Submitted:
13 May 2025
Posted:
13 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Lateral Hemisection of the Spinal Cord
2.3. Spinal Cord Transection
2.4. Histological Analysis of SCI
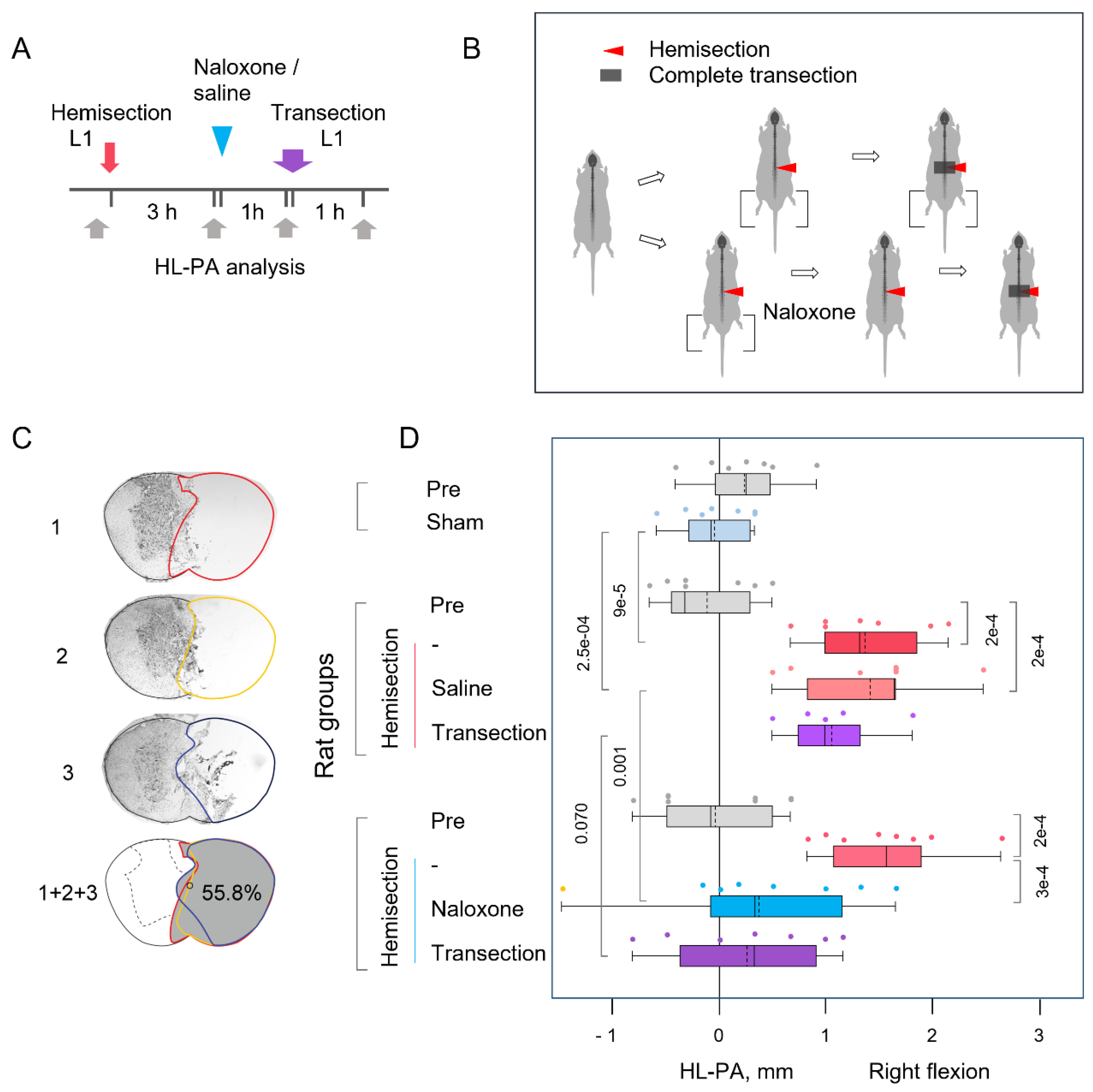
2.5. Analysis of Hindlimb Postural Asymmetry (HL-PA)
2.6. Statistical Analysis
2.7. Declaration of Generative AI and AI-Assisted Technologies in the Writing Process
3. Results
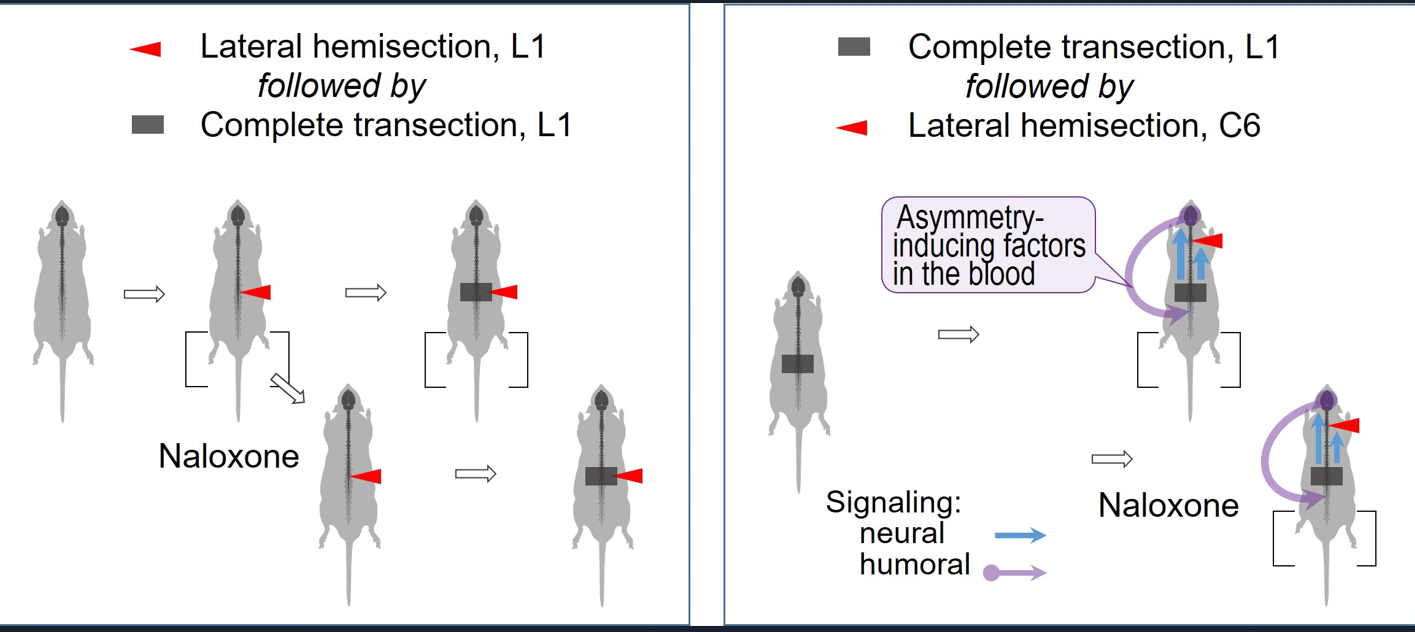
3.1. The Lumbar LHS-Induced HL-PA
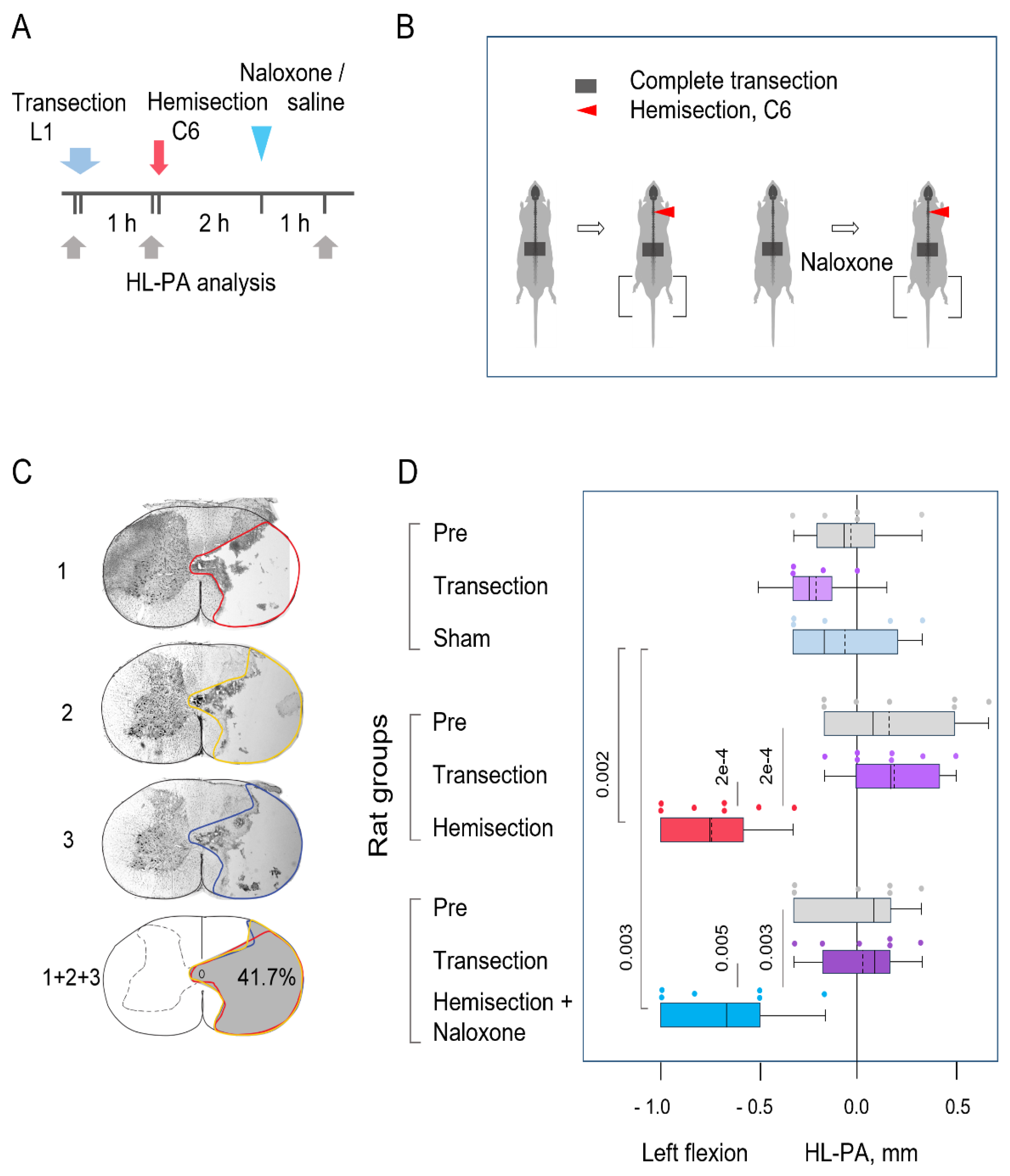
3.2. The Cervical LHS-Induced HL-PA in Rats with Complete Transection of Lumbar Spinal Cords
