Submitted:
03 March 2025
Posted:
04 March 2025
You are already at the latest version
Abstract
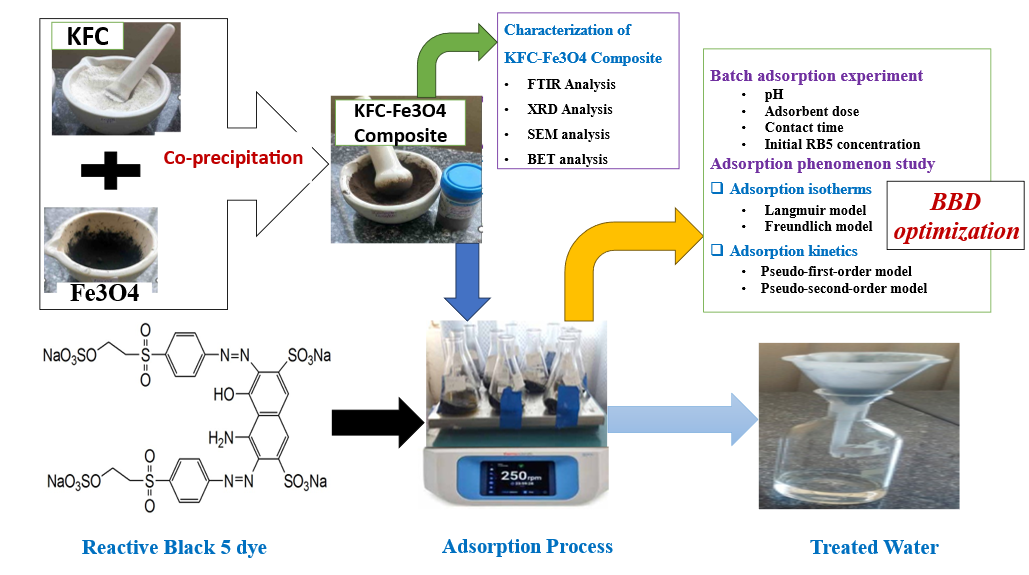
Keywords:
Graphical abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Preparation and Activation of Kaolin Filter Cake (KFC)
2.3. Synthesis of KFC-Fe3O4 Nanocomposite
2.4. Characterization of Synthesized KFC-Fe3O4 NCs
2.5. Point of Zero Charge (pHpzc)
2.6. Batch Adsorption Experiment
2.7. Experimental Design
2.8. Adsorption Isotherm and Kinetic Models
3. Results and Discussion
3.1. Characterization of KFC-Fe3O4 Composite
3.2. Effect of Different Adsorption Parameters
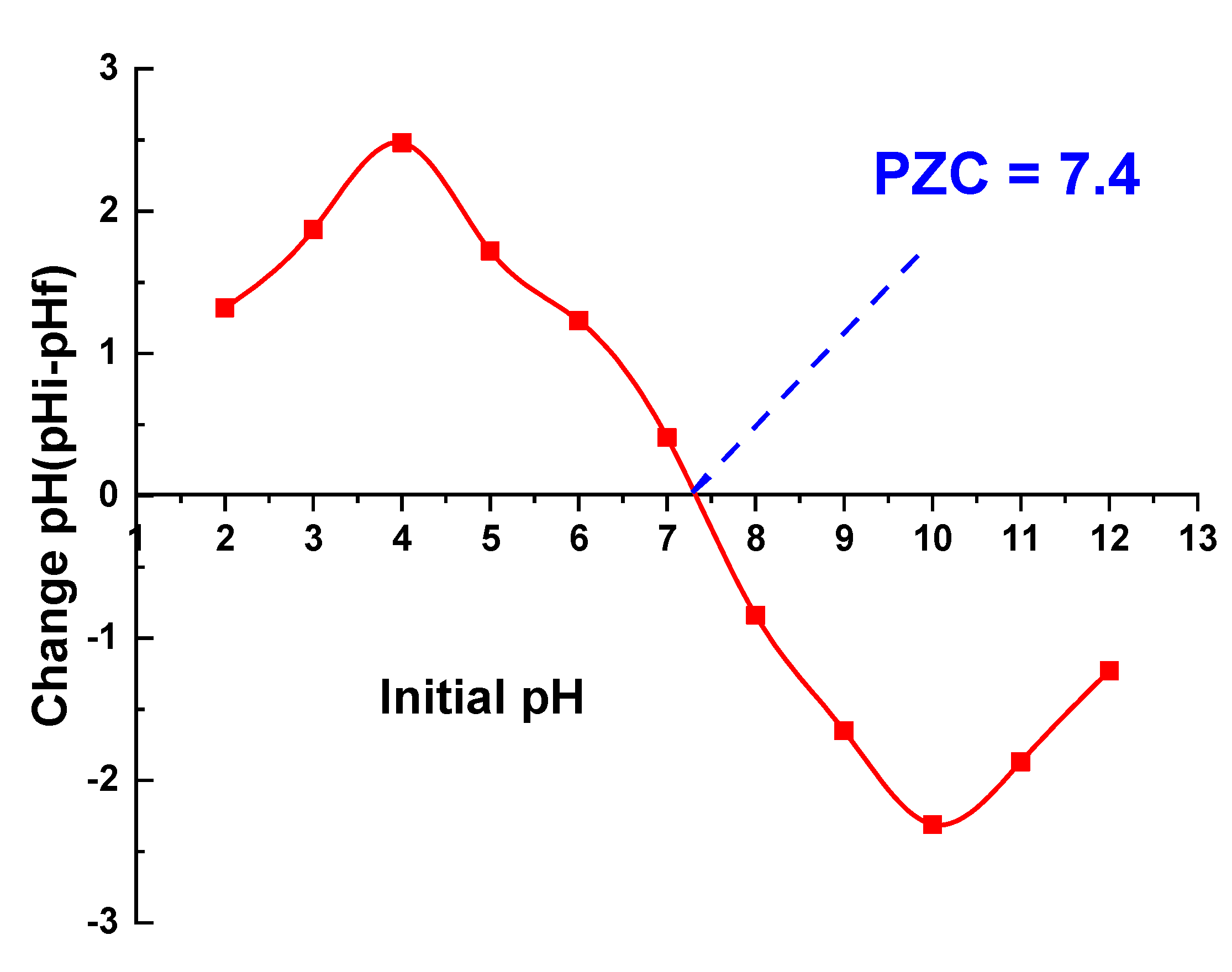
3.2.1. Effect of pH
3.2.2. Effect of Adsorbent Dose
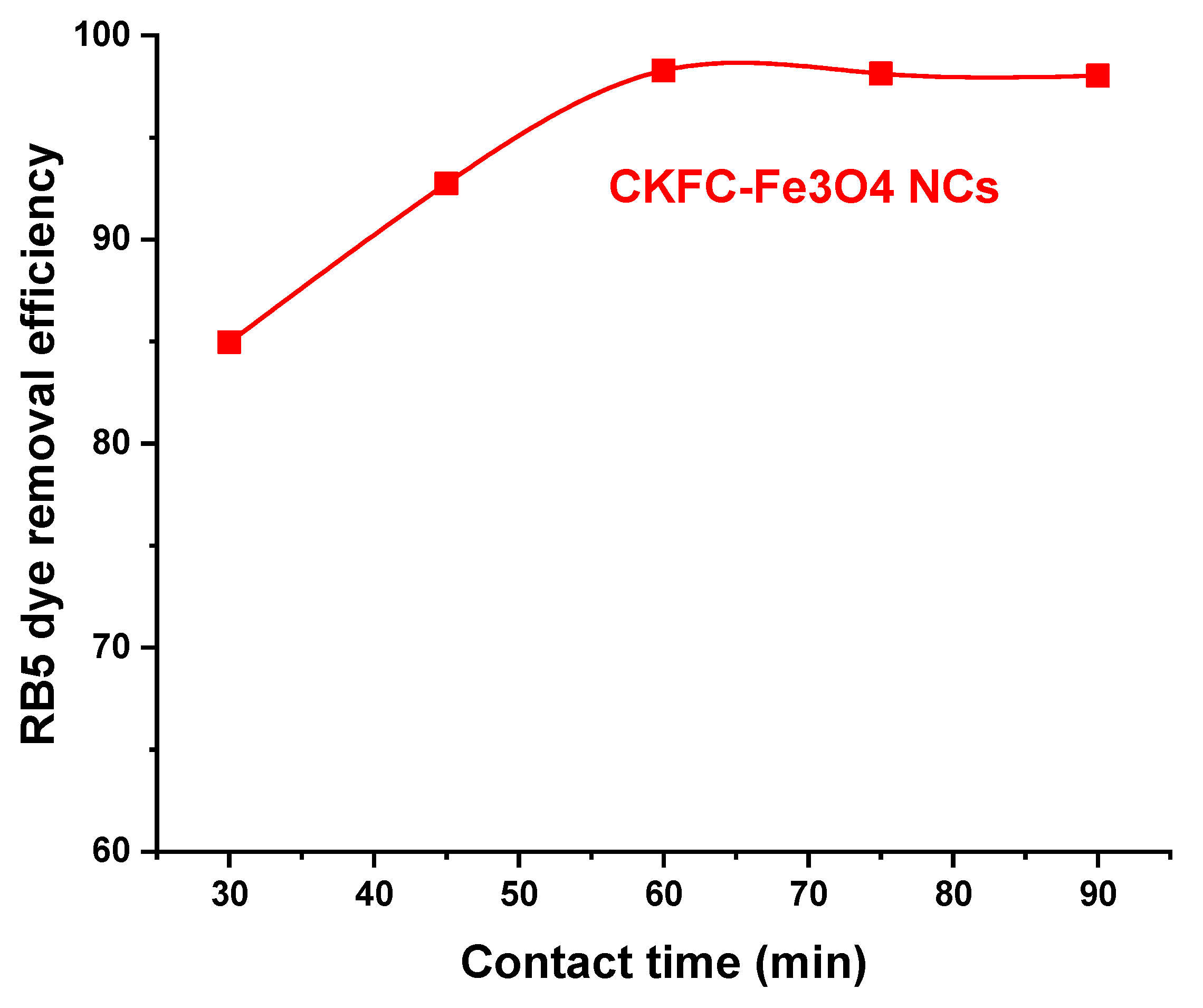
3.2.3. Effect of Contact Time
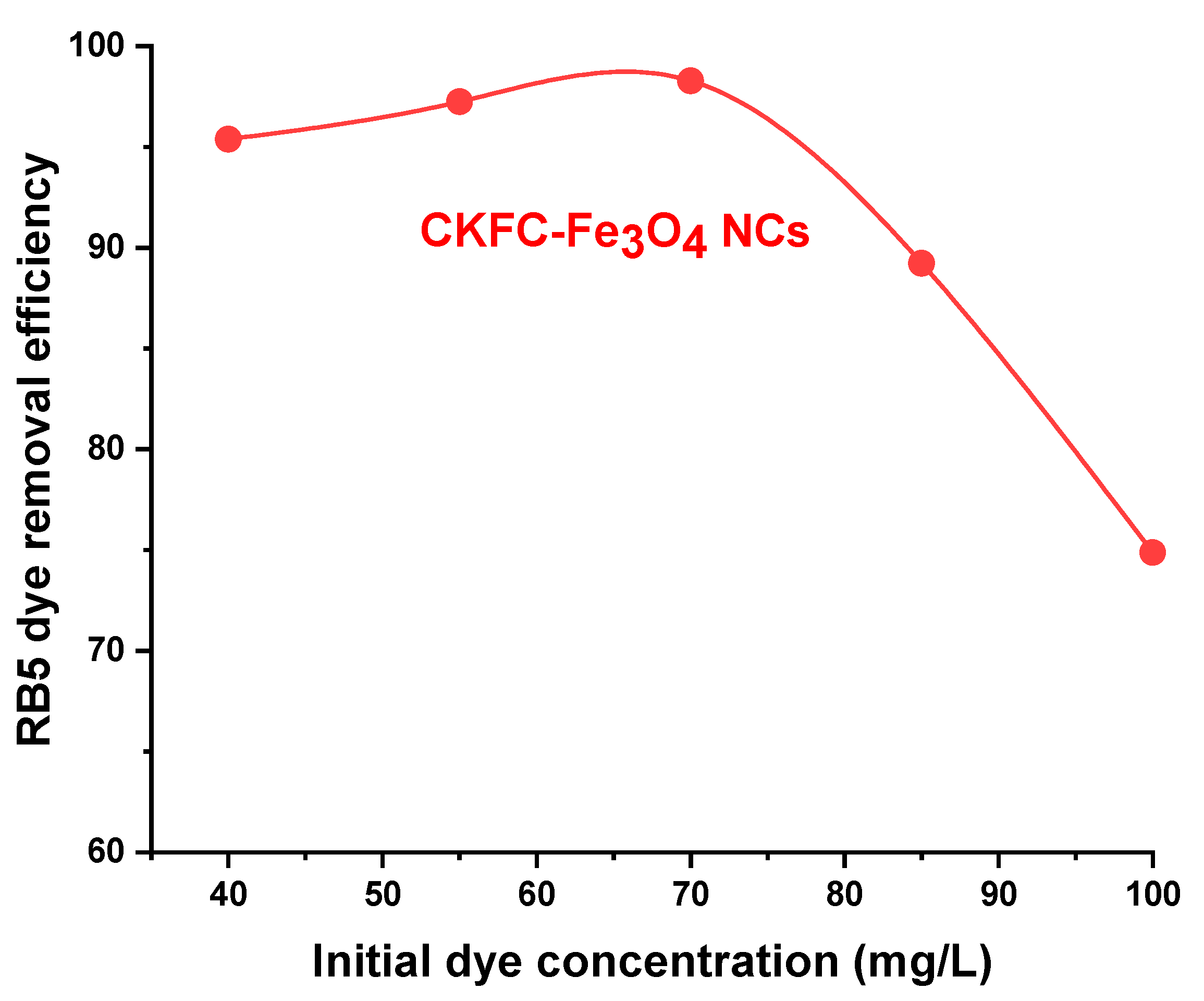
3.2.4. Effect Initial RB5 Dye Concentration
3.3. Box-Behnken Design (BBD) Analysis
3.3.1. Significance of Model Terms
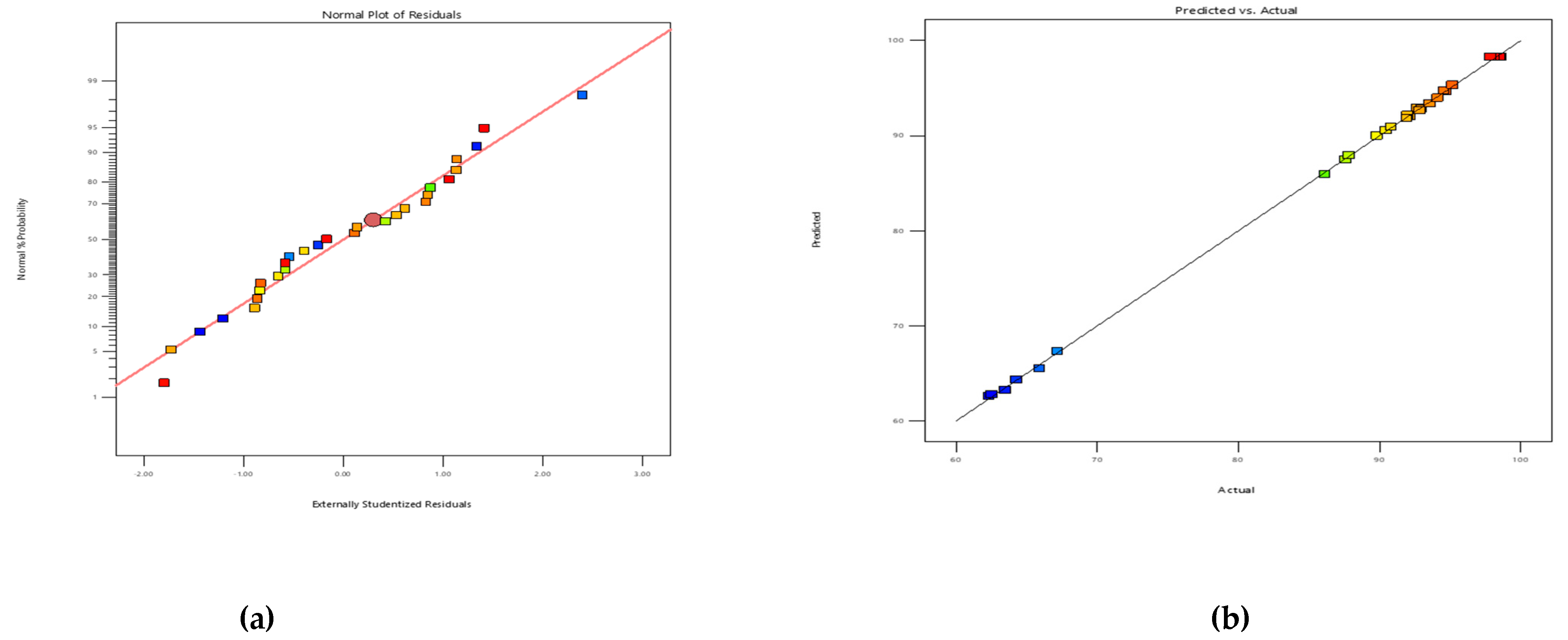
3.3.2. Fit Summary
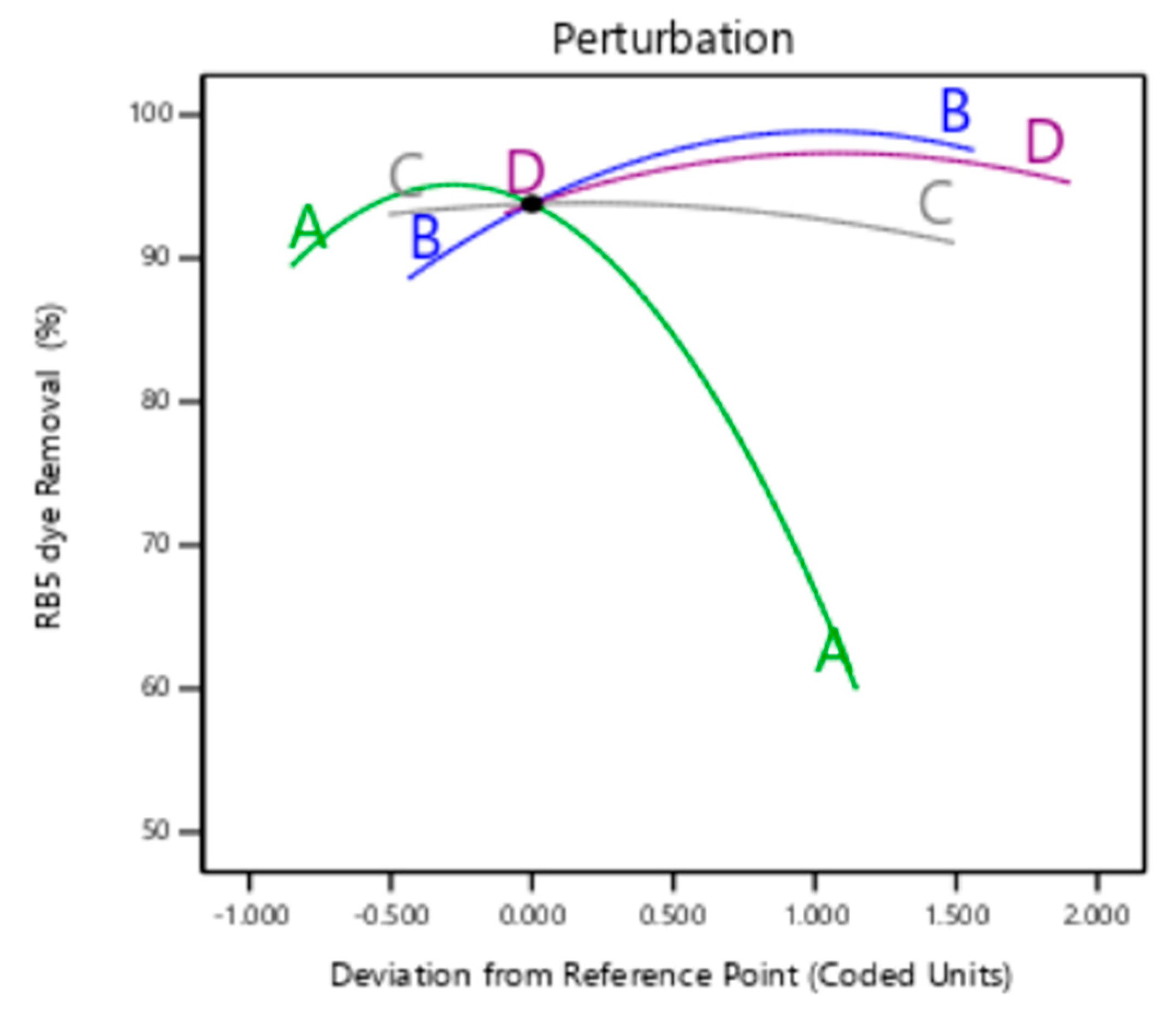
3.3.3. Perturbation Plot
3.4. Process Parameter Optimization for RB5 Removal
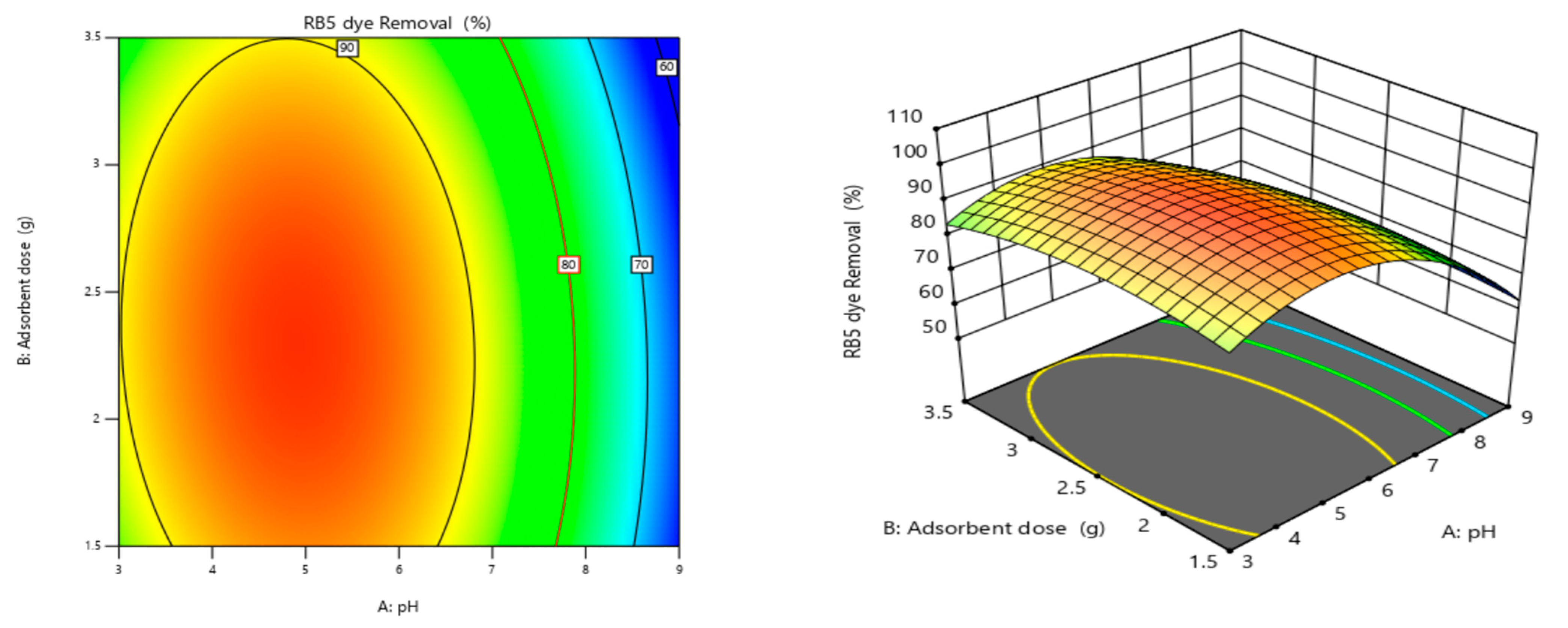
3.4.1. pH and Adsorbent Dose
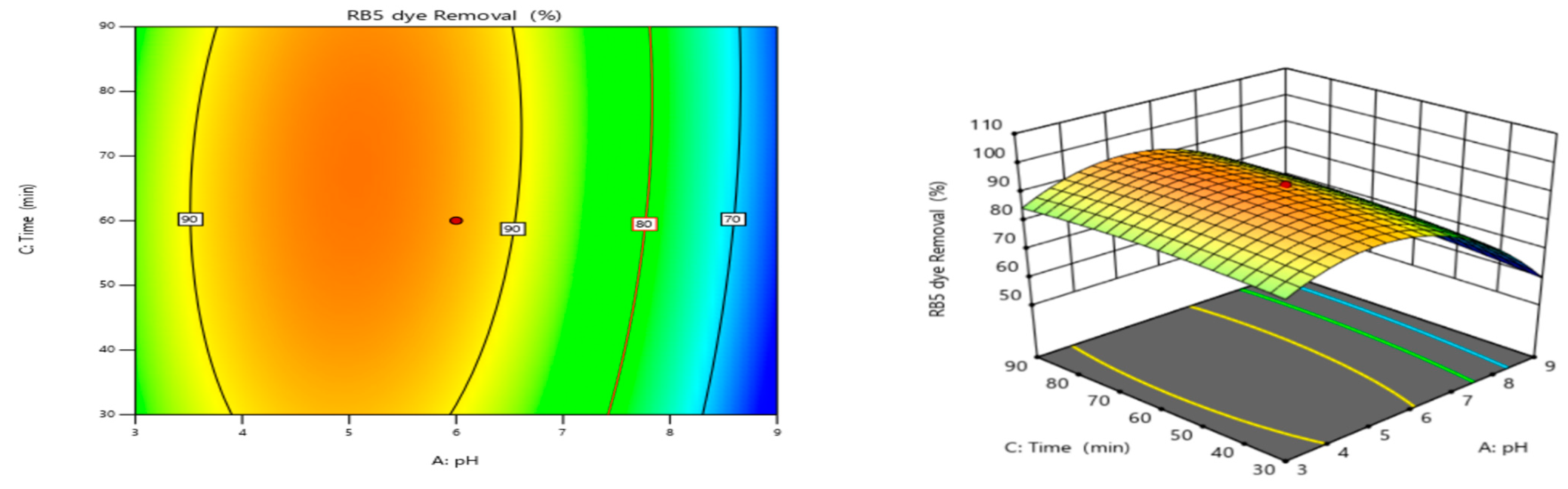
3.4.2. pH and Contact Time
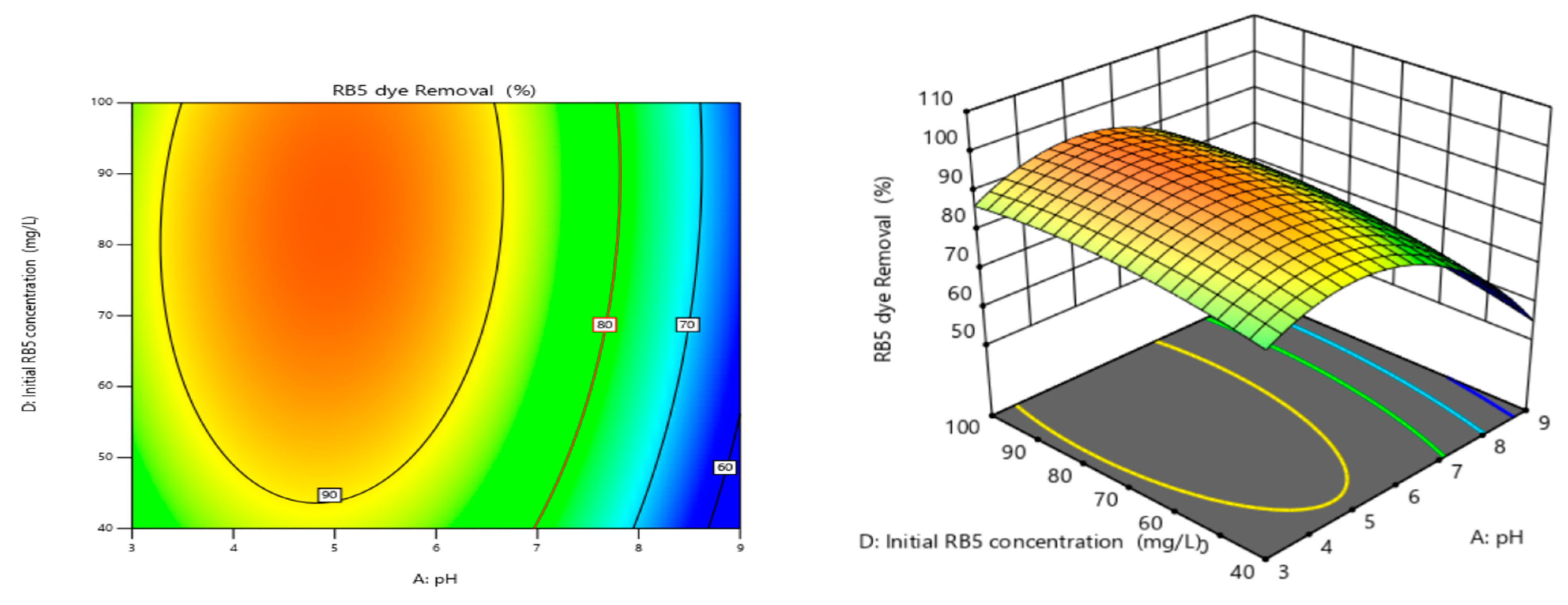
3.4.3. pH and Initial RB5 Concentration
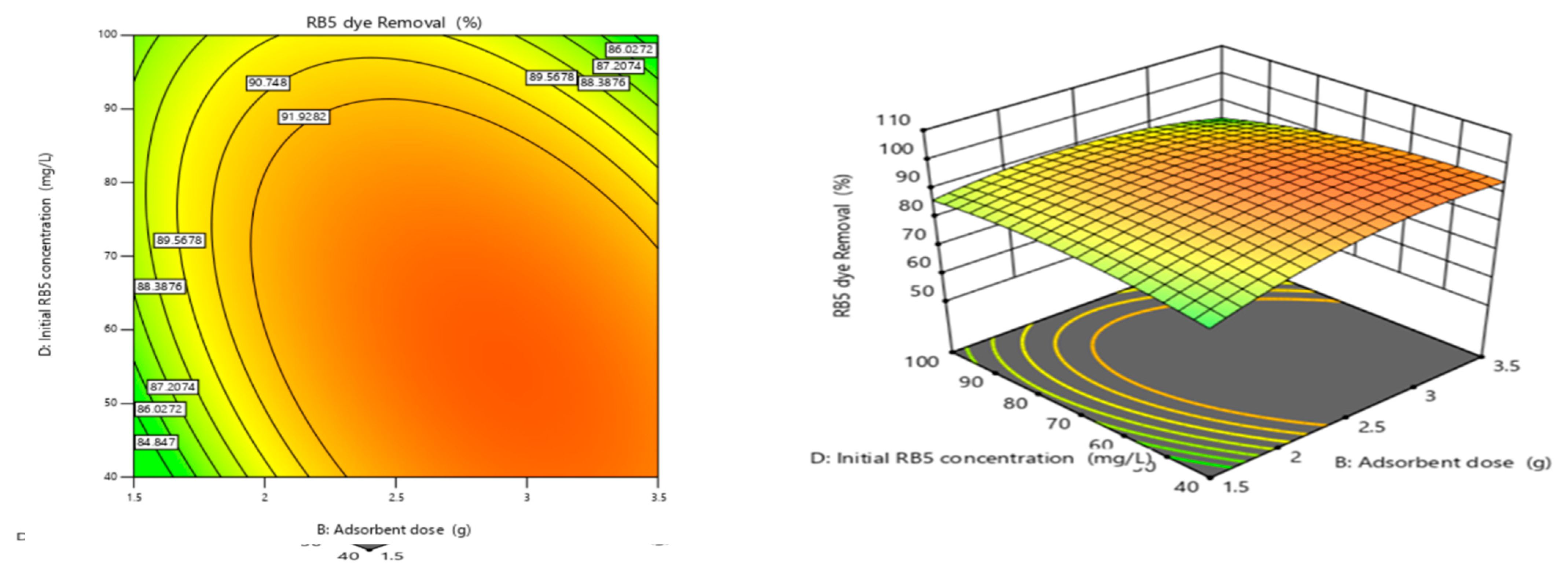
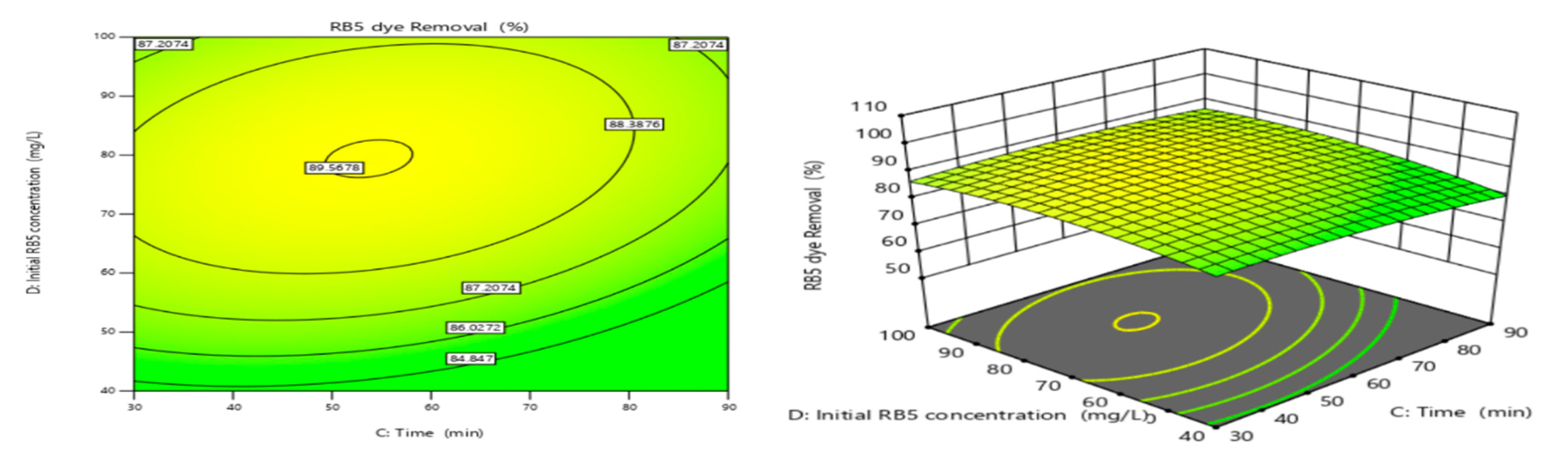
3.4.4. Adsorbent Dose and Initial RB5 Concentration
3.4.5. Contact Time and Initial RB5 Concentration
3.5. Kinetic, Equilibrium and Thermodynamic Studies
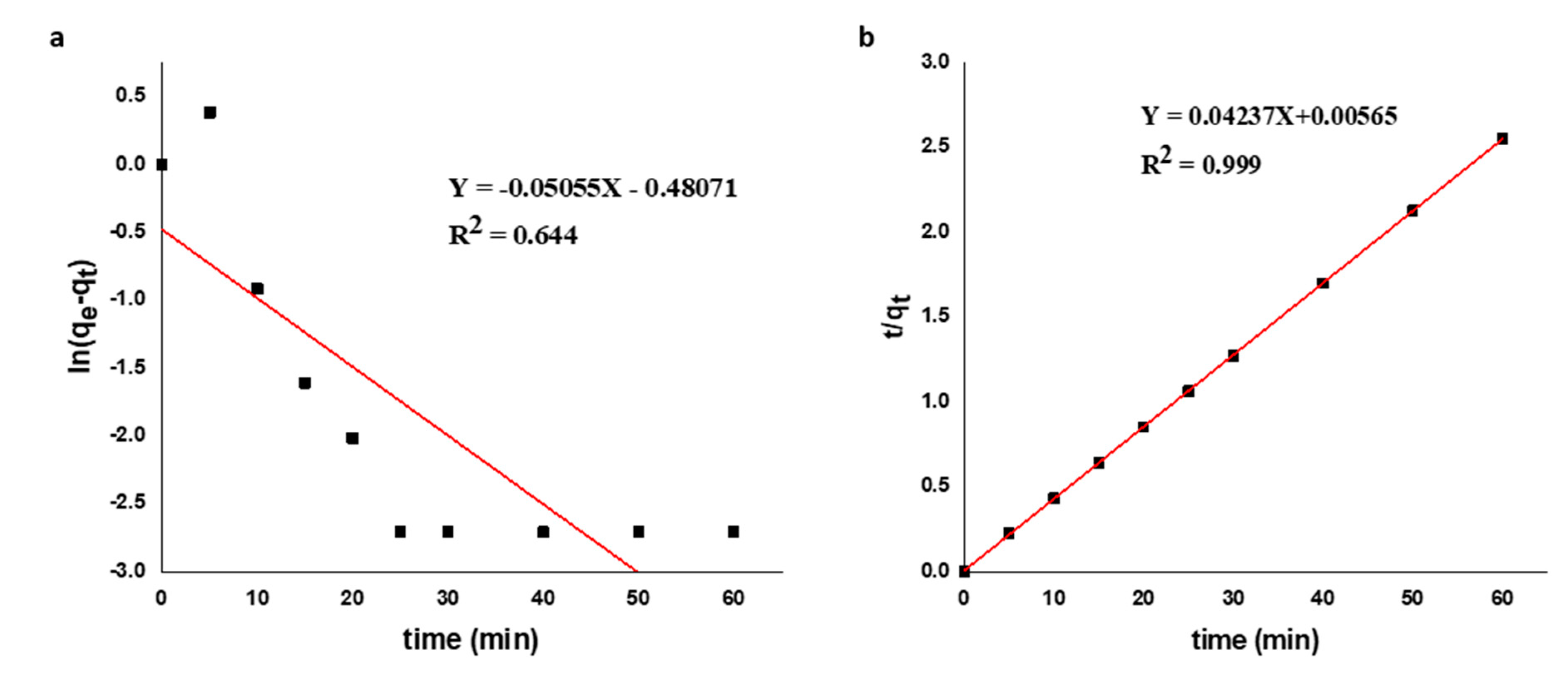
3.5.1. Adsorption Kinetics
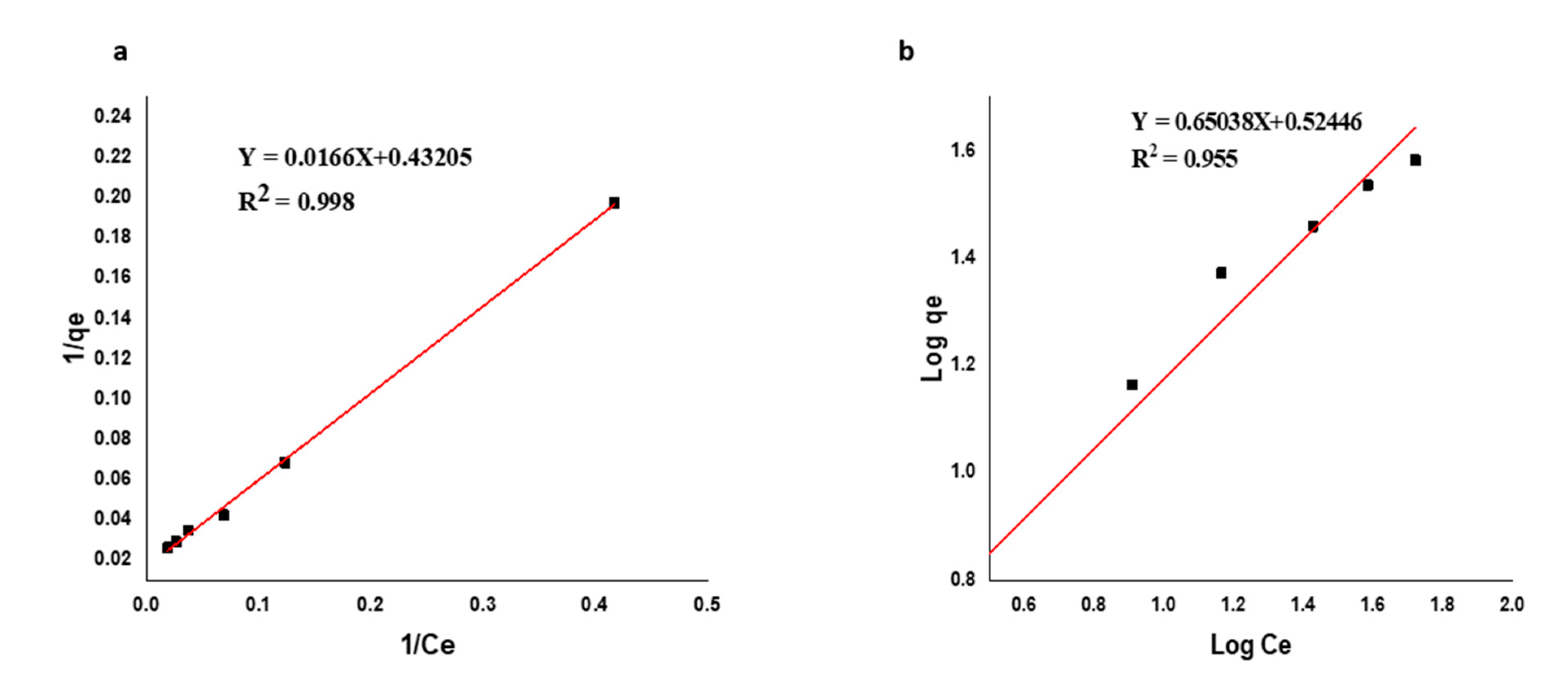
3.5.2. Adsorption Isotherms
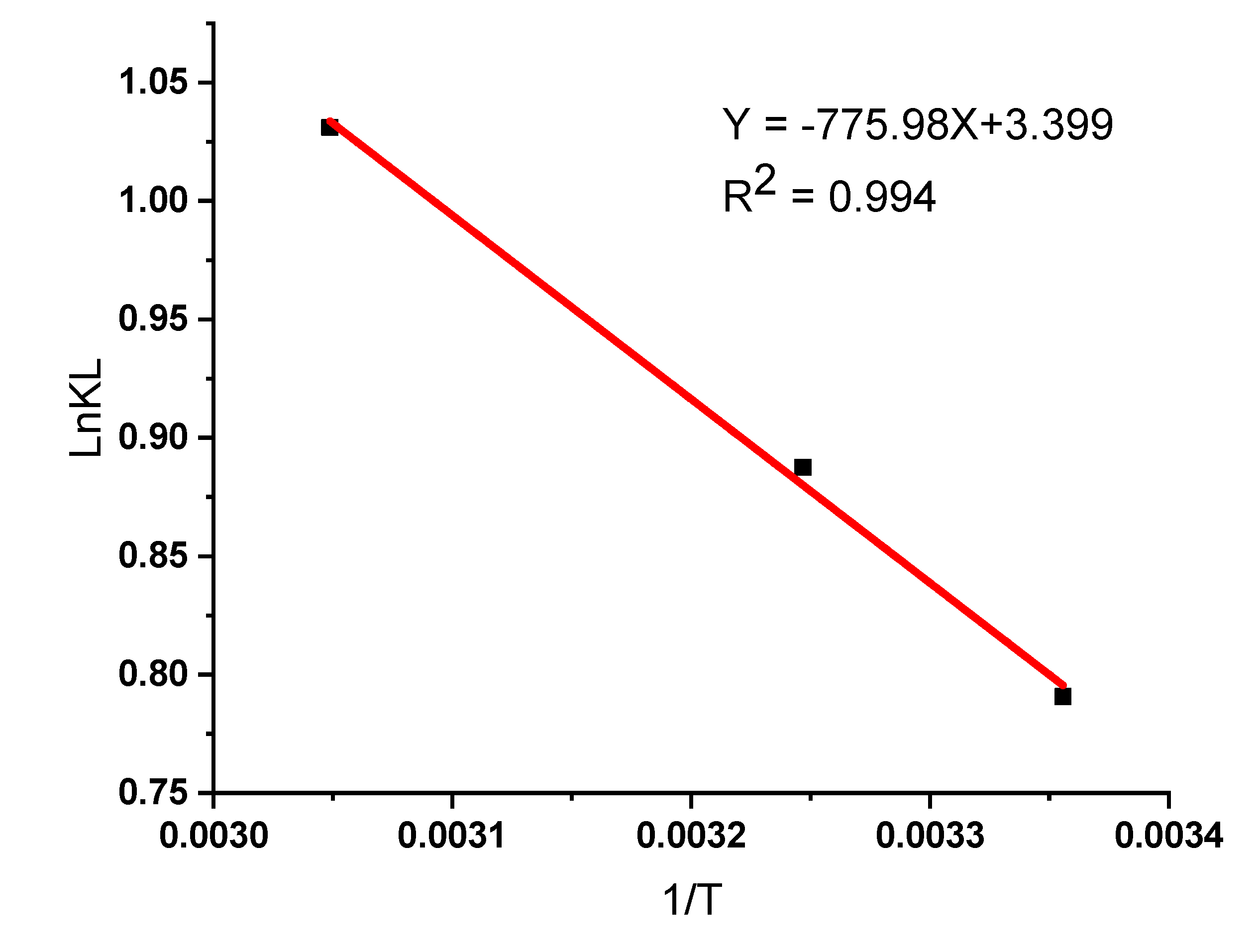
3.5.3. Adsorption Thermodynamic Studies
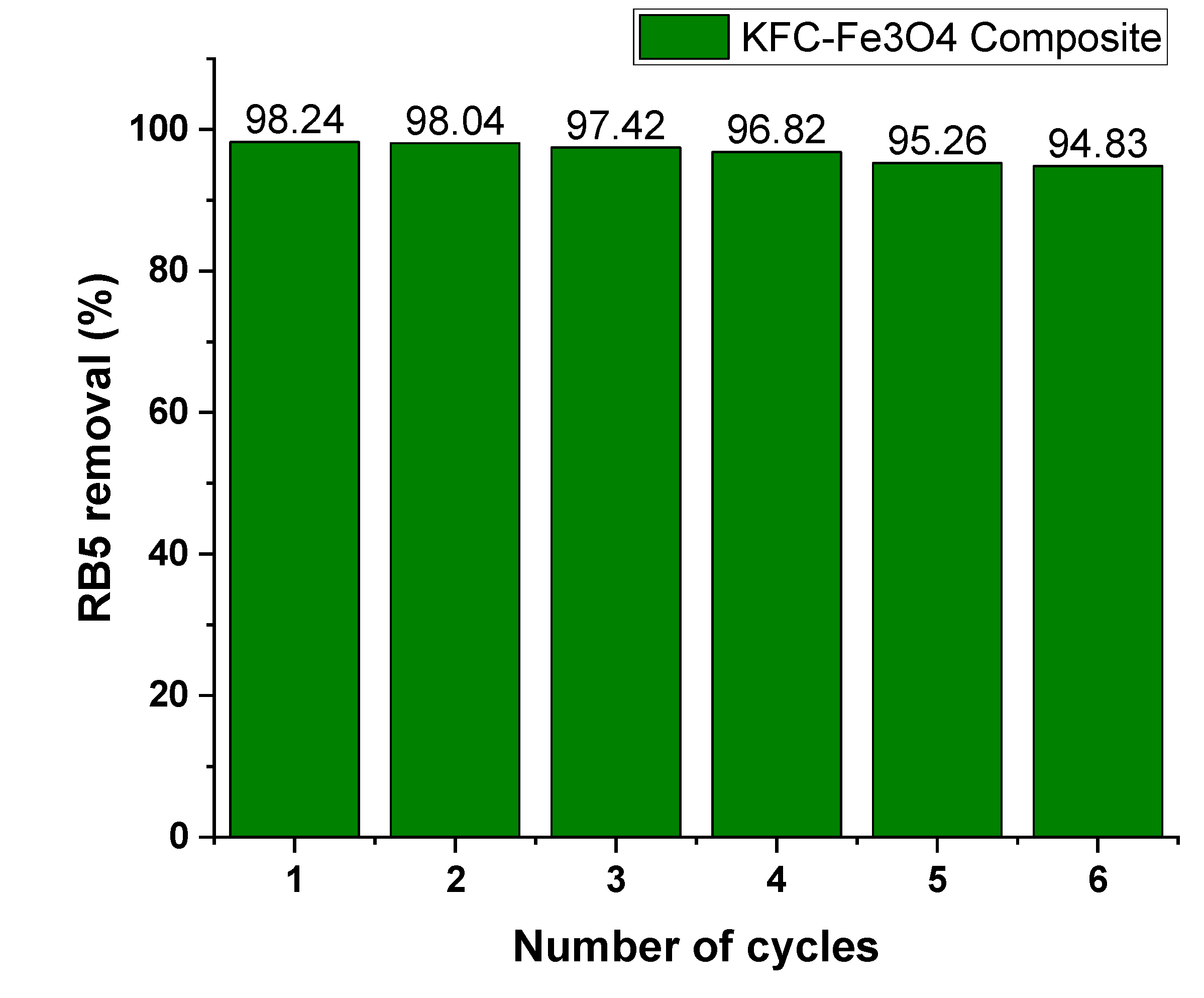
3.6. Reusability of the KFC-Fe3O4 Composite
4. Conclusions
Author Contributions
Data Availability Statement
Acknowledgements
Conflicts of Interest:
References
- Alnasrawy, S.T. Adsorption Efficiency, Isotherms, and Kinetics for Cationic Dye Removal Using Biowaste Adsorbent. J. Hazard. Toxic Radioact. Waste 2023, 27, 04022040. [Google Scholar] [CrossRef]
- Vojnović, B.; Cetina, M.; Franjković, P.; Sutlović, A. Influence of Initial pH Value on the Adsorption of Reactive Black 5 Dye on Powdered Activated Carbon: Kinetics, Mechanisms, and Thermodynamics. Molecules 2022, 27, 1349. [Google Scholar] [CrossRef]
