Introduction
With over 5.3 billion doses administered globally, the mRNA-containing lipid nanoparticle (mRNA–LNP)-based COVID-19 vaccines, Pfizer and BioNTech’s Comirnaty and Moderna’s Spikevax, these vaccines remain the primary tool for maintaining immunity against circulating SARS-CoV-2 variants. Nonetheless, due to the declining threat of the virus and increasing concern over the uniquely broad spectrum and relatively high incidence of adverse events (AEs), some countries and regions have discontinued their use, and the U.S. Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA) have recently limited their recommendations for vaccination with these products.
Regarding the AEs, they are statistically rare on an individual basis (officially reported at 0.03–0.5%) [
1], the vast scale of global vaccination campaigns has resulted vaccine-related injuries in the millions. These adverse outcomes have been collectively described as post-vaccination syndrome (PVS) [
2,
3,
4,
5,
6,
7], a newly recognized condition which -in individuals with persistent health issues or disabilities-, may qualify as an iatrogenic orphan disease [
1]. In some cases, simultaneous inflammation of multiple organs can occur, leading to a highly lethal condition known as multisystem inflammatory response syndrome.
The objective of this review is to focus on the role of endothelial cells (ECs) within the microcirculation as a likely primary site of the development of vaccine-induced injury. A better understanding of these processes is essential for improving the safety, and thereby the future success, of mRNA–LNP platform technology, not only for vaccination but also for other therapeutic purposes.
2. The mRNA–LNP-induced Inflammatory Complications and their Root Cause
Table 1 shows a list of organ-specific inflammatory symptoms or illnesses that were reported in the publicly available 3-month safety surveillance report of Pfizer [
8].The advantage of using these data is that the list of AEs likely excludes symptoms caused by SARS-CoV-2 infection, as the vaccine had not been administered to infected individuals. The list was compiled using data from 52 countries, minimizing the risk of regional bias. Artificial intelligence assisted in the process by scanning over 1,200 entries for terms containing the suffix “-itis”, followed by screening to exclude redundant qualifiers such as “acute”, “chronic”, “(auto)immune”, “bacterial”, or “viral”. Thus, the list does not distinguish among acute or chronic inflammatory conditions due to bacterial or viral infections or autoimmune attack; it just shows that essentially all organs can be inflicted with a form of inflammation starting sometimes within 3 months after vaccination.
The similar kinetics of vaccine-induced inflammatory AEs listed in
Table 1 (developing within days or much later, in months after vaccination) and numerous commonalties in these illnesses, such as the association with reactivation of certain viral strains (
Table 2), point to a common, very fundamental immune abnormality or combination of abnormalities underlying these conditions.
A recent comprehensive theory on this conundrum attributed the phenomenon, at least in part, to plausible consequences of inherent structural and functional properties of mRNA-LNPs [
1,
9]. The concerns related to the design of mRNA vaccines vaccine include: (i) ribosomal synthesis of the antigen (spike protein), which fundamentally alters the natural process of antigen processing and presentation; (ii) extensive chemical modification of the mRNA, rendering the spike protein translation poorly controllable and mutation-prone upon codon–anticodon pairing; (iii) the use of a proinflammatory, fusogenic aminolipid in the LNP, which promotes widespread systemic distribution of the mRNA-LNP entailing transfection of non-target cells to produce a toxin; (iv) the chemical stabilization of the spike protein, despite its known multiorgan, polymodal toxicity; (v) the choice of LNP lipid composition with reduced nanoparticle stability in aqueous environments; (vi) the PEGylation of the LNP surface, despite PEG’s known immune reactivity and immunogenicity; and (vii) recombinant production of the mRNA, which carries a risk for DNA contamination. These features collectively predispose to collateral tissue damage caused by inflammation involving cytotoxic T cell and complement-mediated autoimmune responses. Although nucleoside modifications (e.g., pseudouridine substitution) of the mRNA reduce recognition by innate immune sensors, such as TLR3, TLR7, TLR8, and retinoic acid-inducible gene I (RIG-I) [
10,
11,
12,
13,
14], residual activation of innate immunity by the modified mRNA via RIG-I-like receptors remains a possibility under certain conditions [
15].
Given the multicausal and multiorgan nature of vaccine-induced inflammation, numerous pathological processes are likely involved. This review focuses on microcirculatory inflammation, especially on the endothelium, as a central driver of inflammatory AEs. The task is complicated by tissue-specific differences in microcirculatory structure and function, as described below.
3. Components and Unique Organization of Microcirculation Across Tissues
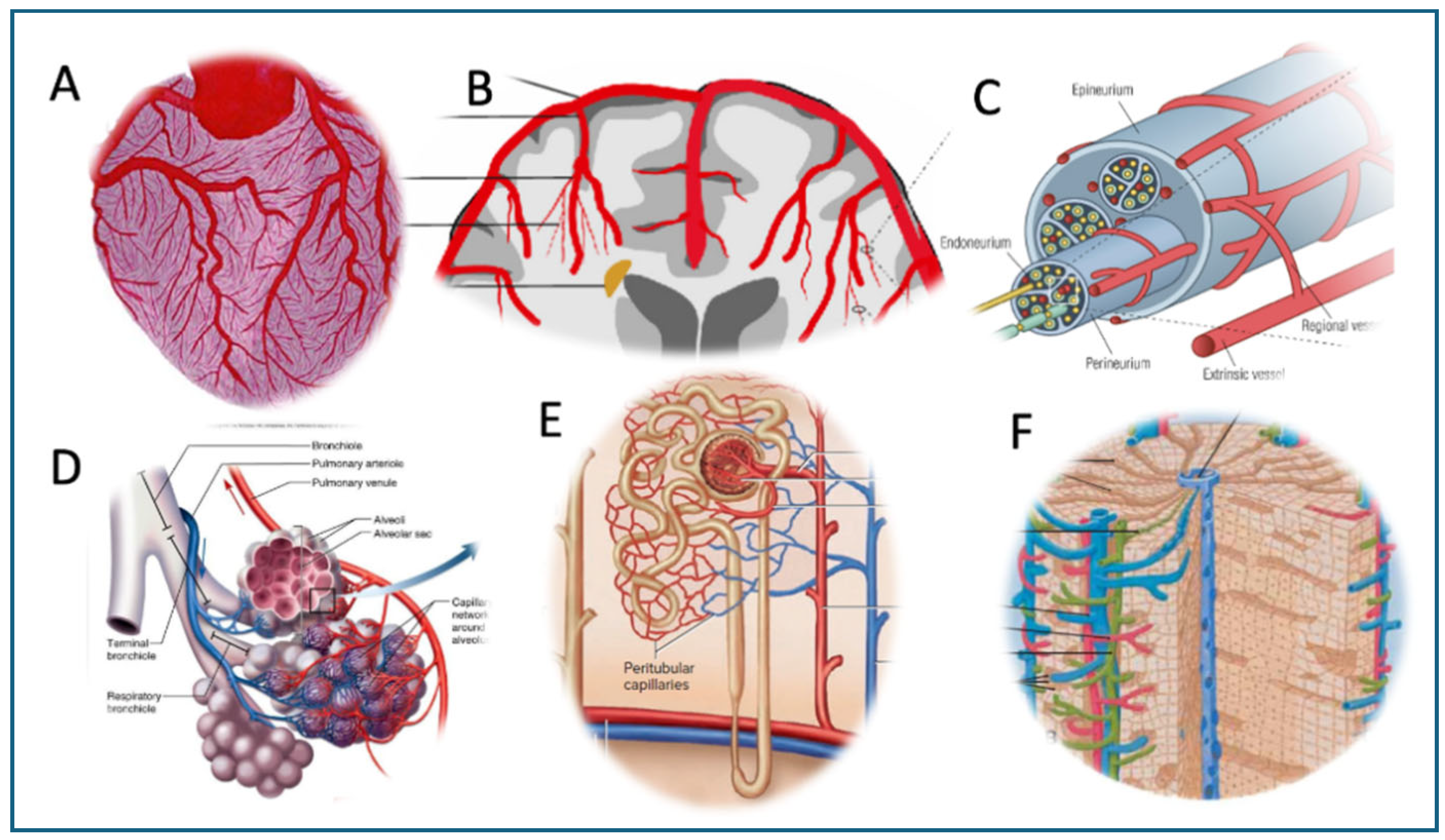
The microcirculation refers to the smallest blood vessels in the body (below ~200 micrometer), including arterioles, capillaries, and venules which collectively responsible for gas and nutrient exchange, waste removal, and immune surveillance at the tissue level. Some morphological features of cells in the microcirculation are illustrated in
Figure 1.
4. Endothelial Cells in the Frontline of Systemic Inflammation
Endothelial cells, being an interface between blood and tissues, are particularly responsive to inflammatory stimuli and are often the first point of contact for systemically circulating immune mediators and nanoparticles, including mRNA-LNPs. Damage to the glycocalyx, and, hence, endothelial integrity, can precipitate secondary cascades such as coagulation, complement activation, immune cell infiltration, all of which contribute to various pathophysiological states, including inflammation, sepsis, ischemia-reperfusion injury, and vascular complications related to infections and vaccinations [
25,
26]. The manifestations of symptoms and illnesses vary widely across organs, reflecting not only differences in immune responses but also organ-specific anatomical factors, discussed below.
5. Distinctive Microcirculatory Architectures Across Organs and Their Impacts on Vaccine-Induced Inflammations
Although all organs share a basic microvascular blueprint, the microcirculatory networks differ markedly due to a variety of anatomical, physiological, and functional factors. These include the density of the capillary network (
Figure 1A), EC ultrastructure and architecture (
Figure 1B,C), and endothelial subtype based on the tightness of intercellular junctions (
Figure 1D) [
25,
27]. Additional control is exerted by local regulatory mechanisms involving adjacent tissue cells, such as pericytes around nerve fibers (
Figure 1C), smooth muscle cells in precapillary sphincters (
Figure 1A), or other resistance elements that modulate blood flow in response to tissue-specific functional demands [
28]. These structural and functional variations render the microcirculation a highly specialized, tissue-specific interface between the bloodstream and parenchymal cells. Consequently, they critically influence the nature and extent of inflammatory responses. In particular, the localization or systemic spread of the spike protein is strongly determined by the special structures and function of microcirculation of various organs.
To illustrate the above differences with examples, the heart’s microcirculation is characterized by an exceptionally dense capillary network providing nearly one-to-one capillary/cardiomyocyte ratio. This allows fast and efficient gas exchange and nutrient supply to the cardiac muscle fibers (
Figure 2A). The high-density capillary network in the myocardium, which is tightly connected to the epicardium, the inner layer of fluid-containing pericardium [
29,
30] provides the structural basis for the frequent vaccine-induced coincidence of pericarditis and myocarditis, a condition referred to as myopericarditis [
31,
32,
33].
Under such conditions the continuous capillaries with tight junctions undergo capillary remodeling, endothelial activation and fenestration (32), underlying the inflammatory symptoms.
In the brain, the ECs in continuous capillaries are connected by tight junctions, forming the blood–brain barrier (BBB), which strictly regulates molecular and cellular trafficking (
Figure 2B). This anatomical configuration is consistent with the emergence of localized inflammatory processes, underlying epileptic, cognitive and other focal AEs, for example optic neuritis, Guillain–Barré syndrome, facial paresis, etc. Rinaldi, 2022 #9042}. The localized, restrained inflammation also helps explain the functional neurological disorders which are characterized by neurological symptoms without detectable abnormalities upon imaging with MRI or CT [
34,
35,
36]. The situation is similar in the spinal cord, for example in transverse myelitis, which entails localized motor, sensory, and autonomic dysfunction.
In peripheral nerves, the endoneurium, which encases the individual nerve fibers (axons) along with their myelin sheath, contains an extensive network of capillaries also with tightly bound ECs (
Figure 2C). Here, too, the inflammatory signals spread locally, along the axons and the perineurial and epineurial tissue, leading to demyelinating neuritis of different afferent or efferent nerves.
In the lung, the microcirculation comprises an extensive and highly permeable capillary network that lies in close association with the alveolar epithelium (
Figure 2D). This anatomical arrangement facilitates the transmission of inflammatory signals to both type I squamous epithelial cells and type II secretory cells, leading to widespread inflammatory involvement of the respiratory tissue. Consequently, typical manifestations of vaccine-induced inflammatory adverse events in the lung include alveolar infiltrates accompanied by respiratory distress, dyspnea, chest tightness, and hypoxia [
37,
38].
In the kidney, the microcirculation consists of two distinct capillary networks: glomerular capillaries, which form high-pressure filtration units, and peritubular capillaries, which surround the renal tubules and facilitate selective reabsorption and urine concentration within the nephron (
Figure 2E). Inflammation of the ECs in both types of capillaries explain the nephrotic syndrome characteristic of vaccine injury, manifested in acute onset edema, hypoalbuminemia, and heavy proteinuria [
39,
40].