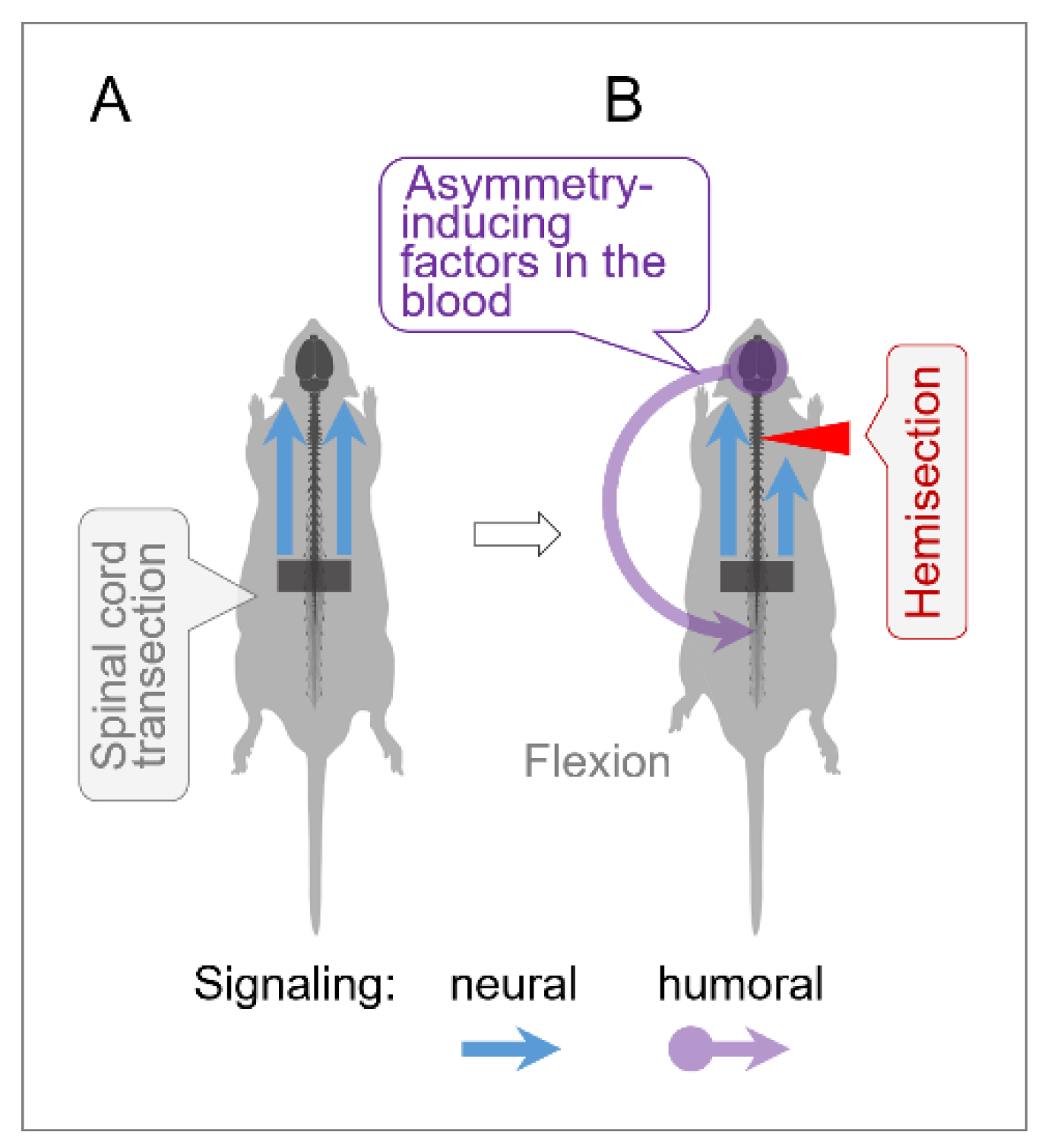
4. Discussion
4.1. LHS-Induced HL-PA
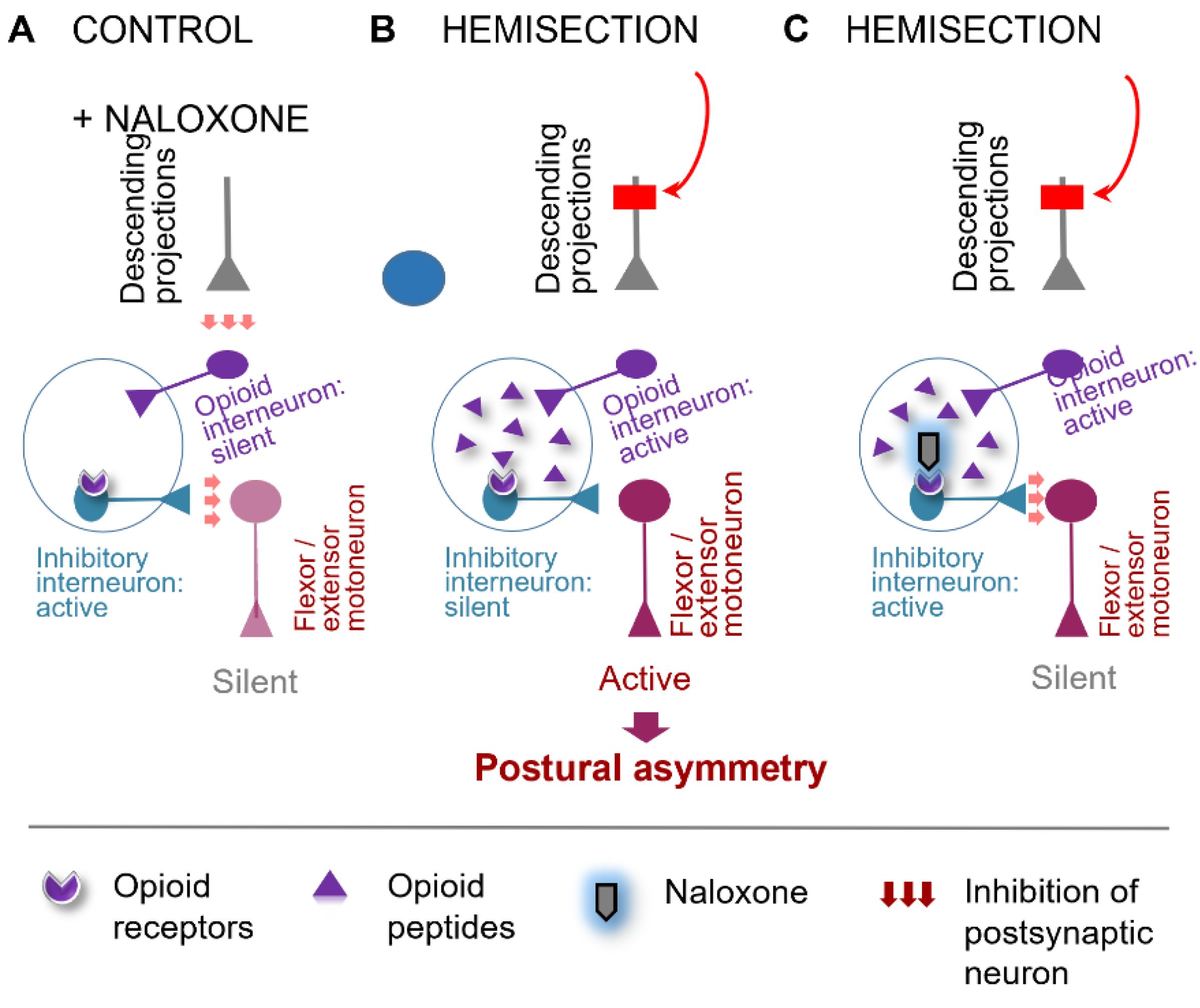
4.2. The Opioid HL-PA Mechanism
4.3. Humoral Signaling in HL-PA Formation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HL-PA | Hindlimb postural asymmetry |
| LHS | Lateral hemisection of the spinal cord |
| SCI | Spinal cord injury () |
References
- Wirz M, Zörner B, Rupp R, Dietz V. Outcome after incomplete spinal cord injury: central cord versus Brown-Sequard syndrome. Spinal Cord. 2010;48:407-414. [CrossRef]
