1. Introduction
Entropy is one of the most fascinating concepts in physics and it plays a crucial role in quantifying the irreversibility of change in thermodynamic systems, which is a consequence of the energy dissipation that takes place during various processes [
1,
2,
3]. The universality of this concept, its applicability irreversible physical processes both near to and far from equilibrium, as well as its connection to statistics, highlights entropy as a bridge between natural sciences and philosophy [
4]. Self-maintenance within a set of catalytic chemical reactions and conversion of biochemical energy into mechanical energy, and the reverse process, occur through intricate molecular interactions between an organism and its environment [
5,
6]. Living cells are profoundly affected by their microenvironment, which alters their mechanical properties and structural composition, and most importantly, their metabolic activities [
7]. This energy self-generation, as well as conversion, and transport, are fundamental to the emergence and maintenance of life, including morphogenesis and wound healing occurring over a range of time-scales. Energy conversion and transport can be optimised by reducing energy dissipation. Peter McClintock [
8] pointed out that a structural organisation capable of supporting the network of molecular processes responsible for controllable production and utilization of energy is a necessary condition for life. In accordance with the fact that life represents a ‘’battle for structural ordering’’ [
9], the emergence of life and its evolution can be discussed in the context of entropy production.
Alterations in energy transformation, coupled with induced energy dissipation, can result in diverse mutations and the emergence of cancerous diseases [
10]. This aligns with the understanding that energy dissipation is primarily an undesirable loss of energy; however, within biochemical processes, it has the potential to modify biochemical pathways and promote the advancement of cancer [
11,
12]. As a result, numerous therapeutic approaches have been developed to enhance the metabolic energy dissipation in tumours [
12]. Cells possess the ability to regulate energy dissipation to a certain degree through mechanisms of cell signalling and gene expression, which in turn influence the remodelling of adhesion contacts between cells and the extracellular matrix, as well as alterations in cell morphology and division [
13,
14,
15,
16].
The main goal of this review is to emphasize the impact of the viscoelasticity of migrating epithelial collectives on the dissipation of mechanical energy and entropy production. Collective cell migration is an integral part of many biological processes such as: morphogenesis, wound healing, and the spreading of cancer. Consequently, a persistent migration of aligned cells, quantified by the velocity correlation length and the persistence of cell migration, can reduce energy dissipation and protect living organisms against various mutations and development of cancer [
17,
18]. This complex phenomenon of will be discussed for model systems such as the collective migration of epithelial monolayers on substrate matrices. Migrating epithelial collectives can be treated as irreversible thermodynamic systems far from equilibrium. The multi-scale nature entropy production is influenced by the viscoelasticity and surface characteristics of multicellular systems, particularly in relation to the epithelial and matrix surface tensions, as well as the interfacial tension that exists between them [
19]. Entropy production at the supracellular level, which take places over a time-scale of hours, is examined through the lens of continuum mechanics, particularly in relation to thermodynamic entropy as influenced by cellular velocity, packing density, and alignment. In contrast, entropy production at the cellular level is analyzed in terms of conformational entropy, which occurs over a time-scale of minutes. The viscoelastic behavior, in conjunction with the surface properties of epithelial monolayers, is contingent upon the interactions between cells, and between cells and the matrix, which have a feedback effect on cell migration [
19,
20]. In this context, three regimes of cell migration will be discussed depending on the interplay between the cell packing density, cell velocity and degree of cell orientation in the direction of migration. They are the convective, conductive (diffusion) and sub-diffusion regimes. Our analysis will focus on energy dissipation and entropy production, drawing upon a range of experimental data from the existing literature, complemented by considerations from multi-scale modeling.
2. Phenomenological description of migrating epithelial monolayers on substrate matrices
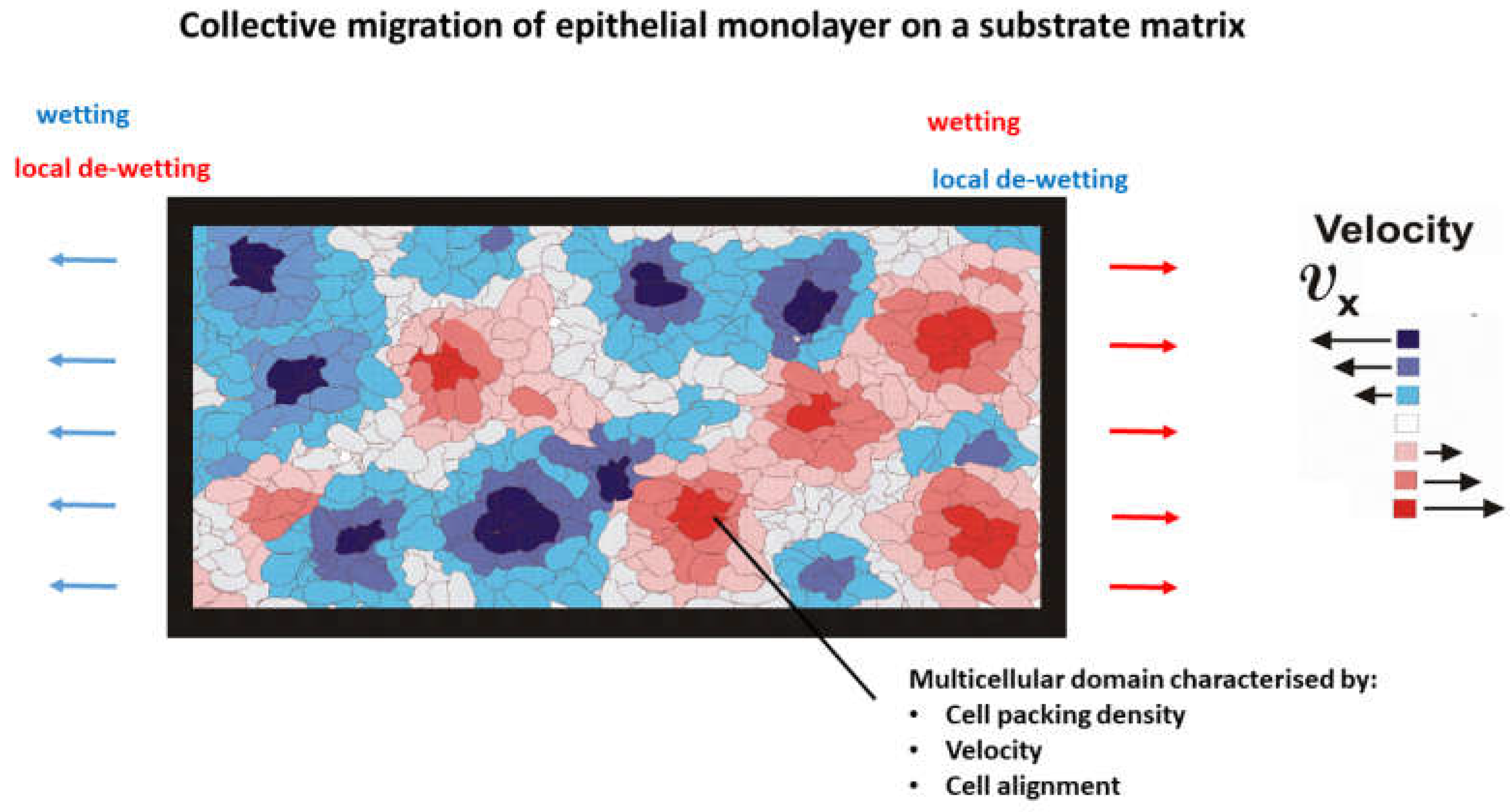
We focus here on internal entropy production during cell rearrangement caused by the collective migration of epithelial monolayers on substrate matrices. The main characteristics of migrating epithelial collectives are the inhomogeneous distribution of cell packing density, degree of cell orientation in the direction of cell migration, cell velocity, corresponding strain and mechanical stress [
19,
20,
21,
22,
23]. Cell packing density and cell velocity vary within collectively migrating epithelial monolayers. Tlili et al. [
23] investigated the active wetting properties of MDCK epithelial monolayers and found that the cell packing density ranged from
to
. An increase in cell packing density from
to
resulted in a decrease in cell velocity from
to zero [
23]. Similarly, Nnetu et al. [
22] noted that the velocity of breast epithelial MCF-10A cells reached zero at a packing density of approximately
. indicating a phenomenon known as cell jamming (
Box 1 and
Box 2). This suggests that multicellular systems can be conceptualised as an ensemble of multicellular domains as shown in
Figure 1.