In the liver, the microcirculation is organized into sinusoidal capillaries with a discontinuous endothelium, allowing free distribution of inflammatory mediators among the blood and hepatocytes. Accordingly, the symptoms of vaccine-induced hepatitis reflect common hepatocyte dysfunction, manifested in acute cholestatic liver injury (biliary obstruction), abdominal pain, pruritus, fever, fatigue, anorexia, nausea and vomiting, laboratory deviations, occasionally fatal outcome in patients with preexisting liver disease [
41,
42,
43,
44,
45].
Given this diversity of AEs in different organ systems, the impact of endothelitis must be tissue-specific, likely responsive to targeted anti-inflammatory therapies.
6. The Journey of mRNA-LNPs from the Deltoid Muscle to the Sites of Inflammations
Intramuscular injection of mRNA–LNP-based vaccines is generally associated with immediate interaction with antigen-presenting cells (APCs) and other immune cells at the injection site and within the draining lymph nodes. These locations are considered the primary sites for T and B cell priming and have remained the central focus of most narratives concerning the immune mechanisms of mRNA-based vaccines [
46,
47,
48,
49]. However, it is well established that LNPs injected intramuscularly rapidly distribute throughout the body, reaching various non-immune organs and cell types, many of which can take up the nanoparticles.
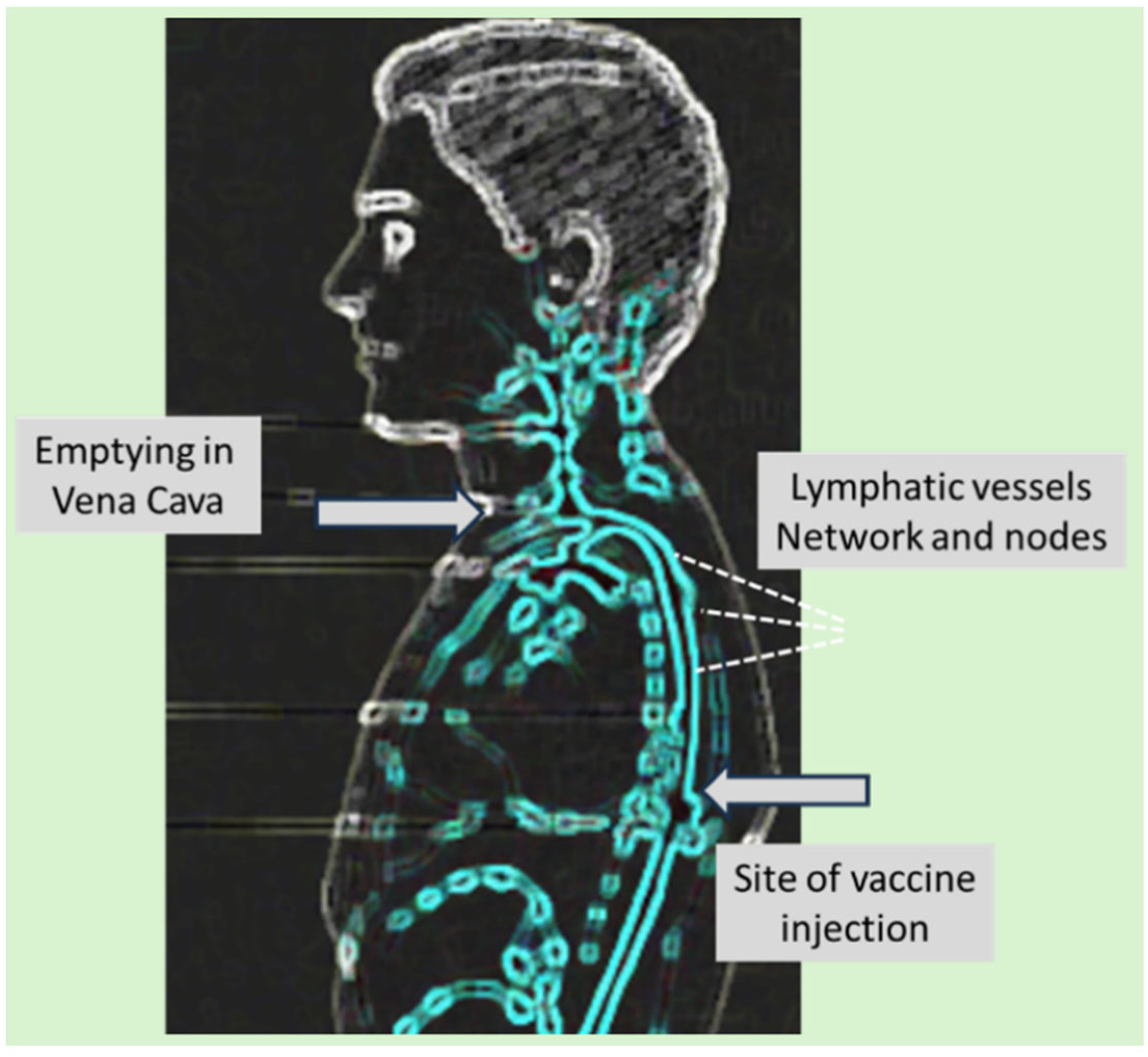
Three main mechanisms have been proposed to explain how mRNA-LNPs reach the systemic circulation. One is an accidental, direct injection into a small vessel. Because the standard immunization protocol does not require aspiration or needle retraction prior to injection [
50], inadvertent intravenous administration and fast entry of vaccine into the brachial vein may occur, particularly in regions with dense capillary networks, such as the deltoid muscle. Second, entry via the lymphatic system which may start through the blind-ended portions of lymphatic vessels, which are leaflet-like endothelial structures that open in response to increased interstitial pressure (Fig. 1A). The 300 µL bolus of vaccine can transiently and locally raise tissue pressure above the typically low pressure within lymphatic vessels, promoting nanoparticle entry. Once inside the lymphatic network, rhythmic contractions and relaxations of the lymphatic vessel walls propel the nanoparticles through valve-separated chambers and lymph nodes of lymphatic vessels into the venous blood [
51,
52,
53,
54].
High-speed video lymphoscintigraphic measurements showed the velocity of lymph flow in the ~2-9 mm/s range, which implies that the small portion (about 3 %) of Comirnaty nanoparticles that does not reside in the deltoid muscle and the draining lymph nodes, reaches the blood within about 1 hour [
55,
56]. As additional variables that contribute to the individual variation of vaccine pharmacokinetics, the pumping activity of lymph microvessels are delicately controlled by nitric oxide and prostaglandins released from the endothelium [
51].
Figure 3 details the anatomical roadmap of mRNA–LNPs transport from the deltoid muscle to the blood.
A third potential route for LNP entry into the bloodstream involves concentration gradient-driven transcapillary transport from the interstitial space into capillaries, particularly in regions where vascular permeability is elevated due to the proinflammatory effects of the vaccine. This process can be considered a reversal of the enhanced permeation and retention (EPR) effect, which is exploited for passive targeting of liposomes in cancer chemotherapy [
57]. As for the extent and kinetics of systemic biodistribution of mRNA-LNPs, Pfizer Australia's preclinical study reported that 2.8% of a radioactive lipid marker remained in the plasma of rats 15 minutes after intramuscular injection of Comirnaty-equivalent LNPs, with plasma levels peaking between 1 and 4 hours [
58].
7. Quantitative Aspects of Vaccine Nanoparticle-Endothelium Engagement
The vascular endothelium constitutes the first biological barrier to the systemic distribution of mRNA–LNPs, prompting the key question of stoichiometry: how many mRNA-LNPs actually reach and interact with the apical surface of each endothelial cell? A calculation at 15 minutes post-injection suggests that approximately 500 to 7,000 mRNA–LNP may potentially interact with each endothelial cell in the body. This implies that a single vaccine dose delivers mRNA–LNPs in substantial excess relative to the number of endothelial cells. The assumptions underlying this estimate are as follows: (i) each 30 µg dose of mRNA in a Comirnaty injection contains approximately 2 × 10⁻⁸ moles of mRNA, based on a molecular weight of ~1.4 MDa, corresponding to roughly 1.2 × 10¹³ mRNA molecules; (ii) approximately 3% of the injected mRNA–LNPs reach the bloodstream within 15 minutes [
58]; (iii) each LNP carries 1–2 mRNA molecules [
59,
60,
61,
62,
63]; (iv) the total endothelial surface area in the human body is 1–7 m², equivalent to 1–7 × 10¹² µm²; and (v) the apical surface area of a typical endothelial cell is 300–600 µm² [
64,
65].
It must be emphasized, however, that this is a snapshot calculation; the number of mRNA–LNPs available for interaction with endothelial cells rapidly declines due to uptake by various organs and depends critically on the rate of entry into the circulation. The key implication of these metrics is that vaccine-induced endothelial transfection, activation, and consequent inflammation could potentially affect any segment of the vascular endothelium. Hence, the small amount of vaccine inoculum does not preclude the possibility of off-target distribution of vaccine nanoparticles.
8. The Pathophysiology and Diagnosis of Vaccine-Induced Vasculitis
Table 3 shows the types and symptoms of vasculitis classified according to the size of the blood vessels affected. Based on the proportional relationship between LNP uptake and capillary surface area, most vasculitis symptoms are associated with inflammation of small vessels, which collectively provide the largest endothelial interface. Nevertheless, the inflammatory diseases involving tissue compartments enriched in medium- and large-sized blood vessels have also been documented to display distinct clinical features [
66,
67,
68,
69] (
Table 3).
The diagnosis of vasculitis-related illnesses on organ level remains challenging. The nonspecific symptoms, such as fever, fatigue, rash, may not pin down the organ affected just as the inflammatory markers (e.g., CRP) and autoimmune indicators (e.g., ANCA, ANA) in blood or cerebrospinal fluid. Imaging studies, including conventional or digital subtraction angiography, MRI with contrast and tissue biopsies support the diagnosis, but a thorough differential diagnosis is essential to exclude infections, malignancies, and thrombotic disorders or ischemic or hemorrhagic [
70]. Advanced techniques like vessel wall imaging (VWI) and magnetic resonance angiography (MRA) may reveal vessel abnormalities [
71].
Focusing on the role of the spike protein in post-vaccination cerebral arteritis, a recent study demonstrated its prolonged presence in cerebral arteries, accompanied by inflammatory cell infiltration [
72]. This chronic cerebral inflammation may disrupt local circulation and oxygen delivery, potentially underlying symptoms such as fatigue, memory loss, dementia, Alzheimer acceleration and other cognitive or psychological complaints following vaccination [
28,
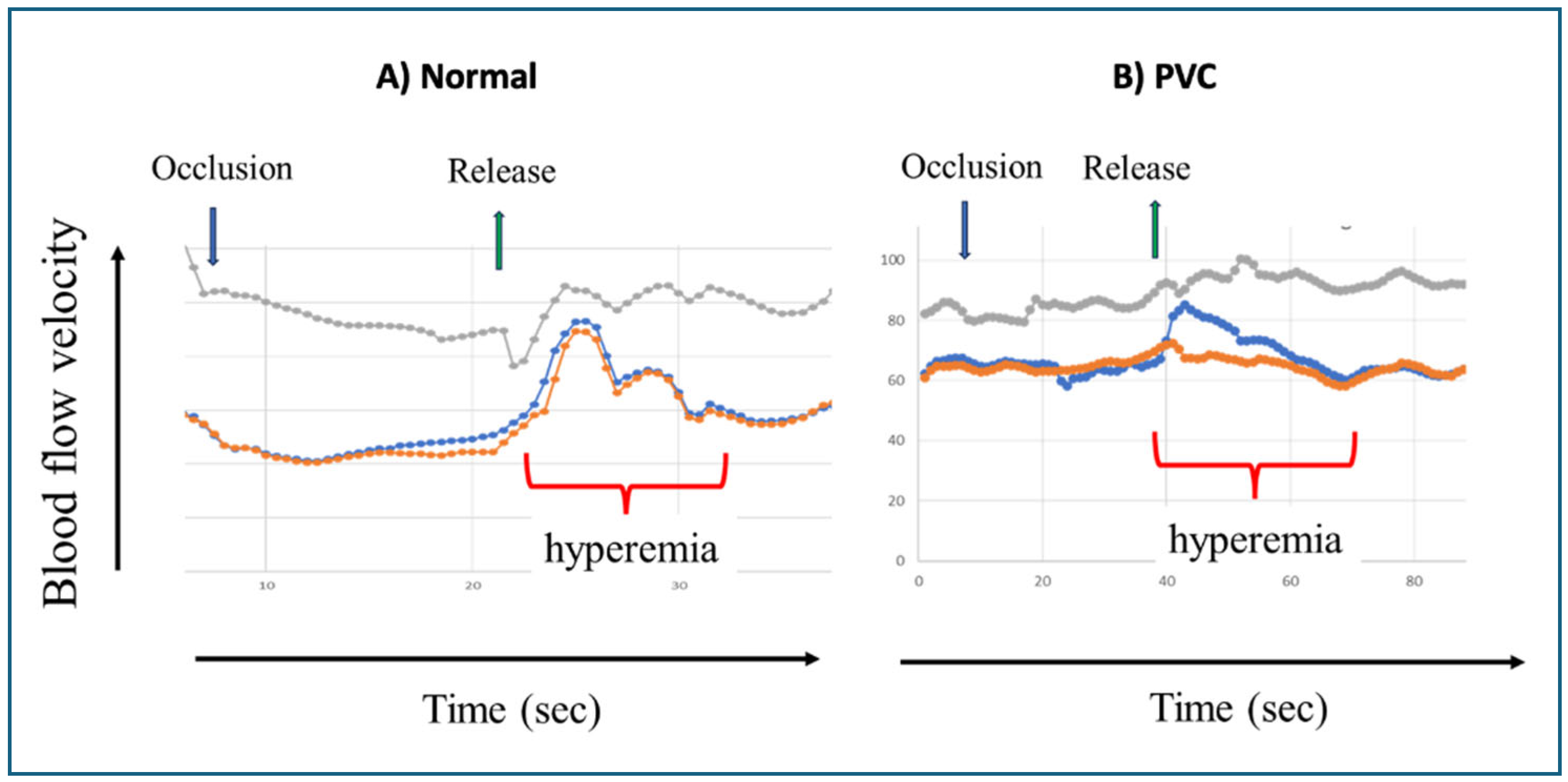
73]. Supporting this, we observed impaired cerebrovascular regulation in individuals with post-COVID condition (whether from infection or vaccination), assessed using transcranial Doppler ultrasound, a non-invasive method that measures blood flow velocity in major cerebral arteries.