- Takeoka A, Arber S. Functional Local Proprioceptive Feedback Circuits Initiate and Maintain Locomotor Recovery after Spinal Cord Injury. Cell Rep. 2019;27(1):71-85.e3. [CrossRef]
- Zorner B, Bachmann LC, Filli L, at al. Chasing central nervous system plasticity: the brainstem’s contribution to locomotor recovery in rats with spinal cord injury. Brain. 2014;137:1716–1732.
- Aminoff, MJ. The life and legacy of Brown-Séquard. Brain. 2017;140(5):1525-1532. [CrossRef]
- Han Q, Ordaz JD, Liu NK, et al. Descending motor circuitry required for NT-3 mediated locomotor recovery after spinal cord injury in mice. Nat Commun 2019;10:5815. [CrossRef]
- Hutson TH, Di Giovanni S. The translational landscape in spinal cord injury: focus on neuroplasticity and regeneration. Nat Rev Neurol. 2019;15:732–745. [CrossRef]
- Asboth L, Friedli L, Beauparlant J, et al., Cortico-reticulo-spinal circuit reorganization enables functional recovery after severe spinal cord contusion. Nat Neurosci. 2018;21(4):576-588. [CrossRef]
- Han Q, Xie Y, Ordaz JD, et al. Restoring Cellular Energetics Promotes Axonal Regeneration and Functional Recovery after Spinal Cord Injury. Cell Metab. 2020;31(3):623-641.e8. [CrossRef]
- Wang D, Tawfik VL, Corder G, Low SA, Francois A, Basbaum AI, Scherrer G. Functional Divergence of Delta and Mu Opioid Receptor Organization in CNS Pain Circuits. Neuron 2018;98:90-108. [CrossRef]
- Clarke RW, Galloway FJ, Harris J, Taylor JS, Ford TW. Opioidergic inhibition of flexor and extensor reflexes in the rabbit. The Journal of physiology 1992;449:493-501. [CrossRef]
- Steffens H, Schomburg ED. Spinal motor actions of the mu-opioid receptor agonist DAMGO in the cat. Neuroscience research. 2011;70:44-54. [CrossRef]
- Faber ES, Chambers JP, Brugger F, Evans RH. Depression of A and C fibre-evoked segmental reflexes by morphine and clonidine in the in vitro spinal cord of the neonatal rat. British journal of pharmacology. 1997;120:1390-1396.