The domains satisfy the following conditions:
Every domain represents a canonical ensemble of cells and can be described by homogeneous distributions of cell packing density, cell velocity, degree of cell orientation in the direction of movement, and cell mechanical stress.
The lifetime of domains is on a time-scale of hours.
Vanishing of the domains is caused by: (i) domain active and passive wetting/de-wetting; and (ii) collisions between neighboring domains.
The domain viscoelasticity and surface characteristics, which depend on cell packing density, and cell velocity have an impact on the internal production of entropy. This is consistent with the fact that change of cell packing density influences cell-cell interactions by changing intra-cellular distances and cellular alignment in the direction of migration [
20,
24].
Box 1. The main characteristics of cell-cell interactions
Two types of cell-cell interaction have been identified in the context of collective cell migration: positional and orientational interactions, which are interrelated [
16,
25]. Positional interactions among cells are influenced by the stretching or compression of adhesion contacts mediated by E-cadherin. As a result, the interaction energy can exhibit repulsive characteristics when the local distance between cells is less than approximately 8 μm, while it becomes attractive at greater distances [
26]. The characteristic time for conformational changes of proteins such as E-cadherin and integrin that represent integral parts of cell-cell and cell-matrix adhesion contacts as well as the turnover time of these proteins corresponds to a time-scale of minutes [
27].
Cell-cell orientational interactions depends on angle between neighbour cells. These interactions are influenced by compressive and shear stress components capable of inducing topological defects of cell alignment, pronounced in overcrowded environment [
28]. Two primary interaction types, namely head-on and glancing interactions, play a crucial role in the rearrangement of cells [
20,
29,
30]. Head-on interactions initiate cell re-polarisation, which is associated with a reduction in both cell-cell and cell-matrix adhesion contacts, a phenomenon referred to as contact inhibition of locomotion [
29]. After re-polarisation, cells establish cell-cell and cell-matrix adhesion contacts and start migration in the opposite direction to each other. In contrast, glancing interactions do not facilitate cell repolarization; however, they induce cell
rotation during the process of realignment, potentially leading to the disruption of cell-matrix adhesion contacts [
19]. The cell re-polarisation occurs on a time-scale of hours [
31].
The cell packing density within migrating epithelial monolayers varies from
to
(where
is the cell packing density,
is the cell packing density in the confluent state, and
is the cell packing density in the cell jamming state). Petitjean et al. [
32] demonstrated that the Madin Darby Canine Kidney (MDCK) cell monolayers attained confluence at a cell packing density of approximately
and a cell velocity of
. The cell packing density
is a few times higher than the cell packing density in the confluent state [
22]. The corresponding cell speed
decreases with the cell packing density from
to
for cells in the jamming state.
Box 2. The cell jamming state transition: the main physical characteristics
The transition to a jamming state in a collection of cells denotes a change from a contractile to a non-contractile state, induced by contact inhibition of locomotion. This effect is particularly significant in overcrowded environments where there is high compressive stress [
33,
34]. The contact inhibition of locomotion, caused by cell orientational, head-on interactions (Box 1), triggers the mechanism of cell re-polarisation by weakening the cell-cell and cell matrix adhesion contacts [29]. In the context of cell response on head-on interactions, two characteristic times should be introduced, i.e., the necessary time for cell re-polarisation, and the time between two successive cell collisions. Notbohm et al. [
31] revealed that the average re-polarisation time of MDCK cell monolayers is 1.28 h. When cells do not have enough time to re-polarise between two collisions, they enter the non-contractile (jamming) state, while the cell velocity drops to zero [
34]. The jamming state transition can be treated as a rigidity state transition [
34]. This aligns with the observation that active (contractile) cells exhibit significantly greater stiffness compared to passive (non-contractile) cells, a phenomenon attributed to the accumulation of contractile energy. Research by Schulze et al. [
35] indicates that the Young's modulus for contractile MDCK cell monolayers is approximately 33.0 ± 3.0 kPa, whereas the modulus for non-contractile cells is approximately half of this value. A reduction in cell-cell adhesion contacts results in energy dissipation, which subsequently decreases the compressive stress. As a result, cells experience an unjamming transition and resume migration.
