Submitted:
26 March 2025
Posted:
26 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
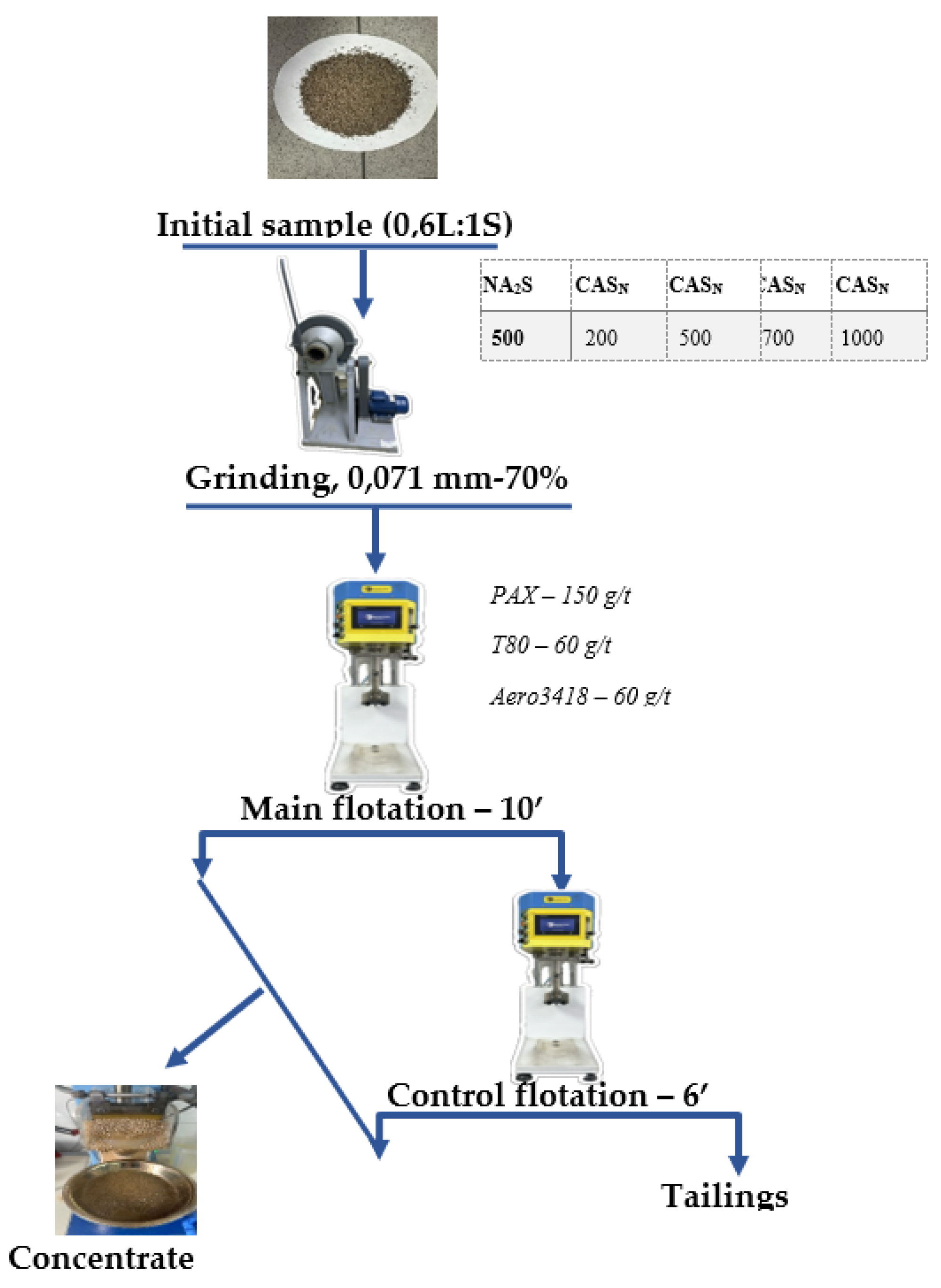
2. Materials and Methods
3. Results
| Dosage, g/t | Product | Yield, % | Content, %/g/t* | Recovery, % | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Pb | Zn | Ag* | Fe | Pb | Zn | Ag | Fe | |||
| Base Test | ||||||||||
| Na2S - 500 | Concentrate | 3.10 | 8.77 | 1.34 | 116.21 | 6.31 | 46.90 | 7.40 | 65.50 | 3.10 |
| Tailings | 96.90 | 0.32 | 0.54 | 1.96 | 6.31 | 53.10 | 92.60 | 34.50 | 96.60 | |
| Initial Sample | 100.0 | 0.58 | 0.56 | 5.50 | 6.31 | 100.0 | 100.0 | 100.0 | 100.0 | |
| Test 1 | ||||||||||
| CaSₙ - 200 | Concentrate | 3.50 | 7.24 | 1.30 | 92.90 | 8.11 | 43.70 | 8.10 | 59.10 | 4.50 |
| Tailings | 96.50 | 0.34 | 0.53 | 2.33 | 6.24 | 56.30 | 91.90 | 40.90 | 95.50 | |
| Initial Sample | 100.0 | 0.58 | 056 | 5.50 | 6.31 | 100.0 | 100.0 | 100.0 | 100.0 | |
| Test 2 | ||||||||||
| CaSₙ - 500 | Concentrate | 3.71 | 7.58 | 1.19 | 92.36 | 6.97 | 48.51 | 7.90 | 62.30 | 4.10 |
| Tailings | 96.29 | 0.31 | 0.54 | 2.15 | 6.28 | 51.49 | 92.10 | 37.70 | 95.90 | |
| Initial Sample | 100.0 | 0.58 | 0.56 | 5.50 | 6.31 | 100.0 | 100.0 | 100.0 | 100.0 | |
| Test 3 | ||||||||||
| CaSₙ - 700 | Concentrate | 3.82 | 8.13 | 1.06 | 95.20 | 6.61 | 53.56 | 7.2 | 66.10 | 4.00 |
| Tailings | 96.18 | 0.28 | 0.54 | 1.94 | 6.30 | 46.44 | 92.80 | 33.90 | 96.00 | |
| Initial Sample | 100.0 | 0.58 | 0.56 | 5.50 | 6.31 | 100.0 | 100.0 | 100.0 | 100.0 | |
| Test 4 | ||||||||||
| CaSₙ - 1000 | Concentrate | 4.02 | 7.39 | 0.99 | 87.84 | 5.65 | 51.23 | 7.10 | 64.20 | 3.60 |
| Tailings | 95.98 | 0.29 | 0.54 | 2.05 | 6.34 | 48.77 | 92.90 | 35.80 | 96.40 | |
| Initial Sample | 100.0 | 0.58 | 0.56 | 5.50 | 6.31 | 100.0 | 100.0 | 100.0 | 100.0 | |
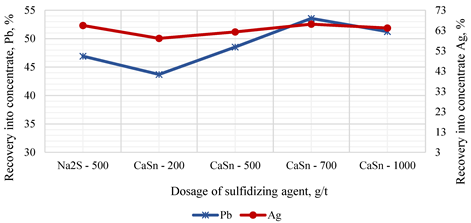
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| РАХ | potassium xanthogenate amyl |