- Balarak, D.; Al-Musawi, T.J.; Mohammed, I.A.; Abasizadeh, H. The Eradication of Reactive Black 5 Dye Liquid Wastes Using Azolla Filiculoides Aquatic Fern as a Good and an Economical Biosorption Agent. SN Appl. Sci. 2020, 2, 1015. [Google Scholar] [CrossRef]
- Kalkan, E.; Nadaroğlu, H.; Celebi, N.; Tozsin, G. Removal of Textile Dye Reactive Black 5 from Aqueous Solution by Adsorption on Laccase-Modified Silica Fume. Desalination Water Treat. 2014, 52, 6122–6134. [Google Scholar] [CrossRef]
- Nure, J.F.; Mengistu, A.; Abewaa, M.; Angassa, K.; Moyo, W.; Phiri, Z.; Mafa, P.J.; Kuvarega, A.T.; Nkambule, T.T. Adsorption of Black MNN Reactive Dye from Tannery Wastewater Using Activated Carbon of Rumex Abysinicus. J. Taiwan Inst. Chem. Eng. 2023, 151, 105138. [Google Scholar]
- Farrokhzadeh, S.; Razmi, H.; Jannat, B. Study of Reactive Red 195 Anionic Dye Adsorption on Calcined Marble Powder as Potential Eco-Friendly Adsorbent. Hum. Health Halal Metr. 2020, 1, 42–56. [Google Scholar] [CrossRef]
- Mokue Mafo, S.G.; Nanssou Kouteu, P.A.; Tchuifon Tchuifon, D.R.; Fotsop, C.G.; Zue Mve, M.; Teikam Kenda, G.; Bopda, A.; Tiotsop Kuete, H.-I.; Sadeu Ngakou, C.; Ndifor-Angwafor, N.G.; et al. Low-Cost Magnetic Carbons-Based Rubber Seed Husks Materials for Highly Efficient Removal for Reactive Black 5 and Reactive Blue 19 Textile Dyes from Wastewater. Int. J. Environ. Anal. Chem. 2023, 0, 1–25. [Google Scholar] [CrossRef]
- Jalali Sarvestani, M.R.; Doroudi, Z. Removal of Reactive Black 5 from Waste Waters by Adsorption: A Comprehensive Review. J. Water Environ. Nanotechnol. 2020, 5, 180–190. [Google Scholar] [CrossRef]
- Suryawan, I.W.; Helmy, Q.; Notodarmojo, S. Textile Wastewater Treatment: Colour and COD Removal of Reactive Black-5 by Ozonation. IOP Conf. Ser. Earth Environ. Sci. 2018, 106, 012102. [Google Scholar] [CrossRef]
- Alula, M.T.; Spende, H.; Aragaw, T.A.; Alene, A.N.; Aragaw, B.A.; Madiba, M. A Highly Stable Silver Nanoparticle Loaded Magnetic Nanocomposite as a Recyclable Catalysts. J. Clust. Sci. 2023, 34, 2205–2214. [Google Scholar] [CrossRef]
- Srivastava, A.; Rani, R.; Kumar, S. Optimization, Kinetics, and Thermodynamics Aspects in the Biodegradation of Reactive Black 5 (RB5) Dye from Textile Wastewater Using Isolated Bacterial Strain, Bacillus Albus DD1. Water Sci. Technol. 2022, 86, 610–624. [Google Scholar] [CrossRef] [PubMed]
- Antunes, M.L.P.; Conceição, F.T.; Navarro, G.R.B.; Fernandes, A.M.; Durrant, S.F. Use of Red Mud Activated at Different Temperatures as a Low Cost Adsorbent of Reactive Dye. Eng. Sanit. E Ambient. 2021, 26, 805–811. [Google Scholar] [CrossRef]