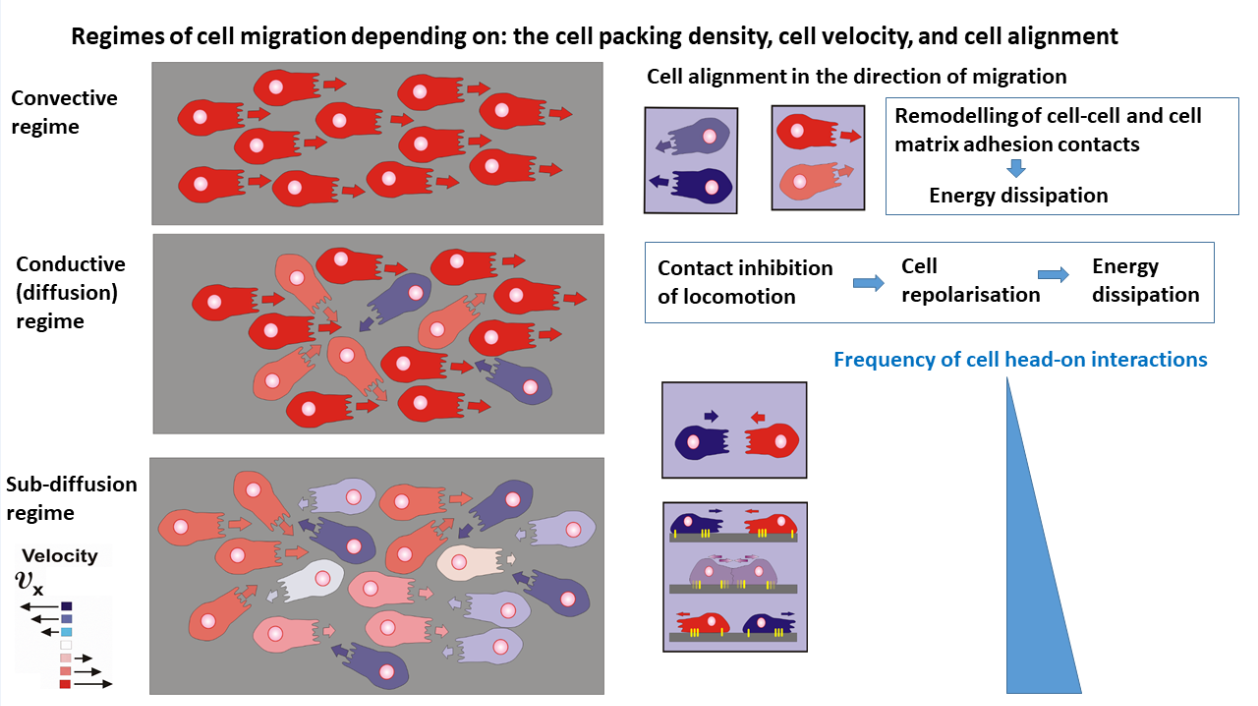
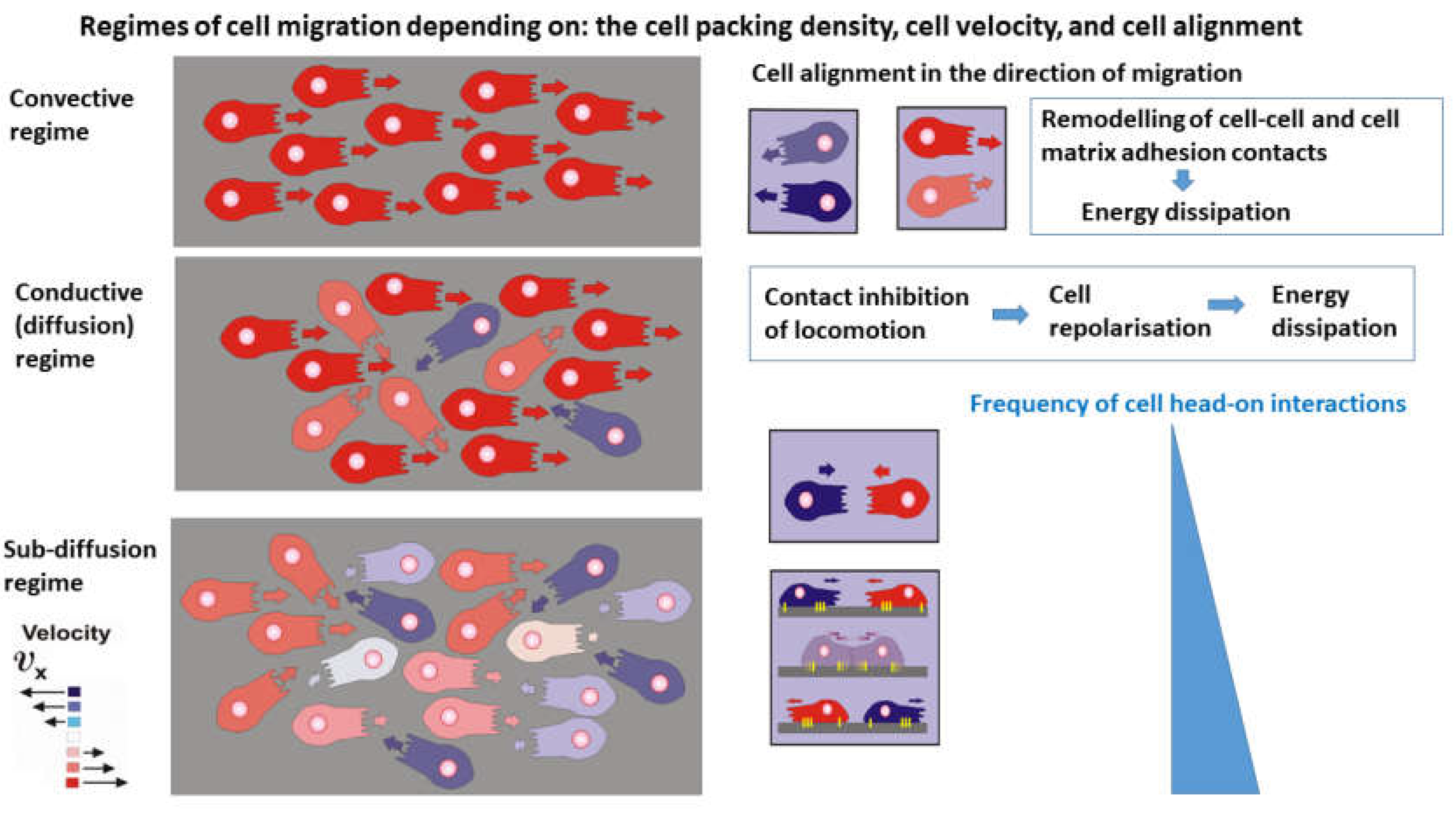
Consequently, the production of entropy can be discussed within three regimes of cell migration: the convective, diffusion, and sub-diffusion regimes as shown in
Figure 2:
The main characteristics of the regimes are given as follows:
The convective mechanism of cell migration arises within domains which satisfy the conditions that the cell packing density is
and the cell speed is
. The velocity correlation length, i.e., the distance over which cells move in a correlated fashion, is about 10 cell lengths [
17], indicating an anisotropic behavior. Contact inhibition of locomotion, caused by cell orientational interactions, is rare. Energy dissipation is caused primarily by the remodeling of cell-cell and cell matrix adhesion contacts that occurs on a time-scale of minutes [
19].
The conductive (diffusion) mechanism of cell migration arises within domains of higher cell packing density, i.e. for cell packing density
and
(where
is the cell packing density under cell jamming). An increase in cell packing density perturbs the cell alignment, quantified by a decrease in the velocity correlation length [
17,
32]. This regime of cell packing densities could be considered as an isotropic cell rearrangement. The perturbation of cell alignment leads to an increase in contact inhibition of locomotion [
25,
29,
30]. Altered cell re-polarisation accompanied by a weakening of cell-cell and cell-matrix adhesion contacts is the main cause of energy dissipation in this regime [
29,
30]. Consequently, energy dissipation occurs on a time-scale of hours. The main characteristic of this regime is that the re-polarisation time is shorter than the time between collisions, which enable cells to finalise the re-polarisation process and continue migration in the opposite direction by preventing cell jamming.
The damped conductive (sub-diffusion) mechanism occurs under high cell packing density near cell jamming within domains satisfying the conditions that
and
. The velocity correlation length corresponds to the length of a single cell [
17] indicating isotropic cell rearrangement. This high cell packing density causes intensive cell-cell interactions leading to collective contact inhibition of locomotion (
Box 1). However, in contrast to the previous case, the time necessary for cell re-polarisation is longer than the time between two cell collisions and cells are unable to finalise the re-polarisation process. The cells then undergo jamming. Consequently, energy dissipation within cell collectives near jamming occurs on a time-scale of hours.
The production of entropy, induced by energy dissipation, occurs on two time-scales. Cell shape changes and the remodeling of cell-cell and cell-matrix adhesion contacts occur on a time-scale of minutes. Cell alignment, re-polarisation, and velocity change accompanied by the corresponding strain, and cell residual stress accumulation occur on a time-scale of hours. Cell division in various types of epithelial cells typically occurs over a time frame of several days and is further extended in overcrowded environments leading to some parts of a multicellular system being in the quiescent (non-proliferative) state [
36,
37,
38].
In the discussion below, we will explore the connection between long-term entropy production and the energy dissipation rate associated with collective cell migration, incorporating relevant physical parameters. The dissipation of energy is influenced by the viscoelastic properties and surface characteristics of epithelial monolayers, which will be examined within the context of the defined regimes of cell migration.
3. Long term change of internal entropy caused by collective cell migration
A migrating epithelial collective can be treated as a closed thermodynamic system far from equilibrium. The energy storage, caused by cell rearrangement, can be expressed in the form of the Helmholtz free energy (where is the internal energy of the multicellular system, T is the temperature, which is about 300 K for biological systems and is the entropy). The internal energy of multicellular systems includes cumulative effects of various biochemical processes influenced by cell contractions and crosstalk between cell-cell and cell-matrix adhesion contacts, which occur through cytoskeleton rearrangement. An isothermal change of the free energy can be expressed as: . The internal energy change can be expressed from the first law of thermodynamics as: (where is a heat primarily generated by frictional effects and is the work of cell rearrangement , is the cell residual mechanical stress generated during collective cell migration, and is the corresponding cell strain). The entropy change can be expressed based on the second law of thermodynamics as: (where is the internally generated entropy during collective cell migration, which satisfies the condition for irreversible processes that ). Consequently, the production of entropy can be expressed as: (where represents the rate of energy dissipation ).