As shown in
Figure 4, the attenuated reactive hyperemic response reflects diminished vasodilatory capacity of cerebral resistance vessels in the brain and was associated with persistent cognitive and mental dysfunction even after ~2 years. Notably, this impairment was alleviated by regular physical activity [
28].
9. Non-Target Organ Uptake of mRNA–LNPs
Beside the classic endocytic uptake of mRNA-LNPs by phagocytic cells in the body, a main mechanism is transfection via fusion. This function of LNPs has been known since the discovery in the late 80-s that the ionizable, positively charged lipids can tightly bind the negatively charged nucleic acids and carry them into the cytoplasm of cells without loss of gene function [
61,
74,
75,
76,
77,
78,
79,
80,
81]. Since then, many different tissues and cells have been shown to be “viable targets” for gene therapies using LNPs [
82], providing rationale for using the LNPs for the delivery of spike protein mRNA to immune cells in the vicinity of the injection; the widely claimed mode of action of mRNA vaccines.
In the background of the selection of the ionizable positively charged aminolipids (ALC-0315 in Comirnaty) as main LNP component in mRNA vaccines, the concept was based, among others, on its nucleic acid-binding capability with fusogenic activity resulting in unique competency for genetic modification of cells. This selection was in keeping with the clinical success of liver-targeted patisiran (Onpattro), the first FDA-approved gene therapy against amyloidosis [
79].
The assumption that Comirnaty delivers mRNA exclusively to antigen-presenting cells (APCs) near to the injection site may have overlooked a 2015 study by Pardi et al, which showed delivery by LNPs and translation of firefly luciferase-mRNA in cells at the injection site and its draining lymph nodes, as well as in the liver, lung and other organs within 5 hours [
83]. In Pfizer Australia's preclinical rat study, beyond the injection site and sentinel lymph nodes, the LNPs were primarily distributed to the liver, adrenal glands, spleen, and ovaries over a 48-hour period, and low-level (<2%) radioactive signals were also detected in 12 additional organs [
58].
In further large-animal studies, Ferraresso et al. [
82] demonstrated that LNPs transfected nearly all major organs in pigs. More recently, Dézsi et al. [
84] reported the presence of both spike protein mRNA and the translated spike protein in multiple organs as early as 6 hours following intravenous injection of Comirnaty. Notably, the uptake of spike protein mRNA showed a close temporal correlation with the transcriptional upregulation of key proinflammatory cytokine genes, including IL-6, TNF-α, and IL-1. These findings align with the known proinflammatory properties of both the mRNA and the spike protein, although it remains unclear whether one plays a dominant role in cytokine induction or whether they act synergistically.
The internalization of LNPs by different cells may occur through multiple pathways, including clathrin- or caveolae-mediated endocytosis, micropinocytosis -particularly in inflamed or activated endothelium- and/or membrane fusion, a hallmark of LNPs containing ionizable aminolipids. As for ECs, experimental evidence for effective LNP delivery of functional mRNA into these cells across various organs is well documented [
85,
86,
87,
88,
89,
90,
91,
92]. However, regardless of the entry mechanism of mRNA-LNPs into the ECs, the internalized mRNA and de novo synthesized spike protein trigger the activation of ECs via a variety of ways, direct and indirect, independent and cooperative, involving innate and adaptive immune responses. These are itemized below.
10. Adverse Impacts of mRNA-LNPs and the Spike Protein on Endothelial Cells
The spike protein functions as a pluripotent toxin [
93,
94,
95,
96,
97], causing oxidative damage of the mitochondria [
98,
99,
100], induction of proinflammatory cytokines [
101,
102] and other self-destructive effects in the cells that produce them.
Further immune-mediated damage to the mRNA-LNP transfected EC is related to the diversification of the intracellular antigen processing of free ribosome-derived spike protein [
9,
103]. Notably, after formation on free ribosomes, which exceeds the number of endoplasmic reticulum (ER)-bound ribosomes in most cells [
104], a part of the intact spike protein molecules in transfected cells undergoes proteasome digestion, which yields antigenic peptides to be transported to MHC Class I molecules on the endothelial cell surface [
103]. This process is known as cross-presentation [
105], since the spike protein degradation peptides are supposed to be presented on MHC-II molecules. [
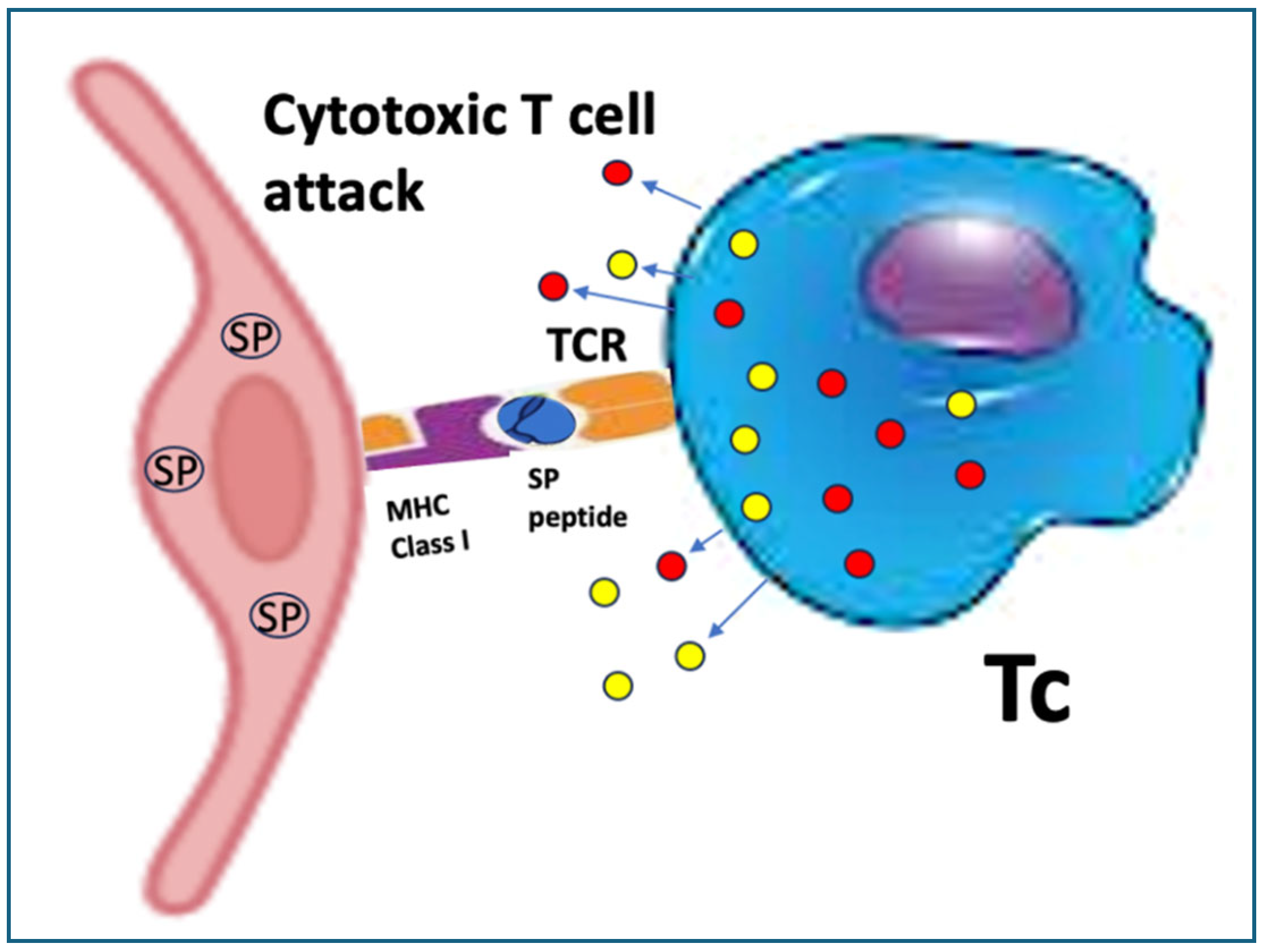
105],. In previously infected or immunized people, MHC Class I-presented peptides are recognized by specific cytotoxic T cells (Tc, CTLs) which attack the EC cells (
Figure 5). This mechanism plays a major role in the widespread autoimmune phenomena observed after mRNA-type vaccination and may help explain why booster injections can elicit more pronounced AEs than primary immunization.
In addition to proteasomal degradation, an alternative trafficking route for the translated spike protein directs it to the cell membrane, i.e., the same destination it naturally follows in SARS-CoV-2-infected cells. As a result, the luminal surface of ECs, along with the outer membrane of other transfected cells, becomes “crowned” with the spike protein, mimicking a true viral infection. The immune system recognizes these “pseudo-infected” cells and targets them through at least two antibody-dependent elimination mechanisms: complement (C) activation (
Figure 6) and antibody-dependent cellular cytotoxicity (ADCC) (
Figure 7).
Regarding C activation, Comirnaty is a strong initiator of this process [
106,
107,
108], a relatively ignored contributor to the AEs of mRNA vaccines [
63]. It came to the focus of attention in this subject because of its role in the acute anaphylactic reactions whose incidence had significantly increased after immunisation with mRNA vaccines [
109]. These investigations [
63,
106,
107,
108,
109] led to the concept that C activation-related pseudoallergy (CARPA) represents a significant contributor to vaccine-induced anaphylaxis [
63]. However, while CARPA is due to fluid phase activation of the proteolytic cascade, the C-mediated cytotoxicity proceeds on cell surfaces, including the ECs. C activation on cell surfaces that express the spike protein and or bind PEGylated LNP can also occur via both the classical and the alternative the pathways [
107]. The former process may be due to anti-PEG antibody binding to EC-adhered mRNA-LNPs, which express PEG on their surface (
Figure 6A). The spike protein is moving from free ribosomes to the cell surface, triggering also classical pathway C activation through the binding of anti-spike protein antibodies
Figure 6B). In addition, fluid phase and cell surface spike protein can induce lectin pathway activation [
96,
110] (
Figure 6C) and the damaged cell membrane can bind C3b directly, the core mechanism of alternative pathway activation (
Figure 6D). Each of these activations can be perpetuated via the alternative pathway amplification loop [
111,
112] fed by activated EC-produced C3, properdin and factor B.
The other antibody-mediated EC cell damage is called antibody-dependent cellular cytotoxicity (ADCC) whereupon effector cells of the immune system recognize and eliminate target cells that have been opsonized by specific antibodies. The cells involved in this action include natural killer (NK) cells, macrophages and neutrophils. The specific antibodies, usually IgG1 or IgG3, bind to antigens expressed on the surface of a target cell and these antibodies are recognized by
the Fcγ receptors (especially FcγRIIIa/CD16) on the above cells (
Figure 7). Binding of FcγR triggers degranulation of these effector cells, just as the binding of CTL to MHC Class I molecules triggering the release of
cytolytic mediators, namely p
erforins and granzymes.
The latter proteins, especially granzyme B, are serine proteases that activate caspases (e.g., caspase-3) that cause DNA fragmentation and apoptosis. Granzyme A induces caspase-independent cell death (via reactive oxygen species, mitochondrial damage). Both NK cells and CTLs use
receptor-ligand interactions leading to cell death, such as the binding of Fas Ligand (FasL, CD95L) to
Fas receptors (CD95) on target cells, activating caspase-8 and causing apoptosis [
113,
114]. The
TNF-related apoptosis-inducing ligand (TRAIL) is also involved in the apoptotic attack of Tc and/or NK on the ECs [
115]. Among the cytokines produced,
IFN-γ enhances macrophage activity and antigen presentation [
116].