References
- Nayak, M.S. Jena, N.R. Mandre. Beneficiation of lead–zinc ores—a review/ Miner. Process. Extr. Metall. Rev., 43 (5), 2022, pp.564-583. [CrossRef]
- Weiping Liu, Zuoxing Wang, Xuming Wang, Jan D. Miller. Smithsonite flotation with lauryl phosphate // Minerals Engineering, Volume 147, 1 March 2020, № 106155. [CrossRef]
- Wengang Liu, Liang Zhao, Wenbao Liu, Yongxing Zheng, Lingyun Huang, Yong Mao, Shengyuan Ding. Enhanced flotation of smithsonite from calcite based on the synergistic action of carboxylated chitosan and sodium carbonate // Advanced Powder Technology, Volume 34, Issue 12, December 2023, № 104261. [CrossRef]
- Sajjad Maghfouri, Mohammad Reza Hosseinzadeh, Abdorrahman Rajabi, Flavien Choulet. A review of major non-sulfide zinc deposits in Iran // Geoscience Frontiers, Volume 9, Issue 1, January 2018, рр. 249-272 . [CrossRef]
- 5. Yahui Yi, Peixuan Li, Ga Zhang, Qicheng Feng, Guang Han, Stepwise activation of hemimorphite surfaces with lead ions and its contribution to sulfidization flotation // Separation and Purification Technology, Volume 299, 15 October 2022, №121679 . [CrossRef]
- Martha Araceli Elizondo-Álvarez, Alejandro Uribe-Salas, Fabiola Nava-Alonso, Flotation studies of galena (PbS), cerussite (PbCO3) and anglesite (PbSO4) with hydroxamic acids as collectors // Minerals Engineering, Volume 155, 15 August 2020, № 106456 . [CrossRef]
- Jiwei Xue, Yaoben Qu, Yao Chen, Chonghui Zhang, Xianzhong Bu, Effective sulfide flotation of cerussite by using trithiocyanuric acid as a novel sulfurizing reagent // Minerals Engineering, Volume 198, July 2023, №108087 . [CrossRef]
- Song Zhang, Yongjun Xian, Shuming Wen, Guanyu Liang, Enhancement of xanthate adsorption on lead-modified and sulfurized smithsonite surface in the presence of ammonia // Minerals Engineering, Volume 189, November 2022, №107872 . [CrossRef]