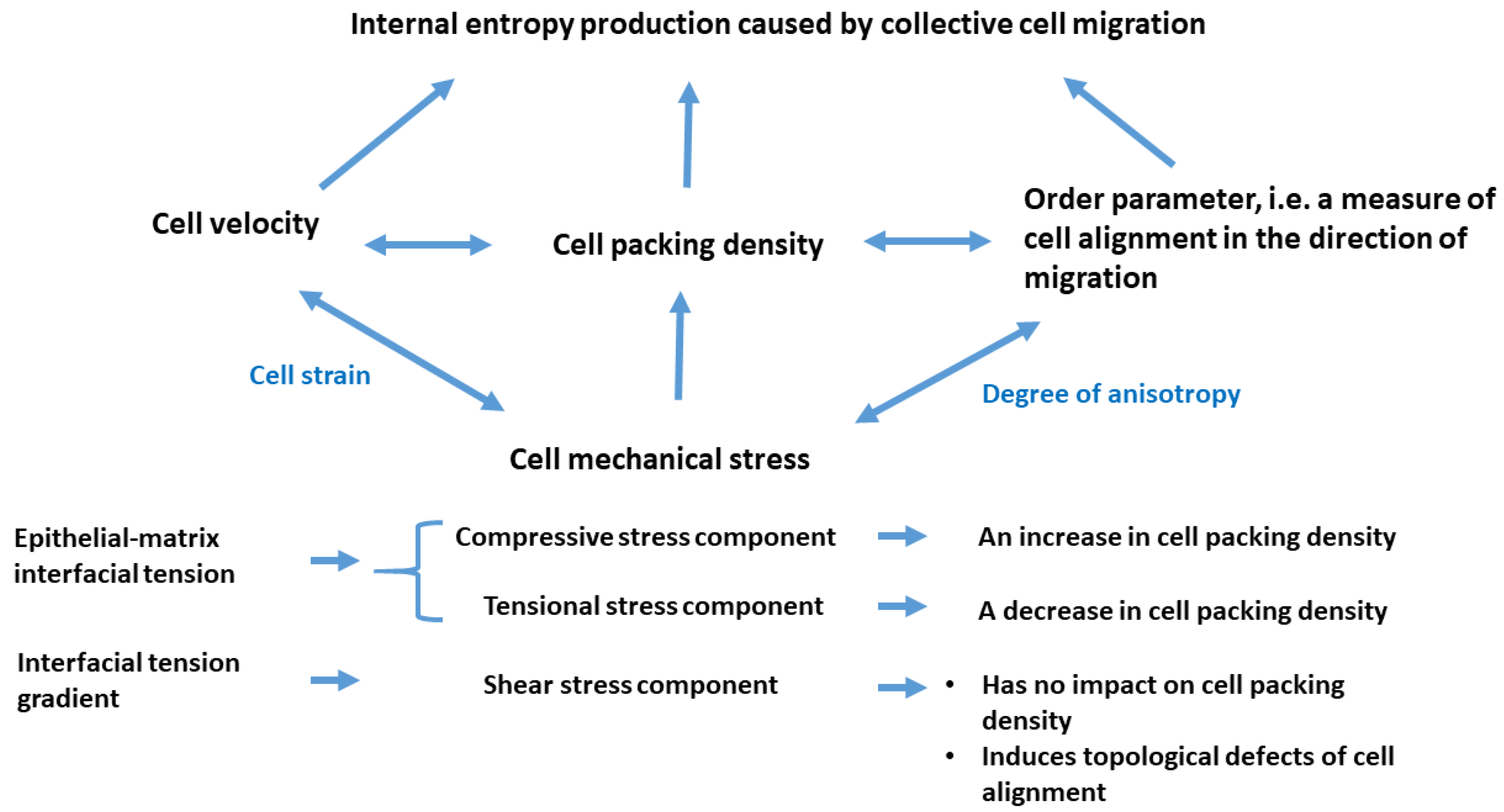
Long-term entropy production depends on the viscoelasticity and surface characteristics of multicellular systems, while the viscoelasticity itself relates to cell velocity, packing density, and cell alignment in the direction of migration. It can be expressed under isothermal conditions as:
were
is the temperature and
is the energy dissipation, while
is the cell velocity,
is the average orientational vector within the domain expressed as:
, and
is the cell packing density). Consequently, the local internal entropy production during cell rearrangement can be expressed as:
where
is the local entropy caused by cell rearrangement and
is the rate of entropy production. The entropy production rate increases with increasing cell packing density
and decreases with an increase in cell speed
and the degree of cell orientation in the direction of migration (the order parameter)
. The order parameter
ranges from 0 for a completely disordered multicellular domain, to 1 for a domain that is completely aligned [
39]. Consequently, the parameter
can be treated as a measure of the domain anisotropy such that
corresponds to isotropic viscoelasticity, while the degree of anisotropy increases with
. For
the viscoelasticity is totally anisotropic.
Box 3. Oscillations of physical parameters: mechanical waves
Collective cell migration generates mechanical waves, as noted by Serra-Picamal et al. [
21] and Notbohm et al. [
31], which have been examined in relation to low Reynolds turbulence [
40]. These mechanical waves are characterized by sustained oscillations in cell velocity, associated strain, and mechanical stress, and they have been analysed in terms of an effective inertia [
26,
31,
40]. The oscillatory nature of mechanical stress results in variations in cell packing density: (i) compressive stress leads to an increase in cell packing density, (ii) tensional stress results in a decrease in cell packing density, and (iii) shear stress does not affect cell packing density.
The evidence suggests that: (1) the cell speed oscillates [
31,
41], (2) variations in cell packing density result from oscillations in cell normal stress (
Box 3) [
19,
28], (3) cell normal stress exhibits long-term oscillatory behaviour [
21,
31], and (4) the order parameter undergoes fluctuations due to repeated perturbations and realignments of cell orientation [
28,
42]. Consequently, it can be inferred that the internal production of entropy
also undergoes oscillations.
The relationship between long-term entropy production, energy dissipation and relevant physical parameters is shown schematically in
Figure 3.
The set of physical parameters established depends on the cumulative effects of cell-cell and cell-matrix interactions, which have a feedback impact on the surface characteristics of epithelial collectives, quantified by epithelial cohesiveness and epithelial-matrix adhesiveness, and viscoelasticity caused by collective cell migration [
19,
20]. These parameters will be formulated and discussed in the next two sections.
3.1. Surface characteristics of epithelial monolayers
The cohesion and adhesion energies of multicellular systems depend on the strength of cell-cell and cell-matrix adhesion contacts and on the crosstalk between the adhesion contacts, managed by the cytoskeleton contractions [
13]. The strength of cell-cell and cell-matrix adhesion contacts relies on cell-cell positional and orientational interactions depending on micro-environmental conditions such as the stiffness and rheological behaviour of the extracellular matrix and cell packing density [
16,
20]. An inhomogeneous distribution of cell packing density accompanied by mechanical stress induces an inhomogeneous distribution in the strength of cell-cell adhesion contacts [
26,
43].
An interrelation between cohesion and adhesion energies, expressed in the form of the spreading factor is responsible for cell active or passive wetting (extension)/de-wetting (compression) of epithelial monolayers on substrate matrices [43,19). Active wetting/de-wetting occurs via collective cell migration, while passive wetting/de-wetting is driven by the gradient of the epithelial matrix interfacial tension [
19]. This phenomenon, known as the Marangoni effect [
44], significantly influences cell rearrangement and the generation of cell shear stress. The spreading factor accompanied by other physical parameters included in the surface characterization of epithelial monolayers are shown in
Table 1:
The contractile properties of epithelial cells contribute to the reinforcement of cell-cell adhesion interactions, thereby enhancing the epithelial surface tension [
50]. Furthermore, the expansion of epithelial monolayers through collective cell migration results in an increase in epithelial surface tension [
51]. Conversely, mechanical compression may disrupt cellular alignment, potentially weakening E-cadherin-mediated adhesion and leading to a reduction in epithelial surface tension [
48]. The variation in the extension of certain multicellular domains, in contrast to the compression observed in others, leads to an inhomogeneous distribution of epithelial surface tension and its long-term change. The matrix surface tension
depends on the flexibility of the polymer chains and the strength of the inter-chain bonds. Cell tractions can induce disordering of chain organization and the establishment of local polymer concentration gradients, which have a feedback impact to the distribution of the matrix surface tension [
48,
52]. The gradient of the matrix surface tension is the one of the driving forces for directional cell migration [
48]. The cell-matrix adhesion energy
depends on the strength of cell-matrix adhesion contacts and on that basis relies on: matrix stiffness, cell velocity and packing density. Non-contractile cells establish weak cell-matrix adhesion contacts. An increase in adhesion energy
leads to a reduction of epithelial-matrix interfacial tension (
Table 1). Inhomogeneous distribution of epithelial and matrix surface tension accompanied by cell-matrix adhesion energy causes an inhomogeneous distribution of the interfacial tension and generation the interfacial tension gradient [
19,
49]. The interfacial tension gradient drives passive extension of multicellular domains from the regions of lower interfacial tension to the regions of higher interfacial tension [
19].
