. J. Med. 2019, 381 (25), 2440–2450.
- Kraus, W. E.; Bhapkar, M.; Huffman, K. M.; Pieper, C. F.; Das, S. K.; Redman, L. M.; Fontana, L. 2 years of calorie restriction and cardiometabolic risk (CALERIE): Exploratory outcomes of a multicentre, phase 2, randomised controlled trial. Lancet Diabetes Endocrinol. 2019, 7 (9), 673–683.
- Centers for Disease Control and Prevention. CDC twenty-four seven. Saving lives, protecting people: heart disease. https://www.cdc.gov/heart-disease/data-research/facts-stats/index.html (accessed Oct 24, 2024).
- Adeva-Andany, M. M.; Domínguez-Montero, A.; Adeva-Contreras, L.; Fernández-Fernández, C.; Carneiro-Freire, N.; González-Lucán, M. Body fat distribution contributes to defining the relationship between insulin resistance and obesity in human diseases. Curr. Diabetes Rev. 2024, 20 (5), 66–96.
- Fontana, L.; Meyer, T. E.; Klein, S.; Holloszy, J. O. Long-term calorie restriction is highly effective in reducing the risk of atherosclerosis in humans. Proc. Natl. Acad. Sci. U.S.A. 2004, 101 (17), 6659–6663. [CrossRef]
- Speakman, J. R.; Mitchell, S. E. Caloric restriction. Mol. Aspects Med. 2011, 32 (3), 159–221. https://www.sciencedirect.com/science/article/pii/S009829971100032X.
- Waziry, R.; Ryan, C. P.; Corcoran, D. L.; Huffman, K. M.; Kobor, M. S.; Kothari, M.; Belsky, D. W. Effect of long-term caloric restriction on DNA methylation measures of biological aging in healthy adults from the CALERIE trial. Nat. Aging 2023, 3 (3), 248–257. https://www.nature.com/articles/s43587-022-00357-y.
- Napoleão, A.; Fernandes, L.; Miranda, C.; Marum, A. P. Effects of calorie restriction on health span and insulin resistance: Classic calorie restriction diet vs. ketosis-inducing diet. Nutrients 2021, 13 (4), 1302. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8071299.
- Ravussin, E.; Redman, L. M.; Rochon, J.; Das, S. K.; Fontana, L.; Kraus, W. E.; CALERIE Study Group. A 2-year randomized controlled trial of human caloric restriction: Feasibility and effects on predictors of health span and longevity. J. Gerontol. A Biol. Sci. Med. Sci. 2015, 70 (9), 1097–1104. https://pubmed.ncbi.nlm.nih.gov/26187233/.
- Ryan, D. H.; Yockey, S. R. Weight loss and improvement in comorbidity: Differences at 5%, 10%, 15%, and over. Curr. Obes. Rep. 2017, 6, 187–194.
- Guh, D. P.; Zhang, W.; Bansback, N.; Amarsi, Z.; Birmingham, C. L.; Anis, A. H. The incidence of co-morbidities related to obesity and overweight: A systematic review and meta-analysis. BMC Public Health 2009, 9, 1–20.
- Wilding, J. P.; Batterham, R. L.; Calanna, S.; Davies, M.; Van Gaal, L. F.; Lingvay, I.; Kushner, R. F. Once-weekly semaglutide in adults with overweight or obesity. N. Engl. J. Med. 2021, 384 (11), 989–1002.
- Ryan, D. H.; Lingvay, I.; Colhoun, H. M.; Deanfield, J.; Emerson, S. S.; Kahn, S. E.; Kushner, R. F.; Marso, S.; Plutzky, J.; Brown-Frandsen, K.; Gronning, M. O.; Hovingh, G. K.; Holst, A. G.; Ravn, H.; Lincoff, A. M. Semaglutide effects on cardiovascular outcomes in people with overweight or obesity (SELECT) rationale and design. Am. Heart J. 2020, 229, 75–85. [CrossRef]
- Wali, S. Financial Incentives for Weight Reduction Study (FIReWoRk) [Internet]; University of California, Los Angeles. ClinicalTrials.gov, May 30, 2023. https://clinicaltrials.gov/ct2/show/NCT03157713 (accessed Jan 10, 2025).