- Alemayehu, E.; Melak, F.; Sharma, S.K.; Van Der Bruggen, B.; Lennartz, B. Use of Porous Volcanic Rocks for the Adsorptive Removal of Copper. Water Environ. J. 2017, 31, 194–201. [Google Scholar] [CrossRef]
- Sarma, G.K.; Gupta, S.S.; Bhattacharyya, K.G. Removal of Hazardous Basic Dyes from Aqueous Solution by Adsorption onto Kaolinite and Acid-Treated Kaolinite: Kinetics, Isotherm and Mechanistic Study. SN Appl. Sci. 2019, 1. [Google Scholar] [CrossRef]
- Mustapha, S.; Tijani, J.; Ndamitso, M.; Abdulkareem, A.; Shuaib, D.; Mohammed, A.K. Adsorptive Removal of Pollutants from Industrial Wastewater Using Mesoporous Kaolin and Kaolin/TiO2 Nanoadsorbents. Environ. Nanotechnol. Monit. Manag. 2021, 15, 100414. [Google Scholar] [CrossRef]
- Mandale, P.; Kulkarni, K.; Jadhav, K.; Kulkarni, A.; Mahajan, R. Adsorption of Malachite Green Dye from Aqueous Solution on Bioadsorbent as Low-Cost Adsorbent. Mater. Today Proc. 2024. [Google Scholar] [CrossRef]
- Muisa, N.; Nhapi, I.; Ruziwa, W.; Manyuchi, M.M. Utilization of Alum Sludge as Adsorbent for Phosphorus Removal in Municipal Wastewater: A Review. J. Water Process Eng. 2020, 35, 101187. [Google Scholar] [CrossRef]
- Temesgen, F.; Kalifa, M.; Jembere, A.; Genet, M. Spent Kaolin Filter Cake as an Effective Adsorbent for the Removal of Hexavalent Chromium [Cr (VI)] From Aqueous Solution: Comparative Study of Wastewater Treatment Methods. South Afr. J. Chem. Eng. 2021, 38. [Google Scholar] [CrossRef]
- Abate, G.Y.; Alene, A.N.; Habte, A.T.; Getahun, D.M. Adsorptive Removal of Malachite Green Dye from Aqueous Solution onto Activated Carbon of Catha Edulis Stem as a Low Cost Bio-Adsorbent. Environ. Syst. Res. 2020, 9, 29. [Google Scholar] [CrossRef]
- Azha, S.F.; Shahadat, M.; Ismail, S.; Ali, S.W.; Ahammad, S.Z. Prospect of Clay-Based Flexible Adsorbent Coatings as Cleaner Production Technique in Wastewater Treatment, Challenges, and Issues: A Review. J. Taiwan Inst. Chem. Eng. 2021, 120, 178–206. [Google Scholar] [CrossRef]
- Ike, J.I.; Babayemi, A.K.; Egbosiuba, T.C.; Jin, C.G.; Mustapha, S.; Yusuff, A.S.; Yadav, V.K.; Igwegbe, C.A. Treated Kaolin Clay Incorporated with Nickel Nanoparticles for Enhanced Removal of Crystal Violet and Methyl Orange from Textile Wastewater. ACS Appl. Eng. Mater. 2024, 2, 1031–1046. [Google Scholar] [CrossRef]
- Amin, M.T.; Alazba, A.A.; Shafiq, M. Adsorptive Removal of Reactive Black 5 from Wastewater Using Bentonite Clay: Isotherms, Kinetics and Thermodynamics. Sustainability 2015, 7, 15302–15318. [Google Scholar] [CrossRef]