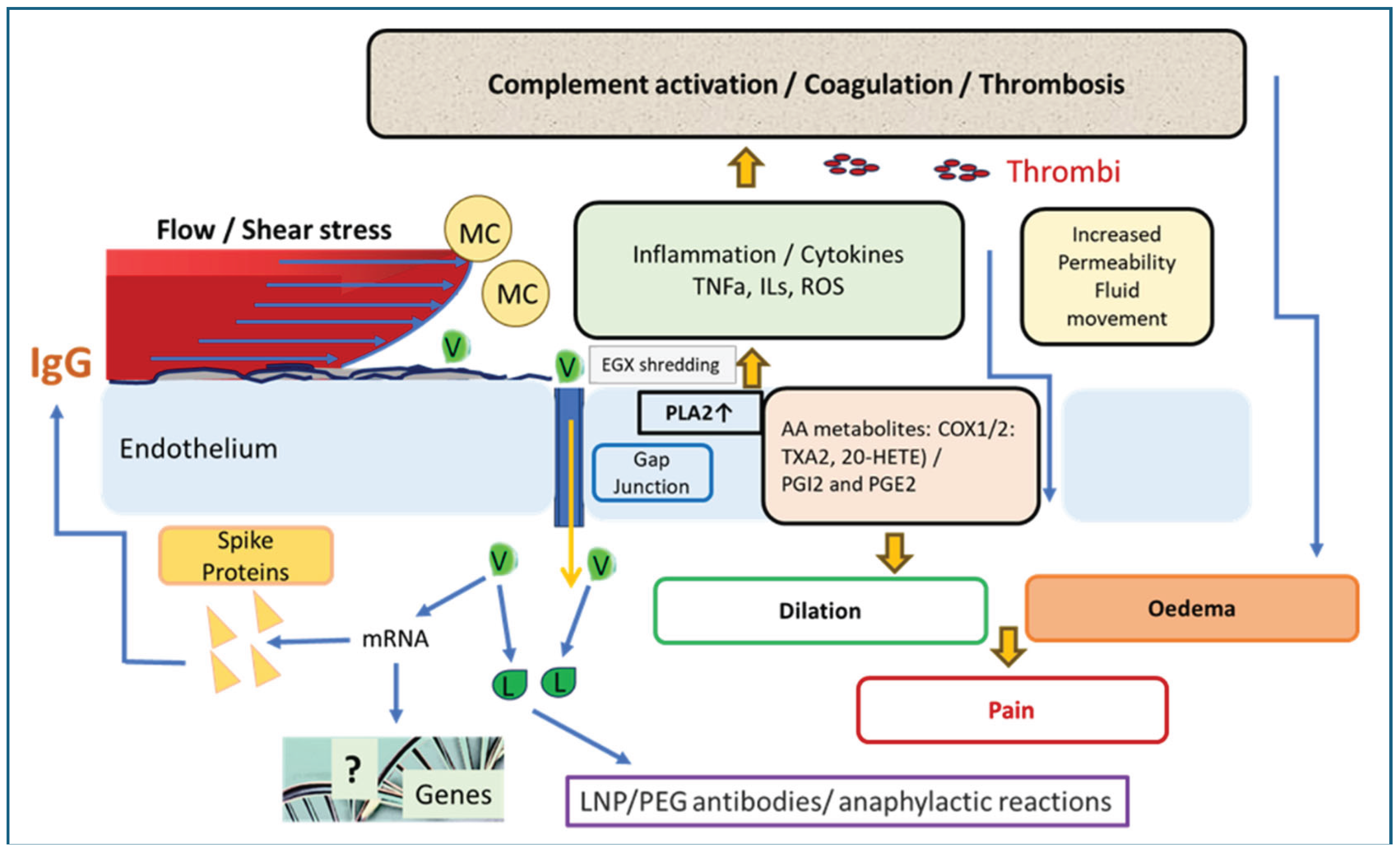
11. Causes and Features of Endothelitis
Figure 8 illustrates the process whereupon the encounter with vaccine nanoparticles entails the activation of ECs. In this process, the glycocalyx derangement enables the deposition of neutrophils, monocytes and macrophages, with the release of oxygen radicals, PLA2, cytokines, leukotrienes, C proteins and other inflammatory mediators. The pathophysiological manifestations of EC activation and damages include increased vascular permeability, opening the gap junctions, release of PLA2, leukotrienes, cytokines and reactive oxygen species, deposition of neutrophils, monocytes and macrophages. The opening of the gap junctions upon vasodilatation (which increases intravascular pressure) is associated with oedema and pain. The increased production of thromboxane A2 and reduced production of prostaglandins and nitric oxide create a pro-coagulant environment promoting thrombogenesis, which is frequently observed after mRNA-type vaccinations. Moreover, damaged ECs release DAMPs, which further activate the immune system and contribute to a cycle of self-perpetuating chronic condition. Endothelial microparticles (exosomes) and anti-endothelial cell antibodies have been identified as diagnostic or prognostic biomarkers of endothelitis.
12. Outlook
Since the introduction and widespread deployment of mRNA-based COVID-19 vaccines, their safety profile has become a focal point of both public discourse and scientific investigation. This cutting-edge technology has raised several unresolved questions, particularly regarding the unusually broad spectrum and relatively high incidence of AEs associated with its use [
1]. According to an evolving paradigm shift, beyond their intended antiviral efficacy, COVID-19 mRNA vaccines may cause collateral tissue damage due to inherent design limitations, as elaborated in
Section 2 of this review on mRNA–LNP-induced inflammatory complications. In essence, the unique properties of the spike protein antigen and its delivery vehicle, the mRNA-LNP lipoplex, can provoke immune responses resembling those of active infection. Thus, the vaccine nanoparticles act as non-replicating, functionally simplified mimics of SARS-CoV-2 infection and, as such, exhibit features akin to live attenuated viruses with potential for reverted pathogenicity [
1,
9].
Although -fortunately-, the AEs affect only a small subset of vaccine recipients, safety concerns have begun to influence vaccination policies. Most recently, the CDC and FDA ceased recommending COVID-19 mRNA vaccination for children and pregnant women, restricting its use primarily to high-risk populations [
117,
118]. At this time, the attention is being centered on the accumulating evidence of AEs and their public health implications, while the biological roots of the health problem is far from being understood. Among scientific investigations into vaccine-related AEs, there is growing recognition that a common pathogenic denominator is vascular inflammation, specifically, endothelitis. This review aims to advance understanding of the problem by focusing on tissue-specific features of vaccine-transfected microcirculation, elucidating the mechanisms of mRNA-LNP intravasation and cellular uptake, and examining the self-injurious consequences of spike protein expression. While acknowledging the public health achievements and benefits of mRNA-LNP-based vaccines, the insights gained from studying their AEs will be instrumental in improving the safety of future vaccines and therapeutics using this platform. A key unresolved question for future research is: what protective mechanisms shield the vast majority of vaccine recipients from these complications?
Funding
The financial supports by the European Union Horizon 2020 project 825828 (Expert) and 2022-1.2.5-TÉT-IPARI-KR-2022-00009 (JS) and Project no. TKP2020-NKA-17 and TKP2021-EGA-37 by the Ministry of Innovation and Technology of Hungary from the National Research, Development and Innovation Fund, and MTA/HAS-Post-Covid 2021-34 and National Research, Development, and Innovation Office of Hungary (OTKA132596, K-19 (AK) are acknowledged.
References
- Szebeni, Janos. "Expanded Spectrum and Increased Incidence of Adverse Events Linked to Covid-19 Genetic Vaccines: New Concepts on Prophylactic Immuno-Gene Therapy, Iatrogenic Orphan Disease, and Platform-Inherent Challenges." Pharmaceutics 17, no. 4 (2025): 450.
- Krumholz, H. M., Y. Wu, M. Sawano, R. Shah, T. Zhou, A. S. Arun, P. Khosla, S. Kaleem, A. Vashist, B. Bhattacharjee, Q. Ding, Y. Lu, C. Caraballo, F. Warner, C. Huang, J. Herrin, D. Putrino, D. Hertz, B. Dressen, and A. Iwasaki. "Post-Vaccination Syndrome: A Descriptive Analysis of Reported Symptoms and Patient Experiences after Covid-19 Immunization." medRxiv posted , 2023. 10 November. [CrossRef]
- Palmer, M., S. Bhakdi, B. Hooker, M. Holland, M. DesBois, D. Rasnick, and C.A. Fitts. Mrna Vvaccine Toxicity, D4ce.Org. https://d4ce.org/mRNA-vaccine-toxicity/, 2023.
- Costa, C.; Moniati, F.; Li, X. The Epidemiology of COVID-19 Vaccine-Induced Myocarditis. Adv. Med. 2024, 2024, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Novak, N.; Tordesillas, L.; Cabanillas, B. Adverse rare events to vaccines for COVID-19: From hypersensitivity reactions to thrombosis and thrombocytopenia. Int. Rev. Immunol. 2021, 41, 438–447. [Google Scholar] [CrossRef] [PubMed]
- Oueijan, R.I.; Hill, O.R.; Ahiawodzi, P.D.; Fasinu, P.S.; Thompson, D.K. Rare Heterogeneous Adverse Events Associated with mRNA-Based COVID-19 Vaccines: A Systematic Review. Medicines 2022, 9, 43. [Google Scholar] [CrossRef] [PubMed]
- Padilla-Flores, T.; Sampieri, A.; Vaca, L. Incidence and management of the main serious adverse events reported after COVID-19 vaccination. Pharmacol. Res. Perspect. 2024, 12, e1224. [Google Scholar] [CrossRef]
- Worldwide Safety. "Cumulative Analysis of Post-Authorization Adverse Event Reports of Pf-07302048 (Bnt162b2) Received through 28-Feb-2021." https://phmpt.org/wp-content/uploads/2021/11/5.3.6-postmarketing-experience.pdf?fbclid=IwAR2tWI7DKw0cc2lj8 (2021).
- Szebeni, János. "The Unique Features and Collateral Immune Effects of Mrna-Based Covid-19 Vaccines: Potential Plausible Causes of Adverse Events and Complications." In Preprints: Preprints, 2025.
- Karikó, K.; Buckstein, M.; Ni, H.; Weissman, D. Suppression of RNA Recognition by Toll-like Receptors: The Impact of Nucleoside Modification and the Evolutionary Origin of RNA. Immunity 2005, 23, 165–175. [Google Scholar] [CrossRef]
- Karikó, K.; Muramatsu, H.; Welsh, F.A.; Ludwig, J.; Kato, H.; Akira, S.; Weissman, D. Incorporation of Pseudouridine Into mRNA Yields Superior Nonimmunogenic Vector With Increased Translational Capacity and Biological Stability. Mol. Ther. 2008, 16, 1833–1840. [Google Scholar] [CrossRef]
- Anderson, B.R.; Muramatsu, H.; Nallagatla, S.R.; Bevilacqua, P.C.; Sansing, L.H.; Weissman, D.; Karikó, K. Incorporation of pseudouridine into mRNA enhances translation by diminishing PKR activation. Nucleic Acids Res. 2010, 38, 5884–5892. [Google Scholar] [CrossRef]
- Anderson, B.R.; Muramatsu, H.; Jha, B.K.; Silverman, R.H.; Weissman, D.; Karikó, K. Nucleoside modifications in RNA limit activation of 2'-5'-oligoadenylate synthetase and increase resistance to cleavage by RNase L. Nucleic Acids Res. Nucleic Acids Res. 2011, 39, 9329–9338. [Google Scholar] [CrossRef]
- Karikó, K.; Muramatsu, H.; Ludwig, J.; Weissman, D. Generating the optimal mRNA for therapy: HPLC purification eliminates immune activation and improves translation of nucleoside-modified, protein-encoding mRNA. Nucleic Acids Res. 2011, 39, e142–e142. [Google Scholar] [CrossRef]
- Rehwinkel, J.; Gack, M.U. RIG-I-like receptors: their regulation and roles in RNA sensing. Nat. Rev. Immunol. 2020, 20, 537–551. [Google Scholar] [CrossRef]
- Lv, Y.; Chang, Y. Cytomegalovirus Proctitis Developed after COVID-19 Vaccine: A Case Report and Literature Review. Vaccines 2022, 10, 1417. [Google Scholar] [CrossRef] [PubMed]
- Chakravorty, S.; Cochrane, A.B.; Psotka, M.A.; Regmi, A.; Marinak, L.N.; Thatcher, A.N.; Shlobin, O.A.; Brown, A.W.; King, C.S.M.; Ahmad, K.; et al. CMV Infection Following mRNA SARS-CoV-2 Vaccination in Solid Organ Transplant Recipients. Transplant. Direct 2022, 8, e1344. [Google Scholar] [CrossRef] [PubMed]
- Herzum, A.; Trave, I.; D’aGostino, F.; Burlando, M.; Cozzani, E.; Parodi, A. Epstein-Barr virus reactivation after COVID-19 vaccination in a young immunocompetent man: a case report. Clin. Exp. Vaccine Res. 2022, 11, 222–225. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Bielsa, A.; Gracia-Cazaña, T.; Aldea-Manrique, B.; Abadías-Granado, I.; Ballano, A.; Bernad, I.; Gilaberte, Y. COVID-19 infection and vaccines: potential triggers of Herpesviridae reactivation. An. Bras. de Dermatol. 2023, 98, 347–354. [Google Scholar] [CrossRef]
- Shafiee, A.; Amini, M.J.; Bahri, R.A.; Jafarabady, K.; Salehi, S.A.; Hajishah, H.; Mozhgani, S.-H. Herpesviruses reactivation following COVID-19 vaccination: a systematic review and meta-analysis. Eur. J. Med Res. 2023, 28, 1–15. [Google Scholar] [CrossRef]
- Maple, P. A. C. "Covid-19, Sars-Cov-2 Vaccination, and Human Herpesviruses Infections." Vaccines (Basel) 11, no. 2 (2023).