- Davies, M.; Færch, L.; Jeppesen, O. K.; Pakseresht, A.; Pedersen, S. D.; Perreault, L.; Rosenstock, J.; Shimomura, I.; Viljoen, A.; Wadden, T. A.; Lingvay, I.; STEP 2 Study Group. Semaglutide 2.4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): A randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial. Lancet 2021, 397 (10278), 971–984. [CrossRef]
- Wadden, T. A.; Bailey, T. S.; Billings, L. K.; Davies, M.; Frias, J. P.; Koroleva, A.; Lingvay, I.; O’Neil, P. M.; Rubino, D. M.; Skovgaard, D.; Wallenstein, S. O.; Garvey, W. T.; STEP 3 Investigators. Effect of subcutaneous semaglutide vs placebo as an adjunct to intensive behavioral therapy on body weight in adults with overweight or obesity: The STEP 3 randomized clinical trial. JAMA 2021, 325 (14), 1403–1413. [CrossRef]
- Rubino, D.; Abrahamsson, N.; Davies, M.; Hesse, D.; Greenway, F. L.; Jensen, C.; Lingvay, I.; Mosenzon, O.; Rosenstock, J.; Rubio, M. A.; Rudofsky, G.; Tadayon, S.; Wadden, T. A.; Dicker, D.; STEP 4 Investigators. Effect of continued weekly subcutaneous semaglutide vs placebo on weight loss maintenance in adults with overweight or obesity: The STEP 4 randomized clinical trial. JAMA 2021, 325 (14), 1414–1425. [CrossRef]
- Garvey, W. T.; Batterham, R. L.; Bhatta, M.; Buscemi, S.; Christensen, L. N.; Frias, J. P.; Jódar, E.; Kandler, K.; Rigas, G.; Wadden, T. A.; Wharton, S.; STEP 5 Study Group. Two-year effects of semaglutide in adults with overweight or obesity: The STEP 5 trial. Nat. Med. 2022, 28 (10), 2083–2091. [CrossRef]
- Kadowaki, T.; Isendahl, J.; Khalid, U.; Lee, S. Y.; Nishida, T.; Ogawa, W.; Tobe, K.; Yamauchi, T.; Lim, S.; STEP 6 Investigators. Semaglutide once a week in adults with overweight or obesity, with or without type 2 diabetes in an East Asian population (STEP 6): A randomised, double-blind, double-dummy, placebo-controlled, phase 3a trial. Lancet Diabetes Endocrinol. 2022, 10 (3), 193–206. [CrossRef]
- Rubino, D. M.; Greenway, F. L.; Khalid, U.; O'Neil, P. M.; Rosenstock, J.; Sørrig, R.; Wadden, T. A.; Wizert, A.; Garvey, W. T.; STEP 8 Investigators. Effect of weekly subcutaneous semaglutide vs daily liraglutide on body weight in adults with overweight or obesity without diabetes: The STEP 8 randomized clinical trial. JAMA 2022, 327 (2), 138–150. PMID: 35015037; PMCID: PMC8753508. [CrossRef]
- Humphreys, S. The unethical use of BMI in contemporary general practice. Br. J. Gen. Pract. 2010, 60 (578), 696–697.
- Pétré, B.; Scheen, A.; Ziegler, O.; Donneau, A. F.; Dardenne, N.; Husson, E.; Guillaume, M. Weight loss expectations and determinants in a large community-based sample. Prev. Med. Rep. 2018, 12, 12–19.
- Flore, G.; Preti, A.; Carta, M. G.; Deledda, A.; Fosci, M.; Nardi, A. E.; Velluzzi, F. Weight maintenance after dietary weight loss: Systematic review and meta-analysis on the effectiveness of behavioural intensive intervention. Nutrients 2022, 14 (6), 1259.