- Co-Precipitation Synthesis of Clay-Magnetite Nanocomposite for Adsorptive Removal of Synthetic Dye in Wastewater of Benang Bintik Batik. Mol. J. Ilm. Kim. 2022, 17, 261–261. [CrossRef]
- Gahlot, R.; Taki, K.; Kumar, M. Efficacy of Nanoclays as the Potential Adsorbent for Dyes and Metal Removal from the Wastewater: A Review. Environ. Nanotechnol. Monit. Manag. 2020, 14, 100339. [Google Scholar] [CrossRef]
- Kassa, A.E.; Shibeshi, N.T.; Tizazu, B.Z.; Prabhu, S.V. Characteristic Investigations on Ethiopian Kaolinite: Effect of Calcination Temperature on Pozzolanic Activity and Specific Surface Area. Adv. Mater. Sci. Eng. 2022, 2022, e2481066. [Google Scholar] [CrossRef]
- Jawad, A.H.; Abdulhameed, A.S. Facile Synthesis of Crosslinked Chitosan-Tripolyphosphate/Kaolin Clay Composite for Decolourization and COD Reduction of Remazol Brilliant Blue R Dye: Optimization by Using Response Surface Methodology. Colloids Surf. Physicochem. Eng. Asp. 2020, 605, 125329. [Google Scholar] [CrossRef]
- Dissanayake, R.E.A.; Premarathne, I.M.; Iqbal, S.S.; Priyantha, N.; Iqbal, M.C.M. Synthesis, Characterization, and Textile Dye Adsorption Studies of a Kaolin-Based Polymer Layer Silicate Composite. Int. J. Environ. Sci. Technol. 2022, 19, 9519–9534. [Google Scholar] [CrossRef]
- Jawad, A.H.; Malek, N.N.A.; Abdulhameed, A.S.; Razuan, R. Synthesis of Magnetic Chitosan-Fly Ash/Fe3O4 Composite for Adsorption of Reactive Orange 16 Dye: Optimization by Box–Behnken Design. J. Polym. Environ. 2020, 28, 1068–1082. [Google Scholar] [CrossRef]
- Magdy, A.; Fouad, Y.O.; Abdel-Aziz, M.H.; Konsowa, A.H. Synthesis and Characterization of Fe 3 O 4 /Kaolin Magnetic Nanocomposite and Its Application in Wastewater Treatment. J. Ind. Eng. Chem. 2017, 56, 299–311. [Google Scholar] [CrossRef]
- Fito, J.; Abewaa, M.; Nkambule, T. Magnetite-Impregnated Biochar of Parthenium Hysterophorus for Adsorption of Cr(VI) from Tannery Industrial Wastewater. Appl. Water Sci. 2023, 13, 78. [Google Scholar] [CrossRef]
- Jawad, A.H.; Sahu, U.K.; Jani, N.A.; ALOthman, Z.A.; Wilson, L.D. Magnetic Crosslinked Chitosan-Tripolyphosphate/MgO/Fe3O4 Nanocomposite for Reactive Blue 19 Dye Removal: Optimization Using Desirability Function Approach. Surf. Interfaces 2022, 28, 101698. [Google Scholar] [CrossRef]
- Mirzapour, P.; Kamyab Moghadas, B.; Tamjidi, S.; Esmaeili, H. Activated Carbon/Bentonite/Fe3O4 Nanocomposite for Treatment of Wastewater Containing Reactive Red 198. Sep. Sci. Technol. 2021, 56, 2693–2707. [Google Scholar] [CrossRef]