- Qi Zuo, Jing Yang, Yunfeng Shi, Dandan Wu, Activating hemimorphite using a sulfidation-flotation process with sodium sulfosalicylate as the complexing agent // Journal of Materials Research and Technology, Volume 9, Issue 5, September–October 2020, рр. 10110-10120 . [CrossRef]
- Shaojun Bai, Pan Yu, Zhan Ding, Chunlong Li, Yongjun Xian, Shuming Wen, Ammonium chloride catalyze sulfidation mechanism of smithsonite surface: Visual MINTEQ models, ToF-SIMS and DFT studies // Minerals Engineering, Volume 146, 15 January 2020, № 106115 . [CrossRef]
- Y.X. Zheng, J.F. Lv, H. Wang, S.M. Wen, J. Pang, Formation of zinc sulfide species during roasting of ZnO with pyrite and its contribution on flotation // Scientific Reports., Volume 8, Issue 1, 1 December 2018, № 7839. [CrossRef]
- B.L. Ge, J. Pang, Y.X. Zheng, J.L. Ning, J.F. Lü, Sulfidation mechanism of cerussite in the presence of sulphur at high temperatures // J. Cent. South Univ., 27 (11) (2020), pp. 3259-3268 . [CrossRef]
- Y. Ke, N. Peng, K. Xue, X.B. Min, L.Y. Chai, Q.L. Pan, Y.J. Liang, R.Y. Xiao, Y.Y. Wang, C.J. Tang, H. Liu, Sulfidation behavior and mechanism of zinc silicate roasted with pyrite // Applied Surface Science., Volume 435, 30 March 2018, рр. 1011-1019. [CrossRef]
- X.Y. Wei, J.W. Han, Y.W. Wang, R. Huang, X.S. Gao, W.Q. Qin, Sulfidation behaviors and phase transformation mechanism of zinc silicate // Chinese Journal of Nonferrous Metals, Volume 32, Issue 12, 28 December 2022, рр. 3811-3822. [CrossRef]
- Hui Liu, Kai Pan, Chenglang Xiang, Dong Ye, Haining Wang, Xiaoqing Gou, Mechanochemical effect of spontaneous combustion of sulfide ore // Fuel, Volume 329, 1 December 2022, №125391. [CrossRef]
- Zhao Li, Min Chen, Peng-wu Huang, Qi-wu Zhang, Shao-xian Song, Effect of grinding with sulfur on surface properties and floatability of three nonferrous metal oxides // Transactions of Nonferrous Metals Society of China, Volume 27, Issue 11, November 2017, рр. 2474-2480. [CrossRef]
