Submitted:
10 June 2025
Posted:
12 June 2025
Read the latest preprint version here
Abstract
Keywords:
Introduction
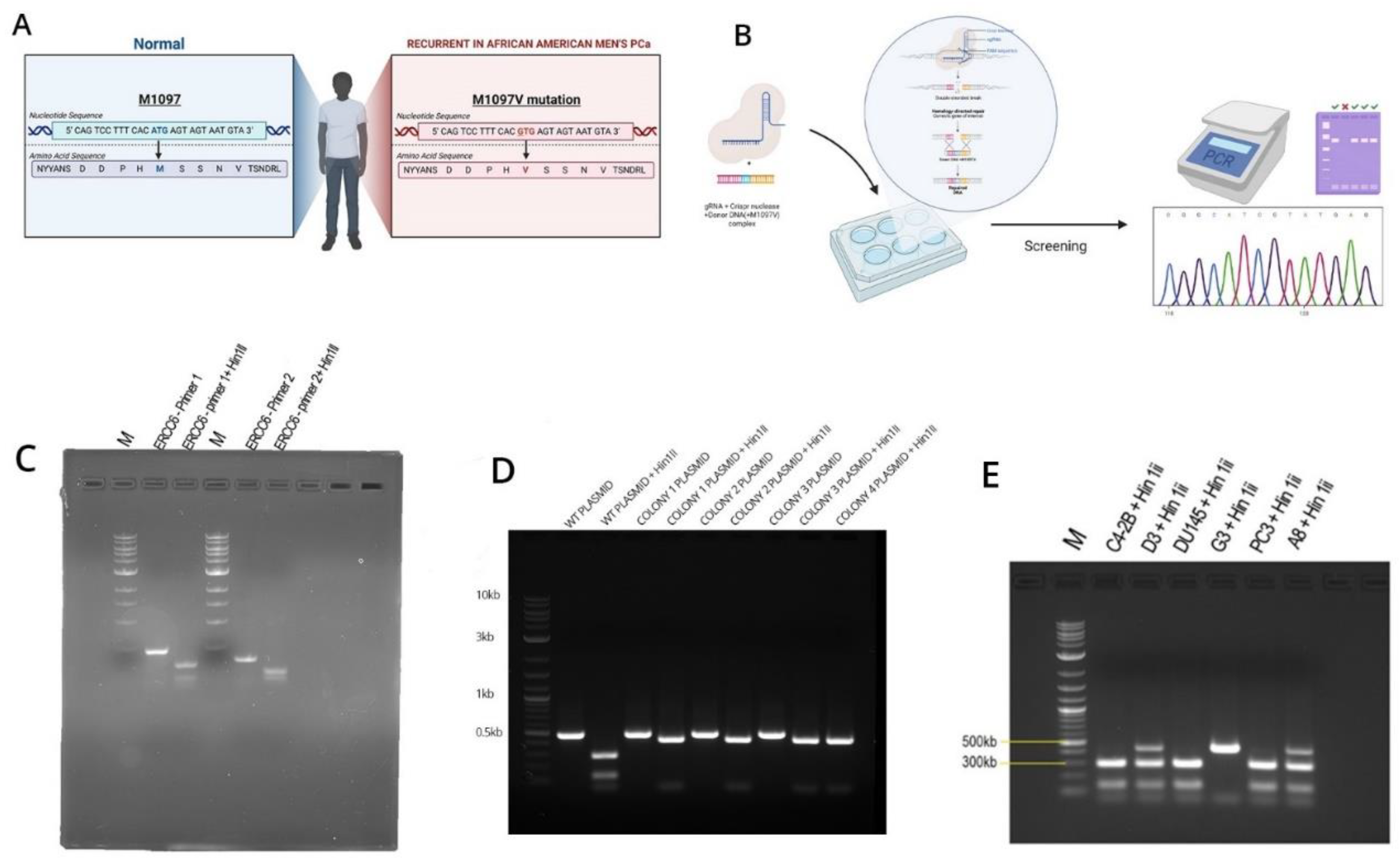
Crispr Site-Directed Mutagenesis Experiment
Gel Electrophoresis
PCR- Restriction Fragment Length Polymorphism(RFLP)
Dot Blot (DNA Southwestern Blot)
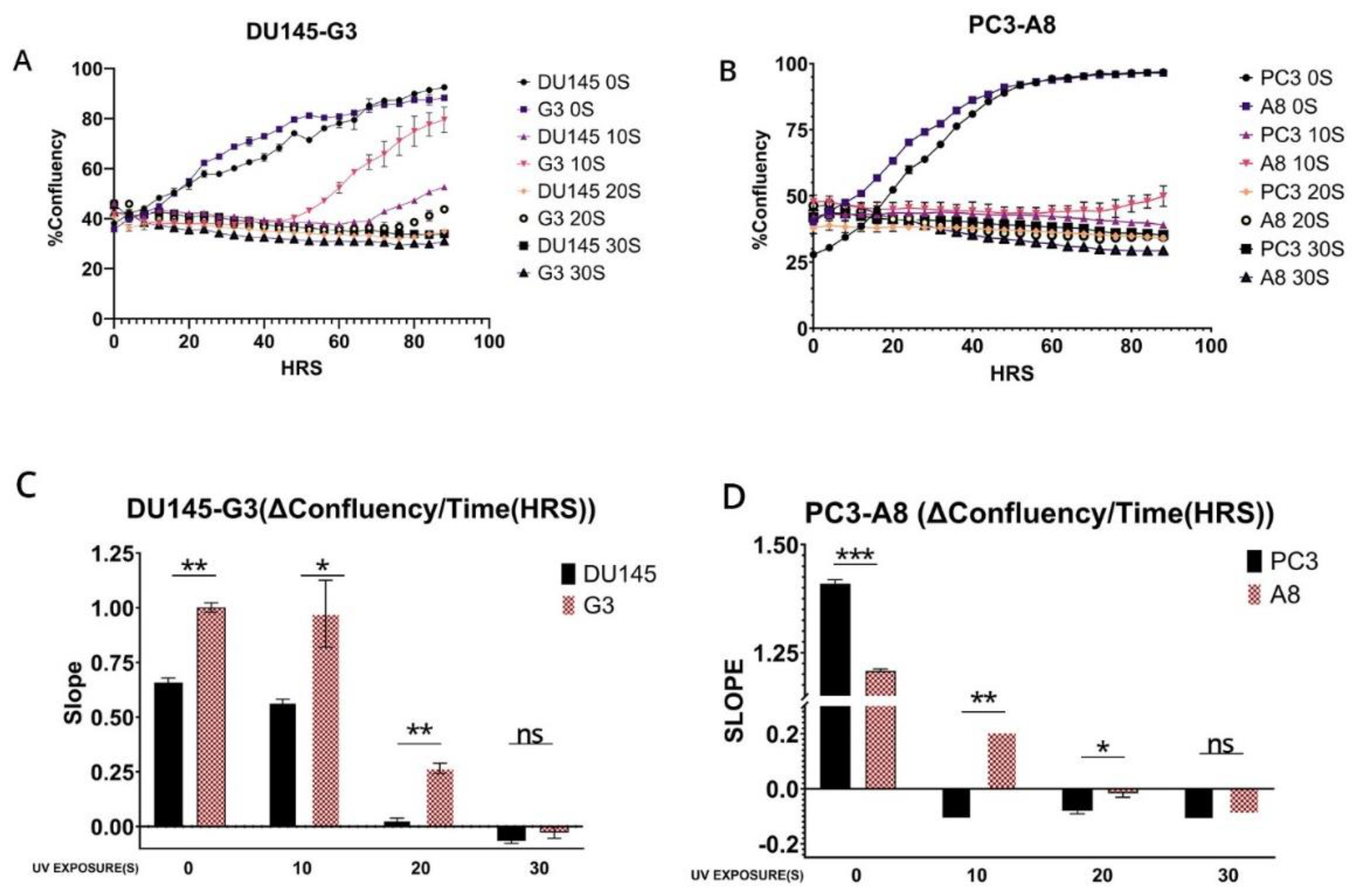