- Sacko, A.; Nure, J.F.; Nyoni, H.; Mamba, B.; Nkambule, T.; Msagati, T.A.M. The Application of Tannic Acid-Coated Magnetite Nanoparticles for Recovery of Microplastics from the Water System. Water Conserv. Sci. Eng. 2024, 9, 41. [Google Scholar] [CrossRef]
- Masuku, M.; Nure, J.F.; Atagana, H.I.; Hlongwa, N.; Nkambule, T.T. Advancing the Development of Nanocomposite Adsorbent through Zinc-Doped Nickel Ferrite-Pinecone Biochar for Removal of Chromium (VI) from Wastewater. Sci. Total Environ. 2024, 908, 168136. [Google Scholar] [PubMed]
- Hasan, M.; Al Biruni, M.T.; Azad, S.; Ahmed, T. Adsorptive Removal of Dye from Textile Wastewater Employing Moringa Oleifera Leaves Biochar as a Natural Biosorbent. Biomass Convers. Biorefinery 2022. [Google Scholar] [CrossRef]
- Mutambyi, V.; Jean Nepo, H.; Rugabirwa, B.; Kwisanga, C.; Beakou, B.H.; Munyegaju, J.; Uwitonze, N.; Nyandwi, V.; Mushirabwoba, B. Removal of Reactive Black 5 from Simulated Textile Effluents by an Electrocoagulation Process: Optimization by Response Surface Methodology. Water Pract. Technol. 2023, 18, 3048–3064. [Google Scholar] [CrossRef]
- Seleman, M.; Sime, T.; Ayele, A.; Sergawie, A.; Nkambule, T.; Fito, J. Isotherms and Kinetic Studies of Copper Removal from Textile Wastewater and Aqueous Solution Using Powdered Banana Peel Waste as an Adsorbent in Batch Adsorption Systems. Int. J. Biomater. 2023, 2023, 1–10. [Google Scholar] [CrossRef]
- Hamri, N.; Imessaoudene, A.; Hadadi, A.; Cheikh, S.; Boukerroui, A.; Bollinger, J.-C.; Amrane, A.; Tahraoui, H.; Tran, H.N.; Ezzat, A.O.; et al. Enhanced Adsorption Capacity of Methylene Blue Dye onto Kaolin through Acid Treatment: Batch Adsorption and Machine Learning Studies. Water 2024, 16, 243. [Google Scholar] [CrossRef]
- Ribeiro, A.C.; Barbosa de Andrade, M.; Quesada, H.B.; Bergamasco Beltran, L.; Bergamasco, R.; Calado Santos Sobral da Fonseca, M.M.; da Costa Neves Fernandes de Almeida Duarte, E. Physico-Chemical and Electrostatic Surface Characterisation of Mica Mineral and Its Applicability on the Adsorption of Safranin Orange and Reactive Black 5 Dyes. Environ. Technol. 2022, 43, 3765–3778. [Google Scholar] [CrossRef]
- Souhassou, H.; Fahoul, Y.; El Mrabet, I.; Iboustaten, E.; Assila, O.; Nahali, L.; Mertah, O.; Kherbeche, A. Optimization of Basic Red 29 Dye Removal onto a Natural Red Clay Using Response Surface Methodology. J. Iran. Chem. Soc. 2024, 21, 275–291. [Google Scholar] [CrossRef]