As a result, both the interfacial tension and its gradient play a significant role in generation of the cell residual stress. In multicellular systems, residual stress is maintained during collective cell migration and shows variations over a period of several hours [
19]. The cell normal residual stress consists of isotropic and deviatoric parts. The isotropic part of the stress is caused by the work of cell-matrix interfacial tension expressed by the Young-Laplace equation. The interfacial tension does work along the biointerface area between epithelial monolayer and substrate matrix in order to reduce the interface area [
48]. The interfacial tension is therefore a key factor influencing the compression and extension of an epithelial monolayer as well as the surrounding matrix. When the epithelial monolayer undergoes extension during its movement, commonly referred to as cell wetting, this process induces compression of the underlying matrix [
48]. Conversely, when the monolayer experiences compression due to de-wetting, it results in the relaxation of matrix strain in the region of the cell-matrix biointerface, which can be characterized as an expansion. Compression of an epithelial monolayer is labelled with the sign “+”, whileits extension is labelledwith a “-“. The deviatoric part of cell normal residual stress is induced by collective cell migration. Consequently, the cell normal residual stress can be expressed as:
where the subscript
for the normal stress (
,
,
is the isotropic part of the cell normal stress equal to
,
is the normal vector of the cell-matrix biointerface, and
is the normal cell residual stress component caused by collective cell migration. The shear residual stress component accounts for two contributions. One contribution is induced by the interfacial tension gradient and described as natural convection, while the other contribution is caused by collective cell migration and described as forced convection [
48]. Consequently, the shear residual stress component is expressed as:
where the subscript
and
is the tangent vector of the epithelial-matrix biointerface. The first term of the right hand side represents the shear stress contribution caused by natural convection, while the second term represents the contribution caused by forced convection, i.e., collective cell migration.
Energy dissipation caused by cell rearrangement via collective cell migration is expressed here as:
where
is the energy dissipation caused by change of the interfacial tension, equal to
, such that the normal stress components satisfy the condition of isotropy, i.e.,
(eq. 3), while
is the shear stress component (eq. 4),
are strain components such that
and
are normal strain components equal to
, (
, and
is the shear strain component equal to
, (
and
),
are components of the epithelial displacement vector
, while the epithelial velocity represents the rate of change the epithelial displacement vector, i.e.
, and
is the energy dissipation caused by collective cell migration.
Energy dissipation
, as a product of the viscoelasticity of a multicellular system, depends on the regime of cell migration. Three regimes have been observed experimentally: convective, conductive (diffusion), and sub-diffusion depending on cell speed and cell packing density [
21,
22,
31,
53]. It is necessary to propose a suitable constitutive model of viscoelasticity for each regime and then to formulate this contribution to the energy dissipation.
3.2.2. Viscoelasticity of epithelial monolayers: the energy dissipation
Each regime of cell migration is related to a particular constitutive model of viscoelasticity described as shown in
Table 2.
Serra-Picamal et al. [
21] and Notbohm et al. [
31] considered the rearrangement of MDCK cell monolayers as a function of cell packing density
and revealed that the long-term cell stress (i.e., the cell residual stress) correlates with the corresponding strain, thereby highlighting the viscoelastic solid behaviour. It is known that epithelial cells establish strong E-cadherin mediated cell-cell adhesion contacts. An additional significant characteristic of epithelial monolayers, particularly relevant to this specific range of cell packing densities, is the ability of cell stress to relax towards the cell residual stress. Khalilgharibi et al. [
56] emphasized that stress relaxation time is on a time-scale of minutes, while the cell residual stress accumulation occurs on a time-scale of hours [
53]. According to the findings presented, Pajic-Lijakovic and Milivojevic [
53] proposed the scenario that cell stress change occurs through many short-time stress relaxation cycles, while cell strain (caused by collective cell migration) and the corresponding cell residual stress change over a time scale of hours. A suitable constitutive model should satisfy the conditions (1) that the stress relaxes exponentially on a time scale of minutes and (2) that the cell residual stress correlates with the corresponding strain, indicating the long-term elastic behaviour. This model is the Zener model, suitable for viscoelastic solids, as shown in
Table 2. In this case, energy dissipation, characteristic of the viscoelastic behaviour of multicellular systems, occurs on a time scale of minutes as a consequence of the remodelling of cell-cell adhesion contacts [
20]. The main characteristic of this regime is pronounced anisotropic behaviour of cell rearrangement. The consequence of this anisotropic viscoelasticity is that cell normal strains in the x- or y-directions contribute to generation of shear stress, while the shear strain also contributes to the generation of normal and shear stress components. Consequently, shear stress is generated within multicellular domains, as well as along the biointerface between neighbour domains. Serra-Picamal et al. [
21] and Tambe et al. [
57] revealed that maximum shear and normal, tensional stress components, generated during free expansion of epithelial monolayers, were about 150 Pa. Notbohm et al. [
31] considered the rearrangement of confluent epithelial monolayers and measured compressive stress up to 300 Pa.
An increase in cell packing density, caused by inhomogeneous wetting/de-wetting of migrating epithelial domains, additionally reduces cell movement and perturbs cell alignment as quantified by a decrease in the velocity correlation length. Garcia et al. [
33] revealed that a decrease in cell velocity from
to
results in a decrease in the velocity correlation length from
to
and consequently induces the disordering of cell movement. Intensive cell-cell interactions, caused by an increase in cell packing density, are responsive to the transition from convective-to-conductive mechanisms, and can be characterized by an isotropic, linear viscoelasticity. The main characteristic of conductive regime is that the cell stress cannot relax, while the energy dissipation occurs on a time scale of hours as a consequence of the increased number of cell re-polarisation events. The constitutive model that satisfies these conditions is the Kelvin-Voigt model [
53] (
Table 2).
An additional rise in cell packing density enhances interactions among cells, which disrupts their alignment and leads to significant orientational interactions between cells. These latter initiate the process of cell re-polarization. Nevertheless, the frequency of cell interactions occurs at a rate that is faster than the time required for re-polarization, resulting in a phenomenon known as jamming (
Box 2). In this regime of cell packing densities, collective cell migration occurs via a non-linear sub-diffusion mechanism. The disordering trend of cell migration, quantified by significant decrease in the velocity correlation length, points to isotropic viscoelasticity, while a non-linear mechanism of cell migration has been described by the Fractional constitutive model (
Table 2). The corresponding energy dissipation, caused by perturbation of cell alignment and cell contractile-to-non-contractile transitions in this regime, occurs on a time-scale of hours.
4. Physical parameters which govern cell rearrangement and entropy production
Interplay between three physical parameters such as: (i) the degree of cell orientation in the direction of cell migration, (ii) cell velocity and (iii) cell packing density influences the long-term generation of entropy during collective cell migration. Cell packing density and cell velocity changes will be discussed based on formulated force balance and mass balance equations, while the degree of cell orientation will be formulated based on thermodynamic phase model. All parameters are interconnected. Detail descriptions of these parameters follow in the next three sections.
4.1. The degree of cell orientation in the direction of cell migration
The degree of cell orientation in the direction of cell migration depends on cell-cell positional and orientational interactions (
Box 1) and the accumulated cell residual stress (eqs. 3-4). A change of this parameter can be expressed in the form of a modified phase model [
58,
59] as:
where
is the free energy per unit volume caused by orientational cell-cell interactions,
is the effective frictional coefficient among cells, and
is the cell residual stress accumulation expressed by eqs. 3-4 and
Table 2 for various cell packing density regimes. The free energy can be expressed in the form of a Landau-Ginsburg functional [
58] as:
(where the parameter
controls the relaxation of cell alignment, the parameter
controls the nearest neighbor alignment, and
is an increment of volume of the multicellular system). The accumulation of cell residual stress perturbs the cell alignment [
28]. The relaxation time
corresponds to the cell re-polarisation time and depends inversely on cell packing density, i.e.
. For cell packing densities near jamming
, the relaxation time tends to infinity, i.e.,
. In accordance with fact that the cell packing density significantly influences the relaxation time of cell alignment and the accumulation of cell residual stress, it is necessary to formulate the mass balance equation.
4.2. The mass balance
The change of cell packing density is induced by collective cell migration and the gradient of interfacial tension. Mass balance can be expressed as [
40,
53]:
where the cell packing density is
and the flux
describes the regime of cell movement (
Table 2). For the convective regime it is the convective flux
, while for the diffusion regime, it is the diffusion flux
(where
is the effective diffusion coefficient). For the sub-diffusion regime, the corresponding flux
represents the sub-diffusion flux
and the mass balance equation (eq. 7) should be formulated by including the fractional derivatives, i.e.