- Martinez-Reviejo, R.; Tejada, S.; Adebanjo, G.A.; Chello, C.; Machado, M.C.; Parisella, F.R.; Campins, M.; Tammaro, A.; Rello, J. Varicella-Zoster virus reactivation following severe acute respiratory syndrome coronavirus 2 vaccination or infection: New insights. Eur. J. Intern. Med. 2022, 104, 73–79. [Google Scholar] [CrossRef]
- Elbaz, M.; Hoffman, T.; Yahav, D.; Dovrat, S.; Ghanem-Zoubi, N.; Atamna, A.; Grupel, D.; Reisfeld, S.; Hershman-Sarafov, M.; Ciobotaro, P.; et al. Varicella-Zoster Virus–Induced Neurologic Disease After COVID-19 Vaccination: A Multicenter Observational Cohort Study. Open Forum Infect. Dis. 2024, 11, ofae287. [Google Scholar] [CrossRef]
- Attwell, D., A. Mishra, C. N. Hall, F. M. O'Farrell, and T. Dalkara. "What Is a Pericyte?" J Cereb Blood Flow Metab 36, no. 2 (2016): 451-5.
- Allport, J.R.; Ding, H.; Collins, T.; Gerritsen, M.E.; Luscinskas, F.W. Endothelial-dependent Mechanisms Regulate Leukocyte Transmigration: A Process Involving the Proteasome and Disruption of the Vascular Endothelial–Cadherin Complex at Endothelial Cell-to-Cell Junctions. J. Exp. Med. 1997, 186, 517–527. [Google Scholar] [CrossRef]
- Ishiko, S.; Koller, A.; Deng, W.; Huang, A.; Sun, D. Liposomal nanocarriers of preassembled glycocalyx restore normal venular permeability and shear stress sensitivity in sepsis: assessed quantitatively with a novel microchamber system. Am. J. Physiol. Circ. Physiol. 2024, 327, H390–H398. [Google Scholar] [CrossRef]
- Gerritsen, M.E.; Bloor, C.M. Endothelial cell gene expression in response to injury. FASEB J. 1993, 7, 523–532. [Google Scholar] [CrossRef]
- Takács, J.; Deák, D.; Koller, A. Higher level of physical activity reduces mental and neurological symptoms during and two years after COVID-19 infection in young women. Sci. Rep. 2024, 14, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, Z.; Cziraki, A.; Szabados, S.; Biri, B.; Keki, S.; Koller, A.; Gaetano, C. Elevated Levels of Asymmetric Dimethylarginine (ADMA) in the Pericardial Fluid of Cardiac Patients Correlate with Cardiac Hypertrophy. PLOS ONE 2015, 10, e0135498. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, Z.; Cziraki, A.; Szabados, S.; Horvath, I.; Koller, A. Pericardial fluid of cardiac patients elicits arterial constriction: role of endothelin-1. Can. J. Physiol. Pharmacol. 2015, 93, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Gill, J.R.; Tashjian, R.; Duncanson, E. Autopsy Histopathologic Cardiac Findings in 2 Adolescents Following the Second COVID-19 Vaccine Dose. Arch. Pathol. Lab. Med. 2022, 146, 925–929. [Google Scholar] [CrossRef]
- Verma, A. K., K. J. Lavine, and C. Y. Lin. "Myocarditis after Covid-19 Mrna Vaccination." N Engl J Med 385, no. 14 (2021): 1332-34.
- Rodriguez, E.R.; Tan, C.D. Structure and Anatomy of the Human Pericardium. Prog. Cardiovasc. Dis. 2017, 59, 327–340. [Google Scholar] [CrossRef]
- Rinaldi, V.; Bellucci, G.; Buscarinu, M.C.; Reniè, R.; Marrone, A.; Nasello, M.; Zancan, V.; Nistri, R.; Palumbo, R.; Salerno, A.; et al. CNS inflammatory demyelinating events after COVID-19 vaccines: A case series and systematic review. Front. Neurol. 2022, 13, 1018785. [Google Scholar] [CrossRef]
- Patone, M.; Handunnetthi, L.; Saatci, D.; Pan, J.; Katikireddi, S.V.; Razvi, S.; Hunt, D.; Mei, X.W.; Dixon, S.; Zaccardi, F.; et al. Neurological complications after first dose of COVID-19 vaccines and SARS-CoV-2 infection. Nat. Med. 2021, 27, 2144–2153. [Google Scholar] [CrossRef]
- Khayat-Khoei, M.; Bhattacharyya, S.; Katz, J.; Harrison, D.; Tauhid, S.; Bruso, P.; Houtchens, M.K.; Edwards, K.R.; Bakshi, R. COVID-19 mRNA vaccination leading to CNS inflammation: a case series. J. Neurol. 2021, 269, 1093–1106. [Google Scholar] [CrossRef]
- Yoshikawa, T., K. Tomomatsu, E. Okazaki, T. Takeuchi, Y. Horio, Y. Kondo, T. Oguma, and K. Asano. "Covid-19 Vaccine-Associated Organizing Pneumonia." Respirol Case Rep 10, no. 5 (2022): e0944.
- Park, J.Y.; Kim, J.-H.; Park, S.; Hwang, Y.I.; Kim, H.I.; Jang, S.H.; Jung, K.-S.; Kim, Y.K.; Kim, H.A.; Lee, I.J. Clinical characteristics of patients with COVID-19 vaccine-related pneumonitis: a case series and literature review. Korean J. Intern. Med. 2022, 37, 989–1001. [Google Scholar] [CrossRef]
- Kervella, D.; Jacquemont, L.; Chapelet-Debout, A.; Deltombe, C.; Ville, S. Minimal change disease relapse following SARS-CoV-2 mRNA vaccine. Kidney Int. 2021, 100, 457–458. [Google Scholar] [CrossRef]
- Zhang, J.; Cao, J.; Ye, Q. Renal Side Effects of COVID-19 Vaccination. Vaccines 2022, 10, 1783. [Google Scholar] [CrossRef] [PubMed]
- Rocco, A.; Sgamato, C.; Compare, D.; Nardone, G. Autoimmune hepatitis following SARS-CoV-2 vaccine: May not be a casuality. J. Hepatol. 2021, 75, 728–729. [Google Scholar] [CrossRef] [PubMed]
- Schinas, G.; Polyzou, E.; Dimakopoulou, V.; Tsoupra, S.; Gogos, C.; Akinosoglou, K. Immune-mediated liver injury following COVID-19 vaccination. World J. Virol. 2023, 12, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Sergi, C.M. COVID-19 vaccination-related autoimmune hepatitis—a perspective. Front. Pharmacol. 2023, 14, 1190367. [Google Scholar] [CrossRef]
- Chen, C.; Xie, D.; Xiao, J. Real-world evidence of autoimmune hepatitis following COVID-19 vaccination: A population-based pharmacovigilance analysis. Front. Pharmacol. 2023, 14, 1100617. [Google Scholar] [CrossRef]
- Kim, J. H., H. B. Chae, S. Woo, M. S. Song, H. J. Kim, and C. G. Woo. "Clinicopathological Characteristics of Autoimmune-Like Hepatitis Induced by Covid-19 Mrna Vaccine (Pfizer-Biontech, Bnt162b2): A Case Report and Literature Review." Int J Surg Pathol 31, no. 6 (2023): 1156-62.
- Liang, F.; Lindgren, G.; Lin, A.; Thompson, E.A.; Ols, S.; Röhss, J.; John, S.; Hassett, K.; Yuzhakov, O.; Bahl, K.; et al. Efficient Targeting and Activation of Antigen-Presenting Cells In Vivo after Modified mRNA Vaccine Administration in Rhesus Macaques. Mol. Ther. 2017, 25, 2635–2647. [Google Scholar] [CrossRef]
- Bettini, E.; Locci, M. SARS-CoV-2 mRNA Vaccines: Immunological Mechanism and Beyond. Vaccines 2021, 9, 147. [Google Scholar] [CrossRef]
- Szabó, G.T.; Mahiny, A.J.; Vlatkovic, I. COVID-19 mRNA vaccines: Platforms and current developments. Mol. Ther. 2022, 30, 1850–1868. [Google Scholar] [CrossRef]
- Buckley, M.; Araínga, M.; Maiorino, L.; Pires, I.S.; Kim, B.; Michaels, K.K.; Dye, J.; Qureshi, K.; Zhang, Y.J.; Mak, H.; et al. Visualizing lipid nanoparticle trafficking for mRNA vaccine delivery in non-human primates. Mol. Ther. 2025, 33, 1105–1117. [Google Scholar] [CrossRef]
- Ndeupen, S.; Qin, Z.; Jacobsen, S.; Bouteau, A.; Estanbouli, H.; Igyártó, B.Z. The mRNA-LNP platform's lipid nanoparticle component used in preclinical vaccine studies is highly inflammatory. iScience 2021, 24, 103479. [Google Scholar] [CrossRef]
- Mizuno, R.; Koller, A.; Kaley, G. Regulation of the vasomotor activity of lymph microvessels by nitric oxide and prostaglandins. Am. J. Physiol. Integr. Comp. Physiol. 1998, 274, R790–R796. [Google Scholar] [CrossRef] [PubMed]
- Koller, A.; Mizuno, R.; Kaley, G. Flow reduces the amplitude and increases the frequency of lymphatic vasomotion: role of endothelial prostanoids. Am. J. Physiol. Integr. Comp. Physiol. 1999, 277, R1683–R1689. [Google Scholar] [CrossRef] [PubMed]
- Breslin, J.W. Mechanical forces and lymphatic transport. Microvasc. Res. 2014, 96, 46–54. [Google Scholar] [CrossRef] [PubMed]
- von der Weid, P. Y. "Lymphatic Vessel Pumping." Adv Exp Med Biol 1124 (2019): 357-77.
- Alazraki, N.; Glass, E.C.; Castronovo, F.; Olmos, R.A.V.; Podoloff, D. ; Society of Nuclear Medicine Procedure guideline for lymphoscintigraphy and the use of intraoperative gamma probe for sentinel lymph node localization in melanoma of intermediate thickness 1. 0.. 2002, 43, 1414–8. [Google Scholar]
- Dixon, J.B.; Greiner, S.T.; Gashev, A.A.; Cote, G.L.; Moore, J.E., Jr.; Zawieja, D.C. Lymph Flow, Shear Stress, and Lymphocyte Velocity in Rat Mesenteric Prenodal Lymphatics. Microcirculation 2006, 13, 597–610. [Google Scholar] [CrossRef]
- Borresen, B., A. E. Hansen, F. P. Fliedner, J. R. Henriksen, D. R. Elema, M. Brandt-Larsen, L. K. Kristensen, A. T. Kristensen, T. L. Andresen, and A. Kjaer. "Noninvasive Molecular Imaging of the Enhanced Permeability and Retention Effect by (64)Cu-Liposomes: In Vivo Correlations with (68)Ga-Rgd, Fluid Pressure, Diffusivity and (18)F-Fdg." Int J Nanomedicine 15 (2020): 8571-81.
- Ltd, Pfizer Australia Pty. "Nonclinical Evaluation Report: Bnt162b2 [Mrna] Covid-19 Vaccine (Comirnatytm)." https://www.tga.gov.au/sites/default/files/foi-2389-06.pdf https://t.co/Zrhakh7Xgv (2021).
- Pardi, N., M. J. Hogan, F. W. Porter, and D. Weissman. "Mrna Vaccines - a New Era in Vaccinology." Nat Rev Drug Discov 17, no. 4 (2018): 261-79.
- Sabnis, S.; Kumarasinghe, E.S.; Salerno, T.; Mihai, C.; Ketova, T.; Senn, J.J.; Lynn, A.; Bulychev, A.; McFadyen, I.; Chan, J.; et al. A Novel Amino Lipid Series for mRNA Delivery: Improved Endosomal Escape and Sustained Pharmacology and Safety in Non-human Primates. Mol. Ther. 2018, 26, 1509–1519. [Google Scholar] [CrossRef]
- Hou, X., T. Zaks, R. Langer, and Y. Dong. "Lipid Nanoparticles for Mrna Delivery." Nat Rev Mater 6, no. 12 (2021): 1078-94.
- Li, S.; Hu, Y.; Li, A.; Lin, J.; Hsieh, K.; Schneiderman, Z.; Zhang, P.; Zhu, Y.; Qiu, C.; Kokkoli, E.; et al. Payload distribution and capacity of mRNA lipid nanoparticles. Nat. Commun. 2022, 13, 1–13. [Google Scholar] [CrossRef]
- Szebeni, J.; Storm, G.; Ljubimova, J.Y.; Castells, M.; Phillips, E.J.; Turjeman, K.; Barenholz, Y.; Crommelin, D.J.A.; Dobrovolskaia, M.A. Applying lessons learned from nanomedicines to understand rare hypersensitivity reactions to mRNA-based SARS-CoV-2 vaccines. Nat. Nanotechnol. 2022, 17, 337–346. [Google Scholar] [CrossRef]
- Jaffe, E. A. "Cell Biology of Endothelial Cells." Hum Pathol 18, no. 3 (1987): 234-9.