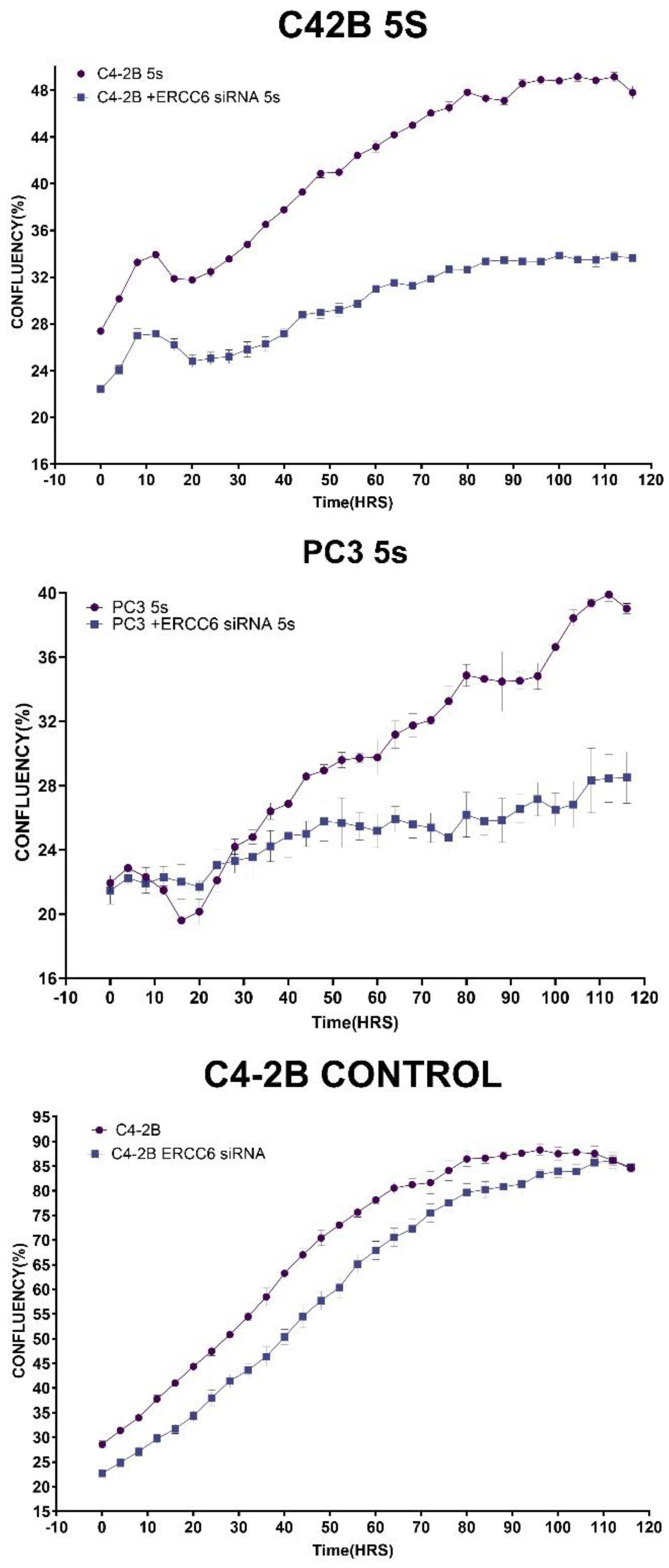
Proliferation Assay
Statistical Analysis
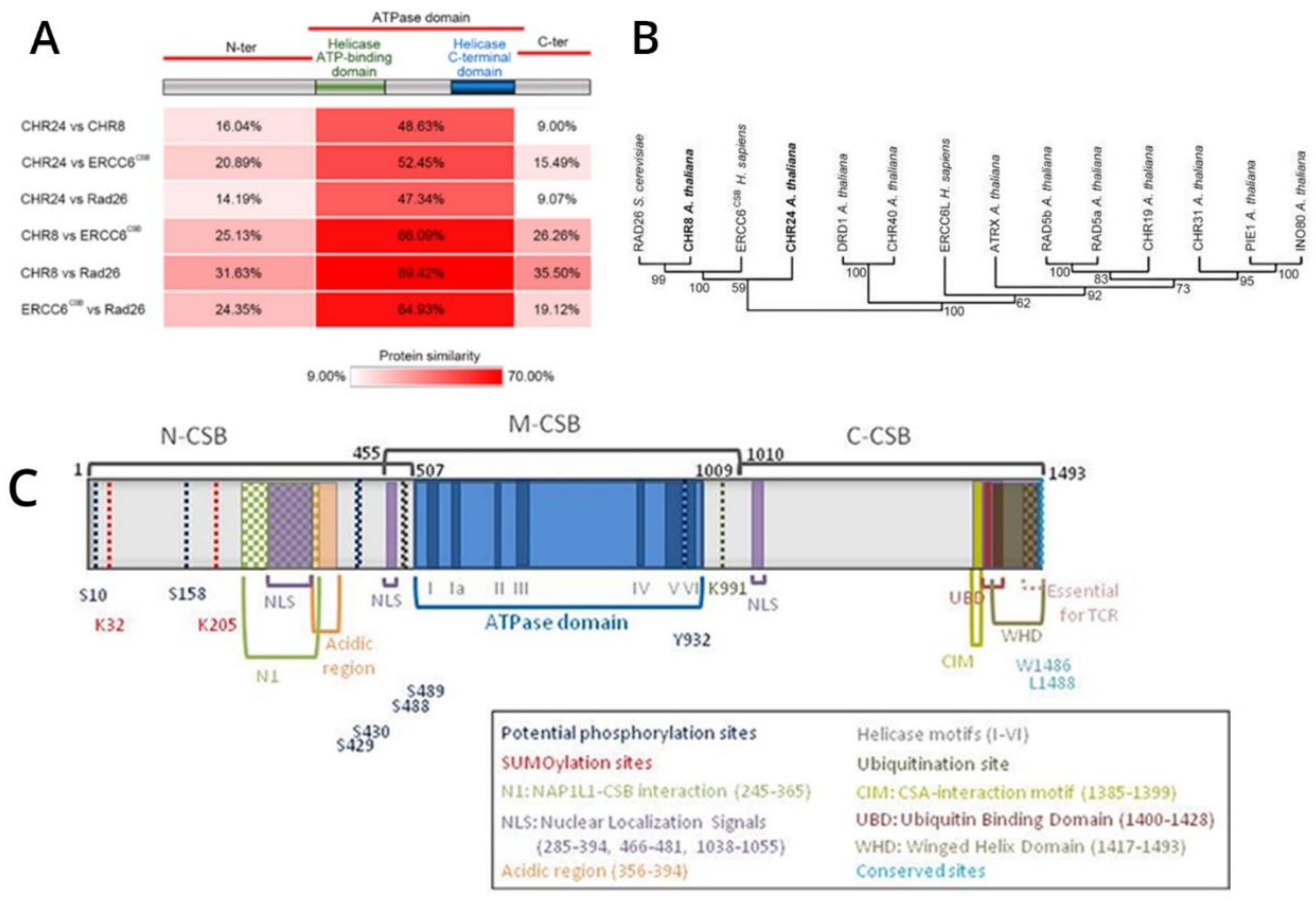
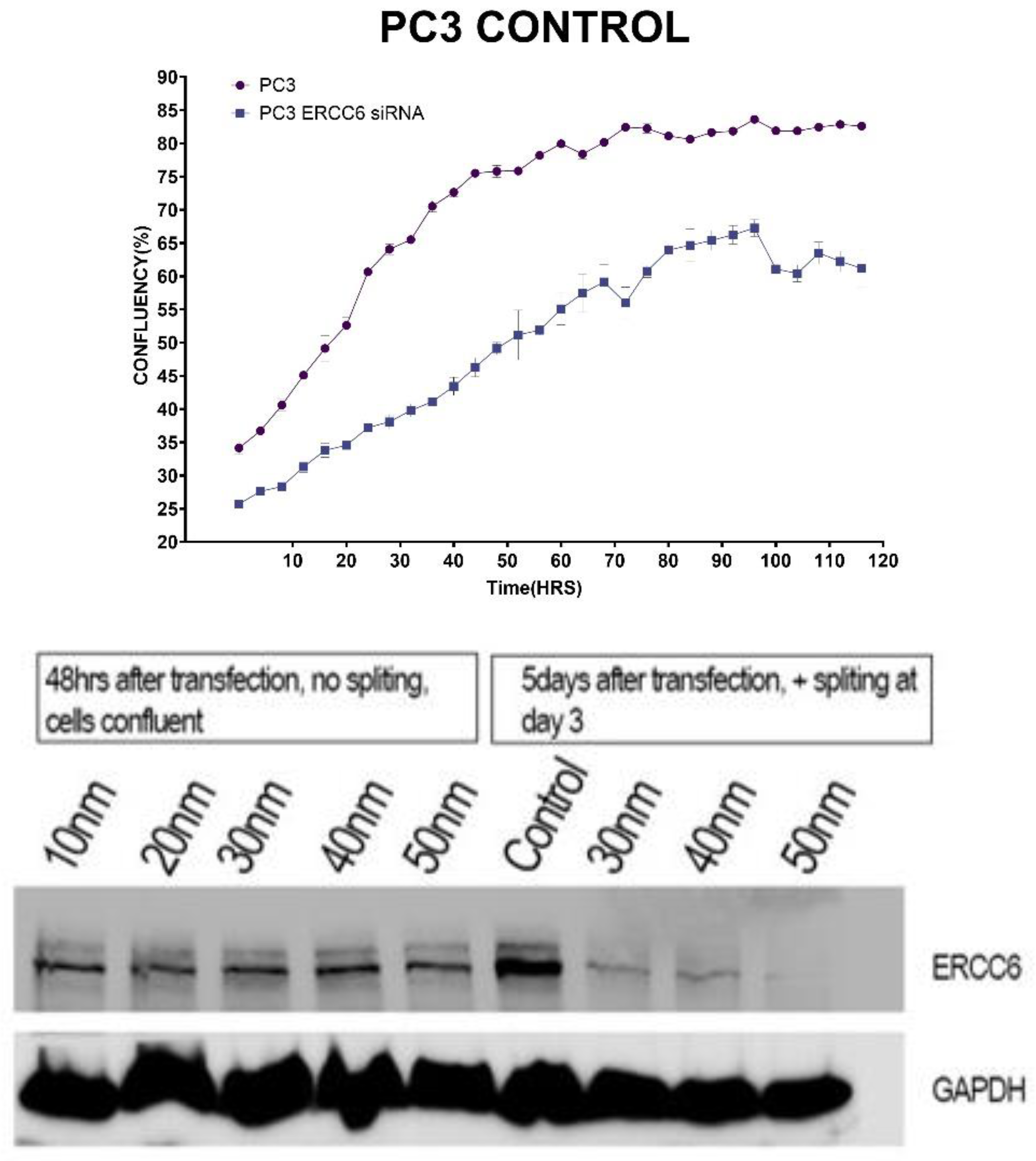
Results
Discussion
- General Activities of ERCC6/CSB:
-
Transcription-Coupled DNA Repair (TC-NER):
- ○
- ERCC6 plays a central role in detecting and initiating the repair of DNA lesions that block transcription.
- ○
- When RNA polymerase II stalls at DNA damage (such as UV-induced cyclobutane pyrimidine dimers), ERCC6 helps recruit repair factors to remove the lesion and resume transcription.
-
Chromatin Remodeling:
- ○
- ERCC6 possesses ATP-dependent chromatin remodeling activity, allowing it to alter nucleosome positioning.
- ○
- This activity is crucial for providing repair machinery access to DNA in compact chromatin regions.
-
Transcription Regulation:
- ○
- In addition to DNA repair, ERCC6 can regulate gene expression by interacting with transcription machinery.
- ○
- It influences RNA polymerase 1 and RNA polymerase II pausing and restart, ensuring proper transcription resumption after repair.
-
Interaction with Other Repair Proteins:
- ○
- ERCC6 interacts with other TC-NER factors such as CSA (ERCC8), XPG, TFIIH, and UVSSA to coordinate the repair process.
-
Response to Oxidative Stress:
- ○
- ERCC6 has been implicated in the repair of oxidative DNA damage, not just UV-induced lesions.
- ○
- It helps maintain mitochondrial function and cellular redox balance under stress conditions.