- . [CrossRef]
- Jankowska, E. Schomburg ED. A leu-enkephalin depresses transmission from muscle and skin non-nociceptors to first-order feline spinal neurones. The Journal of physiology 1998;510:513-525. [CrossRef]
- Terminel, M.N. , Bassil, C., Rau, J. et al. Morphine-induced changes in the function of microglia and macrophages after acute spinal cord injury. BMC Neurosci. 2022;23(1):58. [CrossRef]
- Rau J, Hemphill A, Araguz K, et al. Adverse Effects of Repeated, Intravenous Morphine on Recovery after Spinal Cord Injury in Young, Male Rats Are Blocked by a Kappa Opioid Receptor Antagonist. J Neurotrauma. 2022;39(23-24):1741-1755. [CrossRef]
- Yue WWS, Touhara KK, Toma K, Duan X, Julius D. Endogenous opioid signalling regulates spinal ependymal cell proliferation. Nature. 2024;634(8033):407-414. [CrossRef]
- Lukoyanov N, Watanabe H, Carvalho LS, et al. Left-right side-specific endocrine signaling complements neural pathways to mediate acute asymmetric effects of brain injury. eLife. 2021;10:e65247. [CrossRef]
- Watanabe H, Nosova O, Sarkisyan D, et al. Ipsilesional versus contralesional postural deficits induced by unilateral brain trauma: a side reversal by opioid mechanism. Brain Commun. 2020;2, fcaa208. [CrossRef]
- Watanabe H, Nosova O, Sarkisyan D, et al. Left-right side-specific neuropeptide mechanism mediates contralateral responses to a unilateral brain injury. eNeuro. 2021.
- Watanabe H, Kobikov Y, Nosova O, et al. The Left-Right Side-Specific Neuroendocrine Signaling from Injured Brain: An Organizational Principle. Function (Oxf). 2024;5(4):zqae013.
- . [CrossRef]
- Bakalkin, G. The left-right side-specific endocrine signaling in the effects of brain lesions: questioning of the neurological dogma. Cell Mol Life Sci. 2022;79(11):545. [CrossRef]
- Lin XJ, Wen S, Deng LX, et al. Spinal cord lateral hemisection and asymmetric behavioral assessments in adult rats. J Vis Exp. 2020. [CrossRef]
- Webb AA, Muir GD. Compensatory locomotor adjustments of rats with cervical or thoracic spinal cord hemisections. J Neurotrauma. 2002;19:239-56. [CrossRef]
- Zhang M, Watanabe H, Sarkisyan D, et al. Hindlimb motor responses to unilateral brain injury: spinal cord encoding and left-right asymmetry. Brain Commun. 2020;2(1):fcaa055. [CrossRef]
- Hultborn H, Malmsten J. Changes in segmental reflexes following chronic spinal cord hemisection in the cat. I. Increased monosynaptic and polysynaptic ventral root discharges. Acta Physiol Scand. 1983;119(4):405-422. [CrossRef]
- Gossard JP, Delivet-Mongrain H, Martinez M, Kundu A, Escalona M, Rossignol S. Plastic Changes in Lumbar Locomotor Networks after a Partial Spinal Cord Injury in Cats. J Neurosci. 2015;35(25):9446-55. [CrossRef]
- Miller JF, Paul KD, Lee RH, Rymer WZ, Heckman CJ. Restoration of extensor excitability in the acute spinal cat by the 5-HT2 agonist DOI. J Neurophysiol. 1996 Feb;75(2):620-8. [CrossRef] [PubMed]