- Aird, W. C. "Phenotypic Heterogeneity of the Endothelium: I. Structure, Function, and Mechanisms." Circ Res 100, no. 2 (2007): 158-73.
- Gilio, M. , and G. De Stefano. "Large-Vessel Vasculitis Following the Pfizer-Biontech Covid-19 Vaccine." Intern Emerg Med 17, no. 4 (2022): 1239-41.
- Bostan, E., F. Zaid, N. Akdogan, and O. Gokoz. "Possible Case of Mrna Covid-19 Vaccine-Induced Small-Vessel Vasculitis." J Cosmet Dermatol 21, no. 1 (2022): 51-53.
- Abuhammad, A.; Albandak, M.; Ayyad, M.; Refayeh, E.; Qawasma, B.; Hour, S.; Abu Thraiee, Y.; A Sowaity, Z.; Dukmak, O.; Jobran, A.W.M.; et al. COVID-19 vaccine-associated vasculitis: A systematic review. SAGE Open Med. 2024, 12. [Google Scholar] [CrossRef]
- Abdelmaksoud, A., U. Wollina, S. A. Temiz, and A. Hasan. "Sars-Cov-2 Vaccination-Induced Cutaneous Vasculitis: Report of Two New Cases and Literature Review." Dermatol Ther 35, no. 6 (2022): e15458.
- Rice, C.M.; Scolding, N.J. The diagnosis of primary central nervous system vasculitis. Pr. Neurol. 2019, 20, 109–114. [Google Scholar] [CrossRef]
- Gupta, N., S. B. Hiremath, R. I. Aviv, and N. Wilson. "Childhood Cerebral Vasculitis : A Multidisciplinary Approach." Clin Neuroradiol 33, no. 1 (2023): 5-20.
- Ota, N.; Itani, M.; Aoki, T.; Sakurai, A.; Fujisawa, T.; Okada, Y.; Noda, K.; Arakawa, Y.; Tokuda, S.; Tanikawa, R. Expression of SARS-CoV-2 spike protein in cerebral Arteries: Implications for hemorrhagic stroke Post-mRNA vaccination. . 2025, 136, 111223. [Google Scholar] [CrossRef] [PubMed]
- Takács, J.; Deák, D.; Seregély, B.; Koller, A. Cognitive Slowing, Dysfunction in Verbal Working Memory, Divided Attention and Response Inhibition in Post COVID-19 Condition in Young Adults. Life 2025, 15, 821. [Google Scholar] [CrossRef] [PubMed]
- Felgner, P.L.; Gadek, T.R.; Holm, M.; Roman, R.; Chan, H.W.; Wenz, M.; Northrop, J.P.; Ringold, G.M.; Danielsen, M. Lipofection: a highly efficient, lipid-mediated DNA-transfection procedure. Proc. Natl. Acad. Sci. 1987, 84, 7413–7417. [Google Scholar] [CrossRef] [PubMed]
- Horejs, C. From lipids to lipid nanoparticles to mRNA vaccines. Nat. Rev. Mater. 2021, 6, 1075–1076. [Google Scholar] [CrossRef]
- Holland, John W., Thomas D. Madden, and Pieter R. Cullis. "Bilayer Stabilizing Components and Their Use in Forming Programmable Fusogenic Liposomes.". U.S. Patent and Trademark Office, 1999.
- Cullis, P.R.; Hope, M.J. Lipid Nanoparticle Systems for Enabling Gene Therapies. Mol. Ther. 2017, 25, 1467–1475. [Google Scholar] [CrossRef]
- Kulkarni, J.A.; Darjuan, M.M.; Mercer, J.E.; Chen, S.; van der Meel, R.; Thewalt, J.L.; Tam, Y.Y.C.; Cullis, P.R. On the Formation and Morphology of Lipid Nanoparticles Containing Ionizable Cationic Lipids and siRNA. ACS Nano 2018, 12, 4787–4795. [Google Scholar] [CrossRef]
- Akinc, A.; Maier, M.A.; Manoharan, M.; Fitzgerald, K.; Jayaraman, M.; Barros, S.; Ansell, S.; Du, X.; Hope, M.J.; Madden, T.D.; et al. The Onpattro story and the clinical translation of nanomedicines containing nucleic acid-based drugs. Nat. Nanotechnol. 2019, 14, 1084–1087. [Google Scholar] [CrossRef]
- Pateev, I.; Seregina, K.; Ivanov, R.; Reshetnikov, V. Biodistribution of RNA Vaccines and of Their Products: Evidence from Human and Animal Studies. Biomedicines 2023, 12, 59. [Google Scholar] [CrossRef]
- Dalby, B., S. Cates, A. Harris, E. C. Ohki, M. L. Tilkins, P. J. Price, and V. C. Ciccarone. "Advanced Transfection with Lipofectamine 2000 Reagent: Primary Neurons, Sirna, and High-Throughput Applications." Methods 33, no. 2 (2004): 95-103.
- Ferraresso, F.; Badior, K.; Seadler, M.; Zhang, Y.; Wietrzny, A.; Cau, M.F.; Haugen, A.; Rodriguez, G.G.; Dyer, M.R.; Cullis, P.R.; et al. Protein is expressed in all major organs after intravenous infusion of mRNA-lipid nanoparticles in swine. Mol. Ther. 2024, 32, 101314. [Google Scholar] [CrossRef]
- Pardi, N.; Tuyishime, S.; Muramatsu, H.; Kariko, K.; Mui, B.L.; Tam, Y.K.; Madden, T.D.; Hope, M.J.; Weissman, D. Expression kinetics of nucleoside-modified mRNA delivered in lipid nanoparticles to mice by various routes. J. Control. Release 2015, 217, 345–351. [Google Scholar] [CrossRef]
- Dézsi, L., G. Kökény, G. Szénási, Cs. Révész, T. Mészáros, A.B. Barta, R. Facsko, A. Szilasi, T. Bakos, G.T. Kozma, A. Dobos, B., B. Merkely, T. Radovits, and J. Szebeni. "Acute Anaphylactic and Multiorgan Inflammatory Effects of Comirnaty in Pigs: Evidence of Spike Protein mRNA Transfection and Paralleling Inflammatory Cytokine Upregulation." BioRxiv (2025). https://www.biorxiv.org/cgi/content/short/2025.06.07. 6583. [Google Scholar]
- Sago, C.D.; Lokugamage, M.P.; Paunovska, K.; Vanover, D.A.; Monaco, C.M.; Shah, N.N.; Castro, M.G.; Anderson, S.E.; Rudoltz, T.G.; Lando, G.N.; et al. High-throughput in vivo screen of functional mRNA delivery identifies nanoparticles for endothelial cell gene editing. Proc. Natl. Acad. Sci. 2018, 115, E9944–E9952. [Google Scholar] [CrossRef] [PubMed]
- Francia, V.; Schiffelers, R.M.; Cullis, P.R.; Witzigmann, D. The Biomolecular Corona of Lipid Nanoparticles for Gene Therapy. Bioconjugate Chem. 2020, 31, 2046–2059. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.H.Y.; Leung, J.; Zhang, Y.; Strong, C.; Basha, G.; Momeni, A.; Chen, Y.; Jan, E.; Abdolahzadeh, A.; Wang, X.; et al. Induction of Bleb Structures in Lipid Nanoparticle Formulations of mRNA Leads to Improved Transfection Potency. Adv. Mater. 2023, 35, e2303370. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.W.; Guzman, E.B.; Menon, N.; Langer, R.S. Lipid Nanoparticles for Nucleic Acid Delivery to Endothelial Cells. Pharm. Res. 2023, 40, 3–25. [Google Scholar] [CrossRef]
- Cullis, P.R.; Felgner, P.L. The 60-year evolution of lipid nanoparticles for nucleic acid delivery. Nat. Rev. Drug Discov. 2024, 23, 709–722. [Google Scholar] [CrossRef]
- Yazdi, M.; Pöhmerer, J.; Kafshgari, M.H.; Seidl, J.; Grau, M.; Höhn, M.; Vetter, V.; Hoch, C.C.; Wollenberg, B.; Multhoff, G.; et al. In Vivo Endothelial Cell Gene Silencing by siRNA-LNPs Tuned with Lipoamino Bundle Chemical and Ligand Targeting. Small 2024, 20, e2400643. [Google Scholar] [CrossRef]
- Petersen, D. M. S., R. M. Weiss, K. A. Hajj, S. S. Yerneni, N. Chaudhary, A. N. Newby, M. L. Arral, and K. A. Whitehead. "Branched-Tail Lipid Nanoparticles for Intravenous Mrna Delivery to Lung Immune, Endothelial, and Alveolar Cells in Mice." Adv Healthc Mater 13, no. 22 (2024): e2400225.
- Papp, T.E.; Zeng, J.; Shahnawaz, H.; Akyianu, A.; Breda, L.; Yadegari, A.; Steward, J.; Shi, R.; Li, Q.; Mui, B.L.; et al. CD47 Peptide-Cloaked Lipid Nanoparticles Promote Cell-Specific mRNA Delivery. Mol. Ther. 2025. [Google Scholar] [CrossRef]
- Robles, J. P., M. Zamora, E. Adan-Castro, L. Siqueiros-Marquez, G. Martinez de la Escalera, and C. Clapp. "The Spike Protein of Sars-Cov-2 Induces Endothelial Inflammation through Integrin Alpha5beta1 and Nf-Kappab Signaling." J Biol Chem 298, no. 3 (2022): 101695.
- Yonker, L.M.; Swank, Z.; Bartsch, Y.C.; Burns, M.D.; Kane, A.; Boribong, B.P.; Davis, J.P.; Loiselle, M.; Novak, T.; Senussi, Y.; et al. Circulating Spike Protein Detected in Post–COVID-19 mRNA Vaccine Myocarditis. Circulation 2023, 147, 867–876. [Google Scholar] [CrossRef]
- Avolio, E.; Carrabba, M.; Milligan, R.; Williamson, M.K.; Beltrami, A.P.; Gupta, K.; Elvers, K.T.; Gamez, M.; Foster, R.R.; Gillespie, K.; et al. The SARS-CoV-2 Spike protein disrupts human cardiac pericytes function through CD147 receptor-mediated signalling: a potential non-infective mechanism of COVID-19 microvascular disease. Clin. Sci. 2021, 135, 2667–2689. [Google Scholar] [CrossRef]
- Perico, L.; Morigi, M.; Galbusera, M.; Pezzotta, A.; Gastoldi, S.; Imberti, B.; Perna, A.; Ruggenenti, P.; Donadelli, R.; Benigni, A.; et al. SARS-CoV-2 Spike Protein 1 Activates Microvascular Endothelial Cells and Complement System Leading to Platelet Aggregation. Front. Immunol. 2022, 13, 827146. [Google Scholar] [CrossRef]
- Perico, L.; Morigi, M.; Pezzotta, A.; Locatelli, M.; Imberti, B.; Corna, D.; Cerullo, D.; Benigni, A.; Remuzzi, G. SARS-CoV-2 spike protein induces lung endothelial cell dysfunction and thrombo-inflammation depending on the C3a/C3a receptor signalling. Sci. Rep. 2023, 13, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Van Huynh, T.; Rethi, L.; Lee, T.-W.; Higa, S.; Kao, Y.-H.; Chen, Y.-J. Spike Protein Impairs Mitochondrial Function in Human Cardiomyocytes: Mechanisms Underlying Cardiac Injury in COVID-19. Cells 2023, 12, 877. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, L.; Aparicio-Alonso, M.; Henry, M.; Radman, M.; Attal, R.; Bakkar, A. Toxicity of the spike protein of COVID-19 is a redox shift phenomenon: A novel therapeutic approach. Free. Radic. Biol. Med. 2023, 206, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Sahin, U.; Karikó, K.; Türeci, Ö. mRNA-based therapeutics—Developing a new class of drugs. Nat. Rev. Drug Discov. 2014, 13, 759–780. [Google Scholar] [CrossRef]
- Forsyth, C.B.; Zhang, L.; Bhushan, A.; Swanson, B.; Zhang, L.; Mamede, J.I.; Voigt, R.M.; Shaikh, M.; Engen, P.A.; Keshavarzian, A. The SARS-CoV-2 S1 Spike Protein Promotes MAPK and NF-kB Activation in Human Lung Cells and Inflammatory Cytokine Production in Human Lung and Intestinal Epithelial Cells. Microorganisms 2022, 10, 1996. [Google Scholar] [CrossRef]
- Niu, C.; Liang, T.; Chen, Y.; Zhu, S.; Zhou, L.; Chen, N.; Qian, L.; Wang, Y.; Li, M.; Zhou, X.; et al. SARS-CoV-2 spike protein induces the cytokine release syndrome by stimulating T cells to produce more IL-2. Front. Immunol. 2024, 15, 1444643. [Google Scholar] [CrossRef]
- Pishesha, N.; Harmand, T.J.; Ploegh, H.L. A guide to antigen processing and presentation. Nat. Rev. Immunol. 2022, 22, 751–764. [Google Scholar] [CrossRef]
- Palade, G.E. A SMALL PARTICULATE COMPONENT OF THE CYTOPLASM. J. Cell Biol. 1955, 1, 59–68. [Google Scholar] [CrossRef]
- Embgenbroich, M.; Burgdorf, S. Current Concepts of Antigen Cross-Presentation. Front. Immunol. 2018, 9, 1643. [Google Scholar] [CrossRef]
- Dézsi, L.; Mészáros, T.; Kozma, G.; H-Velkei, M.; Oláh, C.Z.; Patkó, Z.; Fülöp, T.; Hennies, M.; Szebeni, M.; Barta, B.A.; et al. A naturally hypersensitive porcine model may help understand the mechanism of COVID-19 mRNA vaccine-induced rare (pseudo) allergic reactions: complement activation as a possible contributing factor. GeroScience 2022, 44, 597–618. [Google Scholar] [CrossRef]
- Bakos, T., T. Meszaros, G. T. Kozma, P. Berenyi, R. Facsko, H. Farkas, L. Dezsi, C. Heirman, S. de Koker, R. Schiffelers, K. A. Glatter, T. Radovits, G. Szenasi, and J. Szebeni. "mRNA-LNP Covid-19 Vaccine Lipids Induce Complement Activation and Production of Proinflammatory Cytokines: Mechanisms, Effects of Complement Inhibitors, and Relevance to Adverse Reactions." Int J Mol Sci 25, no. 7 (2024).