-
Role in Disease:
- ○
- Mutations in ERCC6 cause Cockayne Syndrome, a rare autosomal recessive disorder characterized by growth failure, neurodegeneration, and premature aging.
- ○
- It is also linked to other neurodevelopmental and progeroid syndromes.
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- CHANG C-H, CHIU C-F, WANG H-C, WU H-C, TSAI R-Y, TSAI C-W, WANG R-F, WANG C-H, TSOU Y-A, BAU D-T: Significant Association of ERCC6 Single Nucleotide Polymorphisms with Bladder Cancer Susceptibility in Taiwan. Anticancer Research 2009, 29(12):5121-5124.
- Forrest IS, Chaudhary K, Vy HMT, Bafna S, Kim S, Won HH, Loos RJF, Cho J, Pasquale LR, Nadkarni GN et al: Genetic pleiotropy of ERCC6 loss-of-function and deleterious missense variants links retinal dystrophy, arrhythmia, and immunodeficiency in diverse ancestries. Hum Mutat 2021, 42(8):969-977. [CrossRef]
- Lin X, Wu Y, Li Q, Yu H, Li X, Li X, Zheng J: Genetic Association of ERCC6 rs2228526 Polymorphism with the Risk of Cancer: Evidence from a Meta-Analysis. Biomed Res Int 2022, 2022:2662666. [CrossRef]
- Putnam CD: Strand discrimination in DNA mismatch repair. DNA Repair (Amst) 2021, 105:103161. [CrossRef]
- Loeb LA: A mutator phenotype in cancer. Cancer Res 2001, 61(8):3230-3239.
- Kakarla M, ChallaSivaKanaka S, Hayward SW, Franco OE: Race as a Contributor to Stromal Modulation of Tumor Progression. Cancers 2021, 13(11):2656. [CrossRef]
- Teng PC, Huang SP, Liu CH, Lin TY, Cho YC, Lai YL, Wang SC, Yeh HC, Chuu CP, Chen DN et al: Identification of DNA Damage Repair-Associated Prognostic Biomarkers for Prostate Cancer Using Transcriptomic Data Analysis. Int J Mol Sci 2021, 22(21). [CrossRef]
- Schmid S, Omlin A, Higano C, Sweeney C, Martinez Chanza N, Mehra N, Kuppen MCP, Beltran H, Conteduca V, Vargas Pivato de Almeida D et al: Activity of Platinum-Based Chemotherapy in Patients With Advanced Prostate Cancer With and Without DNA Repair Gene Aberrations. JAMA network open 2020, 3(10):e2021692-e2021692. 2021. [CrossRef]
- Zhang W, Van Gent DC, Incrocci L, Van Weerden WM, Nonnekens J: Role of the DNA damage response in prostate cancer formation, progression and treatment. Prostate cancer and prostatic diseases 2020, 23(1):24-37. [CrossRef]
- Banks P, Xu W, Murphy D, James P, Sandhu S: Relevance of DNA damage repair in the management of prostate cancer. Current problems in cancer 2017, 41(4):287-301. [CrossRef]
- de Bono J, Mateo J, Fizazi K, Saad F, Shore N, Sandhu S, Chi KN, Sartor O, Agarwal N, Olmos D et al: Olaparib for Metastatic Castration-Resistant Prostate Cancer. New England Journal of Medicine 2020, 382(22):2091-2102. [CrossRef]
- Abida W, Campbell D, Patnaik A, Shapiro JD, Sautois B, Vogelzang NJ, Voog EG, Bryce AH, McDermott R, Ricci F et al: Non-BRCA DNA Damage Repair Gene Alterations and Response to the PARP Inhibitor Rucaparib in Metastatic Castration-Resistant Prostate Cancer: Analysis From the Phase II TRITON2 Study. Clin Cancer Res 2020, 26(11):2487-2496. [CrossRef]
- Perše M: Cisplatin Mouse Models: Treatment, Toxicity and Translatability. Biomedicines 2021, 9(10):1406. [CrossRef]
- Su LJ, Arab L, Steck SE, Fontham ET, Schroeder JC, Bensen JT, Mohler JL: Obesity and prostate cancer aggressiveness among African and Caucasian Americans in a population-based study. Cancer Epidemiol Biomarkers Prev 2011, 20(5):844-853. [CrossRef]
- Baurick T, Younes L, Meiners J: Welcome to "Cancer Alley" where toxic air is about to get worse. In: Propublica. 2019.