- Eric A. Runge, Muammar Mansor, Jeremiah Shuster, Stefan Fischer, Yali Liu, Dominique J. Lunter, Andreas Kappler, Jan-Peter Duda, Sulfidation of nano-magnetite to pyrite: Implications for interpreting paleoenvironmental proxies and biosignature records in hydrothermal sulfide deposits // Earth and Planetary Science Letters, Volume 617, 1 September 2023, №118261. [CrossRef]
- C.X. Li, C. Wei, Z.G. Deng, X.B. Li, M.T. Li, H.S. Xu, Hydrothermal sulfidation and flotation of oxidized zinc–lead ore // Metallurgical and Materials Transactions B, Volume 45, pр. 833–838, (2014). [CrossRef]
- H. Tan, J.X. Jin, Y.C. Shao, D. Zhou, Y. Zhou, Z.X. Wu, M.J. Wu, D.S. Shen, Y.Y. Long, Microwave hydrothermal sulfidation process for zinc-containing plating sludge // Separation Science and Technology, 56(17), рр. 3001–3010. [CrossRef]
- Yuanjia Luo, Leming Ou, Jianhua Chen, Guofan Zhang, Understanding the impact of ammonium salt on the sulfidization of smithsonite from coordination chemistry // Applied Surface Science, Volume 635, 30 October 2023, №157723. [CrossRef]
- X. Zhang, J.S. Deng, Y. Wang, G.Y. Wang, H.X. Xu, Novel insight into the lead sulfide species formed on hemimorphite surface during lead ions improved sulfidation // Colloids and Surfaces A: Physicochemical and Engineering Aspects, 653 (2022), № 129959. [CrossRef]
- Qicheng Feng, Meili Wang, Ga Zhang, Wenjuan Zhao, Guang Han, Enhanced adsorption of sulfide and xanthate on smithsonite surfaces by lead activation and implications for flotation intensification // Separation and Purification Technology, Volume 307, 15 February 2023, №122772. [CrossRef]
- Wenjuan Zhao, Bin Yang, Dianwen Liu, Qicheng Feng, Effect of copper ions on sulfidization flotation of smithsonite: Surface properties and adsorption mechanism // Colloids and Surfaces A: Physicochemical and Engineering Aspects, Volume 650, 5 October 2022, 1№ 29515. [CrossRef]