- Barta, B.A.; Radovits, T.; Dobos, A.B.; Kozma, G.T.; Mészáros, T.; Berényi, P.; Facskó, R.; Fülöp, T.; Merkely, B.; Szebeni, J. Comirnaty-induced cardiopulmonary distress and other symptoms of complement-mediated pseudo-anaphylaxis in a hyperimmune pig model: Causal role of anti-PEG antibodies. Vaccine: X 2024, 19, 100497. [Google Scholar] [CrossRef] [PubMed]
- Kozma, G.T.; Mészáros, T.; Berényi, P.; Facskó, R.; Patkó, Z.; Oláh, C.Z.; Nagy, A.; Fülöp, T.G.; Glatter, K.A.; Radovits, T.; et al. Role of anti-polyethylene glycol (PEG) antibodies in the allergic reactions to PEG-containing Covid-19 vaccines: Evidence for immunogenicity of PEG. Vaccine 2023, 41, 4561–4570. [Google Scholar] [CrossRef] [PubMed]
- Ali, Y. M., M. Ferrari, N. J. Lynch, S. Yaseen, T. Dudler, S. Gragerov, G. Demopulos, J. L. Heeney, and W. J. Schwaeble. "Lectin Pathway Mediates Complement Activation by Sars-Cov-2 Proteins." Front Immunol 12 (2021): 714511.
- Gibson, B.G.; Cox, T.E.; Marchbank, K.J. Contribution of animal models to the mechanistic understanding of Alternative Pathway and Amplification Loop (AP/AL)-driven Complement-mediated Diseases. Immunol. Rev. 2022, 313, 194–216. [Google Scholar] [CrossRef]
- Brouwer, N.; Dolman, K.M.; van Zwieten, R.; Nieuwenhuys, E.; Hart, M.; Aarden, L.A.; Roos, D.; Kuijpers, T.W. Mannan-binding lectin (MBL)-mediated opsonization is enhanced by the alternative pathway amplification loop. Mol. Immunol. 2006, 43, 2051–2060. [Google Scholar] [CrossRef] [PubMed]
- Risso, V.; Lafont, E.; Le Gallo, M. Therapeutic approaches targeting CD95L/CD95 signaling in cancer and autoimmune diseases. Cell Death Dis. 2022, 13, 1–32. [Google Scholar] [CrossRef]
- Ramirez-Labrada, A., C. Pesini, L. Santiago, S. Hidalgo, A. Calvo-Perez, C. Onate, A. Andres-Tovar, M. Garzon-Tituana, I. Uranga-Murillo, M. A. Arias, E. M. Galvez, and J. Pardo. "All About (Nk Cell-Mediated) Death in Two Acts and an Unexpected Encore: Initiation, Execution and Activation of Adaptive Immunity." Front Immunol 13 (2022): 896228.
- Wiley, S.R.; Schooley, K.; Smolak, P.J.; Din, W.S.; Huang, C.-P.; Nicholl, J.K.; Sutherland, G.R.; Smith, T.D.; Rauch, C.; Smith, C.A.; et al. Identification and characterization of a new member of the TNF family that induces apoptosis. Immunity 1995, 3, 673–682. [Google Scholar] [CrossRef]
- Schroder, K., P. J. Hertzog, T. Ravasi, and D. A. Hume. "Interferon-Gamma: An Overview of Signals, Mechanisms and Functions." J Leukoc Biol 75, no. 2 (2004): 163-89.
- Nathan-Kazis, J. , and Follow. "Kennedy’s Edict Results in New, Narrowed Vaccine Guidance from Cdc." https://www.barrons.com/articles/vaccines-cdc-covid-19-pregnant-women-children-kennedy-6f19ab5f?reflink=desktopwebshare_permalink (2025).
- FDA. "Notification for Safety Labeling Change. , 2025." https://www.fda.gov/media/186581/download?attachment, 2025. 17 April.
Figure 1.
Schematic representation of some features of the microcirculation. A, The arterioles and venules are connected by a network of capillaries. Some of the precapillary arterioles have sphincter-like smooth muscle structures, whereas the capillaries have pericytes to control their diameter and thus blood flow through the microcirculatory network [
24]. In the interstitial space dead-end lymphatic capillaries are anchored to the underlying tissue by filaments. B, Endothelial cells composed of a plasma membrane with surface receptors, a cytoskeleton that maintains shape and enables signal transduction, cell junctions for barrier function, and organelles like the nucleus, mitochondria, and endoplasmic reticulum to support metabolism, protein synthesis, and regulatory functions. The glycocalyx is a thin, gel-like layer of glycoproteins and glycolipids that coats the surface of ECs, playing a crucial role in vascular permeability, mechanotransduction, and protection against shear stress and inflammation. C, Cross-section of a capillary, illustrating the spatial arrangement of structural components such as the ECs, basal lamina, pericytes, and tight junction. D, The three types of capillaries specialized for different organ systems; schematic illustration (top); typical electron microscopic images (bottom). From left to right: continuous capillaries with tight junctions (TJ), fenestrated ones with pores (F), and discontinuous capillaries with large gaps (G) between the cells. The illustrations are modifications of publicly available sources, including Google images.
Figure 1.
Schematic representation of some features of the microcirculation. A, The arterioles and venules are connected by a network of capillaries. Some of the precapillary arterioles have sphincter-like smooth muscle structures, whereas the capillaries have pericytes to control their diameter and thus blood flow through the microcirculatory network [
24]. In the interstitial space dead-end lymphatic capillaries are anchored to the underlying tissue by filaments. B, Endothelial cells composed of a plasma membrane with surface receptors, a cytoskeleton that maintains shape and enables signal transduction, cell junctions for barrier function, and organelles like the nucleus, mitochondria, and endoplasmic reticulum to support metabolism, protein synthesis, and regulatory functions. The glycocalyx is a thin, gel-like layer of glycoproteins and glycolipids that coats the surface of ECs, playing a crucial role in vascular permeability, mechanotransduction, and protection against shear stress and inflammation. C, Cross-section of a capillary, illustrating the spatial arrangement of structural components such as the ECs, basal lamina, pericytes, and tight junction. D, The three types of capillaries specialized for different organ systems; schematic illustration (top); typical electron microscopic images (bottom). From left to right: continuous capillaries with tight junctions (TJ), fenestrated ones with pores (F), and discontinuous capillaries with large gaps (G) between the cells. The illustrations are modifications of publicly available sources, including Google images.
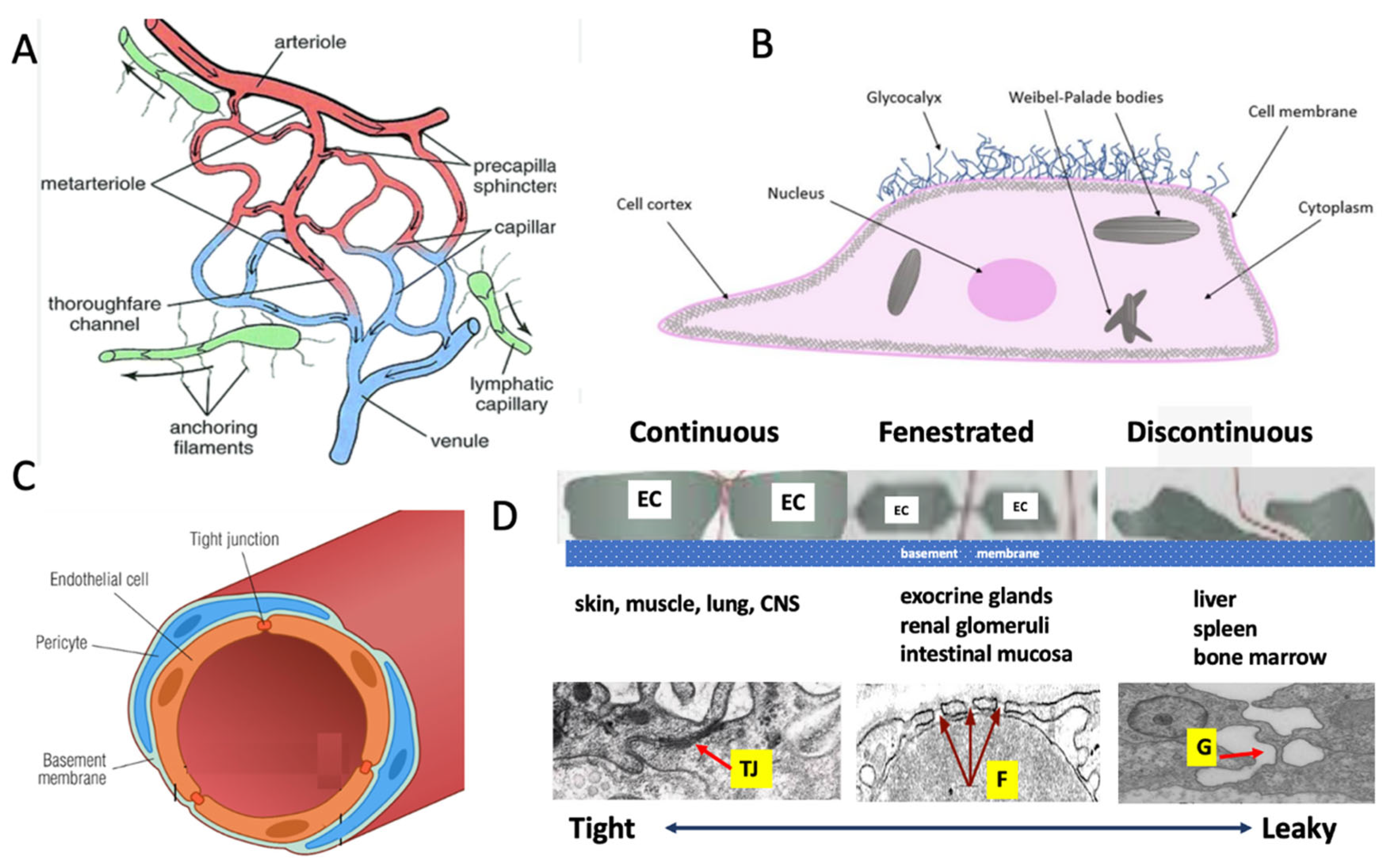
Figure 2.
Special features of the microcirculation in major organs of the human body. A, heart; B, brain; C, nerves; D, lung; E, kidney; F, liver). The illustrations are modifications of publicly available sources, including Google images.
Figure 2.
Special features of the microcirculation in major organs of the human body. A, heart; B, brain; C, nerves; D, lung; E, kidney; F, liver). The illustrations are modifications of publicly available sources, including Google images.
Figure 3.
Sagittal view illustrating the cranioventral transport pathway of mRNA-LNPs from the deltoid muscle to the bloodstream via the lymphatic system. Following i.m. injection into the deltoid muscle, the mRNA-LNPs enter the local interstitial space and are taken up by lymphatic capillaries. These merge into a network of superficial and deep lymphatic vessels whose rhythmic contractions propel the nanoparticles cranioventrally, toward the deltopectoral, lateral, central, and apical axillary lymph nodes. On the left side, this trunk drains into the thoracic duct, which empties at the left venous angle (angle of Pirogoff) at the junction of the left subclavian and internal jugular veins); on the right, drainage occurs via the right lymphatic duct into the right venous angle.
Figure 3.