- Yadav S, Anbalagan M, Baddoo M, Chellamuthu VK, Mukhopadhyay S, Woods C, Jiang W, Moroz K, Flemington EK, Makridakis N: Somatic mutations in the DNA repairome in prostate cancers in African Americans and Caucasians. Oncogene 2020, 39(21):4299-4311. [CrossRef]
- Singh V, Bhoir S, Chikhale RV, Hussain J, Dwyer D, Bryce RA, Kirubakaran S, De Benedetti A: Generation of phenothiazine with potent anti-TLK1 activity for prostate cancer therapy. Iscience 2020, 23(9):101474. [CrossRef]
- Singh V, Jaiswal PK, Ghosh I, Koul HK, Yu X, De Benedetti A: Targeting the TLK1/NEK1 DDR axis with Thioridazine suppresses outgrowth of androgen independent prostate tumors. International journal of cancer 2019, 145(4):1055-1067. [CrossRef]
- Singh V, Jaiswal PK, Ghosh I, Koul HK, Yu X, De Benedetti A: The TLK1-Nek1 axis promotes prostate cancer progression. Cancer letters 2019, 453:131-141. [CrossRef]
- Bhoir S, Ogundepo O, Yu X, Shi R, De Benedetti A: Exploiting TLK1 and Cisplatin Synergy for Synthetic Lethality in Androgen-Insensitive Prostate Cancer. Biomedicines 2023, 11(11):2987. [CrossRef]
- Hinata N, Fujisawa M: Racial Differences in Prostate Cancer Characteristics and Cancer-Specific Mortality: An Overview. World J Mens Health 2022, 40(2):217-227. [CrossRef]
- Kakarla M, ChallaSivaKanaka S, Hayward SW, Franco OE: Race as a Contributor to Stromal Modulation of Tumor Progression. Cancers 2021, 13(11):2656. [CrossRef]
- Teng PC, Huang SP, Liu CH, Lin TY, Cho YC, Lai YL, Wang SC, Yeh HC, Chuu CP, Chen DN et al: Identification of DNA Damage Repair-Associated Prognostic Biomarkers for Prostate Cancer Using Transcriptomic Data Analysis. Int J Mol Sci 2021, 22(21). [CrossRef]
- Schmid S, Omlin A, Higano C, Sweeney C, Martinez Chanza N, Mehra N, Kuppen MCP, Beltran H, Conteduca V, Vargas Pivato de Almeida D et al: Activity of Platinum-Based Chemotherapy in Patients With Advanced Prostate Cancer With and Without DNA Repair Gene Aberrations. JAMA network open 2020, 3(10):e2021692-e2021692. [CrossRef]
- Zhang W, Van Gent DC, Incrocci L, Van Weerden WM, Nonnekens J: Role of the DNA damage response in prostate cancer formation, progression and treatment. Prostate cancer and prostatic diseases 2020, 23(1):24-37. [CrossRef]
- Banks P, Xu W, Murphy D, James P, Sandhu S: Relevance of DNA damage repair in the management of prostate cancer. Current problems in cancer 2017, 41(4):287-301. [CrossRef]
- de Bono J, Mateo J, Fizazi K, Saad F, Shore N, Sandhu S, Chi KN, Sartor O, Agarwal N, Olmos D et al: Olaparib for Metastatic Castration-Resistant Prostate Cancer. New England Journal of Medicine 2020, 382(22):2091-2102. [CrossRef]
- Abida W, Campbell D, Patnaik A, Shapiro JD, Sautois B, Vogelzang NJ, Voog EG, Bryce AH, McDermott R, Ricci F et al: Non-BRCA DNA Damage Repair Gene Alterations and Response to the PARP Inhibitor Rucaparib in Metastatic Castration-Resistant Prostate Cancer: Analysis From the Phase II TRITON2 Study. Clin Cancer Res 2020, 26(11):2487-2496. [CrossRef]
- Perše M: Cisplatin Mouse Models: Treatment, Toxicity and Translatability. Biomedicines 2021, 9(10):1406. [CrossRef]
- Yadav S, Anbalagan M, Baddoo M, Chellamuthu VK, Mukhopadhyay S, Woods C, Jiang W, Moroz K, Flemington EK, Makridakis N: Somatic mutations in the DNA repairome in prostate cancers in African Americans and Caucasians. Oncogene 2020, 39(21):4299-4311. [CrossRef]
- CHANG C-H, CHIU C-F, WANG H-C, WU H-C, TSAI R-Y, TSAI C-W, WANG R-F, WANG C-H, TSOU Y-A, BAU D-T: Significant Association of ERCC6 Single Nucleotide Polymorphisms with Bladder Cancer Susceptibility in Taiwan. Anticancer Research 2009, 29(12):5121-5124.