- Caponi, N.; Collazzo, G.C.; Jahn, S.L.; Dotto, G.L.; Mazutti, M.A.; Foletto, E.L. Use of Brazilian Kaolin as a Potential Low-Cost Adsorbent for the Removal of Malachite Green from Colored Effluents. Mater. Res. 2017, 20, 14–22. [Google Scholar] [CrossRef]
- Khunjan, U.; Kasikamphaiboon, P. Green Synthesis of Kaolin-Supported Nanoscale Zero-Valent Iron Using Ruellia Tuberosa Leaf Extract for Effective Decolorization of Azo Dye Reactive Black 5. Arab. J. Sci. Eng. 2021, 46, 383–394. [Google Scholar] [CrossRef]
- De Luca, P.; B. Nagy, J. Treatment of Water Contaminated with Reactive Black-5 Dye by Carbon Nanotubes. Materials 2020, 13, 5508. [Google Scholar] [CrossRef]
- Bello, A.M.; Muhammad, N.A.; Hamisu, A. RSM Optimized Adsorption of Eriochrome Black T Dye onto Alumina Nanoprticles: Isotherm and Kinetics Studies. Phys. Chem. Res. 2023, 11, 315–326. [Google Scholar] [CrossRef]
- Sawood, G.M.; Mishra, A.; Gupta, S.K. Optimization of Arsenate Adsorption over Aluminum-Impregnated Tea Waste Biochar Using RSM–Central Composite Design and Adsorption Mechanism. J. Hazard. Toxic Radioact. Waste 2021, 25, 04020075. [Google Scholar] [CrossRef]
- Belachew, N.; Fekadu, R.; Ayalew Abebe, A. RSM-BBD Optimization of Fenton-Like Degradation of 4-Nitrophenol Using Magnetite Impregnated Kaolin. Air Soil Water Res. 2020, 13, 1178622120932124. [Google Scholar] [CrossRef]
- Njuhou, S.; Mouafon, M.; Pountouenchi, A.; Njindam, O.R.; Lecomte-Nana, G.L.; Njoya, D. Rice Husks and Kaolin Based Ceramic Membrane for Filtration of Slaughterhouse Wastewater: Optimization Study Using Response Surface Methodology (RSM) and Responses Interdependence Analysis. Trans. Indian Ceram. Soc. 2023, 82, 143–155. [Google Scholar] [CrossRef]
- López-Rodríguez, D.; Micó-Vicent, B.; Jordán-Núñez, J.; Bonet-Aracil, M.; Bou-Belda, E. Uses of Nanoclays and Adsorbents for Dye Recovery: A Textile Industry Review. Appl. Sci. 2021, 11, 11422. [Google Scholar] [CrossRef]
- Hidayat, E.; Mohamad Sarbani, N.M.B.; Yonemura, S.; Mitoma, Y.; Harada, H. Application of Box–Behnken Design to Optimize Phosphate Adsorption Conditions from Water onto Novel Adsorbent CS-ZL/ZrO/Fe3O4: Characterization, Equilibrium, Isotherm, Kinetic, and Desorption Studies. Int. J. Mol. Sci. 2023, 24, 9754. [Google Scholar] [CrossRef]
- Aragaw, T.A.; Angerasa, F.T. Synthesis and Characterization of Ethiopian Kaolin for the Removal of Basic Yellow (BY 28) Dye from Aqueous Solution as a Potential Adsorbent. Heliyon 2020, 6, e04975. [Google Scholar] [CrossRef]