- Musienko PE, Zelenin PV, Orlovsky GN, Deliagina TG. Facilitation of postural limb reflexes with epidural stimulation in spinal rabbits. J Neurophysiol. 2010 Feb;103(2):1080-92. Epub 2009 Dec 16. [CrossRef] [PubMed] [PubMed Central]
- Frigon A, Johnson MD, Heckman CJ. Altered activation patterns by triceps surae stretch reflex pathways in acute and chronic spinal cord injury. J Neurophysiol. 2011 Oct;106(4):1669-78. Epub 2011 Jul 6. [CrossRef] [PubMed] [PubMed Central]
- Zhou HH, Jin TT, Qin B, Turndorf H. Suppression of spinal cord motoneuron excitability correlates with surgical immobility during isoflurane anesthesia. Anesthesiology. 1998 Apr;88(4):955-61. [CrossRef] [PubMed]
- Fuchigami T, Kakinohana O, Hefferan MP, Lukacova N, Marsala S, Platoshyn O, Sugahara K, Yaksh TL, Marsala M. Potent suppression of stretch reflex activity after systemic or spinal delivery of tizanidine in rats with spinal ischemia-induced chronic spastic paraplegia. Neuroscience. 2011 Oct 27;194:160-9.022. Epub 2011 Aug 16. [CrossRef] [PubMed] [PubMed Central]
- Jankowska, E. Interneuronal relay in spinal pathways from proprioceptors. Prog Neurobiol. 1992;38(4):335-78. [CrossRef] [PubMed]
- Valero-Cabré A, Forés J, Navarro X. Reorganization of reflex responses mediated by different afferent sensory fibers after spinal cord transection. J Neurophysiol. 2004 Jun;91(6):2838-48. Epub 2004 Feb 4. [CrossRef] [PubMed]
- Lavrov I, Gerasimenko Y, Burdick J, Zhong H, Roy RR, Edgerton VR. Integrating multiple sensory systems to modulate neural networks controlling posture. J Neurophysiol. 2015 Dec;114(6):3306-14. Epub 2015 Oct 7. [CrossRef] [PubMed] [PubMed Central]
- Gracies, JM. Pathophysiology of spastic paresis. I: Paresis and soft tissue changes. Muscle Nerve. 2005 May;31(5):535-51. [CrossRef] [PubMed]
- Lorentzen J, Pradines M, Gracies JM, Bo Nielsen J. On Denny-Brown's 'spastic dystonia' - What is it and what causes it? Clin Neurophysiol. 2018 Jan;129(1):89-94. Epub 2017 Nov 4. [CrossRef] [PubMed]
- Marinelli L, Currà A, Trompetto C, Capello E, Serrati C, Fattapposta F, Pelosin E, Phadke C, Aymard C, Puce L, Molteni F, Abbruzzese G, Bandini F. Spasticity and spastic dystonia: the two faces of velocity-dependent hypertonia. J Electromyogr Kinesiol. 2017 Dec;37:84-89. Epub 2017 Sep 27. [CrossRef] [PubMed]
- Sheean G, McGuire JR. Spastic hypertonia and movement disorders: pathophysiology, clinical presentation, and quantification. PM R. 2009 Sep;1(9):827-33. [CrossRef] [PubMed]
- Baude M, Nielsen JB, Gracies JM. The neurophysiology of deforming spastic paresis: A revised taxonomy. Ann Phys Rehabil Med. 2019 Nov;62(6):426-430. Epub 2018 Nov 28. [CrossRef] [PubMed]
- Denny-Brown, D. The cerebral control of movement. Liverpool: Liverpool University Press; 1966. p. 210-7.
- Denny-Brown, D. Preface: historical aspects of the relation of spasticity to movement. In: Feldman RG, Young RR, Koella WP, editors. Spasticity: disordered motor control. Chicago: Yearbook Medical 1980. p. 1-16.