Sagittal view illustrating the cranioventral transport pathway of mRNA-LNPs from the deltoid muscle to the bloodstream via the lymphatic system. Following i.m. injection into the deltoid muscle, the mRNA-LNPs enter the local interstitial space and are taken up by lymphatic capillaries. These merge into a network of superficial and deep lymphatic vessels whose rhythmic contractions propel the nanoparticles cranioventrally, toward the deltopectoral, lateral, central, and apical axillary lymph nodes. On the left side, this trunk drains into the thoracic duct, which empties at the left venous angle (angle of Pirogoff) at the junction of the left subclavian and internal jugular veins); on the right, drainage occurs via the right lymphatic duct into the right venous angle.
Figure 4.
Changes in blood flow velocity in the middle cerebral arteries following 10 sec manual carotid artery compression in a healthy, uninfected (Left) and a post-Covid-19 individual (Right). Blue arrow: occlusion, green arrow: Release. Red: reactive hyperemia, Gray: blood pressure (unpublished data).
Figure 4.
Changes in blood flow velocity in the middle cerebral arteries following 10 sec manual carotid artery compression in a healthy, uninfected (Left) and a post-Covid-19 individual (Right). Blue arrow: occlusion, green arrow: Release. Red: reactive hyperemia, Gray: blood pressure (unpublished data).
Figure 5.
Potential autoimmune attack against ECs by spike protein-specific cytotoxic T cells with increasing impact after booster vaccinations. The scheme applies to all body cells transfected with the vaccine and expressing spike protein fragments on their surface Class-I molecules.
Figure 5.
Potential autoimmune attack against ECs by spike protein-specific cytotoxic T cells with increasing impact after booster vaccinations. The scheme applies to all body cells transfected with the vaccine and expressing spike protein fragments on their surface Class-I molecules.
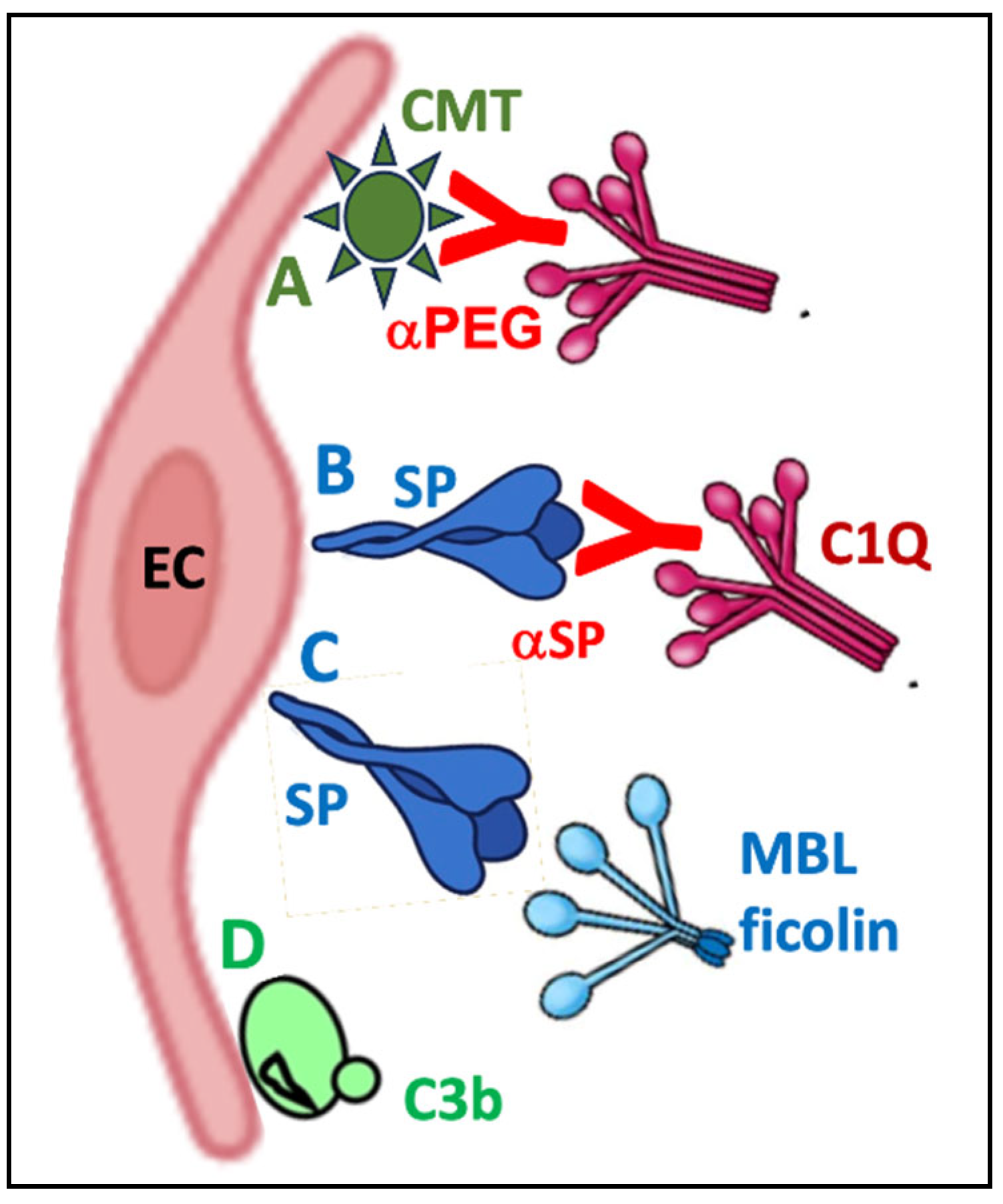
Figure 6.
Potential pathways of complement activation on the surfaces of endothelial cells. A) The polyethylene glycol (PEG) on Comirnaty (CMT) binds anti-PEG antibodies (αPEGs) which initiate C activation via the classical pathway; B) The spike protein expressed on the cell surface binds anti- spike protein antibodies and initiate C activation also via the classical pathway; C) The spike protein expressed on the cell surface binds mannose binding lectin (MBL) or ficolin, and triggers lectin pathway C activation; D) Direct deposition of C3 on membrane surfaces represents standard alternative pathway activation.
Figure 6.
Potential pathways of complement activation on the surfaces of endothelial cells. A) The polyethylene glycol (PEG) on Comirnaty (CMT) binds anti-PEG antibodies (αPEGs) which initiate C activation via the classical pathway; B) The spike protein expressed on the cell surface binds anti- spike protein antibodies and initiate C activation also via the classical pathway; C) The spike protein expressed on the cell surface binds mannose binding lectin (MBL) or ficolin, and triggers lectin pathway C activation; D) Direct deposition of C3 on membrane surfaces represents standard alternative pathway activation.
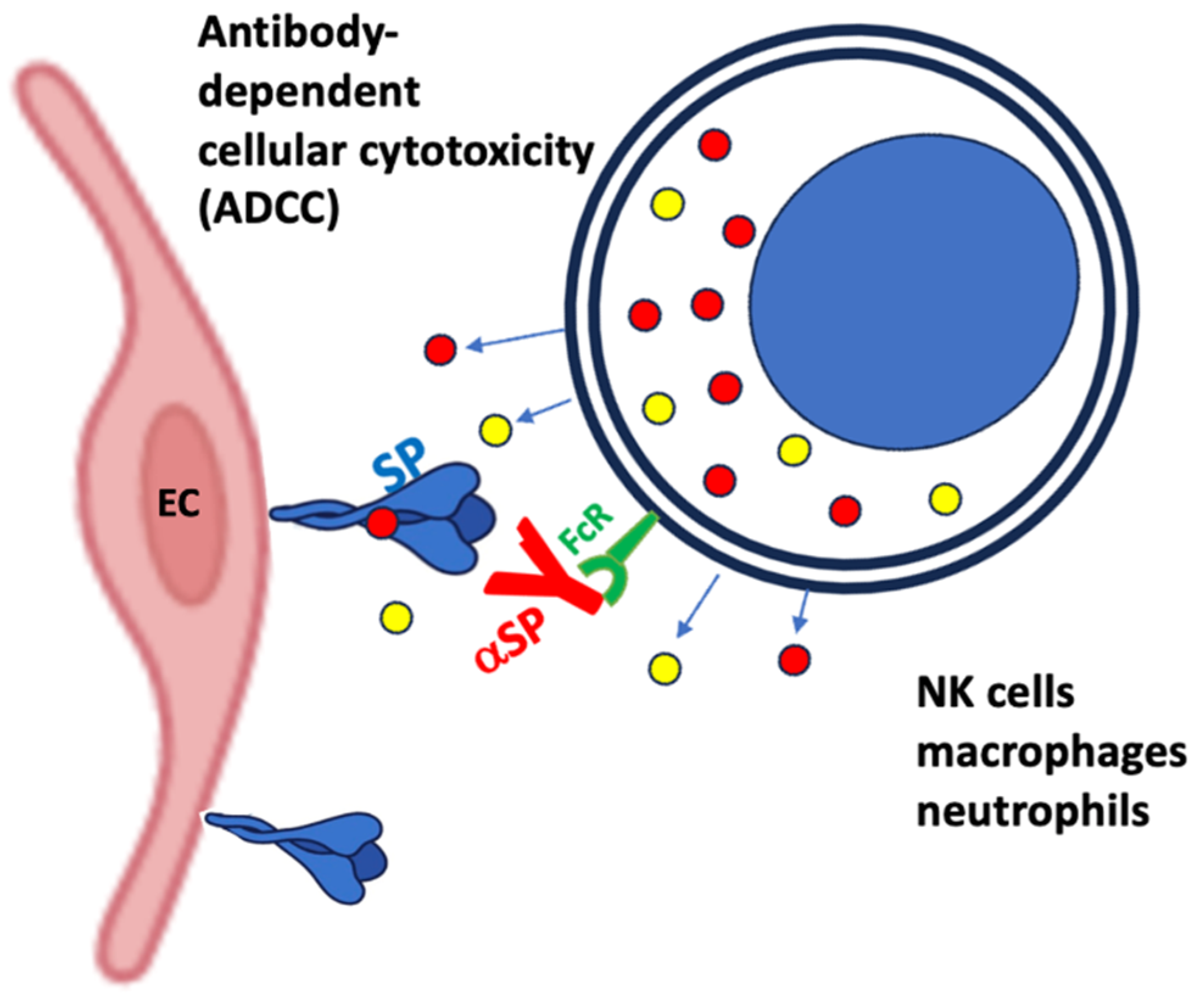
Figure 7.
Potential antibody-dependent autoimmune attacks against endothelial cells (ECs) via antibody-mediated cellular cytotoxicity. Comirnaty; αPEG, anti-PEG antibodies; SP, spike protein; αSP, anti-SP antibody; FcR, Fcγ-receptor; MBL, mannose binding lectin.
Figure 7.
Potential antibody-dependent autoimmune attacks against endothelial cells (ECs) via antibody-mediated cellular cytotoxicity. Comirnaty; αPEG, anti-PEG antibodies; SP, spike protein; αSP, anti-SP antibody; FcR, Fcγ-receptor; MBL, mannose binding lectin.
Figure 8.
Potential consequences of vaccine-induced inflammation at the interface of blood and ECs. The figure illustrates the combination of molecular and cellular changes associated with mRNA-LNP transfection of endothelial cells.
Figure 8.
Potential consequences of vaccine-induced inflammation at the interface of blood and ECs. The figure illustrates the combination of molecular and cellular changes associated with mRNA-LNP transfection of endothelial cells.
Table 1.
Inflammatory complications associated with immunization with mRNA-LNP vaccines.
Table 1.
Inflammatory complications associated with immunization with mRNA-LNP vaccines.
Table 2.
Virus strains whose reactivations may be associated with inflammatory AEs of mRNA vaccines.
Table 2.
Virus strains whose reactivations may be associated with inflammatory AEs of mRNA vaccines.
| Strains |
References |
| Cytomegalovirus (CMV) |
[16,17] |
| Epstein-Barr virus (EBV) |
[18] |
| Herpes simplex virus (HSV) |
[19,20,21] |
| Varicella Zoster virus (VZV), |
[22,23] |
Table 3.
Vasculitis types and symptoms classified according to the size of the blood vessels affected.
Table 3.
Vasculitis types and symptoms classified according to the size of the blood vessels affected.
| Vessel Size |
Types of Vasculitis |
Typical Symptoms |
| Small |
Microscopic polyangiitis (MPA) Granulomatosis with polyangiitis (Wegener’s) Eosinophilic granulomatosis with polyangiitis (Churg-Strauss) IgA vasculitis (Henoch-Schönlein purpura) Cryoglobulinemic vasculitis Hypocomplementemic urticarial vasculitis (anti-C1q vasculitis) Cutaneous leukocytoclastic vasculitis Leukocytoclastic vasculitis ANCA*-associated vasculitis |
Vasodilation Increased vascular permeability Leukocyte adhesion, margination and transmigration Endothelial activation and dysfunction Microthrombus formation Pain tissue hypoxia/ischemia lactate accumulation, acidosis |
| Medium |
|
Skin ulcers Abdominal pain Mononeuritis multiplex Hypertension |
| Large |
|
Headache Visual disturbances Jaw claudication Limb claudication Elevated ESR/CRP |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).