- Lowe, M. R.; Kral, T. V.; Miller-Kovach, K. Weight-loss maintenance 1, 2 and 5 years after successful completion of a weight-loss programme. Br. J. Nutr. 2008, 99 (4), 925–930.
- Hintze, L. J.; Mahmoodianfard, S.; Auguste, C. B.; Doucet, É. Weight loss and appetite control in women. Curr. Obes. Rep. 2017, 6, 334–351.
- Miller-Kovach, K.; Hermann, M.; Winick, M. The psychological ramifications of weight management. J. Women's Health Gend. Based Med. 1999, 8 (4), 477–482.
- Emmerich, S. D.; Fryar, C. D.; Stierman, B.; Ogden, C. L. Obesity and severe obesity prevalence in adults: United States, August 2021–August 2023. NCHS Data Brief 2024, 508. https://www.cdc.gov/nchs/data/databriefs/db508.pdf.
- Maegawa, S.; Lu, Y.; Tahara, T.; Lee, J. T.; Madzo, J.; Liang, S.; Jelinek, J.; Colman, R. J.; Issa, J. P. J. Caloric restriction delays age-related methylation drift. Nat. Commun. 2017, 8 (1), 539. [CrossRef]
- Hahn, O.; Grönke, S.; Stubbs, T. M.; Ficz, G.; Hendrich, O.; Krueger, F.; Andrews, S.; Zhang, Q.; Wakelam, M. J.; Beyer, A.; Reik, W.; Partridge, L. Dietary restriction protects from age-associated DNA methylation and induces epigenetic reprogramming of lipid metabolism. Genome Biol. 2017, 18, 56. [CrossRef]
- Petkovich, D. A.; Podolskiy, D. I.; Lobanov, A. V.; Lee, S. G.; Miller, R. A.; Gladyshev, V. N. Using DNA methylation profiling to evaluate biological age and longevity interventions. Cell Metab. 2017, 25 (4), 954–960.e6. [CrossRef]
- Fontana, L. The scientific basis of caloric restriction leading to longer life. Curr. Opin. Gastroenterol. 2009, 25 (2), 144–150. https://pubmed.ncbi.nlm.nih.gov/19262201/.
- Dutton, G. R.; Perri, M. G.; Dancer-Brown, M.; Goble, M.; Van Vessem, N. Weight loss goals of patients in a health maintenance organization. Eat. Behav. 2010, 11 (2), 74–78. https://pubmed.ncbi.nlm.nih.gov/20188289/.
- Fildes, A.; Charlton, J.; Rudisill, C.; Littlejohns, P.; Prevost, A. T.; Gulliford, M. C. Probability of an obese person attaining normal body weight: Cohort study using electronic health records. Am. J. Public Health 2015, 105 (9), e54–e59. https://pubmed.ncbi.nlm.nih.gov/23747584/.
- Han, S. H.; Safeek, R.; Ockerman, K.; Trieu, N.; Mars, P.; Klenke, A.; Furnas, H.; Sorice-Virk, S. Public interest in the off-label use of glucagon-like peptide 1 agonists (Ozempic) for cosmetic weight loss: A Google Trends analysis. Aesthet. Surg. J. 2024, 44 (1), 60–67. [CrossRef]
- Montero, A.; Sparks, G.; Presiado, M.; Hamel, L. KFF Health Tracking Poll May 2024: The public’s use and views of GLP-1 drugs; Kaiser Family Foundation, May 10, 2024. https://www.kff.org/health-costs/poll-finding/kff-health-tracking-poll-may-2024-the-publics-use-and-views-of-glp-1-drugs/.
- Sochovsky, N.; Miles, H. Global Equity Observer GLP-1: The weight of speculation; Morgan Stanley Investment Management, Dec 2023. https://www.morganstanley.com/im/publication/insights/articles/article_geoglp1_us.pdf.