- Pingel J, Bartels EM, Nielsen JB. New perspectives on the development of muscle contractures following central motor lesions. J Physiol. 2017 Feb 15;595(4):1027-1038. Epub 2016 Dec 7. PMCID: PMC5309377. [CrossRef] [PubMed]
- Bakalkin GIa, Iarygin KN, Trushina ED, Titov MI, Smirnov VN. Predpochtitel'noe razvitie fleksii levoĭ ili pravoĭ zadneĭ konechnosti pod deĭstviem sootvetstvenno metionin-énkefalina i leĭtsin-énkefalina [Preferential development of flexion of the left or right hindlimb as a result of treatment with methionine-enkephalin or leucine-enkephalin, respectively]. Dokl Akad Nauk SSSR. 1980;252(3):762-5. Russian. [PubMed]
- Chazov EI, Bakalkin GYa, Yarigin KN, Trushina ED, Titov MI, Smirnov VN. Enkephalins induce asymmetrical effects on posture in the rat. Experientia. 1981;37(8):887-9. [CrossRef] [PubMed]
- Bakalkin GYa, Kobylyansky AG. Opioids induce postural asymmetry in spinal rat: the side of the flexed limb depends upon the type of opioid agonist. Brain Res. 1989 Feb 20;480(1-2):277-89. [CrossRef] [PubMed]
- Kononenko O, Galatenko V, Andersson M, Bazov I, Watanabe H, Zhou XW, Iatsyshyna A, Mityakina I, Yakovleva T, Sarkisyan D, Ponomarev I, Krishtal O, Marklund N, Tonevitsky A, Adkins DL, Bakalkin G. Intra- and interregional coregulation of opioid genes: broken symmetry in spinal circuits. FASEB J. 2017 May;31(5):1953-1963. Epub 2017 Jan 25. [CrossRef] [PubMed] [PubMed Central]
- Serafin EK, Paranjpe A, Brewer CL, Baccei ML. Single-nucleus characterization of adult mouse spinal dynorphin-lineage cells and identification of persistent transcriptional effects of neonatal hindpaw incision. Pain. 2021;162(1):203-218. [CrossRef]
- Schomburg ED. Spinal sensorimotor systems and their supraspinal control. Neurosci Res. 1990;7(4):265-340. [CrossRef]
- François A, Low SA, Sypek EI, et al. A Brainstem-Spinal Cord Inhibitory Circuit for Mechanical Pain Modulation by GABA and Enkephalins. Neuron. 2017;93(4):822-839.e6. [CrossRef]
- Baskin DS, Hosobuchi Y. Naloxone reversal of ischaemic neurological deficits in man. Lancet. 1981 Aug 8;2(8241):272-5. [CrossRef] [PubMed]
- Hosobuchi Y, Baskin DS, Woo SK. Reversal of induced ischemic neurologic deficit in gerbils by the opiate antagonist naloxone. Science. 1982 Jan 1;215(4528):69-71. [CrossRef] [PubMed]
- Baskin DS, Kieck CF, Hosobuchi Y. Naloxone reversal and morphine exacerbation of neurologic deficits secondary to focal cerebral ischemia in baboons. Brain Res. 1984 Jan 9;290(2):289-96. [CrossRef] [PubMed]
- Jabaily J, Davis JN. Naloxone administration to patients with acute stroke. Stroke. 1984 Jan-Feb;15(1):36-9. [CrossRef] [PubMed]
- Namba S, Nishigaki S, Fujiwara N, Wani T, Namba Y, Masaoka T. Opiate-antagonist reversal of neurological deficits--experimental and clinical studies. Jpn J Psychiatry Neurol. 1986 Mar;40(1):61-79. [CrossRef] [PubMed]
- Skarphedinsson JO, Delle M, Hoffman P, Thorén P. The effects of naloxone on cerebral blood flow and cerebral function during relative cerebral ischemia. J Cereb Blood Flow Metab. 1989 Aug;9(4):515-22. [CrossRef] [PubMed]
- Hans P, Brichant JF, Longerstay E, Damas F, Remacle JM. Reversal of neurological deficit with naloxone: an additional report. Intensive Care Med. 1992;18(6):362-3. [CrossRef] [PubMed]
- Baskin DS, Widmayer MA, Browning JL, Heizer ML, Schmidt WK. Evaluation of delayed treatment of focal cerebral ischemia with three selective kappa-opioid agonists in cats. Stroke. 1994 Oct;25(10):2047-53; discussion 2054. [CrossRef] [PubMed]
- Wang X, Sun ZJ, Wu JL, Quan WQ, Xiao WD, Chew H, Jiang CM, Li D. Naloxone attenuates ischemic brain injury in rats through suppressing the NIK/IKKα/NF-κB and neuronal apoptotic pathways. Acta Pharmacol Sin. 2019 Feb;40(2):170-179. Epub 2018 Jun 14. [CrossRef] [PubMed] [PubMed Central]
- Gironi M, Martinelli-Boneschi F, Sacerdote P, Solaro C, Zaffaroni M, Cavarretta R, Moiola L, Bucello S, Radaelli M, Pilato V, Rodegher M, Cursi M, Franchi S, Martinelli V, Nemni R, Comi G, Martino G. A pilot trial of low-dose naltrexone in primary progressive multiple sclerosis. Mult Scler. 2008 Sep;14(8):1076-83. [CrossRef] [PubMed]
- Bakalkin GYa, Kobylyansky AG, Nagornaya LV, Yarygin KN, Titov MI. Met-enkephalin-induced release into the blood of a factor causing postural asymmetry. Peptides. 1986;7(4):551-556. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