, where
is the fractional derivative expressed as
, and
is the order of the fractional derivative i.e. the damping coefficient of the cell rearrangement near jamming, which satisfies the condition
, the flux
, and
is the the damped-conductive diffusion coefficient which has units of
[
53].
The flux
is the Marangoni flux, which depends on the gradient of the epithelial-matrix interfacial tension and has been formulated as:
, where
quantifies the mobility of epithelial cells along the biointerface and
is the surface gradient [
48]. The Marangoni flux directs the movement of cells from the region of lower interfacial tension toward the region of higher interfacial tension. This phenomenon is observed in numerous soft matter systems, predominantly resulting from variations in temperature and concentration distributions [
44].
A change in cell packing density, caused by the generation of mechanical stress, influences the energy dissipation and entropy production both directly and indirectly by influencing physical parameters such as: the cell velocity and the order parameter. The inter-relationship between cell velocity and other relevant physical parameters will be discussed in the next section in the form of the force balance.
4.3. The force balance
Oscillations of the driving and resistive forces (
Box 3) induce successive establishment and perturbation of dynamical equilibrium, resulting in long-term oscillations in cell velocity. The phenomenon has been observed and discussed in the context of an effective inertia [
31,
40]. The driving forces are the interfacial tension force and the mixing force, whereas the viscoelastic force and traction force constitute the resistive forces. The volumetric force balance can be expressed as [
40]:
where
is the material derivative [
60],
, which satisfies the condition that
, is the average mass of a single cell,
is the mixing force,
is the interfacial tension force,
is the viscoelastic force, and
is the traction force.
The mixing force
arises from the thermodynamic energy changes associated with the mixing of two soft matter systems, specifically the cell monolayer and the substrate hydrogel matrix. This force has been formulated as:
, where
is the average size of single cell. The interfacial tension force
drives cell active/passive wetting and de-wetting. The phenomenon is contingent upon the relationship between the tissue and matrix surface tensions accompanied by the interfacial tension between them. This relationship can be quantitatively represented by the cell spreading factor [
40]. This force was formulated as:
, where
is the spreading factor of epithelial cells and
is the epithelial displacement field [
40]. The spreading factor for free expansion of cell monolayers satisfies the criteria that
and that change with time causes oscillatory wetting [
48].
The traction force is the resistive force formulated by Murray et al. [
45] as
(where
is the number density of cell-matrix adhesion contacts,
is the spring constant of a single cell-matrix adhesion contact, and
is the local displacement of the matrix caused by cell tractions). The establishment of strong cell-matrix adhesion contacts has been shown to diminish cellular motility [
61]. The viscoelastic force as formulated by Murray et al. [
45] was expressed as:
(where
is the cell residual stress and
is the residual stress within a substrate matrix caused by cell tractions). The residual stress within a substrate matrix depends on the matrix viscoelasticity and cell tractions. Cell tractions also influence the matrix surface tension and epithelial-matrix interfacial tension.
The scenario for the oscillatory variation of the velocity is outlined as follows:
An inhomogeneous accumulation of cell compressive residual stress within the epithelial monolayer caused by collective cell migration leads to an increase in the viscoelastic force [40]. The viscoelastic force and traction force causes a decrease in the velocity of epithelial cells and an increase in the epithelial packing density. An induced decrease in the velocity causes a decrease in the traction force [
48].
An increase in the epithelial packing density leads to intensive cell-cell interactions, resulting in energy dissipation within the epithelial monolayer and, consequently, decreases in: (1) the residual stress accumulation within the epithelium; and (2) the epithelial surface tension.
A reduction in the residual stress present in the epithelial monolayer leads to a decrease in viscoelastic force. As a result, the suppression of both resistive forces, namely the viscoelastic and traction forces, contributes to an increase once more in the velocity of the epithelial cells.
Consequently, oscillations of cell velocity are thus a result of variations in energy dissipation alongside the generation of entropy.
In addition to examining the long-term dynamics of internal entropy generation, it is essential to address the short-term dynamics of entropy generation within a multicellular domain that is typical of a convective regime.
5. Short-term dynamics of entropy generation associated with the convective regime
The short-term dynamics of entropy generation occurs within many successive short-term relaxation cycles and is related to the cumulative effects of the remodeling of cell-cell and cell-matrix adhesion contacts and cell shape changes caused by cell contractions [
41]. The average internal entropy of the m-th domain established after single relaxation cycle can be expressed [
62] as:
where
is the number of cells per m-th domain and
is the Boltzmann probability equal to
(where
is the canonical partition function,
is the mechanical energy of a single-cell expressed as:
, while
are the degrees of freedom (DOFs), which describe the phase-space of a single-cell. Cell activity can be simplified by using a few DOFs such as: (i) the cell contractility dimensionless concentration of phosphorylated myosin
(where
is the equilibrium myosin concentration) [
31]; (ii) the dimensionless number density of E-cadherin molecules per single cell
(where
is the equilibrium number density of cadherin molecules per single cell); (iii) the dimensionless number density of integrin molecules bonded to ligands of extracellular matrix per single cell
(where
is the equilibrium number density of integrin molecules per single cell); and (iv) the dimensionless concentration of signaling molecules in the cell surrounding
(where
is the equilibrium concentration of the i-th signaling molecule), which influences the single-cell state and ordering within a domain [
63,
64].
A short-time change of the DOFs can be expressed in the form of a system of Langevin-type equations [
41] as:
where the stochastic random force
is formulated as a white noise with correlation function
while
serves as the equivalent of frictional resistance.
The current equilibrium state after a single short-time relaxation cycle per domain for the time set
can be expressed by the canonical partition function
as:
where integration goes over all dimensionless degrees of freedom (DOFs)
of all cells in the domain and
is the energetic function equal to
,
is the average mechanical energy of single-cell, and
.
Consequently, the short-term energy production occurs during the stress relaxation cycles within the convective regime.
6. Conclusions
Our theoretical investigation reveals that cells can effectively control entropy generation linked to the dissipation of mechanical energy during collective migration. This collective movement of cells triggers both active and passive wetting and de-wetting on the substrate matrix, leading to an accumulation of mechanical stress within epithelial monolayers. This mechanical stress exerts a feedback influence on various parameters, including cell packing density, alignment, and velocity. Notably, an increase in compressive stress results in an increase in cell packing density, while shear stress can create topological defects in cell alignment, which in turn amplify cell-cell interactions. The principal outcomes of this study were derived from the synthesis of physical models and experimental data in the domains of mechanobiology and biological physics. We can summarize them as follows:
Cell-cell interactions cause remodeling of cell-cell and cell matrix adhesion contacts and can induce re-polarisation of cells accompanied by a weakening of adhesion contacts. These interactions are pivotal in the dynamic alteration of energy dissipation within cells, which is associated with the viscoelastic properties and surface characteristics of multicellular systems, ultimately contributing to entropy generation. The regulation of energy dissipation serves as a cellular mechanism to decrease mechanical stress in epithelial monolayers, thereby inhibiting cell migration.
Energy dissipation caused by the remodeling of cell-cell and cell-matrix adhesion contacts during relaxation of migrating epithelial collectives under mechanical stress occurs on a time scale of minutes. This phenomenon of entropy production is indicative of a convective regime. The continuous remodeling of adhesion contacts is a fundamental aspect of the cellular adaptation to varying microenvironmental conditions and occurs repeatedly throughout the process of collective cell migration.
Contact inhibition of locomotion, which is pronounced in an overcrowded environment characterized by an increase in cell packing density in a conductive diffusion regime, leads to energy dissipation during the process of cell re-polarization. This re-polarization, accompanied by a weakening of cell-cell and cell-matrix adhesion contacts, occurs over a time frame of several hours. When cells are afforded sufficient time to complete the re-polarization process and re-establish robust adhesion contacts with both neighboring cells and the extracellular matrix, they initiate movement in the opposite direction.
When the interval between two collisions is less than the duration required for the re-polarization process, which is typical of the sub-diffusion regime, cells experience jamming. This phenomenon results in a transition from a contractile to a non-contractile state, leading to prolonged energy dissipation and a reduction in mechanical stress. The latter facilitates the formation of robust adhesion contacts between cells and the extracellular matrix, subsequently initiating collective migration once more.