- Mouni, L.; Belkhiri, L.; Bollinger, J.-C.; Bouzaza, A.; Assadi, A.; Tirri, A.; Dahmoune, F.; Madani, K.; Remini, H. Removal of Methylene Blue from Aqueous Solutions by Adsorption on Kaolin: Kinetic and Equilibrium Studies. Appl. Clay Sci. 2018, 153, 38–45. [Google Scholar] [CrossRef]
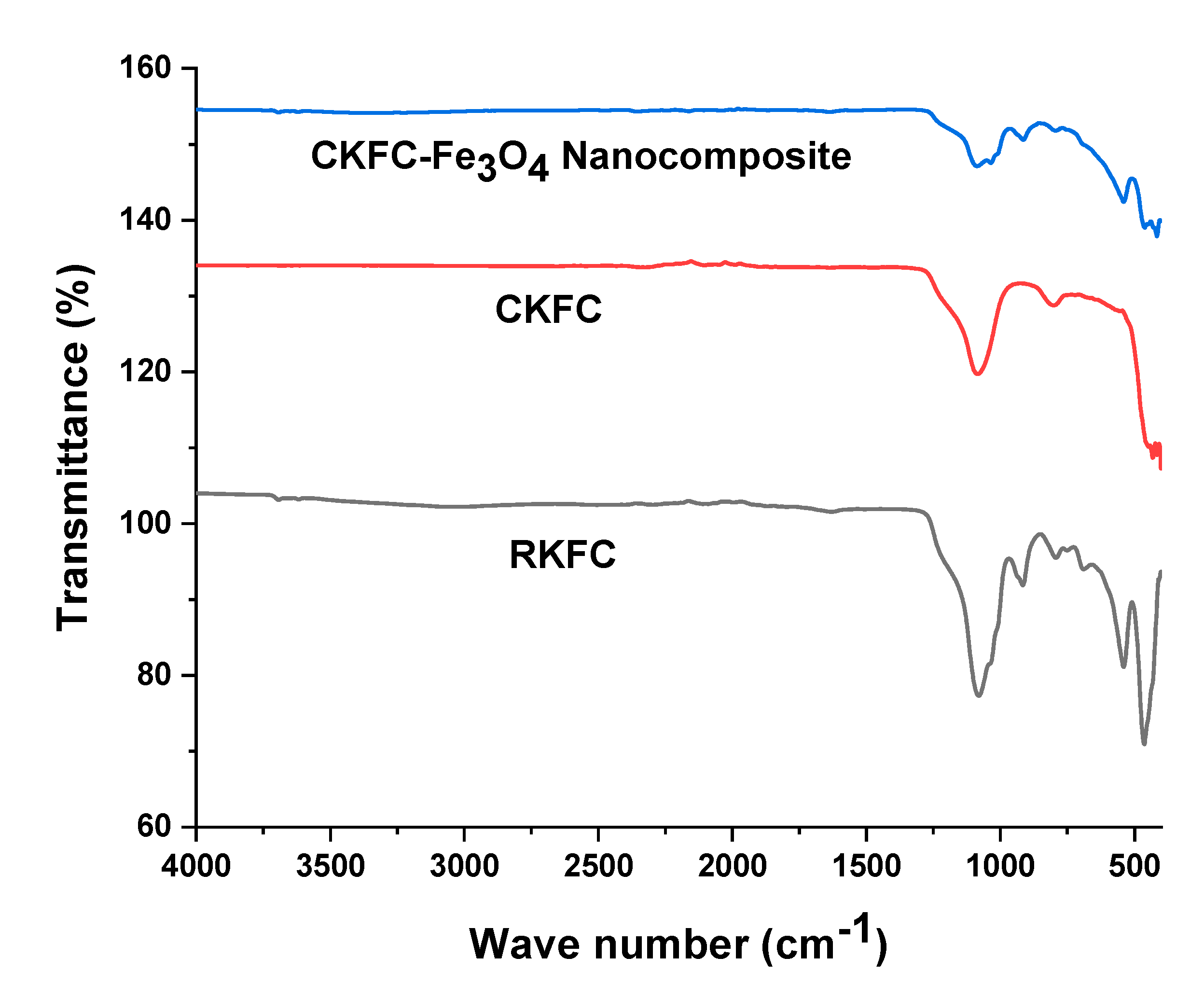
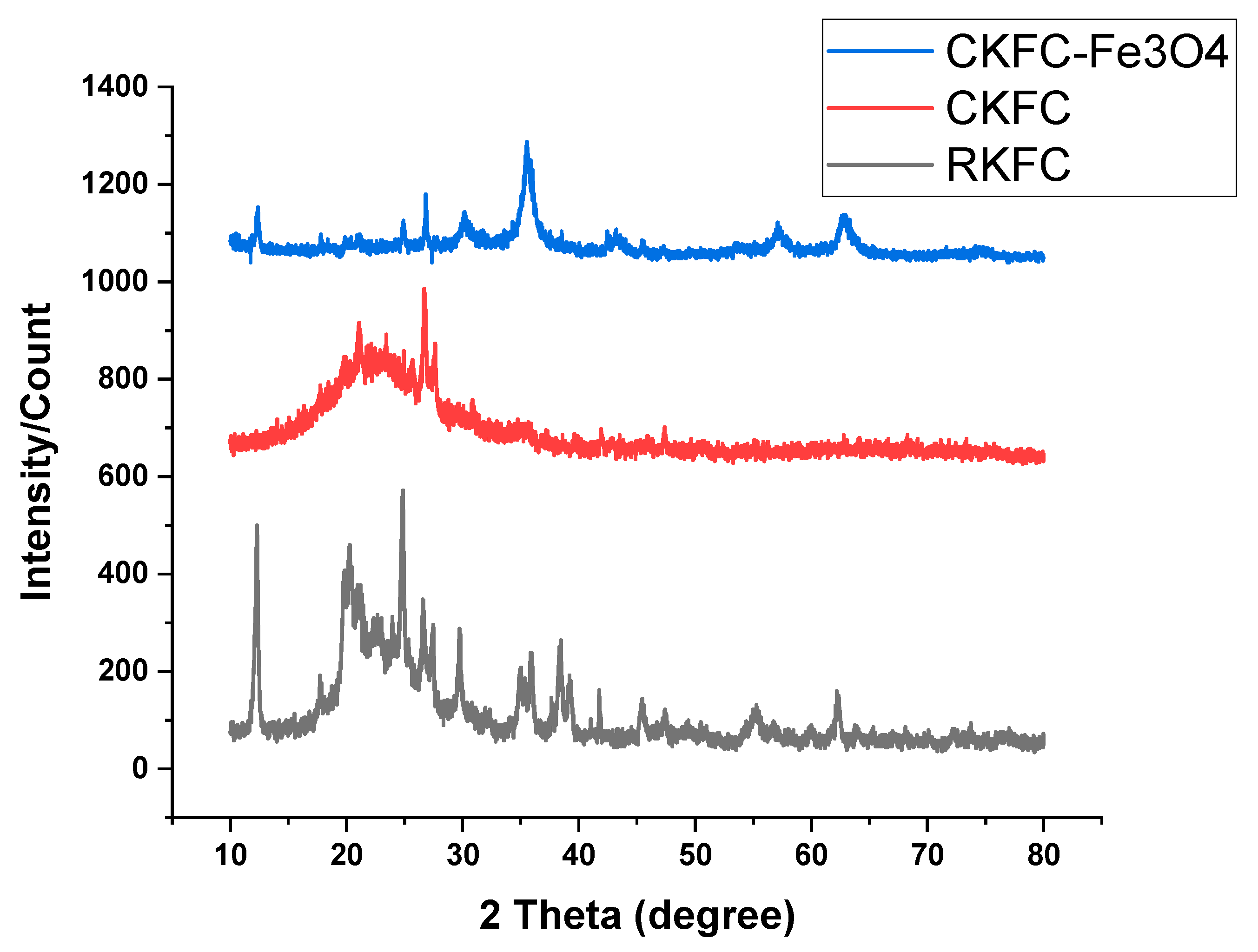

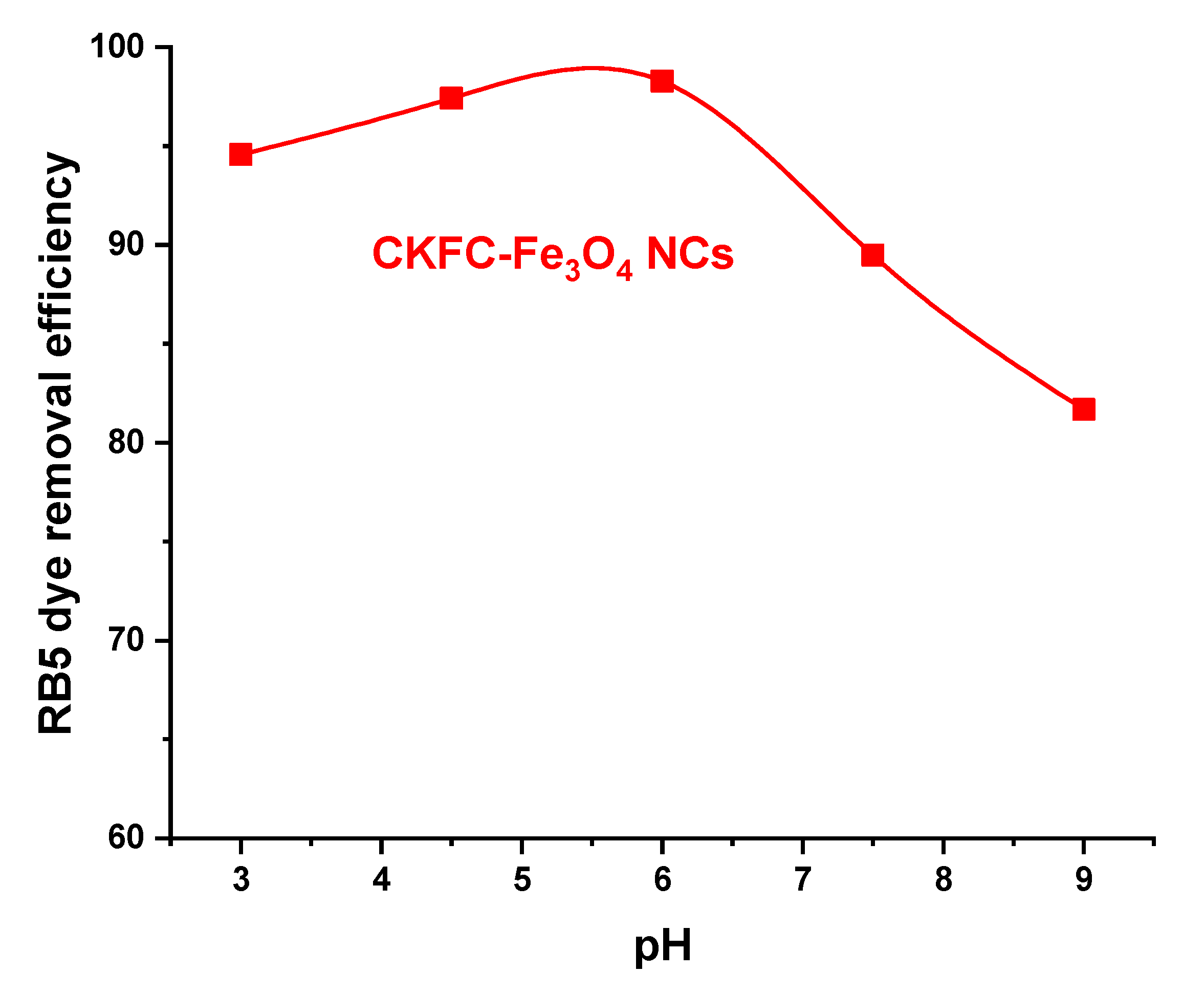
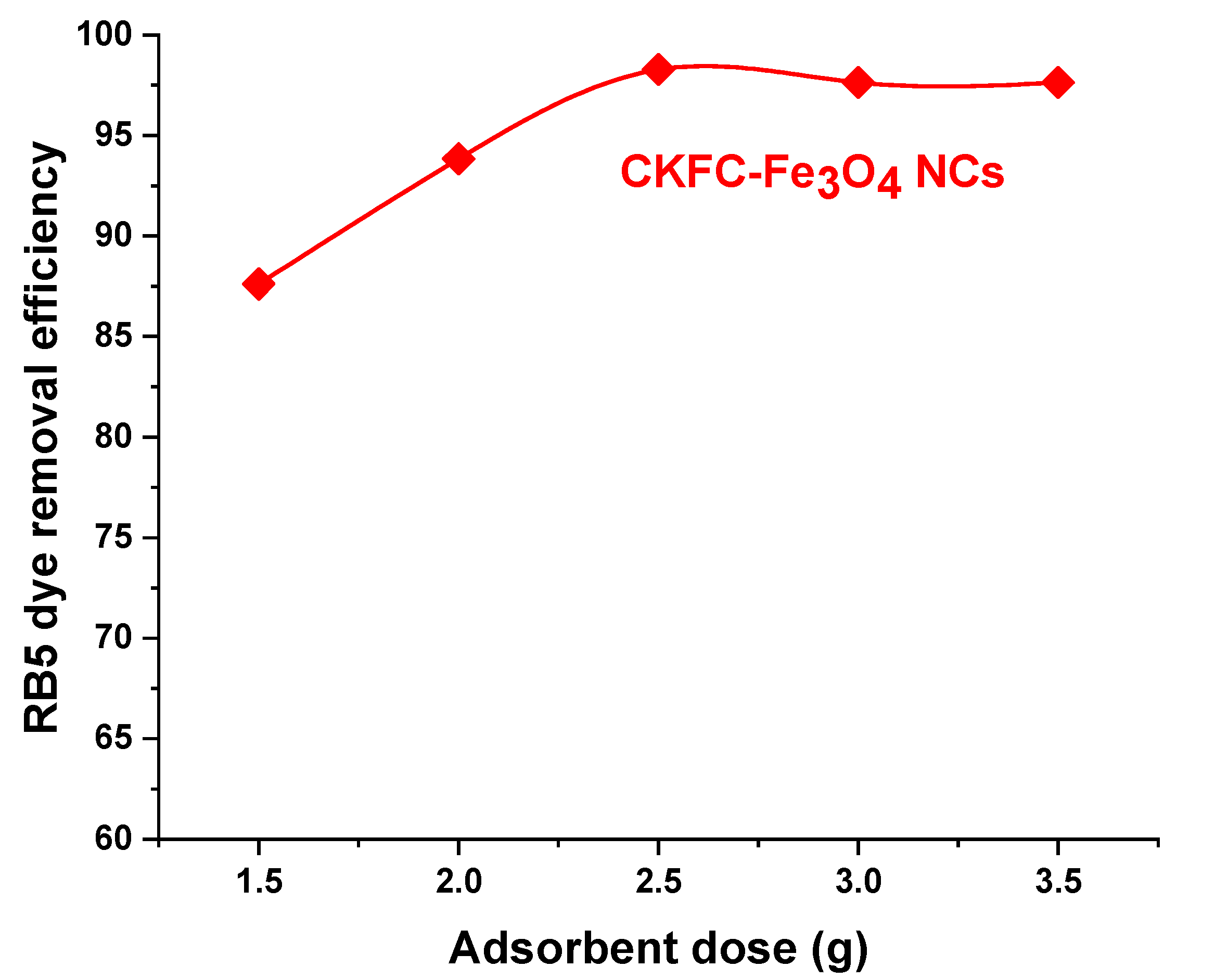
| Codes | Variables | Low (- 1) | Center (0) | High (+1) |
| A | pH | 3 | 6 | 9 |
| B | Adsorbent dose (g) | 1.5 | 2.5 | 3.5 |
| C | Contact time (min) | 40 | 60 | 80 |
| D | Initial dye concentration (mg/L) | 30 | 70 | 100 |
| Run | A: pH | B: Adsorbent dose (g) | C: Time (min) | D: Initial RB5 concentration (mg/l) | RB5 Removal (%) | Predicted RB5 Removal (%) |
| 1 | 6 | 3.5 | 30 | 70 | 92.97 | 92.81 |
| 2 | 3 | 3.5 | 60 | 70 | 92.13 | 92.01 |
| 3 | 6 | 1.5 | 60 | 40 | 86.12 | 85.95 |
| 4 | 9 | 2.5 | 30 | 70 | 64.26 | 64.31 |
| 5 | 6 | 2.5 | 60 | 70 | 98.56 | 98.28 |
| 6 | 6 | 3.5 | 90 | 70 | 93.56 | 93.35 |
| 7 | 6 | 2.5 | 90 | 100 | 94.58 | 94.74 |
| 8 | 6 | 1.5 | 90 | 70 | 90.82 | 90.90 |
| 9 | 3 | 1.5 | 60 | 70 | 87.58 | 87.50 |
| 10 | 9 | 2.5 | 60 | 100 | 65.89 | 65.50 |
| 11 | 6 | 1.5 | 30 | 70 | 90.45 | 90.58 |
| 12 | 6 | 2.5 | 60 | 70 | 98.13 | 98.28 |
| 13 | 3 | 2.5 | 60 | 40 | 92.64 | 92.94 |
| 14 | 9 | 1.5 | 60 | 70 | 62.31 | 62.57 |
| 15 | 3 | 2.5 | 30 | 70 | 94.12 | 93.96 |
| 16 | 6 | 2.5 | 30 | 40 | 94.68 | 94.66 |
| 17 | 6 | 1.5 | 60 | 100 | 92.85 | 92.64 |
| 18 | 9 | 2.5 | 90 | 70 | 67.19 | 67.29 |
| 19 | 6 | 3.5 | 60 | 40 | 95.17 | 95.33 |
| 20 | 3 | 2.5 | 60 | 100 | 89.82 | 89.98 |
| 21 | 9 | 2.5 | 60 | 40 | 63.47 | 63.23 |
| 22 | 6 | 2.5 | 60 | 70 | 98.24 | 98.28 |
| 23 | 3 | 2.5 | 90 | 70 | 91.94 | 91.84 |
| 24 | 6 | 2.5 | 30 | 100 | 91.97 | 92.14 |
| 25 | 6 | 2.5 | 90 | 40 | 92.94 | 92.91 |
| 26 | 6 | 2.5 | 60 | 70 | 97.85 | 98.28 |
| 27 | 9 | 3.5 | 60 | 70 | 62.52 | 62.74 |
| 28 | 6 | 3.5 | 60 | 100 | 87.84 | 87.95 |
| 29 | 6 | 2.5 | 60 | 70 | 98.64 | 98.28 |
| Source | Sum of Squares | df | Mean Square | F-value | p-value | |
| Model | 4277.79 | 14 | 305.56 | 3593.69 | < 0.0001 | significant |
| A-pH | 2202.96 | 1 | 2202.96 | 25909.29 | < 0.0001 | |
| B-Adsorbent dose | 16.47 | 1 | 16.47 | 193.75 | < 0.0001 | |
| C-Time | 0.5547 | 1 | 0.5547 | 6.52 | 0.0229 | |
| D-Initial RB5 concentration | 0.3571 | 1 | 0.3571 | 4.20 | 0.0597 | |
| AB | 4.71 | 1 | 4.71 | 55.38 | < 0.0001 | |
| AC | 6.53 | 1 | 6.53 | 76.78 | < 0.0001 | |
| AD | 6.86 | 1 | 6.86 | 80.73 | < 0.0001 | |