- A.R. Mambetalieva, A. Mukhtarkyzy, M.R. Shautenov. Study of the effect of sulfidization using sodium sulfide on oxidized lead-zinc ores of the Koskuduk deposit. Vestnik KazUTB, 2025. ISSN: 2708-4132. рр.
- N. K. Tussupbayev, A.A. Mukhanova, S.M. Narbekova, L.V. Syemushkina, D.K. Turysbekov. Application of calcium polysulphide as sulfidizing agent at flotation of oxidized lead – bearing ores // Complex Use of Mineral Resources № 4. 2016, рр 12 - 16.
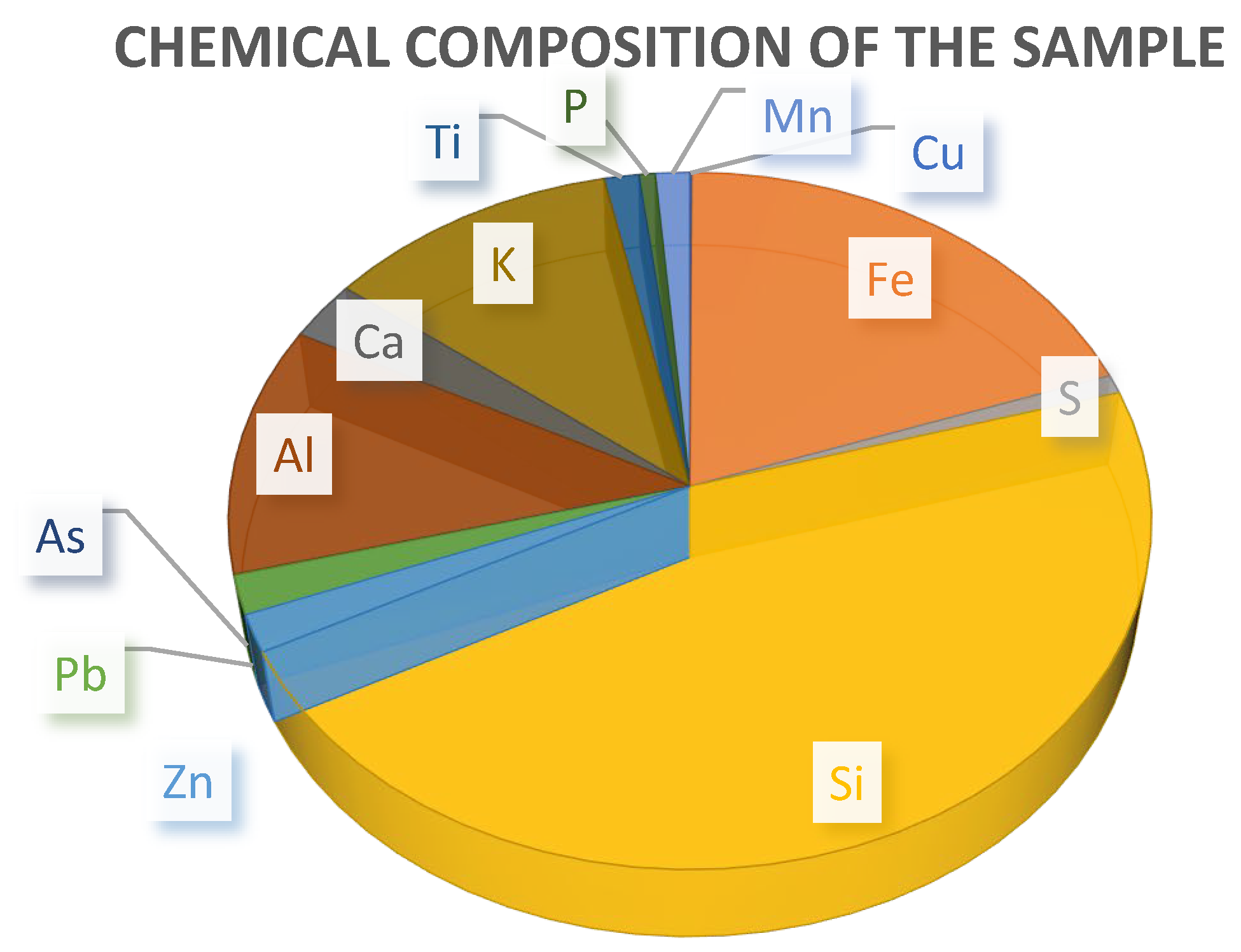

| Component Forms | Absolute Content, % | Relative Content, % |
|---|---|---|
| Zinc Forms: | ||
| - Water-soluble | <0.05 | - |
| - Oxidized | 0.31 | 55.36 |
| - Sulfides | 0.07 | 12.50 |
| - Refractory | 0.18 | 32.14 |
| Total | 0.56 | 100.0 |
| Lead Forms: | ||
| - Oxygen-containing | 0.29 | 50.00 |
| - Galena | 0.13 | 22.41 |
| - Residual forms | 0.16 | 27.59 |
| Total | 0.58 | 100.0 |
| Iron Forms: | ||
| - Sulfides | <0.05 | - |
| - Divalent (Fe²⁺) | 3.06 | 48.60 |
| - Trivalent (Fe³⁺) | 3.24 | 51.40 |
| Total | 6.30 | 100.0 |
| Size Fraction, mm | Yield, % | Content, % | Distribution, % | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Zn | Pb | Fe | Ag | Zn | Pb | Fe | Ag | ||
| -1 + 0.5 | 26.46 | 0.37 | 0.30 | 5.83 | 4.26 | 17.88 | 13.80 | 24.70 | 20.7 |
| -0.5 + 0.2 | 19.90 | 0.51 | 0.48 | 6.21 | 4.79 | 18.62 | 16.62 | 19.77 | 17.5 |
| -0.2 + 0.1 | 32.89 | 0.54 | 0.55 | 5.92 | 5.17 | 32.51 | 31.90 | 31.14 | 31.21 |
| -0.1 + 0.071 | 9.49 | 0.73 | 0.91 | 6.90 | 9.27 | 12.55 | 15.13 | 10.48 | 16.14 |
| -0.071 + 0.045 | 3.31 | 0.83 | 1.06 | 7.43 | 7.00 | 5.00 | 6.16 | 3.93 | 4.24 |
| -0.045 + 0 | 7.96 | 0.93 | 1.17 | 7.84 | 6.99 | 13.43 | 16.39 | 9.98 | 10.20 |
| Total | 100.0 | 0.56 | 0.58 | 6.31 | 5.50 | 100.0 | 100.0 | 100.0 | 100.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