This oscillations of energy storage and energy dissipation cause by collective cell migration result in oscillations in the production of entropy.
Author Contributions
IPL writing-review and editing, MM preparing images. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Ministry of Science, Technological Development and Innovation of the Republic of Serbia (Contract No. 451-03-136/2025-03/200135).
Institutional Review Board Statement
Not applicable.
Data Availability Statement
No new data were created.
Acknowledgments
We thank Peter McClintock (Lancaster University) for discussions which inspired this work and for useful comments that helped us to finalize the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Dugdale, J.S. Entropy and Low Temperature Physics; Hutchinson: London, 1966. [Google Scholar]
- Prigogine, I. Introduction to thermodynamics of irreversible processes, 3rd ed.; John Wiley & Sons: New York, London, Sydney, 1967. [Google Scholar]
- Kleidon, A.; Lorentz, R.D. Non-equilibrium thermodynamics and the production of entropy, Life, Earth, and Beyond; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2005. [Google Scholar]
- Wu, K.; Nan, Q.; Wu, T. Philosophical analysis of the meaning and nature of entropy and negative entropy theories. Complexity 2003, 8769060. [Google Scholar] [CrossRef]
- Ding, C. and Luo, L. Measurement of entropy production in living cells under an alternating electric field. Cell Biol. Int. 2013, 37, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Himeoka, Y. and Kaneko, K. Entropy production of a steady-growth cell with catalytic reactions. Phys. Rev. E 2014, 90, 042714. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.D.; Oloyede, A.; Singh, S.; Gu, Y. Investigation of the effects of extracellular osmotic pressure on morphology and mechanical properties of individual chondrocyte. Cell Biochem. Biophys. 2016, 74, 229–240. [Google Scholar] [CrossRef]
- McClintock, P.V.E. What is life? Contemp. Phys. 2012, 53, 433–435. [Google Scholar] [CrossRef]
- Mayer, C. Life in The Context of Order and Complexity. Life 2020, 10, 5. [Google Scholar] [CrossRef]
- Arango-Restrepo, A. and Rubi, J.M. Predicting cancer stages from tissue energy dissipation. Sci. Rep. 2023, 13, 15894. [Google Scholar] [CrossRef]
- Pelicano, H.; Martin, D.S.; Xu, R.H.; Huang, P. Glycolysis inhibition for anticancer treatment. Oncogene 2006, 25, 4633–4646. [Google Scholar] [CrossRef]
- Brücher, B.L.D.M.; Jamall, I.S. Cell-Cell Communication in the tumor microenvironment, carcinogenesis, and anticancer treatment. Cell Physiol. Biochem. 2014, 34, 213–243. [Google Scholar] [CrossRef]
- Mui, K.L.; Chen, C.S.; Assoian, R.K. The mechanical regulation of integrin–cadherin crosstalk organizes cells, signalling and forces. J. Cell Sci. 2016, 1–8. [Google Scholar]
- Barriga, E.H. and Mayor, R. Adjustable viscoelasticity allows for efficient collective cell migration. Sem. Cell Dev. Biol. 2019, 93, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Iyer, K.V.; Gomez, R.P.; Paijmans, J.; Julicher, F.; Eaton, S. Epithelial viscoelasticity is regulated by mechanosensitive E-cadherin turnover. Curr. Biol. 2019, 29, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Alert, R.; Trepat, X. Physical models of collective cell migration. Annu. Rev. Condens. Matter Phys. 2020, 11, 77–101. [Google Scholar] [CrossRef]
- Petrolli, V.; Boudou, T.; Balland, M.; Cappello, G. Oscillations in collective cell migration, in Viscoelasticity and collective cell migration: An interdisciplinary perspective across levels of organization; Pajic-Lijakovic, I., Barriga, E., Eds.; Academic Press: Cambridge, MA, USA; 2021. [Google Scholar]
- Pajic-Lijakovic, I.; Milivojevic, M.; McClintock, P.V.E. Physics of cancer spreading through epithelium. Contemp. Phys. 2024, 296–314. [Google Scholar] [CrossRef]
- Pajic-Lijakovic, I.; Milivojevic, M.; McClintock, P.V.E. Epithelial cell-cell interactions in an overcrowded environment: Jamming or live cell extrusion. J. Biol. Eng. 2024, 18. [Google Scholar] [CrossRef]
- Pajic-Lijakovic, I.; Milivojevic, M.; McClintock, P.V.E. Physical aspects of epithelial cell-cell interactions: Hidden system complexities. Europ. Biophys. J. 2024, 53, 355–372. [Google Scholar] [CrossRef]
- Serra-Picamal, X.; Conte, V.; Vincent, R.; Anon, E.; Tambe, D.T.; Bazellieres, E.; Butler, J.P.; Fredberg, J.J.; Trepat, X. Mechanical waves during tissue expansion. Nat. Phys. 2012, 8, 628–634. [Google Scholar] [CrossRef]
- Nnetu, K.D.; Knorr, M.; Pawlizak, S.; Fuhs, T.; Kaes, J. Slow and anomalous dynamics of an MCF-10A epithelial cell monolayer. Soft Matter 2013, 9, 9335–9341. [Google Scholar] [CrossRef]
- Tlili, S.; Gauquelin, E.; Li, B.; Cardoso, O.; Ladoux, B.; Delanoë-Ayari, H.; Graner, F. Collective cell migration without proliferation: Density determines cell velocity and wave velocity. R. Soc. Open Sci. 2018, 5, 172421. [Google Scholar] [CrossRef]
- Kang, W.; Ferruzzi, J.; Spatarelu, C.-P.; Han, Y.L.; Sharma, Y.; Koehler, S.A.; Mitchel, J.A.; Khan, A.; Butler, J.P.; Roblyer, D.; et al. A novel jamming phase diagram links tumor invasion to non-equilibrium phase separation. iSci 2021, 24, 103252. [Google Scholar] [CrossRef]
- Lin, S.Z.; Ye, S.; Xu, G.K.; Li, B.; Feng, X.Q. Dynamic migration modes of collective cells. Biophys. J. 2018, 115, 1826–1835. [Google Scholar] [CrossRef] [PubMed]
- Deforet, M.; Hakim, V.; Yevick, H.; Duclos, G.; Silberzan, P. Emergence of collective modes and tri-dimensional structures from epithelial confinement. Nat. Commun. 2014, 5, 3747. [Google Scholar] [CrossRef] [PubMed]
- Lee, P. and Wolgemuth, C.W. Crawling cells can close wounds without purse strings or signalling. PLoS Comp. Biol. 2011, 7, e1002007. [Google Scholar] [CrossRef] [PubMed]
- Saw, T.B.; Doostmohammadi, A.; Nier, V.; Kocgozlu, L.; Thampi, S.; Toyama, Y.; Marcq, P.; Lim, C.T.; Yeomans, J.M.; Ladoux, B. Topological defects in epithelia govern cell death and extrusion. Nature 2017, 544, 212–216. [Google Scholar] [CrossRef]
- Roycroft, A.; Mayor, R. Molecular basis of contact inhibition of locomotion. Cell. Mol. Life Sci. 2016, 73, 1119–1130. [Google Scholar] [CrossRef]
- Zimmermann, J.; Camley, B.A.; Rappel, W.J.; Herbert, L.H. Contact inhibition of locomotion determines cell–cell and cell–substrate forces in tissues. PNAS 2016, 113, 2660–2665. [Google Scholar] [CrossRef]
- Notbohm, J.; Banerjee, S.; Utuje, K.J.C.; Gweon, B.; Jang, H.; Park, Y.; Shin, J.; Butler, J.P.; Fredberg, J.J.; Marchetti, M.C. Cellular contraction and polarization drive collective cellular motion. Biophys. J. 2016, 110, 2729–2738. [Google Scholar] [CrossRef]
- Petitjean, L.; Reffay, M.; Grasland-Mongrain, E.; Poujade, M.; Ladoux, B.; Buguin, A.; Silberzan, P. Velocity fields in a collectively migrating epithelium. Biophys. J. 2010, 98, 1790–1800. [Google Scholar] [CrossRef]