- Forrest IS, Chaudhary K, Vy HMT, Bafna S, Kim S, Won HH, Loos RJF, Cho J, Pasquale LR, Nadkarni GN et al: Genetic pleiotropy of ERCC6 loss-of-function and deleterious missense variants links retinal dystrophy, arrhythmia, and immunodeficiency in diverse ancestries. Hum Mutat 2021, 42(8):969-977. [CrossRef]
- Lin X, Wu Y, Li Q, Yu H, Li X, Li X, Zheng J: Genetic Association of ERCC6 rs2228526 Polymorphism with the Risk of Cancer: Evidence from a Meta-Analysis. Biomed Res Int 2022, 2022:2662666. [CrossRef]
- Su LJ, Arab L, Steck SE, Fontham ET, Schroeder JC, Bensen JT, Mohler JL: Obesity and prostate cancer aggressiveness among African and Caucasian Americans in a population-based study. Cancer Epidemiol Biomarkers Prev 2011, 20(5):844-853. [CrossRef]
- Baurick T, Younes L, Meiners J: Welcome to "Cancer Alley" where toxic air is about to get worse. In: Propublica. 2019.
- Khalil MI, Ghosh I, Singh V, Chen J, Zhu H, De Benedetti A: NEK1 Phosphorylation of YAP Promotes Its Stabilization and Transcriptional Output. Cancers 2020, 12(12):3666. [CrossRef]
- Basei FL, Meirelles GV, Righetto GL, Dos Santos Migueleti DL, Smetana JH, Kobarg J: New interaction partners for Nek4.1 and Nek4.2 isoforms: from the DNA damage response to RNA splicing. Proteome Sci 2015, 13:11. [CrossRef]
- Putnam CD: Strand discrimination in DNA mismatch repair. DNA Repair (Amst) 2021, 105:103161. [CrossRef]
- Loeb LA: A mutator phenotype in cancer. Cancer Res 2001, 61(8):3230-3239.
- Singh V, Bhoir S, Chikhale RV, Hussain J, Dwyer D, Bryce RA, Kirubakaran S, De Benedetti A: Generation of phenothiazine with potent anti-TLK1 activity for prostate cancer therapy. Iscience 2020, 23(9):101474. [CrossRef]
- Singh V, Jaiswal PK, Ghosh I, Koul HK, Yu X, De Benedetti A: Targeting the TLK1/NEK1 DDR axis with Thioridazine suppresses outgrowth of androgen independent prostate tumors. International journal of cancer 2019, 145(4):1055-1067. [CrossRef]
- Singh V, Jaiswal PK, Ghosh I, Koul HK, Yu X, De Benedetti A: The TLK1-Nek1 axis promotes prostate cancer progression. Cancer letters 2019, 453:131-141. [CrossRef]
- Bhoir S, Ogundepo O, Yu X, Shi R, De Benedetti A: Exploiting TLK1 and Cisplatin Synergy for Synthetic Lethality in Androgen-Insensitive Prostate Cancer. Biomedicines 2023, 11(11):2987. [CrossRef]
- Hinata N, Fujisawa M: Racial Differences in Prostate Cancer Characteristics and Cancer-Specific Mortality: An Overview. World J Mens Health 2022, 40(2):217-227. [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