- Colman, R. J.; Beasley, T. M.; Kemnitz, J. W.; Johnson, S. C.; Weindruch, R.; Anderson, R. M. Caloric restriction reduces age-related and all-cause mortality in rhesus monkeys. Nat. Commun. 2014, 5, 3557. https://pubmed.ncbi.nlm.nih.gov/24691430/.
- Mattison, J. A.; Roth, G. S.; Beasley, T. M.; Tilmont, E. M.; Handy, A. M.; Herbert, R. L.; Longo, D. L.; Allison, D. B.; Young, J. E.; Bryant, M.; Barnard, D.; Ward, W. F.; Qi, W.; Ingram, D. K.; de Cabo, R. Impact of caloric restriction on health and survival in rhesus monkeys from the NIA study. Nature 2012, 489 (7415), 318–321. [CrossRef]
- Das, J. K.; Banskota, N.; Candia, J.; Griswold, M. E.; Orenduff, M.; de Cabo, R.; Corcoran, D. L.; Das, S. K.; De, S.; Huffman, K. M.; Kraus, V. B.; Kraus, W. E.; Martin, C. K.; Racette, S. B.; Redman, L. M.; Schilling, B.; Belsky, D. W.; Ferrucci, L. Calorie restriction modulates the transcription of genes related to stress response and longevity in human muscle: The CALERIE study. Aging Cell 2023, 22 (12), e13963. PMID: 37823711; PMCID: PMC10726900. [CrossRef]
- Gutin, I. In BMI we trust: Reframing the body mass index as a measure of health. Soc. Theory Health 2018, 16 (3), 256–271. https://pubmed.ncbi.nlm.nih.gov/31007613/.
- McEvedy, S. M.; Sullivan-Mort, G.; McLean, S. A.; Pascoe, M. C.; Paxton, S. J. Ineffectiveness of commercial weight-loss programs for achieving modest but meaningful weight loss: Systematic review and meta-analysis. J. Health Psychol. 2017, 22 (12), 1614–1627. https://pubmed.ncbi.nlm.nih.gov/28810454/.
- Hayashi, D.; Edwards, C.; Emond, J. A.; Gilbert-Diamond, D.; Butt, M.; Rigby, A.; et al. What is food noise? A conceptual model of food cue reactivity. Nutrients 2023, 15 (22), 4809. PMID: 38004203; PMCID: PMC10674813. [CrossRef]
- Blaine, B. E.; Rodman, J.; Newman, J. M. Weight loss treatment and psychological well-being: A review and meta-analysis. J. Health Psychol. 2007, 12 (1), 66–82. https://pubmed.ncbi.nlm.nih.gov/22888821/.
- Jackson, S. E.; Steptoe, A.; Beeken, R. J.; Kivimaki, M.; Wardle, J. Psychological changes following weight loss in overweight and obese adults: A prospective cohort study. PLoS One 2014, 9 (8), e104552. https://pubmed.ncbi.nlm.nih.gov/30326501/.
- Swencionis, C.; Wylie-Rosett, J.; Lent, M. R.; Ginsberg, M.; Cimino, C.; Wassertheil-Smoller, S.; et al. Weight change, psychological well-being, and vitality in adults participating in a cognitive-behavioral weight loss program. Health Psychol. 2013, 32 (4), 439–446. https://pubmed.ncbi.nlm.nih.gov/25098417/.
- Chlabicz, M.; Dubatówka, M.; Jamiołkowski, J.; Sowa, P.; Łapińska, M.; Raczkowski, A.; et al. Subjective well-being in non-obese individuals depends strongly on body composition. Sci. Rep. 2021, 11 (1), 21797. https://www.nature.com/articles/s41598-021-01205-6.
- Tahrani, A. A.; Morton, J. Benefits of weight loss of 10% or more in patients with overweight or obesity: A review. Obesity (Silver Spring) 2022. [CrossRef]
- Ryan, D. H.; Lingvay, I.; Deanfield, J.; et al. Long-term weight loss effects of semaglutide in obesity without diabetes in the SELECT trial. Nat. Med. 2024, 30, 2049–2057. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