- Garcia, S.; Hannezo, E.; Elgeti, J.; Joanny, J.-F.; Silberzan, P.; Gov, N.S. Physics of active jamming during collective cellular motion in a monolayer. PNAS 2015, 112, 15314–15319. [Google Scholar] [CrossRef]
- Pajic-Lijakovic, I. and Milivojevic, M. The role of viscoelasticity in long time cell rearrangement. Progr. Biophys. Mol. Boil. 2022, 173, 60–71. [Google Scholar] [CrossRef]
- Schulze, K.D.; Zehnder, S.M.; Urueña, J.M.; Bhattacharjee, T.; Sawyer, W.G.; Angelini, T.E. Elastic modulus and hydraulic permeability of MDCK monolayers. J. Biomech. 2017, 53, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.-C.C.; Eaton, M.J.; Karasek, M.A. Growth characteristics of human epidermal kerationcytes from newborn foreskin in primary and serial cultures. In Vitro 1979, 15, 813–822. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.J.; Thompson, D.P.; Cramer, C.T.; Vidmar, T.J.; Scieszka, J.F. The Madin Darby canine kidney (MDCK) epithelial cell monolayer as a model cellular transport barrier. Pharm. Res. 1989, 6, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Adak, A.; Unal, Y.C.; Yucel, S.; Vural, Z.; Turan, F.B.; Yalcin-Ozuysal, O.; Ozcivici, E.; Mese, G. Connexin 32 induces pro-tumorigenic features in MCF10A normal breast cells and MDA-MB-231 metastatic breast cancer cells. Biochim. Biophys. Acta Mol. Cell Res. 2020, 1867, 118851. [Google Scholar] [CrossRef]
- Volfson, D.; Cookson, S.; Hasty, J.; Tsimring, L.S. Biomechanical ordering of dense cell populations. PNAS 2008, 105, 15346–15351. [Google Scholar] [CrossRef]
- Pajic-Lijakovic, I.; Milivojevic, M.; McClintock, P.V.E. Role of viscoelasticity in the appearance of low-Reynolds turbulence: Considerations for modelling. J. Biol. Eng. 2024, 18, 24. [Google Scholar] [CrossRef]
- Pajic-Lijakovic, I. and Milivojevic, M. Multiscale nature of cell rearrangement caused by collective cell migration. Europ. Biophys. J. 2021, 50, 1–14. [Google Scholar] [CrossRef]
- Ohsawa, S.; Vaughen, J.; Igaki, T. Cell Extrusion: A Stress-Responsive Force for Good or Evil in Epithelial Homeostasis. Develop. Cell 2018, 44, 284–296. [Google Scholar] [CrossRef]
- Pérez-González, C.; Alert, R.; Blanch-Mercader, C.; Gómez-González, M.; Kolodziej, T.; Bazellieres, E.; Casademunt, J.; Trepat, X. Active wetting of epithelial tissues. Nat. Phys. 2019, 15, 79–88. [Google Scholar] [CrossRef]
- Karbalaei, A.; Kumar, R.; Cho, H.J. Thermocapillarity in microfluidics—A review. Micromachines 2016, 7, 13. [Google Scholar] [CrossRef]
- Murray, J.D.; Maini, P.K.; Tranquillo, R.T. Mechanochemical models for generating biological pattern and form in development. Phys. Rep. 1988, 171, 59–84. [Google Scholar] [CrossRef]
- Stirbat, T.V.; Mgharbel, A.; Bodennec, S.; Ferri, K.; Mertani, H.C.; Rieu, J.P.; Delanoe-Ayari, H. Fine tuning of tissues’ viscosity and surface tension through contractility suggests a new role for a-Catenin. PLoS ONE 2013, 8, e52554. [Google Scholar] [CrossRef] [PubMed]
- Koride, S.; Loza, A.J.; Sun, S.X. Epithelial vertex models with active biochemical regulation of contractility can explain organized collective cell motility. APL Bioeng. 2018, 2, 031906. [Google Scholar] [CrossRef] [PubMed]
- Pajic-Lijakovic, I. and Milivojevic, M. Physics of collective cell migration. Europ. Biophys. J. 2023, 52, 625–640. [Google Scholar] [CrossRef]
- Gsell, S.; Tlili, S.; Merkel, M.; Lenne, P.-F. Marangoni-like tissue flows enhance symmetry breaking of embryonic organoids. bioRxiv 2023. [Google Scholar] [CrossRef]
- Devanny, A.J.; Vancura, M.B.; Kaufman, LJ. Exploiting differential effects of actomyosin contractility to control cell sorting among breast cancer cells. Mol. Biol. Cell 2021, 32, ar24. [Google Scholar] [CrossRef]
- Guevorkian, K.; Brochard-Wyart, F.; Gonzalez-Rodriguez, D. Flow dynamics of 3D multicellular systems into capillaries. In Viscoelasticity and collective cell migration: An interdisciplinary perspective across levels of organizatio; Pajic-Lijakovic, I., Barriga, E., Eds.; Academic Press: Cambridge, MA, USA, 2021; p. 193. [Google Scholar]
- Clark, A.G.; Maitra, A.; Jacques, C.; Bergert, M.; Pérez-González, C.; Simon, A.; Lederer, L.; Diz-Muñoz, A.; Trepat, X.; Voituriez, R.; et al. Self-generated gradients steer collective migration on viscoelastic collagen networks. Nat. Mat. 2022, 21, 1200–1210. [Google Scholar] [CrossRef]
- Pajic-Lijakovic, I. and Milivojevic, M. Viscoelasticity and cell jamming state transition. Europ. Phys. J. Plus 2021, 136, 750. [Google Scholar] [CrossRef]
- Colinas-Armijo, N.; DiPaola, M.; Pinnola, F.P. Fractional characteristic times and dissipated energy in fractional linear viscoelasticity. Commun. Nonlinear Sci. Numer. Simulat. 2016, 37, 14–30. [Google Scholar] [CrossRef]
- Podlubny, I. Fractional Differential Equations, Mathematics in Science and Engineering; Academic Press: London, UK, 1999; Volume 198, p. 78. [Google Scholar]
- Khalilgharibi, N.; Fouchard, J.; Asadipour, N.; Yonis, A.; Harris, A.; Mosaff, P.; Fujita, Y.; Kabla, A.; Baum, B.; Muñoz, J.J.; et al. Stress relaxation in epithelial monolayers is controlled by actomyosin. Nat. Phys. 2019, 15, 839–847. [Google Scholar] [CrossRef]
- Tambe, D.T.; Croutelle, U.; Trepat, X.; Park, C.Y.; Kim, J.H.; Millet, E.; Butler, J.P.; Fredberg, J.J. monolayer stress microscopy: Limitations, artifacts, and accuracy of recovered intercellular stresses. PLoS ONE 2013, 8, e55172. [Google Scholar] [CrossRef] [PubMed]
- Ala-Nissila, T.; Majaniemi, K.; Elder, K. Phase-field modeling of dynamical interface phenomena in fluids, Lecture Notes in Physics 640; Springer: Berlin/Heidelberg, Germany, 2004; pp. 357–388. [Google Scholar]
- Tanaka, H. A viscoelastic model of phase separation. Phys. Rev. 1997, 56, 4451–4462. [Google Scholar] [CrossRef]
- Bird, R.B.; Stewart, W.E.; Lightfoot, E.N. Transport Phenomena; John Wiley & Sons Inc.: Cambridge, MA, USA, 1960. [Google Scholar]
- Fuhrmann, A.; Banisadr, A.; Beri, P.; Tlsty, T.D.; Engler, A.J. Metastatic state of cancer cells may be indicated by adhesion strength. Biophys. J. 2017, 112, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Bent, H.A. The second law: An introduction to classical and statistical thermodynamics; Oxford University Press: Oxford, UK, 1965. [Google Scholar]
- Blanchard, G.B.; Fletcher, A.G.; Schumacher, LJ. The devil is in the mesoscale: Mechanical and behavioural heterogeneity in collective cell movement. Sem. Cell Dev. Biol. 2019, 93, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Petrungaro, G.; Morelli, L.; Uriu, K. Information flow in the presence of cell mixing and signaling delays during embryonic development. Sem. Cell Dev. Biol. 2019, 93, 26–35. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).