| BC | 0.0121 | 1 | 0.0121 | 0.1423 | 0.7117 | |
| BD | 49.42 | 1 | 49.42 | 581.25 | < 0.0001 | |
| CD | 4.73 | 1 | 4.73 | 55.64 | < 0.0001 | |
| A² | 1945.25 | 1 | 1945.25 | 22878.36 | < 0.0001 | |
| B² | 147.04 | 1 | 147.04 | 1729.36 | < 0.0001 | |
| C² | 16.94 | 1 | 16.94 | 199.26 | < 0.0001 | |
| D² | 60.54 | 1 | 60.54 | 711.96 | < 0.0001 | |
| Residual | 1.19 | 14 | 0.0850 | |||
| Lack of Fit | 0.7734 | 10 | 0.0773 | 0.7421 | 0.6814 | not significant |
| Pure Error | 0.4169 | 4 | 0.1042 | |||
| Cor Total | 4278.98 | 28 |
| Source | Sum of Squares | df | Mean Square | F-value | p-value | |
| Mean vs Total | 2.206E+05 | 1 | 2.206E+05 | |||
| Linear vs Mean | 2220.34 | 4 | 555.09 | 6.47 | 0.0011 | |
| 2FI vs Linear | 72.26 | 6 | 12.04 | 0.1091 | 0.9942 | |
| Quadratic vs 2FI | 1985.18 | 4 | 496.29 | 5836.99 | < 0.0001 | Suggested |
| Cubic vs Quadratic | 0.7129 | 8 | 0.0891 | 1.12 | 0.4580 | Aliased |
| Residual | 0.4775 | 6 | 0.0796 | |||
| Total | 2.249E+05 | 29 | 7754.04 | |||
| Model summary statistics | ||||||
| Source | Std. Dev. | R² | Adjusted R² | Predicted R² | PRESS | |
| Linear | 9.26 | 0.5189 | 0.4387 | 0.3169 | 2922.87 | |
| 2FI | 10.50 | 0.5358 | 0.2779 | -0.2191 | 5216.64 | |
| Quadratic | 0.2916 | 0.9997 | 0.9994 | 0.9988 | 5.11 | Suggested |
| Cubic | 0.2821 | 0.9999 | 0.9995 | 0.9978 | 9.37 | Aliased |
| Order of reaction | Parameters | CKFC-Fe3O4 Composite | RKFC |
| qe, exp (mg/gm) | 23.53 | 9.20 | |
| Pseudo-first-order | qe, cal (mg/g) | 0.51 | 0.85 |
| k1 (min-1) | -7.60×10-4 | -9.75×10-4 | |
| R2 | 0.5448 | 0.6205 | |
| Pseudo-second-order | qe, cal (mg/g) | 23.60 | 9.32 |
| k2 (g mg-1min-1) | 0.32 | 0.17 | |
| R2 | 0.9999 | 0.9996 |
| Type of isotherm | Parameters | RKFC | CKFC-Fe3O4 Composite |
| Langmuir | qmax (mg/g) | 60.04 | 92.84 |
| KL (L/mg) | 0.0147 | 0.0283 | |
| RL | 0.5748 | 0.4138 | |
| R2 | 0.9996 | 0.9998 | |
| Freundlich | Kf | 2.37 | 4.45 |
| 1/n | 0.5864 | 0.5300 | |
| R2 | 0.9167 | 0.9250 |
| Adsorbents | Dye | qmax (mg/g) | References |
| Residue from the aluminum industry | RB5 | 0.98 | [8] |
| NaOH-treated activated sludge | RB5 | 118.2 | [2] |
| Beneficiated Kaolin | BY 28 | 1.896 | [50] |
| Natural untreated clay | BY 28 | 76.92 | [20] |
| KFC-Fe3O4 Composite | RB5 | 92.84 | This Study |
| Adsorbent | Temp(K) | KL | ΔGo(KJmol-1) | ΔHo(KJmol-1) | ΔSo(KJmol-1) | R2 |
| KFC-Fe3O4Composite | 298 | 2.205 | -1.95908 | 6.4524 | 28.2632 | 0.9942 |
| 308 | 2.429 | -2.27258 | ||||
| 328 | 2.804 | -2.81166 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




